Introduction
Related research showed that cerebral aneurysm is
one of the most common malignant tumors at the present stage in
clinic, the specific mechanism of the occurrence and development of
cerebral aneurysm is still not clear (1). However, most scholars considered that
there was a close correlation between hemodynamic abnormality and
the occurrence and development of aneurysms or angiorrhexis
(2). With the development and
progress of science and technology, research including animal
experiments, clinical imaging observation, in vitro model
study all have played a very important role during the study of
cerebral aneurysm hemodynamic (3).
Research of cerebral aneurysm hemodynamic has gradually become the
research emphasis at the present stage along with the rapid
advances of computer technology and the improvement of computer
operational performance and calculation software (4). The study of hemodynamic has good
controllability and a high economic advantage in general, so it has
wide application (5). Thus, we
decided to assess the clinical value of numerical simulation in
diagnosing cerebral aneurysm based on the analysis of numerical
simulation of hemodynamic model.
Materials and methods
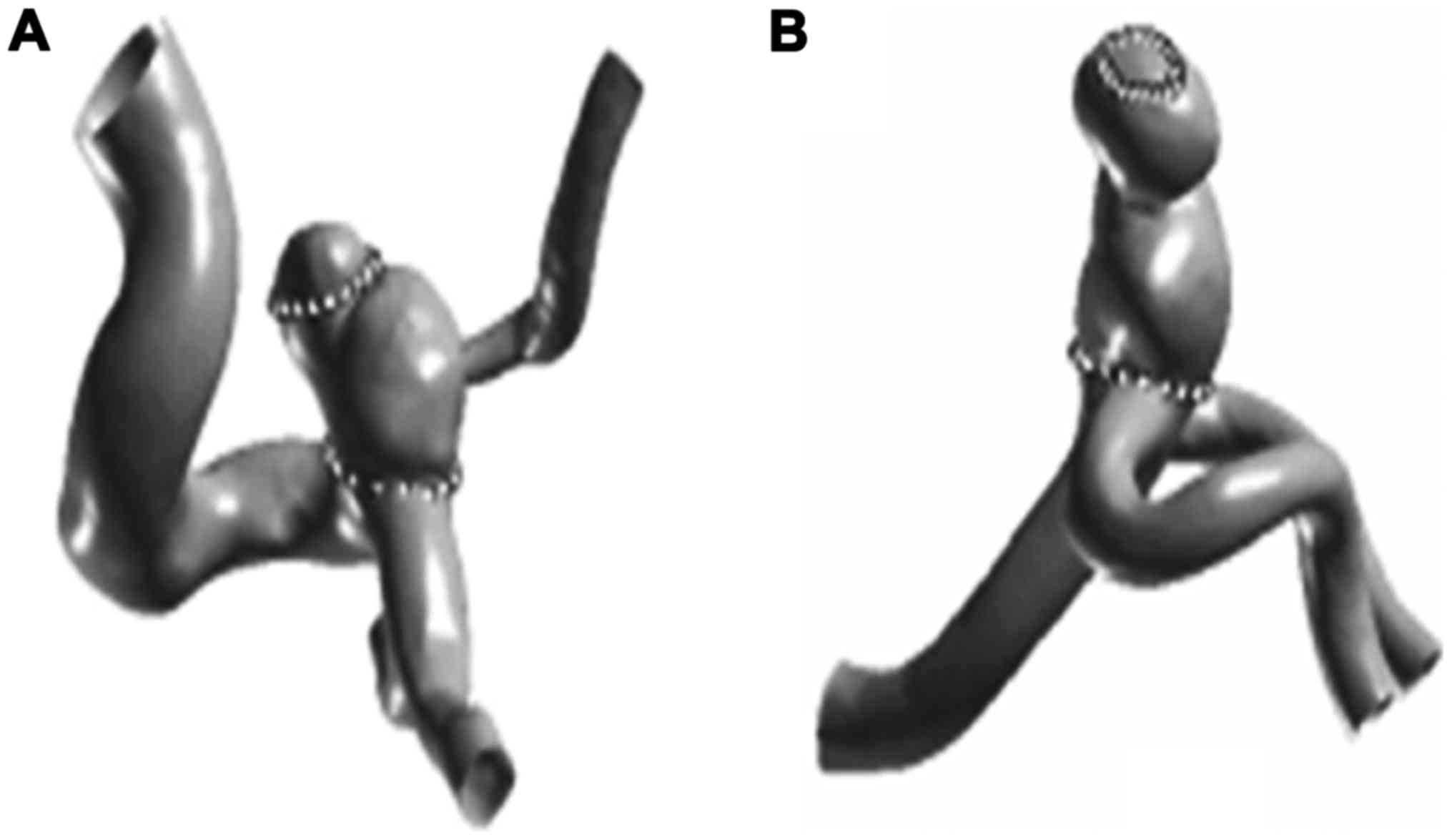
Model reconstruction
In this study, three-dimensional spiral angiography
machine (Phillips, New York, NY, USA) was used to collect the DSA
image, and the image on the three-dimensional angiography
workstation was reconstructed, we chose suitable windows,
intercepted the peripheral artery of cerebral aneurysm and branch,
then read image data with Geomagic Studio 9.0 (Geomagic,
Morrisville, NC, USA), for the data file.
Coordinates extraction
MATLAB6.5 software (MathWorks, Natick, MA, USA) was
used to create and import the bmp files, write the bmp path of
monochromatic aneurysm into data mat, and to obtain the
two-dimensional coordinates of every points. Coordinate point value
was copied and was saved in txt file as data formatted text
file.
Mesh generation
Gambit software (Ansys, Pittsburgh, PA, USA) was
used to create and import the point coordinates file, reproduce the
tumor contour of every point, connect every point with lines,
confirm the boundary contour, mark the region of interest, such as
tumor wall, and the tumor neck.
Boundary definition
The velocity inlet boundary and flow exit boundary
were set of as two entrance boundaries, solid wall boundary was set
as another boundary. Flow exit boundary and velocity inlet boundary
were set of Para tumor, other settings were like the boundary
setting of apical aneurysm boundary.
Mesh dividing
All the contour images were changed into a plane,
the plane was dived by two-dimensional mesh, and saved as mesh
format. Then the lateral aneurysm plan image was opened with Gambit
software, split aneurysm and blood vessels with virtual connection
line, and continued to divide two-dimensional mesh, the number of
units which were divided of CA1 and CA2 were 1507070 and
874595.
Blood flow behavior simulation
The Fluent software (Ansys) was run to import the
msh file of aneurysm, to set tumor boundary conditions and
assumptions, to calculate the blood flow. Tube diameter/tube
diameter length = scale factor, actual velocity of single tumor was
the blood flow speed. First, the viscous parameter of flowing blood
in the vessel (0.002 Pa/sec) and the density of flowing blood in
the vessel (1,059 kg/m3) were set, then a cardiac cycle
iteration was completed, pressure, velocity, shear stress time
curve were found, selection of the point of interest, was chosen
for every point according to the curvilinear trend, and then
analyzed.
Results
Results of the points
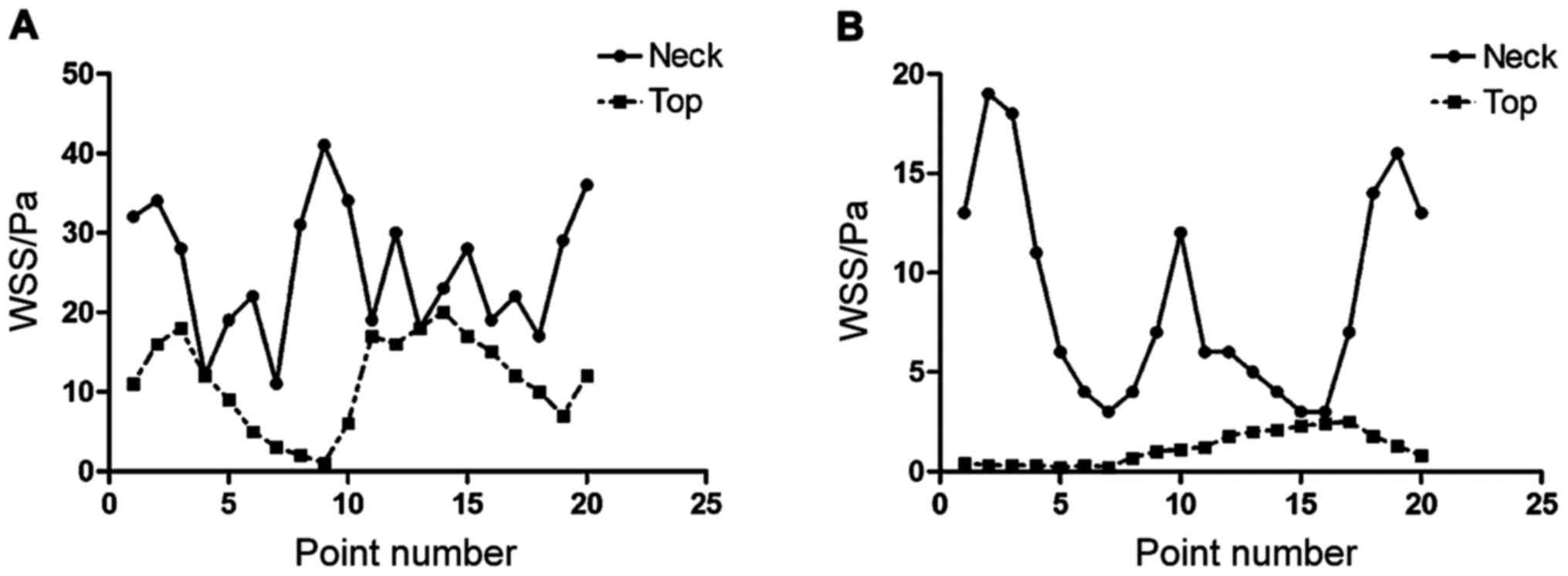
In this study, 20 points were taken on the top and
neck of cerebral aneurysm, the wall shear stress (WSS) and total
pressure of every point during systolic time were drawn
respectively, as shown in Fig.
1.
WSS results of each point
The results of this study showed that the WSS value
on the top of CA1 was significantly lower than the WSS value on the
top (P<0.05), the WSS value of every points on the tumor top of
CA2 were significantly lower than the WSS value of tumor neck
(P<0.05), and the WSS value of tumor neck has more heterogeneous
character than the WSS value of tumor top, as shown in Fig. 2.
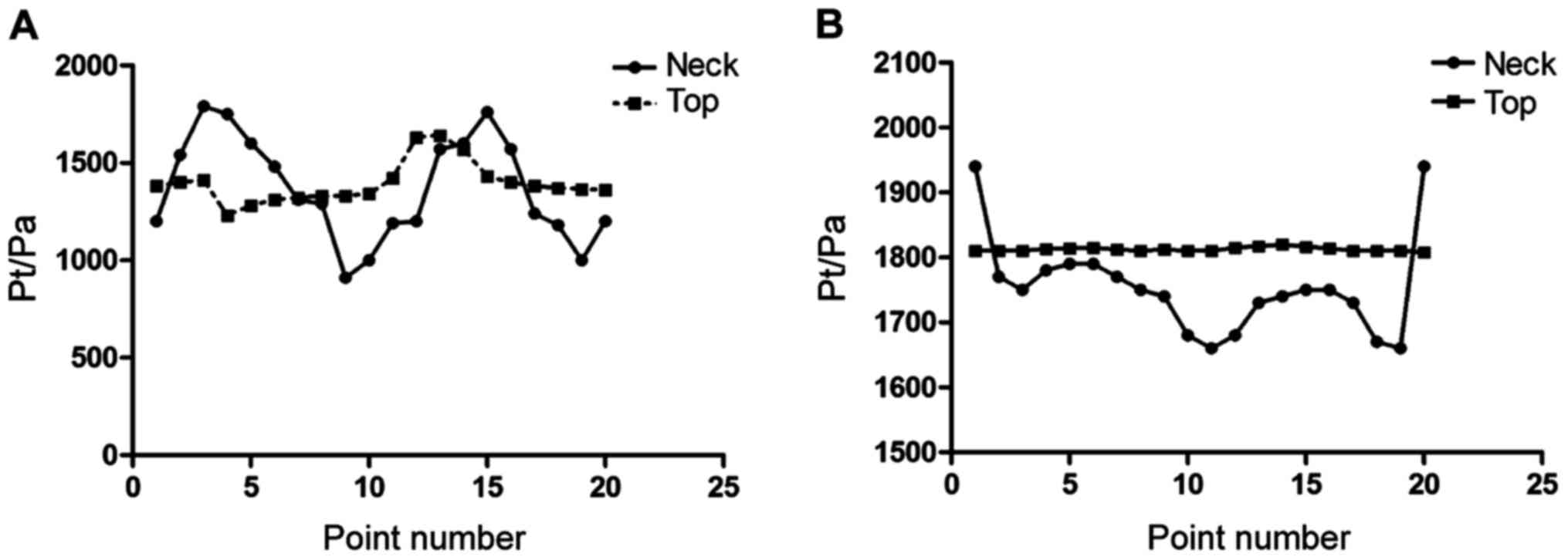
Pressure value calculating results of
each point
The results of this study showed that there was no
significant difference between the pressure of tumor neck (CA1 and
CA2) and the pressure on the tumor top (P>0.05), as shown in
Fig. 3.
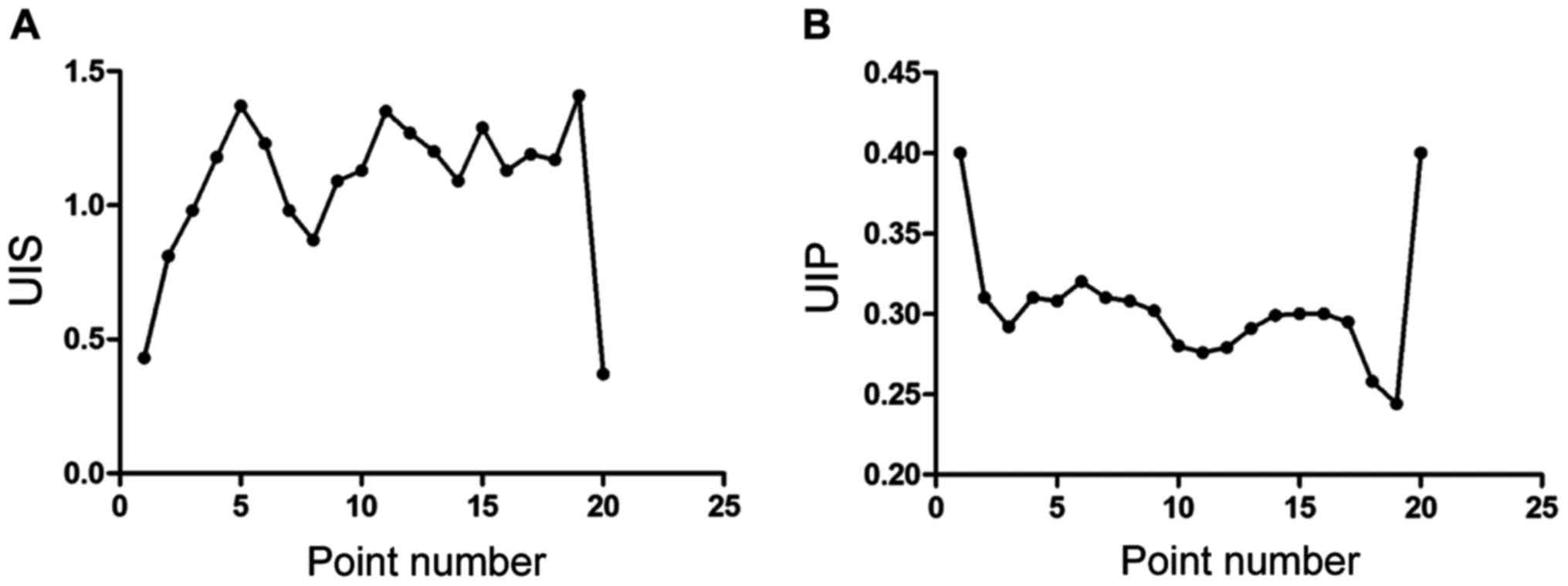
UIS and UIP calculation of results of
each point
The fluctuation of WSS and pressure caused by the
change of time were quantitative analyzed by unsteady index of
shear (UIS) and unsteady index of pressure (UIP). The results
showed that there has significant difference of UIS value changes
of these 20 points, 0.6–1.5 was the wave range. The UIP value of
each point was significantly lower than the UIS value, 0.25–0.40
was the wave range, as shown in Fig.
4.
Discussion
Related research showed that the main reason which
caused the occurrence of aneurysm were elastic friction in arterial
wall and destruction, relaxation, defect of smooth muscle layer
(6). The extra vascular elastic
membrane (EEM) plays a vital role in maintaining the elasticity,
length, bending degree of arterial wall (7). Jain et al injected elastase into
adventitial coat of abdominal aorta, spindle aneurysm can be seen
in 24 h, and the dissolving of middle-level elastic lamina,
endomembrane tissue injury and the disorganization of smooth
muscular tissue can also be seen, they reached the peak in the
third day, and the aortic aneurysm is significantly shrunken after
42 days (8). Some scholars pointed
out that the appearance of irreversible damage of smooth muscle
cells may cause the formation of aneurysm, and medial smooth muscle
may be more important than elastic film, elastase can help smooth
muscle cells to be regenerated (9).
The occurrence of vascular abnormalities is related to protein,
cytokines, apoptosis and inflammation. The extracellular matrix
(ECM) of artery includes reticular fibers and collagen fibers,
elastic fibers, noncollagenous glycoproteins, it can maintain the
tension and elasticity of the arterial wall effectively, and can
also maintain fixed shape under the impact of blood. Related
research results showed that, total collagen protein on the tumor
wall of patients with intracranial aneurysm reduced significantly
compared with total cerebral aneurysms (10). A large amount of collagen can be seen
distributing between adventitia of normal cerebral artery and
tunicae media, but collagen distribution trends to diffuse on
aneurysm wall, the immunofluorescence staining experiment result
showed that the immunofluorescence staining intensity of collagen
on aneurysm wall significantly abated. In normal cerebral artery
wall, the expression of fibronectin (FN) was strongest in
endothelial basement membrane (BM), and the expression of FN can be
seen both in endothelial basement adventitial membrane and tunicae
media membrane, but there was almost no expression of FN on the
wall of human intracranial aneurysm (11). In normal cerebral arteries, type IV
collagen is mainly distributed in subendothelial BM, it can be seen
with low expression around smooth muscle cell in media tunicae, the
distribution of type IV collagen were destroyed on the wall of
tumor, the expression of type IV collagen debated, distribution
dispersion can be seen, all of these caused significant reduction
of type IV collagen expression in original endothelium.
Hemodynamics may also be an important factor which caused the
occurrence and development of cerebral aneurysm, some researchers
analyzed the rat model and found that on side ligaturing the
cephalic artery can cause renal hypertension of rats, the
occurrence of cystic aneurysm, spindle aneurysm can be seen after
ligaturing the cephalic artery 12 months later, so we can conclude
that the change of homodynamic has close correlation with the
occurrence of aneurysm (10–12). Research has found that the pressure,
shear stress and tensile stress all were arterial wall stress.
In-depth research is required to clarify how the above stress
changed into biological signal when arterial wall was affected by
mechanical signal (13,14). In general, endothelial cells can
change into biochemical reaction and electrophysiology when they
meet mechanical force, leading to genetic expression changes and
biological change. Some researchers pointed out that hemodynamic
changes were the primary factors which caused the occurrence and
development of aneurysml (12–14). It
has been shown that internal pressure of aneurysm mainly act on the
tumor wall and then cause the occurrence of stretch stress, and
aneurysmal radius and intra tumor pressure all were positively
correlated with stretch stress, stretch stress presented negative
correlation with tumor wall thickness (15). Some researchers found that the stress
in tumor wall was 10 times higher than normal level (16). Along with the growth of aneurysm,
aneurysm tumor intramural degenerative change was always
accompanied by the increasing of tumor radius, the reducing of
tumor wall thickness, causing high intratumoral pressure and the
occurrence of progressive tensile stress. At the present stage, the
research on hemodynamics mainly include pathological vector model
analysis and the analysis of inner flow field of artery with
aneurysm artery, the analysis of inner flow field of artery with
aneurysm artery can be studied according hydrodynamic to analyze
the character of hemodynamics, seek for the relations between
hemodynamics and pressure distribution, velocity distribution, wall
shear force, this process has two parts: Numerical calculation and
in vitro mode test. Now, with the rapid development of
computer technology and imaging technology, the research on
numerical simulation of 3D imaging in clinic has increased. Related
research showed, the model has realistic geometry structure, can
make a bridge between clinic and experimental research (2,14–17). It
was pointed out that the growth and break of cerebral aneurysm had
close correlation with the distribution of WSS and pressure
(2,15–18). In
general, low WSS can cause aneurysm rupture, high WSS cause the
growth of aneurysm. The results of this research showed that the
WSS maximum value can be found in the position of vascular
bifurcation and tumor neck, and the WSS minimum value can be found
on the top of aneurysm, so the position with daughter tumor of
aneurysm will rupture easily, all the studies above were consistent
with the results of this study. Some researchers pointed out that
the growth and break of aneurysm had close correlation with the
change of pressure and shearing stress in vascular wall, so we
investigated the fluctuation of pressure and WSS with time using
UIS and UIP. The results of this research showed that the change
regulation of UIS, UP or WSS were not always in the same manner,
the results suggested that further research to analyze the
relativity of aneurysm's growth and break and UIS, UIP and WSS.
In conclusion, the research of cerebral aneurysm
hemodynamic can help doctors to judge the risk of aneurysm more
accurate, and help to grasp the opportunity of treatment and
developing treatment strategies.
References
|
1
|
Campodeaño L, Oliveira MSN and Pinho FT: A
review of computational hemodynamics in middle cerebral aneurysms
and rheological models for blood flow. Appl Mech Rev.
67:0308012015. View Article : Google Scholar
|
|
2
|
Futami K, Nambu I, Kitabayashi T, Sano H,
Misaki K, Uchiyama N and Nakada M: Inflow hemodynamics evaluated by
using four-dimensional flow magnetic resonance imaging and the size
ratio of unruptured cerebral aneurysms. Neuroradiology. 59:411–418.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mizuma A, Ishikawa T, Kajihara N, Takano
H, Endo K, Kawakata M, Shibukawa S, Nakamura T, Nishio H, Horie T,
et al: Dynamic cross-sectional changes of the middle cerebral
artery in atherosclerotic stenosis detected by 3·0-Tesla MRI.
Neurol Res. 36:795–799. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sarrami-Foroushani A, Villa-Uriol MC,
Esfahany M Nasr, Coley SC, Di Marco LY, Frangi AF and Marzo A:
Modeling of the acute effects of primary hypertension and
hypotension on the hemodynamics of intracranial aneurysms. Ann
Biomed Eng. 43:207–221. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhang Y, Jing LK and Zhang QQ: Analysis of
morphological and hemodynamic characteristics of unruptured
posterior communicating artery aneurysms with oculomotor nerve
paralysis. Chin J Neurosurg. 32:604–606. 2016.
|
|
6
|
Ivanov D, Dol A and Polienko A:
Patient-specific hemodynamics and stress-strain state of cerebral
aneurysms. Acta Bioeng Biomech. 18:9–17. 2016.PubMed/NCBI
|
|
7
|
Krings T, Piske RL and Lasjaunias PL:
Intracranial arterial aneurysm vasculopathies: Targeting the outer
vessel wall. Neuroradiology. 47:931–937. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Jain K, Jiang J, Strother C and Mardal KA:
Transitional hemodynamics in intracranial aneurysms - Comparative
velocity investigations with high resolution lattice Boltzmann
simulations, normal resolution ANSYS simulations, and MR imaging.
Med Phys. 43:6186–6198. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Frolov SV, Sindeev SV, Potlov AY and
Liepsch D: Numerical modeling of the effects of a flow-diverting
stent on hemodynamic characteristics in a cerebral aneurysm. Biomed
Eng (NY). 50:363–366. 2017. View Article : Google Scholar
|
|
10
|
Turek G, Lewszuk A, Kochanowicz J,
Zielińska-Turek J, Wilk P and Mariak Z: Evaluation of the
effectiveness of endovascular embolization for the treatment of
ruptured cerebral aneurysms. Pol Merkur Lekarski. 42:76–80.
2017.(In Polish). PubMed/NCBI
|
|
11
|
Qian Z, Feng X, Kang H, Wen X, Xu W, Li Y,
Jiang C, Wu Z and Liu A: Dissecting aneurysms of the distal segment
of the posterior cerebral artery: Clinical presentation and
endovascular management. Chin Neurosurg J. 3:72017.(In Chinese).
View Article : Google Scholar
|
|
12
|
Navratil O, Duris K, Juran V, Neuman E,
Svoboda K and Smrcka M: Middle cerebral artery aneurysms with
intracerebral hematoma-the impact of side and volume on final
outcome. Acta Neurochir (Wien). 159:543–547. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhang Y and Yang XJ: Advances in the study
of hemodynamics of stent and coil embolization in the treatment of
intracranial aneurysms. Chin J Cerebrovasc Dis. 13:372–375.
2016.
|
|
14
|
Saho T and Onishi H: Quantitative
comparison of hemodynamics in simulated and 3D angiography models
of cerebral aneurysms by use of computational fluid dynamics.
Radiological Phys Technol. 8:258–265. 2015. View Article : Google Scholar
|
|
15
|
Wang C, Tian Z, Liu J, Jing L, Paliwal N,
Wang S, Zhang Y, Xiang J, Siddiqui AH, Meng H, et al: Flow diverter
effect of LVIS stent on cerebral aneurysm hemodynamics: A
comparison with Enterprise stents and the Pipeline device. J Transl
Med. 14:1992016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Turjman AS, Turjman F and Edelman ER: The
role of fluid dynamics and inflammation in intracranial aneurysm
formation. Circulation. 129:373–382. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Huang C, Shi B and Chai ZGZ: Multi-GPU
based Lattice Boltzmann method for hemodynamic simulation in
patient-specific cerebral aneurysm. Commun Comput Phys. 17:960–974.
2015. View Article : Google Scholar
|
|
18
|
Nair P, Chong BW, Indahlastari A, Ryan J,
Workman C, Babiker M Haithem, Farsani H Yadollahi, Baccin CE and
Frakes D: Hemodynamic characterization of geometric cerebral
aneurysm templates treated with embolic coils. J Biomech Eng.
138:0210112016. View Article : Google Scholar : PubMed/NCBI
|