Introduction
Stroke is the third-leading cause of mortality in
developed countries with an incidence of 250-400 cases per 100,000
people and a mortality rate of ~30% (1). The prevalence and cost of stroke are
expected to rise as the global population ages and in less
developed countries, the incidence and mortality rates of stroke
are increasing due to rapidly changing lifestyles and population
structures (2).
Ischemic cerebral infarction is the cause of ~80% of
strokes (3). Ischemic stroke occurs
following the obstruction of blood vessels supplying blood to the
brain. Due to the limited regeneration capacity of central nervous
tissue, patients with brain damage resulting from ischemic stroke
suffer from lifelong disabilities (4).
Stem cell therapy is a potential novel strategy of
treating patients that have experienced strokes. It has been
demonstrated to have therapeutic potential in animal models of
neurological disorders (5,6). Previous studies have also performed
cell transplantation in models of ischemic infarct. Different types
of stem cells, including rodent mesenchymal stem cells (7), human bone marrow stem cells (8), human umbilical cord blood cells
(9) and rodent embryonic hippocampal
formation cells (10) ameliorate
neurological deficits induced by experimental brain ischemia. The
present study investigated the effects of primary rodent neural
stem cell (NSC) administration on in vitro and in
vivo stroke models and determined the role of the
phosphoinositide 3-kinase/protein kinase b/glycogen synthase kinase
3β (PI3K/Akt/GSK-3) signaling pathway in the neuroprotective
effects of pre-treatment with NSCs.
Materials and methods
Primary culture of NSCs
Complete NSC medium was prepared by mixing
Neuralbasal medium (20 ml/brain; Thermo Fisher Scientific, Inc.,
Waltham, MA, USA) with 10 ng/ml B27 (Invitrogen; Thermo Fisher
Scientific, Inc.), 10 ng/ml basic fibroblast growth factor and 20
ng/ml epidermal growth factor (both Gibco; Thermo Fisher
Scientific, Inc.). The dissociated brain tissue from one animal was
then diluted in 20 ml of culture medium. The mixture was
supplemented with 1% antibiotics (penicillin/streptomycin) and
incubated at 37°C with 5% CO2 and 95% humidity.
A dissection microscope (Fisher Science Education™
Advanced Digital Stereomicroscopes; cat. no. S71012B; Thermo Fisher
Scientific, Inc.) was used to harvest Sprague-Dawley rat embryos at
day 13 (E13; the copulatory plug day was defined as E0). Rats (age,
3-5 weeks; weight, 250±30 g) were obtained from Changhai Hospital
Animal Center (Shanghai, China) and housed under controlled
environmental conditions (temperature, 23±1°C; humidity, 55±5% and
kept under a 12 h light/dark cycle; commercial food and water were
freely available). Trypsin (0.25%) was used to homogenize whole
brain samples (obtained from rat embryos). Neurospheres were
cultured as described previously (11). Cells were incubated at 37°C with 5%
CO2. NSCs were identified using immunofluorescence
labeling with anti-nestin antibody (1:1,000; cat. no. 14-9843-82;
Thermo Fisher Scientific, Inc.) and were then passaged for three
generations prior to administration.
Primary culture of brain microvascular
endothelial cells (BMECs)
Rat BMECs were isolated from Sprague-Dawley rats,
following a previously published protocol with some modifications
(12). Briefly, rat brains were
collected and homogenates of isolated cerebral cortices were then
digested using 0.1% collagenase II (Sigma-Aldrich; Merck KGaA,
Darmstadt, Germany) and 30 U/ml DNase I (cat. no. 10104159001;
Roche Diagnostics, Indianapolis, IN, USA) at 37°C for 1.5 h. The
pellet was then resuspended in 20% bovine serum albumin (BSA;
Sigma-Aldrich; Merck KGaA; 2 ml) and centrifuged at 1,000 × g for
20 min at 4°C. Microvessel pellets in the lower layer were digested
using 0.1% collagenase/dispase (Roche Diagnostics) and 20 U/ml
DNase I at 37°C for 1 h. Cells were incubated at 37°C with 5%
CO2 and a humidity of 95%. BMECs were identified using
immunofluorescence labeling with anti-factor VIII antibody
(1:1,000; cat. no. MA1-43037; Thermo Fisher Scientific, Inc.).
Fluorescence microscope was used to identify cell samples.
Oxygen-glucose deprivation (OGD) cell
model
A Transwell chamber (EMD Millipore, Billerica, MA,
USA) indirect co-culture system was used to identify the effect of
NSCs on BMECs under OGD. The groups for the OGD cell model were as
follows: An NSC group, a control group and an OGD group. For the
NSC group, the upper compartments of the Transwell inserts were
removed following co-culture of NSCs and BMECs (each
4×105/well) in Dulbecco's Modified Eagle medium
(DMEM)-F12 (Thermo Fisher Scientific, Inc.) mixed with 2% B27 and
1% fetal bovine serum (Sigma-Aldrich; Merck KGaA) for 72 h at 37°C.
BMECs were treated with DMEM comprising of glucose-free balanced
salt solution and cultured for 6 h in a hypoxic chamber under an
adjusted atmosphere comprising of 95% N2 and 5%
CO2 at 37°C; the control group consisted of BMECs
without NSCs that had not been exposed to OGD; and the OGD group
consisted of BMECs without NSCs that had been exposed to OGD, which
served as an internal control.
Animal model of middle cerebral
arterial occlusion (MCAO)
MCAO surgery was performed to induce focal cerebral
ischemic injury. Adult male Sprague-Dawley rats aged 6-8 weeks old
and weighing 250-350 g were obtained from the Second Military
Medical University (SMMU) Laboratory Animal Center (Shanghai,
China). Following intraperitoneal anesthesia with 10% chloral
hydrate (360 mg/kg; Sigma-Aldrich; Merck KGaA), focal cerebral
ischemia was induced using an operating microscope via right-sided
endovascular MCAO using a piece of monofilament nylon suture with a
blunted tip coated with poly-lysine. The filament was inserted
through the right common carotid artery and advanced along the
internal carotid artery until the tip occluded the proximal stem of
the middle cerebral artery. Induction of ischemic brain injury was
confirmed using behavioral tests.
Implantation of NSCs in a rat
model
NSCs were collected using centrifugation (800 × g
for 5 min at room temperature) and resuspended in PBS. Rats were
intraperitoneally anesthetized with 10% chloral hydrate (400
mg/kg). A stereotaxic instrument was then used to fix the animal's
head with the lambda point set at 0: Anteroposterior (AP)=16 mm;
medial lateral (ML)=21.6 mm; dorsal ventral (DV)=12.9 mm; and
hippocampus (point of NSCs administration): AP=19 mm; ML=23.6 mm;
DV=14.9 mm (Read coordinate from behind, back, and top,
anteroposterior=AP, medial lateral=ML, and dorsal ventral=DV). Each
rat received 10 µl total fluid using microsyringes over a 10 min
period immediately following MCAO. Following confirmation of the
induction of ischemic brain injury using behavioral tests, rats
were randomized into three groups for transplantation as follows:
i) The NSC group, consisting of rats that underwent transplantation
of ~4×105 NSCs into the hippocampus around the ischemic
boundary zone by injection following MCAO, ii) the MCAO group,
consisting of rats that underwent MCAO alone with PBS
transplantation and iii) the sham group, consisting of rats that
underwent MCAO without insertion of the filament into the MCA and
transplantation of PBS. Mice were anesthetized by inhaling 2.5%
isoflurane before Perform cervical dislocation. At 3 days after
MCAO, mice were sacrificed and samples were collected for further
study. All animals were treated in accordance with the NIH
guidelines for the Care and Use of Laboratory Animals (13). All experimental procedures in the
current study were approved by the Committee on Ethics of
Biomedicine, The Second Military Medical University.
Caspase-3 activity assay
Caspase-3 is one of the key enzymes that serve a
role in apoptosis. A caspase-3 colorimetric assay (Caspase 3
Activity assay kit, Colorimetric; Sigma-Aldrich; Merck KGaA) was
performed, which is based on the hydrolysis of the peptide
substrate acetyl-Asp-Glu-Val-Asp p-nitroanilide (Ac-DEVD-pNA) by
caspase-3, a process that results in the release of the
p-nitroaniline (pNA) moiety. Samples and standards were processed
as indicated in the manufacturer's protocol. Cells were homogenized
in lysis buffer (included in the kit) and centrifuged for 15 min at
a speed of 13,400 × g at 4°C and the supernatant was collected. The
reaction was initiated by adding Ac-DEVD-pNA to each well and the
mixture was shaken gently. The microplate was incubated at 37°C for
3 h and absorbance was measured at a wavelength of 405 nm using a
microplate reader.
Lactate dehydrogenase (LDH) release
assay demonstrating cell cytotoxicity
Cell cytoxicity following OGD treatment was
determined based on the release of LDH using the CytoTox-one
Homogeneous Membrane Integrity assay kit (Promega Corporation,
Madison, WI, USA) following the manufacturer's protocol.
Cytotoxicity was calculated using the following formula:
Cytotoxicity (%)=100×(experimental-culture medium
background)/(maximum LDH release-culture medium background).
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR) in brain tissue from rat
models
RT-qPCR was performed as previously described to
determine the expression of PI3k, Akt and GSK-3β mRNA (14). Briefly, 1 µg total RNA was isolated
using TRIzol reagent (Thermo Fisher Scientific, Inc.) and was then
reverse transcribed into cDNA with a commercially available Reverse
transcription kit (Tiangen Biotech, Co., Ltd., Beijing, China)
following the manufacturer's protocol. qPCR was performed using a
SYBR Green PCR Master mix (Tiangen Biotech, Co., Ltd.) on an ABI
7300 PCR Instrument (Applied Biosystems; Thermo Fisher Scientific,
Inc.). The following primers were used: PI3K, forward,
5′-CTCTCCTGTGCTGGCTACTGT-3′ and reverse,
5′-GCTCTCGGTTGATTCCAAACT-3′; Akt, forward,
5′-TCAGGATGTGGATCAGCGAGA-3′ and reverse,
5′-CTGCAGGCAGCGGATGATAA-3′; GSK-3β, forward,
5′-TTCTCGGTACTACAGGGCACCA-3′, and reverse,
5′-GTCCTAGCAACAATTCAGCCAACA-3′ and β-actin, forward,
5′-GGAGATTACTGCCCTGGCTCCTA-3′ and reverse,
5′-GACTCATCGTACTCCTGCTTGCTG-3′. The following cycling conditions
were used: 10 min at 95°C for polymerase activation, followed by
95°C for 30 sec and 60°C for 30 sec. Rat β-actin was used for
normalization. Relative fold changes in RNA expression normalized
to β-actin were calculated using the 2−ΔΔCq method
(15).
Western blot analysis
Total protein lysates from the ischemic penumbra
area tissues were prepared with ice-cold lysis buffer (50 mM Tris,
pH 7.6, 150 mM NaCl, 10% Triton-X 100 and 100× Roche protease
inhibitor cocktail protease and phosphatase inhibitors). Brain
tissues were then centrifuged in a microcentrifuge at a speed of
13,400 × g rpm for 1 min at 4°C. Protein concentration was
determined using a BCA assay kit (Promega Corporation). A total of
40 µg proteins were loaded per lane and resolved using 10%
SDS-polyacrylamide gel electrophoresis for 2 h at 120 V prior to
being transferred to PVDF membranes using wet tank electro
transfer, membranes were blocked with 5% BSA in TBST overnight at
4°C. Membranes were then incubated with primary antibodies against
phosphorylated (p)-PI3k (1:1,000; cat no. 4228), p-Akt (1:1,000;
cat no. 4060) and p-GSK3β (1:1,000; cat no. 9323; all Cell
Signaling Technology, Inc., Danvers, MA, USA) in 5% BSA blocking
buffer overnight at 4°C. Membranes were also incubated with the
following antibodies obtained from Abcam: PI3K (cat. no. ab86714,
1:1,000 dilution), Akt (cat. no. ab126811, 1:1,000 dilution), GSK3β
(cat. no. ab18893, 1:1,000 dilution) and GAPDH (cat. no. ab8245,
1:2,000 dilution). These were then blocked using 5% skimmed milk at
room temperature for 2 h. Subsequently membranes were incubated
with the horseradish-peroxidase-conjugated secondary antibody
(Horeseradish peroxidase conjugated-goat anti-Rabbit immunoglobulin
G; 1:4,000; cat. no. AB_2533967; Thermo Fisher Scientific, Inc.) in
blocking buffer for 1 h at room temperature. Samples were
visualized using an ECL kit (cat. no. Ab65623; Abcam).
Quantification of inflammatory
cytokines
The expression of cytokines within the brain
[Leptin, L-selectin, monocyte chemotactic protein-1 (MCP-1), tissue
inhibitor of metalloproteinases 1 (TIMP-1), tumor necrosis factor
(TNF)-α] were measured using a Multiplexed Sandwich ELISA-based
Quantitative array-quantibody Rat Cytokine Array 2 kit (cat. no.
QAR-CYT-2; RayBiotech, Inc., Norcross, GA, USA). The protein
molecules were quantified according to the manufacturer's protocol.
A total of 5 animals per group were used for cytokine analysis.
Hematoxylin and eosin (H&E)
staining
Tissue was removed and fixed immediately in 4%
paraformaldehyde at room temperature for 24 h. Brain sections (4
µm) were mounted on gelatin-coated slides. Mounted sections were
then rehydrated in distilled water and submerged in Hematoxylin for
5 min at room temperature. Sections were then rinsed and submerged
in eosin for 2 min at room temperature. Following eosin treatment,
sections were dehydrated in a graded ethanol series prior to
immersion in xylene. A light microscope was used to visualize the
H&E stained sections.
Nissl staining
Tissue was fixed in 4% paraformaldehyde at room
temperature for 24 h. Coronal brain sections (4 µm) were mounted on
gelatin-coated glass slides. Nissl assays were performed following
a previously described protocol (16). Briefly, samples were rehydrated using
decreasing ethanol concentrations and endogenous peroxidase
activity was quenched with 2% H2O2. The
slides were then processed for Nissl staining, according to
standard protocols. A light microscope was used to visualize the
Nissl stained sections (magnification, ×400).
Immunohistochemistry
Immunohistochemical staining of the brain tissue
samples was performed 4 days following MCAO surgery using an
antigen retrieval method, following a previously described protocol
(7). Brain sections were incubated
with anti-microtubule-associated protein 2 (MAP-2) antibody
(1:1,000; cat no. ab32454; Abcam, Cambridge, MA, USA), anti-growth
associated protein-43 (GAP-43) antibody (1:1,000; cat no. ab117265;
Abcam), anti-synaptophysin antibody (1:1,000; cat no. ab175533;
Abcam), anti-glial fibrillary acidic protein (GFAP) antibody
(1:1,000; cat no. HPA056030; Sigma-Aldrich; Merck KGaA),
anti-ionized calcium binding adaptor protein (IBA-1) antibody
(1:1,000; cat no. HPA056030; Sigma-Aldrich; Merck KGaA),
anti-microtubule-associated protein light chain 3 (MAP1LC3A)
antibody (1:1,000; cat no. SAB1306673; Sigma-Aldrich; Merck KGaA)
or anti-Beclin-1 antibody (1:1,000; cat no. SAB1306484;
Sigma-Aldrich; Merck KGaA) overnight at 4°C. Brain samples were
then incubated with biotinylated goat anti-rabbit immunoglobulin G
(cat no. A0208; Beyotime Institute of Biotechnology, Haimen,
China), followed by streptavidin-peroxidase (Beyotime Institute of
Biotechnology) for 40 min at room temperature. The data were
analyzed using Image J software version 1.48 (National Institutes
of Health, Bethesda, MD, USA). A total of 5 animals per group were
used for immunohistochemical analysis, 5 fields of the brain of
each rat at ×200 magnification were selected and mean optical
density was calculated.
Behavioral tests
The modified foot-fault test and adhesive removal
test were employed 3 days following MCAO rat model establishment,
which aimed to measure forelimb placement dysfunction and
somatosensory deficits, respectively (17,18). The
total number of steps that the rat used to cross the grid, left
forelimb foot faults and the mean time required to remove each
stimulus from the limbs was recorded.
Statistical analysis
Statistical significance among the groups was
analyzed using one-way analysis of variance and Tukey's post hoc
test. Data are expressed as the mean ± standard deviation.
P<0.05 was determined to indicate statistically significant
difference. Statistical analysis was performed using SPSS 19.0 (IBM
Corp., Armonk, NY, USA).
Results
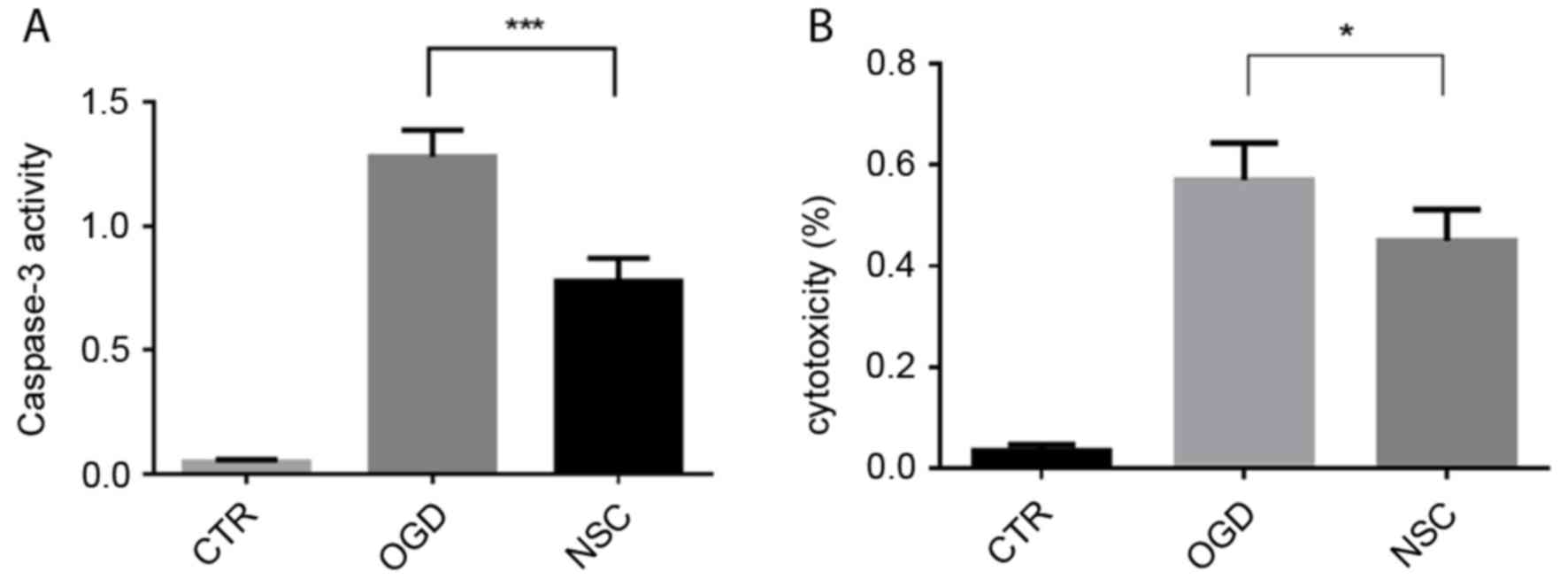
NSCs significantly suppress caspase-3
activity and decrease the release of LDH in BMECs
Caspase-3 activation serves a key role in apoptosis
and may be used as a marker of apoptosis. Caspase-3 activity was
increased ~1.4-fold in the OGD group compared with the NSC group
(P<0.001; Fig. 1A). LDH is an
indicator of cytotoxicity, which is released into the cell culture
supernatant when cell membranes are damaged. The effect of NSCs on
OGD-induced BMEC cytotoxicity was evaluated using an LDH release
assay (Fig. 1B). The cytotoxicity of
the NSC group was significantly decreased compared with the OGD
group (P<0.05).
Effects of NSCs on PI3k/Akt/GSK-3β
signaling pathway activation
Our previous in vitro study demonstrated that
phosphorylation levels of PI3k/Akt/GSK-3β were altered in neurons
following NSCs treatment under OGD (unpublished data). The results
of the in vivo experiments in the current study further indicated
that the PI3k/Akt/GSK-3β signaling pathway may be at least
partially involved in the neuroprotective effects conferred by
NSCs. The expression of p-PI3k, p-Akt and p-GSK3β protein was
measured using western blotting (Fig.
2A). The expressions of p-PI3k at Tyr458 (P<0.01), p-Akt at
Ser473 (P<0.05) and p-GSK-3β at Ser9 (P<0.001) were
significantly increased in the NSC group compared with the MCAO
group (Fig. 2B) RT-qPCR analysis was
performed simultaneously to measure the level of expression of
PI3k, Akt and GSK-3β mRNA (Fig. 2C).
The results indicated that the levels of PI3k and Akt mRNA were
increased in the NSC group compared with the MCAO group (both
P<0.01). By contrast, the expression of GSK-3β mRNA was
significantly decreased (P<0.05) in the NSC group compared with
the MCAO group.
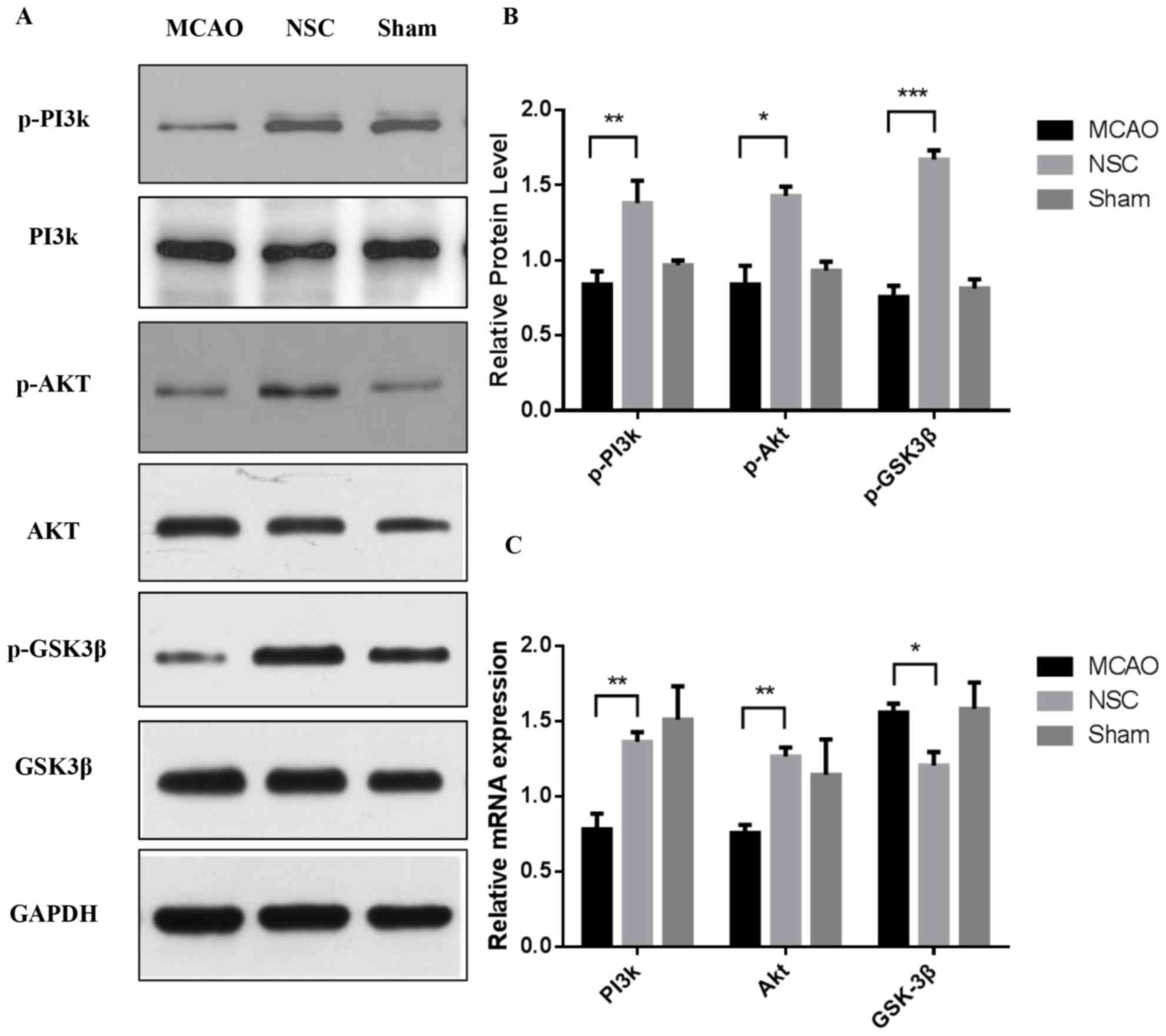 | Figure 2.Western blotting and reverse
transcription-quantitative polymerase chain reaction analysis of
the expression of p-PI3k, p-Akt and p-GSK3β in brain tissues from
an MCAO rat model. (A) Levels of p-PI3k, p-Akt and p-GSK3β protein
were analyzed using western blotting and GAPDH was used as a
loading control. (B) Quantitative analysis of western blotting.
Expression of p-PI3k, p-Akt and p-GSK3β were normalized to total
PI3k, Akt and GSK3β. (C) Quantitative analysis of the relative
levels of PI3k, Akt and GSK3β mRNA. Data are presented as the mean
± standard deviation. All experiments were performed in triplicate.
*P<0.05, **P<0.01, ***P<0.001. p-, phosphorylated; PI3k,
phosphoinositide 3-kinase; Akt, protein kinase b; GSK3β, glycogen
synthase kinase 3β; MCAO, middle cerebral artery occlusion group;
NSC, neural stem cell group. |
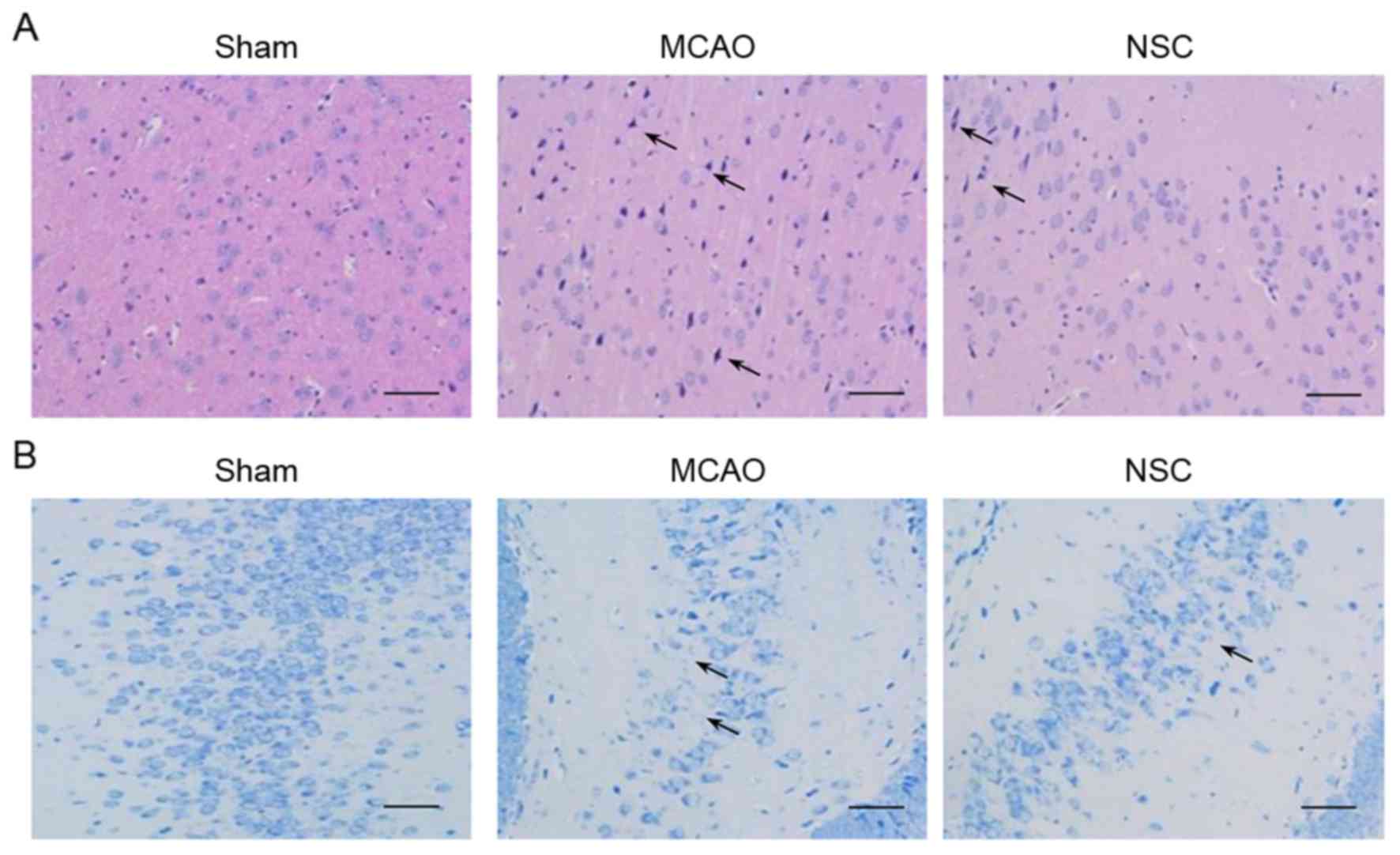
H&E and Nissl staining
The results of H&E staining of the dentate gyrus
of the hippocampus in the MCAO group identified the following
typical neuropathological changes in rats: Neuronal cell loss,
ambiguous structures, nuclear shrinkage and dark neuron staining.
NSC treatment significantly attenuated these ischemic
neuropathological changes (Fig. 3A).
The adjacent hippocampal sections were subjected to Nissl staining
(Fig. 3B). The reduction in Nissl
staining is indicative of neuronal degeneration. A greater
reduction of Nissl staining was evident in the tissues from the
MCAO group compared with tissues from the NSC group.
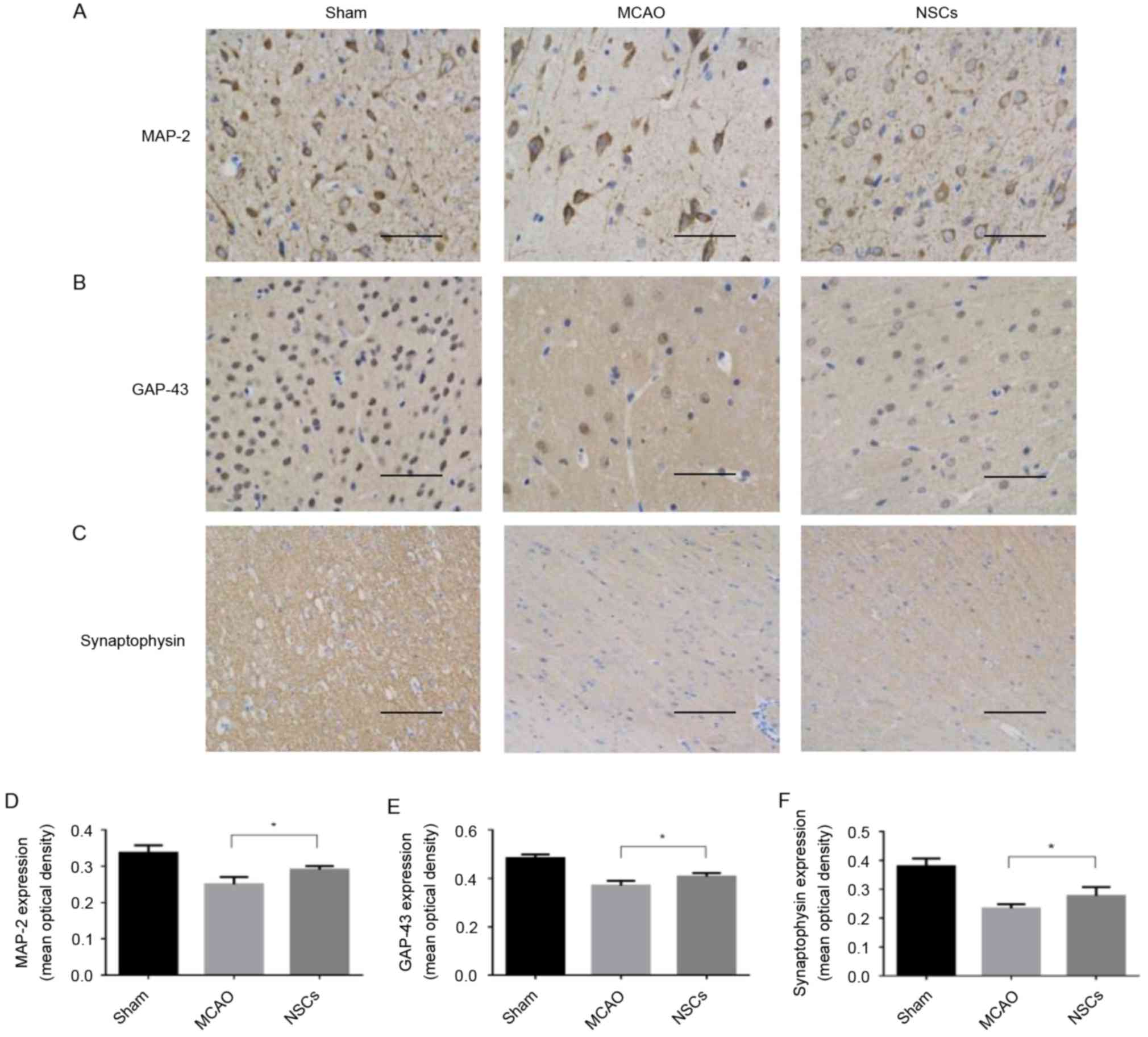
Synaptic remodeling is mediated by
NSCs in the MCAO animal model
MAP-2 is a cytoskeletal phosphoprotein primarily
associated with microtubules and postsynaptic densities (19). GAP-43 is an intracellular
membrane-associated phosphoprotein expressed in neuronal growth
cones and is involved in axonal growth, synaptogenesis, synaptic
remodeling and neurotransmitter release (20). Synaptophysin is a 38-kDa
calcium-binding glycoprotein present in the membranes of
neurotransmitter-containing presynaptic vesicles and is used as a
specific protein marker for the presynaptic terminal (21). Levels of synaptophysin serve as an
index of synaptic number and density (22). The mean average intensity of MAP-2,
GAP-43 and synaptophysin staining were significantly increased in
the NSC group compared with the MCAO group (all P<0.05; Fig. 4).
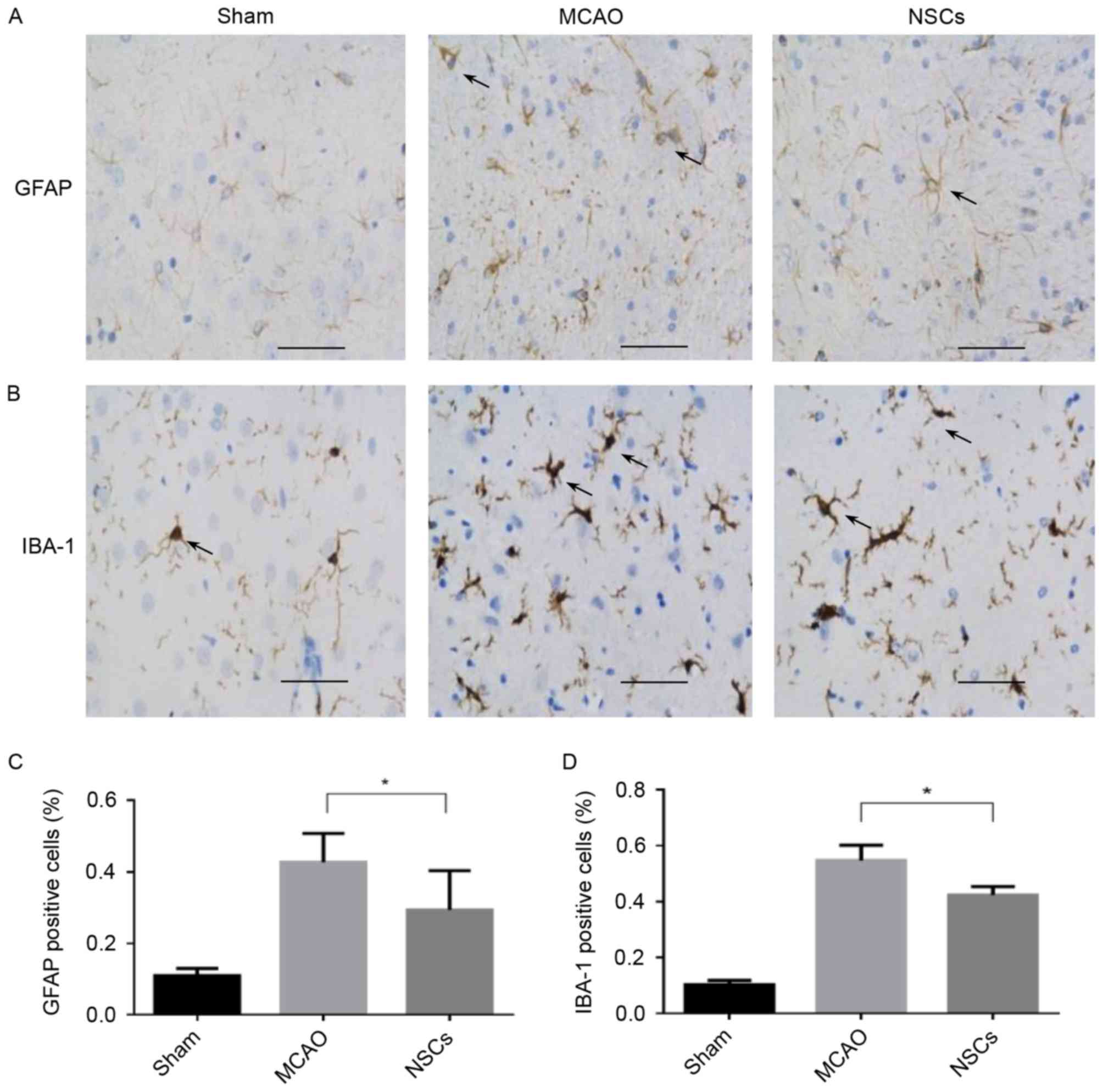
Immunomodulatory properties of
NSCs
The expression of IBA-1 in the microglia and GFAP in
the astrocytes increase in response to a wide variety of
pathological stimuli in the central nervous system (CNS). Microglia
and astrocyte activation commonly occur during the early response
of the CNS to a wide variety of pathological stimuli, including
axotomy, trauma, inflammation and degeneration (23). There was a significant decrease in
the number of IBA-1 and GFAP-positive cells in the peri-ischemic
area in the NSC group compared with the MCAO group (both P<0.05;
Fig. 5).
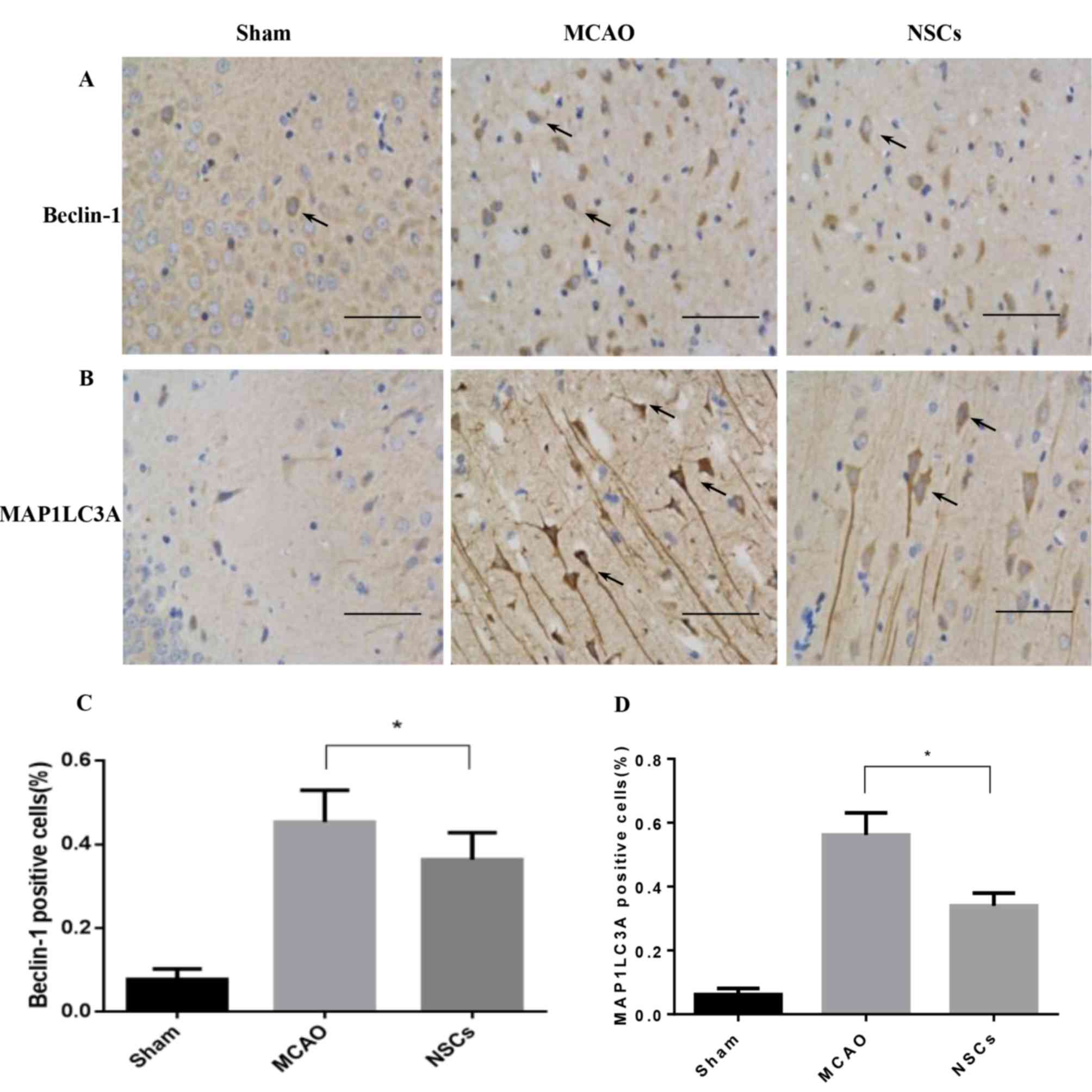
Autophagy marker levels decrease
following NSC treatment
MAP1LC3 and Beclin-1 serve a pivotal role in
mammalian autophagy, which is increased during periods of cell
stress and extinguished during the cell cycle (24). In ischemic neurons, Beclin-1 markers
were dispersed in the soma and dendrites (Fig. 6A), while MAP1LC3 was primarily
located in the soma (Fig. 6B). The
number of MAP1LC3-positive and Beclin-1-positive immune-reactive
puncta were significantly decreased in the NSC group compared with
the MCAO group (both P<0.05; Fig.
6C).
NSCs suppress ischemia-triggered
inflammatory cytokines
Ischemic stroke is accompanied by increased
inflammatory cytokine secretion; therefore basal cytokine secretion
levels in homogenized tissue samples were measured using a protein
array in which levels of different cytokines were quantified
simultaneously. The secretion of pro-inflammatory cytokines,
including L-selectin, leptin, MCP-1 and TNFα was increased in the
MCAO group compared with the sham group. NSCs further increased the
release of anti-inflammatory cytokine TIMP-1 (Fig. 7). These results demonstrate that NSCs
are able to shift the balance of the inflammatory response from
pro-inflammatory to anti-inflammatory.
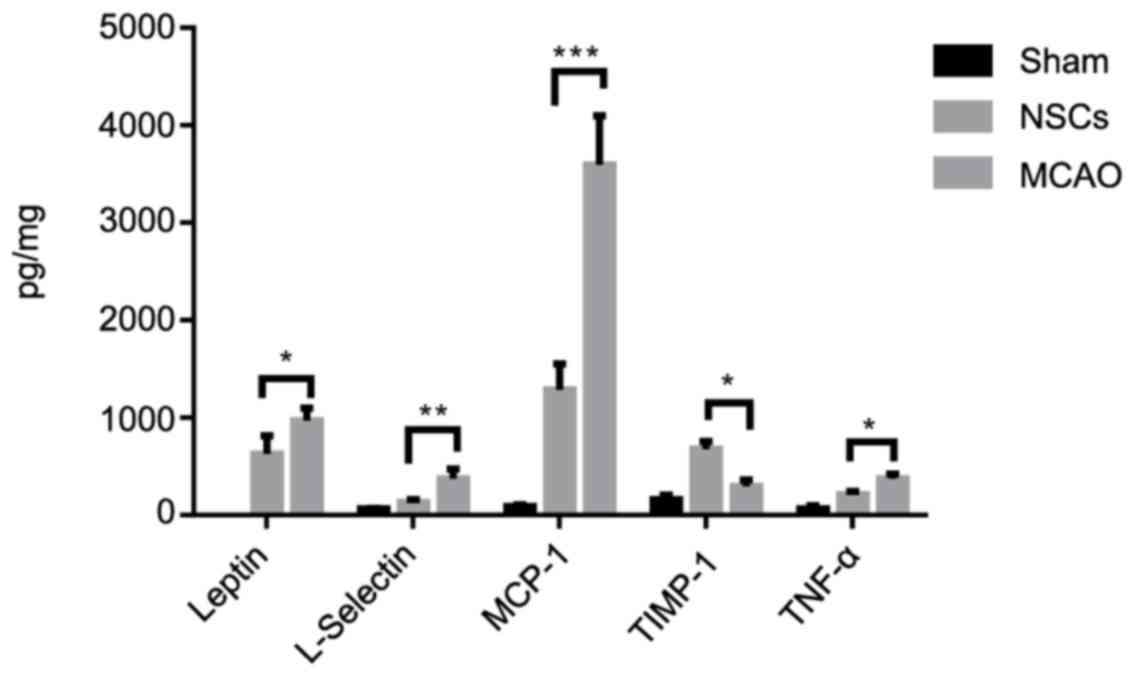 | Figure 7.Cytokine analysis of brain tissues
from an MCAO rat model. Among the analyzed cytokines, NSCs
treatment attenuated the increase of the pro-inflammatory cytokines
L-selectin, leptin, MCP-1, TNFα and further increased the release
of anti-inflammatory cytokine TIMP-1. Data are presented as the
mean ± standard deviation (n=5). *P<0.05, **P<0.01,
***P<0.001. MCP-1, monocyte chemotactic protein 1; TNFα, tumor
necrosis factor α, TIMP-1, tissue inhibitor of metalloproteinases
1. |
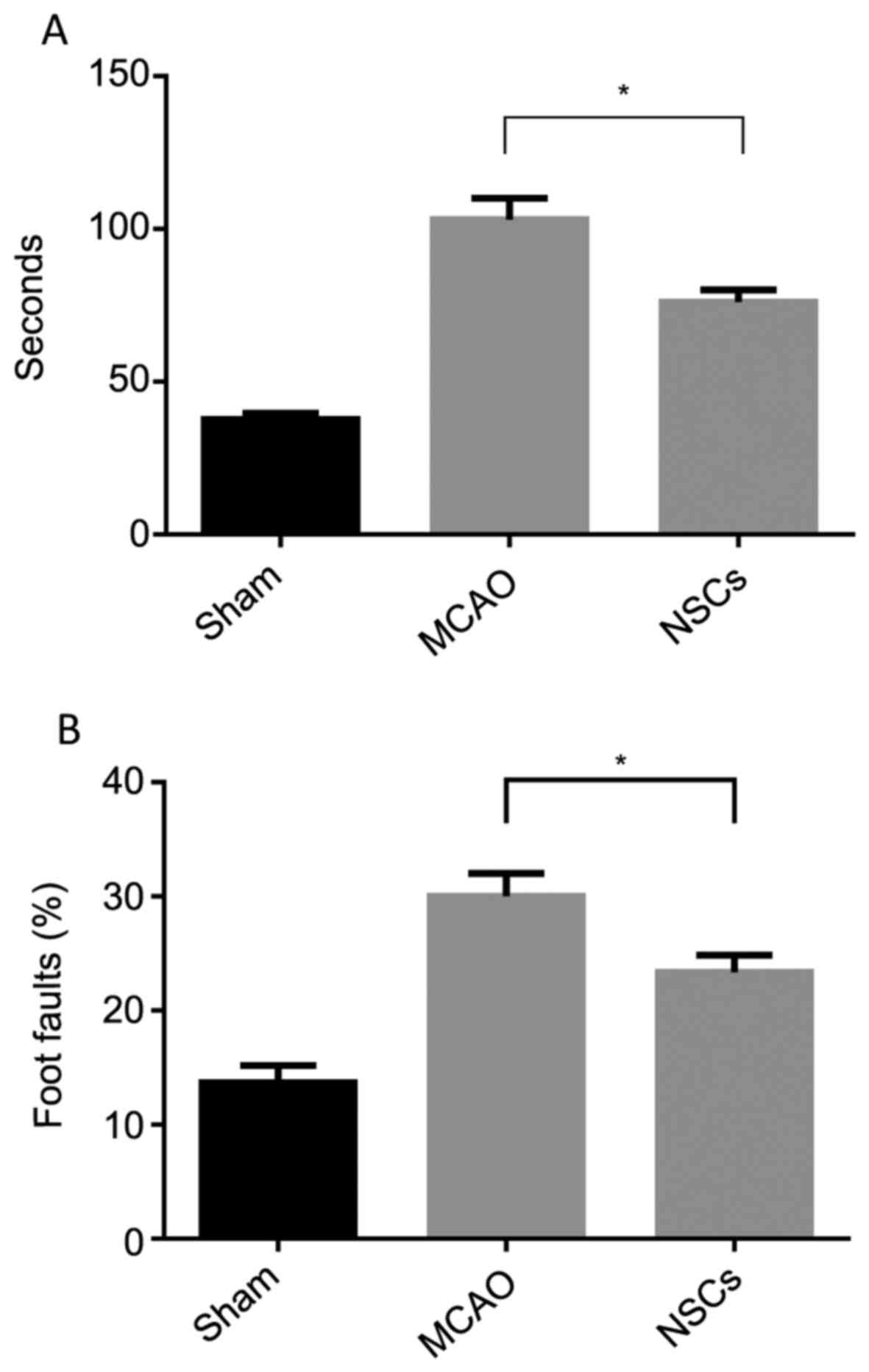
NSCs improve functional outcomes
The foot-fault and adhesive removal tests conducted
3 days following stroke onset indicated that all MCAO and
MSC-treated rats exhibited severe neurological deficits. Treatment
with NSCs initiated 24 h following stroke onset significantly
improved performance on the adhesive removal test (P<0.05;
Fig. 8A) and foot-fault test
(P<0.05; Fig. 8B) compared with
the MCAO group. These results indicate that NSCs may have the
capacity to improve functional outcomes in mice with stroke.
Discussion
Ischemic stroke consists of a complex array of
pathological processes and induces highly variable outcomes
modified by many factors. The rationale for using NSCs as potential
candidates in cell therapy for patients who have experienced stroke
is based on a range of studies that have demonstrated or suggested
the advantages conferred by NSCs following their intravenous or
intracerebral administration in vivo through
immunomodulation, signal amplification and neural repair (11,25,26).
NSCs are regarded as one of the most promising stem cell candidates
to be used as a therapy for many diseases where treatment is
challenging (27). Patients who have
experienced stroke face medical and economic burdens due to
limitations in the number of available treatments (28). Stem cell based replacement therapy
has therefore become attractive due to its potential to solve these
problems.
In the current study, NSCs were administered to the
area surrounding the lateral ventricle in the stroke-affected
hemisphere. The hippocampus was identified as the ideal site for
this type of intervention, as it is one of only two regions in the
adult mammalian brain in which neurogenesis is ongoing (29). Furthermore, it exhibits an abundance
of migration, proliferation, differentiation and integration
signals (30). Thus, the hippocampus
may be a potential target for treatments of neurological conditions
in humans and serves as a gateway for effective clinical
interventions.
Apoptosis is a specific process that leads to
programmed cell death. Cytosolic cytochrome c release and
caspase activation have been observed during ischemia-induced
apoptosis (31) and NSCs are able to
attenuate caspase-3 activation and impairment in OGD-treated BMECs.
It has been demonstrated in vivo that NSCs affect the
PI3K/Akt/GSK3β pathway, which regulates cell proliferation,
survival and migration (14,32,33).
Ischemia increases autophagosome formation and
activate autophagy. MAP1LC3 and Beclin-1 are involved in the
signaling pathway that activates autophagy and the initial step of
autophagosome formation (34). The
in vivo results of the current study demonstrate that the
number of MAP1LC3-positive and Beclin-1-positive immuno-reactive
puncta were significantly decreased in the NSC group compared with
the MCAO group. This indicates that NSCs may suppress autophagic
reactions in the ischemic brain and that this role may be
associated with the PI3K/Akt/GSK3β pathway, as the mechanistic
target of rapamycin is downstream of this pathway and integrates
signals from nutrients, energy regulators and growth factors to
regulate autophagy (33).
The current study demonstrated that NSCs
transplanted into the hippocampus around the ischemic boundary zone
of an ischemic lesion facilitate the functional recovery from
strokes. Cell survival promoted by NSCs may contribute to this
recovery. NSCs also reduce the number of GFAP and IBA1-expressing
cells, which indicates that they have immunomodulatory properties.
These results are consistent with those of previously published
studies (9,11,26).
NSCs possess a strong anti-inflammatory capacity in
a variety of disease models including Alzheimer's disease,
Parkinson's disease and traumatic brain injury models (14). The current study demonstrated that
NSC administration may modulate inflammation by reducing
L-selectin, leptin, MCP-1 and TNFα production and increasing TIMP-1
levels.
In conclusion, the present study suggests that in
addition to increasing cell survival to improve functional recovery
following stroke in rats, NSCs may also activate the
PI3k/Akt/GSK-3β pathway and attenuate the inflammatory process and
autophagy. These effects may contribute to the underlying
beneficial effects exerted by NSCs in ischemic stroke. Further
investigation into the precise molecular mechanisms governing these
effects may lead to the development of novel preventive and
therapeutic strategies for managing ischemic injury in the
future.
Acknowledgements
The authors would like to thank Shibo Wang for
graphical assistance and Jiangbo Gong for proofreading.
Funding
The present study was supported by the Shanghai
Pujiang Program (grant no. 18PJD058), SMMU Youth Fund (grant nos.
2017QN10, 2017QN11).
Availability of data and materials
The analyzed data sets generated during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
XY and XT conceived and planned the experiments, XY
and XW carried out the experiments. SZ contributed to the
interpretation of the results. XY took the lead in writing the
manuscript. All authors provided critical feedback and helped shape
the research, analysis and manuscript.
Ethics approval and consent to
participate
All experimental procedures in the current study
were approved by the Committee on Ethics of Biomedicine, The Second
Military Medical University.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Arsić S, Konstantinović L, Eminović F and
Pavlović D: Correlation between demographic characteristics,
cognitive functioning and functional independence in stroke
patients. Srp Arh Celok Lek. 144:31–37. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kes VB, Jurašić MJ, Zavoreo I, Lisak M,
Jelec V and Matovina LZ: Age and gender differences in acute stroke
hospital patients. Acta Clin Croat. 55:69–78. 2016.PubMed/NCBI
|
|
3
|
Nentwich LM and Grimmnitz B: Neurologic
emergencies in the elderly. Emerg Med Clin North Am. 34:575–599.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mietto BS, Mostacada K and Martinez AM:
Neurotrauma and inflammation: CNS and PNS responses. Mediators
Inflamm. 2015:2512042015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Descloux C, Ginet V, Clarke PG, Puyal J
and Truttmann AC: Neuronal death after perinatal cerebral
hypoxia-ischemia: Focus on autophagy-mediated cell death. Int J Dev
Neurosci. 45:75–85. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Dugo L, Collin M and Thiemermann C:
Glycogen synthase kinase 3beta as a target for the therapy of shock
and inflammation. Shock. 27:113–123. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yin F, Meng C, Lu R, Li L, Zhang Y, Chen
H, Qin Y and Guo L: Bone marrow mesenchymal stem cells repair
spinal cord ischemia/reperfusion injury by promoting axonal growth
and anti-autophagy. Neural Regen Res. 9:1665–1671. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhao LR, Duan WM, Reyes M, Keene CD,
Verfaillie CM and Low WC: Human bone marrow stem cells exhibit
neural phenotypes and ameliorate neurological deficits after
grafting into the ischemic brain of rats. Exp Neurol. 174:11–20.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Arien-Zakay H, Lecht S, Bercu MM, Tabakman
R, Kohen R, Galski H, Nagler A and Lazarovici P: Neuroprotection by
cord blood neural progenitors involves antioxidants, neurotrophic
and angiogenic factors. Exp Neurol. 216:83–94. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Onifer SM and Low WC: Spatial memory
deficit resulting from ischemia-induced damage to the hippocampus
is ameliorated by intra-hippocampal transplants of fetal
hippocampal neurons. Prog Brain Res. 82:359–366. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Roitbak T, Li L and Cunningham LA: Neural
stem/progenitor cells promote endothelial cell morphogenesis and
protect endothelial cells against ischemia via HIF-1alpha-regulated
VEGF signaling. J Cereb Blood Flow Metab. 28:1530–1542. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chou CH, Sinden JD, Couraud PO and Modo M:
In vitro modeling of the neurovascular environment by coculturing
adult human brain endothelial cells with human neural stem cells.
PLoS One. 9:e1063462014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Committee for the Update of the Guide for
the Care and Use of Laboratory Animals, Institute for Laboratory
Animal Research, Division on Earth and Life Studies, : Guide for
the Care and Use of Laboratory Animals. 8th edition. The National
Academies Press; Washington, DC: 2011, PubMed/NCBI
|
|
14
|
Golpich M, Amini E, Hemmati F, Ibrahim NM,
Rahmani B, Mohamed Z, Raymond AA, Dargahi L, Ghasemi R and
Ahmadiani A: Glycogen synthase kinase-3 beta (GSK-3beta) signaling:
Implications for Parkinson's disease. Pharmacol Res. 97:16–26.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang X, Yu X, Xie C, Tan Z, Tian Q, Zhu D,
Liu M and Guan Y: Rescue of brain function using tunneling
nanotubes between neural stem cells and brain microvascular
endothelial cells. Mol Neurobiol. 53:2480–2488. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Schallert T, Upchurch M, Lobaugh N, Farrar
SB, Spirduso WW, Gilliam P, Vaughn D and Wilcox RE: Tactile
extinction: Distinguishing between sensorimotor and motor
asymmetries in rats with unilateral nigrostriatal damage. Pharmacol
Biochem Behav. 16:455–462. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang R, Wang Y, Zhang L, Zhang Z, Tsang
W, Lu M, Zhang L and Chopp M: Sildenafil (Viagra) induces
neurogenesis and promotes functional recovery after stroke in rats.
Stroke. 33:2675–2680. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Liu Y, Saad RS, Shen SS and Silverman JF:
Diagnostic value of microtubule-associated protein-2 (MAP-2) for
neuroendocrine neoplasms. Adv Anat Pathol. 10:101–106. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Grasselli G and Strata P: Structural
plasticity of climbing fibers and the growth-associated protein
GAP-43. Front Neural Circuits. 7:252013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang X, Xing A, Xu C, Cai Q, Liu H and Li
L: Cerebrovascular hypoperfusion induces spatial memory impairment,
synaptic changes, and amyloid-β oligomerization in rats. J
Alzheimer's Dis. 21:813–822. 2010. View Article : Google Scholar
|
|
22
|
Song SH and Augustine GJ: Synapsin
isoforms and synaptic vesicle trafficking. Mol Cells. 38:936–940.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhao J, Mou Y, Bernstock JD, Klimanis D,
Wang S, Spatz M, Maric D, Johnson K, Klinman DM, Li X, et al:
Synthetic oligodeoxynucleotides containing multiple telemeric
TTAGGG motifs suppress inflammasome activity in macrophages
subjected to oxygen and glucose deprivation and reduce ischemic
brain injury in stroke-prone spontaneously hypertensive rats. PLoS
One. 10:e01407722015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wen YD, Sheng R, Zhang LS, Han R, Zhang X,
Zhang XD, Han F, Fukunaga K and Qin ZH: Neuronal injury in rat
model of permanent focal cerebral ischemia is associated with
activation of autophagic and lysosomal pathways. Autophagy.
4:762–769. 2014. View Article : Google Scholar
|
|
25
|
Pacey L, Stead S, Gleave J, Tomczyk K and
Doering L: Neural stem cell culture: Neurosphere generation,
microscopical analysis and cryopreservation. Protoc Exchange.
2006.
|
|
26
|
Weidenfeller C, Svendsen CN and Shusta EV:
Differentiating embryonic neural progenitor cells induce
blood-brain barrier properties. J Neurochem. 101:555–565. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Akesson E, Wolmer-Solberg N, Cederarv M,
Falci S and Odeberg J: Human neural stem cells and astrocytes, but
not neurons, suppress an allogeneic lymphocyte response. Stem Cell
Res. 2:56–67. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kankeu HT, Saksena P, Xu K and Evans DB:
The financial burden from non-communicable diseases in low- and
middle-income countries: A literature review. Health Res Policy
Syst. 11:312013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Walker T, Huang J and Young K: Neural stem
and progenitor cells in nervous system function and therapy. Stem
Cells Int. 2016:18905682016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Curtis MA, Low VF and Faull RL:
Neurogenesis and progenitor cells in the adult human brain: A
comparison between hippocampal and subventricular progenitor
proliferation. Dev Neurobiol. 72:990–1005. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Thiel A, Cechetto DF, Heiss WD, Hachinski
V and Whitehead SN: Amyloid burden, neuroinflammation, and links to
cognitive decline after ischemic stroke. Stroke. 45:2825–2829.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mao Y, Ge X, Frank CL, Madison JM, Koehler
AN, Doud MK, Tassa C, Berry EM, Soda T, Singh KK, et al: Disrupted
in schizophrenia 1 regulates neuronal progenitor proliferation via
modulation of GSK3beta/beta-catenin signaling. Cell. 136:1017–1031.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kang EB and Cho JY: Effect of treadmill
exercise on PI3K/AKT/mTOR, autophagy, and Tau hyperphosphorylation
in the cerebral cortex of NSE/htau23 transgenic mice. J Exerc Nutri
Biochem. 19:199–209. 2015. View Article : Google Scholar
|
|
34
|
Li H, Gao A, Feng D, Wang Y, Zhang L, Cui
Y, Li B, Wang Z and Chen G: Evaluation of the protective potential
of brain microvascular endothelial cell autophagy on blood-brain
barrier integrity during experimental cerebral ischemia-reperfusion
injury. Transl Stroke Res. 5:618–626. 2014. View Article : Google Scholar : PubMed/NCBI
|