Introduction
Asthma is a chronic inflammatory disorder that
affects >300 million people annually worldwide (1), and the worldwide incidence of pediatric
asthma is currently increasing. Asthma is known to be a
heterogeneous pulmonary disease characterized by chronic airway
inflammation, leading to respiratory swelling and dyspnea (2–4). It has
been demonstrated that mast cells, eosinophil granulocytes,
cytokines and T cells serve essential roles in the initiation and
progression of pediatric asthma (5,6).
Previous studies have also indicated that the inflammatory factors
matrix metalloproteinase-9 (MMP-9) and transforming growth factor-β
(TGF-β) induced by immunocytes are closely correlated with airway
inflammation and airway remodeling (7,8).
In recent years, several studies have observed that
the incidence of pediatric asthma presents an increasing trend
according to clinical investigation (9,10).
Pediatric asthma was reported as the most common chronic
respiratory disease in children, and this disease deleteriously
impacts all aspects of systemic function (11). In addition, it has been suggested
that patients with pediatric asthma exhibit behavioral and
psychological problems that contribute to the risk for functional
impairments and communication difficulties (12). Furthermore, environmental factors
have been regarded as one of the main preventable contributors for
the epidemiology of pediatric asthma (13). Notably, although various treatments
have been proposed for pediatric asthma, numerous patients remain
refractory under current clinical therapies. Therefore, novel and
efficient treatments need to be urgently identified for patients
with pediatric asthma.
Latrophilin agents, a novel family of receptors,
have been reported to present beneficial effects on airway smooth
muscle cells by regulating airway smooth muscle cell adhesion,
proliferation and contraction (14).
However, their physiological function remains unclear (15). Therefore, the present clinical trial
hypothesized that recombinant human latrophilin 3 antibody
(rhLPHN3Ab) may exert a beneficial effect on the recovery of
patients with pediatric asthma due to its efficacy on the
improvement of airway smooth muscle cell contraction, determined by
forced expiratory volume (FEV), forced vital capacity (FVC) and
peak expiratory flow (PEF).
Several signaling pathways involved in the
initiation and development of pediatric asthma have been proposed,
including c-Jun N-terminal kinase, tumor necrosis factor-α (TNF-α),
protein kinase A (PKA)/nuclear factor-κB (NF-κB), MAP
kinase-interacting serine/threonine-protein kinase 1/eukaryotic
initiation factor 4E, extracellular signal-regulated kinase
(ERK)1/2 and receptor for advanced glycation end products/ERK
pathways (16–18). However, the mechanism(s) of
rhLPHN3-mediated signaling pathway has not been reported in
previous studies.
In the present study, the efficacy and underlying
mechanism of LPHN3 in the treatment of pediatric asthma was
investigated. It was observed that rhLPHN3 Ab decreased the
expression levels of inflammatory mediators, and contributed to
airway remodeling and bronchodilation through the PKA-induced NF-κB
signaling pathway. Notably, the results indicated that rhLPHN3 Ab
was associated with increased expression of E-selectin and mucin
5AC (MUC5AC). Given the clinical outcomes of the patients in the
current study, rhLPHN3 Ab may be a potential agent for future
application in the treatment of pediatric asthma.
Materials and methods
Ethics statement
This phase-I study (no. YSCOHLY20110812R1) was
conducted between August 2011 and June 2015, in strict accordance
with the recommendations and guidelines of the Yishui Central
Hospital of Linyi (Linyi, China). This study was approved by the
ethics committee of Yishui Central Hospital of Linyi. All patients
and their guardians were required to review trial protocols and
amendments, and provided informed consent prior to
participation.
Patients and treatments
A total of 342 children with capillary bronchitis
and asthma (age, 4–11-years-old) were recruited into the present
study, and the 22-item sinonasal outcome test (SNOT-22) and
Lund-Kennedy endoscopy scores (LKES) were used to evaluate the
status of pediatric asthma (19).
Patients were subjected to rhLPHN3, salbutamol or montelukast once
a day in a double-blind trial at Yishui Central Hospital of Linyi.
No others clinical syndromes were observed in this analysis. All
children were instructed to wash their mouths thoroughly with
normal saline before treatment. The children were randomly divided
into three groups and received treatment with salbutamol (12
mg/day; Tianjin Lisheng Pharmaceutical Co., Ltd., Tianjin, China),
montelukast (12 mg/day; Hangzhou MSD Pharmaceutical Co., Ltd.,
Hangzhou, China) or rhLPHN3 Ab (1:100 dilution with 0.9% NaCl, 12
mg/day; cat. no. BA7210, Boao Biological Pharmaceutical Co., Ltd.,
Tianjin, China) by nasal aerosolization for a total of 16
weeks.
Study design
Double-blind investigation was conducted at two time
points: At the baseline stage, and at 16 weeks of treatment for
patients with pediatric asthma. The patients continued treatment
with the dose of salbutamol (12 mg/day), montelukast (12 mg/day) or
rhLPHN3 Ab (12 mg/day) during the maintenance period.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total mRNA from airway smooth muscle cells (cells
obtained by primary culture of smooth muscle tissue) was isolated
using an mRNeasy Extraction kit (Qiagen, Inc., Valencia, CA, USA).
Extracted mRNA (1 µg) was transcribed into cDNA using a reverse
transcription kit (Qiagen, Inc.). The cDNA (10 ng) was used for
qPCR using the SYBR Green Master mix system (Bio-Rad Laboratories,
Inc., Hercules, CA, USA). All the forward and reverse primers for
E-selectin and MUC5AC were synthesized by Invitrogen (Thermo Fisher
Scientific, Inc.): E-selectin, forward
5′-CATTGGAGAGAAAGGAAAGTGTG-3′ and reverse
5′-GCTTGCATGTACGAAGAGGAT-3′; MUC5AC, forward
5′-TCCAACTACTACCAAGAACTGAA-3′ and reverse
5′-CAAGGAAATAGACGATAGCCAA-3′; β-actin, forward
5′-GTGGGCGCCCAGGCACCA-3′ and reverse 5′-CTCCTTAATGTCACGCACGATTT-3′.
PCR amplification followed preliminary denaturation at 94°C for 2
min, followed by 38 cycles of 95°C for 30 sec, annealing
temperature reduced to 62°C for 30 sec and 72°C for 10 min by
volume of 20 µl containing 50 ng of genomic DNA, 200 µM dNTP, 2.5
units of Taq DNA polymerase, and 200 µM primers. Relative mRNA
expression changes were calculated by 2−ΔΔCq (20).
Western blot analysis
Airway smooth muscle cells were isolated from
pediatric asthma patients by primary culture of smooth muscle
tissue and homogenized in lysate buffer containing
protease-inhibitor (Invitrogen; Thermo Fisher Scientific, Inc.),
then centrifuged at 7,104 × g at 4°C for 10 min. Supernatants were
collected and the protein concentration was detected using a
Bio-Rad protein assay kit (cat. no. 500-0002; Bio-Rad Laboratories,
Inc.) Protein (30 µg) was separated by 10% SDS-PAGE assays followed
by transference onto polyvinylidene difluoride membranes. The
membranes were blocked in Tris-buffered saline buffer (50 mmol/l
NaCl, 10 mmol/l Tris, pH 7.4) containing 5% nonfat milk for 2 h at
room temperature as previously described (20). For western blotting, the following
rabbit anti-human primary antibodies were used: pPKA (cat. no.
14270-1-AP, 1:1,000, Proteintech) and β-actin (cat. no. 66009-1-Ig,
1:5,000, Proteintech). Primary antibodies were incubated overnight
at 4°C. Membranes were then incubated with secondary antibodies
(cat. no. 4410, 1:1,000, Cell Signaling Technology) for 24 h at
4°C. The results were visualized using an enhanced
chemiluminescence detection system (Thermo Fisher Scientific,
Inc.). BandScan 5.0 software (Glyko, Inc., Novato, CA, USA) was
used for the quantification of proteins following western blot
analysis.
NF-κB activation
Airway smooth muscle cells were collected and lysed
by three freeze-thaw steps in 200 µl of 0.25 M Tris-HCl (pH 7.9)
plus 1 mM dithiothreitol. Cell extracts were clarified in a
microcentrifuge (7,104 × g), and 20 µl of each extract was
incubated with 350 µl of reaction buffer A [25 mM glycyl-glycine
(pH 7.8), 5 mM ATP (pH 7.5), 4 mM EGTA (pH 8.0), 15 mM MgSO4] and
then mixed with 100 µl 0.25 mM luciferin (Sigma-Aldrich; Merck
KGaA) in reaction buffer A. A TD-20/20 luminometer (Turner Designs,
Sunnyvale, CA, USA) was used for analyzing NF-κB activation using
an assay of the photons produced (measured in relative light
units).
DNA binding activity
Airway smooth muscle cells were isolated from
pediatric asthma patients on week 0 and 16. DNA binding activity
was determined by DNA binding and antitrypanosomal activity as
reported previously (21).
Outcome measurement
SNOT-22 and LKES tests were used for assessing the
efficacy of salbutamol, montelukast and rhLPHN3 Ab treatments in
children with pediatric asthma at the baseline and post-treatment
at week 16. Clinical pediatric asthma scores were evaluated as
descripted in previous studies (22,23).
Lung function testing
Lung function tests, including the FEV percentage,
FVC and PEF, were conducted using a Jaeger MasterScreen Pulmonary
Function Testing system (Jaeger; BD Biosciences, Franklin Lakes,
NJ, USA) to evaluate the tidal breathing flow volume at the
baseline and post-treatment. The procedures were performed
according to the manufacturer's instructions.
Efficacy and safety assessments
Subjective and objective outcome measurements were
used to assess the efficacy of salbutamol, montelukast and rhLPHN3
Ab on children patients with pediatric asthma. In order to minimize
bias, a single physician measured the scores at the preoperative
baseline and post-treatment. Efficacy assessments, including the
median percent reduction scores and response rate, were analyzed in
children with pediatric asthma from baseline during the 16-week and
double-blind period in the presence of rhLPHN3 Ab (12 mg/day). The
safety assessments of the most frequent treatment-emergent adverse
events were evaluated in all children with pediatric asthma who
received the study drug.
ELISA for detection of plasma
concentration
The levels of interleukin (IL)-10 (BMS614-2), IL-17
(39-8170-65), IL-4 (39-8041-65), MMP-9 (BMS2016-2), interferon-γ
(IFN-γ; BMS216TEN) and TGF-β (BMS249-4TEN) in the serum of children
patients with asthma were investigated using commercial ELISA kits
(Thermo Fisher Scientific, Inc.), according to the manufacturer's
protocols. Absorption was measured at 450 nm with an ELISA reader,
and was subsequently converted to the concentrations of IL-10,
IL-17, IL-4, MMP-9, IFN-γ and TGF-β (24).
Evaluation of toxicity
The median overall duration of treatment for
dose-limiting toxicity (DLT) and maximum tolerated dose (MTD) was
16 weeks for rhLPHN3 Ab dosing cohorts (1, 6, 12, 24 and 30 mg) and
each group had 20 patients. Toxicity was graded using the National
Cancer Institute Common Toxicity Criteria (version 3.0). DLT and
MTD of rhLPHN3 Ab were evaluated by hypertension and proteinuria
grade, as described previously (25).
Statistical analysis
All data are reported as the mean ± standard error
of the mean. Differences between mean values were assessed by
Student's t-test for unpaired data. Comparisons of data between
multiple groups were performed with analysis of variance. Responder
rates and treatment-emergent adverse events were analyzed by
χ2 test. P<0.05 was considered as an indicator of
statistically significant differences.
Results
Characteristics of patients with
pediatric asthma
In total,342 patients with pediatric asthma were
recruited into the present analysis. The characteristics of the
children patients are summarized in Table I. Out of 342 patients, 134 received
salbutamol, 108 received montelukast and 100 received rhLPHN3 Ab
treatment. The number of male and female patients was approximately
equal. In addition, the scores of asthma in children were analyzed
by SNOT-22 and LKES determination in order to evaluate the extent
of asthma at the baseline and following salbutamol, montelukast and
rhLPHN3 Ab treatment. The patients did not receive any other
medications during the treatment period.
 | Table I.Characteristics of study
population. |
Table I.
Characteristics of study
population.
| Characteristic | Salbutamol | Montelukast | rhLPHN3 Ab |
|---|
| No. of patients, n
(%) | 134 (39.2) | 108 (31.6) | 100 (29.2) |
| Gender, n |
|
|
|
|
Male | 60 | 58 | 46 |
|
Female | 74 | 50 | 54 |
| Mean age,
years | 6.5±2.2 | 6.6±2.8 | 6.8±2.0 |
| BMI | 16.8±3.4 | 17.4±2.6 | 17.2±3.0 |
| SNOT-22 | 30.1±4.8 | 32.2±5.6 | 31.5±4.6 |
| LKES | 8.2±2.5 | 7.6±3.1 | 8.3±1.9 |
| FVC, l | 1.52±2.1 | 1.47±2.6 | 1.49±2.4 |
| PEF, % | 68.4±7.8 | 66.5±9.5 | 67.2±8.6 |
| FEV, % | 65.2±6.2 | 64.7±7.6 | 66.2±8.1 |
Duration of treatment, DLT and MTD of
rhLPHN3 Ab
The overall duration of salbutamol, montelukast and
rhLPHN3 Ab treatments were identified as 16 weeks for all
treatments. The various doses of rhLPHN3 Ab were 1, 6, 12, 24 and
30 mg, which were used to evaluate the optimal dosage. Data
presented in Table II revealed that
the DLT and MTD of rhLPHN3 Ab were 24 and 30 mg, respectively.
Furthermore, analysis indicated that the common treatment-emergent
adverse events of rhLPHN3 Ab treatment were hypertension,
proteinuria, fatigue, hypertriglyceridemia, constipation and edema
peripheral (Table II). Notably, the
majority of patients with pediatric asthma required reduction of
the drug dose due to cumulative toxicity following treatment with
the MTD dose of rhLPHN3 Ab. Therefore, pediatric asthma patients
included in the present study received a dose of 12 mg/day rhLPHN3
Ab. Following administration of the last dose of rhLPHN3 Ab (12
mg), it was observed that the most common treatment-emergent
adverse events were hypertension and proteinuria (≥10% each;
Table III). Among the total cohort
of patients, all pediatric asthma patients completed the overall
maintenance period of the phase I study (n=100).
 | Table II.Treatment-emergence adverse events of
rhLPHN3 Ab after 16 weeks treatment. |
Table II.
Treatment-emergence adverse events of
rhLPHN3 Ab after 16 weeks treatment.
|
|
| Dose of rhLPHN3
Ab |
|---|
|
|
|
|
|---|
| Adverse event | Total (n=100) | 1–6 mg/day
(n=40) | 12–24 mg/day
(n=40) | 30 mg/day
(n=20) |
|---|
| Hypertension | 8 | 2 | 3 | 3 |
| Proteinuria | 5 | 1 | 2 | 2 |
| Fatigue | 3 | 0 | 1 | 2 |
|
Hypertriglyceridemia | 3 | 1 | 1 | 1 |
| Constipation | 4 | 1 | 1 | 2 |
| Edema
peripheral | 4 | 1 | 1 | 2 |
 | Table III.Hypertension and proteinuria grades
after 16 weeks treatment with rhLPHN3 Ab. |
Table III.
Hypertension and proteinuria grades
after 16 weeks treatment with rhLPHN3 Ab.
|
|
| Dose of rhLPHN3
Ab |
|---|
|
|
|
|
|---|
| Adverse event | Total (n=37) | 1–10 mg/day
(n=12) | 15 mg/day
(n=15) | 20 mg/day
(n=10) |
|---|
| Hypertension | 8 | 2 | 3 | 3 |
| Grade 1 | 4 | 1 | 2 | 1 |
| Grade 2 | 2 | 0 | 1 | 1 |
| Grade 3 | 2 | 1 | 0 | 1 |
| Proteinuria | 5 | 1 | 2 | 2 |
| Grade 1 | 2 | 0 | 1 | 1 |
| Grade 2 | 2 | 1 | 1 | 0 |
| Grade 3 | 1 | 0 | 0 | 1 |
Efficacy of rhLPHN3 Ab treatment in
patients with pediatric asthma
In order to investigate the efficacy of rhLPHN3 Ab
treatment on the improvement of pediatric asthma, the therapeutic
effects in 100 patients using the SNOT-22 and LKES tests. The onset
frequency, duration of capillary bronchitis and asthma were
compared among patients receiving treatments of salbutamol,
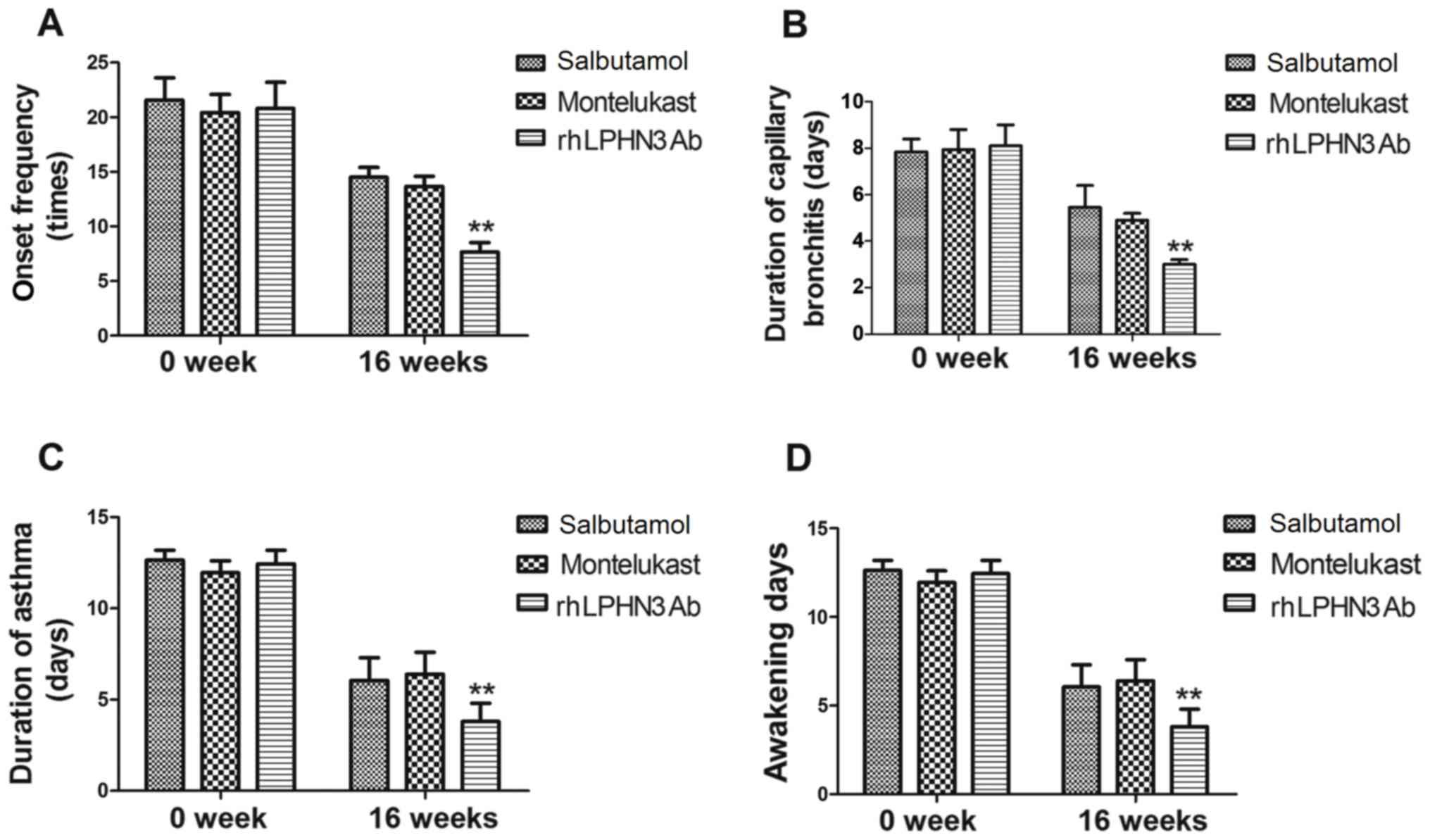
montelukast and rhLPHN3 Ab. As shown in Fig. 1A, the present study data demonstrated
that the onset frequency was significantly decreased in the three
groups, while the outcomes in the rhLPHN3 Ab group were better as
compared with the salbutamol and montelukast groups (P<0.01).
The results shown in Fig. 1B
revealed that the duration of capillary bronchitis was relieved and
63 patients completely recovered subsequent to rhLPHN3 Ab
treatment. Meanwhile, the duration of asthma was decreased
following treatment with salbutamol, montelukast and rhLPHN3 Ab
(Fig. 1C). It was also observed that
the number of days patients were awakened during the night was
evidently decreased subsequent to 16-week treatment in the three
groups (Fig. 1D). These data suggest
that rhLPHN3 Ab is more efficient compared with salbutamol and
montelukast for the treatment of pediatric asthma.
Analysis of inflammatory factors in
pediatric asthma patients following treatment with rhLPHN3 Ab
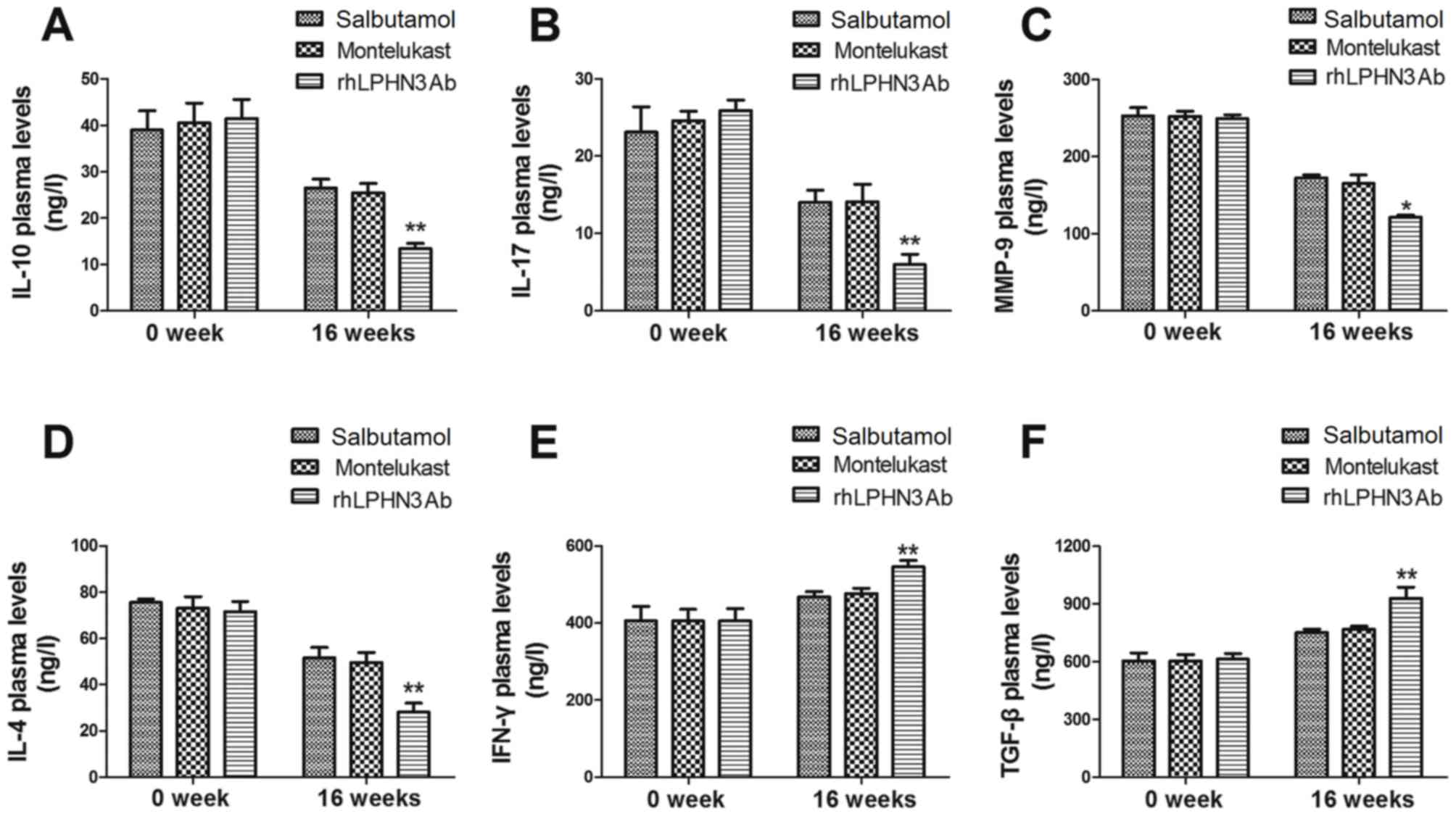
The levels of various inflammatory factors (IL-10,
IL-17, IL-4, MMP-9, IFN-γ and TGF-β) in the patients with pediatric
asthma were analyzed in the present clinical study. As shown in
Fig. 2A, the results demonstrated
that IL-10 concentration was upregulated following treatment with
salbutamol, montelukast and rhLPHN3 Ab.By contrast, the plasma
concentration of IL-17 was decreased in all treatment groups, while
IL-17 levels were significantly lower in the rhLPHN3 Ab group
compared with salbutamol and montelukast groups (P<0.01,
Fig. 2B). In addition, rhLPHN3 Ab
treatment downregulated MMP-9 plasma levels, which may contribute
to inhibition of the inflammatory response (Fig. 2C). It was also observed that IL-4
plasma concentration levels were significantly downregulated in
rhLPHN3 Ab group compared with the salbutamol and montelukast
groups (Fig. 2D). Furthermore, it
was observed that IFN-γ and TGF-β concentration levels were higher
in rhLPHN3 Ab group compared with the salbutamol and montelukast
groups (Fig. 2E and F). These
observations indicated that the inflammatory factor levels in the
children were improved following treatment with rhLPHN3 Ab.
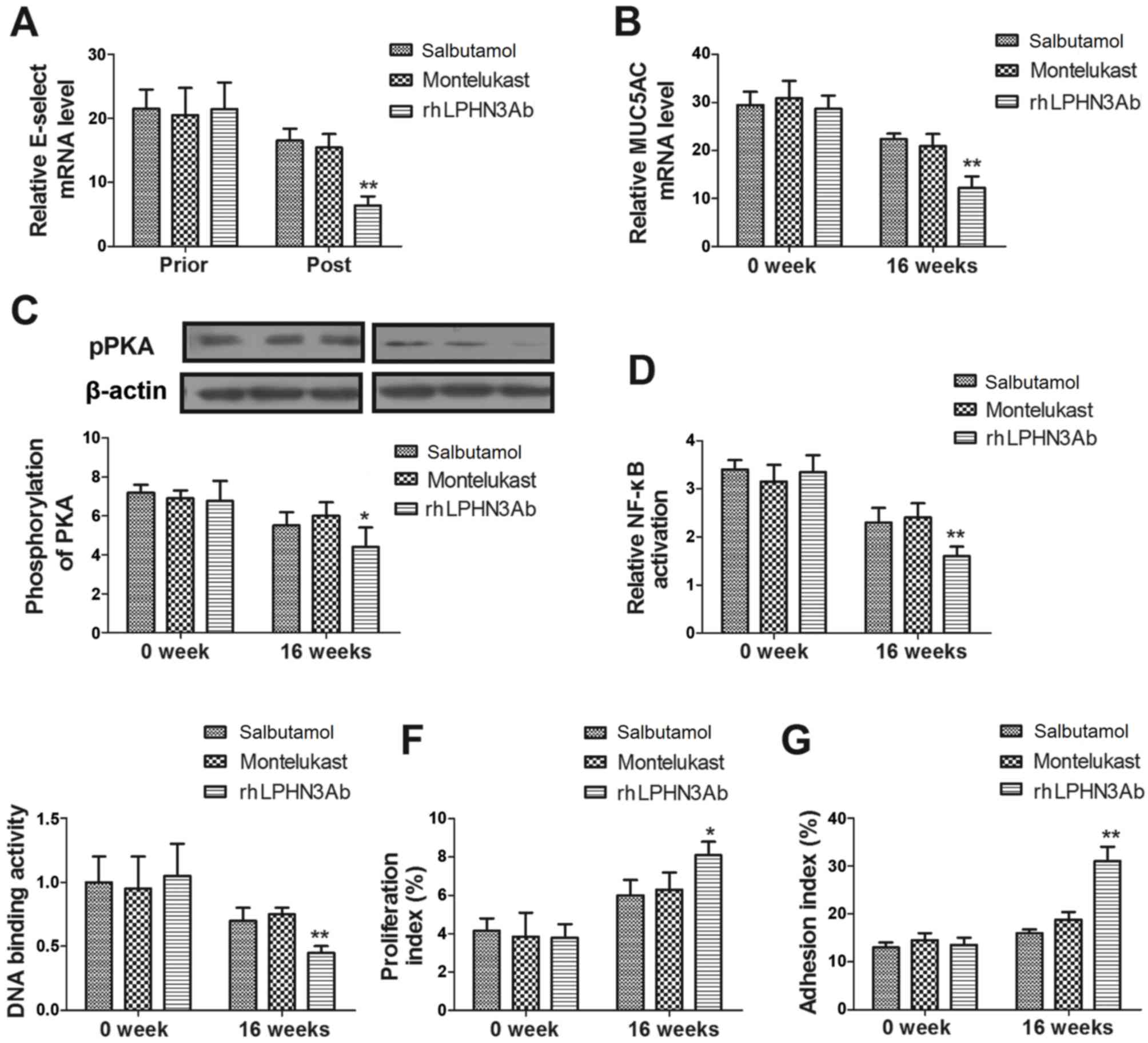
Analysis of potential mechanism
mediated by rhLPHN3 A bin airway smooth muscle cells
Previous studies have proposed numerous signaling
pathways that participate in the development and progression of
pediatric asthma (26,27). In the present study, the rhLPHN3
Ab-mediated signaling pathway in airway smooth muscle cells was
investigated. Initially, the E-selectin and MUC5AC expression
levels in these cells were investigated. As shown in Fig. 3A-B, rhLPHN3 Ab treatment
significantly decreased E-selectin and MUC5AC expression levels in
the airway smooth muscle cells as compared with the salbutamol and
montelukast groups. In addition, data revealed that rhLPHN3 Ab
treatment markedly reduced the phosphorylation of PKA in the airway
smooth muscle cells compared with the other treatment agents
(Fig. 3C). Furthermore, NF-κB
activation and DNA binding activity were decreased in the cells
following rhLPHN3 Ab treatment (Fig. 3D
and E). rhLPHN3Ab also markedly improved the adhesion and
proliferation of airway smooth muscle cells obtained from patients
with pediatric asthma (Fig. 3F and
G). These observations suggest that rhLPHN3 Ab treatment may
improve pediatric asthma via the PKA-induced NF-κB signaling
pathway.
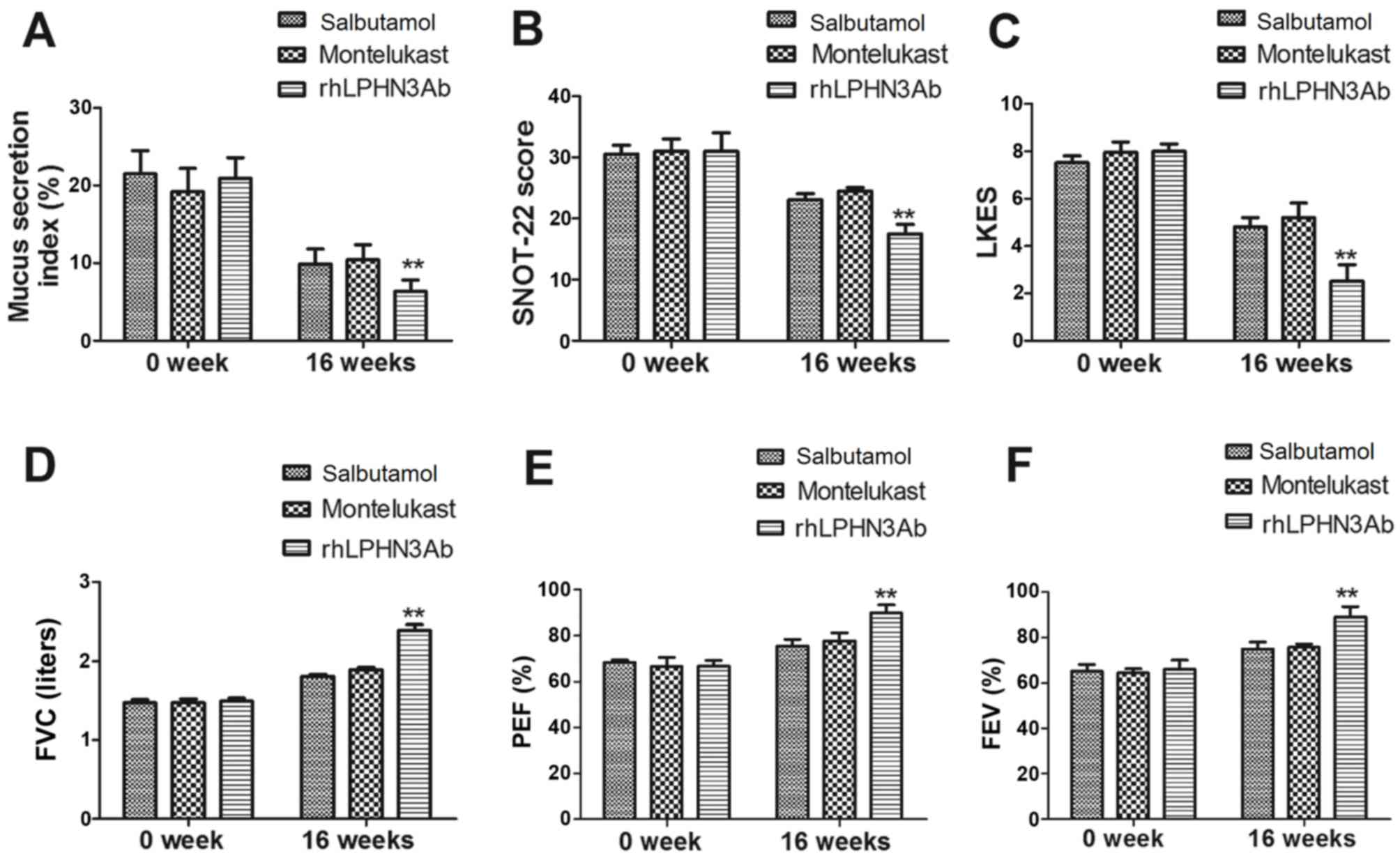
rhLPHN3Ab treatment improves the
function of airway smooth muscle cells and lung contraction
The current study further analyzed the morphologic
changes induced in airway smooth muscle cells after a 16-week
treatment. As shown in Fig. 4A,
rhLPHN3 Ab markedly inhibited airway mucus secretion when compared
with that in the salbutamol and montelukast groups. It was also
observed that the lung function of patients was improved following
treatment with rhLPHN3 Ab, as determined by SNOT-22 and LKES
testing (Fig. 4B and C). The results
further revealed that rhLPHN3 Ab treatment markedly improved the
contraction of the airways compared with the salbutamol and
montelukast groups, as demonstrated by the FVC, PEF and FEV values
(Fig. 4D-F). These results suggest
that rhLPHN3 Ab contributes to the improvement of pediatric asthma
by regulating the activity of the airway smooth muscle cells.
Discussion
According to previous studies, immune dysfunction
and abnormal expression of inflammation factors are closely
associated with capillary bronchial asthma, bronchitis and other
infant asthmatic diseases (28–30).
Notably, Li et al (31) have
demonstrated that the PKA-dependent NF-κB signaling pathway is a
novel target for drug action in asthma therapy. In addition,
evidence indicated that latrophilin receptors are associated with
heterogeneous behavioral disorder (32), which is also a novel target in asthma
(14,33). In the present study, the changes in
inflammation factor levels and the mechanism underlying the rhLPHN3
Ab-mediated signaling pathway were analyzed in patients with
pediatric asthma. The clinical outcomes revealed that rhLPHN3 Ab
treatment decreased the levels of plasma pro-inflammatory factors,
pulmonary pathological alterations, MUC5AC and E-selectin
expression levels and mucus hyper-secretion in patients with
pediatric asthma. In addition, data in the present analysis also
revealed that rhLPHN3 Ab significantly facilitated the
proliferation and adhesion of airway smooth muscle cells in
patients with pediatric asthma after 16-week treatment.
Furthermore, it has been suggested that rhLPHN3 Ab decreased
NF-κBlevels and improved the contraction of the airways through
regulation of the PKA-mediated NF-κB signaling pathway. These
findings indicate that rhLPHN3 Ab may be an efficient agent for the
treatment of pediatric asthma.
Currently, pediatric asthma presents the
characteristics of chronic inflammatory responses and systolic
dysfunction of the airway, in which marked changes in airway smooth
muscle cell contraction are observed. In a recent study, a
systematically review revealed the associations between
inflammatory responses and asthma severity in pediatric asthma,
which indicated that IFN-γ, IL-10 and IL-17 may serve as prognostic
indicators of pediatric asthma (34). In addition, various strategies
targeting inflammatory responses to improve airway remodeling in
asthma physiopathology have been investigated, and the results of
these studies support the hypothesis that modulating the allergic
inflammation improved asthma physiopathology in patients with
asthma in animals model and clinical trials (35–38).
Furthermore, previous reports have indicated that the pathogenesis
of airway inflammation in asthma patients may be associated with
the normal function and maintenance of the airway smooth muscle
cells (39–41). To investigate the therapeutic effects
of rhLPHN3 Ab in the treatment of pediatric asthma, the current
study analyzed the cytokine expression levels in the plasma of
patients. It was observed that rhLPHN3 Ab significantly inhibited
IL-17, MMP-9 and IL-10 levels, while it enhanced IL-4, IFN-γ and
TGF-β expression levels in the peripheral blood. This inhibitory
effect may contribute to the morphological changes of airway smooth
muscle cells. It was indicated that 16-week rhLPHN3 Ab treatment
improved BMI, SNOT-22, LKES, FVC, PEF and FEV.
Clinical manifestations of asthma include the first
onset of capillary bronchitis, typical bronchial asthma, and
chronic inflammation of the airway, which have been regarded as
evaluation criteria for patients with asthma (42–44). In
addition, studies have indicated that the number of times patients
were awakened by asthma during the night is also an important
factor that evaluates the efficacy of the childhood asthma
treatment (45,46). Furthermore, contraction of the
airways is another factor to evaluate the extent of capillary
bronchitis, which may be an early signal of bronchial asthma in
children (47,48). In the current study, the efficacy of
rhLPHN3 Ab on the onset frequency, duration of capillary
bronchitis, asthma and times awakened was investigated and compared
with the effects of salbutamol and montelukast serving as the
controls. The findings revealed that rhLPHN3 Ab evidently improved
the clinical manifestations in patients with pediatric asthma.
Notably, it has been reported that PKA-mediated
NF-κB signaling pathway may be associated with the progression of
pediatric asthma (49,50). In addition, previous studies
demonstrated that liraglutide possesses an anti-inflammatory
potential through inactivation of TNF-α expression and activation
of NF-κB signaling pathway in the progression of pediatric asthma
(51,52). Furthermore, a previous report
indicated that inhibition of NF-κB signaling pathway led to the
amelioration of inflammation and airway hyperresponsiveness in a
mouse asthma model (53). Similarly,
the current study observed that rhLPHN3 Ab inhibited the
inflammatory responses through PKA-mediated NF-κB signaling
pathway, which may contribute to the recovery of patients with
pediatric asthma.
In conclusion, the clinical outcomes of patients
included in the present study suggested that clinical treatment
with rhLPHN3 Ab exerted various beneficial effects on the secretion
of inflammatory mediators in pediatric asthma, which contributes to
the recovery of typical bronchial asthma, and chronic inflammation
of the airway in patients with pediatric asthma. These findings
contribute to better understand the immunologic mechanism
underlying the rhLPHN3 Ab-mediated treatment for pediatric
asthma.
References
|
1
|
Rubin BK: Asthma 2015: The year in review.
Respir Care. 61:556–559. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rodriguez-Villamizar LA, Berney C,
Villa-Roel C, Ospina MB, Osornio-Vargas A and Rowe BH: The role of
socioeconomic position as an effect-modifier of the association
between outdoor air pollution and children's asthma exacerbations:
An equity-focused systematic review. Rev Environ Health.
31:297–309. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rodrigo GJ and Plaza V: Once-daily
fluticasone furoate and vilanterol for adolescents and adults with
symptomatic asthma: A systematic review with meta-analysis. Ann
Allergy Asthma Immunol. 116:565–570. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Pols DH, Wartna JB, Moed H, van Alphen EI,
Bohnen AM and Bindels PJ: Atopic dermatitis, asthma and allergic
rhinitis in general practice and the open population: A systematic
Review. Scand J Prim Health Care. 34:143–150. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Paone G, Leone V, Conti V, De Marchis L,
Ialleni E, Graziani C, Salducci M, Ramaccia M and Munafò G: Blood
and sputum biomarkers in COPD and asthma: A review. European Rev
Med Pharmacol Sci. 20:698–708. 2016.
|
|
6
|
Nwaru BI, McCleary N, Erkkola M, Kaila M,
Virtanen SM and Sheikh A: Assisted reproductive technology and risk
of asthma and allergy in the offspring: Protocol for a systematic
review and meta-analysis. BMJ Open. 6:e0106972016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Oh S, Ji H, Barzman D, Lin PI and Hutton
J: Pediatric asthma and autism-genomic perspectives. Clin Transl
Med. 4:372015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wu DJ, Hipolito E, Bilderback A, Okelo SO
and Garro A: Predicting future emergency department visits and
hospitalizations for asthma using the pediatric asthma control and
communication instrument-emergency department version (PACCI-ED). J
Asthma. 53:387–391. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Doymaz S, Schneider J and Sagy M: Early
administration of terbutaline in severe pediatric asthma may reduce
incidence of acute respiratory failure. Ann Allergy Asthma Immunol.
112:207–210. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Myers TR: Pediatric asthma epidemiology:
Incidence, morbidity, and mortality. Respir Care Clin N Am. 6:1–14.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Doymaz S and Schneider J: Safety of
terbutaline for treatment of acute severe pediatric asthma. Pediatr
Emerg Care. Mar 8–2016.(Epub ahead of print). View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Booster GD, Oland AA and Bender BG:
Psychosocial factors in severe pediatric asthma. Immunol Allergy
Clin North Am. 36:449–460. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Idris IB, Ghazi HF, Zhie KH, Khairuman KA,
Yahya SK, Abd Zaim FA, Nam CW, Rasid HZ Abdul and Isa ZM:
Environmental air pollutants as risk factors for asthma among
children seen in pediatric clinics in UKMMC, Kualalumpur. Annals
Glob Health. 82:202–208. 2016. View Article : Google Scholar
|
|
14
|
Faiz A, Donovan C, Nieuwenhuis MA, van den
Berge M, Postma DS, Yao S, Park CY, Hirsch R, Fredberg JJ, Tjin G,
et al: Latrophilin receptors: Novel bronchodilator targets in
asthma. Thorax. 72:84–82. 2017. View Article : Google Scholar
|
|
15
|
Volynski KE, Silva JP, Lelianova VG,
Rahman M Atiqur, Hopkins C and Ushkaryov YA: Latrophilin fragments
behave as independent proteins that associate and signal on binding
of LTX(N4C). EMBO J. 23:4423–4433. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gao GX, Li QM and Shen HH: Effect of
astragali-cordyceps mixtura on TGF-beta/Smad signal pathway in the
lung of asthma airway remodeling. J Ethnopharmacol. 125:68–74.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Li CC, Lin L, Wang XL, Guan XJ, Su MS,
Xiang QW, Han H, Zhang WX and Li MR: Role of c-Jun N-terminal
kinase signal transduction pathway in the course of airway
remodeling of asthma rat. Zhonghua er ke za zhi. Zhonghua Er Ke Za
Zhi. 46:535–539. 2008.(In Chinese).
|
|
18
|
Xie M, Liu XS, Xu YJ, Zhang ZX, Bai J, Ni
W and Chen SX: Role of the extracellular signal-regulated kinase
1/2 signaling pathway in regulating the secretion of bronchial
smooth muscle cells in a rat model of chronic asthma. Chin Med J
(Engl). 121:73–77. 2008.PubMed/NCBI
|
|
19
|
Caminha GP, Junior JT Melo, Hopkins C,
Pizzichini E and Pizzichini MM: SNOT-22: Psychometric properties
and cross-cultural adaptation into the Portuguese language spoken
in Brazil. Braz J Otorhinolaryngol. 78:34–39. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) methods. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Janockova J, Zilecka E, Kasparkova J,
Brabec V, Soukup O, Kuca K and Kozurkova M: Assessment of
DNA-binding affinity of cholinesterase reactivators and
electrophoretic determination of their effect on topoisomerase I
and II activity. Mol Biosyst. 12:2910–2920. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Rudmik L, Soler ZM, Mace JC, DeConde AS,
Schlosser RJ and Smith TL: Using preoperative SNOT-22 score to
inform patient decision for Endoscopic sinus surgery. Laryngoscope.
125:1517–1522. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Huang J, Chen Y, Long Z, Zhou X and Shu J:
Clinical efficacy of tiotropium in children with asthma. Pak J Med
Sci. 32:462–465. 2016.PubMed/NCBI
|
|
24
|
Diaz I and Mateu E: Use of ELISPOT and
ELISA to evaluate IFN-gamma, IL-10 and IL-4 responses in
conventional pigs. Vet Immunol Immunopathol. 106:107–112. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Mehravi B, Alizadeh AM, Khodayari S,
Khodayari H, Ashtari K, Mohseni M, Anaraki NI, Dana EA, Safari S
and Amanlou M: Acute toxicity evaluation of glycosylated Gd3+-based
silica nanoprobe. Mol Imaging Biol. 19:522–530. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kim SP, Lee SJ, Nam SH and Friedman M: Elm
Tree (Ulmus parvifolia) Bark Bioprocessed with Mycelia of Shiitake
(Lentinus edodes) Mushrooms in Liquid Culture: Composition and
mechanism of protection against allergic asthma in mice. J Agric
Food Chem. 64:773–784. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Bardin P, Kanniess F, Gauvreau G,
Bredenbroker D and Rabe KF: Roflumilast for asthma: Efficacy
findings in mechanism of action studies. Pulm Pharmacol Ther. 35
Suppl:S4–S10. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Johnathan M, Gan SH, Ezumi MF, Faezahtul
AH and Nurul AA: Phytochemical profiles and inhibitory effects of
Tiger Milk mushroom (Lignosus rhinocerus) extract on
ovalbumin-induced airway inflammation in a rodent model of asthma.
BMC Complement Altern Med. 16:1672016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
de Araujo CC, Marques PS, Silva JD, Samary
CS, da Silva AL, Henriques I, Antunes MA, de Oliveira MV,
Goldenberg RC, Morales MM, et al: Regular and moderate aerobic
training before allergic asthma induction reduces lung inflammation
and remodeling. Scand J Med Sci Sports. 26:1360–1372. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Phillips JE, Renteria L, Burns L, Harris
P, Peng R, Bauer CM, Laine D and Stevenson CS: Btk inhibitor RN983
delivered by dry powder nose-only aerosol inhalation inhibits
bronchoconstriction and pulmonary inflammation in the ovalbumin
allergic mouse model of asthma. J Aerosol Med Pulm Drug Deliv.
29:233–241. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Li Z, Zheng J, Zhang N and Li C: Berberine
improves airway inflammation and inhibits NF-κB signaling pathway
in an ovalbumin-induced rat model of asthma. J Asthma. 53:999–1005.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Choudhry Z, Sengupta SM, Grizenko N,
Fortier ME, Thakur GA, Bellingham J and Joober R: LPHN3 and
attention-deficit/hyperactivity disorder: Interaction with maternal
stress during pregnancy. J Child Psychol Psychiatry. 53:892–902.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tobaben S, Sudhof TC and Stahl B: Genetic
analysis of alpha-latrotoxin receptors reveals functional
interdependence of CIRL/latrophilin 1 and neurexin 1 alpha. J Biol
Chem. 277:6359–6365. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Vanders RL, Gibson PG, Wark PA and Murphy
VE: Alterations in inflammatory, antiviral and regulatory cytokine
responses in peripheral blood mononuclear cells from pregnant women
with asthma. Respirology. 18:827–833. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Mauffray M, Domingues O, Hentges F, Zimmer
J, Hanau D and Michel T: Neurturin influences inflammatory
responses and airway remodeling in different mouse asthma models. J
Immunol. 194:1423–1433. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Vanoirbeek JA, De Vooght V, Nemery B and
Hoet PH: Multiple challenges in a mouse model of chemical-induced
asthma lead to tolerance: Ventilatory and inflammatory responses
are blunted, immunologic humoral responses are not. Toxicology.
257:144–152. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lim Y, Vasu VT, Valacchi G, Leonard S,
Aung HH, Schock BC, Kenyon NJ, Li CS, Traber MG and Cross CE:
Severe vitamin E deficiency modulates airway allergic inflammatory
responses in the murine asthma model. Free Radical Res. 42:387–396.
2008. View Article : Google Scholar
|
|
38
|
Bhandari V, Choo-Wing R, Chapoval SP, Lee
CG, Tang C, Kim YK, Ma B, Baluk P, Lin MI and McDonald DM:
Essential role of nitric oxide in VEGF-induced, asthma-like
angiogenic, inflammatory, mucus, and physiologic responses in the
lung. Proc Natl Acad Sci USA. 103:pp. 11021–11026. 2006; View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Gaga M, Zervas E and Loukides S: The
usefulness of inflammatory markers in monitoring treatment
responses in asthma. Clin Exp Allergy. 33:855–858. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Boulay ME and Boulet LP: Lower airway
inflammatory responses to repeated very-low-dose allergen challenge
in allergic rhinitis and asthma. Clin Exp Allergy. 32:1441–1447.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Foster PS, Ming Y, Matthei KI, Young IG,
Temelkovski J and Kumar RK: Dissociation of inflammatory and
epithelial responses in a murine model of chronic asthma. Lab
Invest. 80:655–662. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Provotorov VM, Budnevsky AV and Filatova
YI: Clinical manifestations of asthma during combination therapy
using ceruloplasmin. Ter Arkh. 88:36–39. 2016.(In Russian).
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Hartmann K, Escribano L, Grattan C,
Brockow K, Carter MC, Alvarez-Twose I, Matito A, Broesby-Olsen S,
Siebenhaar F, Lange M, et al: Cutaneous manifestations in patients
with mastocytosis: Consensus report of the European Competence
Network on Mastocytosis; the American Academy of Allergy, Asthma
& Immunology; and the European Academy of Allergology and
Clinical Immunology. J Allergy Clin Immunol. 137:35–45. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Ghaffari J, Hossiani R Farid, Khalilian A,
Nahanmoghadam N, Salehifar E and Rafatpanah H: Vitamin e
supplementation, lung functions and clinical manifestations in
children with moderate asthma: A randomized double blind
placebo-controlled trial. Iran J Allergy Asthma Immunol. 13:98–103.
2014.PubMed/NCBI
|
|
45
|
García-Martín E, García-Menaya J, Sánchez
B, Martínez C, Rosendo R and Agúndez JA: Polymorphisms of
histamine-metabolizing enzymes and clinical manifestations of
asthma and allergic rhinitis. Clin Exp Allergy. 37:1175–1182. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Kim TB, Kim YK, Chang YS, Kim SH, Hong SC,
Jee YK, Cho SH, Min KU and Kim YY: Association between
sensitization to outdoor spider mites and clinical manifestations
of asthma and rhinitis in the general population of adults. J
Korean Med Sci. 21:247–252. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Araujo MI and de Carvalho EM: Human
schistosomiasis decreases immune responses to allergens and
clinical manifestations of asthma. Chem Immunol Allergy. 90:29–44.
2006.PubMed/NCBI
|
|
48
|
Khamidova ZN: Clinical manifestations and
a course of bronchial asthma complicated by gastroduodenal
pathology. Probl Tuberk. 44–45. 2002.(In Russian). PubMed/NCBI
|
|
49
|
Zhou Y, Wang GF, Yang L, Liu F, Kang JQ,
Wang RL, Gu W and Wang CY: Treatment with 1,25(OH)2D3 induced HDAC2
expression and reduced NF-κB p65 expression in a rat model of
OVA-induced asthma. Braz J Med Biol Res. 48:654–664. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Pan C and Dong Z: Antiasthmatic Effects of
Eugenol in a Mouse Model of Allergic Asthma by Regulation of
Vitamin D3 Upregulated Protein 1/NF-κB Pathway. Inflammation.
38:1385–1393. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Garcia-Garcia FJ, Mullol J, Perez-Gonzalez
M, Pujols L, Alobid I, Roca-Ferrer J and Picado C: Signal
transduction pathways (MAPKs, NF-κB, and C/EBP) regulating COX-2
expression in nasal fibroblasts from asthma patients with aspirin
intolerance. PLoS One. 7:e512812012. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Shimizu K, Konno S, Ozaki M, Umezawa K,
Yamashita K, Todo S and Nishimura M: Dehydroxymethylepoxyquinomicin
(DHMEQ), a novel NF-kappaB inhibitor, inhibits allergic
inflammation and airway remodelling in murine models of asthma.
Clin Exp Allergy. 42:1273–1281. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Lim JC, Goh FY, Sagineedu SR, Yong AC,
Sidik SM, Lajis NH, Wong WS and Stanslas J: A semisynthetic
diterpenoid lactone inhibits NF-κB signalling to ameliorate
inflammation and airway hyperresponsiveness in a mouse asthma
model. Toxicol Appl Pharmacol. 302:10–22. 2016. View Article : Google Scholar : PubMed/NCBI
|