Introduction
We recently described a procedure for human muscle
biopsy that we termed tiny percutaneous needle biopsy (TPNB) and we
demonstrated that it is an excellent method for obtaining human
skeletal muscle specimens with the least trauma for the patient
(1). The classically and commonly
used needle biopsy technique is needle aspiration biopsy (NAB),
using a Bergstrom needle (2,3).
Similar to NAB, TPNB involves a percutaneous approach (thus no need
for an invasive skin incision) and the use of a penetrating needle.
However, TPNB differs significantly in the considerably smaller
size of the sample and in the more automated and rapid penetration
of the needle into the depths of the muscle, resulting in a
considerably less traumatic approach and relatively rapid and
orderly regeneration of the tissue (1). To support the low invasiveness of
TPNB, we previously analyzed nuclear magnetic resonance (NMR)
images of muscles following TPNB (unpublished data). At this low
level of resolution, the muscle showed no apparent traces of
lesions or wounding. This evidence has convinced us that TPNB has
great potential for use in human muscle studies, particularly in
horizontal studies, requiring repeated samples from the same
subject, i.e., before and after a specific stimulus, such as a
training period. The positive aspects of this less invasive method
may prove to be advantageous when the volunteers are medium-high
level athletes.
Although only a few milligrams of muscle are
collected using TPNB, the quality of these specimens allows their
use in a large variety of cellular and molecular approaches,
including cell culture, functional studies of single dissociated
muscle fibers and RNA and protein analysis in transcriptional and
proteomic studies (4–6). However, the issue of whether the
very small biopsy samples can be used for structural and/or
ultrastructural analysis, remains unresolved. The cutting of the
muscle fibers with a blade that is part of both open surgery and
needle biopsy procedures, promotes the immediate depolarization of
the fibers, and hence their contraction. The fibers, however, relax
after a very brief period and if, as in a well-managed open biopsy
(7), they are restrained by
ligating to a suitable retaining support prior to excision, they
maintain their resting length and alignment. In a needle biopsy of
any size, the fibers are detached from the surrounding muscle
tissue and after the initial contraction they will remain at a
shorter length as the connective tissue will impede their passive
lengthening to the initial resting length. Additional considerable
disarrangement ensues as bundles of fibers within the sample are
variously oriented. To avoid these major drawbacks of needle biopsy
for structural studies, we explored the possibility that, if
properly handled, an excellent structural preservation of the small
sample could be obtained. The procedure involves the transferring
of the section of muscle from a needle biopsy to a solution that
mimics the intracellular medium (high potassium, low calcium) and
keeps the fibers depolarized and thus not excitable, and in the
appropriate sample micromanipulation in order to allow restoration
to the initial ‘resting’ sarcomere length or close to it. To that
effect, using light microscopy and thin section electron
microscopy, we analyzed muscle samples obtained from mice both by
standard dissection and by TPNB, as well as samples from human
subjects obtained by TPNB. The aim of this study was to clearly
demonstrate that good ultrastructural preservation can be obtained
from TPNB samples.
Materials and methods
Muscles from euthanized mice (under IACUC protocol
approved by the University of Pennsylvania) were used in two
different sets of experiments. One was in determining the effects
of two different high potassium solutions on the ultrastructure of
intact muscle fibers, and the second in devising the best approach
for the preservation of needle biopsy material. First, the extensor
digitorum longus (EDL) muscles were carefully dissected, pinned to
a Sylgard dish, exposed to a solution that replaced extracellular
sodium with potassium and chelated extracellular calcium. Two
different solutions were used: either a ‘KCl’ solution [150 mM KCl,
5 mM MgCl2, 3 mM ethylene glycol tetraacetic acid
(EGTA), 10 mM phosphate buffer, pH 7.2] or a ‘K acetate’ solution
(150 mM K acetate, 5 mM MgSO4, 10 mM EGTA, 10 mM
phosphate buffer, pH 7.2). Muscles were kept for ~3 min at room
temperature in one of these two solutions and then fixed in freshly
prepared 3.5% glutaraldehyde in 0.1 M cacodylate buffer, pH 7.2 for
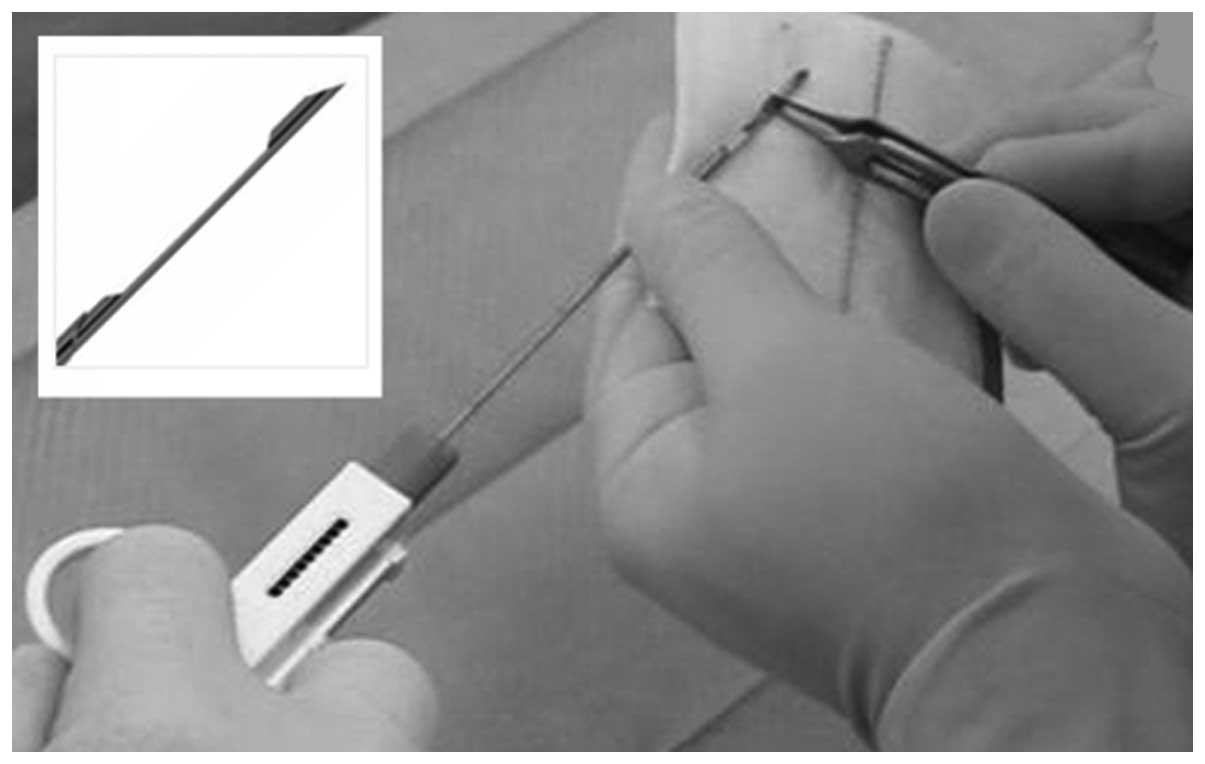
~1 h. Secondly, using a small size needle (semiautomatic 14-gauge
needle biopsy device: Vantage GS, Zamar S.r.l., Suzzara, Italy;
Precisa 1310, HS, Precisa Medico Corp., Latina, Italy) (Fig. 1) biopsies were taken from the
gastrocnemius and the masseter. The latter muscle was more suitable
as the needle was relatively large for the leg muscle of the mouse.
The samples that were expelled directly from the needle into the ‘K
acetate’ solution at room temperature contained several small
coherent bundles of fibers with different orientations and the
fibers appeared mostly wavy. Individual bundles of parallel fibers
were separated by gentle teasing, straightened out by ‘combing’
and/or stretching out with tweezers or syringe needles while
viewing under a dissecting microscope and then immersed in fixative
solution as described above. The fixed bundles were stored at 4°C,
for different periods of time.
We further obtained three small biopsy samples from
the vastus lateralis (VL) muscle from 32–66-year-old male healthy
volunteers (indicated as M32, 32 years old; M33, 41 years old; M34,
66 years old, respectively) using the TPNB procedure as described
in our previous study [Pietrangelo et al(1)]. The study was approved by the Ethics
Committee of the University of Chieti-Pescara (approval protocol
no. 1233/06 COET). Each subject provided written informed consent.
Biopsy was performed under local anesthesia using 2 ml carbocaine
(20 mg/ml; AstraZeneca S.P.A., Basiglio, Italy) following skin
sterilization with betadine. We used semiautomatic needle biopsy
devices as for the mouse, but 13-gauge in size. The cylinder of the
muscle thus obtained had a cross-sectional area of ~3
mm2 and a length of ~4 mm. Despite the small needle
diameter, the TPNB VL muscle specimens were of adequate size and
good quality. The specimens were immediately immersed in ‘K
acetate’ solution and then treated as for the mouse.
All fixed muscles were further rinsed in buffer,
post-fixed in buffered 2% osmium tetroxide (OsO4), and
the block was then stained in saturated uranyl acetate and embedded
in Epon 812. Sections (~40 nm thick) were cut using a Leica
Ultracut R microtome (Leica Microsystems, Vienna, Austria) using a
Diatome diamond knife (Diatome Ltd., Biel, Switzerland) and stained
with lead citrate solution. The sections were imaged in using a
Phillips 410 electron microscope (Philips Electron Optics, Mahwak,
NJ, USA) with a Hamamatsu C4742-95 digital camera (Advanced
Microscopy Techniques, Chazy, NY, USA).
Some fibers were separated from the mouse K-acetate
treated biopsy bundles following osmium fixation. After gentle
teasing to separate them, the fibers were whole-mounted in 100%
glycerol under a coverslip and imaged using a Nikon microscope
equipped with phase contrast optics and a Nikon Digital Sight
DS-Fi1 camera.
The canonical number of three individuals for the
human samples is sufficient to assess the quality of structural
preservation based on the criteria shown.
Results
Effects of extracellular K and Cl on
muscle ultrastructure
The initial immersion of the small biopsy samples in
a ‘high potassium’ balanced salt solution requires some caution, as
it is known that the exposure of a cell (or muscle fiber) to a
solution in which sodium has been replaced by potassium,
instantaneously depolarizes the cell and subsequent to
depolarization, the cell will take up permeant anions (chloride
ions if present) through the sarcolemma from the extracellular
space, thereby swelling (8).
Thus, it is important to substitute the extracellular space anion
with one that is not permeable through the plasmalemma (e.g.,
acetate). Fig. 2 graphically
illustrates the effects of exposing a whole, undamaged mouse muscle
to a solution containing 150 mM of either K acetate (Fig. 2A) or KCl (Fig. 2B). In the K acetate solution, the
muscle fibers have the classical well preserved structural
appearance. Triads (arrowheads) are located at the A–I junctions,
the sarcoplasmic reticulum (SR) elements in between are in the form
of a continuous network (reticulum) constituted of elongated
tubules, with slightly wavy but mostly longitudinal orientations.
The mitochondria (M) have a compact structure. The cross striation
is orderly and there are no excessive empty spaces between the
myofibrils. By contrast, Fig. 2B,
shows the damaging effects of exposure to a high KCl solution. The
most obvious effect is the swelling and vacuolization of the SR;
the whole network is fragmented into large empty vacuoles. Some
triads (arrowheads) are still partially identifiable, but the
calsequestrin-filled cisternae are often enlarged into spherical
sacs (short arrows). The mitochondria (M) have dilated cristae, and
the normal close proximity between the mitochondria and triads
(9) is markedly affected. Large
empty spaces are also created between the myofibrils (data not
shown). Despite these changes, the cross striation remains well
aligned.
Orderly structure of muscle fibers from
TPNB biopsy at the light microscope level
If appropriately handled, as described in Materials
and methods, the small biopsy specimens show long stretches of
fibers that maintain excellent order, with no indication of
distortions or contractures. This is shown in whole mount
preparations of fibers teased from fixed mouse biopsies and
observed under phase contrast optics, as described in the study by
Boncompagni et al(10).
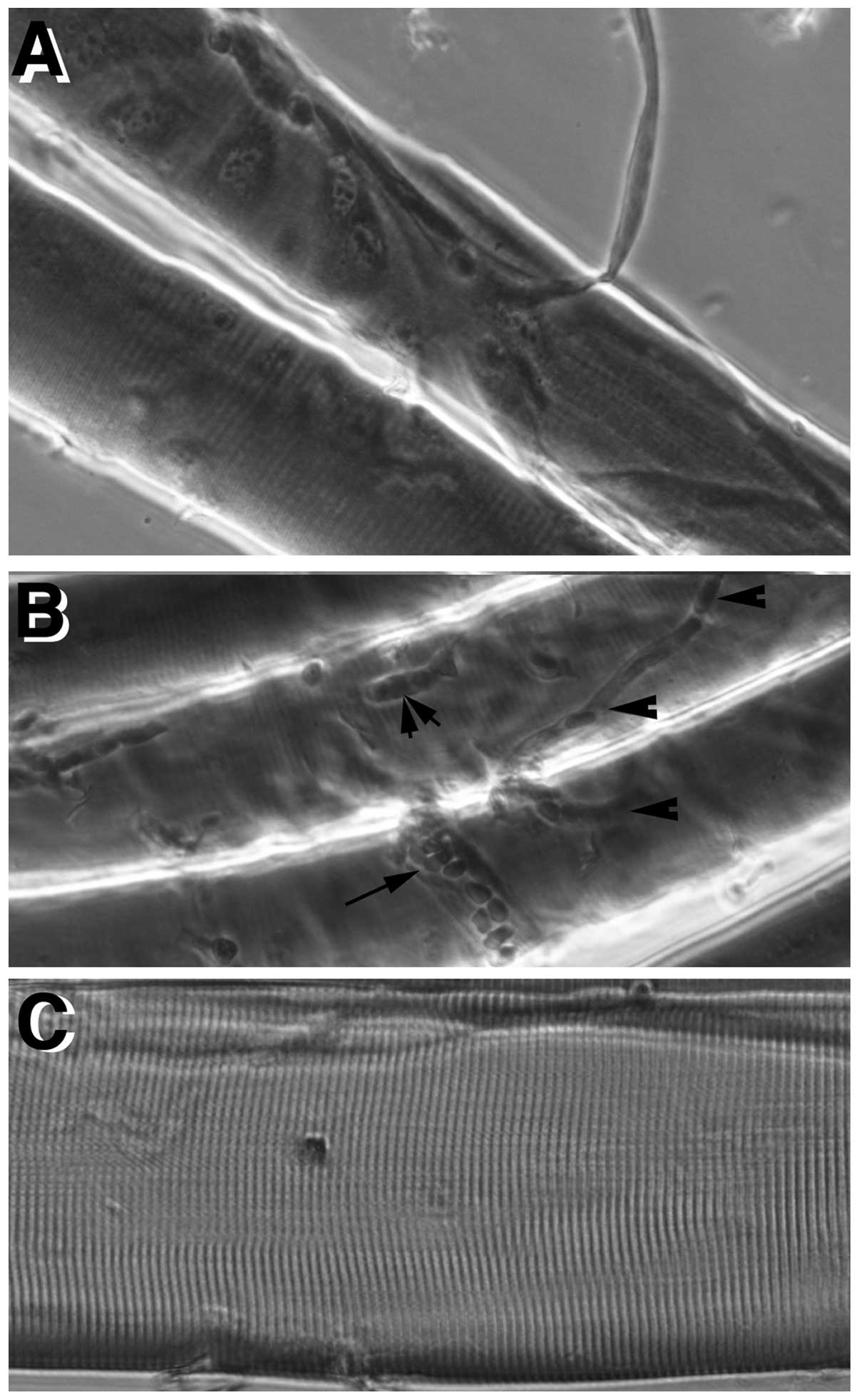
Fig. 3A shows terminal branches
of nerve endings in proximity of neuromuscular synapses; Fig. 3B illustrates small capillaries
(arrowheads), a slightly larger blood vessel filled with blood
cells (small arrow) and a satellite cell (double arrow-head) over
the fiber surface. Noticeably, cross striation has a very regular
spacing (barely visible in Fig. 3A
and B, but clearly visible in Fig. 3C).
Optimum preservation of muscle
ultrastructure is possible in TPNB samples
Successful electron microscopy depends on the
appropriate orientation of the muscle fibers in the embedding and
this was obtained by our technique of selecting small ordered
bundles of fibers from the center of the biopsy sample, and
improving their orientation by gently ‘combing’ and mechanically
stretching the bundle. The image of the mouse biopsy samples shown
in Fig. 4A confirms well aligned
cross striations and an orderly arrangement of the mitochondria,
observed as dense structures aligned on either side of the Z
line.
At higher magnification (Fig. 4B) the ordered sarcomeres show a
well defined I band and a sharp edge of the A band. Triads (triple
arrows) are well positioned and show the appropriate association
between the central T tubules and the two SR lateral sacs. Most
important is the fact that the continuity of the SR network and the
detailed positioning of its longitudinal tubules, fenestrated
collar opposite the centre of the A band and terminal cisternae at
the triads are well preserved. This is clearly visible where the SR
is cut tangentially near the mitochondria. The close association
between the mitochondria and the lateral sacs of the triad
[Boncompagni et al(9)] is
maintained. Fragmentation and vesiculation of the SR of the type
shown in Fig. 2 is not present in
the central regions of the biopsy bundle treated with K acetate,
although these alterations of course occur very close to the cut
ends.
Biopsy samples obtained from human
subjects by TPNB
The TPNB human biopsy samples shown in Fig. 5 were discarded from the needle
into a ‘K acetate’ solution and treated as the mouse biopsies. Low
magnification images of TPNB samples (Fig. 5A) show the regular striation
pattern of the fibers and no sign of local contractures as already
evinced by light microscopy, although the final sarcomere length
varied between 2.3 and 3.0 μm in the various samples. This is due
to different amounts of stretching during the preparation step,
prior to fixation, a good indication that the fibers were relaxed
at that point. An advantage of this approach is that the amount of
stretching applied to the small muscle bundles can be modulated by
the operator during the pre-fixation step.
The details of the sarcomeres confirm optimal
preservation, showing well aligned bands and well preserved
sarcomere details, with aligned filaments, a fairly straight Z line
and a prominent M line (Fig. 5B).
The architecture of the membrane components is retained: the triads
(Fig. 5C, arrows) have the
appropriate orientation at the edges of the A band and the
mitochondria (M) are appropriately positioned near them. The SR is
well differentiated into terminal cisternae, filled with
calsequestrin, and a tubular longitudinal SR segment. The empty
spaces between the SR elements contain glycogen granules (data not
shown), lost during the post-fixation protocol.
Discussion
In this study, we describe a simple procedure for
preserving the ultrastructural details and for appropriately
orienting the muscle fibers for electron microscopy observation
within muscle bundles obtained by small needle muscle biopsy (TPNB
procedure). We focused on TPNB as it presents an ultimate challenge
to ultrastructural preservation due to the very small size of the
sample. The procedure that we devised can of course be applied to
standard needle biopsy samples obtained with larger size needles.
It was generally considered that short damaged segments of fibers
fixed without restraint would be liable to be distorted and thus
unusable for analysis. The novelty of our procedure is in keeping
the fibers in the biopsy sample quiescent by depolarizing them in a
high potassium solution and in using careful micromanipulation in
order to obtain small, but well oriented bundles of fibers that are
straight and stretched to various sarcomere lengths. We also used
EGTA to chelate extracellular calcium, thus decreasing the
contracting effect of the prolonged depolarization. Due to the slow
diffusion of calcium within the muscle cytoplasm (0.014
μm2/msec) (11) we do
not expect entry of extracellular calcium from the cut ends to
induce contractures within a few microns from the cut surface.
TPNB is a relatively easy, rapid and inexpensive
method of obtaining samples using semiautomatic needles developed
for clinical investigations of other organs; however, thus far it
has not been implemented for basic human muscle research. If used
for muscle biopsies, the procedure that we developed depends on a
strict collaboration between the surgeon and an electron
microscopist who has access to a good dissecting microscope within
a very short period of time and is capable of fine
micromanipulations. Two important considerations are that the small
bundles of fibers to be fixed and embedded should be well oriented
and that the solution used for depolarizing the fibers should
contain an impermeant anion in place of Cl− in order to
avoid fiber swelling. If a fairly immediate means of dissection is
not available, the biopsy samples should be immersed in the high K
acetate solution and aerated until ready for use. A prolonged delay
is not advisable.
The value of the TPNB procedure lies not only in its
feasibility, but also in its minimal invasiveness, which makes it
particularly useful for vertical studies of muscle adaptation, such
as in athletes. In addition, we visualize the technique as being
particularly appropriate for the sampling of sarcopenic skeletal
muscle of elderly subjects. Sarcopenia is a condition that is
defined by progressive atrophy of the skeletal muscle (12), although it is not yet fully
understood, particularly in terms of the ultrastructural
adaptation. This has largely been due to the difficulties in
obtaining samples from fragile subjects, such as the elderly and in
pathological conditions resulting in severe muscle atrophy where
the sampling has to be reduced as much as possible. However, the
usefulness of the procedure is limited in the case of some
pathological conditions where large spatial variations in the
distribution of connective tissue and fat exist, so that individual
small biopsyies may collect little muscle tissue. The problem can
be solved by collecting several samples, taking advantage of the
minimal trauma involved in each, and using echographic imaging.
Acknowledgements
The authors wish to thank Dr Luigi D’Amelio and Dr
Vittore Verratti for the surgical procedures. This study was
supported by grants [NIH AR055104 to K.G. Beam, subcontract to
C.F.A and WP 1.5.02 GMP-A.S.I (Italian Space Agency) to G.F–I].
Abbreviations:
|
EGTA
|
ethylene glycol tetraacetic acid
|
|
NAB
|
needle aspiration biopsy
|
|
NMR
|
nuclear magnetic resonance
|
|
SR
|
sarcoplasmic reticulum
|
|
TPNB
|
tiny percutaneous needle biopsy
|
|
VL
|
vastus lateralis
|
References
|
1
|
Pietrangelo T, D’Amelio L, Doria C,
Mancinelli R, Fulle S and Fano G: Tiny percutaneous needle biopsy:
an efficient method for studying cellular and molecular aspects of
skeletal muscle in humans. Int J Mol Med. 27:361–367. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bergstrom J: Muscle electrolytes in man.
Scand J Clin Lab Invest. 14:5111962.
|
|
3
|
Bergstrom J: Percutaneous needle biopsy of
skeletal muscle in physiological and clinical research. Scand J
Clin Lab Invest. 35:609–616. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mancinelli R, Pietrangelo T, La Rovere R,
et al: Cellular and molecular responses of human skeletal muscle
exposed to hypoxic environment. J Biol Regul Homeost Agents.
25:635–645. 2011.PubMed/NCBI
|
|
5
|
Pietrangelo T, Mancinelli R, Toniolo L, et
al: Effects of local vibrations on skeletal muscle trophism in
elderly people: mechanical, cellular, and molecular events. Int J
Mol Med. 24:503–512. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Pietrangelo T, Mancinelli R, Toniolo L, et
al: Transcription profile analysis of vastus lateralis muscle from
patients with chronic fatigue syndrome. Int J Immunopathol
Pharmacol. 22:795–807. 2009.PubMed/NCBI
|
|
7
|
Engel AG: The muscle biopsy. Myology.
Engel AG and Franzini-Armstrong C: 1. 2nd edition. McGraw-Hill; New
York, NY: pp. 822–831. 1994
|
|
8
|
Hodgkin AL and Horowicz P: The influence
of potassium and chloride ions on the membrane potential of single
muscle fibres. J Physiol. 148:127–160. 1959. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Boncompagni S, Rossi AE, Micaroni M, et
al: Mitochondria are linked to calcium stores in striated muscle by
developmentally regulated tethering structures. Mol Biol Cell.
20:1058–1067. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Boncompagni S, Loy RE, Dirksen RT and
Franzini-Armstrong C: The I4895T mutation in the type 1 ryanodine
receptor induces fiber-type specific alterations in skeletal muscle
that mimic premature aging. Aging Cell. 9:958–970. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kushmerick MJ and Podolsky RJ: Ionic
mobility in muscle cells. Science. 166:1297–1298. 1969. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fulle S, Protasi F, Di Tano G, et al: The
contribution of reactive oxygen species to sarcopenia and muscle
ageing. Exp Gerontol. 39:17–24. 2004. View Article : Google Scholar : PubMed/NCBI
|