Introduction
Glaucoma filtration surgery has been established as
an effective therapeutic method for intraocular pressure control
(1). However, excess proliferation
of fibroblasts in the filtration area following surgery leads to
scarring and is the predominant cause of surgery failure (2–4). To
suppress the overreactive wound healing process and reduce bleb
scarring in high risk cases, antimetabolic agents, including
mitomycin-c and 5-fluorouracil (5-FU), may be used during surgery
(5). Clinically, the use of
antimetabolites may ameliorate postoperative effects in numerous
cases that demonstrate a high risk of failure (1,5). These
agents are thought to improve the success rate of surgery; however,
they are also associated with notable complications, including
hypotony, bleb leaks, blebitis and endophthalmitis (5).
It has been reported that transfer of the herpes
virus thymidine kinase (TK) gene into tumor cells could lead to
cell death following administration of a prodrug (6). The Eschericha coli gene coding
for the cytosine deaminase (CD) enzyme was identified to serve as
another suicide gene (7). These two
gene-directed enzyme prodrug therapies have been investigated
extensively, including studies in clinical trials (8). The double suicide gene system, TK-CD,
exhibited a stronger effect on cell death compared to the TK or CD
genes alone (7,9).
Viral vectors have been mostly used for suicide gene
therapy; however, certain limitations remain. For example,
retroviruses only transduce a dividing cell, lentiviruses may
induce oncogenesis and herpes simplex virus (HSV) may cause
inflammation (9–11). Nanoparticles exhibited unique
properties that may provide a platform to overcome the limitations
of viral vectors (12). The
spherical-like nanoparticles may be used to carry DNA for
protecting their cargo from degradation and regulating their
release (12). Furthermore, the use
of nanotechnology in drug delivery has been growing rapidly
recently (13). Advances in
molecular biology and genetic engineering have allowed suicide gene
therapy to represent a potential approach for regulating scarring
following glaucoma filtration surgery (8). The present study investigated the
antiproliferative effect of a double suicide gene, TK combined with
CD, using non-viral vector generation 5-polyamidoamine dendrimers
(G5-PAMAM-D) on human Tenon's capsule fibroblasts (HTFs) in
vitro.
Materials and methods
Cells, reagents and instruments
HTFs were obtained from Tenon's capsule tissues of 5
patients (age range, 36–61 years; mean age, 47.4±12.3 years; 4
males and 1 female, sample collection between October 2015 and
January 2016) undergoing surgery to treat glaucoma at Tianjin Eye
Hospital (Tianjin, China). Tissues were prepared for culture in 35
mm Petri tissue-culture dishes that contained Dulbecco's modified
Eagle's medium (DMEM; Tianjin Haoyang Biotechnology Co., Ltd.,
Tianjin, China), supplemented with 10% fetal bovine serum (Tianjin
Haoyang Biotechnology Co., Ltd.) Cells were incubated at 37°C in a
humidified environment of 95% air and 5% CO2. Following
tissue culture, third to sixth generation cells were prepared for
experiments. The present study was approved by the Institutional
Review Board of Tianjin Eye Hospital (Tianjin, China) and the Human
Research Ethics Committee of Tianjin Eye Hospital (Tianjin, China).
Patient informed consent was obtained prior to initiation of the
study.
The restriction enzymes EcoRI, XhoI,
HindIII and KpnI, and DNA ladder marker (1 kbp) were
purchased from Takara Bio, Inc., (Otsu, Japan). T4 DNA ligase,
Probest DNA polymerase and protein marker (2–212 kDa) were
purchased from New England Biolabs, Inc., (Ipswich, MA, USA). An
agarose gel DNA extraction kit was purchased from Tiangen Biotech
Co., Ltd., (Beijing, China). Ganciclovir (GCV) was purchased from
Shanghai New Pioneer Pharmaceutical Co., Ltd., (Shanghai, China)
and 5-flurocytosine (5-FC) was purchased from Shanghai Shunqiang
Biotechnology Co., Ltd., (Shanghai, China). MTT and dimethyl
sulfoxide (DMSO) were purchased from Sigma-Aldrich (Merck KGaA,
Darmstadt, Germany).
The instruments that were used in the present study
were as follows: GeneAmp9600PCR (PerkinElmer, Inc., Waltham, MA,
USA), OLYMPUS Ix53 optical microscope (Olympus Corporation, Tokyo,
Japan), an OLYMPUS IX71 inverted fluorescence microscope and camera
system (Olympus Corporation), BD FACS Calibur Flow Cytometer (BD
Biosciences, San Jose, CA, USA) and Philips EM400ST transmission
electron microscope (Philips Healthcare, Amsterdam, The
Netherlands).
Vector and plasmid construction
G5-PAMAM-D, plasmid pAcGFP1-Hyg-CD and E.
coli DH5α were kindly provided by the Pharmacy College of
Nankai University (Tianjin, China). The HSV/TK gene plasmid,
pMD18-TK, was obtained from Takara Bio, Inc. The CD expression
plasmid, pAcGFP1-Hyg-CD, was obtained by polymerase chain reaction
(PCR) and restriction enzyme digestion from the CD gene.
The TK gene fragment was amplified from the plasmid
pMD18-TK by PCR with Probest DNA polymerase (New England Biolabs,
Inc.). The thermocycling conditions for the reaction were as
follows: 94°C for 5 min, followed by 35 cycles of 94°C for 30 sec,
52°C for 30 sec, 72°C for 90 sec; followed by 72°C for 10 min. The
following primers were used: TK-S1, forward primer, 5′-GCT
AAGCTT ATG GCC TCG TAC CCC GGC CA-3′ (italicized part
indicates the cutting site of HindIII) and TK-A1, reverse
primer, 5′-CGC GGATCC TCA GTT AGC CTC CCC CAT-3′ (italicized
part indicates the cutting site of BamHI). The amplified TK
gene PCR fragment (1,143 bp) and the expression vector
pAcGFP1-HygN1 (5.8 kb; Clontech Laboratories, Inc., Mountainview,
CA, USA) were digested with BamHI and HindIII and
then ligated to obtain the pAcGFP1-Hyg-TK product (37°C for 4
h).
The TK gene fragment was further amplified by PCR
with Probest DNA polymerase. The thermocycling conditions for the
reaction were as described above. The following primers were used:
TK-S2, forward primer, 5′-CCG CTCGAG ATG GCC TCG TAC CCC GGC
CAT CAA CA-3′ (italicized part indicates the cutting site of
XhoI) and TK-A2, reverse primer, 5′-CGA AAGCTT ACC
AGA ACC ACC GTT AGC CTC CCC CAT CTC CCG GG CA-3′ (italicized part
indicates the cutting site of HindIII). The TK gene PCR
fragment (1,143 bp) and expression vector, pAcGFP1-Hyg-CD (6,262
bp), were digested by XhoI and HindIII and then
ligated to obtain the final plasmid, pAcGFP1-Hyg-TK-CD. Finally,
the plasmid sequence was verified by DNA sequencing, and the
plasmid was further amplified and purified by a
QIAfilter™ plasmid maxi kit (Qiagen China Co., Ltd.,
Shanghai, China), according to the manufacturer's protocol.
HTF plasmid transfection
A total of 2.0 µg/ml pAcGFP1-Hyg-TK-CD plasmid and
PAMAM-D were mixed (mass ratio, 1:2) and incubated for 30 min at
25°C. Logarithmic growth phase HTFs were trypsinized and plated in
24-well plates (5×104/well). After 24 h when the cell
confluency reached 70–75%, cells were divided into experimental
(G5-PAMAM-D/TK-CD), positive control (Lipofectamine 2000
transfection and Lipo/TK-CD) and negative control
(pAcGFP1-Hyg-TK-CD plasmid alone) groups, and each assay was
repeated in triplicate. Lipofectamine® 2000 was
purchased from Invitrogen (Thermo Fisher Scientific, Inc.).
TK and CD gene expression
analysis
After 48 h of transfection, transfection efficiency
was assessed by observation of green fluorescent protein
expression. Total cell RNA was extracted from transfected HTFs 48 h
after transfection with TRIzol reagent (Invitrogen; Thermo Fisher
Scientific, Inc.) and then one-step reverse transcription (RT)-PCR
was performed using a Transcriptor One-step RT-PCR kit (Roche
Diagnostics, Basel, Switzerland), according to the manufacturer's
protocol. The thermocycling conditions for the RT reaction were as
follows: 42°C for 2 h, 95°C for 5 min, preserved at 20°C. PCR
reaction conditions were as follows: 95°C for 5 min, followed by 32
cycles of 94°C for 50 sec, 52°C for 50 sec, 72°C for 1 min, then
72°C for 8 min. The following primers were used: Forward,
5-GGGTCTAGAATGGCTTCGTACCCC-3 and reverse,
5-TCTGTTAACTCAGTTAGCCTCCCCCATCTCCCG-3. The TK-CD PCR product was
analyzed by 1% agarose gel electrophoresis and an Image Master
Total Lab gel imaging analytical system version 2.0 (TotalLab Ltd.,
Newcastle upon Tyne, UK) was used to analyze nucleic acid
bands.
Cell viability assay
MTT colorimetric assay was used to evaluate the
lethal effect and optimal concentration of a prodrug on HTF
and HTF-TK-CD cells. Cells were plated in 96-well plates (cell
concentration lx104 cells/ml) in 200 µl DMEM. After 24 h
of culture, HTF-TK-CD and HTF cells were observed to exhibit robust
growth under a light microscope. The concentration of GCV was
tested at 0, 1, 2, 3, 4, 5, 8 or 10 µg/ml, and the concentration of
5-FC was tested at 0, 50, 100, 200, 400, 600, 800 or 1,000 µg/ml.
The blank control, experimental and control groups were set up in
four replicate groups. In total, 72 h after GCV and 5-FC treatment
at 37°C, the cell viability was determined using the MTT assay. A
total of 20 µl MTT solution (5 mg/ml) was added to each well and
incubated for 4 h at 37°C. The culture medium was then removed,
followed by the addition of 150 µl DMSO to each well and incubation
for an additional 10 min. Subsequently, the absorbance (optical
density; OD) of each well was measured using an enzyme immunoassay
analyzer at a wavelength of 490 nm. The cell survival rate (%) was
calculated according to the following formula: (Experimental group
OD value/blank control group OD value) x100.
To evaluate the bystander effect of the TK-CD gene,
untransfected cells were mixed in an increased proportion of
HTF-TK-CD cells at 10, 20, 30, 40, 50, 60, 70, 80, 90 or 100%. The
cells were then plated into 96-well plates at lx104
cells/well in four replicate groups.
To each well, 3 µg/ml GCV and/or 200 µg/ml 5-FC were
added in DMEM. After 72 h at 37°C, the cell inhibition ratio was
determined using the MTT colorimetric assay, where the cell
inhibition rate (%) was calculated according to the following
formula: (1-experimental group OD value/blank control group OD
value) x100.
Light and transmission electron
microscopy
A total of 1×105 HTFs cells or HTF-TK-CD
cells were divided into groups A, B, C and D and plated into 6-well
culture plates. GCV and/or 5-FC in DMEM media was added as follows:
Group A, GCV 3 µg/ml; group B, 5-FC 200 µg/ml; group C, GCV 3 µg/ml
and 5-FC 200 µg/ml; and group D, DMEM without prodrug. Each
condition was prepared in duplicate. Furthermore, the morphological
changes of the cells were observed using an inverted microscope
after 24 h (37°C) of prodrug treatment.
For transmission electron microscopy, after 72 h
(37°C) of cell culture, cells were washed with PBS three times,
trypsinized and collected. Cells were then centrifuged at 251.5 × g
(room temperature) for 2 min, washed with 4°C precooled PBS twice,
fixed in 4°C precooled 3% glutaraldehyde for 4 h and rinsed with
PBS for 10 min three times. Subsequently, cells were fixed with 1%
osmium tetroxide for 2 h at room temperature, rinsed with PBS for
10 min three times, dehydrated with 30–100% alcohol and embedded
(at 37°C) with SPI-PON 812 resin. Following this, the samples were
cut into slices (100 nm thick) with a LKBV-type ultramicrotome,
double-stained with 3% uranyl acetate for 3 min and 2% lead citrate
for 20 min at room temperature and observed under a transmission
electron microscope (magnification, ×3,400 or ×5,700).
Statistical analysis
Statistical analysis was performed using SPSS
version 19.0 (IBM Corp., Armonk, NY, USA). Data were presented as
the mean ± standard deviation. The effects of different
concentrations of GCV and 5-FC on the two groups were analyzed
using linear regression analysis. Differences between the combined
and individual treatment groups in cell survival were compared
using two-way analysis of variance (ANOVA) for each concentration.
The Student-Newman-Keuls (SNK) method was used for pair-wise
comparisons and comparisons among various groups were performed
using Dunnett's test. P<0.05 was considered to indicate a
statistically significant difference.
Results
TK-CD plasmid verification and
expression in HTF cells
To verify the constructed plasmid
(pAcGFP1-Hyg-TK-CD), the plasmid was digested with XhoI and
KpnI and the 1,617 bp fragment of the TK-CD gene was
confirmed by DNA gel electrophoresis on a 1% agarose gel (Fig. 1A and B). RT-PCR analysis indicated
that the expected band (403 bp) was observed in HTF cells
transfected with the pAcGFP1-Hyg-TK-CD plasmid, while β-actin (561
bp) was present in the transfected and non-transfected groups
(Fig. 1C). TK and CD gene expression
mediated by either Lipofectamine® 2000 or G5-PAMAM-D was
further accessed by fluorescence microscopy. As indicated in
Fig. 1D, TK-CD-green fluorescent
protein expression was observed in Lipofectamine®
2000-transfected and G5-PAMAM-D-mediated HTF cells.
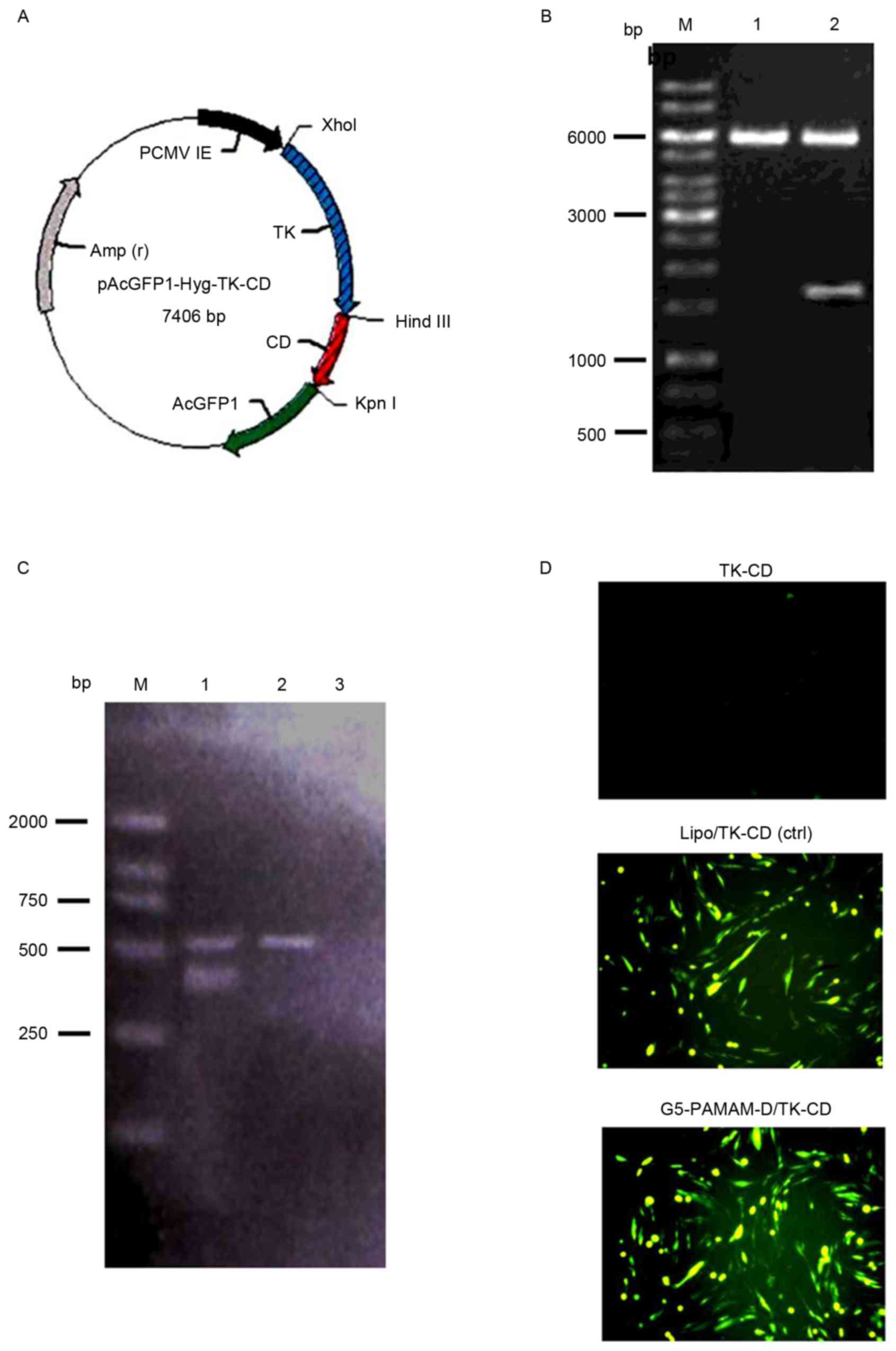 | Figure 1.TK and CD gene expression in HTF
cells. (A) The plasmid map showing the expression vector,
pAcGFP1-Hyg, carrying the TK and CD genes. (B) The 1,617 bp
fragment of the TK-CD gene was confirmed by restriction enzyme
digestion of the plasmid with XhoI and KpnI followed
by 1% DNA gel electrophoresis. Lane M, DNA marker; lane 1, vector
pAcGFP1-Hyg; and lane 2, pAcGFP1-Hyg-TK-CD. (C) Reverse
transcription-polymerase chain reaction analysis of HFT cells
transfected with pAcGFP1-Hyg-TK-CD. Lane M, DL2000 marker; lane 1,
fragment of TK-CD (403 bp) and β-actin (561 bp) in the transfected
cells; lane 2, untransfected; and lane 3, negative control groups.
(D) TK and CD gene expression mediated either by Lipo or G5-PAMAM-D
was accessed by fluorescence microscopy (magnification, ×100). TK,
thymidine kinase; CD, cytosine deaminase; HTFs, human Tenon's
capsule fibroblasts; G5-PAMAM-D, 5-polyamidoamine dendrimers; bp,
base pairs; Lipo, Lipofectamine 2000; ctrl, control. |
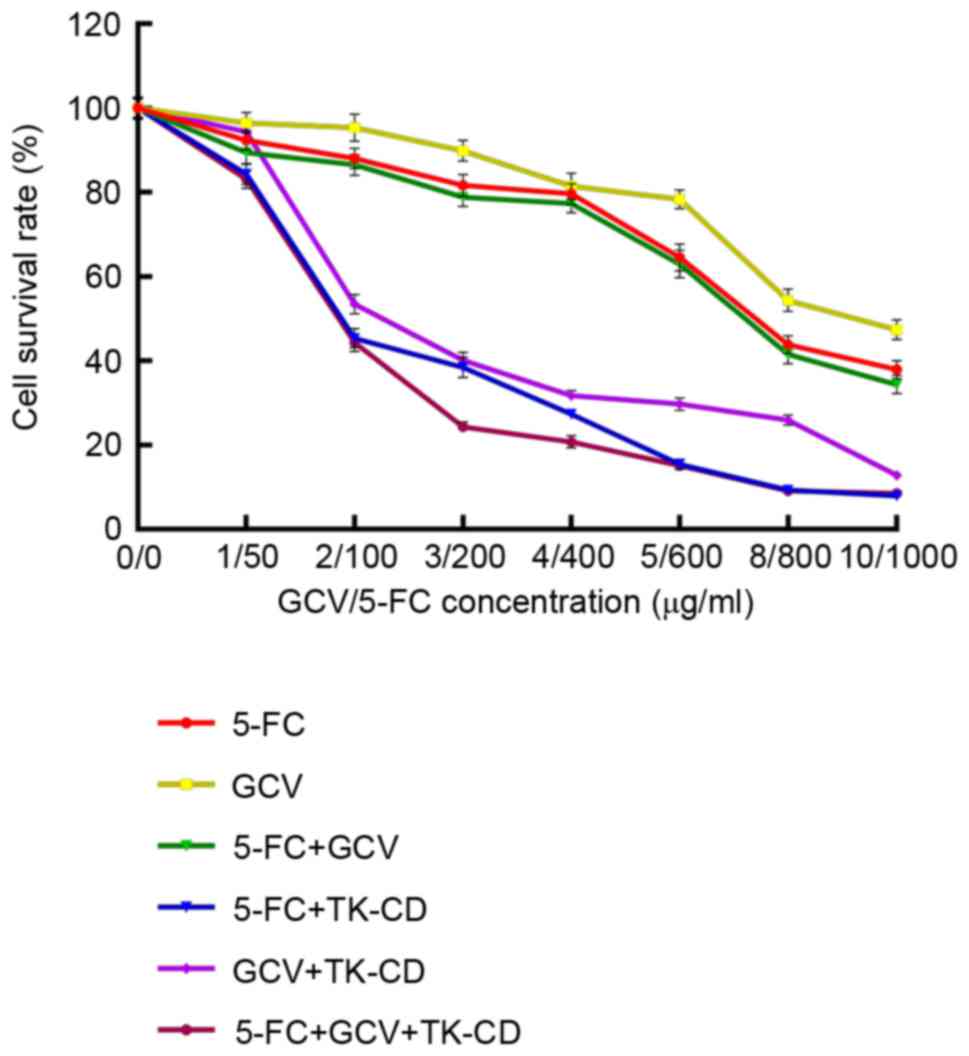
Dose optimization of prodrug on HTF
and HTF-TK-CD cells
The MTT assay in HTF cells indicated that the cell
survival rate significantly decreased as the concentration of GCV
and/or 5-FC added increased (all P<0.05; Fig. 2). A significant linear correlation
(YGCV=103.81–5.67X, P<0.05;
Y5-FC=97.06–0.06X, P<0.05;
YGCV+5-FC=112.68–9.18X, P<0.05) was detected by
simple linear regression analysis. Similarly, the survival rate of
HTF-TK-CD cells significantly decreased as the concentration of GCV
and/or 5-FC added increased (all P<0.05); however, no linear
correlation was detected. ANOVA analysis (with SNK method)
indicated that the F values for 5-FC, GCV and GCV+5-FC were
3,150.07, 3,125.22 and 3,114.35, respectively. The cell survival
rate was significantly decreased (P<0.05) when the 5-FC
concentration was <400 µg/ml. When the concentration for the
5-FC group was 100, 200 or 1,000 µg/ml, the HTF cell survival rate
was 88.07, 81.66 or 40.03%, respectively. In the HTF-TK-CD group,
100 µg/ml of 5-FC appeared to be toxic to the cells as its cell
survival rate decreased to 45.38% when compared to 88.07% of the
cell survival rate in the HTF group. Similarly, a cell survival
rate of 38.46% at 200 µg/ml of 5-FC in the HTF-TK-CD group was
comparable to a cell survival rate of 40.03% at 1,000 µg/ml of 5-FC
in the HTF group. When the 5-FC concentration was >200 µg/ml in
the HTF-TK-CD group (400, 600, 800 and 1000 µg/ml), the cell
survival rate was decreased, but it was not significantly different
(all P>0.05). By contrast, when the 5-FC concentration was ≤200
µg/ml, the cell survival rate was significantly different (all
P<0.05).
Therefore, 200 µg/ml of 5-FC was selected for
subsequent experiments as this concentration could prevent
5-FC-mediated cytotoxicity and effectively exert an inhibitory
effect on the suicide gene. Similarly, 3 µg/ml of GCV was also
selected based on the same criteria. In total, 200 µg/ml 5-FC
and/or 3 µg/ml GCV represented an optimal concentration and were
used in the present study.
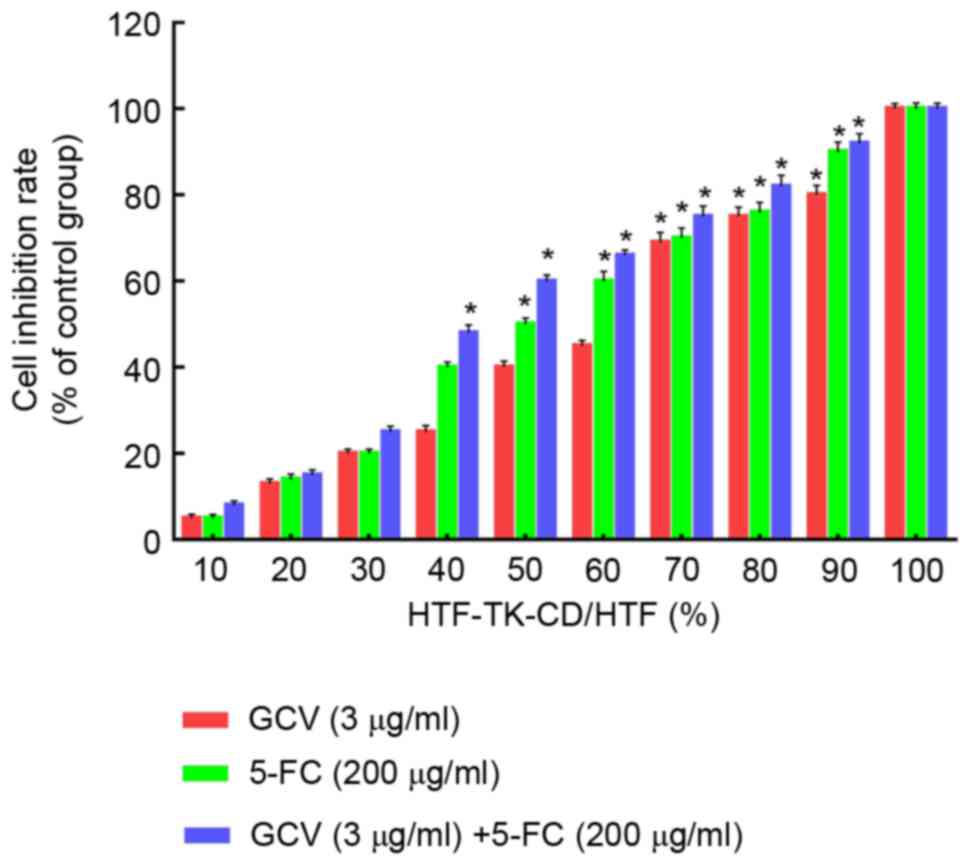
Bystander effect of the TK-CD double
suicide gene
The bystander effect of the TK-CD suicide gene was
further evaluated. As indicated in Fig.
3, 3 µg/ml GCV and/or 200 µg/ml 5-FC demonstrated an increased
inhibition of cell proliferation when HTF cells were mixed with an
increased number of HTF-TK-CD cells. When the ratio of transfected
cells was 40% (HTF-TK-CD: HTF ratio=4:6), the combined treatment
with 5-FC/GCV revealed a significant inhibition of 48.6±1.28%
compared to the control (P<0.05); however, a significant
difference was not observed with 5-FC (38.2±0.47%) or GCV
(24.6±0.86%) treatment alone. Similarly, when the ratio of
transfected cells was >70%, all three groups of 5FC/GCV combined
group, 5FC group and GCV group revealed significantly different
cell proliferation inhibition rates, and the 5-FC/GCV combination
group appeared to be the most significantly different among the
three groups (Tcombined with 5-FC=6.37, Tcombined
with GCV=11.32; P<0.05). These data indicated that the
bystander effect of 5FC/GCV combined group was greater than 5-FC or
GCV treatment alone.
Light and transmission electron
microscopy
The effect of 5-FC and GCV on HTF cell morphology
was accessed by light microscopy. The morphology of HTF cells
following treatment with 3 µg/ml GCV and/or 200 µg/ml 5-FC was
comparable to non-treated HTF cells (Fig. 4A and B). HTF cells were relatively
transparent, adherent and healthy following treatment with GCV
and/or 5-FC. The number of cells and cellular proliferation
demonstrated no clear difference amongst the three groups (GCV,
5-FC or GCV/5-FC). Furthermore, changes of morphology were observed
in HTF-TK-CD cells following treatment with GCV and/or 5-FC as
compared with the control group (Fig.
4C). Additionally, the quantity of intracellular granules
increased, cells shrunk, exhibited lower adherence and appeared
less healthy. The number of cells was also reduced among the three
groups as compared with the number in the control group.
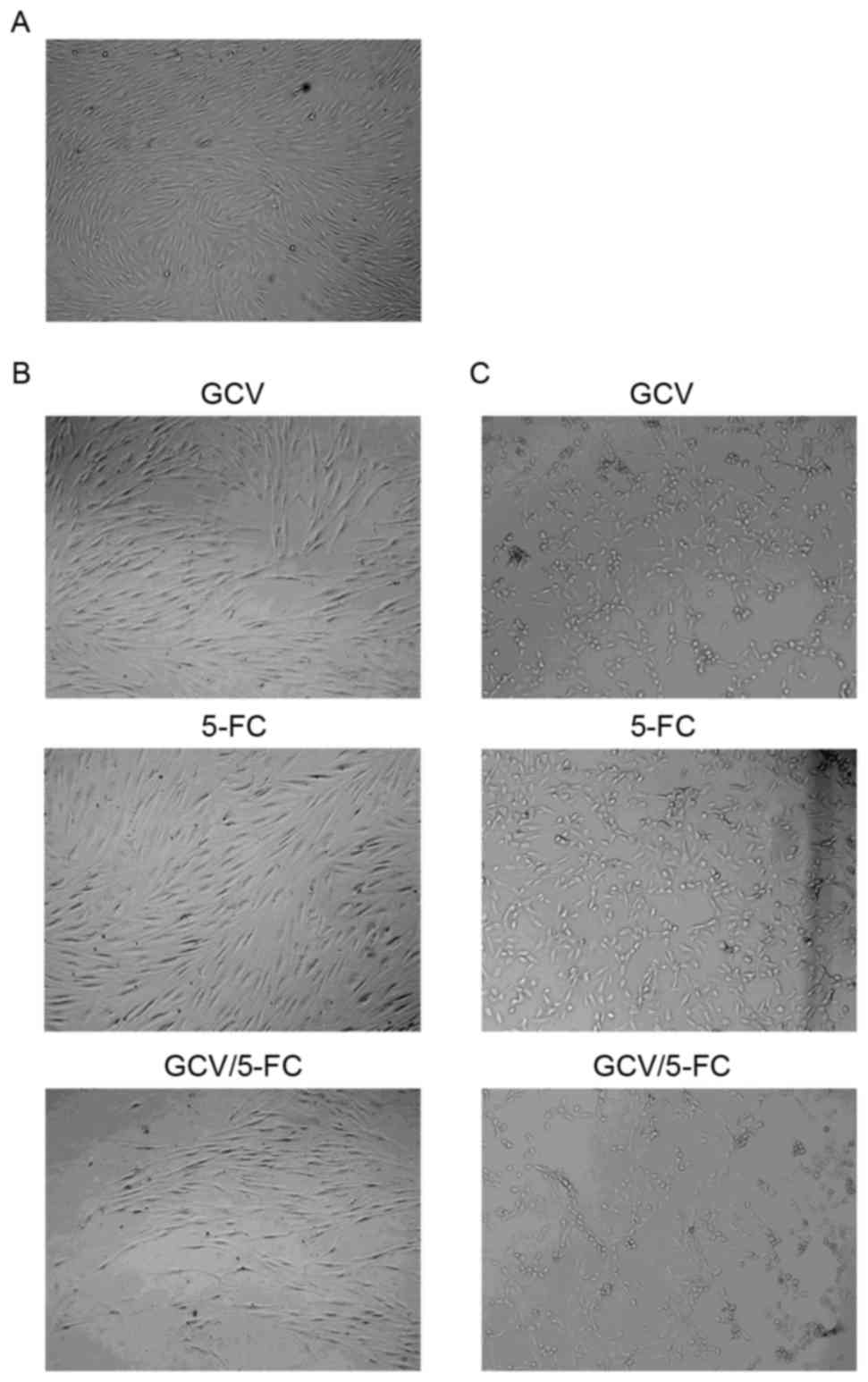 | Figure 4.Effect of 5-FC and GCV on HTF cell
morphology was accessed by light microscopy. (A) Normal HTF cells
demonstrated typical array polarity (magnification, ×100). (B) HTF
cells treated with GCV, 5-FC or GCV/5-FC (magnification, ×100) (C)
HTF-TK-CD cells treated with GCV, 5-FC or GCV/5-FC (magnification,
×100). 5-FC, 5-flurocytosine; GCV, ganciclovir; HTFs, human Tenon's
capsule fibroblasts; TK, thymidine kinase; CD, cytosine
deaminase. |
The ultrastructure of the HTF cells was further
evaluated using a transmission electron microscope. As indicated in
Fig. 5A and B, the normal HTF cells
demonstrated a spindle shape. The oval-shaped nucleus was large,
and represented 1/3 of the cell, which also contained one, two or
multiple nucleoli. Furthermore, the cells had a clearly dilated
cystic shape with rough endoplasmic reticulum that contained
isopycnic fine particles or an isotropic substance. The
intracellular space was filled with free Palade granules and
ribosomes. By contrast, morphological changes typical of apoptosis
could be observed in the HTF-TK-CD cell group treated with GCV and
5-FC for 72 h. The characteristics of apoptosis included cell
pyknosis, chromatin condensation, disappearance of organelles,
cytomorphosis and typical apoptotic bodies. However, the cell
membrane retained its integrity (Fig. 5C
and D).
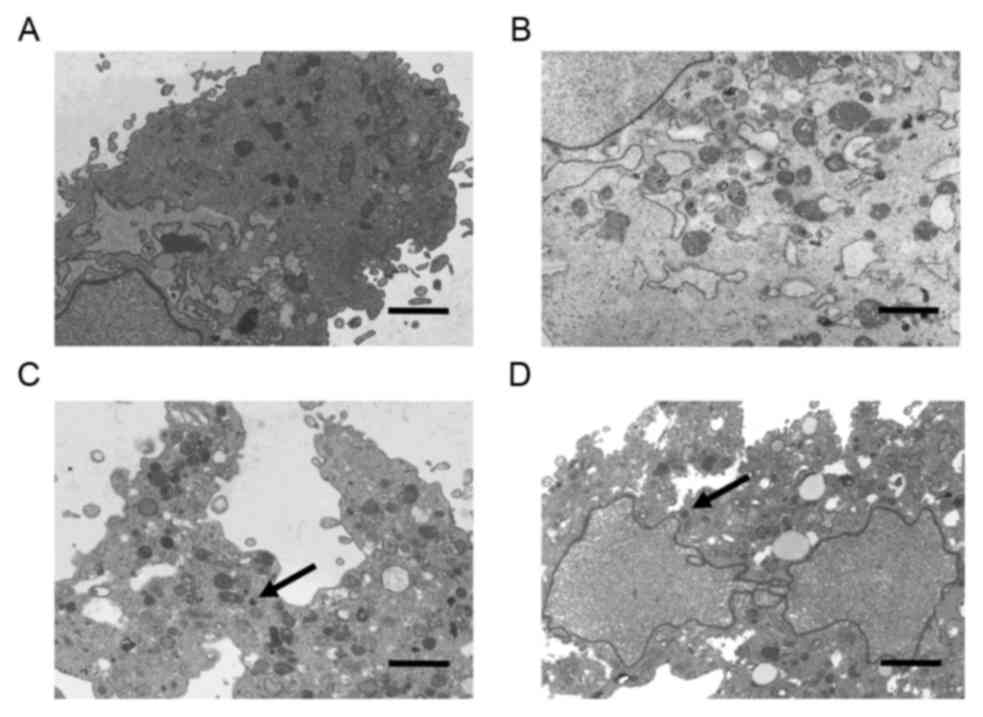 | Figure 5.Effect of 5-FC and GCV on HTF cells
was accessed by transmission electron microscopy. (A and B)
Ultrastructure of normal HTFs. (C and D) Larger regular chromatin
ball typical for apoptosis was observed in HTFs-TK-CD treated with
the prodrug for 72 h (apoptotic body, black arrows). Scale bar, 2
µm. Cells were double-stained with 3% uranyl acetate and lead
citrate. Magnification: A, ×3,400; B, ×5,700; C, ×3,400; D, ×3,400.
5-FC, 5-flurocytosine; GCV, ganciclovir; HTFs, human Tenon's
capsule fibroblasts; TK, thymidine kinase; CD, cytosine
deaminase. |
Discussion
Suicide gene therapy is a gene-directed enzyme
prodrug therapy that is based on the introduction of a viral or
bacterial gene that encodes a metabolic enzyme into target cells,
which allows the conversion of a non-toxic compound into a lethal
drug, causing the death of target cells (14). The ‘bystander effect’ is a phenomenon
whereby the transduction of a small fraction of target cells with
the suicide gene may result in widespread target cell death,
including the non-transduced cells (14,15). The
most frequently used suicide gene therapy for clinical trials
approved by the Food and Drug Administration is the (HSV-TK)/GCV
system along with the CD/5-FC system (8). GCV is an antiviral drug, which may be
transformed into its phosphorylated form by the enzyme when
introduced into cells. The cells that express TK make it
susceptible to GCV drug-induced cytotoxicity (16). However, the drug has no or low
toxicity for normal mammalian cells. 5-FC is an antifungal drug
compound that is non-cytotoxic but may be transformed into 5-FU
following deamination by the CD gene, which may act to inhibit
cellular proliferation (14).
It has been reported that suicide gene therapy may
inhibit HTF proliferation when delivered using a viral vector, more
evidently for single suicide gene transfection (17). The disadvantages of the viral vector
system represent potential immunogenicity, carcinogenicity,
inability to load long fragments and the generation of virion
particles during viral recombination (18,19).
Additionally, there are several advantages when using
PAMAM-D-mediated gene delivery compared with using a viral vector.
Firstly, it is avirulent to an organism, lacks immunogenicity and
is able to load long fragments of DNA (20,21).
Secondly, it protects the target gene from destruction by
complement system in plasma or histocytes (20,21).
Lastly, it may mediate exogenous gene integration in host cell
chromosomal DNA, which may result in long-lasting and stable
expression of a transgene (12,21).
Double suicide gene therapy represents the genetic
integration of two types of suicide genes that express a fusion
gene product with a double gene encoding enzyme activity in the
target cell (21). In the present
study, the plasmid of the double suicide gene, pAcGFP1-Hyg-TK-CD,
was used to transfect HTF cells with a G5-PAMAM-D vector. The
results of the MTT assay indicated that the cell survival ratio was
significantly reduced when an increased concentration of the
prodrug was added to the TK-CD-transfected HTF cells. Furthermore,
the cell survival rate in the drug combination group was evidently
lower than that in the single drug group. When 3 µg/ml GCV + 200
µg/ml 5-FC was administered, the cell survival rate was 24.35%,
which was lower than that of the single administration of 3 µg/ml
of GCV or 200 µg/ml of 5-FC (40.18 and 38.46%, respectively).
Furthermore, inhibition of transfected cells with the drug
combination was greater than that of cells treated with a single
drug. This result indicated that the TK-CD gene combination could
exhibit a synergetic effect due to differentially-expressed enzymes
that possess different sites of action, and the present data are
consistent with the result from a previous study (22). There are several possible reasons
that may account for this synergetic effect (7,9).
Firstly, TK but not CD is a cell cycle-specific agent and the TK-CD
combination could kill cells in dividing and quiescent stages.
Secondly, TK/GCV exerts its ‘bystander effect’ on cellular gap
junctions, while 5-FC acts on cell membranes, and CD/5-FC was
independent of cellular gap junctions, allowing the combination to
kill target cells with and without gap junctions (7). Lastly, the activity of GCV
phosphorylation with HSV/TK was significantly increased in the
presence of 5-FC.
In the present study, by observing HTF cells
transfected with TK-CD and exposed to the prodrug GCV and 5-FC,
deterioration of cell growth and shrinkage were observed, as well
as a reduction in the number of cells. For the GCV and 5-FC
combination compared with prodrug alone, cell growth was diminished
and there were fewer cells left following prodrug treatment.
HTF-TK-CD cell nuclear chromatin margination, cytoplasmic
condensation and cell deformation were observed by transmission
electron microscopy. Additionally, typical apoptotic bodies were
visible while the cell membrane was intact, indicating that the
cells were in an ongoing apoptotic process. Altogether, these
observations indicated that the TK-CD double suicide gene system
could initiate apoptosis in HTF cells.
While G5-PAMAM-D-mediated gene transfection
efficiency requires further improvement, the notable characteristic
of the suicide gene/prodrug system is the existence of a ‘bystander
effect’, whereby the transduction of a small fraction of target
cells with the suicide gene may result in widespread target cell
death, including non-transduced cells, causing exacerbation of the
lethal effect. A preliminary study of the mechanism of the
‘bystander effect’ was reported previously (23). Different suicide genes may act by
different mechanisms, for example the TK gene, but not the CD gene,
was localized to gap junctions (7).
Furthermore, the system may be effective without reaching high
transfection efficiency. The ‘bystander effect’ of double suicide
genes is more powerful than that of a single suicide gene (24). Nevertheless, further theoretical and
experimental studies are warranted to demonstrate that double
suicide genes act by synergism or accumulation (24).
In summary, the present study demonstrated that the
G5-PAMAM-D nanoparticle-mediated double suicide gene TK and CD
system inhibited HTF cell proliferation in vitro, and
exerted a ‘bystander effect’. The present study may provide a novel
framework for gene therapy to treat filtering bleb scarring
following glaucoma filtering surgery. HTF-specific delivery by
G5-PAMAM-D-mediated double suicide TK-CD gene with the addition of
specific fibroblast enhancers or promoters upstream of the suicide
gene may be used as an antiscarring agent and provide a therapeutic
potential for patients receiving glaucoma filtration surgery.
Acknowledgements
The present study was supported by Tianjin Municipal
Science and Technology Commission (grant no. 13JCYBJC21500).
References
|
1
|
Law SK, Shih K, Tran DH, Coleman AL and
Caprioli J: Long-term outcomes of repeat vs initial trabeculectomy
in open-angle glaucoma. Am J Ophthalmol. 148:685–695.e1. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Steplewski A and Fertala A: Inhibition of
collagen fibril formation. Fibrogenesis Tissue Repair. 5 Suppl
1:S292012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gedde SJ, Heuer DK and Parrish RK II: Tube
Versus Trabeculectomy Study Group: Review of results from the tube
versus trabeculectomy study. Curr Opin Ophthalmol. 21:123–128.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Seet LF, Su R, Barathi VA, Lee WS, Poh R,
Heng YM, Manser E, Vithana EN, Aung T, Weaver M, et al: SPARC
deficiency results in improved surgical survival in a novel mouse
model of glaucoma filtration surgery. PLoS One. 5:e94152010.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lama PJ and Fechtner RD: Antifibrotics and
wound healing in glaucoma surgery. Surv Ophthalmol. 48:314–346.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Moolten FL: Tumor chemosensitivity
conferred by inserted herpes thymidine kinase genes: Paradigm for a
prospective cancer control strategy. Cancer Res. 46:5276–5281.
1986.PubMed/NCBI
|
|
7
|
Lee YJ, Galoforo SS, Battle P, Lee H,
Corry PM and Jessup JM: Replicating adenoviral vector-mediated
transfer of a heat-inducible double suicide gene for gene therapy.
Cancer Gene Ther. 8:397–404. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Karjoo Z, Chen X and Hatefi A: Progress
and problems with the use of suicide genes for targeted cancer
therapy. Adv Drug Deliv Rev. 99:113–128. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Moriuchi S, Wolfe D, Tamura M, Yoshimine
T, Miura F, Cohen JB and Glorioso JC: Double suicide gene therapy
using a replication defective herpes simplex virus vector reveals
reciprocal interference in a malignant glioma model. Gene Ther.
9:584–591. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Martín F, Chowdhury S, Neil S, Phillipps N
and Collins MK: Envelope-targeted retrovirus vectors transduce
melanoma xenografts but not spleen or liver. Mol Ther. 5:269–274.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Vargas J Jr, Klotman ME and Cara A:
Conditionally replicating lentiviral-hybrid episomal vectors for
suicide gene therapy. Antiviral Res. 80:288–294. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Eichman JD, Bielinska AU, Kukowska-Latallo
JF and Baker JR Jr: The use of PAMAM dendrimers in the efficient
transfer of genetic material into cells. Pharm Sci Technolo Today.
3:232–245. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Andonova VY: A new direction in ophthalmic
development: Nanoparticle drug delivery systems. Curr Pharm Des.
22:6313–6329. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chaszczewska-Markowska M, Stebelska K,
Sikorski A, Madej J, Opolski A and Ugorski M: Liposomal formulation
of 5-fluorocytosine in suicide gene therapy with cytosine
deaminase-for colorectal cancer. Cancer Lett. 262:164–172. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Duarte S, Carle G, Faneca H, de Lima MC
and Pierrefite-Carle V: Suicide gene therapy in cancer: Where do we
stand now? Cancer Lett. 324:160–170. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Candice LW, Django S and Margaret EB: The
role of herpes simplex virus-1 thymidine kinase alanine 168 in
substrate specificity. Open Biochem J. 2:60–66. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang JB, Ge J, Liu BQ, Huang B and Wei YT:
Anti-proliferative effect of herpes simplex virus thymidine kinase
gene system on human Tenon capsule fibroblasts in vitro. Zhonghua
Yan Ke Za Zhi. 42:212–217. 2006.PubMed/NCBI
|
|
18
|
Larocca C and Schlom J: Viral vector-based
therapeutic cancer vaccines. Cancer J. 17:359–371.. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kajiwara E, Kawano K, Hattori Y, Fukushima
M, Hayashi K and Maitani Y: Long-circulating liposome-encapsulated
ganciclovir enhances the efficacy of HSV-TK suicide gene therapy. J
Control Release. 120:104–110.. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Taghavi Pourianazar N and Gunduz U: CpG
oligodeoxynucleotide-loaded PAMAM dendrimer-coated magnetic
nanoparticles promote apoptosis in breast cancer cells. Biomed
Pharmacother. 78:81–91.. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen Y, Wang G, Kong D, Zhang Z, Yang K,
Liu R, Zhao W and Xu Y: In vitro and in vivo double-enhanced
suicide gene therapy mediated by generation 5 polyamidoamine
dendrimers for PC-3 cell line. World J Surg Oncol. 10:32012.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Choi JS, Nam K, Park JY, Kim JB, Lee JK
and Park JS: Enhanced transfection efficiency of PAMAM dendrimer by
surface modification with L-arginine. J Control Release.
99:445–456. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Denny WA: Prodrugs for gene-directed
enzyme-prodrug therapy (Suicide Gene Therapy). J Biomed Biotechnol.
2003:48–70. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Jia W, Mei L, Wang Y, Liu L and Che G:
Double suicide genes selectively kill human umbilical vein
endothelial cells. Virol J. 8:742011. View Article : Google Scholar : PubMed/NCBI
|