Introduction
Seaweed has been steadily utilized in food
supplements and as human food, feed for fishery organisms and
biofuel (1,2). The Bangiales (Rhodophyta), in
particular Pyropia sp. and Porphyra sp., are cultured
widely in East Asia, including Korea and Japan. Thus far, numerous
studies have concentrated on the availability of seaweed with
regard to the nutritional value of food and medical material.
Previously, the effectiveness of seaweed has become known through
biological activity experiments conducted using seaweed extract
in vivo and in vitro (3–10).
In particular, proteins isolated from Pyropia yezoensis
(formerly known as Porphyra yezoensis) showed a positive
effect on anti-inflammation (9)
and protection from cell toxicity (7). However, the majority of these
studies investigated the expression of physiological activity in
vivo or in vitro. To the best of our knowledge, no study
has reported the association between the molecular structure and
function of seaweed with various bioactive substances.
P. yezoensis is a model marine plant for
physiological and genetic studies in seaweed (11). In addition to expressed sequence
tag (EST) analyses (12,13), the draft nuclear genome sequence
(14) and the complete plastid
genome sequence (15) of P.
yezoensis have recently been published. Hwang et al
(7) reported that the 14-kDa
protein-containing fraction of P. yezoensis has
chemoprotective functions. In the present study, the association
between the chemical structure and chemoprotective activity of the
P. yezoensis protein (PYP) was investigated. PYP components
resolved by sodium dodecyl sulfate-polyacrylamide gel
electrophoresis (SDS-PAGE) were chemically and mass
spectrometrically identified and characterized. The synthetic
N-terminal fragment of the major PYP protein was determined to be a
chemoprotectant. The possible physiological role of PYP and the
mode of chemoprotective action will be discussed.
Materials and methods
Preparation of PYP
The PYP fraction was prepared from P.
yezoensis farmed in Wando Gun (Jeolla Nam-Do, Korea), as
described by Hwang et al (7). In brief, 40 g of powdered P.
yezoensis was dissolved in 1l distilled water (DW) at room
temperature for 3 h. The extract was filtered with a 5-μm Muller
gauze and subsequently three volumes of ethanol were added. The
ethanol extract was filtered (no. 3; Advantec, Tokyo, Japan),
pelleted with ammonium sulfate, dissolved in DW, dialysed in DW
overnight and concentrated at 40°C in a rotary evaporator
(Eyela/Tokyo Rikakikai, Tokyo, Japan). The concentrated solution
was redissolved in double DW and freeze-dried (Eyela FDU-2100; Lab
Corporation, Seoul, Korea). The protein content was determined
using a bicinchoninic acid protein assay kit (Pierce Biotechnology,
Rockford, IL, USA).
Protein sequencing
SDS-PAGE was performed as described by Laemmli
(16) using a 1.5-mm-thick 18%
acrylamide gel. In brief, PYP (20 mg) was dissolved in 1 ml water,
and dark-colored insoluble material was removed by centrifugation
(15,000 × g for 10 min). The clear supernatant (5 μl) was mixed
with an equal volume of Laemmli buffer [125 mM Tris-HCl (pH 6.8),
4% SDS, 20% glycerol, 10% 2-mercaptoethanol and 0.002% bromophenol
blue], and incubated for 5 min at 95°C. SDS-PAGE standard (Broad
Range; Bio-Rad, Hercules, CA, USA) was used as the molecular weight
marker. The molecular weight of each protein band was determined by
the method of Weber and Osborn (17). Electroblotting was performed in 25
mM Tris-HCl (pH 8.3) and 192 mM glycine at 100 V for 30 min using a
Criterion Transblot Cell (Bio-Rad). The polyvinylidene fluoride
(PVDF) membrane Sequi-Blot (Bio-Rad) was used for transfer. Protein
bands were visualized by staining with Coomassie Brilliant Blue
R-250 Staining solution (Bio-Rad) for 1 min and destaining with 50%
methanol for 10 min. Protein bands were excised from the membrane
and destained with 90% methanol prior to protein sequencing. The
N-terminal amino acid sequence and initial yield of
phenylthiohydantoin-amino acid (PTH-AA) of each protein band was
analyzed using a gas phase protein sequencer PPSQ-33A (Shimadzu,
Kyoto, Japan).
Mass spectrometry
PYP proteins separated by SDS-PAGE were visualized
with Coomassie Brilliant Blue G-250 Staining solution in accordance
with the manufacturer’s instructions. The major band (corresponding
to the 10-kDa section in Fig. 1)
was excised using a utility knife. In-gel tryptic digestion and
peptide extraction were performed as previously described (18), except that the destaining step
(prior to in-gel digestion) was omitted to avoid loss of small
proteins. Tandem mass spectrometry (MS/MS) spectra were obtained
using a matrix-assisted laser desorption/ionization-quadrupole ion
trap-time of flight (MALDI-QIT-TOF) mass spectrometer (AXIMA
Resonance; Shimadzu) in the positive mode. All the spectra were
externally calibrated using human angiotensin II (m/z:
1,046.54) and human adrenocorticotropic hormone (18–39) fragment
(m/z: 2,465.20) in a ProteoMass® Peptide &
Protein MALDI-MS Calibration kit (Sigma-Aldrich, St. Louis, MO,
USA). Protein identification was carried out by MS/MS ion searches
using MASCOT® version 2.3 (Matrix Science, London, UK)
against EST_rhodophyta 2011_11 (2,715,402 sequences; 367,566,596
residues) and Pyropia yezoensis CDS (61,962 sequences;
17,483,926 residues), downloaded from the Pyropia yezoensis
Genome Annotation ver. 1, Fisheries Research Agency, Japan
(http://nrifs.fra.affrc.go.jp/ResearchCenter/5_AG/genomes/nori/),
SwissProt 2012_09 (540,958 sequences; 192,206,270 residues,
http://www.uniprot.org/downloads), and
the non-redundant sequence database from the National Center for
Biotechnology Information (NCBInr 20130721; 31,240,556 sequences;
10,789,480,161 residues, ftp://ftp.ncbi.nlm.nih.gov/blast/db/FASTA/nr.gz) in
our own MASCOT server. Search parameters used were as follows:
Enzyme, trypsin; fixed modifications, none; variable modifications,
oxidation (M); mass values, monoisotopic; peptide mass tolerance,
±0.3 Da; fragment mass tolerance, ±0.2 Da; and max missed cleavage,
2. Positive identification was assigned with MASCOT scores above
the threshold level (P<0.05 was considered to indicate a
statistically significant difference) and with at least two
peptides (protein score >44).
Computational analysis
Homology searches were performed using the Basic
Local Alignment Search Tool (BLAST) at the National Center for
Biotechnology Information (http://blast.ncbi.nlm.nih.gov/Blast.cgi). Multiple
sequence alignments were performed using CLUSTALW ver. 2 (19) and refined manually for
representation. Sequence similarity (%) and identity (%) were
calculated using a global pairwise alignment tool (Needle program)
from EMBOSS (20). Theoretical
mass, theoretical isoelectric point, grand average of hydropathy
(GRAVY) and amino acid composition were calculated from the mature
protein sequence using the ProtParm program in the ExPASy proteomic
tools (http://web.expasy.org/protparam/). Secondary structure
was predicted with the Jpred program (http://www.compbio.dundee.ac.uk/www-jpred/).
Hydropathy plots and FoldIndex plots were created using the
ProtScale program (http://web.expasy.org/protscale/) with a window size
of 9, and FoldIndex (http://bioportal.weizmann.ac.il/fldbin/findex) with a
window size of 12, respectively. Subcellular localization of the
protein sequence was analyzed using WoLF PSORT (21). PYP sequence motifs were searched
using MotifScan (http://myhits.isb-sib.ch/cgi-bin/motif_scan).
Peptide synthesis
The N-terminal 20 residues of PYP1
(ALEGGKSSGGGEATRDPEPT), designated PYP1 (1–20), were synthesized by
Peptron (Daejeon, Korea). Purification of PYP1 (1–20) was performed
on a Shimadzu Prominence high-performance liquid chromatography
apparatus and controlled using the software package Class-VP, 6.14
using a C18 column (Shiseido Capcell Pak; Shiseido, Tokyo, Japan)
in 0.1% trifluoroacetic acid (TFA)/water and a gradient of 10–70%
acetonitrile in 0.1% TFA, with a flow rate of 1 ml/min and
ultraviolet detection at 220 nm. The molecular mass of PYP1 (1–20)
was confirmed to be 1,916 Da (matched with the sequence mass) by
mass analysis (HP 1100 Series LC/MSD; Agilent Technologies, Santa
Clara, CA, USA).
Cell culture
The Chang liver cell line (CCL-13; American Type
Culture Collection, Manassas, VA, USA) was maintained at 37°C in a
humidified atmosphere with 5% CO2. Cells were cultured
in minimum essential medium supplemented with 10% fetal bovine
serum (Hyclone, Logan, UT, USA), 100 U/ml penicillin and 100 mg/ml
streptomycin.
Cell proliferation and cytotoxicity
Cell proliferation was measured using a CellTiter
96® aqueous non-radioactive cell proliferation assay
(Promega, Madison, WI, USA), based on the cleavage of MTS into a
formazan product soluble in tissue culture medium. Cells were
seeded onto 96-well plates at 2×104 cells/well and
maintained for 24 h. Medium was replaced with serum-free medium
(SFM). After 24 h, SFM was replaced with PYP1 (1–20; 250 or 500
ng/ml) containing 15 mM acetaminophen (APAP) for 24 h.
Subsequently, cells were added to MTS/phenazine methosulfate
solution for 30 min at 37°C. Cell proliferation was measured at 490
nm absorbance using a microplate reader (Benchmark; Bio-Rad).
Statistical analysis
Significant differences in the effects of each
factor on cell viability were examined using an analysis of
variance (ANOVA), followed by the Duncan test. Differences with a
probability value (P)<0.05 were considered significant.
Results
Protein separation and identification of
the PYP fraction
In our previous study, SDS-PAGE of the PYP fraction
showed a single 14-kDa protein band on a 15% polyacrylamide gel
(7). Since N-terminal protein
sequencing analysis of the 14-kDa band yielded a composite and
unreadable sequence, the PYP fraction was further resolved using an
18% polyacrylamide gel and electroblotted onto a PVDF membrane;
protein bands were detected by Coomassie Brilliant Blue staining.
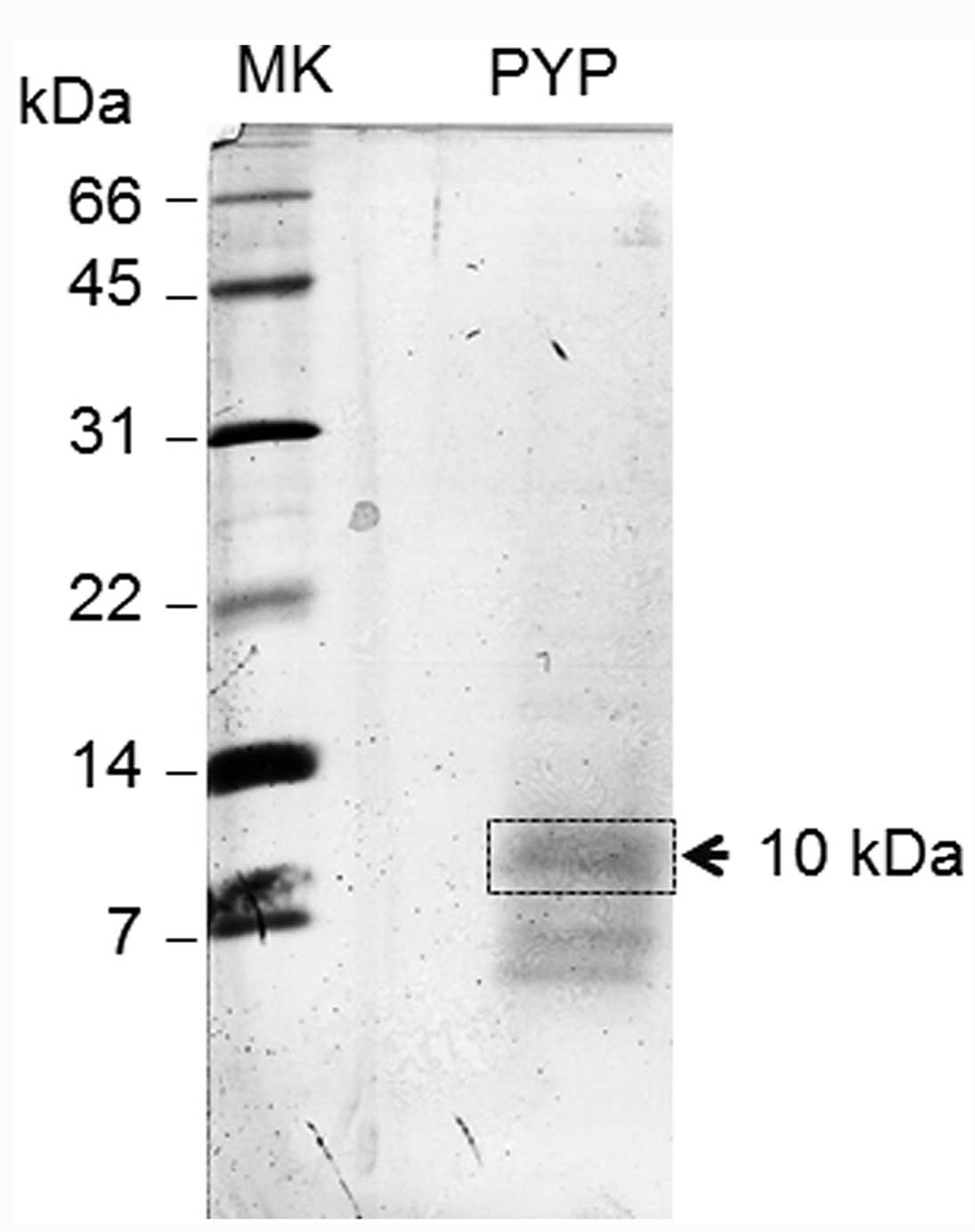
As shown in Fig. 1, the PYP
fraction showed a diffuse 10-kDa band and a ~7-kDa doublet band.
These proteins were easily lost from the gel during destaining when
the gel was stained with Coomassie Brilliant Blue R-250. The 10-kDa
band of the blot was excised as a section (Fig. 1) and sequenced with a gas phase
protein sequencer, yielding two unambiguous sequences:
Ala-Leu-Glu-Gly-Gly-Lys-Ser-Ser-Gly-Gly-Gly-Glu-Ala-Thr-Arg-Asp-Pro-Glu-Pro-Thr
(PTH-AA yield: 23.6 pmol/band) and
Glu-Thr-Xaa-Tyr-Ala-Asn-Val-Pro-Phe-Leu (PTH-AA yield: 7.6
pmol/band). Thus, these two proteins were tentatively designated
PYP1 (N-terminus: Ala-Leu-Glu-) and PYP2 (Glu-Thr-Xaa-),
respectively. The protein sequence data reported will appear in the
UniProt Knowledgebase (http://www.uniprot.org/uniprot) under the accession
numbers C0HJG2 (PYP1) and C0HJG3 (PYP2).
To identify the genes encoding PYP1 and PYP2, the
corresponding 10-kDa band was further analyzed by an MS/MS ions
search following in-gel tryptic digestion. As summarized in
Table I and shown in Fig. 2, two proteins encoded by EST
clones (the nuclear-encoded genes of P. yezoensis) were
identified. Availability of the gene sequences was evaluated with
Rhodophyta EST, Pyropia yezoensis CDS, SwissProt, and NCBInr
databases. Due to partial sequences of predicted gene models in
Pyropia yezoensis CDS annotation ver. 1 (10,327 gene models)
(14), the MASCOT protein score
(sum of the peptide score) was less than that of the Rhodophyta EST
(Table I). The cross-species
MS/MS ions search against the SwissProt and NCBInr databases failed
to identify genes for PYP1 and PYP2 (Table I). Therefore, precursor protein
sequences for PYP1 and PYP2 were deduced from the Rhodophyta EST,
and their homologs were searched from the NCBInr database to
predict the structure and function. As shown in Fig. 2A, PYP1 showed significant sequence
similarity with the hypothetical function-unknown proteins of
Rhodophyta (Porphyra purpurea and Chondrus crispus)
and a Haptophyceae (Emiliania huxleyi), and the N-terminal
domain of a putative fructose 1,6-bisphosphate aldolase class I
from E. huxleyi. Thus, PYP1 is a novel protein conserved in
Rhodophyta and Haptophyceae.
 | Figure 2Multiple sequence alignment of protein
sequences of (A) Pyropia yezoensis protein (PYP)1 and (B)
PYP2 with homologous sequences from Rhodophyta, Haptophyceae and
Stramenopiles. Gray letters indicate putative chloroplast targeting
signal sequences (transit peptides). Dashes denote blanks or gaps.
Underlined sequences are N-terminal protein sequences of mature
proteins determined experimentally. Sequences highlighted in gray
represent tryptic peptides assigned by tandem mass spectrometry
(MS/MS) ions searches. Asterisks, colons and dots represent
identical amino acids, conserved substitutions, and semiconserved
substitutions, respectively. Chain length of the mature protein
(that of the precursor in parenthesis), and percentage identity (I)
and similarity (S) are shown after the C-terminus. Abbreviations
and accession numbers: Ppu_hypo, hypothetical protein from
Porphyra purpurea (esContig7275); Ccr_hypo, unnamed protein
product from Chondrus crispus (CDF35169); Hhu_hypo,
hypothetical protein from Emiliania huxleyi (AFE02913);
Hhu_FBPA1N, N-terminal domain of putative fructose 1,6-bisphosphate
aldolase class I from Emiliania huxleyi (AFE02906);
Gja_PsbU, Photosystem II 12-kDa extrinsic protein (PsbU) from
Griffithsia japonica (AAP80721); Ccr_PsbU, PsbU from C.
crispus (CDF33931); Cgr_PsbU, PsbU from Chaetoceros
gracilis (BAG85212). Precursor protein sequences of PYP1 and
PYP2 were deduced from expressed sequence tags (ESTs) (AV429800 and
DR907360, respectively). Due to the lack of an N-terminal transit
peptide-coding region in the PYP2 EST clone (DR907360), a putative
corresponding sequence (small letters) is supplemented by another
EST clone (AV438439). ‘>>’ indicates that the N-terminal
domain homologous to PYP1 is followed by a putative fructose
1,6-bisphosphate aldolase class I. |
 | Table ITandem mass spectrometry (MS/MS) ion
search of in-gel tryptic digest from the 10-kDa gel section. |
Table I
Tandem mass spectrometry (MS/MS) ion
search of in-gel tryptic digest from the 10-kDa gel section.
| | | | | | Accession no. |
|---|
| | | | | |
|
|---|
| Protein ID | m/z | Deltaa, Da | Missed cleav.b | Scorec | Sequenced | Rhod_ ESTe | Pye_ CDSf | Swiss Prot | NCBInr |
|---|
| PYP1 | 1687.96 | 0.08 | 0 | 40 |
QQAIHVAPSFADYLK | AV429545 | 20922_g5127 | NAg | NAg |
| 2197.09 | 0.09 | 1 | 20 |
SSGGGEATRDPEPT
AVDPNDPK | AV429545 | NAg | NAg | NAg |
| PYP2 | 1382.86 | 0.03 | 0 | 29 | IPGLTDLQIATLK | DR907360 | 19303_g4763 | NAg | NAg |
| 1589.05 | 0.20 | 0 | 36 | LVALTPTPEYELDK | DR907360 | 19303_g4763 | NAg | NAg |
| 2820.43 | 0.04 | 0 | 49 | YPGMYPTVAGLIAT
NGPFETVSDLYK | DR907360 | 19303_g4763 | NAg | NAg |
Cell proliferation and cytotoxicity
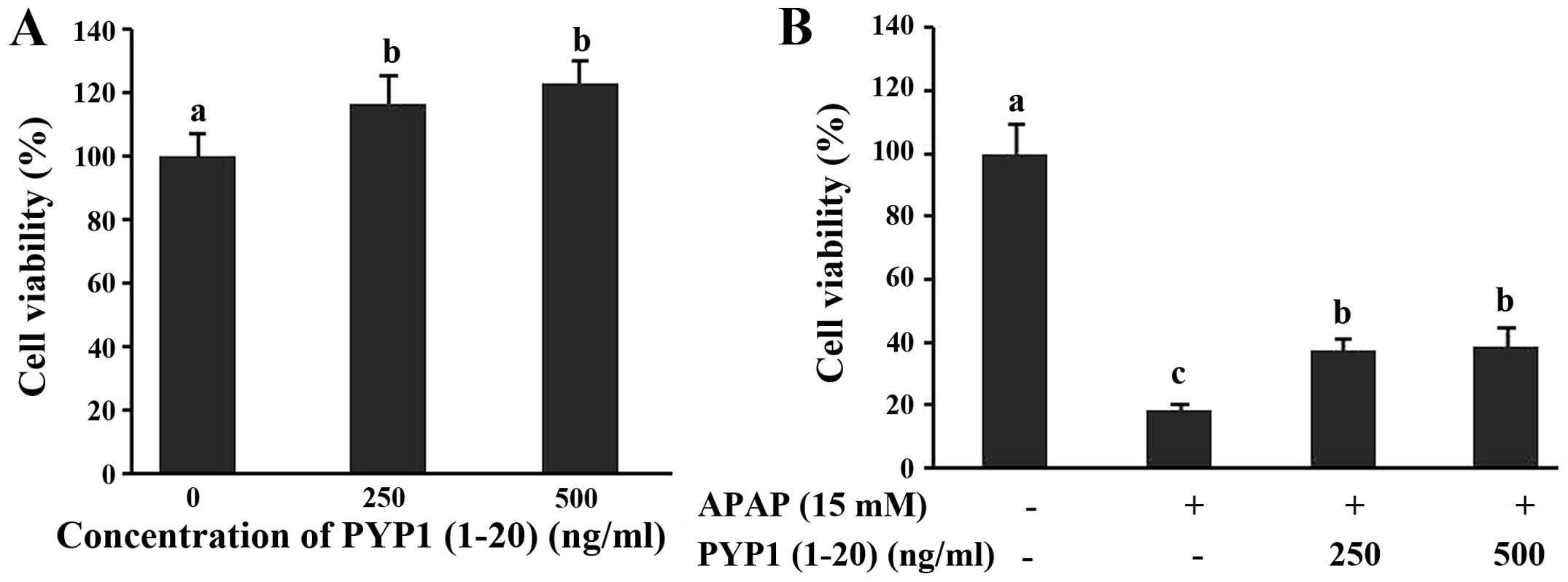
PYP1 (1–20), the synthetic 20-residue peptide of the
N-terminus of PYP1, was used to investigate the effect of Chang
liver cell proliferation, as determined by the CellTiter 96®
aqueous non-radioactive cell proliferation assay. Cells were
treated with various concentrations (250 or 500 ng/ml) of PYP1
(1–20) for 24 h (Fig. 3A). PYP1
(1–20) did not induce cytotoxicity or proliferation in Chang liver
cells. Subsequently, Chang liver cells were investigated for the
protective effect of PYP1 (1–20) against hepatotoxicity following
treatment with 15 mM APAP. As a result, cell viability decreased
80% when treated with 15 mM APAP. However, cell viability recovered
~38% when treated with PYP1 (1–20;
Fig. 3B). Therefore, PYP1 (1–20)
has a protective effect on hepatotoxicity caused by APAP in Chang
liver cells.
Discussion
As the PYP1 precursor has a putative
chloroplast-targeting signal, PYP1 is most likely a
chloroplast-localized protein. Although PYP1 was identified from
the 10-kDa band following SDS-PAGE, the sequence mass of mature
PYP1 was calculated as 5,763.3 Da. This observation indicates that
PYP1 forms an SDS-resistant dimer, as reported in certain other
proteins (22,23). As reported previously, PYP
proteins are heat-soluble (7).
The GRAVY of PYP1 is −1.086, and the amino acid composition is a
preponderance of Ala (16%), Gly (13%), Lys (14%), Glx (13%), Asx
(11%), Thr/Ser (9%) and Pro (9%), and lacks Trp and Cys. The
hydrophilic N-terminal 33 residues and C-terminal 12 residues of
PYP1 were predicted to be intrinsically disordered regions and the
short internal hydrophobic region to be structured, respectively.
Additionally, the physicochemical characteristic of PYP1 is similar
to that of late embryogenesis abundant (LEA) proteins, which
protect protein denaturation from desiccation, freezing, heat, salt
and osmotic stress (24). A
LEA-like protein from Arabidopsis thaliana also plays a role
in the protection against oxidative stress (25). LEA proteins are heat stable,
intrinsically disordered proteins, and have high hydrophilicity
with GRAVY (often, <−1.0), a preponderance of Ala, Gly, Glu,
Lys/Arg and Ser/Thr, and a lack or low proportion of Cys and Trp.
Although structurally known motifs of the LEA family were not
identifiable from the PYP1 sequence, PYP1 may be a novel LEA-like
protein. PYP2 showed significant sequence similarity with PsbU
(Fig. 2B). PsbU, an extrinsic
protein localized in the luminal side of photosystem II, has been
identified in the majority of cyanobacteria and red algae, but not
in green algae and higher plants (26). Notably, PsbU contributes to the
thermal stability of photosystem II (27) and enhances its structural
stability, protecting it from reactive oxygen species (ROS), which
are produced as an inevitable by-product of photosynthesis
(28,29); therefore, PYP2 (i.e., P.
yezoensis PsbU) could also have such functions. Since
APAP-induced cell injury is caused by oxidative stress through ROS
production (30), PYP1 (putative
novel LEA-like) and PYP2 (PsbU) may also contribute to the
chemoprotective activity though an uncharacterized protection
mechanism from ROS attack.
For the first time, Hwang et al (7) reported that PYP demonstrated
chemoprotective effects against APAP-induced liver injury. Also,
antioxidant and anti-inflammatory effects of a glycoprotein from
P. yezoensis were demonstrated in
lipopolysaccharide-stimulated RAW 264.7 mouse macrophages (9). Despite various bioactive substances
of P. yezoensis, no information was available regarding a
specific peptide with bioactivity in PYP. In the present study,
PYP1 (1–20), the synthetic 20-residue peptide of the N-terminus of
PYP1, was used to investigate the effect of Chang liver cell
proliferation. As a result, PYP1 (1–20) was shown to not induce
cytotoxicity, as well as having the effect of the proliferation in
Chang liver cells. These data indicate that PYP1 is a novel
LEA-like protein with chemoprotective activity at the N-terminal
portion. Taking into account the chemoprotective activity of PYP
in vivo and in vitro (7), PYP1 may have an effect on protection
against liver cell injury.
Acknowledgements
The authors thank Ms. Natsumi Kawano and Ms. Yuri
Shima for their technical assistance. The present study was
supported by the Basic Science Research Program through the
National Research Foundation of Korea (NRF), funded by the Ministry
of Education (no. 2012R1A6A1028677), and in part by a Grant-in-Aid
for Scientific Research (no. 24580303) to K.Y. from the Ministry of
Education, Culture, Sports, Science and Technology, Japan.
References
|
1
|
Velasquez-Orta SB, Curtis TP and Logan BE:
Energy from algae using microbial fuel cells. Biotechnol Bioeng.
103:1068–1076. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Choi YH, Kim KW, Han HS, Nam TJ and Lee
BJ: Dietary Hizikia fusiformis glycoprotein-induced IGF-I and
IGFBP-3 associated to somatic growth, polyunsaturated fatty acid
metabolism, and immunity in juvenile olive flounder Paralichthys
olivaceus. Com Biochem Phys A. 167:1–6. 2014. View Article : Google Scholar
|
|
3
|
Hirayasu H, Yoshikawa Y, Tsuzuki S and
Fushiki T: Sulfated polysaccharides derived from dietary seaweeds
increase the esterase activity of a lymphocyte tryptase, granzyme
A. J Nutr Sci Vitaminol. 51:475–477. 2005. View Article : Google Scholar
|
|
4
|
Maeda H, Hosokawa M, Sashima T, Funayama K
and Miyashita K: Fucoxanthin from edible seaweed, Undaria
pinnatifida, shows antiobesity effect through UCP1 expression in
white adipose tissues. Biochem Biophys Res Commun. 332:392–397.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yuan YV and Walsh NA: Antioxidant and
antiproliferative activities of extracts from a variety of edible
seaweeds. Food Chem Toxicol. 44:1144–1150. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hwang HJ, Kim IH and Nam TJ: Effect of a
glycoprotein from Hizikia fusiformis on acetaminophen-induced liver
injury. Food Chem Toxicol. 46:3475–3481. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hwang HJ, Kwon MJ, Kim IH and Nam TJ:
Chemoprotective effects of a protein from the red algae Porphyra
yezoensis on acetaminophen-induced liver injury in rats. Phytother
Res. 22:1149–1153. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Go H, Hwang HJ and Nam TJ: A glycoprotein
from Laminaria japonica induces apoptosis in HT-29 colon cancer
cells. Toxicol In Vitro. 24:1546–1553. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Shin ES, Hwang HJ, Kim IH and Nam TJ: A
glycoprotein from Porphyra yezoensis produces anti-inflammatory
effects in liposaccharide-stimulated macrophages via the TLR4
signaling pathway. Int J Mol Med. 28:809–815. 2011.PubMed/NCBI
|
|
10
|
Kim YM, Kim IH and Nam TJ: Induction of
apoptosis signaling by glycoprotein of Capsosiphon fulvescens in
human gastric cancer (AGS) cells. Nutr Cancer. 64:761–769. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Saga N and Kitade Y: Porphyra: a model
plant in marine sciences. Fish Sci. 68(Suppl): 1075–1078. 2002.
|
|
12
|
Lee EK, Seo SB, Kim TH, et al: Analysis of
expressed sequence tags of Porphyra yezoensis. Mol Cells.
10:338–342. 2000.PubMed/NCBI
|
|
13
|
Nikaido I, Asamizu E, Nakajima M, Nakamura
Y, Saga N and Tabata S: Generation of 10,154 expressed sequence
tags from a leafy gametophyte of a marine red alga, Porphyra
yezoensis. DNA Res. 7:223–227. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Nakamura Y, Sasaki N, Kobayashi M, et al:
The first symbiont-free genome sequence of marine red alga,
Susabi-nori (Pyropia yezoensis). PLoS One. 8:e571222013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang L, Mao Y, Kong F, et al: Complete
sequence and analysis of plastid genomes of two economically
important red algae: Pyropia haitanensis and Pyropia yezoensis.
PLoS One. 8:e659022013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Laemmli UK: Cleavage of structural
proteins during the assembly of the head of bacteriophage T4.
Nature. 227:680–685. 1970. View
Article : Google Scholar : PubMed/NCBI
|
|
17
|
Weber K and Osborn M: The reliability of
molecular weight determinations by dodecyl sulfate-polyacrylamide
gel electrophoresis. J Biol Chem. 244:4406–4412. 1969.PubMed/NCBI
|
|
18
|
Yamaguchi K: Preparation and proteomic
analysis of chloroplast ribosomes. Chloroplast Research
Arabidopsis. Javis RP: 775. Humana Press; New York, NY: pp.
241–264. 2011, View Article : Google Scholar
|
|
19
|
Larkin MA, Blackshields G, Brown NP, et
al: Clustal W and Clustal X version 2.0. Bioinformatics.
23:2947–2948. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Rice P, Longden I and Bleasby A: EMBOSS:
the European Molecular Biology Open Software Suite. Trends Genet.
16:276–277. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Horton P, Park KJ, Obayashi T, et al: WoLF
PSORT: protein localization predictor. Nucleic Acids Res. 35(Web
Server issue): W585–W587. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Cabra V, Arreguin R, Vazquez-Duhalt R and
Farres A: Effect of temperature and pH on the secondary structure
and processes of oligomerization of 19 kDa alpha-zein. Biochim
Biophys Acta. 1764:110–1118. 2006.
|
|
23
|
Tatsuno R, Yamaguchi K, Takatani T and
Arakawa O: RT-PCR- and MALDI-TOF mass spectrometry-based
identification and discrimination of isoforms homologous to
pufferfish saxitoxin- and tetrodotoxin-binding protein in the
plasma of non-toxic cultured pufferfish (Takifugu rubripes). Biosci
Biotechnol Biochem. 77:208–212. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Battaglia M, Olvera-Carrillo Y,
Garciarrubio A, Campos F and Covarrubias AA: The enigmatic LEA
proteins and other hydrophilins. Plant Physiol. 148:6–24. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Mowla SB, Cuypers A, Driscoll SP, et al:
Yeast complementation reveals a role for an Arabidopsis thaliana
late embryogenesis abundant (LEA)-like protein in oxidative stress
tolerance. Plant J. 48:743–756. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Roose JL, Wegener KM and Pakrasi HB: The
extrinsic proteins of Photosystem II. Photosynth Res. 92:369–387.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Nishiyama Y, Los DA and Murata N: PsbU, a
protein associated with photosystem II, is required for the
acquisition of cellular thermotolerance in Synechococcus species
PCC 7002. Plant Physiol. 120:301–308. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Balint I, Bhattacharya J, Perelman A, et
al: Inactivation of the extrinsic subunit of photosystem II, PsbU,
in Synechococcus PCC 7942 results in elevated resistance to
oxidative stress. FEBS Lett. 580:2117–2122. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Abasova L, Deák Z, Schwarz R and Vass I:
The role of the PsbU subunit in the light sensitivity of PSII in
the cyanobacterium Synechococcus 7942. J Photochem Photobiol B.
105:149–156. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Adamson GM and Harman AW: Oxidative stress
in cultured hepatocytes exposed to acetaminophen. Biochem
Pharmacol. 45:2289–2294. 1993. View Article : Google Scholar : PubMed/NCBI
|