Introduction
Aging is associated with a progressive decline in a
number of physiopathological processes, leading to health
complication and disease, including cardiovascular diseases, cancer
and neurodegenerative diseases (1).
With the continuous increasing global aging population, aging has
already become a huge society burden. How to reverse aging has
become one of the biggest challenges in clinical and biological
research. In order to understand the basic mechanism of aging as
well as the physiopathological and behavioral effects of aging,
aging studies range from the molecular level to the global organism
level, from cell to the whole body, which are extensively
investigated worldwide (2,3). Animal aging models are important for
aging study, and several standard models have been established,
including fruit fly, fish, birds, mouse, rats and dogs (4,5).
Generally, aging models can be classified into two categories:
naturally aging model and accelerated aging models (6,7). Due to
practical condition of the research projects, such as duration and
budget, accelerated aging models are widely and commonly used
(6,8).
Among mammal aging models, beagle dogs have been
generally used. They have moderate life spans, varying from
approximately 12–15 years, and they are easy to handle and present
in abundance (9). Aged beagle dogs
are an excellent model in many respects, since dogs and human share
a number of chronic age-associated ailments, including mobility
decline, cardiac dysfunction, neurodegenerative disorders and other
chronic organ failure (10–12). Despite, as an aging model, the
duration of aging studies using beagle dogs is up to 6–8 years,
which is time consuming and leads to a huge expenses. Therefore,
establishment of accelerated aging models is important for
improving the efficiency of aging studies using beagle dogs.
Experimental-induced aging by chemical compounds, such as
D-galactose, has been used as accelerated aging models in mouse and
rats (13,14). D-galactose is a reducing sugar and an
important nutrient that react with amino acids to form glycation
end products through non-enzymatic glycation. D-galatcose also
contributes to generation of reactive oxygen species (ROS) via
metabolism of D-galactose (15). It
has been showed that D-galactose induced aging mice is observed
progressive declines in learning and memory ability, mobility and
cardiac dysfunction, which is similar to naturally aging mice
(16). However, there are very few
studies using D-galactose induced aging in beagle dogs.
In the present study, we induced aging in young
adult beagle dogs using D-galactose. This D-galactose induced aging
model was compared with naturally aging beagle dogs, and young
adult beagle dog was used as control. Our data revealed that
D-galactose induced aging group had an increased MDA level and
decreased SOD and GSH-Px level, which is similar to the naturally
aging group in comparison to young control group. Parallel
histopathological features were observed in both D-galactose
induced aging and naturally aging group compared with young control
group. Reduced expression level of proliferating cell nuclear
antigen (PCNA), and increased expression levels of P16 and P21 in
the hippocampus were observed in both naturally ageing group and
induced aging group compared with the young control group. Our
observations provides a strong piece of evidence supporting that
D-galactose induced aging model as a well-established model in mice
and rats, can also be applied in beagle dogs. In addition, our
comparison study extends the understanding of the etiology of
aging-related progression in different organs and also the
important implication in the investigations on age-related
diseases.
Materials and methods
Animals and treatment
Beagle dogs of both genders were provided by
Guangzhou General Pharmaceutical Research Institute, Ltd.
(Guangzhou, China) and housed in the Guangdong Medical University
Animal Center (Zhanjiang, China). Total 14 dogs were used in this
study, including 4 dogs at 10 years of age (2 male and 2 female) as
naturally aging group, 6 dogs at 2 years of age (3 male and 3
female) as induced aging group and 4 dogs at 2 years of age (2 male
and 2 female) as young control group. They were kept under
controlled conditions (room temperature, 22–24°C; relative
humidity, 40–60%) and accustomed to the housing environment for 1
week prior to the experiment with free access to food and distilled
water. The induced aging group was given D-galactose at 50 mg/kg
daily subcutaneous injections for 90 days. Blood samples from each
group were collected every four weeks. Animals were sacrificed on
the last day of treatment, and blood, sera and organs were
immediately collected for bioassay or stored at −70°C for later
use. All the experimental procedures were conducted following the
approval of the Animal Ethics Committee of Guangdong Medical
University.
MDA content, SOD activity and GSH-Px
activity measurement
serum sample and homogenates from brain tissue were
assayed for the determination of oxidative stress-related factors
including malondialdehyde (MDA) and superoxide dismutase (SOD) and
glutathione peroxidase (GSH-Px) by kits (Nanjing Jiancheng
Bioengineering Institute, Nanjing, China) using an automatic
chemical analyzer according to manufacturer's instructions.
Hematoxylin and eosin (H&E)
staining
A segment of tissue (liver, kidney, heart, lung and
spleen and brain) was transferred to the fixative (4% formaldehyde
and 1% glutaraldehyde in PBS, pH 7.4). The fixed segment of tissue
was embedded in paraffin, 5 µm thick sections were cut by Sliding
microtome Leica SM2010 R (Leica Store Shanghai, Shanghai, China).
Sections were dehydrated by gradient ethanol and stained with
H&E. Images were captured by microscope.
Nissl staining
Hippocampus tissue was removed from the brain and
fixed with 4% paraformaldehyde for 48 h. Sections (10-µm thick)
were cut on a cryostat (Leica Microsystems GmbH, Wetzlar, Germany).
The sections were hydrated in 1% toluidine blue at 50°C for 20 min
for Nissl staining. Then they were dehydrated and mounted with
Permount (Thermo Fisher Scientific, Inc., Waltham, MA, USA) after
rinsing with distilled water. The slide was observed under a
microscope.
Immunohistochemical staining
Immunostaining of iNOS, COX-2 and NF-κB was carried
out on the tissue from liver, kidney, heart, lung and spleen and
brain with PBS containing 0.05 M EDTA followed by 4%
paraformaldehyde. Then 5-µm sections were incubated with blocking
reagent followed by primary antibody anti-iNOS or anti-COX-2 or
anti-NF-κB (17) in the presence of
10% rabbit serum overnight at 4°C, followed by biotin-conjugated
goat anti-rabbit Ig, avidin-linked HRP complex and
3,3′-diaminobenzidine as substrate. Slides were counterstained with
hematoxylin, dehydrated, and examined under a light microscope. NIH
ImageJ was used for macrophage and neutrophil quantification.
Western blot analysis
Tissue samples from the brain were harvested and
stored at −80°C. For western blot analysis, frozen samples were
sonicated on ice twice for 5 sec in 50 mM lysis buffer (pH 7.4)
containing 3.1 mM sucrose, 1 mM DTT, 10 µg/ml leupeptin, 10 µg/ml
soybean trypsin inhibitor, 2 µg/ml aprotinin, and 0.1% Triton
X-100. Homogenates were centrifuged at 10,000 × g at 4°C for 20 min
and the supernatant was collected. The total protein concentration
was measured using the Bradford protein assay (Bio-Rad
Laboratories, Inc., Hercules, CA, USA). Protein lysates (20 µg) was
separated using 12% SDS-PAGE and transferred to a PVDF membrane.
After blocking with 5% nonfat milk, the PVDF membrane was incubated
overnight with the primary antibody as follows: rabbit anti-PCNA,
rabbit anti-p21 and rabbit anti-p16 solute in TBS-T. Membranes were
washed in TBS-T (10 min ×3) and then probed with the appropriate
secondary antibody (1:10,000; Abcam, Cambridge, UK). Membranes were
developed using Versa Doc 5000 and band densities were measured
with Quantity One 4.6 software (Bio-Rad Laboratories, Inc.). Equal
protein loading was additionally verified by measurement of GAPDH
level with rabbit polyclonal antibody.
Gene expression analysis using
qPCR
The total RNA from tissue samples was extracted by
using TRIzol kit (Invitrogen; Thermo Fisher Scientific, Inc.)
according to the manufacture instruction. A total of 1 µg RNA was
reverse transcribed into complementary (c)DNA using oligo (dT) and
III reverse transcriptase is a version of M-MLV RT. Real-time PCR
was carried out with an ABI 7500 Real-Time PCR system. Target cDNA
levels were determined by SYBR Green-based real-time PCR in 20 µl
reactions containing 10 µl Power SYBR Green Master Mix (Applied
Biosystems; Thermo Fisher Scientific, Inc.). PCNA forward,
5′-TGTCGATAAGGAGGAGGAAGC-3′ and reverse,
5′-CTGTAGGAGAGAGCGGAGTGG-3′; p16 forward, 5′-ACCCCGTCACCCTCACC-3′
and reverse, 5′-GCTCCTCAGCCAGGTCCAC-3′; p21 forward,
5′-GGGGACAGCTCAGGAGGA-3′ and reverse, 5′-CGGCGTTTGGAGTGATAGA-3′ and
GAPDH forward, 5′-GGTCGGAGTGAACGGATTT-3′ and reverse,
5′-CATTTGATGTTGGCGGGA-3′. The general PCR cycle was performed as
follows: 2 min at 94°C, 29 to 32 cycles of 95°C for 30 sec, 57–60°C
for 45 sec, 72°C for 1 min, 72°C for 5 min and hold at 4°C. The
qPCR data were analyzed with a 2−ΔΔCt method and
normalized using GAPDH as an internal control.
Statistical analysis
Statistical calculations were performed using Prism
6 (GraphPad Software, Inc., La Jolla, CA, USA). Data are presented
as the mean ± SEM. Student's t-tests were used for comparisons
between 2 groups, and one-way or two-way ANOVA was used for
comparisons among multiple groups. P<0.05 or P<0.01 were
considered to indicate a statistically significant result.
Results
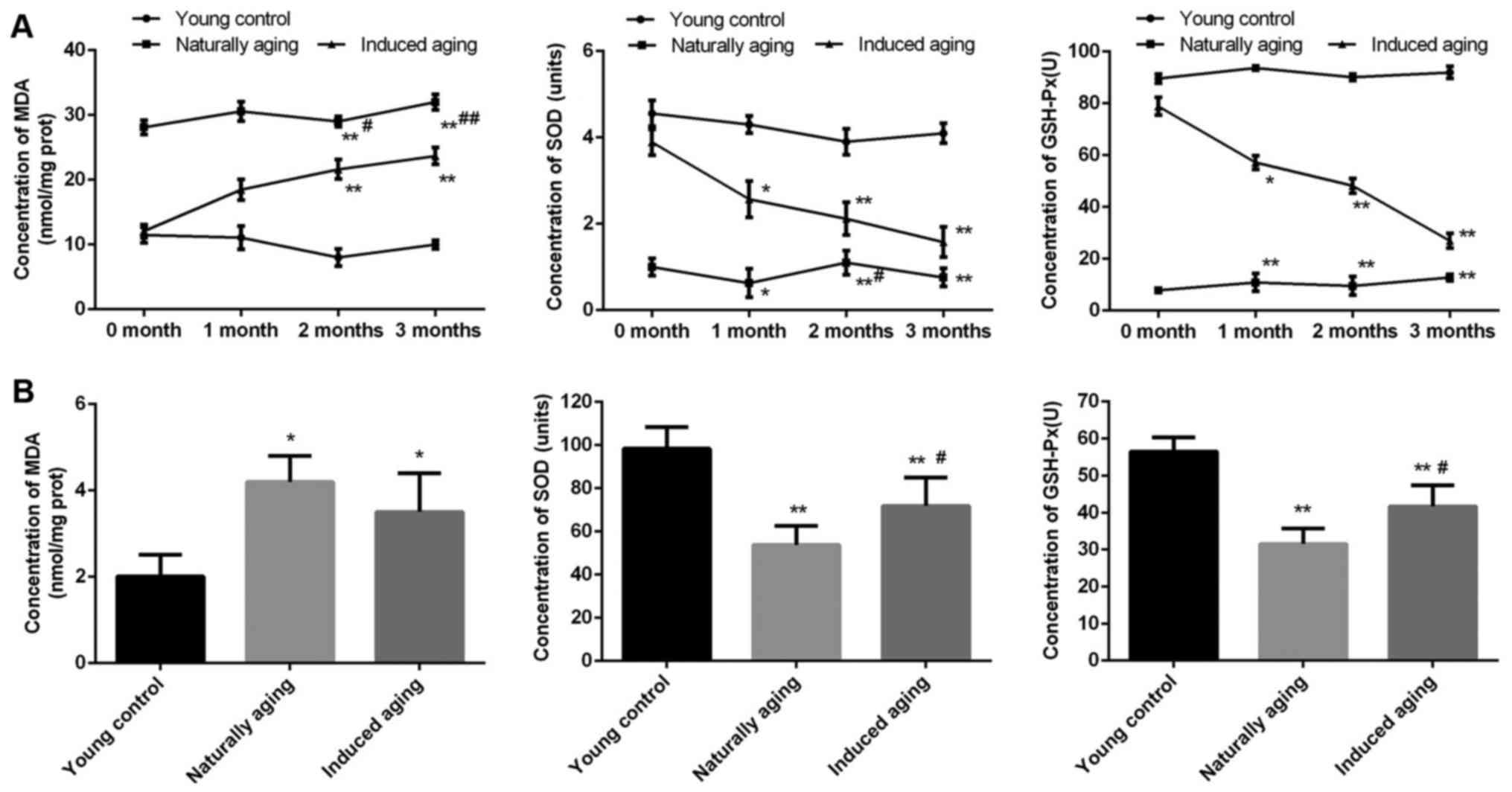
Comparison of oxidative stress
markers
In order to evaluate the level of oxidative stress
in the induced aging group and naturally aging group, MDA content,
SOD activity and GSH-Px activity as oxidative stress markers were
determined. In the induced aging group, MDA level was progressively
increased from about 60% after one month D-galactose treatment to
almost 120% after three months treatment in comparison to young
control group. While SOD activity and GSH-Px activity were
gradually decreased by approximately 100 and 75% after three months
treatment compared with young control group, representatively. In
the naturally aging group, steady high level of MDA and steady low
level of SOD activity and GSH-Px activity were observed, compared
with young control group (Fig. 1A).
Similar results were obtained from the brain tissue sample.
Significantly increased MDA level was detected, along with
remarkably reduced SOD activity and GSH-Px activity in comparison
to young control group (Fig.
1B).
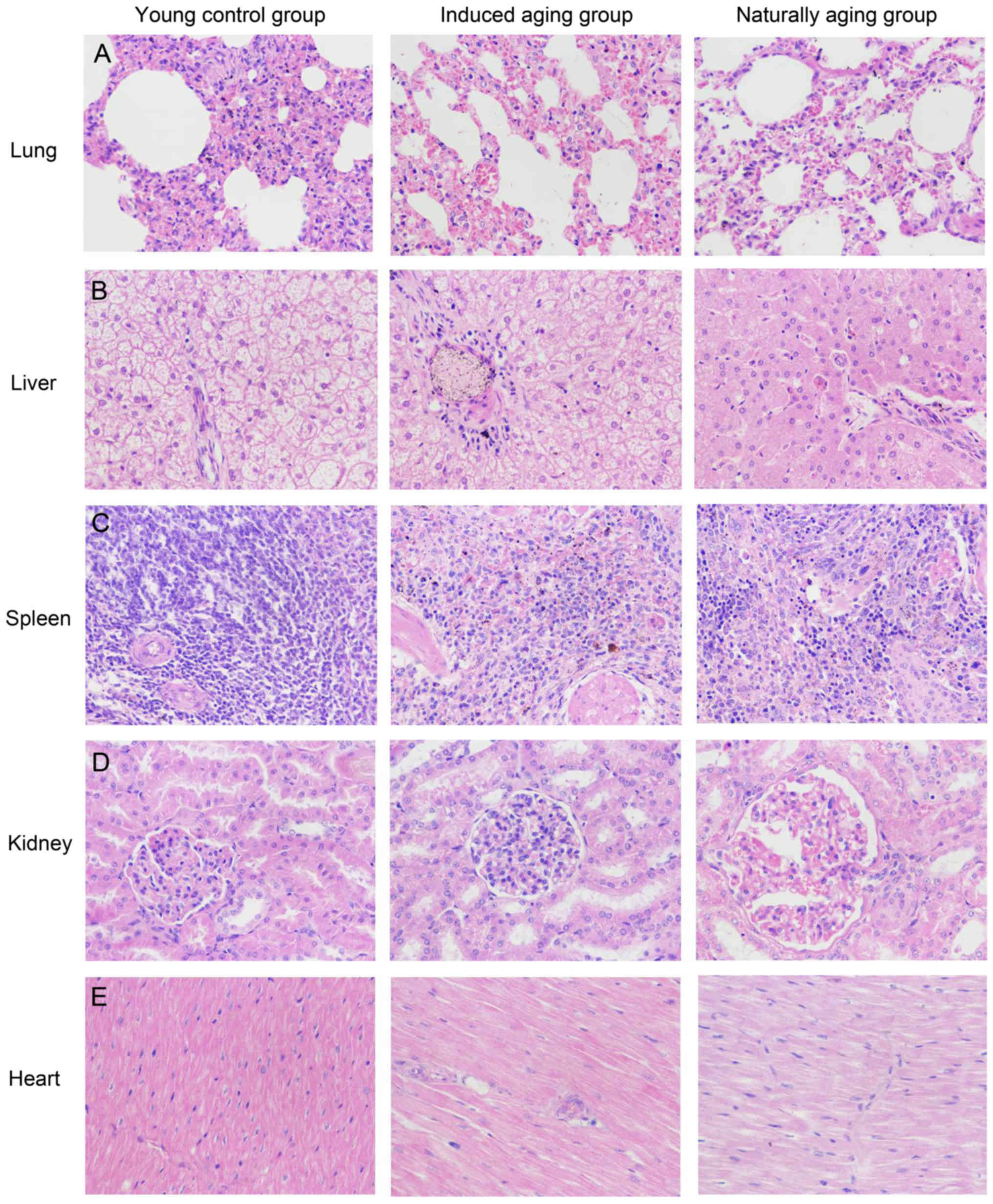
Histologic expressions and
histopathological features in the induced aging model
To determine histologic expressions and
histopathological features in the induced aging model, H&E
staining was used for tissue morphology in liver, kidney, heart,
lung and spleen and brain tissue, and nissl staining was used for
morphology and pathology of neural tissue, and immunohistochemical
staining was used to evaluate the expression of PCNA, p16 and p21.
H&E staining and nissl staining revealed that induced aging
group and naturally aging group have parallel histopathological
features in liver, kidney, heart, lung and spleen and brain: i) in
H&E stained section from lung tissue, lung injury such as
inflammatory cell infiltration and alveolar wall destruction was
observed in both induced aging group and naturally aging group,
compared to the normal lung architecture in young control group
(Fig. 2A); ii) in H&E stained
section from liver tissue, microabscess involving a few hepatocytes
with inflammatory cells and necrotic debris was accumulated in the
liver tissue in induced aging group and naturally aging group
(Fig. 2B); iii) in H&E stained
section from spleen tissue, we observed high degree of morphologic
organization in the young control group, while density of
inflammatory cells was diminished in both induced aging group and
naturally aging group (Fig. 2C); iv)
in H&E stained section from kidney tissue, young control group
exhibited normal histology, and induced aging and naturally aging
group revealed enlarged vascular glomeruli, widening the glomerular
capsular space and destructed flat epithelium lining of Bowman's
capsule (Fig. 2D); v) in H&E
stained section from heart tissue, disordered arrangement and
enlarged size of cardiomyocytes was observed, indicating a cardiac
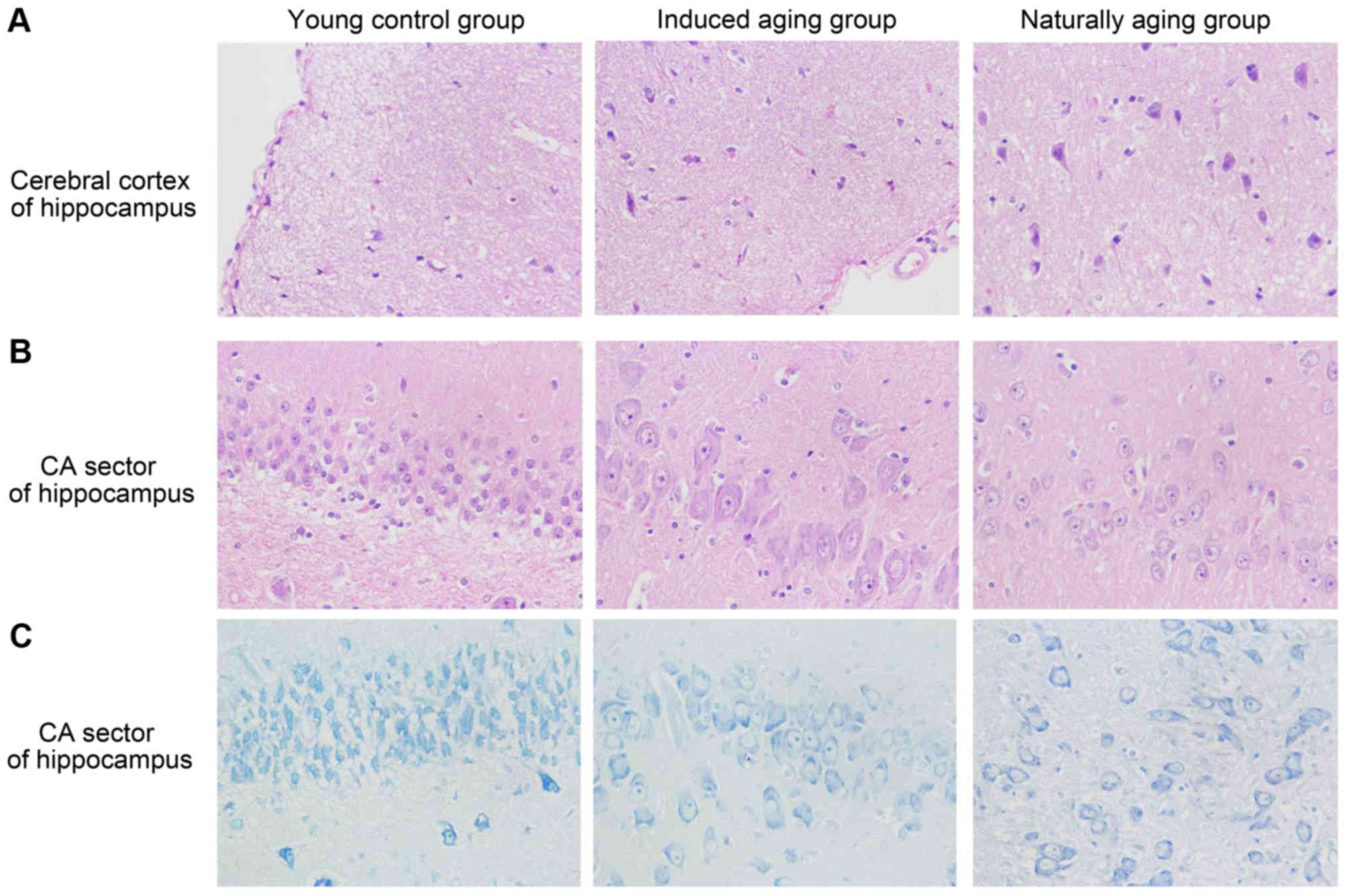
remodeling event in both aging groups (Fig. 2E); and vi) in sections from brain
tissue, H&E stained cerebral cortex sections from both aging
groups revealed high level of inflammatory cell in comparison to
young control group, which indicate that inflammation was
progressing in the cerebral cortex. In H&E stained section of
hippocampus AC area, young control group showed well organized
pyramidal cells, and different types of the neuralgia cells are
scattered inside the neuropil matrix, while both aging groups
showed apparent decrease in the number of pyramidal cells with
presence of some shrunken degenerated cells. Similar results were
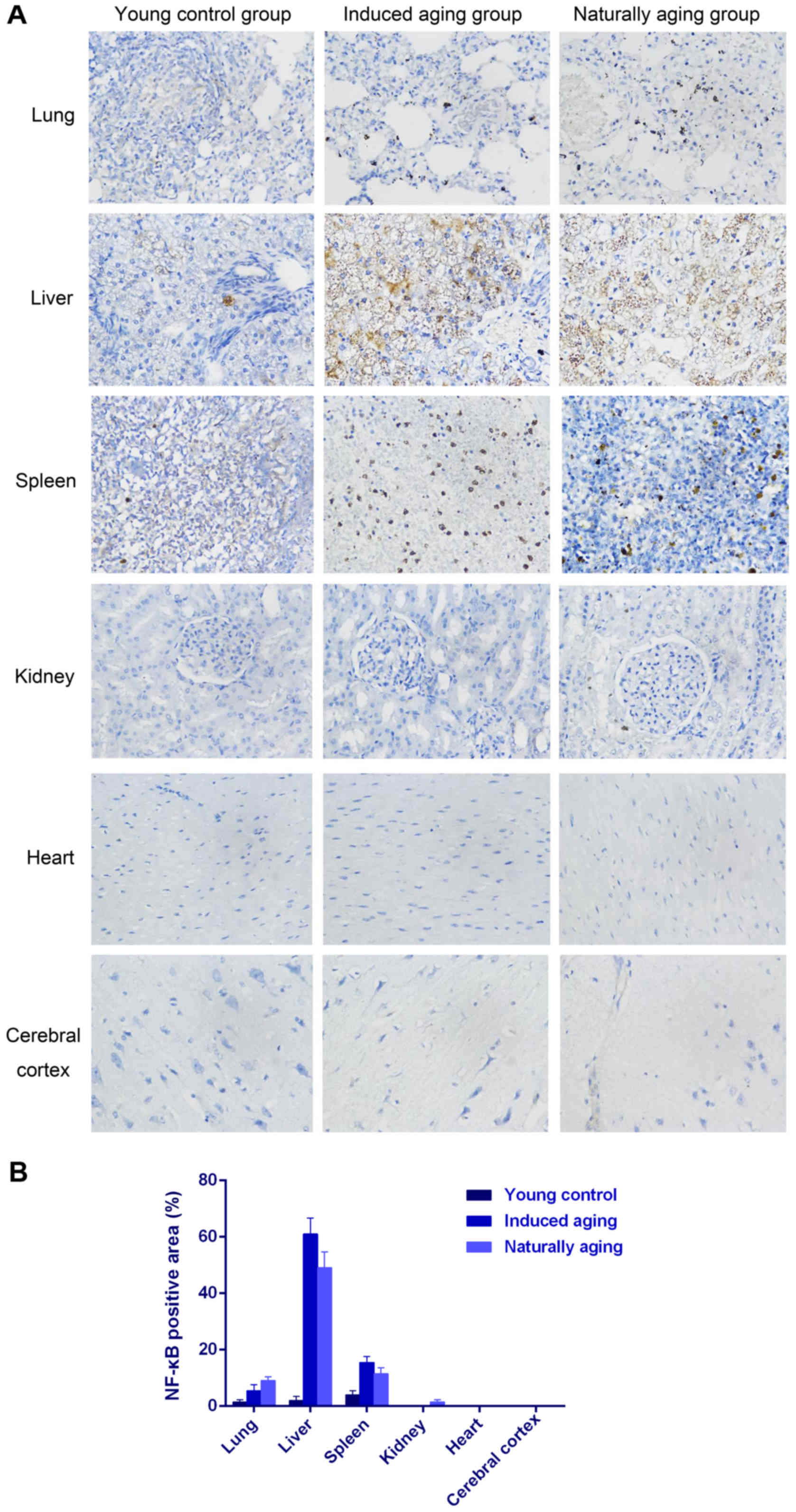
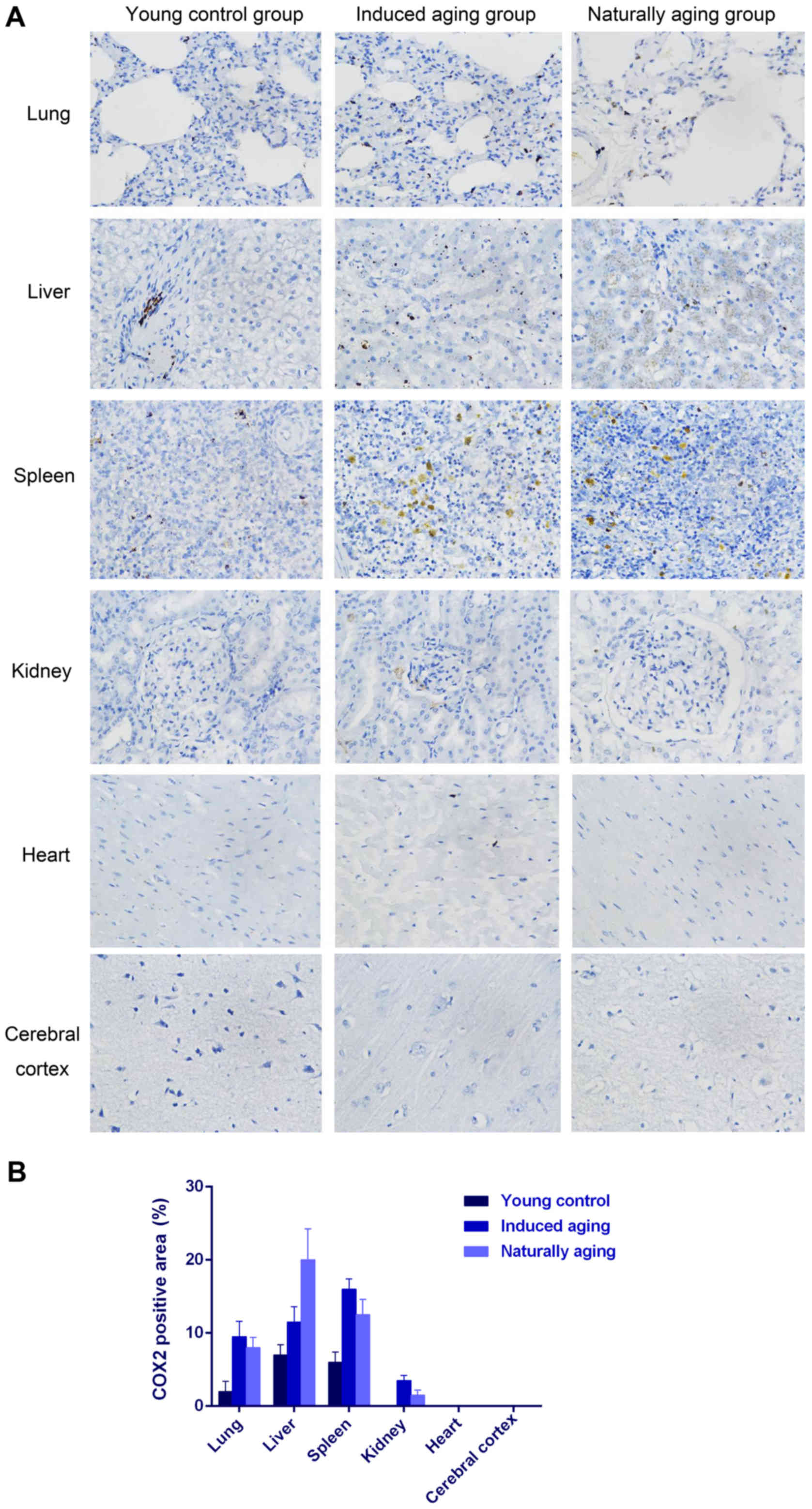
also observed in nissl staining sections (Fig. 3). Immunohistochemical staining
indicated increased levels of NF-Κb, iNOS and COX2 in lung, liver
and spleen of both aging groups in comparison to young control
group (Figs. 4–6). P<0.05, significantly different from
the control groups.
Comparison of the expression of
age-associated factors
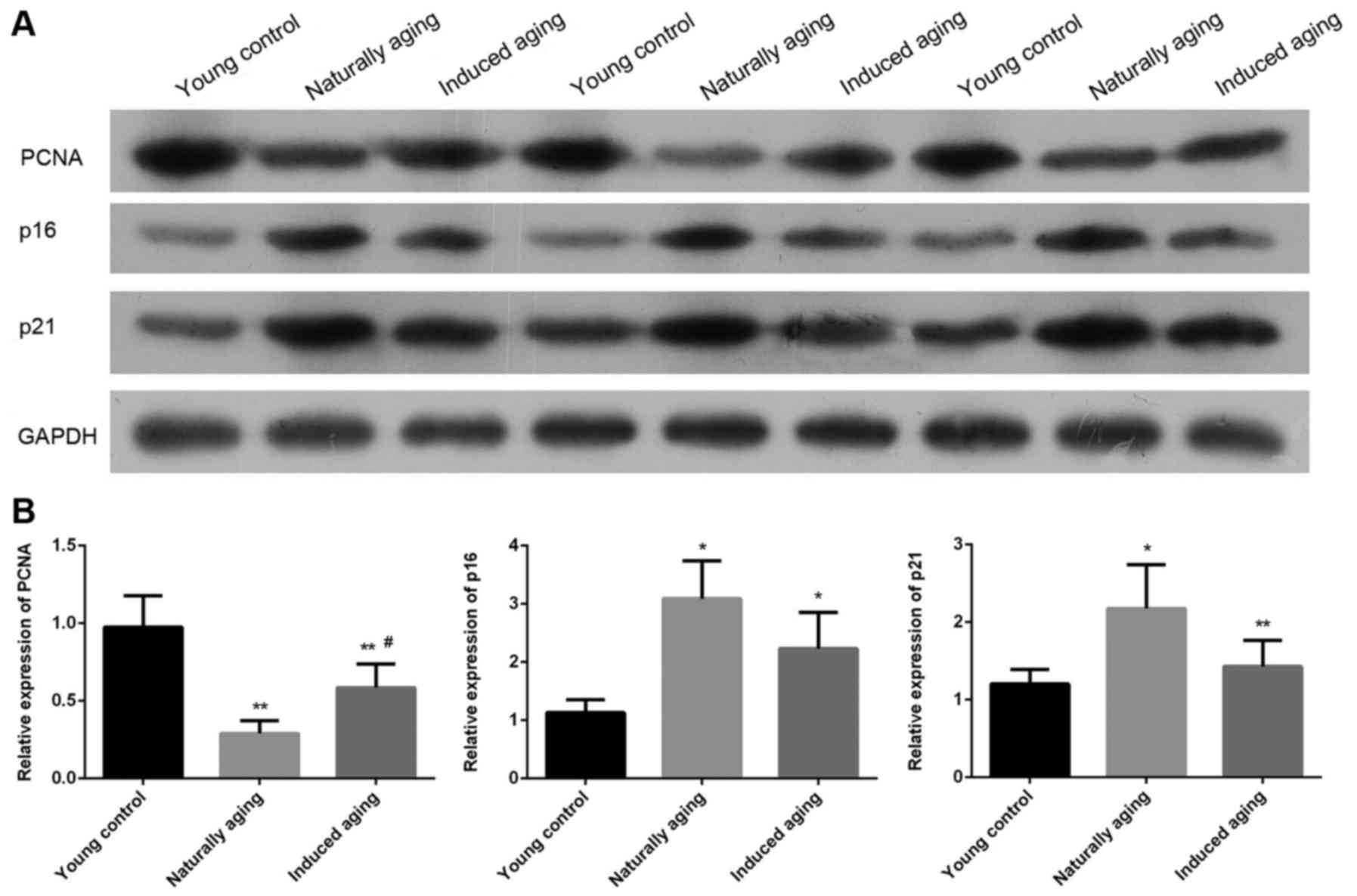
The expression level of PCNA, p16 and p21 were
determined by western blot and RT-qPCR. Western blot analysis
indicated that the expression level of PCNA was significantly
reduced in induced aging group and naturally aging group compared
with the young control group. In contrast, lower levels of p16 and
p21 were detected in both aging groups in comparison to young
control group. Similar results were also obtained by qPCR (Fig. 7). P<0.05, significantly different
from the control groups.
Discussion
With the growing aging population, the burden of
aging and aging-associated diseases will continue to be huge
medical and financial problems for the society. Aging-associated
pathological conditions are critical risk factors for a variety of
diseases, including cancers, neurodegenerative diseases, chronic
organs failure, metabolic diseases and Loss of mobility (18,19).
Determining the fundamental underlying cellular and molecular
mechanisms of aging progression is crucial for the development of
anti-aging strategies if we are to meet the increasing healthcare
needs of the growing aging population. Aging models are commonly
used in aging studies, since the duration of the study is a major
difficulty in human aging. In order to establish a robust induced
aging model in beagle dogs, in the present study, D-galactose was
used to induce aging in beagle dogs as a induced aging model, and
we compared histologic expressions, histopathological features and
age-related factors expression with naturally aging beagle dogs
model. This is the first study on comparison of naturally aging and
D-galactose induced aging in beagles. Our results showed that
induced aging model and naturally aging model exhibited parallel
morphology and histology as well as similar level of oxidative
stress related factors (MDA, SOD, GSH-Px and iNOS), age-related
factors (PCNA, p16, p21 and COX2), providing a piece of evidence to
support that D-galactose induced aging beagle dogs can be used for
aging research in order to shorten the duration of study.
Animal aging models have been at the forefront of
aging research, and obtained a wealth of information, supporting
investigators to discover pathways that drive human aging (4). Successful animal aging model include
mice, rats, zebrafish and dogs (5).
In the present study, we used beagle dogs as model animal. Dogs as
a human-company pet are ideal model for aging study, since they
share the same living environment and similar food recourse. The
aged dogs naturally develop age-related decline in a variety of
tissue and organs and exhibit human-like individual variability in
aging progression, such as muscular and neurological decline, as
well as cardiovascular disease (20–23).
Rodent, howerver, do not develop neurodegeneration with age
(24). Therefore, dogs might be
particularly interesting in the study of neurodegenerative diseases
in aging (25). On the other hand,
though dogs have human-like aging phenotype that makes them an
attractive model, the average 10 years of life span is still
problematic. Therefore, accelerated aging model or induced aging
models in dogs are necessary to develop for aging studies.
Accelerated aging model has been well established in mice and rats
(6), and these models can be
experimental induced by compounds such as dihydrotachysterol
(26), O3 (27) and D-galactose (28). D-galactose contributes to generation
of reactive oxygen species (ROS) via metabolism of D-galactose.
Murata et al have reported that degeneration in the retinal
capillaries of galactose-fed beagle dogs results from apoptosis
(29). However, there has not been a
study investigating D-galactose induced accelerated aging in beagle
dogs. Therefore, we treated beagle dogs with D-galactose to develop
an induced accelerated aging model.
Aging-related decline or aging-associated
pathological conditions contain many aspects including oxidative
stress damage, chronic low-level inflammation, destructed cells and
tissue lining and dysfunction of organs. Studies have showed that a
significant increased level of MDA and decreased level of SOD
activity and GSH-Px activity in D-galactose induced aging mice
(30) and D-galactose induced aging
rats (31). Our data revealed
similar results in D-galactose induced aging dogs, compared with
young control and naturally aging dogs, suggesting that aging dogs
have more oxidative stress and free radical but less anti-oxidant
and enzymes to handle these stress resulting in serious damage in
tissue and organs. Furthermore, decreased level of PCNA and
elevated level of p16 and p21 has been considered as a standard
event in aging (32). Results from
the present study confirmed that D-galactose induced aging dogs has
a similar aging-related factors expression profile to naturally
aging dogs, including reduced level of PCNA, and high level of
NF-κB, iNOS, COX2, p16 and p21. These aging-related markers are
driving a number of apoptosis, necrosis, tissue remodeling and
inflammatory response signaling pathways that consisting of the
complex senescence processes leading to aging.
To the best of our knowledge, our study showed for
the first time D-galactose induced aging beagle dogs used as an
aging model. These results provides a strong piece of evidence
supporting that D-galactose induced aging model as a
well-established model in mice and rats, can also be applied in
beagle dogs. This accelerated induced aging model might be
promising tool for human aging in the future. In addition, our
comparison study extends the understanding of the etiology of
aging-related progression in different organs and also the
important implication in the investigations on age-related
diseases.
Acknowledgements
This study was funded by Provincial Science and
Technology Project of Guangdong Province (no. 2015A030302076,
2016A030303056 and 2016A020215224) and the Natural Science
Foundation Project of Guangdong Province (no. 2015A030310046 and
2016A030313674).).
References
|
1
|
Dillin A, Gottschling DE and Nyström T:
The good and the bad of being connected: The integrons of aging.
Curr Opin Cell Biol. 26:107–112. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Niedernhofer LJ, Kirkland JL and Ladiges
W: Molecular pathology endpoints useful for aging studies. Ageing
Res Rev. 35:241–249. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Marchal J, Pifferi F and Aujard F:
Resveratrol in mammals: Effects on aging biomarkers, age-related
diseases, and life span. Ann NY Acad Sci. 1290:67–73. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mitchell SJ, Scheibye-Knudsen M, Longo DL
and de Cabo R: Animal models of aging research: Implications for
human aging and age-related diseases. Annu Rev Anim Biosci.
3:283–303. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lees H, Walters H and Cox LS: Animal and
human models to understand ageing. Maturitas. 93:18–27. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Harkema L, Youssef SA and de Bruin A:
Pathology of mouse models of accelerated aging. Vet Pathol.
53:366–389. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Liao CY and Kennedy BK: Mouse models and
aging: Longevity and progeria. Curr Top Dev Biol. 109:249–285.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gurkar AU and Niedernhofer LJ: Comparison
of mice with accelerated aging caused by distinct mechanisms. Exp
Gerontol. 68:43–50. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Head E: A canine model of human aging and
Alzheimer's disease. Biochim Biophys Acta. 1832:1384–1389. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bosch MN, Pugliese M, Gimeno-Bayón J,
Rodríguez MJ and Mahy N: Dogs with cognitive dysfunction syndrome:
A natural model of Alzheimer's disease. Curr Alzheimer Res.
9:298–314. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Araujo JA, Nobrega JN, Raymond R and
Milgram NW: Aged dogs demonstrate both increased sensitivity to
scopolamine impairment and decreased muscarinic receptor density.
Pharmacol Biochem Behav. 98:203–209. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Vasilevko V and Head E: Immunotherapy in a
natural model of Abeta pathogenesis: The aging beagle. CNS Neurol
Disord Drug Targets. 8:98–113. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Shahroudi MJ, Mehri S and Hosseinzadeh H:
Anti-aging effect of nigella sativa fixed oil on
D-galactose-induced aging in mice. J Pharmacopuncture. 20:29–35.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liu C, Hu J, Mao Z, Kang H, Liu H, Fu W,
Lv Y and Zhou F: Acute kidney injury and inflammatory response of
sepsis following cecal ligation and puncture in d-galactose-induced
aging rats. Clin Interv Aging. 12:593–602. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Parameshwaran K, Irwin MH, Steliou K and
Pinkert CA: D-galactose effectiveness in modeling aging and
therapeutic antioxidant treatment in mice. Rejuvenation Res.
13:729–735. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ho SC, Liu JH and Wu RY: Establishment of
the mimetic aging effect in mice caused by D-galactose.
Biogerontology. 4:15–18. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Soria-Valles C, Osorio FG,
Gutiérrez-Fernández A, De Los Angeles A, Bueno C, Menéndez P,
Martín-Subero JI, Daley GQ, Freije JM and López-Otín C: NF-kB
activation impairs somatic cell reprogramming in ageing. Nat Cell
Biol. 17:1004–1013. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
Harada CN, Natelson Love MC and Triebel
KL: Normal cognitive aging. Clin Geriatr Med. 29:737–752. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Maes C, Gooijers J, de Xivry Orban JJ,
Swinnen SP and Boisgontier MP: Two hands, one brain, and aging.
Neurosci Biobehav Rev. 75:234–256. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Fast R, Schütt T, Toft N, Møller A and
Berendt M: An observational study with long-term follow-up of
canine cognitive dysfunction: Clinical characteristics, survival,
and risk factors. J Vet Intern Med. 27:822–829. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Freeman LM: Cachexia and sarcopenia:
Emerging syndromes of importance in dogs and cats. J Vet Intern
Med. 26:3–17. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kim SA, Lee KH, Won HY, Park S, Chung JH,
Jang Y and Ha JW: Quantitative assessment of aortic elasticity with
aging using velocity-vector imaging and its histologic correlation.
Arterioscler Thromb Vasc Biol. 33:1306–1312. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kraus C, Pavard S and Promislow DE: The
size-life span trade-off decomposed: Why large dogs die young. Am
Nat. 181:492–505. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
24
|
Jucker M: The benefits and limitations of
animal models for translational research in neurodegenerative
diseases. Nat Med. 16:1210–1214. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
25
|
Borras D, Ferrer I and Pumarola M:
Age-related changes in the brain of the dog. Vet Pathol.
36:202–211. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Schriefer JA and Spratto GR: Examination
of dihydrotachysterol-induced progeria as a model for aging changes
in carbohydrate metabolism. J Pharmacol Methods. 3:297–304. 1980.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Xing H, Hu X, Liu H, Li Y and Chen Y:
Study on DNA oxidative damage of O3 aging model in mice. Hua Xi Yi
Ke Da Xue Xue Bao. 32:229–231. 2001.(In Chinese). PubMed/NCBI
|
|
28
|
Budni J, Garcez ML, Mina F,
Bellettini-Santos T, da Silva S, Luz APD, Schiavo GL, Batista-Silva
H, Scaini G, Streck EL and Quevedo J: The oral administration of
D-galactose induces abnormalities within the mitochondrial
respiratory chain in the brain of rats. Metab Brain Dis.
32:811–817. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Murata M, Ohta N, Fujisawa S, Tsai JY,
Sato S, Akagi Y, Takahashi Y, Neuenschwander H and Kador PF:
Selective pericyte degeneration in the retinal capillaries of
galactose-fed dogs results from apoptosis linked to aldose
reductase-catalyzed galactitol accumulation. J Diabetes
Complications. 16:363–370. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Calvert GD and Scott PJ: Properties of two
pig low density lipoproteins prepared by zonal ultracentrifugation.
Atherosclerosis. 22:583–599. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Li M, Ouyang W, Wu X, Zheng Y, Wei Y and
An L: Kinetin inhibits apoptosis of aging spleen cells induced by
D-galactose in rats. J Vet Sci. 15:353–359. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Stein GH, Drullinger LF, Soulard A and
Dulić V: Differential roles for cyclin-dependent kinase inhibitors
p21 and p16 in the mechanisms of senescence and differentiation in
human fibroblasts. Mol Cell Biol. 19:2109–2117. 1999. View Article : Google Scholar : PubMed/NCBI
|