Introduction
A wide range of food or dietary supplements that are
derived from plants have been shown to be able to modify the
functions of the central nervous system. Ginsenosides, the
secondary plant metabolites produced by Panax ginseng, are
classified into two major groups in terms of the number and
position of sugar moieties: 20 (S)-protopanaxadiol (PPD) and 20
(S)-protopanaxatriol (PPT) saponins. The diversity of individual
ginsenosides may be responsible for their specific pharmacological
effects (1). Increasing evidence
has indicated the beneficial effects of ginsenosides on the central
nervous system (2–6). However, the majority of these
studies have focused on the beneficial effects of ginsenosides
Rg1 and Rb1.
Over the past three decades, an increasing number of
studies have focused on the correlation between neurogenesis and
memory formation (7–9). The vast majority of these studies
describe neurogenesis in the subregions of the hippocampus [the
subventricular zone and subgranular zone (SGZ)] (10,11), whereas only a few studies have
investigated neuronal survival (12). Several factors that affect
hippocampal neurogenesis cause corresponding changes in cognitive
performance. For example, voluntary running improves performance in
the Morris water maze task by increasing cell proliferation in the
SGZ (13). Aged animals and
animals under stress display impaired memory and learning in
hippocampal-dependent tasks (14–16). Therefore, the putative function of
neurogenesis in the SGZ in learning and memory is considered an
index for the evaluation of substrates that exert beneficial
effects. On the other hand, brain-derived neurotrophic factor
(BDNF) has been shown to promote the differentiation and survival
of neurons in the adult brain (17).
In this regard, only a few studies have reported the
effects of the oral administration of metabolites (18,19) of the two ginsenosides,
Rg1 and Rb1, such as ginsenosides
Rh1, PPT, compound K and PPD. Thus, the question remains
of whether ginsenoside Rh1 can affect learning and
memory ability. This issue is not only of academic interest but
also has a number of practical implications for future research and
product development. Therefore, in the present study, we
investigated the effects of the long-term administration of
ginsenoside Rh1 on memory and learning in the adult
mouse brain.
Materials and methods
Animals and housing conditions
Male ICR mice, 6 months of age were housed in a
temperature-controlled animal room with a 12-h light-dark cycle and
allowed access to food and water ad libitum. All experiments
were performed in strict accordance with the Guide for the Care and
Use of Laboratory Animals issued by the National Institutes of
Health (USA), and were approved and monitored by the Ethics
Committee of Animal Experiments at Chungnam National University,
Daejeon, Korea. Prior to the experiments, the mice were left
undisturbed for 7 days and were randomly assigned to 1 of 4
experimental groups: i) the saline (0.9% NaCl)-treated group
(n=16); ii) the group treated with 5 mg/kg Rh1 (n=16);
iii) the group treated with 10 mg/kg Rh1 (n=16); and iv)
the behavioral test group (n=30, passive avoidance test; n=30,
water maze test). The behavioral test group was subdivided into 3
groups (control, 5 mg/kg Rh1 and 10 mg/kg
Rh1; 10 mice per group) and the other 3 groups were
subdivided into 2 groups (n=8 per group) for the evaluation of
neurogenesis and cell survival in the hippocampus.
Bromodeoxyuridine (BrdU) and ginsenosides
protocol
Mice in the saline-treated group (0.9% NaCl) and the
ginsenoside (FuSong County Natural Biotechnology, Co., Ltd.,
Fusong, China)-treated groups (Rh1, 5 and 10 mg/kg body
weight) were orally administrated saline and ginsenoside,
respectively for a period of 3 months. Rh1 doses were
converted between adult human (60 kg) and mouse (20 g) body
weights, using the body surface area normalization method, as
previously described (20). The
selected doses corresponded to 2–3 and 5–6 g of ginseng per day in
an adult human (60 kg body weight). Considering that the long-term
intravenous administration of ginsenosides would cause inflammation
or anxiety, we selected oral administration, even though this would
be associated with less bioactivity.
The thymidine analogue, BrdU, is incorporated into
the DNA of dividing cells and can be detected immunohistochemically
in their progeny. The behavioral tests were performed at the end of
the drug administration. To determine the effects of ginsenoside
Rh1 on neurogenesis, BrdU (100 mg/kg body weight) was
administered to the mice twice per day for 3 consecutive days prior
to sacrifice; to determine the effects of ginsenoside
Rh1 on cell survival, BrdU was administered to the mice
twice per day for 3 consecutive days (20 days prior to sacrifice)
during the the 3rd month of the treatment period. Following the
evaluation of neurogenesis and cell survival, the animals (8 per
group) were sacrificed, the brains were excised and the brain
tissue was then subjected to immunohistochemical and protein
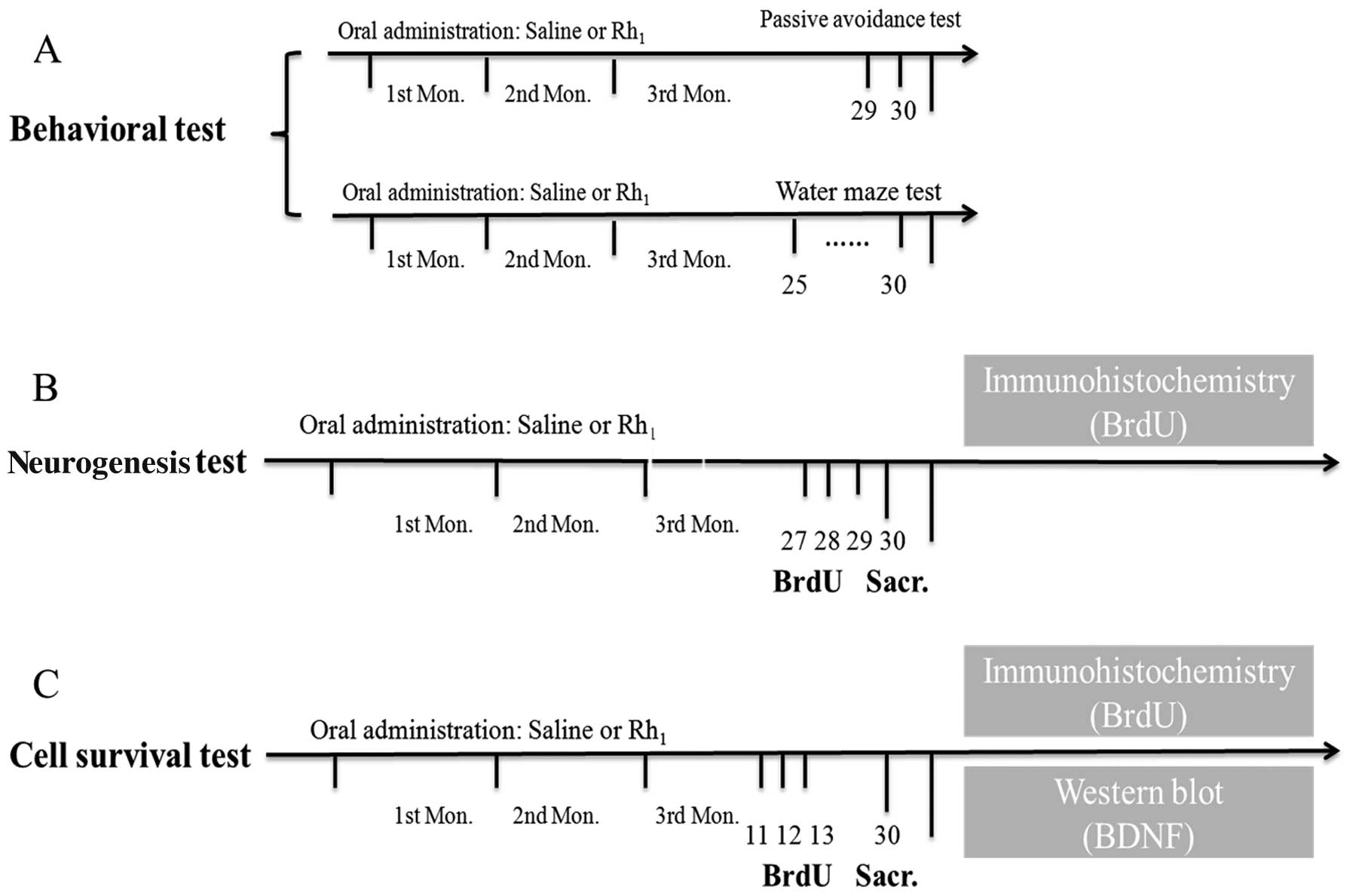
expression analysis. The overall experimental protocol is presented
in Fig. 1.
Morris water maze test
Mice from the different groups were subjected to a
Morris water maze test for 5 consecutive days in the terminal phase
of the administration process. The escape platform (diameter, 10
cm; height, 24 cm) was hidden 1 cm below the surface of the water,
which had been made opaque by the addition of non-toxic black
paint. Each animal was subjected to 4 experimental trails per day,
each lasting 60 sec and each time commencing from 4 different
starting points that randomly varied each day. If an animal was not
able to find the platform it was manually place on it at the end of
the trail. The animals were allowed to rest on the platform for 15
sec. A probe test was performed on day 6.
Passive avoidance test
The passive avoidance test was performed in
identical compartments. The illuminated compartment (20×20×20 cm)
contained a 100 W bulb, and the floor of the non-illuminated
compartment (20×20×20 cm) was composed of 2 mm stainless steel rods
at 1 cm intervals. These 2 compartments were separated by a
guillotine door (5×5 cm). For the acquisition trials, the mice were
initially placed in the illuminated compartment and the door was
opened 15 sec later. When the mouse entered the non-illuminated
compartment, the door was closed and an electrical foot shock (0.5
mA) of 3 sec in duration was delivered through the stainless steel
rods. Twenty-four hours after the acquisition trial, the mice were
again placed in the illuminated compartment for the retention
trials. The time taken for a mouse to enter the non-illuminated
compartment after the opening of the door was termed as the
step-through latency time in the retention trials. If a mouse did
not enter the non-illuminated compartment within 180 sec, it was
assumed that the mouse had remembered the single training
trial.
Immunohistochemistry
The mice were sacrificed and the brains were removed
after the final BrdU injection. The brains were fixed in 4%
phosphate-buffered paraformaldehyde for 12 h. The brain tissues
were then embedded in paraffin and cut into sections. The sections
were mounted on glass slides and stored overnight at 42°C.
Following deparaffinization with xylene and rehydration in a graded
series of ethanol, the sections were rinsed in 0.01 M
phosphate-buffered saline (PBS). BrdU is a widely used S-phase
marker of neurogenesis.
For BrdU-immunostaining, the sections were
hydrolyzed with 2 N hydrochloride (HCl) in PBS (pH 7.4) at 37°C for
15 min, and then stained using the Invitrogen BrdU staining kit
(Invitrogen, Carlsbad, CA, USA). The sections were incubated in
serum blocking solution, in a 1:50 dilution of a mouse monoclonal
antibody against BrdU (Santa Cruz Biotechnology, Inc., Santa Cruz,
CA, USA) and incubated overnight at 4°C in biotinylated secondary
antibody at room temperature for 30 min, and finally in
streptavidin-peroxidase conjugate at room temperature for 20 min.
After each step, the sections were rinsed with PBS. The sections
were then incubated in 3,3′-diaminobenzidine (DAB) solution.
Subsequently, the sections were incubated in 1% ferric chloride
solution at room temperature for 5 min. BrdU-positive nuclei
exhibited deposits of dark brown or black-colored precipitates. The
sections were counterstained with hematoxylin and cover-slipped
with histomount.
Microscopy and cell counting
Every 10th section throughout the hippocampus was
processed for BrdU immunohistochemistry. Using this spacing ensures
that the same neuron will not be counted in two sections. All
BrdU-labeled cells in the dentate gyrus (granule cell layer) and
hilus were counted in each section. To distinguish single cells
within clusters, all counts were performed at ×400 magnification
under a light microscope (Olympus BX-41; Olympus, Tokyo, Japan),
omitting cells in the outermost focal plane. A cell was counted as
being in the SGZ of the dentate gyrus if it was touching or in the
SGZ. Cells that were located more than two cells away from the SGZ
were classified as hilar. The total number of BrdU-labeled cells
per section was determined and multiplied by 6 to obtain the total
number of cells per dentate gyrus.
Western blot analysis
The mice were anesthetized and decapitated after 3
months of treatment. The hippocampus dissected from each animal was
homogenized ultrasonically in protein extraction buffer (PRO-PREP™
17081; iNtRON Biotechnology, Seongnam, Korea). The supernatant was
collected after centrifugation at 15,000 rpm for 30 min at 4°C.
Following quantification, the samples (20 μg protein per lane) were
subjected to preparative sodium dodecyl sulfate-polyacrylamide gel
electrophoresis in a 15% gel and electrophoretically transferred
onto PVDF membranes (Millipore, Billerica, MA, USA) using a
trans-blot device (Bio-Rad, Hercules, CA, USA) at a 15 V constant
current overnight at 4°C. The PVDF membranes were soaked in 5% skim
milk in PBS solution for 2 h at room temperature to block
non-specific binding, rinsed in PBST, and incubated with a rabbit
polyclonal anti-SNAP-25 antibody (diluted 1:300 in 5% skim milk in
TBST; Santa Cruz Biotechnology, Inc.) overnight at 4°C. The
membranes were then washed 3 times for 10 min each in PBST and
incubated for 2 h with a secondary antibody, goat anti-rabbit IgG
(1:10,000; Santa Cruz Biotechnology, Inc.). After washing twice for
15 min in PBST, the signal was detected using an ECL system.
Western blot analysis for β-actin was performed using the same
procedure using a goat polyclonal anti-actin antibody (1:1,000;
Santa Cruz Biotechnology, Inc.) as the primary antibody. The blots
were quantified using image analysis software (ImageJ). Bond
intensity values were expressed as a percentage of the control
average.
Statistical analysis
Data are expressed as the means ± SEM. Statistical
differences were assessed by one-way analysis of variance (ANOVA)
using repeated measures where appropriate. The post-hoc Duncan’s
test was carried out where appropriate. The level for a
statistically significant difference was set at P<0.05.
Results
Behavioral test
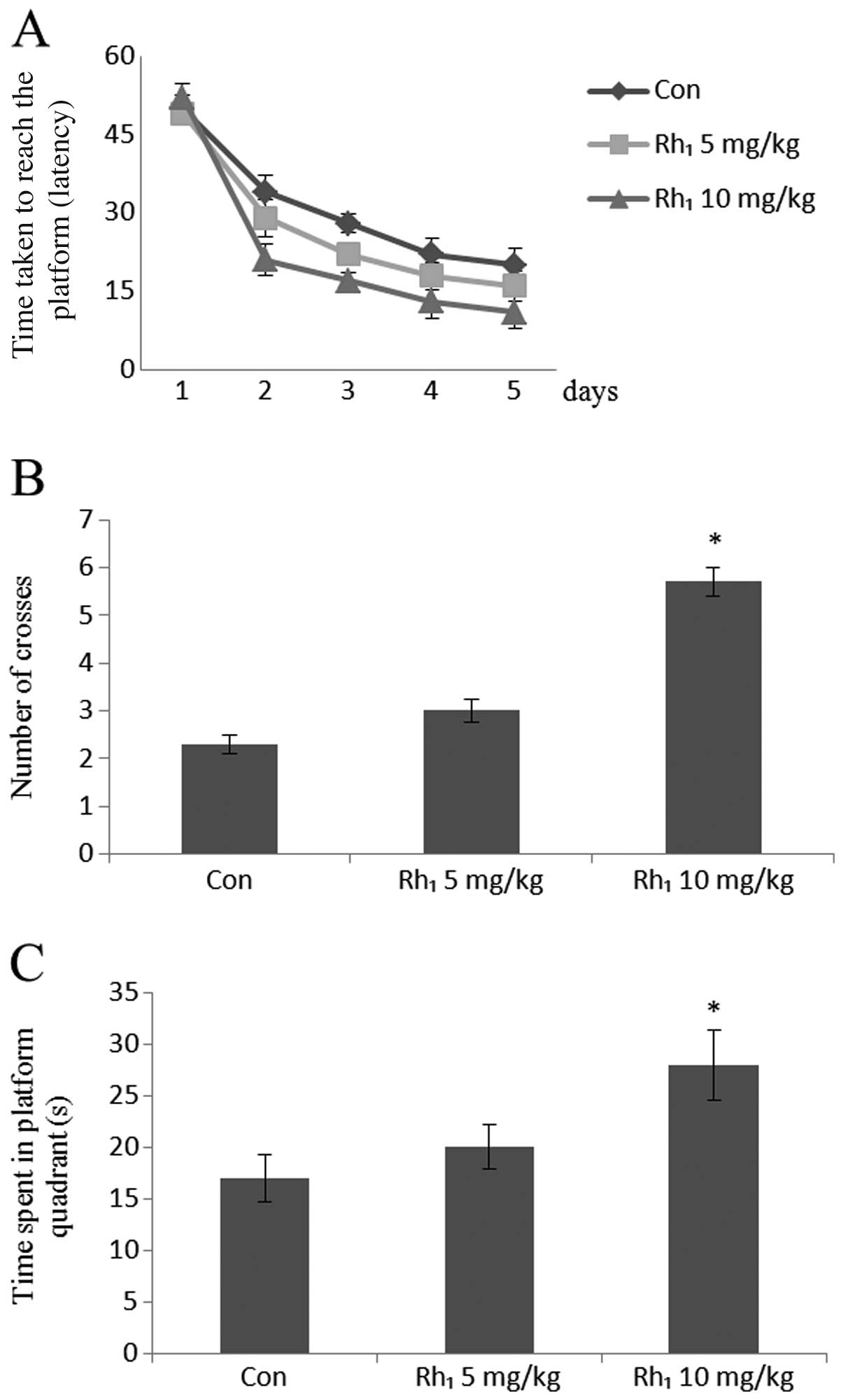
For the Morris water maze, repeated ANOVA (time ×
group) revealed a significant effect decrease in escape latency (in
days) following treatment with Rh1 [5 mg/kg,
F(2,24)=3.72, P=0.067; 10 mg/kg, F(3,42)=11.65, P<0.0001].
In fact, all groups, including the control group
showed a general decrease in overall latency throughout the
acquisition phase (Fig. 2A).
In the probe tests, the group treated with 10 mg/kg
ginsenoside Rh1 showed a significant increase in the
number of crosses (number of times mouse crosses the platform
location) in the target quadrant (P<0.05), whereas the group
treated with 5 mg/kg ginsenoside Rh1, did not show
statistically significant results (P>0.48) compared with the
control group (Fig. 2B).
ANOVA for the time spent in the platform quadrant
yielded significant results for the groups treated with
Rh1 [5 mg/kg, F(1,14)=13.62, P=0.071; 10 mg/kg,
F(1,14)=17.85, P<0.002]. Post-hoc comparisons revealed that a
general increase in the time spent in the target quadrant
throughout the acquisition phase (Fig. 2C).
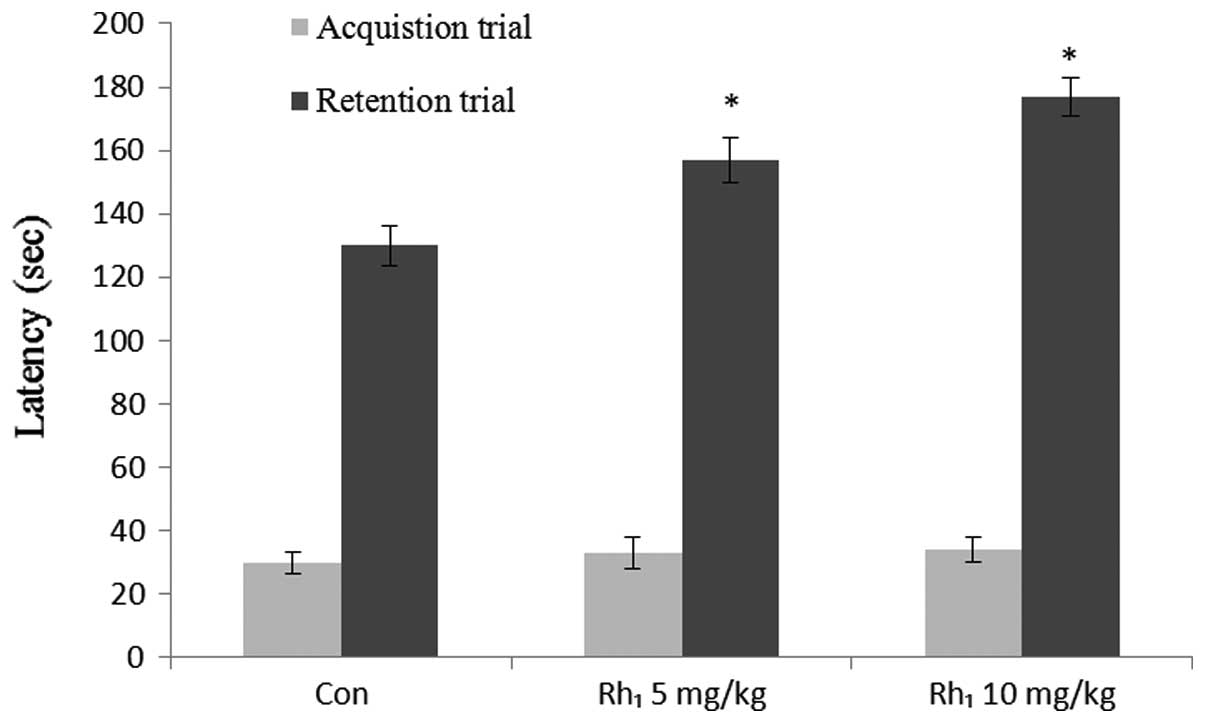
For the passive avoidance tests, no differences were
observed among all the groups in the step-through latency during
the acquisition trials (Fig. 3).
ANOVA for the step-through latency during the retention trials
revealed significant differences among the groups treated with
Rh1 [5 mg/kg, F(1,16)=0.59, P<0.031; 10 mg/kg,
F(1,16)=0.40, P<0.01].
Effects of long-term ginsenoside
Rh1 administration on cell proliferation in the mouse
hippocampus
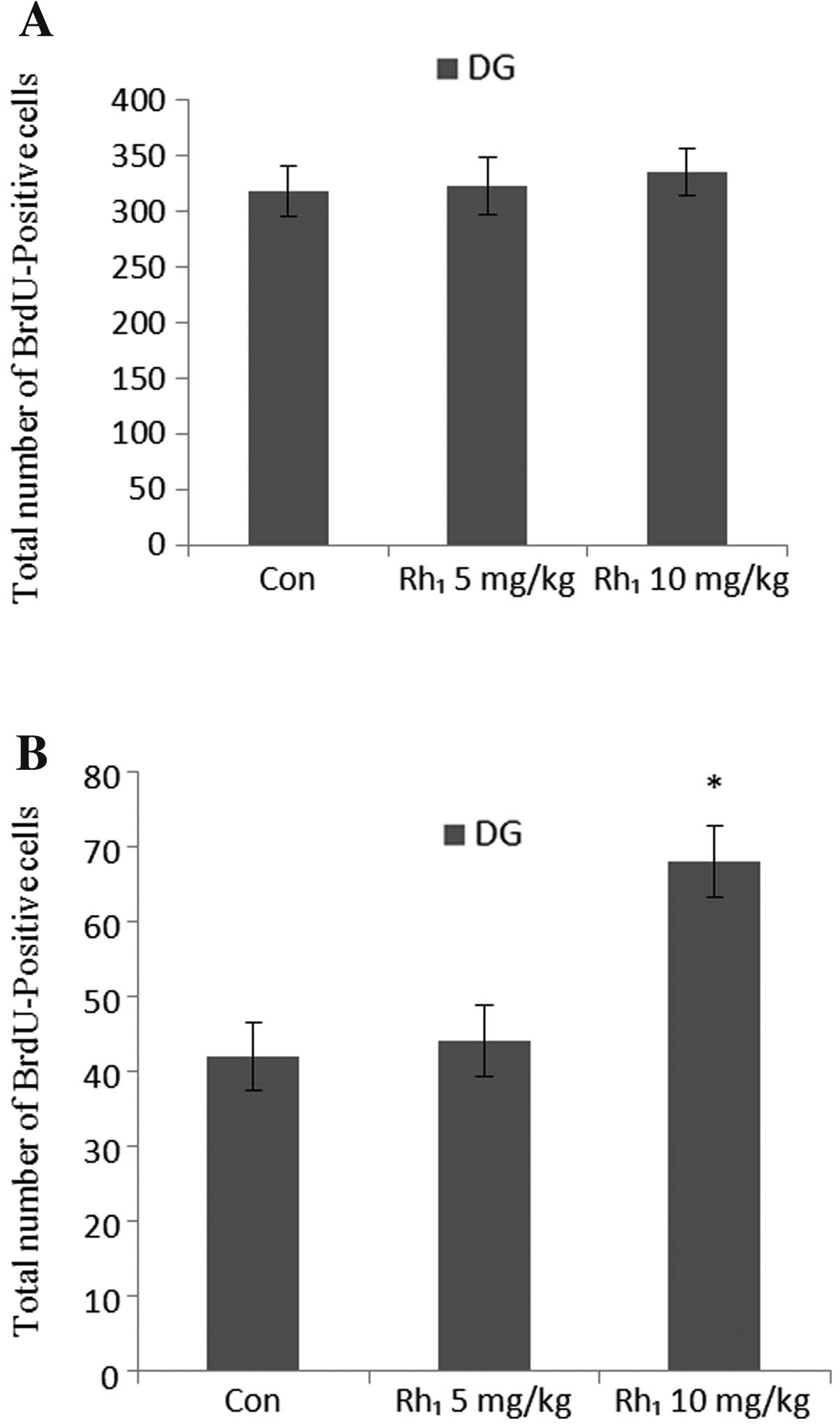
The animals were administered ginsenoside
Rh1 for 3 months and sacrificed after the final BrdU
injection. Analysis of the number of BrdU-labeled cells
demonstrated that the long-term administration of ginsenoside
Rh1 had no statistically significant effect on the
number of BrdU-positive cells in the dentate gyrus (5 mg/kg,
P>0.30; 10 mg/kg, P>0.47) (Fig.
4A).
Effects of long-term ginsenoside
Rh1 administration on cell survival in the mouse
hippocampus
To specially determine the effects of long-term
ginsenoside Rh1 administration on cell survival, a BrdU
injection was administered on the 1st day of the 3rd month. ANOVA
revealed an overall significant effect in the number of
BrdU-positive cells [F(2,18)=51.87, P<0.0003], and post-hoc
tests revealed that treatment with 10 mg/kg Rh1 yielded
significant results (P<0.05) compared with the control group
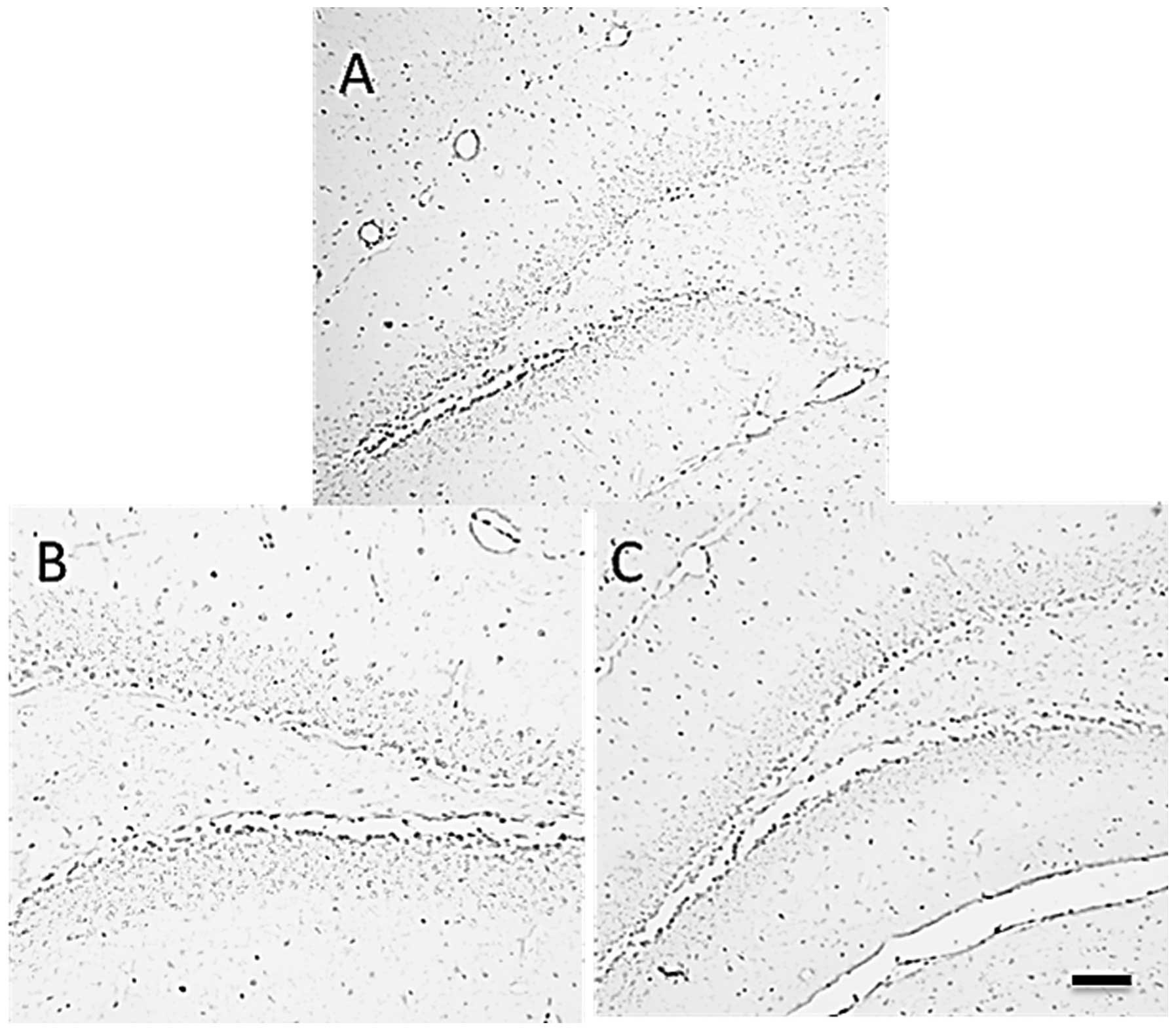
(Fig. 4B). The results of BrdU
immunohistochemistry are presented in Fig. 5. These results suggest that the
long-term administration of Rh1 increased cell survival
in the hippocampus.
Effects of long-term ginsenoside
Rh1 administration on BDNF expression levels
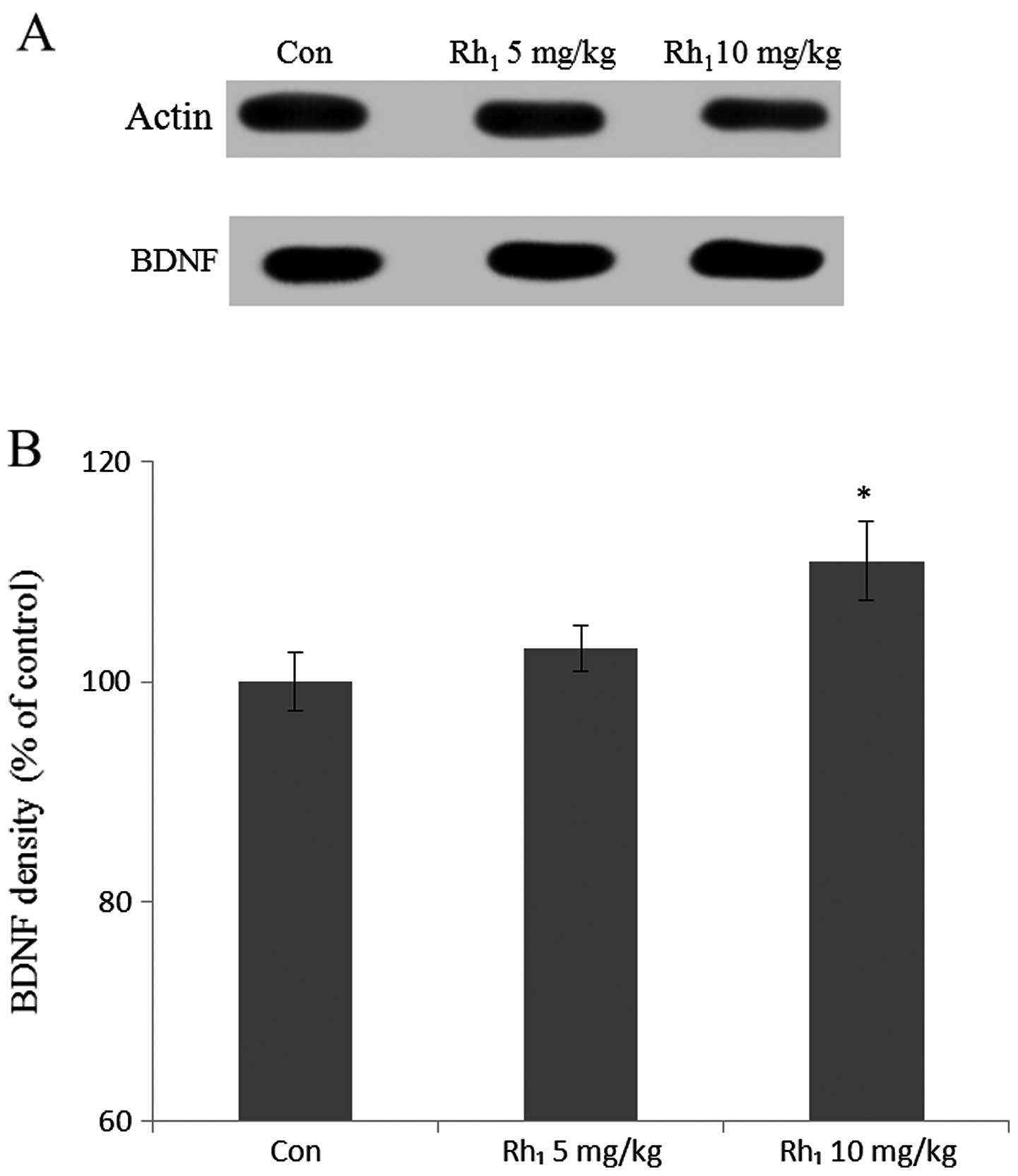
The mice were sacrificed and BDNF protein expression
was quantified by western blot analysis. BDNF density was measured
in the hippocampus (Fig. 6). BDNF
density in the control group was 100±2.8% and in the groups treated
with 5 mg/kg and 10 mg/kg Rh1 was 103.2±2.4 and 112±3.7%
of the control, respectively. Treatment with 10 mg/kg
Rh1 yielded statistically significant results compared
with the control group (P<0.05).
Discussion
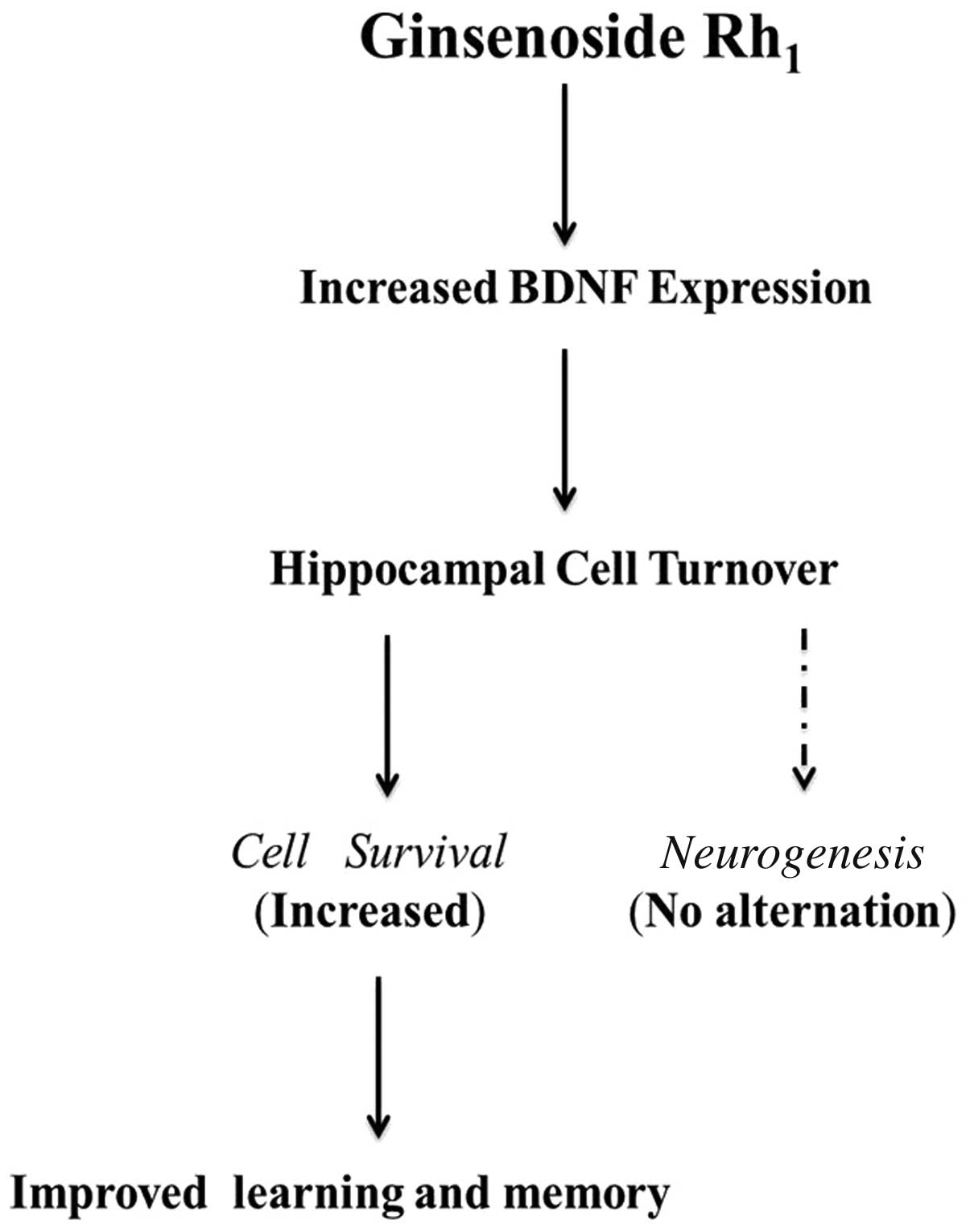
In this study, we report that the long-term
administration of ginsenoside Rh1 enhances spatial
recognition memory, as shown by a Morris water maze test and a
passive avoidance test. Both of these tasks require the involvement
of the hippocampus; thus, we observed a significant increase in
hippocampal cell survival in the treated animals, as shown by the
increase in the number of BrdU-labeled cells. These findings are
consistent with those from a previous study, demonstraring that the
long-term administration of ginsenoside Rb1 enhanced spatial
learning and memory (21). To the
best of our knowledge, only a few studies have reported the
pharmacological functions of ginsenoside Rh1 and PPT
(22) in learning and memory. In
the present study, we invesgigated the potential pharmacological
effects of ginsenoside Rh1 (Fig. 7).
In fact, a relatively higher dose of Rh1
is required for optimal memory and learning in a water maze task.
However, we observed a trend for the enhancing effects on memory to
be more pronounced in the group administered the higher dose vs.
the group administered the lower dose, although there was no
significant difference observed between the 2 Rh1
treatment groups in the passive avoidance test.
The regulation of neurogenesis can occur at
different stages, including cell proliferation, differentiation and
survival. Several experimental methods have been conducted to
investigate the role of adult hippocampal neurogenesis in learning
and memory, such as low-dose brain irradiation (23,24), various types of stress (25) and methylazoxymethanol acetate
treatment (8). These methods have
been shown to significantly reduce neurogenesis, as well as
hippocampal-dependent tasks, whereas these studies suggest some
potential roles for neurogenesis in learning and memory. In
addition, the decreased survival of proliferating cells in the
hippocampus is associated with a decline in spatial memory, as
observed in a previous study (26). Hence, to elucidate the memory
enhancing effects of treatment with ginsenoside Rh1, we
hypothesized that this treatment would have an impact on
hippocampal neurogenesis. A significant increase in cell survival
in the hippocampus was observed in the treated groups compared with
the control group. By contrast, the number of BrdU-positive cells
in the hippocampus did not differ between the treated groups and
the control group, suggesting that cell proliferation at the time
of BrdU injection was unaffected. Nonetheless, further studies are
required to determine whether the treatment-induced increase in
cell survival underlies the enhancement of memory and learning
which was observed.
It is also possible that this increase was
consistent with the mechanisms observed in previous studies, as in
the central nervous system, BDNF regulates neuronal activity and is
important for the positive selection and survival of functionally
active neurons (27) and protects
newborn neurons from death during the differentation process from
immature to mature neurons (28).
In the present study, ginsenoside Rh1 enhanced the
survival of cells in the dentate gyrus following the increase in
BDNF expression. In this perspective, this result may partly
explain the mechanisms by which ginsenoside Rh1 improves
the learning and memory process.
Apart from the neurogenesis factor, there are still
other factors affecting the learning and memory process. A critical
role for T cell-derived interleukin (IL)-4 in the regulation of
learning and memory through the meningeal myeloid cell phenotype
and BDNF expression has been indicated (29). Stress and corticosteroid hormones
are known to affect learning and memory processes (30). Plasticity levels in the gray and
white matter of the brain change during the learning process
(31). The activation of
cAMP-response element binding protein (CREB)-gene expression has a
significant impact on memory (32).
On the other hand, Alzheimer’s disease is a
progressive neurologic disease that results in the irreversible
loss of neurons, particularly in the cortex and hippocampus.
Parkinson’s disease is the second most common neurodegenerative
disorder, after Alzheimer’s disease. It is characterized
pathologically by the loss of neurons. According to a previous
study (33), ginsenoside
Rg1 significantly inhibits β-secretase activity in
vitro and protects against Aβ-induced cytotoxicity in PC12
cells. In our study, ginsenoside Rh1, a metabolite of
the major ginsenoside Rg1, demonstrated great potential
as a therapeutic agent by promoting cell survival.
In conclusion, the long-term administration of
ginsenoside Rh1 resulted in improved behavioral
performance in hippocampal-dependent tasks. Although ginsenoside
Rh1 is able to promote cell survival in the dentate
gyrus of the mouse hippocampus, it is likely that a combination of
increased cell survival, as well as unknown factors, contribute to
the enhanced performance induced by the long-term administration of
ginsenoside Rh1. Further studies are required for the
analysis of the differentiation of survived cells and other
possible factors.
References
|
1
|
Christensen LP: Ginsenosides chemistry,
biosynthesis, analysis, and potential health effects. Advances in
Food and Nutrition Research. Taylor S: 55. Academic Press; New
Yotk: pp. 1–99. 2008
|
|
2
|
Van Kampen J, Robertson H, Hagg T and
Drobitch R: Neuroprotective actions of the ginseng extract G115 in
two rodent models of Parkinson’s disease. Exp Neurol. 184:521–529.
2003.PubMed/NCBI
|
|
3
|
Kennedy DO and Scholey AB: Ginseng:
potential for the enhancement of cognitive performance and mood.
Pharmacol Biochem Behav. 75:687–700. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Reay JL, Scholey AB and Kennedy DO: Panax
ginseng (G115) improves aspects of working memory performance and
subjective ratings of calmness in healthy young adults. Hum
Psychopharmacol. 25:462–471. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kim EJ, Jung IH, Van Le TK, Jeong JJ, Kim
NJ and Kim DH: Ginsenosides Rg5 and Rh3 protect scopolamine-induced
memory deficits in mice. J Ethnopharmacol. 146:294–299. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Quan Q, Wang J, Li X and Wang Y:
Ginsenoside Rg1 decreases Aβ(1–42) level by upregulating PPARγ and
IDE expression in the hippocampus of a rat model of Alzheimer’s
disease. PloS One. 8:e591552013.
|
|
7
|
van Praag H, Schinder AF, Christie BR,
Toni N, Palmer TD and Gage FH: Functional neurogenesis in the adult
hippocampus. Nature. 415:1030–1034. 2002.PubMed/NCBI
|
|
8
|
Shors TJ, Miesegaes G, Beylin A, Zhao M,
Rydel T and Gould E: Neurogenesis in the adult is involved in the
formation of trace memories. Nature. 410:372–376. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Aimone JB, Wiles J and Gage FH: Potential
role for adult neurogenesis in the encoding of time in new
memories. Nat Neurosci. 9:723–727. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Johansson CB, Svensson M, Wallstedt L,
Janson AM and Frisén J: Neural stem cells in the adult human brain.
Exp Cell Res. 253:733–736. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Leuner B, Gould E and Shors TJ: Is there a
link between adult neurogenesis and learning? Hippocampus.
16:216–224. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Drapeau E, Montaron MF, Aguerre S and
Abrous DN: Learning-induced survival of new neurons depends on the
cognitive status of aged rats. J Neurosci. 27:6037–6044. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Olson AK, Eadie BD, Ernst C and Christie
BR: Environmental enrichment and voluntary exercise massively
increase neurogenesis in the adult hippocampus via dissociable
pathways. Hippocampus. 16:250–260. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Klempin F and Kempermann G: Adult
hippocampal neurogenesis and aging. Eur Arch Psychiatry Clin
Neurosci. 257:271–280. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mirescu C and Gould E: Stress and adult
neurogenesis. Hippocampus. 16:233–238. 2006. View Article : Google Scholar
|
|
16
|
Rinwa P and Kumar A: Piperine potentiates
the protective effects of curcumin against chronic unpredictable
stress-induced cognitive impairment and oxidative damage in mice.
Brain Res. 1488:38–50. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Quadrato G, Benevento M, Alber S, et al:
Nuclear factor of activated T cells (NFATc4) is required for
BDNF-dependent survival of adult-born neurons and spatial memory
formation in the hippocampus. Proc Natl Acad Sci. 109:E1499–E1508.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lai L, Hao H, Liu Y, et al:
Characterization of pharmacokinetic profiles and metabolic pathways
of 20(S)-ginsenoside Rh1 in vivo and in vitro. Planta Med.
75:797–802. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Joh EH, Lee IA, Jung IH and Kim DH:
Ginsenoside Rb1 and its metabolite compound K inhibit IRAK-1
activation-the key step of inflammation. Biochem Pharmacol.
82:278–286. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yang L, Zhang J, Zheng K, Shen H and Chen
X: Long-term ginsenoside Rg1 supplementation improves age-related
cognitive decline by promoting synaptic plasticity associated
protein expression in C57BL/6J mice. J Gerontol A Biol Sci Med Sci.
July.5–2013.(Epub ahead of print).
|
|
21
|
Liu L, Hoang-Gia T, Wu H, et al:
Ginsenoside Rb1 improves spatial learning and memory by regulation
of cell genesis in the hippocampal subregions of rats. Brain Res.
1382:147–154. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang YZ, Chen J, Chu SF, et al:
Improvement of memory in mice and increase of hippocampal
excitability in rats by ginsenoside Rg1’s metabolites ginsenoside
Rh1 and protopanaxatriol. J Pharmacol Sci. 109:504–510.
2009.PubMed/NCBI
|
|
23
|
Santarelli L, Saxe M, Gross C, et al:
Requirement of hippocampal neurogenesis for the behavioral effects
of antidepressants. Science. 301:805–809. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Snyder JS, Hong NS, McDonald RJ and
Wojtowicz JM: A role for adult neurogenesis in spatial long-term
memory. Neuroscience. 130:843–852. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Shors TJ, Mathew J, Sisti HM, Edgecomb C,
Beckoff S and Dalla C: Neurogenesis and helplessness are mediated
by controllability in males but not in females. Biol Psychiatry.
62:487–495. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wati H, Kudo K, Qiao C, Kuroki T and Kanba
S: A decreased survival of proliferated cells in the hippocampus is
associated with a decline in spatial memory in aged rats. Neurosci
Lett. 399:171–174. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Thoenen H: Neurotrophins and neuronal
plasticity. Science. 270:593–598. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Bergami M, Rimondini R, Santi S, Blum R,
Götz M and Canossa M: Deletion of TrkB in adult progenitors alters
newborn neuron integration into hippocampal circuits and increases
anxiety-like behavior. Proc Natl Acad Sci USA. 105:15570–15575.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Derecki NC, Cardani AN, Yang CH, et al:
Regulation of learning and memory by meningeal immunity: a key role
for IL-4. J Exp Med. 207:1067–1080. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Schwabe L, Schächinger H, de Kloet ER and
Oitzl MS: Corticosteroids operate as a switch between memory
systems. J Cogn Neurosci. 22:1362–1372. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zatorre RJ, Fields RD and Johansen-Berg H:
Plasticity in gray and white: neuroimaging changes in brain
structure during learning. Nat Neurosci. 15:528–536. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Benito E and Barco A: CREB’s control of
intrinsic and synaptic plasticity: implications for CREB-dependent
memory models. Trends Neurosci. 33:230–240. 2010.
|
|
33
|
Wang YH and Du GH: Ginsenoside Rg1
inhibits β-secretase activity in vitro and protects against
Abeta-induced cytotoxicity in PC12 cells. J Asian Nat Prod Res.
11:604–612. 2009.
|