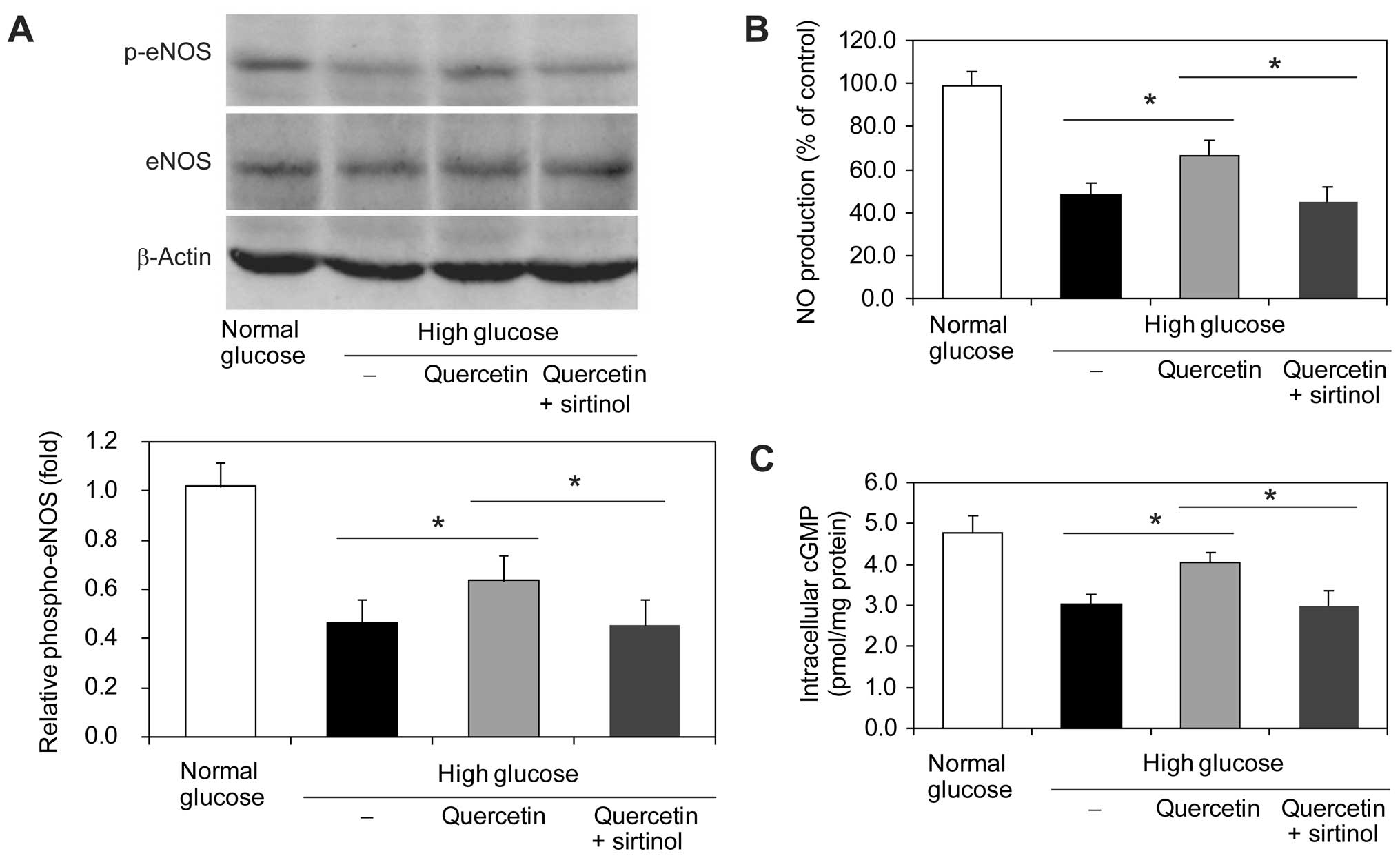
|
1
|
Asahara T, Murohara T, Sullivan A, et al:
Isolation of putative progenitor endothelial cells for
angiogenesis. Science. 275:964–967. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hristov M, Erl W and Weber PC: Endothelial
progenitor cells: mobilization, differentiation, and homing.
Arterioscler Thromb Vasc Biol. 23:1185–1189. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hristov M and Weber C: Endothelial
progenitor cells: characterization, pathophysiology, and possible
clinical relevance. J Cell Mol Med. 8:498–508. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Loomans CJ, de Koning EJ, Staal FJ, et al:
Endothelial progenitor cell dysfunction: a novel concept in the
pathogenesis of vascular complications of type 1 diabetes.
Diabetes. 53:195–199. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Fadini GP, Miorin M, Facco M, et al:
Circulating endothelial progenitor cells are reduced in peripheral
vascular complications of type 2 diabetes mellitus. J Am Coll
Cardiol. 45:1449–1457. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sorrentino SA, Bahlmann FH, Besler C, et
al: Oxidant stress impairs in vivo reendothelialization capacity of
endothelial progenitor cells from patients with type 2 diabetes
mellitus: restoration by the peroxisome proliferator-activated
receptor-gamma agonist rosiglitazone. Circulation. 116:163–173.
2007. View Article : Google Scholar
|
|
7
|
Rosso A, Balsamo A, Gambino R, et al: p53
Mediates the accelerated onset of senescence of endothelial
progenitor cells in diabetes. J Biol Chem. 281:4339–4347. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hamed S, Brenner B and Roguin A: Nitric
oxide: a key factor behind the dysfunctionality of endothelial
progenitor cells in diabetes mellitus type-2. Cardiovasc Res.
91:9–15. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
van Dam RM, Naidoo N and Landberg R:
Dietary flavonoids and the development of type 2 diabetes and
cardiovascular diseases: review of recent findings. Curr Opin
Lipidol. 24:25–33. 2013.PubMed/NCBI
|
|
10
|
Nicolle E, Souard F, Faure P and
Boumendjel A: Flavonoids as promising lead compounds in type 2
diabetes mellitus: molecules of interest and structure-activity
relationship. Curr Med Chem. 18:2661–2672. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Formica JV and Regelson W: Review of the
biology of Quercetin and related bioflavonoids. Food Chem Toxicol.
33:1061–1080. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kim JH, Kang MJ, Choi HN, Jeong SM, Lee YM
and Kim JI: Quercetin attenuates fasting and postprandial
hyperglycemia in animal models of diabetes mellitus. Nutr Res
Pract. 5:107–111. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Margina D, Gradinaru D, Manda G, Neagoe I
and Ilie M: Membranar effects exerted in vitro by polyphenols -
quercetin, epigallocatechin gallate and curcumin - on HUVEC and
Jurkat cells, relevant for diabetes mellitus. Food Chem Toxicol.
61:86–93. 2013. View Article : Google Scholar
|
|
14
|
Mahmoud MF, Hassan NA, El Bassossy HM and
Fahmy A: Quercetin protects against diabetes-induced exaggerated
vasoconstriction in rats: effect on low grade inflammation. PloS
One. 8:e637842013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zahedi M, Ghiasvand R, Feizi A, Asgari G
and Darvish L: Does quercetin improve cardiovascular risk factors
and inflammatory biomarkers in women with type 2 diabetes: a
double-blind randomized controlled clinical trial. Int J Prev Med.
4:777–785. 2013.PubMed/NCBI
|
|
16
|
Marrotte EJ, Chen DD, Hakim JS and Chen
AF: Manganese superoxide dismutase expression in endothelial
progenitor cells accelerates wound healing in diabetic mice. J Clin
Invest. 120:4207–4219. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang P, Xu TY, Guan YF, Su DF, Fan GR and
Miao CY: Perivascular adipose tissue-derived visfatin is a vascular
smooth muscle cell growth factor: role of nicotinamide
mononucleotide. Cardiovasc Res. 81:370–380. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wang P, Guan YF, Du H, Zhai QW, Su DF and
Miao CY: Induction of autophagy contributes to the neuroprotection
of nicotinamide phosphoribosyltransferase in cerebral ischemia.
Autophagy. 8:77–87. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Liu BQ, Du ZX, Zong ZH, et al:
BAG3-dependent noncanonical autophagy induced by proteasome
inhibition in HepG2 cells. Autophagy. 9:905–916. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Garg AD, Dudek AM, Ferreira GB, et al:
ROS-induced autophagy in cancer cells assists in evasion from
determinants of immunogenic cell death. Autophagy. 9:1292–1307.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhang XH, Lei H, Liu AJ, Zou YX, Shen FM
and Su DF: Increased oxidative stress is responsible for severer
cerebral infarction in stroke-prone spontaneously hypertensive
rats. CNS Neurosci Ther. 17:590–598. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Schmeisser H, Fey SB, Horowitz J, et al:
Type I interferons induce autophagy in certain human cancer cell
lines. Autophagy. 9:683–696. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Luciani A, Villella VR, Esposito S, et al:
Targeting autophagy as a novel strategy for facilitating the
therapeutic action of potentiators on DeltaF508 cystic fibrosis
transmembrane conductance regulator. Autophagy. 8:1657–1672. 2012.
View Article : Google Scholar
|
|
24
|
Wang P, Zhang RY, Song J, et al: Loss of
AMP-activated protein kinase-alpha2 impairs the insulin-sensitizing
effect of calorie restriction in skeletal muscle. Diabetes.
61:1051–1061. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang P, Xu TY, Guan YF, et al:
Nicotinamide phosphoribosyltransferase protects against ischemic
stroke through SIRT1-dependent adenosine monophosphate-activated
kinase pathway. Ann Neurol. 69:360–374. 2011. View Article : Google Scholar
|
|
26
|
Oh JM, Choi EK, Carp RI and Kim YS:
Oxidative stress impairs autophagic flux in prion protein-deficient
hippocampal cells. Autophagy. 8:1448–1461. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhang T, Li Y, Park KA, et al:
Cucurbitacin induces autophagy through mitochondrial ROS production
which counteracts to limit caspase-dependent apoptosis. Autophagy.
8:559–576. 2012. View Article : Google Scholar
|
|
28
|
Song YM, Song SO, Jung YK, et al: Dimethyl
sulfoxide reduces hepatocellular lipid accumulation through
autophagy induction. Autophagy. 8:1085–1097. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhao YD, Courtman DW, Deng Y, Kugathasan
L, Zhang Q and Stewart DJ: Rescue of monocrotaline-induced
pulmonary arterial hypertension using bone marrow-derived
endothelial-like progenitor cells: efficacy of combined cell and
eNOS gene therapy in established disease. Circ Res. 96:442–450.
2005. View Article : Google Scholar
|
|
30
|
Duda DG, Fukumura D and Jain RK: Role of
eNOS in neovascularization: NO for endothelial progenitor cells.
Trends Mol Med. 10:143–145. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Mattagajasingh I, Kim CS, Naqvi A, et al:
SIRT1 promotes endothelium-dependent vascular relaxation by
activating endothelial nitric oxide synthase. Proc Natl Acad Sci
USA. 104:14855–14860. 2007. View Article : Google Scholar
|
|
32
|
Chao CL, Hou YC, Chao PD, Weng CS and Ho
FM: The antioxidant effects of quercetin metabolites on the
prevention of high glucose-induced apoptosis of human umbilical
vein endothelial cells. Br J Nutr. 101:1165–1170. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Bournival J, Francoeur MA, Renaud J and
Martinoli MG: Quercetin and sesamin protect neuronal PC12 cells
from high-glucose-induced oxidation, nitrosative stress, and
apoptosis. Rejuvenation Res. 15:322–333. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Shi Y, Liang XC, Zhang H, Wu QL, Qu L and
Sun Q: Quercetin protects rat dorsal root ganglion neurons against
high glucose-induced injury in vitro through Nrf-2/HO-1 activation
and NF-kappaB inhibition. Acta Pharmacol Sin. 34:1140–1148. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chen YH, Lin SJ, Lin FY, et al: High
glucose impairs early and late endothelial progenitor cells by
modifying nitric oxide-related but not oxidative stress-mediated
mechanisms. Diabetes. 56:1559–1568. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kobayashi S, Xu X, Chen K and Liang Q:
Suppression of autophagy is protective in high glucose-induced
cardiomyocyte injury. Autophagy. 8:577–592. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Thum T, Fraccarollo D, Schultheiss M, et
al: Endothelial nitric oxide synthase uncoupling impairs
endothelial progenitor cell mobilization and function in diabetes.
Diabetes. 56:666–674. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Herranz D and Serrano M: SIRT1: recent
lessons from mouse models. Nat Rev Cancer. 10:819–823. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Balestrieri ML, Rienzo M, Felice F, et al:
High glucose downregulates endothelial progenitor cell number via
SIRT1. Biochim Biophys Acta. 1784:936–945. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Servillo L, D’Onofrio N, Longobardi L, et
al: Stachydrine ameliorates high-glucose induced endothelial cell
senescence and SIRT1 downregulation. J Cell Biochem. 114:2522–2530.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Davis JM, Murphy EA, Carmichael MD and
Davis B: Quercetin increases brain and muscle mitochondrial
biogenesis and exercise tolerance. Am J Physiol Regul Integr Comp
Physiol. 296:R1071–R1077. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Casuso RA, Martinez-Lopez EJ, Nordsborg
NB, et al: Oral quercetin supplementation hampers skeletal muscle
adaptations in response to exercise training. Scand J Med Sci
Sports. Oct 14–2013.(E-pub ahead of print).
|