Introduction
T regulatory (Treg) cells are important suppressive
immune regulatory cells that exert their immunosuppressive effects
by producing cytokines with inhibitory functions and have an
important role in the maintenance of immune homeostasis (1). Treg cells facilitate immune evasion via
two mechanisms: Immune anergy and immunosuppression; they reduce
the recognition of tumor antigens by the organism, induce immune
tolerance of tumor antigens associated with immune anergy and
negatively regulate the anti-tumor immune response associated with
immunosuppression, resulting in immune escape of malignancies
(2). Treg cells account for 5–10% of
CD4+ T cells and may be divided into natural Treg
(nTreg) cells and inducible Treg (iTreg) cells (3). nTreg mainly refers to
CD4+CD25+ Treg cells and iTreg refers to
transforming growth factor (TGF)-β-secreting T helper lymphocyte 3
(Th3) and IL-10-secreting type 1 regulatory T (Tr1) cells (4–6).
Furthermore, immunosuppressive CD8+ Treg and natural
killer T cells, which exhibit bidirectional immunity, represent two
categories of Tregs (7,8). Previous studies on Treg cells mainly
focused on CD4+CD25+ Treg, Th3 and Tr1 cells.
In recent years, a novel subset of Treg cells with an
immunosuppressive function was identified based on the presence of
cell surface marker latency-associated peptide (LAP), which were
therefore designated as LAP+CD4+ T cells
(9). Certain animal studies indicate
that LAP+CD4+ T cells participate in the
initiation and development of several autoimmune and inflammatory
diseases (10,11). In clinical practice, a close
association between LAP+CD4+ T cells and
tumors has been indicated (12).
However, to date, the distribution of
LAP+CD4+ T cells in hepatocellular carcinoma
(HCC) has remained to be determined.
In the present study, flow cytometric (FCM) analysis
was performed to determine the proportion of
LAP+CD4+ T cells among peripheral blood
mononuclear cells (PBMCs) and changes of
LAP+CD4+ T cell levels in the peripheral
blood after curative resection of HCC. Furthermore,
immunohistochemical staining with double enzyme labeling was
applied to determine the abundance of
LAP+CD4+ T cells in HBV-infected HCC tissue,
peri-cancer tissue and HBV-infected hepatic tissues around benign
lesions. The differences in the distribution characteristics of
LAP+CD4+ T cells in HCC tissue, peri-cancer
tissue and HBV-infected hepatic tissues around benign lesions were
analyzed. The function of LAP+CD4+ T cells in
the local immune microenvironment of HCC and the expression
characteristics of LAP+CD4+ T cells among
PBMCs were also analyzed.
Materials and methods
Clinical information
The pre-operative group included 30 patients with
HCC who were preliminarily diagnosed at the department of
Hepatobiliary and Laparoscopic Surgery, Shenzhen Hospital (Peking
University, Shenzhen, China) between October 2011 and December
2012, including 17 males and 13 females at the age of 42–60 years
(median age, 51.4 years). Patients exhibited a chronic hepatitis B
virus (HBV) infection and did not receive any treatment (including
transcatheter hepatic arterial chemoembolization) prior to surgery.
They were diagnosed with HCC after post-operative pathological
examination. The post-operative group comprised 28 patients with
HCC from the pre-operative group (two patients were unable to
undergo the surgery due to metastasis in other organs), including
16 males and 12 females (age range, 42–60 years; median age, 52.1
years). The peripheral blood control group included 30 HBV-carrying
volunteers (16 males, 14 female; age range, 39–57 years; median
age, 48.3 years) who presented at the aforementioned hospital with
chronic cholecystitis, inguinal hernia and chronic appendicitis
between October 2011 and December 2012. The histopathology control
group contained 28 HBV-carrying patients who received hepatic
resection due to benign lesions, including 16 males and 12 females
(age range, 35–52 years; median age, 45.7 years). None of the
patients included in the present study had diabetes,
hyperthyroidism or any autoimmune diseases. The differences in
gender and age between the HCC pre or post-operative group and the
peripheral blood control group, and the differences between the HCC
or peri-cancer group and the hepatic tissue control group were not
statistically significant.
Sample collection
The blood specimens were collected using disposable
vacuum blood collection tubes. Each sample contained 6 ml venous
blood from the subject that was drawn in the morning after the
subject had fasted. All specimens were processed within 4 h
following collection and analyzed within 16 h. The blood samples
were collected three days prior to the operation for the
pre-operative group and 10 days after the operation for the
post-operative group.
Fresh hepatic tissue was collected from patients
after hepatectomy, fixed in formalin, embedded in conventional
paraffin and continuously cut into 4-µm sections. Hepatic tumor
samples and peri-cancer tissue samples (collected ≤2 cm away from
the carcinoma nodule) were collected separately from patients with
HCC, avoiding the necrotic region, and assigned to the HCC group
and the hepatocellular peri-cancer group. For the control group,
HBV-infected hepatic tissues around benign lesions were collected
following hepatectomy due to benign lesions.
Analysis of
LAP+CD4+ T cells in PBMCs by FCM
analysis
The following major reagents and instruments were
used: CD4-fluorescein isothiocyanate (FITC) monoclonal antibody
(mAb; cat. no. 553650) and negative control group immunoglobulin
(IgG)2α-FITC antibody (cat. no. 550056) for fluorescence staining
were purchased from BD Biosciences (Franklin Lakes, NJ, USA). Flow
Cytometry Staining Buffer (1×; cat. no. FC001), monoclonal
LAP-phycoerythrin (PE) antibody (cat. no. 246-LP-025) and the
negative negative control group IgG1-PE (cat. no. FAB110P) were
purchased from R&D Systems (Minneapolis, MN, USA). Lymphocyte
separation medium (cat. no. B6385-10G) was provided by
Sigma-Aldrich (Merck KGaA, Darmstadt, Germany). Prior to
experimentation, the flow cytometer was calibrated to recognize
positively stained cells using FITC calibration particles (5 peaks;
BD Biosciences, Franklin Lakes, NJ, USA; cat. no. 559123). A
four-color flow cytometer (Beckmann-Coulter, Brea, CA, USA) was
used for FCM analysis.
The blood sample (3 ml) was diluted with an equal
volume of PBS. The lymphocytes were isolated by mixing 3 ml sample
with lymphocyte separation medium, followed by centrifugation at a
speed of 500 × g for 20 min at 25°C. The cells were re-suspended
with 200 µl buffer and 100 µl of this lymphocyte suspension was
added to Tube A (experimental tube) and Tube B (isotype tube),
followed by addition of monoclonal CD4-FITC antibodies (1:5,000) to
each tube. The samples were incubated for 20 min at 25°C in the
dark, fixed with 500 µl of 4% paraformaldehyde for 10 min at 25°C
and washed twice with PBS. LAP-PE mAb (10 µl; 1:5,000) was added to
Tube A and isotype control IgG1-PE (10 µl; 1:5,000) was added to
Tube B. The samples were incubated for 45 min at 25°C in the dark.
After one wash with PBS, the lymphocytes were re-suspended in 500
µl PBS and analyzed with the four-color flow cytometer. At least
50,000 lymphocytes were collected for each acquisition. Flow jo
software (flowjo 7.6.1; BD Biosciences) was used to analyze the
results.
Analysis of
LAP+CD4+ T cells in HCC tissue by
immunohistochemical staining with double enzyme labeling
As the major reagents, goat anti-human LAP
polyclonal antibody (cat. no. AF-246-NA) was purchased from R &
D Systems and was diluted at 1:100 for use furthermore, ready-to
use rabbit anti-human CD4 mAb (cat. no. YB-0766R), enzyme-labeled
rabbit anti-goat IgG (cat. no. EHJSW-135238),
streptavidin-peroxidase (cat. no. SP KIT-D1),
3-amino-9-ethylcarbazole (AEC) color reagent kit (cat. no.
HPBIO-JC583), and diaminobenzidine (DAB) color reagent kit (cat.
no. YYJK-1009) were purchased from Fuzhou Maixin (Fuzhou, Fujian,
China).
The tissue sections were de-waxed, incubated with
pepsin (1:10,000; cat. no. P7012-250MG; Sigma-Aldrich; Merck KGaA)
in a 37°C incubator for 25 min, washed with PBS and then soaked in
3% H2O2 for 20 min at room temperature.
Samples were further washed with PBS twice for 5 min, to discard
the H2O2 solution and then 10% fetal calf
serum (cat. no. KL-P0015; Sigma-Aldrich; Merck KGaA) was added and
the samples were incubated in a 37°C incubator for 30 min. Ready-to
use rabbit anti-human CD4 monoclonal antibody (1:100) was added and
the samples were incubated at 4°C overnight. After being washed
with PBS, the samples were incubated with enzyme-labeled rabbit
anti-goat IgG (1:100) in a 37°C incubator for 30 min. After washing
with PBS, streptavidin-peroxidase was added and the samples were
incubated in a 37°C incubator for 30 min. The samples were then
washed with PBS, followed by incubation coloration with DAB for 4
min at 25°C and soaking in 3% H2O2 for 20 min
at room temperature. After another wash with PBS, the samples were
incubated with 10% fetal calf serum in a 37°C incubator for 30 min.
Subsequently, the samples were incubated with goat anti-human LAP
polyclonal antibody at a dilution of 1:100 at 4°C overnight. The
samples were washed with PBS and incubated with enzyme-labeled
rabbit anti-goat IgG (1:100) in a 37°C incubator for 30 min. After
another wash with PBS, the samples were incubated with
streptavidin-peroxidase in a 37°C incubator for 30 min. After being
washed with PBS, the samples were incubated with AEC for 4 min to
visualize the antibodies. Subsequently, the samples were
dehydrated, cleared, sealed and examined under a microscope.
LAP+CD4+ T cells were
identified as follows: Complete cellular morphology, clear
structure, brown/yellow and purple/red granules specifically
co-localized in the cytoplasm and on cell the membrane (brown
granules). CD4+ T cells were identified as follows:
Complete morphology, clear structure, brown/yellow granules
specifically located in the cytoplasm and on the cell membrane. The
slides were observed under a microscope by an observer blinded to
the sample identity. First, the whole slide was observed at a low
magnification. Subsequently, 5 high-power fields (magnification,
×400) were randomly selected for quantification of stained cells
and calculation of the mean value.
Statistical analysis
Values are expressed as the mean ± standard
deviation of the percentage. A paired Student's t-test was used for
comparisons between HCC and corresponding peri-cancer samples, as
well as between HCC pre-operative peripheral blood samples and
their corresponding post-operative samples. Two-way analysis of
variance was performed for multi-group comparisons involving the
control and the HCC groups, and Tukey's post-hoc test was used for
comparisons between the control and the HCC groups (peripheral
blood control group vs. pre/post-operative group for FCM results
and hepatic tissue control group vs. HCC/peri-cancer group for
immunohistochemistry results). SPSS 17.0 software (SPSS, Inc.,
Chicago, IL, USA) was used for data processing. P<0.05 was
considered to indicate a statistically significant difference.
Results
LAP+CD4+T cells
are increased in the PBMCs of patients with HCC, and are reduced
after surgical resection
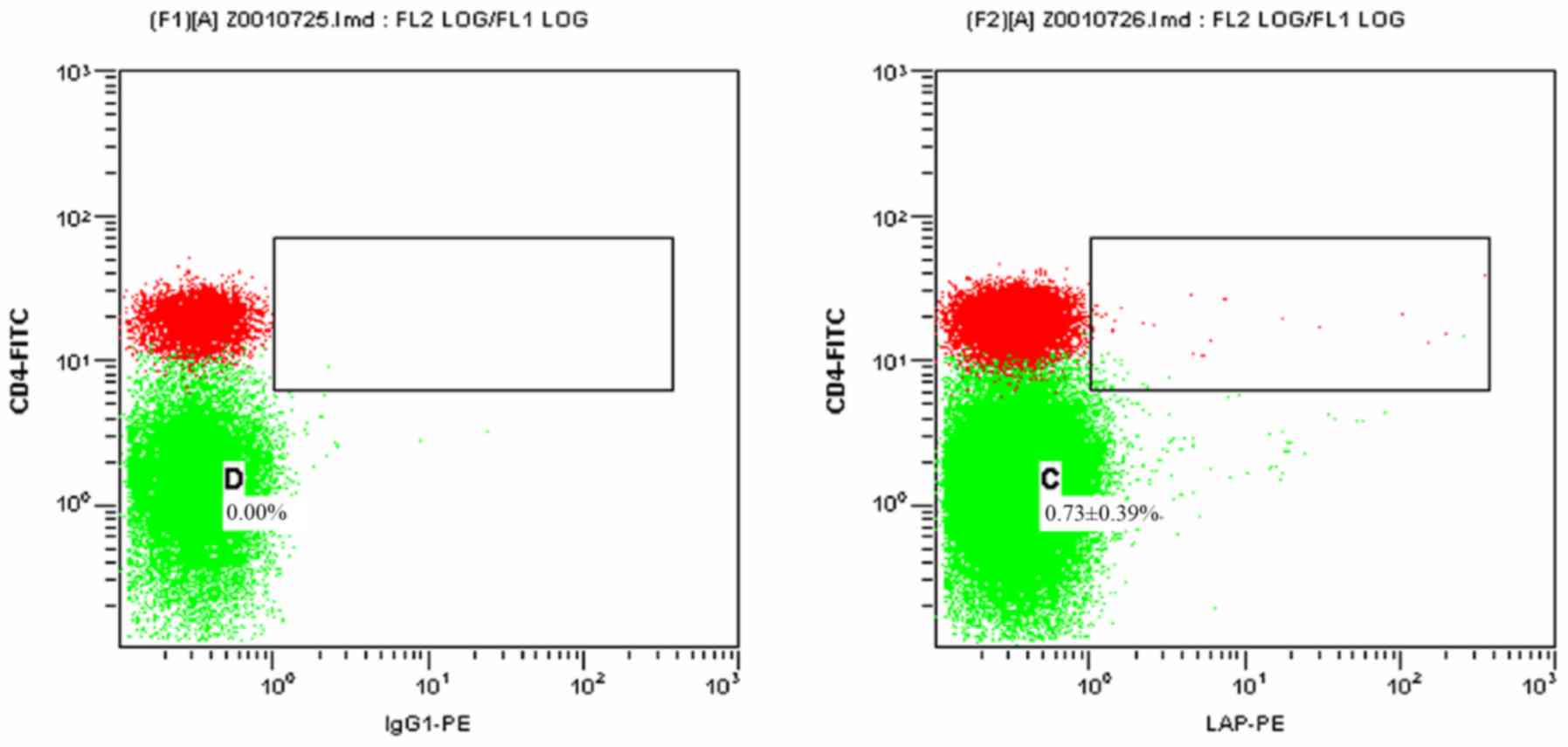
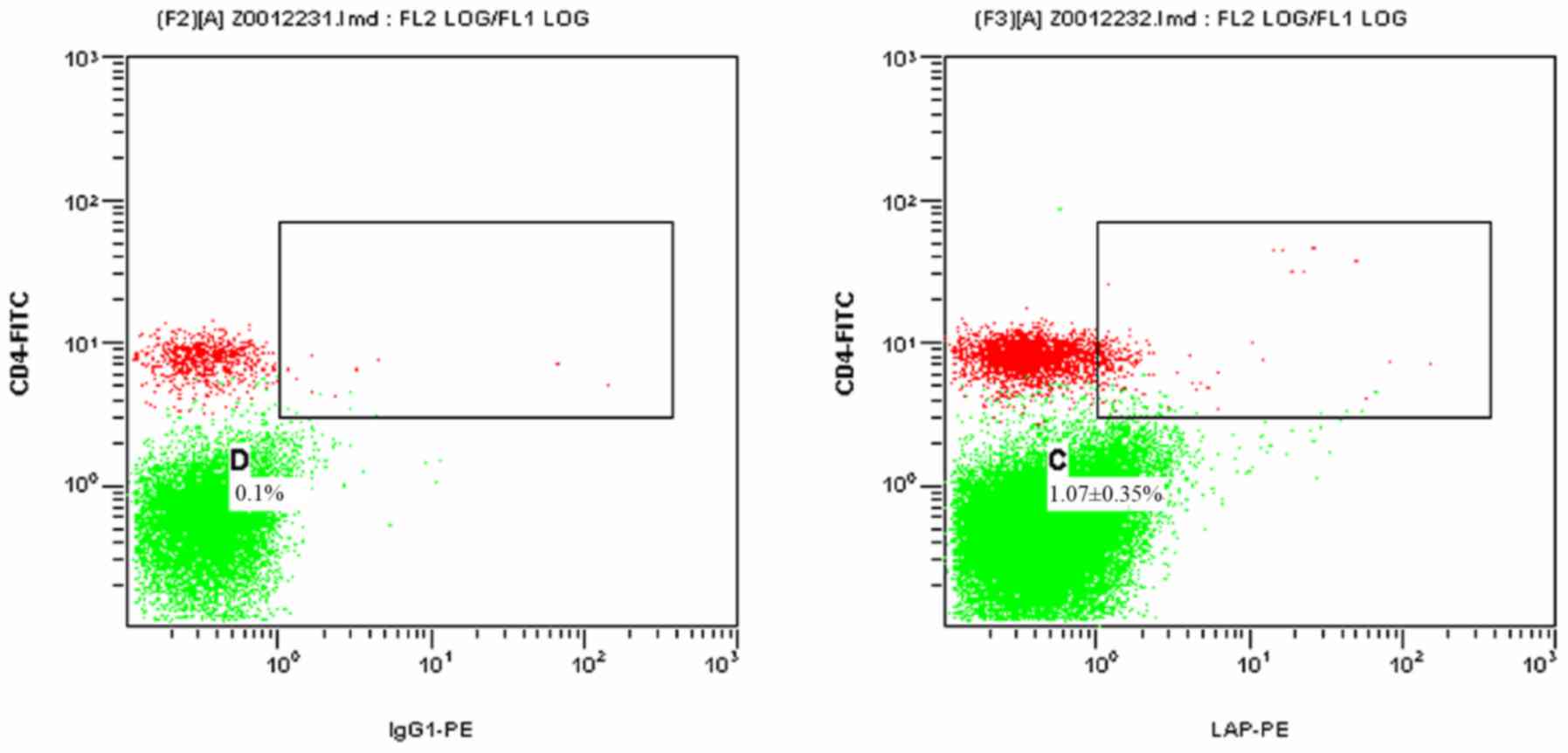
Figs. 1 and 2 present scatter plots of
LAP+CD4+ T cells in PBMCs from the control
group and the HCC pre-operative group, respectively, detected by
FCM. As indicated in Table I, the
proportion of LAP+CD4+ T cells among the
PBMCs was 0.73±0.39% in the control group and 1.84±0.85% in the HCC
pre-operative group. The proportion of
LAP+CD4+ T cells in the PBMCs of the HCC
pre-operative group was significantly higher than that in the
control group (P=0.019).
 | Table I.Proportion of
LAP+CD4+ T cells among the peripheral blood
mononuclear cells of the control group as well as the HCC
pre-operative and post-operative groups. |
Table I.
Proportion of
LAP+CD4+ T cells among the peripheral blood
mononuclear cells of the control group as well as the HCC
pre-operative and post-operative groups.
| Group | N | CD4+
cells (%) |
LAP+CD4+ T cells
(%) |
|---|
| Control | 30 | 32.11±5.12 | 0.73±0.39 |
| HCC
pre-operative | 30 | 21.91±6.62 |
1.84±0.85a |
| HCC
post-operative | 28 | 29.58±6.24 |
1.07±0.35b |
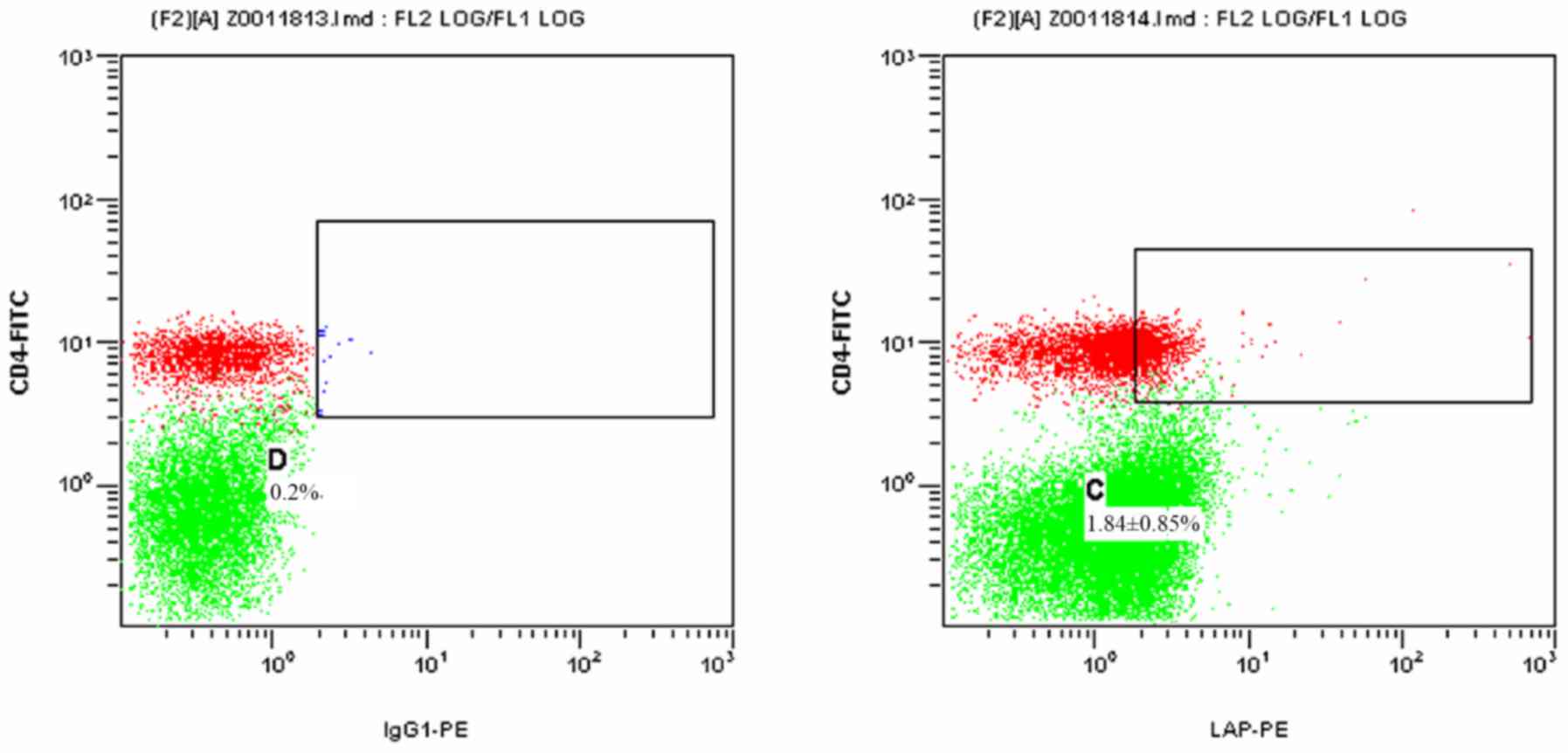
Fig. 3 presents a
2-dimensional scatter plot displaying
LAP+CD4+ T cells among PBMCs from the HCC
post-operative group detected by FCM. As displayed in Table I, the proportion of
LAP+CD4+ T cells among the PBMCs was
1.07±0.35% in the HCC post-operative group, which was significantly
declined compared with that in the HCC pre-operative group
(1.84±0.85; P=0.021), while it remained slightly, but not
significantly higher than that in the control group (0.73±0.39%;
P=0.342).
Histopathological analysis of
LAP+CD4+ T cells in HCC tissues
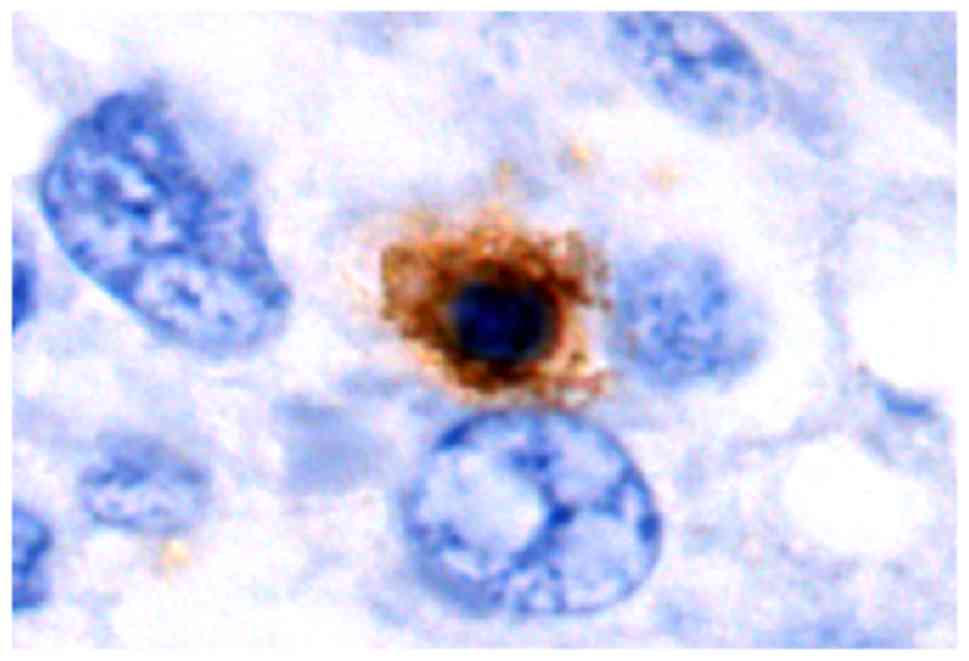
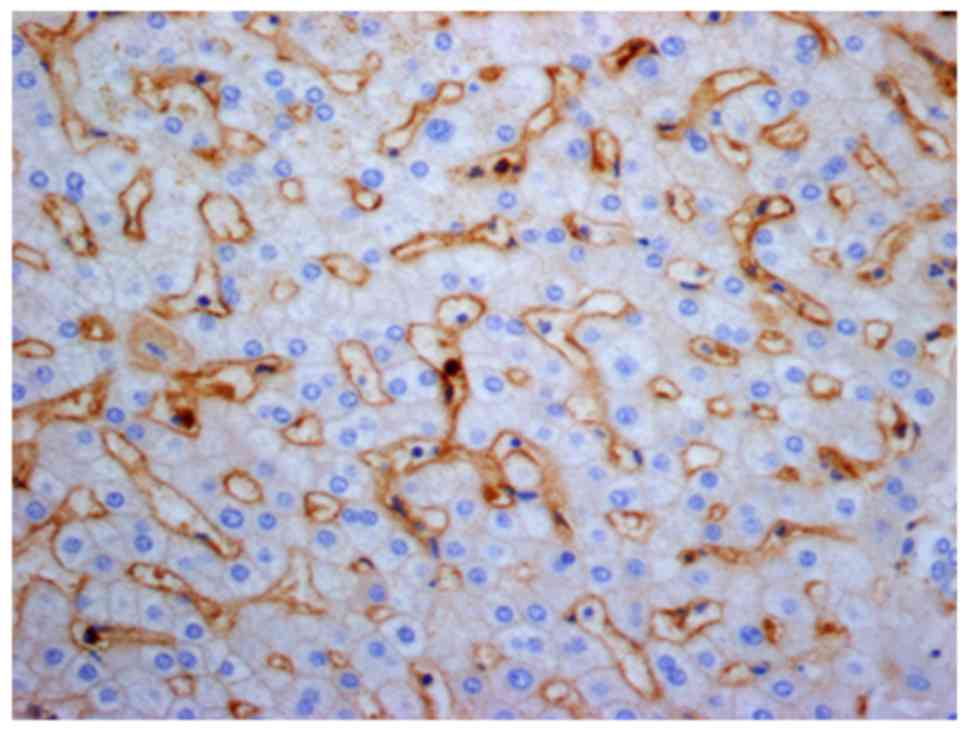
As exemplified in the representative image in
Fig. 4, LAP was localized in the
cytoplasm and on the cell membrane of CD4+ T cells in
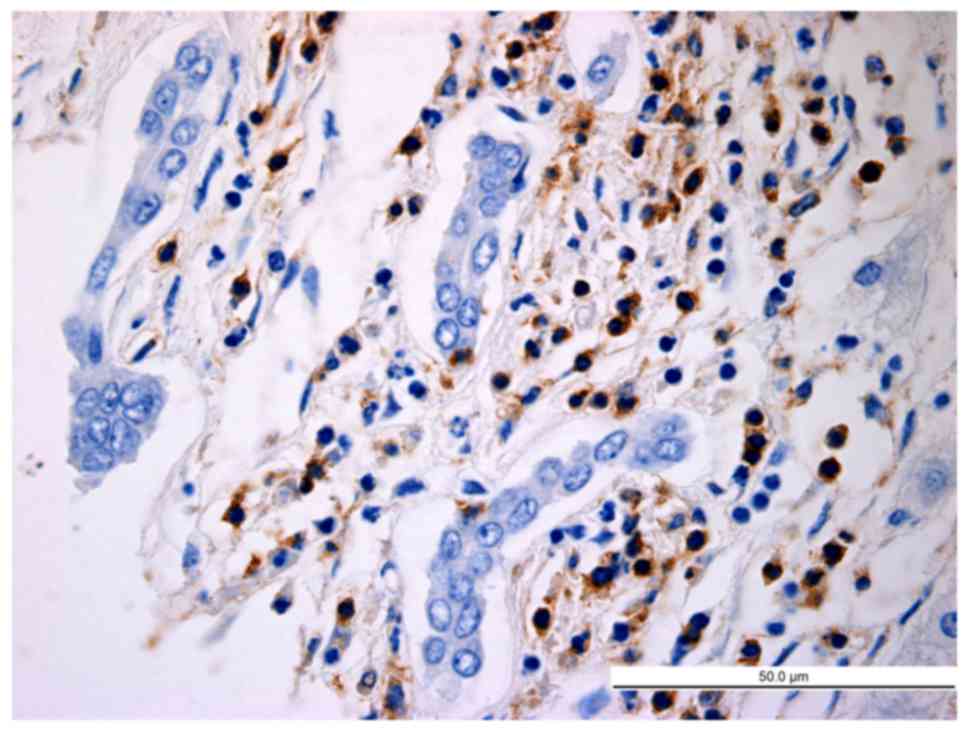
HCC tissues. In normal hepatic tissues and peri-cancer tissue,
LAP+CD4+ T cells were scattered, but were
mostly clustered in tumor stroma and located close to other
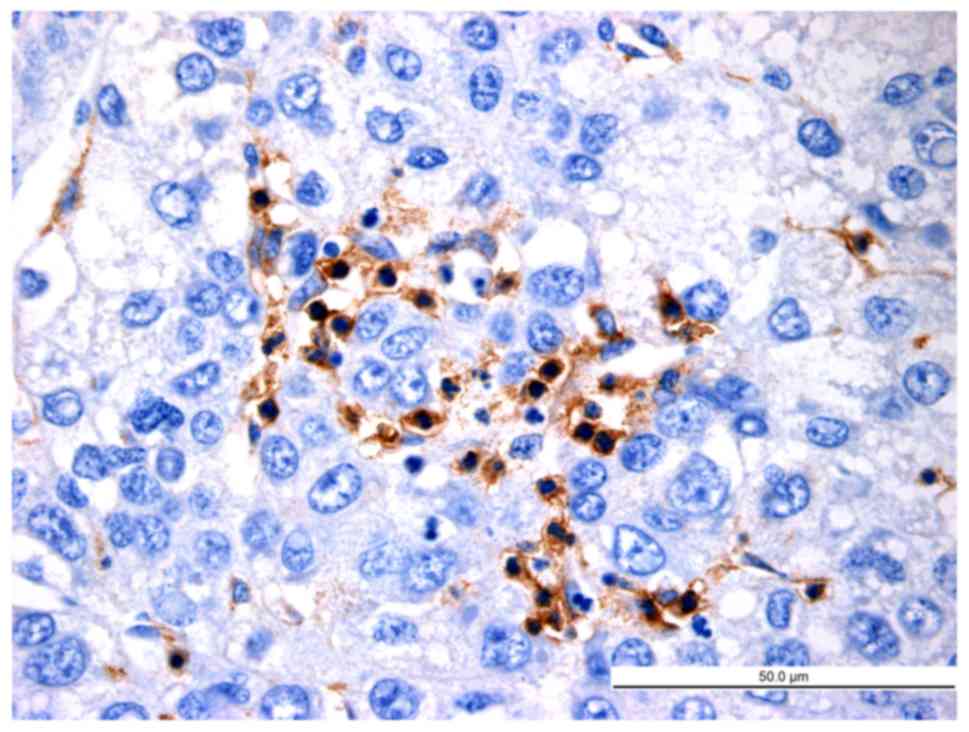
lymphocytes (Figs. 5–7). Of note, a large number of
LAP+CD4+ T cells were observed in HCC nests
(cancer nests have a clear boundary and are primarily composed of
heavily stained basal cells. Cancer cells are also well
differentiated and nuclear fission is rare) and they were in close
contact with CD4+ T cells (Fig. 8).
As presented in Table
II, the average number of LAP+CD4+ T
cells per high-power field in the HCC group, peri-cancer group and
normal control group was 11.25±3.00, 5.75±1.00 and 2.61±0.83,
respectively. The difference in the abundance of
LAP+CD4+ T cells was significant between the
HCC group and the control group (P=0.013), between the peri-cancer
group and the control group (P=0.025), and between the peri-cancer
group and the HCC group (P=0.018).
 | Table II.Distribution of
LAP+CD4+ T cells in HCC, peri-cancer and
control HBV-infected hepatic tissues. |
Table II.
Distribution of
LAP+CD4+ T cells in HCC, peri-cancer and
control HBV-infected hepatic tissues.
| Group | N | Average number of
LAP+CD4+T cells |
|---|
| Control | 28 | 2.61±0.83 |
| HCC | 28 |
11.25±3.00a |
| Peri-cancer | 28 |
5.75±1.00b |
Discussion
In 1995, Sakaguchi et al (13) first reported that 10% of
CD4+ T cells in the peripheral blood of normal adult
non-immune mice with T lymphocyte defects can express the α chain
(CD25) of interleukin (IL)-2. They named these
CD4+CD25+ T cells Treg cells and demonstrated
that these cells inhibit the activation of other T cells. To date,
various types of Treg cell have been identified among
CD4+ T cells, but the most widely studied are
CD4+CD25+forkhead box protein 3
(FOXP3)+ Treg cells (14). FOXP3 is the specific transcription
factor of Treg cells and is specifically expressed on their
surface. It is the most specific surface marker of Treg cells and
regulates their development, activation and functions (15).
LAP was first discovered by Miyazono et al
(16) in 1993. It is a pro-peptide
that binds non-covalently to the amino terminus of TGF-β. TGF-β is
a multifunctional polypeptide growth factor that is usually
secreted out of the cells in its inactive or latent precursor form
and exerts its biological activity after activation and binding to
TGF-β receptor (TβR). Pre-activated complexes of TGF-β include
TGF-β homodimer, as well as those with LAP and latent TGF-β binding
protein (LTBP). LAP remains connected to TGF-β via a non-covalent
bond after being cleaved from TGF-β precursor by a specific
protease and forms an inactive complex with LTBP to prevent
uncontrolled activation of TβR (17). In addition to keeping TGF-β in a
latent state, LAP also has an important role in releasing and
targeting latent TGF-β to the extracellular matrix, whereas LTBP
guides the assembly and secretion of latent TGF-β complexes.
Activation of TGF-β is achieved by partial or total enzymatic
cleavage of LAP (18).
In 2001, Nakamura et al (19) reported that TGF-β precursor is
expressed in mouse CD4+ T cells, drawing attention to
the functions of LAP in CD4+ T cells. Oida et al
(20) indicated that CD4+
T cells express LAP on their surface regardless of whether CD25 is
expressed. A previous study also suggested that CD25 expression in
CD4+CD25+ Treg cells is closely linked to the
regulatory activity of these cells (14). However, Nakamura et al
(21) demonstrated that
LAP+ T cells with TGF-β1 on their cell surface exert
inhibitory effects, which is independent of the expression of CD25.
Therefore, they reasoned that LAP as a surface marker of Treg cells
has more advantages than CD25. Chen et al (9) performed a study on
CD4+CD25+LAP+ Treg cells from
mice, indicating that TGF-β and TβR were expressed on their
surface. The immune regulatory function of these
CD4+CD25+LAP+ Treg cells is more
effective than that of
CD4+CD25+LAP− T cells due to
intercellular contact and TGF-β-dependent mechanisms.
The immunosuppressive effects of
LAP+CD4+ T cells have been demonstrated in
mouse models of cerebrospinal meningitis, allergic inflammation,
type II diabetes, colitis, arthritis, systemic lupus erythematosus
and atherosclerosis. Oida et al (20) identified CD4+ T cells that
express LAP+ on the cell surface by using goat LAP
antibody. Compared with LAP−CD4+ T cells,
LAP+CD4+ T cells produce more TGF-β and
IL-10, cytokines which are important for the immunomodulatory
effects of Treg cells in several systems. In a mouse model of
autoimmune encephalomyelitis, Ochi et al (22) identified that
CD4+CD25+LAP+ T cells express
TGF-β and more FOXP3 and cytotoxic T-lymphocyte-associated protein
4 than CD4+CD25+LAP− T cells and
exert stronger immune inhibitory effects in vitro and in
vivo. In a mouse model of arthritis,
LAP+CD4+ T cells were induced to secrete
IL-10 in the joint to inhibit arthritis-specific reactive T-cell
proliferation, the generation of specific antibodies and the
development of arthritis (23).
Different from the LAP+CD4+ T cells in the
mouse model of arthritis, LAP+CD4+ T cells
induced in models of systemic lupus erythematosus and diabetes
inhibited the development of T helper cells in a TGF-β-dependent
manner to alleviate the disease (24,25). The
above studies not only reveal the generality of immunosuppressive
action, but also suggest the differentiation of immunosuppression.
For instance, TGF-β mainly works in models of systemic lupus
erythematosus, diabetes and encephalomyelitis, high levels of IL-10
are secreted in arthritis models to inhibit effector T cells, and
TGF-β and IL-10 work jointly in the colitis model.
Previous studies (9,19,22,23)
have primarily focused on the functional mechanism of
LAP+CD4+ T cells, the results of which in
animal models are promising, but only few clinical studies are
available. Mahalingam et al (12) have assessed
LAP+CD4+ T cells in the frame of clinical
tumor research, revealing that the levels of
LAP+CD4+ T cells in the PBMCs and solid tumor
mass of colorectal carcinoma patients are markedly higher than
those in non-tumor tissue. The present study indicated that in
patients with HCC, the proportion of LAP+CD4+
T cells among the PBMCs is higher than that in the normal control
group. The mechanism may be associated with IL-8, TGF-β and/or
Foxp3. Akiba et al (26) also
reported that IL-8 was abnormally increased in the peripheral blood
of patients with HCC and in seven HCC cell lines and proved that
HCC cells secrete large amounts of IL-8. In the study by Gandhi
et al (27) IL-8 was the most
effective stimulator of LAP+CD4+ T-cell
activation among other cytokines. While increases of IL-8 activated
LAP+CD4+ T cells, the levels of active
LAP+CD4+ T cells exhibited a marked decline
when IL-8 mAb was provided. These results indicate that IL-8 in the
peripheral blood of patients with HCC acts as a modulator to
activate LAP+CD4+ T cells and is an important
factor involved in the negative regulation of tumor immunology via
LAP+CD4+ T cells. Almost all types of tumor
cell secrete TGF-β. The HCC and HepG2 human hepatoma cell line also
secretes TGF-β actively (17). Chen
et al (9) reported that
activated LAP+CD4+ T cells secrete TGF-β and
that the negative regulatory effects of
LAP+CD4+ T cells are contact-dependent and
TGF-β-dependent. Therefore, it was further indicated that TGF-β is
important in the negative immune regulation of
LAP+CD4+ T cells. FOXP3+ Treg
cells are a T-cell subset with a marked negative regulatory
cellular immunologic function. Abnormal expression of
FOXP3+ Treg cells has been identified in various
inflammatory pathologies and tumor types (28). FOXP3+ Treg cells are able
to inhibit the anti-tumor immunity and increase the immune
tolerance of tumor cells, which is important for the immune evasion
of tumors. Duan et al (10)
indicated that in a mouse model of asthma-associated pneumonia,
elevated levels of LAP+CD4+ T cells were
accompanied with an increased number of FOXP3+ Treg
cells. They reasoned that FOXP3 expressed on
LAP+CD4+ T cells may have an important
regulatory function.
The present results have demonstrated that the
proportion of LAP+CD4+ T cells among the
PBMCs of patients with HCC at the post-operative stage was
significantly declined compared with that at the pre-operative
stage, while it remained slightly but not significantly higher
compared with that in the control group. However, the advanced
stage of these two patients with metastasis may have affected the
results of flow cytometry. This result indicates that radical
resection affects the levels of LAP+CD4+ T
cells in the peripheral blood of patients with HCC and it balanced
out the immunologic derangement, reducing
LAP+CD4+ T cells to the normal level. The
possible mechanisms include the following: i) In the absence of
tumor antigens, the anti-tumor immune response, which mostly
comprises cellular immunity, decreases and the inhibitory effect of
LAP+CD4+ T cells on the anti-tumor immune
response also decreases. ii] Tumor cells secrete large amounts of
IL-8, TGF-β and other cytokines. Radical resection may reduce the
levels of these cytokines and change the microenvironment where
LAP+CD4+ T cells exert their function,
leading to the inhibition of LAP+CD4+ T-cell
activation. Thus, this provides a valuable direction of future
research. Radical resection affects the levels of
LAP+CD4+ T cells in the peripheral blood of
patients with HCC and reduces LAP+CD4+ T
cells to the normal level. As LAP+CD4+ T
cells in the blood of patients with HCC also (partially) originate
from the tumor environment itself, where upon they are released
into the circulation, it can be hypothesized that these trends may
be more marked in hepatic tissues. Due to the difficulty of hepatic
tissue sampling from patients with HCC following radical resection,
it is difficult to validate this hypothesis. Future studies should
therefore determine the difference in
LAP+CD4+ T cell expression in hepatic tissue
following radical resection in animal models.
The immunity of patients with primary HCC is weak,
and the local immune microenvironment of the tumor is abnormal,
resulting in failure of immune defense. It is an important factor
for the immune evasion, recurrence and metastasis of HCC (29). A previous study revealed that
immunosuppressive Treg cells were abnormally increased in the
peripheral blood and tumor tissues of patients with HCC (30). Therefore, the extent of Treg cell
infiltration in tumor tissues is representative of the suppression
of anti-tumor immunity and is a key mechanism resulting in tumor
immune tolerance and immune escape. In the present study,
immunohistochemical staining with double enzyme labeling indicated
that i) LAP is located in the cytoplasm and on the membrane of
CD4+ T cells; ii) LAP+CD4+ T cells
are clustered in HCC tissues; and iii) the extent of
LAP+CD4+ T-cell infiltration in HCC,
peri-cancer and normal control groups is significantly different.
These results suggest the presence of
LAP+CD4+ T cell infiltration in HCC tissues,
which increased with the proximity to the tumor tissue. The results
of the present study indicate that the
LAP+CD4+ T cell infiltration (to reduce the
body's anti-tumor immune response) was highest in HCC tissue but
lower in peri-cancer tissue. However, this result is unexpected as
it would be more likely that LAP+CD4+T cell
infiltration is highest at the border between tumor and normal
tissue (peri-cancer tissue), where the body's anti-tumor immune
response is most inhibited. (31)
This may be due to tumor cells secreting large quantities of IL-8
(which is increased in the peripheral blood and microenvironments
of various types of cancer), TGF-β and other cytokines;
furthermore, IL-8 is closely associated with immune cell chemotaxis
and cell proliferation (26,27) as aforementioned. Therefore, it is
hypothesized that LAP+CD4+ T cells migrate to
HCC tissues via the IL-8 chemotactic effect and that
LAP+CD4+ T cells alter the quantity and
function of infiltrating lymphocytes within HCC tissues, thereby
inhibiting tumor-specific and non-specific immune responses and
promoting tumor progression, invasion or metastasis. The full
elucidation of this mechanism should thus be assessed in future
studies. Immunohistochemical analysis indicated that
LAP+CD4+ cells were present not only in the
stroma but at the surface of sinusoids or capillaries. This may be
due to the breakage of certain LAP+CD4+ T
cells during the preservation and fixation process of the tissue
sections, resulting in positive staining in sinusoids and
capillaries. Therefore, it may be speculated that
LAP+CD4+ T cells are gathered in local tissue
of HCC to negatively regulate effector T cells, which then inhibit
specific and non-specific anti-tumor immune responses, possibly
resulting in changes in the local microenvironment of the tumor in
favor of tumor cell proliferation and migration.
Treg cells mediate the immune evasion of tumors via
multiple molecular mechanisms. At present, the immune regulation
effects of Treg cells and the underlying mechanisms have remained
to be fully elucidated. An in vitro study indicated that
CD4+CD25+ Treg cells may mediate
immunosuppression by cell-to-cell contact-dependent and
cytokine-independent mechanisms, whereas the subsequent in
vivo study indicates that a cytokine-dependent mechanism may
also exist. For instance, IL-10 has an important role in immune
inhibition and TGF-β may participate in this process (32). Studies on animal models with
autoimmune diseases and inflammation indicated that
LAP+CD4+ T cells exert their
immunosuppressive activity by inhibiting effector T cells (23,24). At
present, little is known regarding LAP+CD4+ T
cells in the tumor microenvironment. The present study indicated
that in HCC tissues, LAP+CD4+ T cells are
mostly clustered and localize close to lymphocytes; furthermore,
CD4+ T cells accumulate in HCC nests. Therefore, the
present study suggests that LAP+CD4+ T cells
may inhibit the proliferation of effector T cells by cell-to-cell
contact and facilitate the evasion of HCC cells of the anti-tumor
immune response of the body.
The results of the present study allow for the
following conclusions: i) High levels of
LAP+CD4+ T cells with immune inhibitory
effects exist in the peripheral blood and tumor tissues of patients
with HCC, which is important for the negative regulation of the
local microenvironment of HCC and is one of the mechanisms of HCC
immune evasion. ii) After curative resection, the level of
LAP+CD4+ T cells in the peripheral blood of
patients with HCC decreased and approached the normal level, which
may aid in the attenuation of the immunosuppressive state and
improve the microenvironment of the local tissue. iii)
LAP+CD4+ T cells are closely associated with
CD4+ T cells in HCC tissues, suggesting that
LAP+CD4+ T cells may inhibit local anti-tumor
immunity by cell-cell contact and have an important role in the
immune escape of HCC. Further research is required to study the
cytokines and molecular mechanisms involved in the function of
LAP+CD4+ T cells in the tumor
microenvironment. Zhong et al (33) observed a positive correlation between
the proportion of LAP+CD4+ T cells and the
tumor-nodes-metastasis stage, the presence of distant metastasis
and the serum levels of carcinoembryonic antigen. In the present
study, the distribution of LAP+CD4+ T cells
in patients with HCC with different grades of differentiation and
clinical stages was not analyzed due to the limited funding and the
number of cases included; this will be investigated in a future
study by our group. Elucidation of the functions of
LAP+CD4+ T cells in the tumor
microenvironment and the underlying regulatory mechanisms, as well
as identification of methods to regulate
LAP+CD4+ T-cell proliferation, may lead to
the development of more effective anti-tumor immunotherapies.
Acknowledgements
The authors would like to thank Professor Yao-Ting
Gui and Dr Cai-Ling Li from the Department of Central Laboratory at
Shenzhen Hospital (Peking University, Shenzhen, China) for their
respective scientific consultation and discussion, and help with
laboratory analysis.
Funding
The present study was supported by the Science and
Technology Development Fund Project of Shenzhen (grant nos.
JC200903180670A, JCYJ20150403091443302 and JCYJ20160428164539088),
the Sanming Project of Medicine in Shenzhen (grant no.
SZSM201612021) and the Science and Technology Developing Project of
Guangdong Province (grant no. 2017B090904010).
Availability of data and materials
The datasets used and/or analyzed during the
current study are available from the corresponding author on
reasonable request.
Authors' contributions
XO conceived and designed the work that led to the
submission, carried out literature search, data acquisition,
manuscript editing and played an important role in interpreting the
results. XPL carried out the concepts, design, definition of
intellectual content, literature search, data acquisition, data
analysis and manuscript preparation. JG collected the blood
specimens, performed the flow cytometry and participated in the
data acquisition, data analysis and statistical analysis. JSC
collected the tissue samples, performed the immunohistochemical
analysis and participated in the data acquisition, data analysis
and statistical analysis. JCY, PKT and JKL carried out the
literature search, data acquisition and manuscript editing, and
participated in data analysis and statistical analysis. LPN
participated in the flow cytometry, assisted in the data
acquisition, data analysis and data interpretation. YZ and GYY
participated in the immunohistochemical analysis, assisted in the
data acquisition, data analysis and data interpretation.
Ethical approval and consent to
participate
This study was approved by the Ethics Committee of
Shenzhen Hospital (Peking University, Shenzhen, China). All
patients and volunteers who participated in the study were informed
of the possible health risks and potential effects of blood
sampling and provided written informed consent.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Sayour EJ, Mclendon P, Mclendon R, de Leon
G, Reynolds R, Kresak J, Sampson JH and Mitchell DA: Increased
proportion of FoxP3+ regulatory T cells in tumor infiltrating
lymphocytes is associated with tumor recurrence and reduced
survival in patients with glioblastoma. Cancer Immunol Immunother.
64:419–427. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yuan CH, Sun XM, Zhu CL, Liu SP, Wu L,
Chen H, Feng MH, Wu K and Wang FB: Amphiregulin activates
regulatory T lymphocytes and suppresses CD8+T cell-mediated
anti-tumor response in hepatocellular carcinoma cells. Oncotarget.
6:32138–32153. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bluestone JA and Abbas AK: Natural versus
adaptive regulatory T cells. Nat Rev Immunol. 3:253–257. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Weiner HL: Oral tolerance for the
treatment of autoimmune diseases. Annu Rev Med. 48:341–351. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Roncarolo MG and Levings MK: The role of
different subsets of T regulatory cells in controlling
autoimmunity. Curr Opin Immunol. 12:676–683. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Weiner HL, da Cunha AP, Quintana F and Wu
H: Oral tolerance. Immunol Rev. 241:241–259. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Filaci G, Fenoglio D and Indiveri F:
CD8(+) T regulatory/suppressor cells and their relationships with
autoreactivity and autoimmunity. Autoimmunity. 44:51–57. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wong CH and Kubes P: Imaging natural
killer T cells in action. Immunol Cell Biol. 91:304–310. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chen ML, Yan BS, Bando Y, Kuchroo VK and
Weiner HL: Latency-associated peptide identifies a novel CD4+CD25+
regulatory T cell subset with TGFbeta-mediated function and
enhanced suppression of experimental autoimmune encephalomyelitis.
J Immunol. 180:7327–7337. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Duan W, So T, Mehta AK, Choi H and Croft
M: Inducible CD4+LAP+Foxp3-regulatory T cells suppress allergic
inflammation. J Immunol. 187:6499–6507. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ilan Y, Maron R, Tukpah AM, Maioli TU,
Murugaiyan G, Yang K, Wu HY and Weiner HL: Induction of regulatory
T cells decreases adipose inflammation and alleviates insulin
resistance in ob/ob mice. Proc Natl Acad Sci USA. 107:9765–9770.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mahalingam J, Lin YC, Chiang JM, Su PJ,
Fang JH, Chu YY, Huang CT, Chiu CT and Lin CY: LAP+CD4+ T cells are
suppressors accumulated in the tumor sites and associated with the
progression of colorectal cancer. Clin Cancer Res. 18:5224–5233.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sakaguchi S, Sakaguchi N, Asano M, Itoh M
and Toda M: Immunologic self-tolerance maintained by activated T
cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a
single mechanism of self-tolerance causes various autoimmune
diseases. J Immunol. 155:1151–1164. 1995.PubMed/NCBI
|
|
14
|
Sakaguchi S: Naturally arising
Foxp3-expressing CD25+CD4+ regulatory T cells in immunological
tolerance to self and non-self. Nat Immunol. 6:345–352. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Huang CT, Workman CJ, Flies D, Pan X,
Marson AL, Zhou G, Hipkiss EL, Ravi S, Kowalski J, Levitsky HI, et
al: Role of LAG-3 in regulatory T cells. Immunity. 21:503–513.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Miyazono K, Ichijo H and Heldin CH:
Transforming growth factor-beta: Latent forms, binding proteins and
receptors. Growth Factors. 8:11–22. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Keski-Oja J, Koli K and von Melchner H:
TGF-beta activation by traction? Trends Cell Biol. 14:657–659.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
McMahon GA, Dignam JD and Gentry LE:
Structural characterization of the latent complex between
transforming growth factor beta 1 and beta 1-latency-associated
peptide. Biochem J. 313:343–351. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Nakamura K, Kitani A and Strober W: Cell
contact-dependent immunosuppression by CD4(+)CD25(+) regulatory T
cells is mediated by cell surface-bound transforming growth factor
beta. J Exp Med. 194:629–644. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Oida T, Zhang X, Goto M, Hachimura S,
Totsuka M, Kaminogawa S and Weiner HL:
CD4+CD25− T cells that express
latency-associated peptide on the surface suppress
CD4+CD45RBhigh-induced colitis by a TGF-beta-dependent
mechanism. J Immunol. 170:2516–2522. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Nakamura K, Kitani A, Fuss I, Pedersen A,
Harada N, Nawata H and Strober W: TGF-beta 1 plays an important
role in the mechanism of CD4+CD25+ regulatory
T cell activity in both humans and mice. J Immunol. 172:834–842.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ochi H, Abraham M, Ishikawa H, Frenkel D,
Yang K, Basso AS, Wu H, Chen ML, Gandhi R, Miller A, et al: Oral
CD3-specific antibody suppresses autoimmun encephalomyelitis by
inducing CD4+ CD25-LAP+ T cells. Nat Med. 12:627–635. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wu HY, Maron R, Tukpah AM and Weiner HL:
Mucosal anti-CD3 monoclonal antibody attenuates collagen-induced
arthritis that is associated with induction of LAP+ regulatory T
cells and is enhanced by administration of an emulsome-based
Th2-skewing adjuvant. J Immunol. 185:3401–3407. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wu HY, Quintana FJ and Weiner HL: Nasal
anti-CD3 antibody ameliorates lupus by inducing an IL-10-secreting
CD4+ CD25-LAP+ regulatory T cell and is associated with
down-regulation of IL-17+ CD4+ ICOS+ CXCR5+ follicular helper T
cells. J Immunol. 181:6038–6050. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ishikawa H, Ochi H, Chen ML, Frenkel D,
Maron R and Weiner HL: Inhibition of autoimmune diabetes by oral
administration of anti-CD3 monoclonal antibody. Diabetes.
56:2103–2109. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Akiba J, Yano H, Ogasawara S, Higaki K and
Kojiro M: Expression and function of interleukin-8 in human
hepatocellular carcinoma. Int J Oncol. 18:257–264. 2001.PubMed/NCBI
|
|
27
|
Gandhi R, Farez MF, Wang Y, Kozoriz D,
Quintana FJ and Weiner HL: Cutting edge: Human latency-associated
peptide+ T cells: A novel regulatory T cell subset. J Immunol.
184:4620–4624. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Huang Y, Wang FM, Wang T, Wang YJ, Zhu ZY,
Gao YT and Du Z: Tumor-infiltrating FoxP3+Tregs and
CD8+T cells affect the prognosis of hepatocellular
carcinoma patients. Digestion. 86:329–337. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Brunner SM, Itzel T, Rubner C, Kesselring
R, Griesshammer E, Evert M, Teufel A, Schlitt HJ and Fichtner-Feigl
S: Tumor-infiltrating B cells producing antitumor active
immunoglobulins in resected HCC prolong patient survival.
Oncotarget. 8:71002–71011. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhou Y, Wang B, Wu J, Zhang C, Zhou Y,
Yang X, Zhou J, Guo W and Fan J: Association of preoperative EpCAM
circulating tumor cells and peripheral treg cell levels with early
recurrence of hepatocellular carcinoma following radical hepatic
resection. BMC Cancer. 16:5062016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zhang L, Jiang G, Yao F, Liang G, Wang F,
Xu H, Wu Y, Yu X and Liu H: Osthole promotes anti-tumor immune
responses in tumor-bearing mice with hepatocellular carcinoma.
Immunopharmacol Immunotoxicol. 37:301–307. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Miyara M and Sakaguchi S: Natural
regulatory T cells: Mechanisms of suppression. Trends Mol Med.
13:108–116. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhong W, Jiang ZY, Zhang L, Huang JH, Wang
SJ, Liao C, Cai B, Chen LS, Zhang S, Guo Y, et al: Role of
lap+cd4+t cells in the tumor microenvironment of colorectal cancer.
World J Gastroenterol. 23:455–463. 2017. View Article : Google Scholar : PubMed/NCBI
|