Introduction
Alcohol abuse is a worldwide problem, and about 3.2%
of deaths each year in the world are associated with alcohol,
causing great medical burdens. Alcoholic liver disease (ALD) is the
most important clinical manifestation of alcohol abuse, of which
the spectrum includes alcoholic fatty liver, alcoholic hepatitis
and alcoholic cirrhosis (1).
Alcoholic fatty liver is present in almost all the heavy drinkers,
with the pathological changes reversible, and the patients can
recover after the alcohol temperance of a few weeks (2). 15–20% of patients with alcoholic fatty
liver disease will develop into alcoholic hepatitis, and the course
is usually progressive; even if complete temperance is adopted,
only 27% patients show pathological self-healing, and about 20–50%
alcoholic hepatitis patients will be further developed into
cirrhosis (3).
After the activated STAT enters into the nucleus, it
activates the expressions of various genes including the abnormal
genes, which can effectively reduce liver inflammation and cell
damage in alcohol liver (4). While
after the knockout of STAT3 gene, liver regeneration capacity is
weakened, and insulin resistance is increased (5). Interestingly, the selective knockdown
of STAT3 in murine hepatocyte may aggravate the degree of ALD,
whereas the selective knockdown of STAT3 from endothelial cells in
mice can significantly reduce endothelial and hepatic damage
(6). The studies in the effects of
STAT3 on different cells show that STAT3 plays a role of
proinflammatory cytokine in hepatocytes, which may be related to
the release of various cytokines and regulatory proteins in
stimulated hepatocytes; STAT3 in macrophages and neutrophils from
bone marrow has the anti-inflammatory effect, and plays a leading
role in ALD (5,7).
Akt gene is in the core of PI3K/Akt signaling
pathway, and Akt is involved in cell growth and proliferation. It
is both a signal of cell survival and an essential factor for cell
survival. The continuous existence of Akt can avoid cell damage
(8). Akt signal is activated under
the stimulation of hypoxia or cytokine (9). The main targets of Akt signal are the
apoptotic family members, Bax/Bcl-2 and caspases. In addition, Akt
can also regulate intracellular glucose metabolism, in order to
make the cells adapted to environmental changes, and enhance cell
viability (9).
The pathogenic factor of ALD is long-term
large-quantity alcohol intake. But its pathogenesis is not entirely
clear (10). It has been confirmed
that long-term alcohol intake can activate NF-κB in liver tissue,
and the activated NF-κB enters into the nucleus, bond to the κB
sequence in promoter regions of various inflammatory factors, to
promote the transcriptions of inflammatory cytokines (TNF-1, IL-2,
IL-6, etc.), adhesion molecules and NO synthase, leading to further
inflammation, necrosis, apoptosis and fibrosis of hepatocytes
(11). In addition, the metabolism
of alcohol in the liver results in a large number of free radicals,
which inhibits fatty acid oxidation, accumulates intrahepatic fat
when beyond the body's clearing ability, and also leads to lipid
peroxidation of liver cell membrane, resulting in liver cell damage
(12). At the same time, these
reactive oxygen species will increase the level of endotoxin, and
lead to the release of inflammatory mediators, which can also make
NF-κB activated, thus generating a large number of inflammatory
mediators, to have an inflammatory cascade amplification effect,
aggravating liver damage (7).
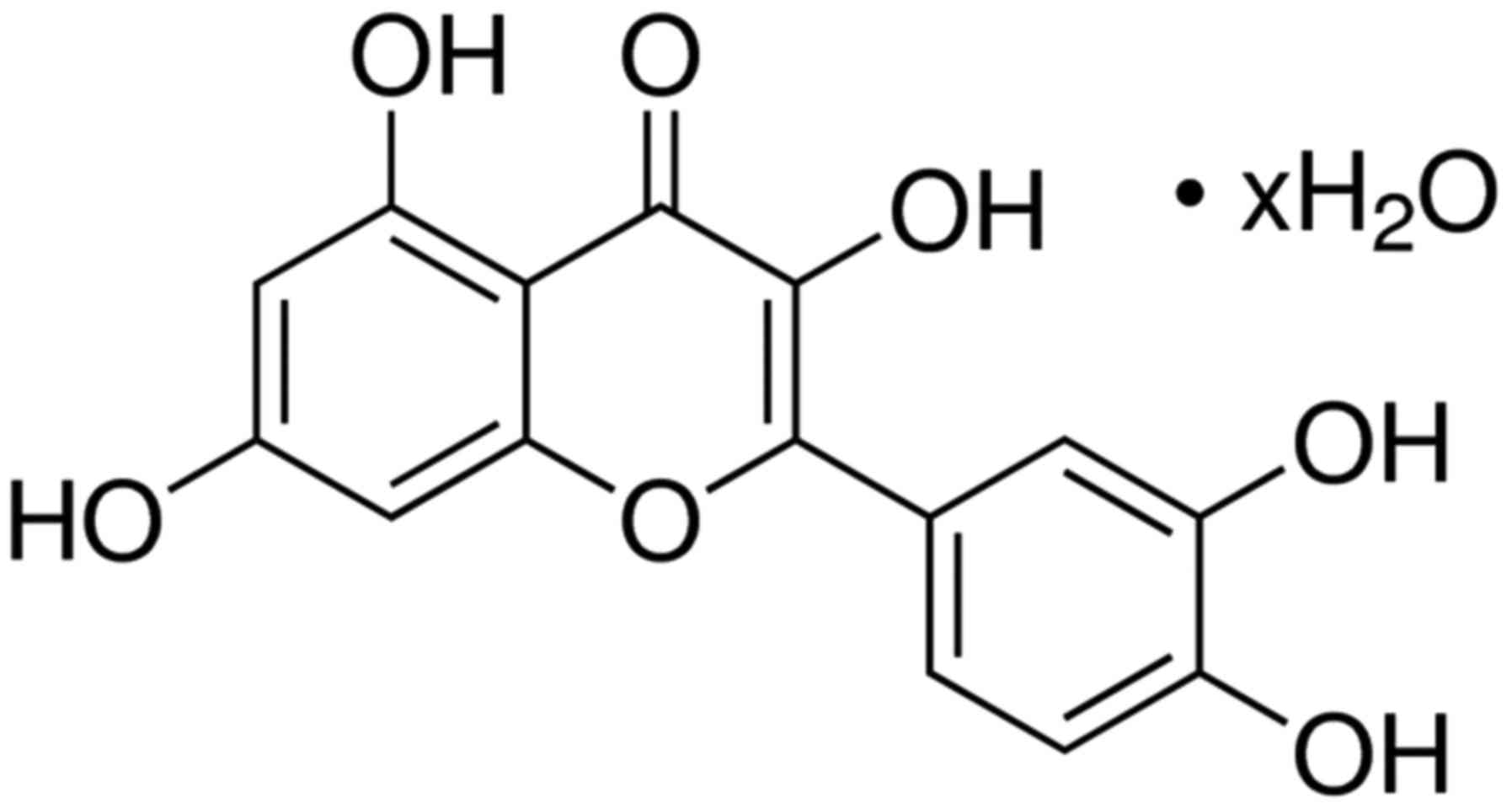
Quercetin (Fig. 1) is
a kind of flavonoid antioxidant substance, present in a variety of
plants such as apples, green tea, onions, grape skins and so on.
The red wine also contains a lot, and it is not easily soluble in
water (13). It is regarded to have
protective effect on cardiovascular and respiratory systems.
Moreover, it has been shown that quercetin can reduce carcinogens
and inhibit the growth of cancer cells (14). It is found that the quercetin flavors
extracted from French red wine can effectively inhibit the
proliferation of prostate cancer cells and only a small dose is
enough, which is equivalent to the amount contained in two cups of
red wine every day (15). Quercetin
has significant antioxidant and anti-inflammatory activities, so as
to be used to treat a variety of diseases (16). Therefore, these fruits were selected
to evaluate the protective role of quercetin prevents
alcohol-induced liver injury.
Materials and methods
Animal and grouping
Eight-week-old male C57 mice (n=30) were obtained
from Anhui Medical University. The animal protocol was approved by
the Institutional Animal Care and Use Committee of Anhui Medical
University. Mice were maintained in a room with a controlled
temperature of 22±0.5°C, 50–60% relative humidity and were allowed
free access to a basal pellet diet and tap water. 30 mice were
randomly divided into three groups (very group=10): Control group,
model group and quercetin group. Mice of model group and quercetin
group were irrigated with 35% v/v ethanol (3 g/kg) for 1 week, 40%
v/v ethanol (4 g/kg) for 1 week, and 52% v/v ethanol (5 g/kg) for 1
week. Meanwhile, we used staining for alcohol-duced liver injury
model. Meanwhile, mice of quercetin group were irrigated with 60
mg/kg of quercetin for 3 weeks. Then, the animals were
anesthetized, and liver tissues and blood samples were
collected.
Liver oil red O staining
After anesthesia, liver tissue was acquired, washed
with PBS and fixed in 10% formalin for 24 h. Then, tissue was
embedded in paraffin, cut into 0.4 µM and tained with Oil Red O
(17).
Measurement of hepatic injury in the
serum
The blood samples were centrifuged at 2,000 × g for
10 min to separate the serums. ELISA assays to determine AST, ALT,
TBIL, TG, SOD, GSH-Px, MDA, IL-1β, IL-6, IL-10, iNOS and caspase-3
contents.
Western blot analysis
Liver tissues samples were collected and homogenized
and treated with RIPA lysis buffer (Dingguo, China) and protease
inhibitor (1:100, Dingguo, China). Proteins contents were
determined using BCA kit (Dingguo, China). 25–50 µg protein were
electrophoresed on 6–10% sodium dodecyl sulfate-polyacrylamide gel
(SDS-PAGE) and transferred onto PVDF membranes (0.45 µm, Millipore,
BioRad). Membranes were blocked with 5% BSA in TBST for 1 h at 37°C
and incubated with iNOS, Bax/Bcl-2, PARP, p-STAT3, NF-κB, p-Akt and
GAPDH overnight at 4°C. Membranes washed with TBST for 3 times and
ncubated for 2 h at room temperature with secondary HRP-conjugated
sheep anti-rabbit antibody. Immunoreactivity was detected by
enhanced chemiluminescence (ECL kit, Millipore, USA) and measured
using the Bio-Rad Laboratories Quantity One software.
Statistical analysis
The data were presented as mean ± standard
deviation. Differences among multiple groups were analyzed by
D'Agostino's K-squared test followed by Tukey's test. P<0.05 was
considered to be statistically significant.
Results
The effect of quercetin on liver
injure
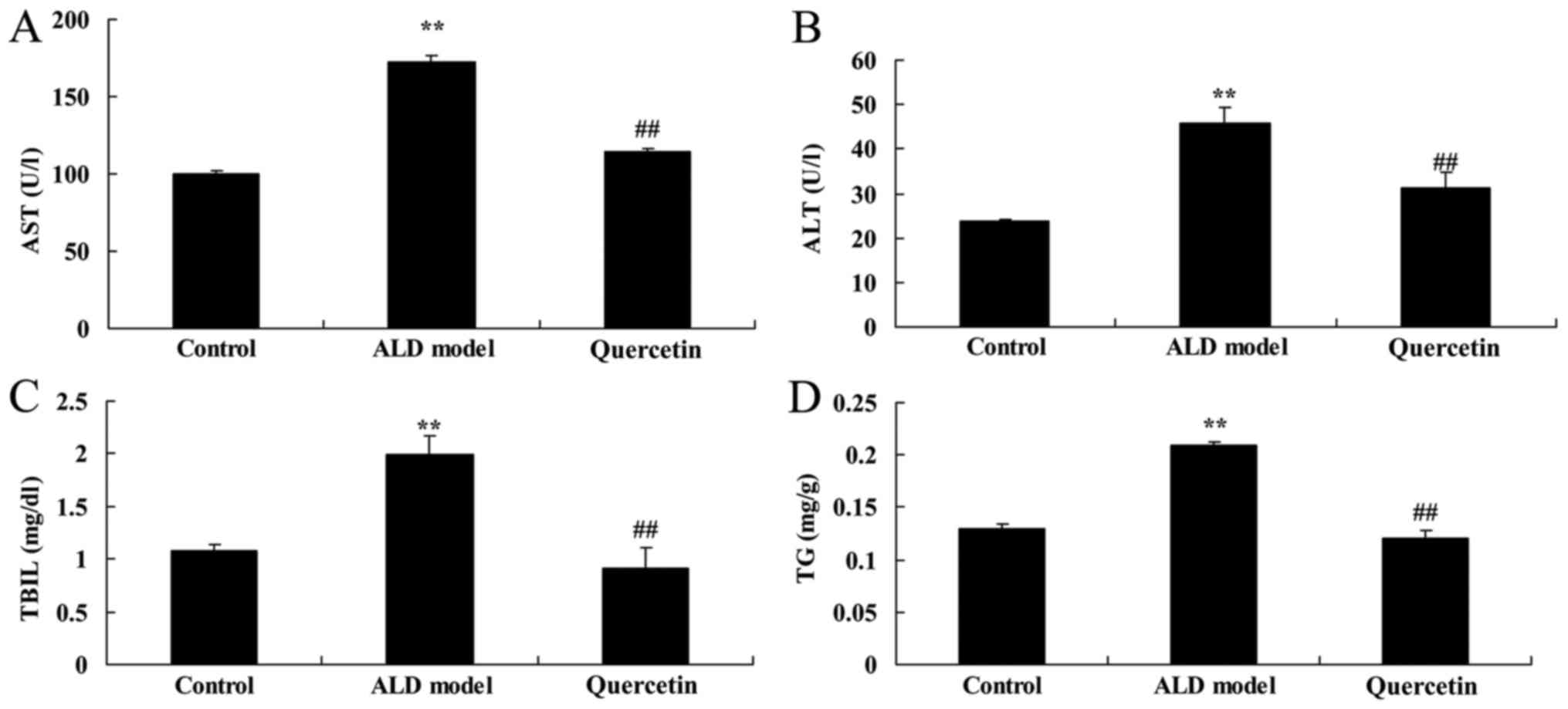
As showed in Fig. 2,
AST, ALT, TBIL and TG content of ALD model group were higher than
those of control group. Quercetin significantly reduced AST, ALT,
TBIL and TG content in ALD mice, compared with ALD model group
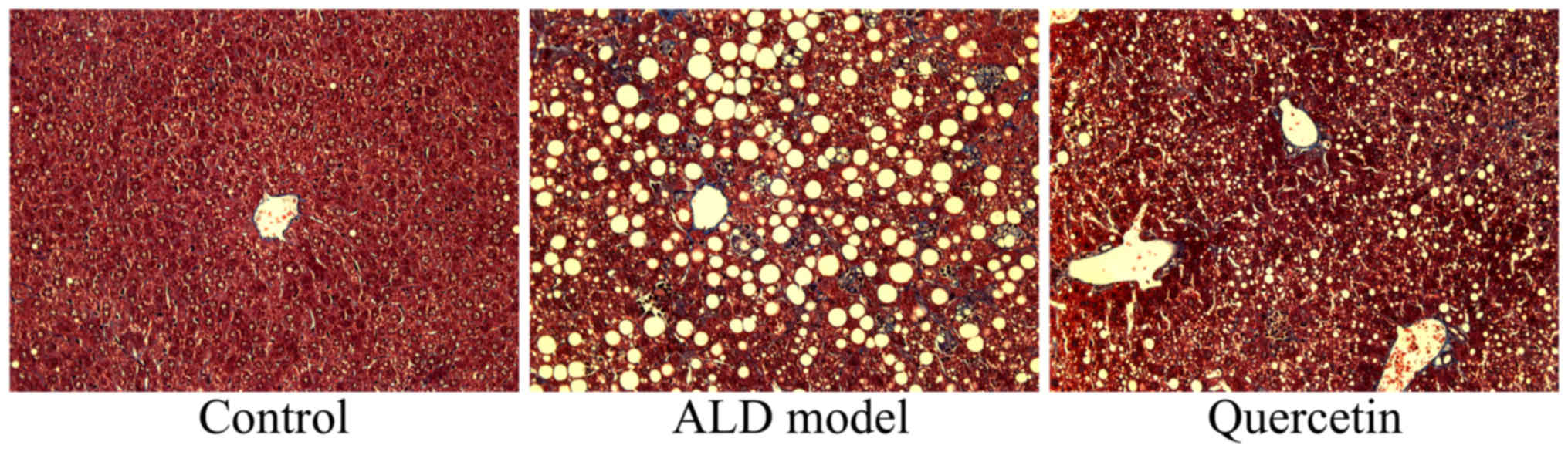
(Fig. 2). Meanwhile, we used red oil
assay to stained liver tissue and found that liver injure were
significantly increased in ALD model group, compared with control
group (Fig. 3). However, treatment
with quercetin significantly inhibited liver injure in ALD mice,
compared with ALD model group (Fig.
3).
The effect of quercetin on SOD, GSH-Px
and MDA contents
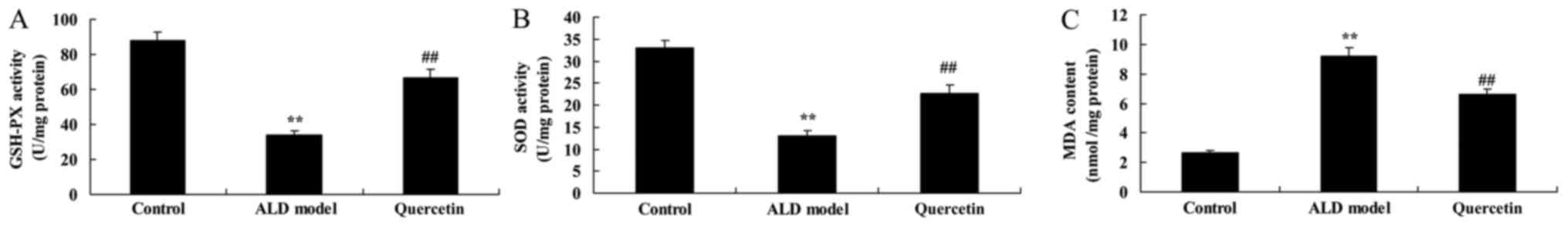
To investigate the antioxidant effects of quercetin
in alcohol-induced liver injury mice, SOD, GSH-Px and MDA contents
were measured using ELISA Kits. The SOD and GSH-Px contents were
reduced and MDA content was increased in ALD model group, compared
with control group (Fig. 4).
Quercetin significantly increased SOD and GSH-Px contents and
inhibited MDA content in ALD mice, compared with ALD model group
(Fig. 4). These data indicate that
quercetin might inhibit oxidative stress of ALD mice.
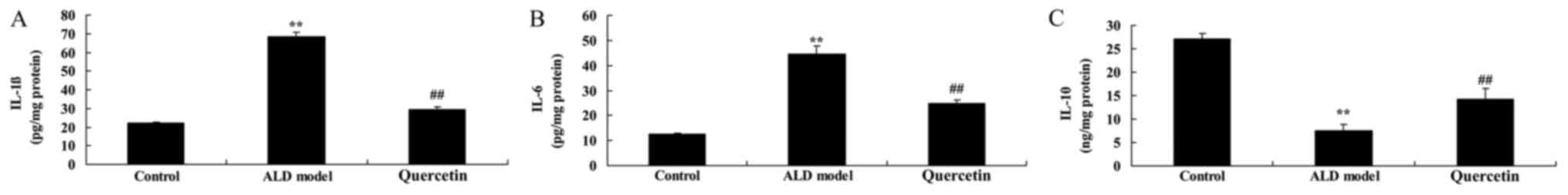
The effect of quercetin on IL-1β,
IL-6, IL-10 contents
To investigate the anti-inflammation effects of
quercetin in alcohol-induced liver injury mice, IL-1β, IL-6, IL-10
contents were measured using ELISA Kits. The levels of IL-1β and
IL-6 were promoted and IL-10 level was decreased in ALD model
group, compared with control group (Fig.
5). Quercetin significantly also reduced IL-1β and IL-6 levels
and increased IL-10 level in ALD mice, compared with ALD model
group (Fig. 5). Our study showed
that quercetin reduced inflammation of ALD, and it mechanism need
to measure.
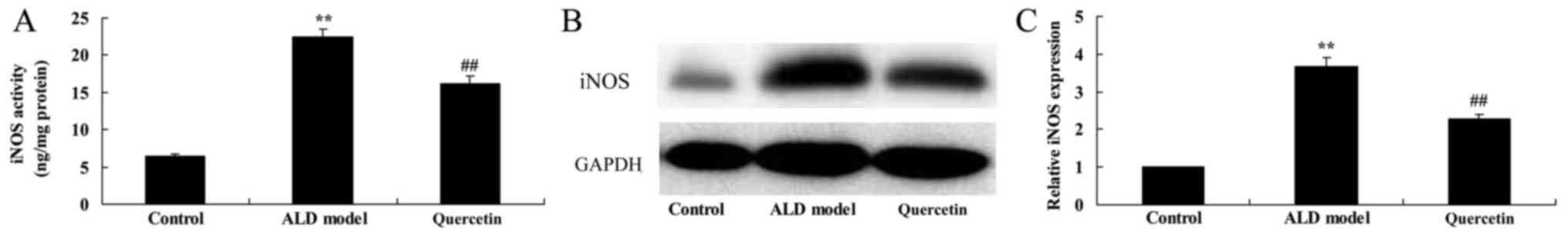
The effect of quercetin on iNOS contents and protein
expression. Nevertheless, we also found that iNOS contents and
protein expression of ALD model group were higher than that of
control group (Fig. 6). Treatment
with quercetin significantly iNOS contents and protein expression
in ALD mice, compared with ALD model group (Fig. 6), suggesting that quercetin inhibited
iNOS contents and protein expression to prevent ALD.
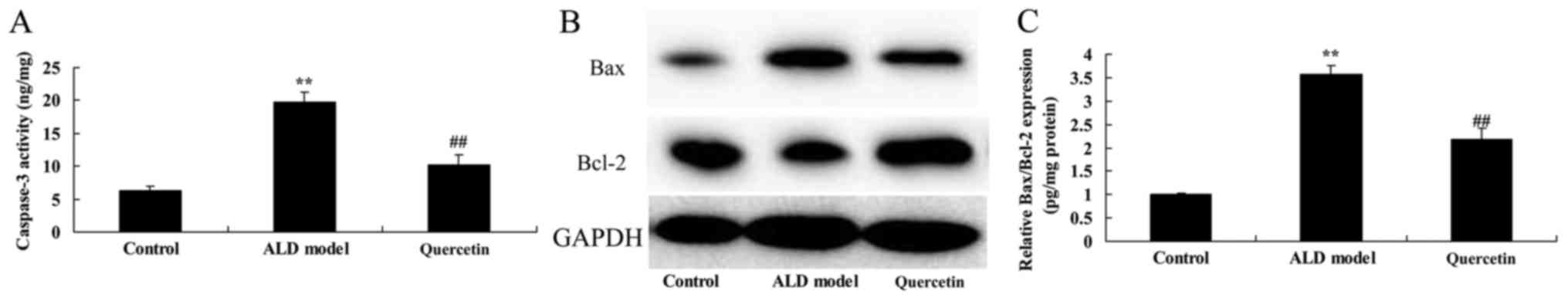
The effect of quercetin on caspase-3
contents and Bax/Bcl-2 protein expression
In order to explore the anti-apoptosis of quercetin
in alcohol-induced liver injury mice, caspase-3 contents and
Bax/Bcl-2 protein expression were measured in ALD mice. As showed
in Fig. 7, caspase-3 contents and
Bax/Bcl-2 protein expression were promoted in ALD model group,
compared with control group. Treatment with quercetin significantly
suppressed caspase-3 contents and Bax/Bcl-2 protein expression in
ALD mice, compared with ALD model group (Fig. 7). The results indicate that quercetin
possess anti-apoptosis effects in treatment of ALD.
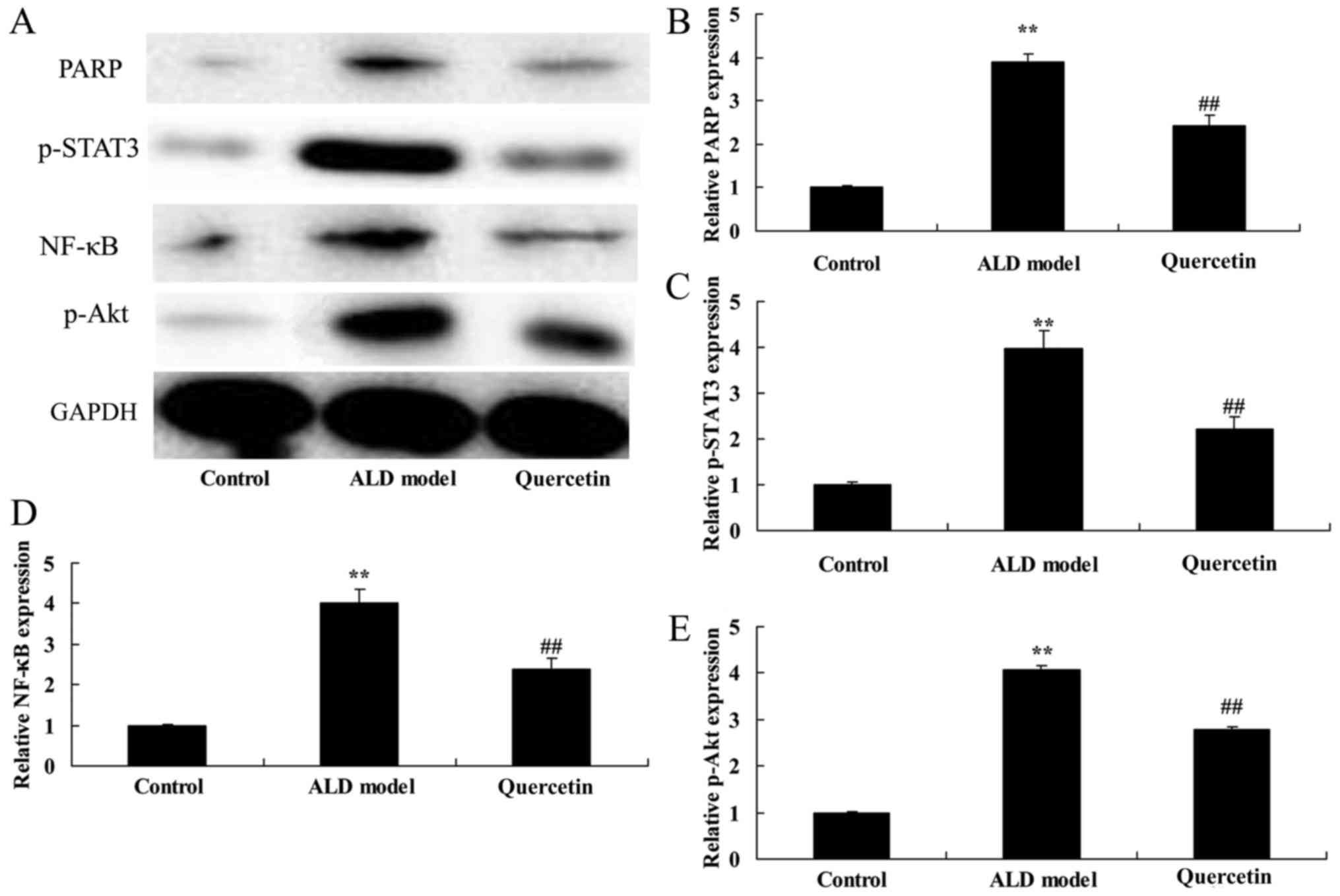
The effect of quercetin on p-STAT3,
NF-κB, PARP and p-Akt protein expression
In order to determine the potential mechanism of
magnesium isoglycyrrhizinat in alcohol-induced liver injury mice,
p-STAT3, NF-κB, PARP and p-Akt protein expression was measured
using Western blot analysis. As showed in Fig. 8, p-STAT3, NF-κB, PARP and p-Akt
protein expression was induced in ALD model group, compared with
compared with control group. p-STAT3, NF-κB, PARP and p-Akt protein
expression was significantly suppressed by magnesium
isoglycyrrhizinat in ALD mice, compared with ALD model group
(Fig. 8). Ours showed that quercetin
inhibited inflammation and apoptosis through STAT3/NF-κB and
PARP/Akt protein expression in treatment of ALD.
Discussion
About 90% individuals in those who have been
drinking over a long period of time develop into alcoholic fatty
liver disease, and only a few will develop into alcoholic hepatitis
or alcoholic cirrhosis (18). The
current accepted interpretation of this phenomenon is the ‘second
strike theory’. A large number of alcohol intake induces the
formation of alcoholic fatty liver as a ‘first strike’, and the
alcoholic liver injury requires a second strike, including the
change in nutrition, the effect of drug poisons, viral infections
and genetic factors (19). The
result showed that Quercetin significantly reduced AST, ALT, TBIL
and TG content in ALD mice.
In the past, the pathogenesis of ALD has been
extensively studied. The main pathogenetic causes of ALD are
oxidative damage, inflammatory injury, mitochondrial damage,
apoptosis damage, metabolic damage and aldehyde toxicity (20). The literature shows that the liver
cells generate large amounts of reactive oxygen in the process of
metabolizing alcohol through alcohol metabolism and mitochondrial
pathway (21). When beyond the scope
of the antioxidant defense system, the oxidative stress occurs
(21). Alcohol metabolism can also
generate a large number of toxic products, acetaldehyde, and then
mediate toxicity (21). Inflammatory
reactions are also involved in the pathogenesis of alcoholic liver
injury, and inflammatory factors can further promote oxidative
stress injury (22). Excessive
alcohol exposure leads to oxidative stress, acetaldehyde toxicity
and inflammation, which can lead to the damage of important
cellular organelle structure or function, such as endoplasmic
reticulum stress and mitochondrial dysfunction, so that the liver
cell function shows disorder or even induces apoptosis or necrosis
of liver cell, ultimately affecting liver function and leading to
ALD (20). The present study
demonstrated that quercetin significantly increased SOD and GSH-Px
contents and inhibited MDA content, and reduced IL-1β and IL-6
levels and increased IL-10 level in ALD mice. Xue et al
revealed that quercetin inhibits LPS-induced inflammation in
atherosclerosis (16).
Abnormality of cytokine metabolism is another
feature of ALD, especially TNF-α. Studies have shown that alcohol
perfusion can cause a significant increase in serum LPS level,
which is bond to the LPS CD14/toll-like receptor 4 complex on the
surface of Kupffer cells to activate NF-κB and TNF-α expression
(23,24). The level of TNF-α in patients with
ALD is significantly increased and the severity of ALD is
positively correlated with the severity of ALD (24). Several research groups have
demonstrated that TNF-α antibody therapy can prevent
alcohol-induced liver injury in rats.
Clinical experimental researches suggest that
apoptosis of hepatocytes exists in ALD, and studies show the
inhibition of hepatocyte apoptosis can relieve alcohol-induced
liver injury in experimental animals (25). Pathologic studies show oxidative
stress and hepatocyte apoptosis are involved in ALD cells
apoptosis. The apoptosis of a large number of hepatocytes is a
major feature of fulminant hepatitis and the inhibition of
excessive hepatocyte apoptosis can be used as a means of treating
fulminant hepatic failure (26).
Previous studies have shown that the anti-apoptotic effect is
realized by the activation of STAT3 signaling pathways to induce
anti-apoptotic protein expression (26,27). In
the present study, quercetin significantly suppressed p-STAT3
protein expression in ALD mice. Xue et al revealed that
quercetin inhibits LPS-induced inflammation in atherosclerosis
through suppression of STAT3 protein expression (16).
TNF-α and other inflammatory factors cause liver
injury, and IL-6, IL-10 and others can protect the liver through
inflammatory response (25). IL-6
up-regulates the expression of multiple anti-injury genes in liver
cells by activating STAT-3 pathway; IL-10 inhibits hepatic
inflammatory response by activating STAT3 in Kupffer cells
(25). The imbalance and re-balance
process of proinflammatory and anti-inflammatory factors is the
development and regression of ALD (28).
Interestingly, although the inflammation is more
obvious after the knockout of IL-10 gene from mice liver, but liver
cell damage and steatosis are decreased, and the IL-6/STAT3
expression in liver is increased. This phenomenon may be related to
liver cell damage and weakened steatosis (23). Some studies have shown that IL-10 can
reduce the activation of NF-κB after stimulation. IL-10 inhibits
the binding of NF-κB with DNA activity from the cytoplasm to the
nucleus by inhibiting the activity of nuclear factor (IKK),
inhibits the NF-κB activity, and reduces the release of TNFα,
thereby reducing the inflammatory response (7). Thus, the data suggest that quercetin
significantly suppressed NF-κB in ALD mice.
Apoptosis refers to the cell death under a certain
physiological or pathological condition, with special morphological
and biochemical characteristics, which is regulated by gene
(22). Physiological apoptosis is
the normal life phenomenon, by which the body or cell adapts to the
environment or growth and differentiation, but pathological
apoptosis may cause function damage of the corresponding tissue and
structure, which may lead to the failure of relevant organs
(29).
Sperm exposure also leads to abnormal expression of
apoptosis-related genes, such as the Bcl-2 protein family.
Bax/Bcl-2 ratio can also reflect the degree of apoptosis to some
extent (30). Bcl-2 protein can also
promote the synthesis of GSH and inhibit the consumption of
mitochondria GSH, to play an antioxidant role, in order to maintain
mitochondrial redox homeostasis. It is also found that the lower
expression level of Bcl-2 means the higher level of lipid
peroxidation (30). Caspase-3 is an
important executive molecule in the process of apoptosis (9). The activity of Caspase-3 is often
indicative of the degree of apoptosis in tissues or cells.
Caspase-3 activity is increased during ALD. This result suggests
that quercetin significantly suppressed caspase-3 contents and
Bax/Bcl-2 protein expression in ALD mice. Du et al indicate
that quercetin attenuates neuronal apoptosis via caspase-3 and Bax
levels (13).
The PI3K/Akt pathway is an important component of
the insulin signaling pathway, which is also a classic cell
survival signaling pathway and glucose metabolism regulation
pathway that activates NF-κB and increases the expression of
anti-apoptotic proteins to improve cell survival (9). In addition, it is also reported that
the activation of Akt can also inhibit the activity of Caspase-3
(9). It is also shown from
literature that the activation of Akt inhibits the binding of
Bcl-xL to Bcl-xL and finally inhibits apoptosis (8). In the current study, we found that
quercetin significantly inhibited p-Akt protein expression in ALD
mice. Lu et al showed that quercetin attenuates high
fructose feeding-induced atherosclerosis through inhibition of
PI3K/AKT signaling pathway (31).
However, in this study, we not used PI3K/Akt/NF-κB or STAT3
signaling pathway inhibitor to verify the function of
PI3K/Akt/NF-κB or STAT3 signaling pathway in effects of quercetin
on alcohol-induced liver injury, which are limitations of the study
and these need to execute in further study.
In summary, our study showed that quercetin prevents
alcohol-induced liver injury through its antioxidant,
anti-inflammation and anti-apoptosis effects by STAT3, Akt and
NF-κB pathway. These results suggest that quercetin is a promising
therapeutic target for treating alcohol-induced liver injury in
further clinical.
References
|
1
|
Kirpich IA, Solovieva NV, Leikhter SN,
Shidakova NA, Lebedeva OV, Sidorov PI, Bazhukova TA, Soloviev AG,
Barve SS, McClain CJ and Cave M: Probiotics restore bowel flora and
improve liver enzymes in human alcohol-induced liver injury: A
pilot study. Alcohol. 42:675–682. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Michel MC, Radziszewski P, Falconer C,
Marschall-Kehrel D and Blot K: Unexpected frequent hepatotoxicity
of a prescription drug, flupirtine, marketed for about 30 years. Br
J Clin Pharmacol. 73:821–825. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wu Y, Pan X, Zhang S, Wang W, Cai M, Li Y,
Yang F and Guo H: Protective effect of corn peptides against
alcoholic liver injury in men with chronic alcohol consumption: A
randomized double-blind placebo-controlled study. Lipids Health
Dis. 13:1922014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Li S, Zhang J, Wang Z, Wang T, Yu Y, He J,
Zhang H, Yang T and Shen Z: MicroRNA-17 regulates autophagy to
promote hepatic ischemia/reperfusion injury via suppression of
signal transductions and activation of transcription-3 expression.
Liver Transp. 22:1697–1709. 2016. View
Article : Google Scholar
|
|
5
|
Kim HY, Jhun JY, Cho ML, Choi JY, Byun JK,
Kim EK, Yoon SK, Bae SH, Chung BH and Yang CW: Interleukin-6
upregulates Th17 response via mTOR/STAT3 pathway in
acute-on-chronic hepatitis B liver failure. J Gastroenterol.
49:1264–1273. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chen X, Meng Q, Wang C, Liu Q, Sun H, Huo
X, Sun P, Yang X, Peng J and Liu K: Protective effects of calycosin
against CCl4-induced liver injury with activation of FXR and STAT3
in mice. Pharm Res. 32:538–548. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tan Z, Luo M, Yang J, Cheng Y, Huang J, Lu
C, Song D, Ye M, Dai M, Gonzalez FJ, et al: Chlorogenic acid
inhibits cholestatic liver injury induced by
α-naphthylisothiocyanate: Involvement of STAT3 and NFκB signalling
regulation. J Pharm Pharmacol. 68:1203–1213. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yang J, Chen Q, Tian S, Song S, Liu F,
Wang Q and Fu Z: The role of 1,25-dyhydroxyvitamin D3 in mouse
liver ischemia reperfusion injury: Regulation of autophagy through
activation of MEK/ERK signaling and PTEN/PI3K/Akt/mTORC1 signaling.
Am J Transl Res. 7:2630–2645. 2015.PubMed/NCBI
|
|
9
|
Suo L, Kang K, Wang X, Cao Y, Zhao H, Sun
X, Tong L and Zhang F: Carvacrol alleviates ischemia reperfusion
injury by regulating the PI3K-Akt pathway in rats. PLoS One.
9:e1040432014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Xie YL, Chu JG, Jian XM, Dong JZ, Wang LP,
Li GX and Yang NB: Curcumin attenuates
lipopolysaccharide/d-galactosamine-induced acute liver injury by
activating Nrf2 nuclear translocation and inhibiting NF-kB
activation. Biomed Pharmacother. 91:70–77. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li Z, Zhang J, Mulholland M and Zhang W:
mTOR activation protects liver from ischemia/reperfusion-induced
injury through NF-kappaB pathway. FASEB J. 31:3018–3026. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Choudhury S, Ghosh S, Mukherjee S, Gupta
P, Bhattacharya S, Adhikary A and Chattopadhyay S: Pomegranate
protects against arsenic-induced p53-dependent ROS-mediated
inflammation and apoptosis in liver cells. J Nutr Biochem.
38:25–40. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Du G, Zhao Z, Chen Y, Li Z, Tian Y, Liu Z,
Liu B and Song J: Quercetin attenuates neuronal autophagy and
apoptosis in rat traumatic brain injury model via activation of
PI3K/Akt signaling pathway. Neurol Res. 1–8. 2016.(Epub ahead of
print). PubMed/NCBI
|
|
14
|
Liu Y, Gong W, Yang ZY, Zhou XS, Gong C,
Zhang TR, Wei X, Ma D, Ye F and Gao QL: Quercetin induces
protective autophagy and apoptosis through ER stress via the
p-STAT3/Bcl-2 axis in ovarian cancer. Apoptosis. 22:544–557. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tsai PH, Cheng CH, Lin CY, Huang YT, Lee
LT, Kandaswami CC, Lin YC, Lee KP, Hung CC, Hwang JJ, et al:
Dietary flavonoids luteolin and quercetin suppressed cancer stem
cell properties and metastatic potential of isolated prostate
cancer cells. Anticancer Res. 36:6367–6380. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xue F, Nie X, Shi J, Liu Q, Wang Z, Li X,
Zhou J, Su J, Xue M, Chen WD and Wang YD: Quercetin inhibits
LPS-induced inflammation and ox-LDL-induced lipid deposition. Front
Pharmacol. 8:402017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ge X, Leung TM, Arriazu E, Lu Y, Urtasun
R, Christensen B, Fiel MI, Mochida S, Sørensen ES and Nieto N:
Osteopontin binding to lipopolysaccharide lowers tumor necrosis
factor-α and prevents early alcohol-induced liver injury in mice.
Hepatology. 59:1600–1616. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Dart RC, Green JL, Kuffner EK, Heard K,
Sproule B and Brands B: The effects of paracetamol (acetaminophen)
on hepatic tests in patients who chronically abuse alcohol-a
randomized study. Aliment Pharmacol Ther. 32:478–486. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Garcia-Gil FA, Serrano MT, Fuentes-Broto
L, Arenas J, García JJ, Güemes A, Bernal V, Campillo A, Sostres C,
Araiz JJ, et al: Celsior versus University of Wisconsin preserving
solutions for liver transplantation: Postreperfusion syndrome and
outcome of a 5-year prospective randomized controlled study. World
J Surg. 35:1598–1607. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Xu LQ, Xie YL, Gui SH, Zhang X, Mo ZZ, Sun
CY, Li CL, Luo DD, Zhang ZB, Su ZR and Xie JH: Polydatin attenuates
d-galactose-induced liver and brain damage through its
anti-oxidative, anti-inflammatory and anti-apoptotic effects in
mice. Food Funct. 7:4545–4555. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wu CH, Chen CC, Lai CY, Hung TH, Lin CC,
Chao M and Chen SF: Treatment with TO901317, a synthetic liver X
receptor agonist, reduces brain damage and attenuates
neuroinflammation in experimental intracerebral hemorrhage. J
Neuroinflammation. 13:622016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yu HJ, Lin BR, Lee HS, Shun CT, Yang CC,
Lai TY, Chien CT and Hsu SM: Sympathetic vesicovascular reflex
induced by acute urinary retention evokes proinflammatory and
proapoptotic injury in rat liver. Am J Physiol Renal Physiol.
288:F1005–F1014. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Jung J, Moon JW, Choi JH, Lee YW, Park SH
and Kim GJ: Epigenetic alterations of IL-6/STAT3 signaling by
placental stem cells promote hepatic regeneration in a rat model
with CCl4-induced liver injury. Int J Stem Cells. 8:79–89. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Cui X, Chen Q, Dong Z, Xu L, Lu T, Li D,
Zhang J, Zhang M and Xia Q: Inactivation of Sirt1 in mouse livers
protects against endotoxemic liver injury by acetylating and
activating NF-κB. Cell Death Dis. 7:e24032016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Grant L, Shearer KD, Czopek A, Lees EK,
Owen C, Agouni A, Workman J, Martin-Granados C, Forrester JV,
Wilson HM, et al: Myeloid-cell protein tyrosine phosphatase-1B
deficiency in mice protects against high-fat diet and
lipopolysaccharide-induced inflammation, hyperinsulinemia, and
endotoxemia through an IL-10 STAT3-dependent mechanism. Diabetes.
63:456–470. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Obayashi Y, Arisaka H, Yoshida S, Mori M
and Takahashi M: Proline protects liver from D-galactosamine
hepatitis by activating the IL-6/STAT3 survival signaling pathway.
Amino Acids. 43:2371–2380. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wan Y, Jiang S, Lian LH, Bai T, Cui PH,
Sun XT, Jin XJ, Wu YL and Nan JX: Betulinic acid and betulin
ameliorate acute ethanol-induced fatty liver via TLR4 and STAT3 in
vivo and in vitro. Int Immunopharmacol. 17:184–190. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang H, Shi P, Huang C and Liu Q: Maresin
1 ameliorates iron-deficient anemia in IL-10(−/-) mice with
spontaneous colitis by the inhibition of hepcidin expression though
the IL-6/STAT3 pathway. Am J Transl Res. 8:2758–2766.
2016.PubMed/NCBI
|
|
29
|
Zhang M, Wang C, Wang C, Zhao H, Zhao C,
Chen Y, Wang Y, McClain C and Feng W: Enhanced AMPK phosphorylation
contributes to the beneficial effects of Lactobacillus rhamnosus GG
supernatant on chronic-alcohol-induced fatty liver disease. J Nutr
Biochem. 26:337–344. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Song E, Fu J, Xia X, Su C and Song Y:
Bazhen decoction protects against acetaminophen induced acute liver
injury by inhibiting oxidative stress, inflammation and apoptosis
in mice. PLoS One. 9:e1074052014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lu XL, Zhao CH, Yao XL and Zhang H:
Quercetin attenuates high fructose feeding-induced atherosclerosis
by suppressing inflammation and apoptosis via ROS-regulated
PI3K/AKT signaling pathway. Biomed Pharmacother. 85:658–671. 2017.
View Article : Google Scholar : PubMed/NCBI
|