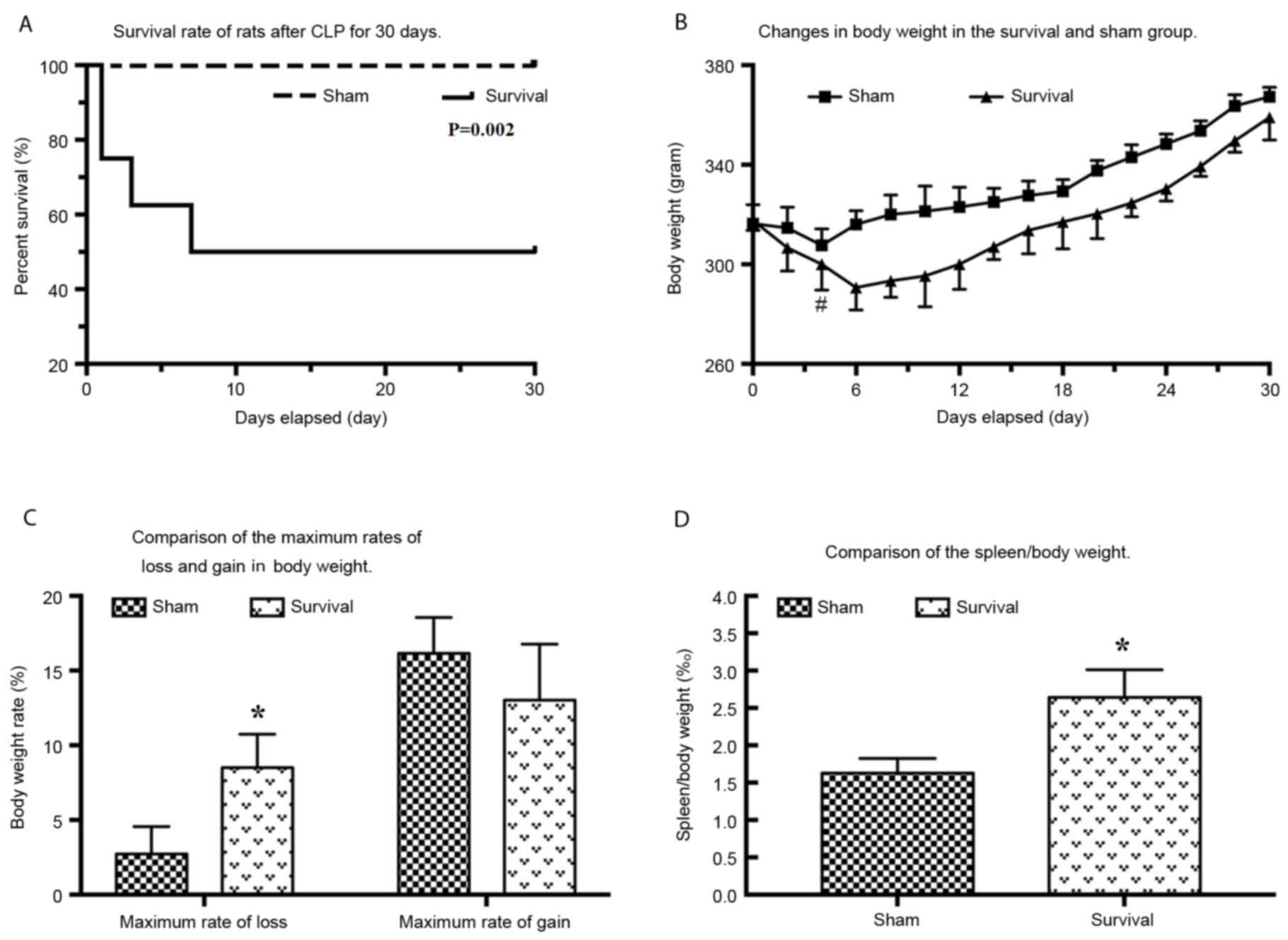
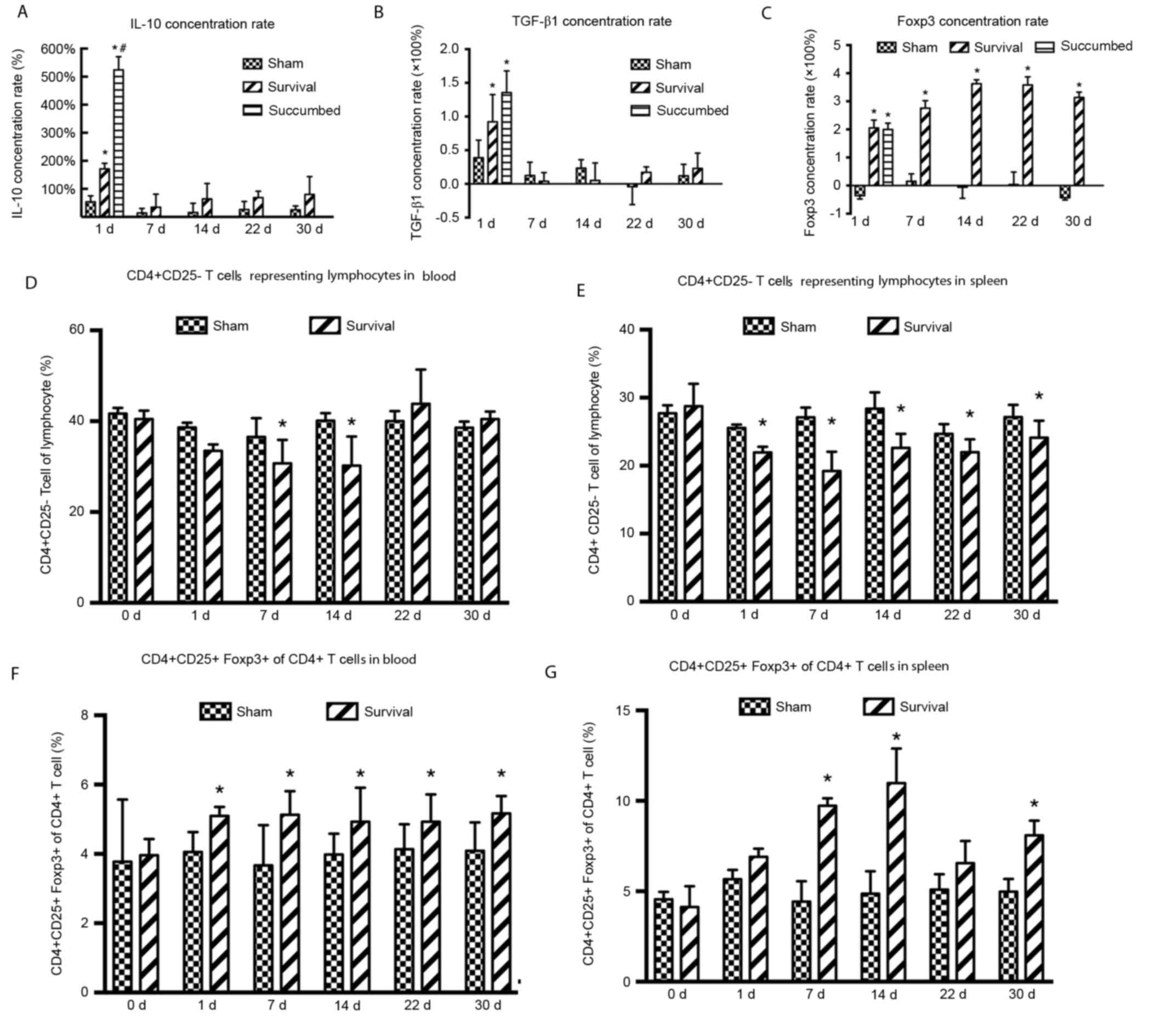
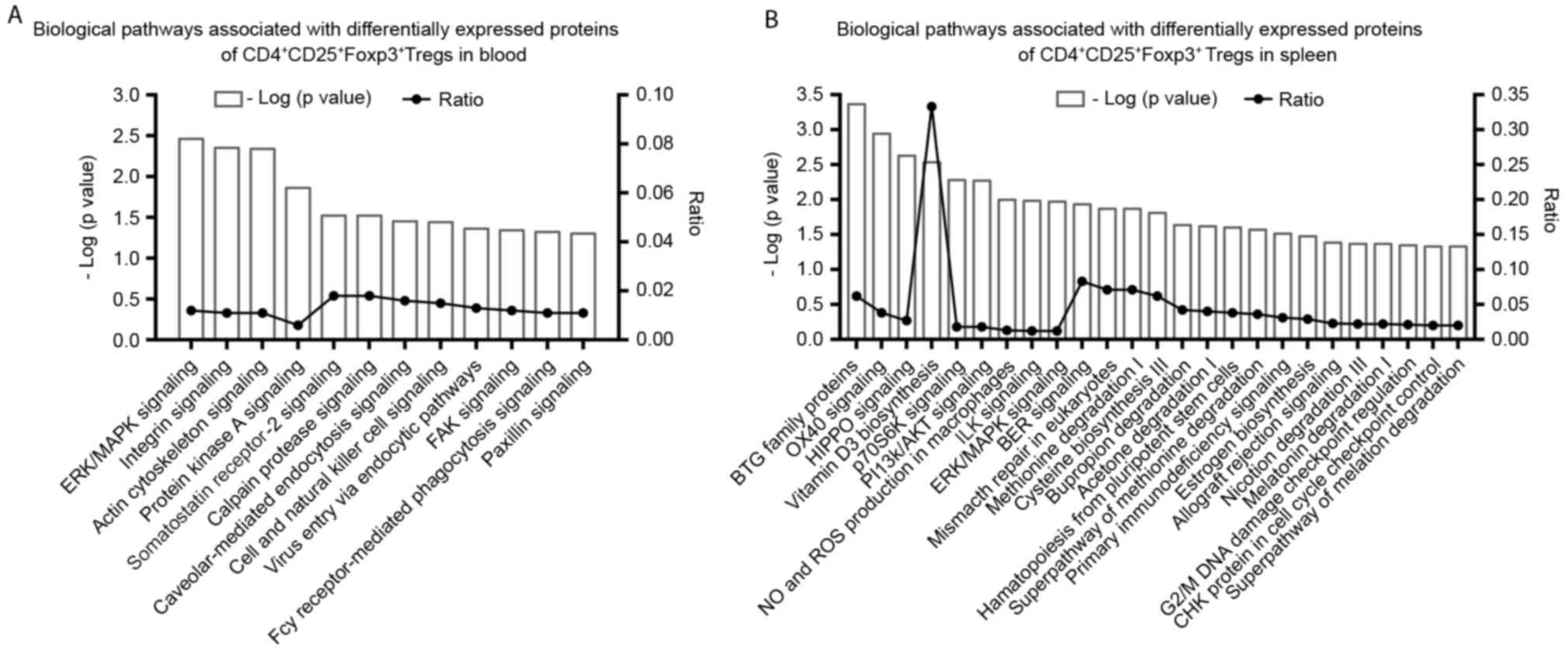
|
1
|
Singer M, Deutschman CS, Seymour CW,
Shankar-Hari M, Annane D, Bauer M, Bellomo R, Bernard GR, Chiche
JD, Coopersmith CM, et al: The third international consensus
definitions for sepsis and septic shock (Sepsis-3). JAMA.
315:801–810. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Stoller J, Halpin L, Weis M, Aplin B, Qu
W, Georgescu C and Nazzal M: Epidemiology of severe sepsis:
2008–2012. J Crit Care. 31:58–62. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Yadav H and Cartin-Ceba R: Balance between
Hyperinflammation and Immunosuppression in Sepsis. Semin Respir
Crit Care Med. 37:42–50. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hotchkiss RS, Monneret G and Payen D:
Sepsis-induced immunosuppression: From cellular dysfunctions to
immunotherapy. Nat Rev Immunol. 13:862–874. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hotchkiss RS, Monneret G and Payen D:
Immunosuppression in sepsis: A novel understanding of the disorder
and a new therapeutic approach. Lancet Infect Dis. 13:260–268.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Attridge K and Walker LS: Homeostasis and
function of regulatory T cells (Tregs). in vivo: Lessons from
TCR-transgenic Tregs. Immunol Rev. 259:23–39. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Guo J and Zhou X: Regulatory T cells turn
pathogenic. Cell Mol Immunol. 12:525–532. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Leavy O: Regulatory T Cells: Distinct role
in tissue repair. Nat Rev Immunol. 15:596–597. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cao C, Ma T, Chai YF and Shou ST: The role
of regulatory T cells in immune dysfunction during sepsis. World J
Emerg Med. 6:5–9. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Nascimento DC, Alves-Filho JC, Sônego F,
Fukada SY, Pereira MS, Benjamim C, Zamboni DS, Silva JS and Cunha
FQ: Role of regulatory T cells in long-term immune dysfunction
associated with severe sepsis. Crit Care Med. 38:1718–1725. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Rudiger A, Dyson A, Felsmann K, Carré JE,
Taylor V, Hughes S, Clatworthy I, Protti A, Pellerin D, Lemm J, et
al: Early functional and transcriptomic changes in the myocardium
predict outcome in a long-term rat model of sepsis. Clin Sci
(Lond). 124:391–401. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Brealey D, Karyampudi S, Jacques TS,
Novelli M, Stidwill R, Taylor V, Smolenski RT and Singer M:
Mitochondrial dysfunction in a long-term rodent model of sepsis and
organ failure. Am J Physiol Regul Integr Comp Physiol.
286:R491–R497. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lilley E, Armstrong R, Clark N, Gray P,
Hawkins P, Mason K, López-Salesansky N, Stark AK, Jackson SK,
Thiemermann C and Nandi M: Refinement of animal models of sepsis
and septic shock. Shock. 43:304–316. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Fink MP: Animal models of sepsis.
Virulence. 5:143–153. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
He X, Wei Z, Zhou E, Chen L, Kou J, Wang J
and Yang Z: Baicalein attenuates inflammatory responses by
suppressing TLR4 mediated NF-κB and MAPK signaling pathways in
LPS-induced mastitis in mice. Int Immunopharmacol. 28:470–476.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kim J, Yang HY and Jang YS: A G
protein-associated ERK pathway is involved in LPS-induced
proliferation and a PTK-associated p38 MAPK pathway is involved in
LPS-induced differentiation in resting B cells. Mol Immunol.
43:1232–1242. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gonzalo S, Grasa L, Arruebo MP, Plaza MÁ
and Murillo MD: Extracellular signal-regulated kinase (ERK) is
involved in LPS-induced disturbances in intestinal motility.
Neurogastroenterol Motil. 23:e80–e90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Asati V, Mahapatra DK and Bharti SK:
PI3K/Akt/mTOR and Ras/Raf/MEK/ERK signaling pathways inhibitors as
anticancer agents: Structural and pharmacological perspectives. Eur
J Med Chem. 109:314–341. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yamashita M, Shinnakasu R, Asou H, Kimura
M, Hasegawa A, Hashimoto K, Hatano N, Ogata M and Nakayama T:
Ras-ERK MAPK cascade regulates GATA3 stability and Th2
differentiation through ubiquitin-proteasome pathway. J Biol Chem.
280:29409–29419. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Knight T and Irving JA: Ras/Raf/MEK/ERK
pathway activation in childhood acute lymphoblastic leukemia and
its therapeutic targeting. Front Oncol. 4:1602014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Fischer TH, Gatling MN, McCormick F, Duffy
CM and White GC II: Incorporation of Rap 1b into the platelet
cytoskeleton is dependent on thrombin activation and extracellular
calcium. J Biol Chem. 269:17257–17261. 1994.PubMed/NCBI
|
|
22
|
Naci D and Aoudjit F: Alpha2beta1 integrin
promotes T cell survival and migration through the concomitant
activation of ERK/Mcl-1 and p38 MAPK pathways. Cell Signal.
26:2008–2015. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Gendron S, Couture J and Aoudjit F:
Integrin alpha2beta1 inhibits Fas-mediated apoptosis in T
lymphocytes by protein phosphatase 2A-dependent activation of the
MAPK/ERK pathway. J Biol Chem. 278:48633–48643. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Iwamoto DV and Calderwood DA: Regulation
of integrin-mediated adhesions. Curr Opin Cell Biol. 36:41–47.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Mattila PK, Batista FD and Treanor B:
Dynamics of the actin cytoskeleton mediates receptor cross talk: An
emerging concept in tuning receptor signaling. J Cell Biol.
212:267–280. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hirata H, Chiam KH, Lim CT and Sokabe M:
Actin flow and talin dynamics govern rigidity sensing in
actin-integrin linkage through talin extension. J R Soc Interface.
11:pii: 20140734. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Critchley DR and Gingras AR: Talin at a
glance. J Cell Sci. 121:1345–1347. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Priddle H, Hemmings L, Monkley S, Woods A,
Patel B, Sutton D, Dunn GA, Zicha D and Critchley DR: Disruption of
the talin gene compromises focal adhesion assembly in
undifferentiated but not differentiated embryonic stem cells. J
Cell Biol. 142:1121–1133. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhao P, Ma W, Hu Z, Zang L, Tian Z and
Zhang K: Filamin A (FLNA) modulates chemosensitivity to docetaxel
in triple-negative breast cancer through the MAPK/ERK pathway.
Tumour Biol. 37:5107–5115. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Shao QQ, Zhang TP, Zhao WJ, Liu ZW, You L,
Zhou L, Guo JC and Zhao YP: Filamin A: Insights into its Exact Role
in Cancers. Pathol Oncol Res. 22:245–252. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Truong T, Shams H and Mofrad MR:
Mechanisms of integrin and filamin binding and their interplay with
talin during early focal adhesion formation. Integr Biol (Camb).
7:1285–1296. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
De Franceschi N and Ivaska J: Integrin
bondage: Filamin takes control. Nat Struct Mol Biol. 22:355–357.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Modarres HP and Mofradt MR: Filamin: A
structural and functional biomolecule with important roles in cell
biology, signaling and mechanics. Mol Cell Biomech. 11:39–65.
2014.PubMed/NCBI
|
|
34
|
Winkler GS: The mammalian
anti-proliferative BTG/Tob protein family. J Cell Physiol.
222:66–72. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Berthet C, Guéhenneux F, Revol V, Samarut
C, Lukaszewicz A, Dehay C, Dumontet C, Magaud JP and Rouault JP:
Interaction of PRMT1 with BTG/TOB proteins in cell signalling:
Molecular analysis and functional aspects. Genes Cells. 7:29–39.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Liu C, Tao T, Xu B, Lu K, Zhang L, Jiang
L, Chen S, Liu D, Zhang X, Cao N and Chen M: BTG1 potentiates
apoptosis and suppresses proliferation in renal cell carcinoma by
interacting with PRMT1. Oncol Lett. 10:619–624. 2015.PubMed/NCBI
|
|
37
|
Voo KS, Foglietta M, Percivalle E, Chu F,
Nattamai D, Harline M, Lee ST, Bover L, Lin HY, Baladandayuthapani
V, et al: Selective targeting of Toll-like receptors and OX40
inhibit regulatory T-cell function in follicular lymphoma. Int J
Cancer. 135:2834–2846. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Vu MD, Xiao X, Gao W, Degauque N, Chen M,
Kroemer A, Killeen N, Ishii N and Li XC: OX40 costimulation turns
off Foxp3+ Tregs. Blood. 110:2501–2510. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Hawse WF, Gloor BE, Ayres CM, Kho K, Nuter
E and Baker BM: Peptide modulation of class I major
histocompatibility complex protein molecular flexibility and the
implications for immune recognition. J Biol Chem. 288:24372–24381.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Rudolph MG, Stanfield RL and Wilson IA:
How TCRs bind MHCs, peptides and coreceptors. Annu Rev Immunol.
24:419–466. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Liu B, Zheng Y, Yin F, Yu J, Silverman N
and Pan D: Toll receptor-mediated hippo signaling controls innate
immunity in drosophila. Cell. 164:406–419. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Ye S and Eisinger-Mathason TS: Targeting
the Hippo pathway: Clinical implications and therapeutics.
Pharmacol Res. 103:270–278. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Mo JS, Park HW and Guan KL: The Hippo
signaling pathway in stem cell biology and cancer. EMBO Rep.
15:642–656. 2014.PubMed/NCBI
|
|
44
|
Pan D: The hippo signaling pathway in
development and cancer. Dev Cell. 19:491–505. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Yu FX and Guan KL: The Hippo pathway:
Regulators and regulations. Genes Dev. 27:355–371. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Tumaneng K, Schlegelmilch K, Russell RC,
Yimlamai D, Basnet H, Mahadevan N, Fitamant J, Bardeesy N, Camargo
FD and Guan KL: YAP mediates crosstalk between the Hippo and
PI(3)K-TOR pathways by suppressing PTEN via miR-29. Nat Cell Biol.
14:1322–1329. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Liko D and Hall MN: mTOR in health and in
sickness. J Mol Med (Berl). 93:1061–1073. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Sandilands E, Schoenherr C and Frame MC:
p70S6K is regulated by focal adhesion kinase and is required for
Src-selective autophagy. Cell Signal. 27:1816–1823. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Tabe Y, Jin L, Konopleva M, Shikami M,
Kimura S, Andreeff M, Raffeld M and Miida T: Class IA PI3K
inhibition inhibits cell growth and proliferation in mantlecell
lymphoma. Acta Haematol. 131:59–69. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Vivanco I and Sawyers CL: The
phosphatidylinositol 3-kinase AKT pathway in human cancer. Nat Rev
Cancer. 2:489–501. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
51
|
Ribeiro PS, Josué F, Wepf A, Wehr MC,
Rinner O, Kelly G, Tapon N and Gstaiger M: Combined functional
genomic and proteomic approaches identify a PP2A complex as a
negative regulator of Hippo signaling. Mol Cell. 39:521–534. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Zeng Q, Zhang H, Qin J, Xu Z, Gui L, Liu
B, Liu C, Xu C, Liu W, Zhang S, et al: Rapamycin inhibits
BAFF-stimulated cell proliferation and survival by suppressing
mTOR-mediated PP2A-Erk1/2 signaling pathway in normal and
neoplastic B-lymphoid cells. Cell Mol Life Sci. 72:4867–4884. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Li Z, Tang J and Guo F: Identification of
14-3-3 proteins phosphopeptide-binding specificity using an
affinity-based computational approach. PLoS One.
11:e1474672016.
|
|
54
|
Bikle D: Nonclassic actions of vitamin D.
J Clin Endocrinol Metab. 94:26–34. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Verstuyf A, Carmeliet G, Bouillon R and
Mathieu C: Vitamin D: A pleiotropic hormone. Kidney Int.
78:140–145. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Baeke F, Takiishi T, Korf H, Gysemans C
and Mathieu C: Vitamin D: Modulator of the immune system. Curr Opin
Pharmacol. 10:482–496. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Cheng JB, Levine MA, Bell NH, Mangelsdorf
DJ and Russell DW: Genetic evidence that the human CYP2R1 enzyme is
a key vitamin D 25-hydroxylase. Proc Natl Acad Sci USA. 101:pp.
7711–7715. 2004, View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Zawaira A, Gallotta M, Beeton-Kempen N,
Coulson L, Marais P, Kuttel M and Blackburn J: Exhaustive
computational search of ionic-charge clusters that mediate
interactions between mammalian cytochrome P450 (CYP) and
P450-oxidoreductase (POR) proteins. Comput Biol Chem. 34:42–52.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Gocek E, Marchwicka A, Bujko K and
Marcinkowska E: NADPH-cytochrome P450 reductase is regulated by
all-trans retinoic acid and by 1,25-dihydroxyvitamin D3 in human
acute myeloid leukemia cells. PLoS One. 9:e917522014. View Article : Google Scholar : PubMed/NCBI
|