Introduction
Traumatic optic nerve injury is one of the most
common neuropathies, affecting an increasing number of individuals
worldwide and leading to the loss of neural cells in the eyes
(1). The 'optic nerve,' the
bundle of retinal ganglion cell (RGC) axons, is part of the central
nervous system (CNS). RGCs are the output neurons that relay visual
signals from the eye, extending along axons via the optic nerve to
the brain. Traumatic optic neuropathies often lead to the
retrograde degeneration and apoptosis of RGCs, potentially
resulting in visual impairment and neurological dysfunction. For
the treatment of traumatic optic neuropathy, it is necessary to
promote axonal regeneration or prevent the death of injured neurons
(2).
Establishing an effective animal model of optic
nerve injury is important in order to better understand the
mechanisms responsible for RGC degeneration and neuroprotection,
and to further explore novel drug targets and treatment options for
optic nerve functional recovery and regeneration. 'Optic nerve
crush' (ONC) injury is a popular experimental model of traumatic
optic nerve injury, typically characterized by sudden visual loss
attributable to irreversible disturbances (1).
Regenerative medicine using human stem cells is a
new and promising field which has the potential to for use in the
treatment of various intractable diseases and damaged organs,
including difficult-to-treat optic nerve diseases. Many scientists
have demonstrated the abilities of various stem cells to ameliorate
optic nerve damage (1–7) in animal models of optic nerve
compression. Mesenchymal stem cells (MSCs) have great potential for
use as cell therapy in regenerative medicine, including the
treatment of nerve injury. Umbilical cord blood (UCB) mononuclear
cells (CB-MNCs), which reportedly contain stem cells (8–12),
have been widely used as a hematopoietic source, and have been
shown to possess therapeutic potential for repairing neurological
impairment (13–19). A previous study using an
experimental animal model of ONC injury demonstrated the
therapeutic effects of intravitreally administering CB-MNCs, with
the incorporation of these cells into the neurological lesion
(1). UCB cells have been banked
worldwide, and no harmful effects have been reported with their use
(10,11). In terms of the therapeutic
management of optic nerve injury, UCB has potential based on its
known neuroprotective properties due to its anti-inflammatory and
anti-apoptotic activities (10,11,13,18).
In contrast to other MSCs, including bone
marrow-derived MSCs (BM-MSCs) and adipose tissue-derived MSCs
(AD-MSCs), placenta-derived mesenchymal stem cells (PD-MSCs) are
obtainable in massive numbers and have potent immunological
properties (20,21). Human PD-MSCs were classified over
the past decade and have become the focus of attention in stem cell
research (22) due to the
following reasons: they were the first of the adult stem cells to
appear, they have great potential for proliferation,
differentiation and self-renewal, they are readily available, and
they are easily procured without any invasive procedures (10,23). Several types of PD-MSCs, such as
amnion-derived MSCs (AMSCs), chorionic plate-derived MSCs
(CP-MSCs), Wharton's jelly-derived MSCs (WJ-MSCs) and chorionic
villi-derived MSCs (CV-MSCs), have been described and may exert
therapeutic effects through anti-fibrotic mechanisms (20). It has been confirmed that the
cells derived from the chorioamniotic membrane of the full term
placenta are MSCs and these cells have been characterized as
CP-MSCs and have been shown to exert several therapeutic effects
(10,20,21,23).
Although research dedicated to the management of
traumatic optic nerve injury using various measures is ongoing,
novel therapeutic strategies based on the complex underlying major
mechanism(s) responsible for this type of injury, such as
inflammation and/or ischemia, are required.
Thus, in the present study, we established an animal
model of traumatic optic neuropathy (namely ONC injury) using
Sprague-Dawley (SD) rats and examined the variations in axon
counts. Using this rat model, we aimed to examine the efficacy of
treatment using UCB (namely CB-MNCs), compared to treatment using
CP-MSCs.
Materials and methods
Animals
All experimental procedures conformed to the
Association for Research in Vision and Ophthalmology (ARVO)
Statement regarding the Use of Animals in Ophthalmic and Vision
Research. Animal protocols were approved by the Institutional
Animal Care and Use Committee of CHA Bundang Medical Center
(Seongnam-si, Korea).
All animals were housed in a standard animal
facility with food and water provided ad libitum at a
constant temperature of 21°C. Six-week-old male SD rats (Orient Bio
Inc., Seongnam-si, Korea) were maintained in an air-conditioned
animal house under specific pathogen-free conditions. The animals
were maintained for at least 1 week in this environment prior to
being subjected to the surgical procedures, which were performed on
one eye of each rat, while the contralateral eye served as the
sham-operated control. Topical ophthalmic ointment (Ciproxacin,
Tarivid®; Santen Pharmaceutical, Co, Tokyo, Japan) was
applied immediately after the surgical procedures.
Establishment of a rat model of ONC
injury
Ninety male SD rats (Orient Bio Inc.), weighing
200–250 g, were anesthetized by an intraperitoneal injection of
Zoletil (1:1 tiletamine:zolazepam; 30 mg/kg) and Rompun (xylazine
10 mg/kg). Following the topical application of 0.5% proparacaine
hydrochloride, a lateral canthotomy and conjunctival incision were
made, and the tissues surrounding the optic nerve were dissected
such that the optic nerve was exposed without damaging the adjacent
blood supply. Extra-fine self-closing forceps were used to crush
the optic nerve 2 mm behind the globe for 5 sec. The canthal
incision was then sutured. To examine the temporal response
following ONC injury and to establish the model of ONC injury, 15
rats were administered a lethal dose of anesthetic at 1, 2 or 4
weeks (n=5 rats for each time point).
Assessment of axonal density and axon
survival ratio
After 1, 2 and 4 weeks, the eyeballs were enucleated
to assess the axonal density following an intraperitoneal injection
of Zoletil (1:1 tiletamine:zolazepam; 30 mg/kg) and Rompun
(xylazine, 10 mg/kg). The excised axons and the eyeballs were fixed
in Karnovsky's solution, processed with 1% osmium tetroxide and
embedded in Epon-Araldite resin (Ladd Research Industries,
Burlington, VT, USA). Semi-thin (<1.0 µm) cross-sections
of the optic nerve (obtained from the midpoint of the sample,
approximately 1 mm from the stump) were stained with 1% toluidine
blue in 1% sodium borate. Digital images were obtained at x40
magnification using a light microscope (Olympus BX40; Olympus
Optical Co. Ltd., Tokyo, Japan) for the histological examination of
axonal density using ImageJ software (NIH, http://rsb.info.nih.gov/ij/index.html).
We regarded the following axons as non-viable: i)
visible swollen axons; ii) shrunken axons or axons with
layer-splitting of myelin sheaths; and iii) extensive fibrotic
axons. The number of the viable axons was counted and axon survival
rates were defined as the axon density ratio between the normal
side and the ONC injured side in each SD rat at the time of
evaluation.
Preparation of CB-MNCs
Human heterogeneic UCB units were selected from the
affiliated UCB bank, CHA Medical Center Cord Blood Bank
(Seongnam-si, Korea). The out-of-specification UCB units consisted
of >3×107/kg total nucleated cells, which were washed
to eliminate dimethyl sulfoxide, and the CB-MNCs were counted.
Prior to the administration of UCB (CB-MNCs), human leukocyte
antigen (HLA) typing of the CB-MNCs, vs. the CP-MSCs, was
performed; specifically, HLA-A -B, -C (#555552, BD Biosciences, San
Diego, CA, USA), HLA-DR (#555811, BD Biosciences) and HLA-G
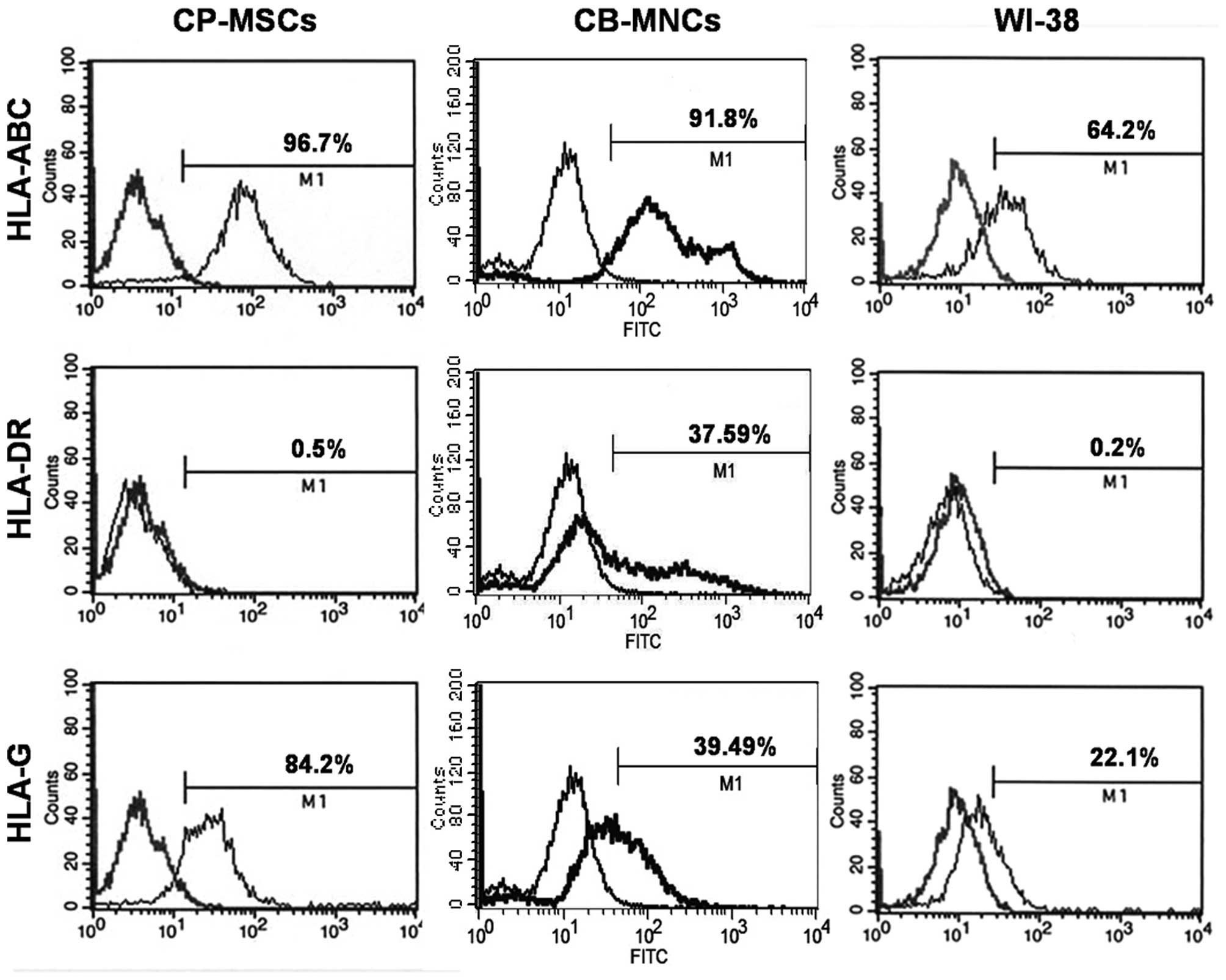
(#ab7904, Abcam, Cambridge, MA, USA) typing (Fig. 1).
Cultivation of CP-MSCs
Sample collection and use for research purposes were
approved by the Institutional Review Board of CHA General Hospital,
Seoul, Korea. All participants provided written informed consent
prior to sample collection. The placentas were collected from
female subjects who were free of medical, obstetric and surgical
complications and who delivered at term (>37 gestational weeks).
The CP-MSCs were harvested as previously described (22). Briefly, the CP-MSCs were collected
from the inner side of the chorioamniotic membrane of the placenta
and treated with 0.5% collagenase IV (C7661; Sigma-Aldrich, St.
Louis, MO, USA) and cultured in MEM-α modification (#32561;
Gibco-BRL, NY, USA) supplemented with 10% fetal bovine serum (FBS,
#16000-044), 1% penicillin/streptomycin (#15140-122) (all from
Invitrogen, Carlsbad, CA, USA), 25 ng/ml human fibroblast growth
factor-4 (hFGF-4; #AF-100-31; Peprotech, Inc., Rocky Hill, NJ, USA)
and 1 µg/ml heparin (H3149; Sigma-Aldrich).
In addition, the normal fibroblast cell line, WI-38,
was purchased from ATCC (#CCL-75, ATCC; Manassas, VA, USA). The
cells were cultured in α-MEM containing 10% FBS, 1%
penicillin/streptomycin (P/S; 100 mg/ml, Gibco-BRL), and 2 mM
L-glutamine (Gibco-BRL).
FACS analysis
To analyze the expression of HLA surface markers,
the CB-MNCs and CP-MSCs (106 cells) were collected and
washed with PBS containing 2% FBS. They were then incubated with
isotype control immunoglobulin G (IgG) or antigen-specific
antibodies [HLA-ABC (BD Biosciences), HLA-DR (BD Biosciences) and
HLA-G (Abcam)] as well as fluorescence-conjugated anti-human IgG
antibodies (1:200;, #550931; BD Biosciences) for 30 min at room
temperature. They were then stained with 5 ng/ml propidium iodide
(#4170; Sigma-Aldrich) to identify non-viable cells. FACS analysis
was performed using a FACSCalibur Flow Cytometer (#342975; BD
Biosciences). The characterization of CP-MSCs (passages 8–10) is
standard in our laboratory and has been proven desirable (24).
Stem cell injection
To analyze the engraftment of the cells, the CB-MNCs
and the CP-MSCs were labeled using a PKH26 Red Fluorescent Cell
Linker kit (MINI-26; Sigma-Aldrich) according to the manufacturer's
instructions. The procedures are standard in our laboratory and
have been proven efficient (24).
The PKH26-labeled CP-MSCs (106 cells, passage 8–10, 0.5
ml) and PKH26-labeled CB-MNCs (106 cells, 0.5 ml) were
each injected into the left internal carotid artery of each animal
(n=30). Non-transplanted (NTP) rats (n=15) were injected with an
equal volume of normal saline alone as the sham-operated
controls.
To determine the stem cell response, the animals
were euthanized at 1, 2 or 4 weeks after the cell injection. The
eyeballs were enucleated to assess the axon survival ratio and the
immunohistochemical evaluation of the normal right side and the ONC
injured left side in each rat. The optic nerves were collected at
1, 2 or 4 weeks post-injection from the animals in each of the 3
groups (NTP group, CB-MNC-transplanted group and
CP-MSC-transplanted group). In addition, liver, lung and kidney
specimens were obtained from 10 rats from the groups injected with
the stem cells groups following enucleation at 1 week
post-implantation. Liver, lung and kidney samples were embedded in
Tissue-Tek OCT compound (Sakura Finetechnical Co., Ltd., Tokyo,
Japan) to observe the infiltration of the injected stem cells
following the transplantation of the CP-MSCs or CB-MNCs. Cryostat
sections (10 µm-thick) were assessed for PKH26-labeled cells
using a fluorescence microscope.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
The growth-associated protein-43 (GAP-43), and
hypoxia-inducible factor-1α (HIF-1α) mRNA levels were measured by
RT-qPCR. Equal amounts of protein from individual animals were
pooled from the rats in the control (NTP) group (n=15), the rats in
the CP-MSC-transplanted group at 1 week (n=10), the
CP-MSC-transplanted group at 2 weeks (n=10), the
CP-MSC-transplanted group at 4 weeks (n=10), as well as the rats in
the CB-MNC-transplanted group at 1 week (n=10), the
CB-MNC-transplanted group at 2 weeks (n=10) and the rats in the
CB-MNC-transplanted group at 4 weeks (n=10). Total RNA was isolated
using TRIzol reagent. The concentration and quality of the RNA were
determined using an ND-1000 spectrophotometer (NanoDrop, Waltham,
MA, USA). Reverse-transcription to cDNA was conducted using the
iScript cDNA synthesis kit (Bio-Rad Laboratories, Hercules, CA,
USA) according to the manufacturer's instructions. The cDNA used
for qPCR was normalized to β-actin. qPCR was performed using
IQ SYBR-Green supermix (Bio-Rad Laboratories). Gene expression was
quantified using the ΔCT method. All reactions were performed in
triplicate. Data are expressed as the means ± SD of triplicate
experiments.
Histological examination
Enucleation was performed to allow the pathological
examination of the optic nerve. Tissue slices (5-µm-thick)
were cut and fixed in 4% paraformaldehyde. The tissue slices were
then embedded in paraffin. The sections were dipped into gradient
ethanol, stained with hematoxylin for 10 min, differentiated in 1%
(v/v) hydrochloric acid alcohol for 5–10 sec, incubated in
insaturated lithium carbonate solution for 5–10 sec, and stained
with eosin for 10 min. The sections were rinsed with distilled
water for each interval step. The sections were then dehydrated
with gradient ethanol, cleared with xylene, mounted with neutral
gum, dried and observed for the general morphology and composition
of the optic nerve using light microscopy. Immunohistochemical
staining was performed on the 5-µm-thick sections, cut from
formalin-fixed, paraffin wax-embedded tissues, using the anti-human
nuclei and chromosomes, histone H1 protein antibody (Cat. no.
MAB1276, mouse monoclonal, clone 1415-1, 1:200 dilution; Millipore,
Billerica, MA, USA). The visualization system used was the
BenchMark XT with heat-induced epitope retrieval (CC1 solution) and
the ultraView Universal DAB Detection kit (all from Ventana,
Tucson, AZ, USA).
RNA assay
The isolated optic nerves, including the crush
lesion were homogenized using TRIzol reagent (Invitrogen, Carlsbad,
CA, USA) for RNA extraction. To synthesize the cDNA, 3 µg
total RNA from each sample was reverse transcribed using
SuperScript III reverse transcriptase (Invitrogen). RT-PCR was
performed using the Solg 2X Taq PCR Pre-Mix (Solgent, Daejeon,
Korea). The PCR conditions were as follows: initial melting (95°C,
2 min), 40 cycles of amplification (95°C for 20 sec, 45–55°C for 40
sec, and 72°C for 1 min) and a final extension (72°C for 5 min).
β-actin was amplified for normalization using the following
PCR conditions: initial melting (95°C for 2 min), 40 cycles of
amplification (95°C for 20 sec, 50°C for 40 sec, and 72°C for 1
min) and a final extension (72°C for 5 min). The ERM-like protein
(ERMN), lecithin-retinol acyltransferase (LRAT) and
SLIT-ROBO Rho GTPase activating protein 2 (SRGAP2) primer
sets used were as follows: ERMN FP, 5′-AAA ACC CAG AAG AGA
AAC-3′ and RP, 5′-ATC ATT ATT GCA ATT ATC-3′; LRAT FP,
5′-AGG CGA CGT GCT GGA GGT-3′ and RP, 5′-AGT GCT CGC AGT TGT
TCC-3′; SRGAP2 FP, 5′-GAA TTT TTC CTG CAA CTG GA-3′ and RP,
5′-AAT GAG CAT ATT TGG CTT TG-3′.
Western blot analysis
The optic nerve tissues were lysed in PRO-PREP
solution (Intron, Seongnam-si, Korea). The protein lysates were
loaded onto 10% sodium dodecyl sulfate-polyacrylamide gels, and the
separated proteins were transferred onto polyvinylidene difluoride
(PVDF) membranes, blocked, and then incubated with antibodies to
SRGAP2 and ERMN (both from Abcam, Cambridge, UK), neurofilament
(NF; Cell Signaling Technology, Danvers, MA, USA) and oncomodulin
(OCM; Santa Cruz Biotechnology, Santa Cruz, CA, USA) overnight at
4°C at dilutions of 1:200 or 1:1,000. The membranes were washed and
then incubated with anti-rabbit or anti-goat IgG secondary antibody
(Santa Cruz Biotechnology) for 3 h at room temperature using an
orbital shaker. After washing, the bands were detected using an
enhanced chemiluminescence reagent (Bio-Rad Laboratories).
Statistical analysis
In the present study, statistical analyses were
performed using SPSS software (version 19.0; IBM, Chicago, IL, USA)
and Prism 5.0 (GraphPad Software, San Diego, CA, USA). Depending on
the normality of the data, they are presented as the means ± SD.
Differences among the groups were analyzed using one-way ANOVA,
followed by a Student's t-test. A p-value <0.05 was considered
to indicate a statistically significant difference. ANOVA or the
Kruskal-Wallis and Mann-Whitney U tests were used to compare
outcomes between independent groups, and a paired t-test was used
to analyze intragroup differences.
Results
FACS analysis
Our results revealed differences in the expression
levels of the HLA surface markers between the CP-MSCs and CB-MNCs.
The CP-MSCs were negative for hematopoietic markers, such as
HLA-DR, and positive for non-hematopoietic markers, such as
HLA-ABC, similar to the WI-38 cells. Furthermore, the surface
expression of HLA-G was detected in the CP-MSCs. However, the
CB-MNCs were positive for hematopoietic markers, such as HLA-DR.
The characterization of CP-MSCs (passages 8–10) is standard in our
laboratory and has been proven desirable (Fig. 1).
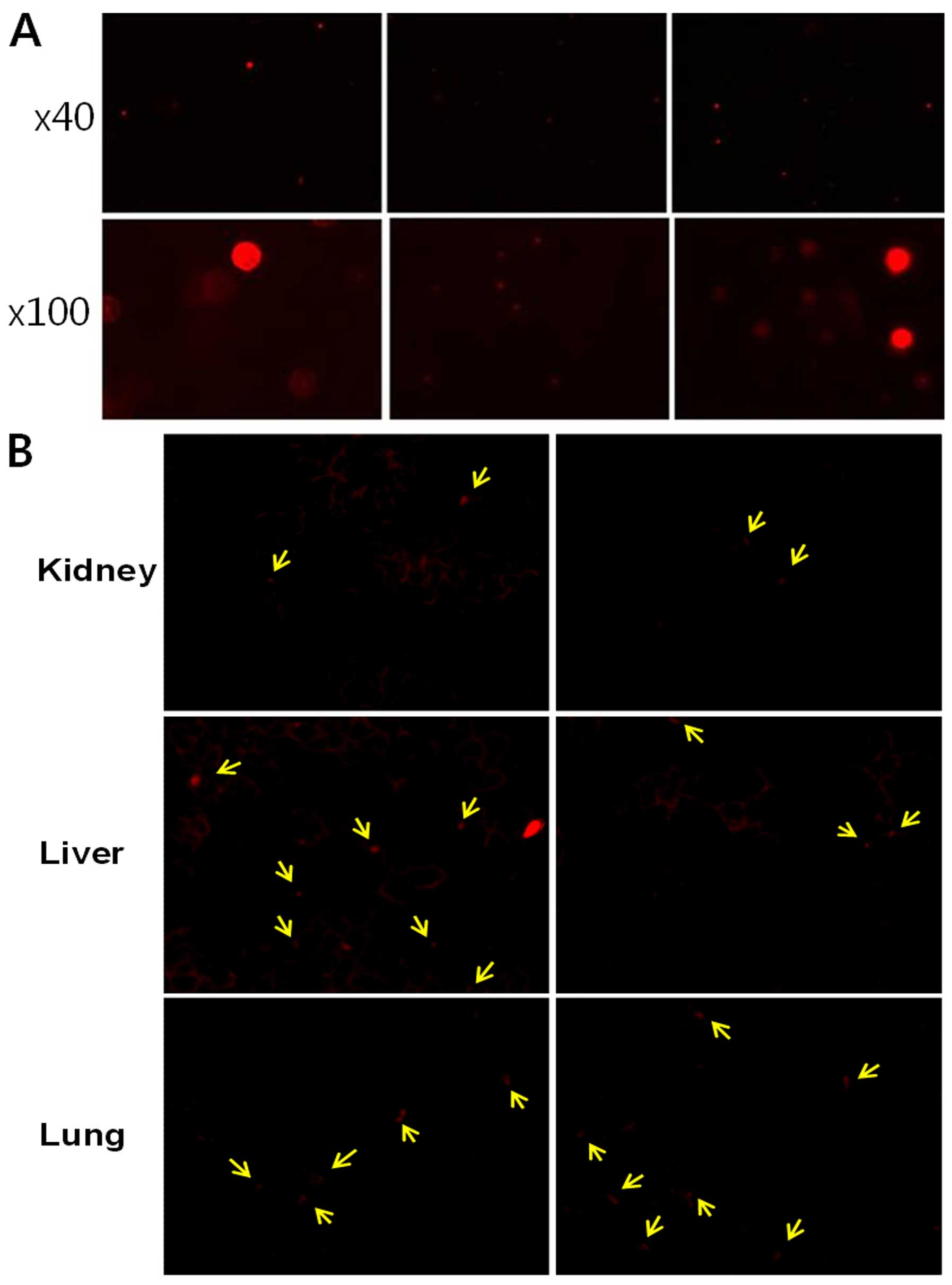
Engraftment of CB-MNCs and CP-MSCs
To confirm the infiltration of the injected CP-MSCs
or CB-MNCs, liver, lung and kidney samples were examined.
PKH26-labeled red fluorescent cells were observed in the liver,
lungs and kidneys at 1 week post-transplantation (Fig. 2).
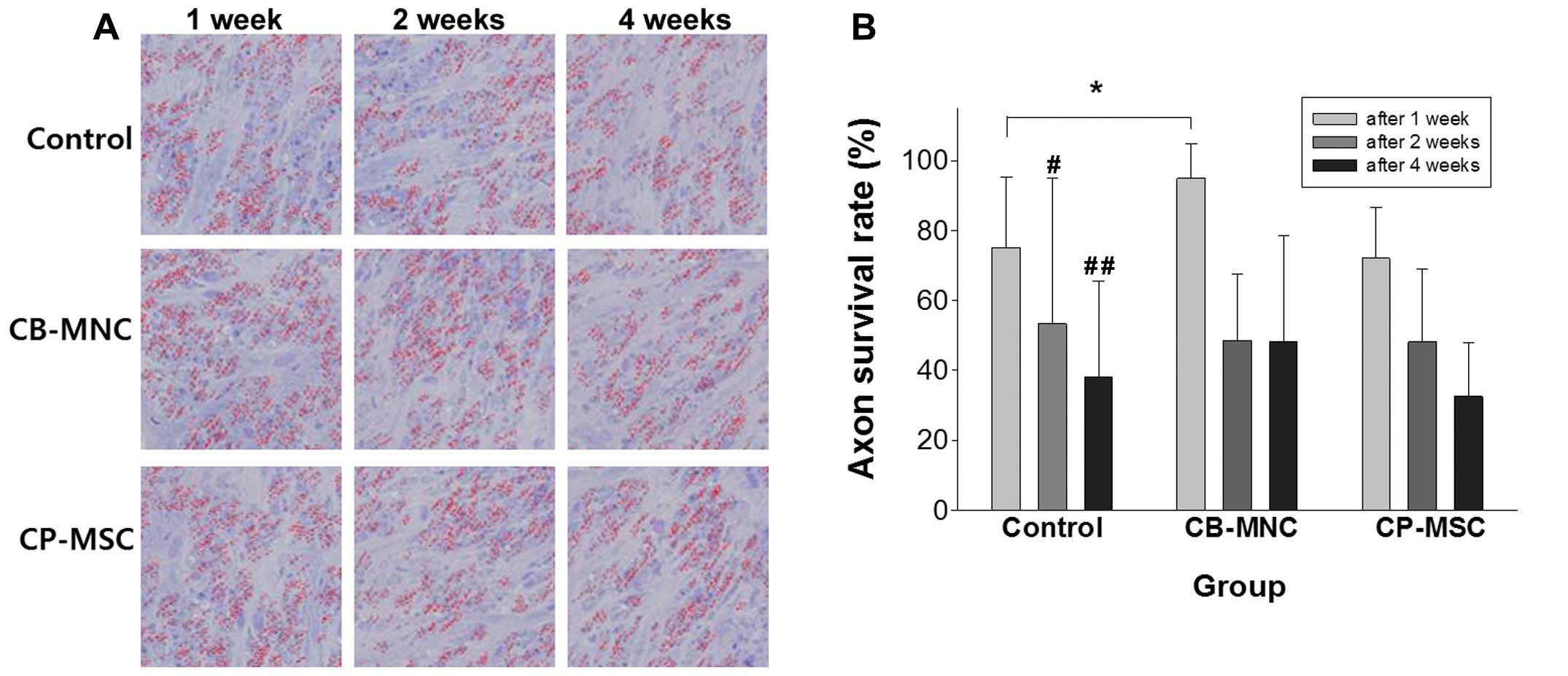
Axon survival ratio in the rat model of
ONC injury
The axon survival rates were defined as the axon
density ratio between the normal side and the ONC injured side in
each rat at the time of sacrifice. Compared with the normal side,
the axon density decreased significantly on the ONC injured side
(p<0.05). In the rats with ONC injury, the axon survival rate
was approximately 80% at 1 week and decreased to approximately 60%
at 2 weeks and to approximately 40% at 4 weeks following ONC
injury. The axon survival rates between 1 and 4 weeks differed
significantly (p=0.029) (Fig. 3B,
control). In the rat model of ONC injury, the axon survival rate
may be used as an outcome measure for evaluating diagnosis and
treatment.
Axon survival ratios in rats with ONC
injury following the injection of CB-MNCs or CP-MSCs
Axon counting revealed the low survival rate in the
NTP group compared to the CB-MNC group and CP-MSC group, with the
representative images of each group at each time point shown in
Fig. 3A. The axon survival rates
were 75.1±18.05% at 1 week, 61.15±38.12% at 2 weeks and
38.08±26.02% at 4 weeks following ONC injury in the control group.
Compared with the control group, the axon survival rates were
87.46±16.34% at 1 week (p<0.05), 48.56±17.92% at 2 weeks, and
42.68±30.13% at 4 weeks following ONC injury in the
CB-MNC-transplanted group. The axon survival rates were
72.08±13.70% at 1 week, 48.17±19.57% at 2 weeks, and 39.96±24.88%
at 4 weeks following ONC injury in the CP-MSC-transplanted group
(Fig. 3B).
Thus, in this SD rat model of traumatic optic
neuropathy (ONC injury), the axon survival ratio increased
significantly at 1 week post CB-MNC transplantation.
CB-MNC and CP-MSC transplantation
upregulates GAP-43 expression
Generally, an inflammatory response coincides with
damage and repair processes in tissues, causing a hypoxic condition
that acts as a triggering factor for cell damage and also as a
necessary factor during optic nerve regeneration (25,26). In this study, to investigate the
potential anti-inflammatory effects of CB-MNC and CP-MSC
transplantation, we analyzed neuronal regeneration in the optic
nerve by measuring the expression levels of GAP-43 using
RT-qPCR. In the rats in the NTP group, no significant difference
was observed between the normal side and the ONC inuured side at 1
week post-injury. Furthermore, GAP-43 was expressed at low
levels in the optic nerve in the NTP group rats, whereas the
transplantation of the CP-MSCs resulted in a marked increase in
GAP-43 expression in the ONC injured optic nerve at 1 week
post transplantation (p<0.05; Fig.
4A). GAP-43 expression then decreased in the ONC injured
side in the CB-MNC-transplanted group or CP-MSC-transplanted group
compared to the NTP group at 4 weeks post-transplantation
(p<0.05; Fig. 4A). These
findings suggest that the transplantation of the CB-MNCs and
CP-MSCs increases the expression of axonal growth-associated
protein, mainly during the first stages of acute injury.
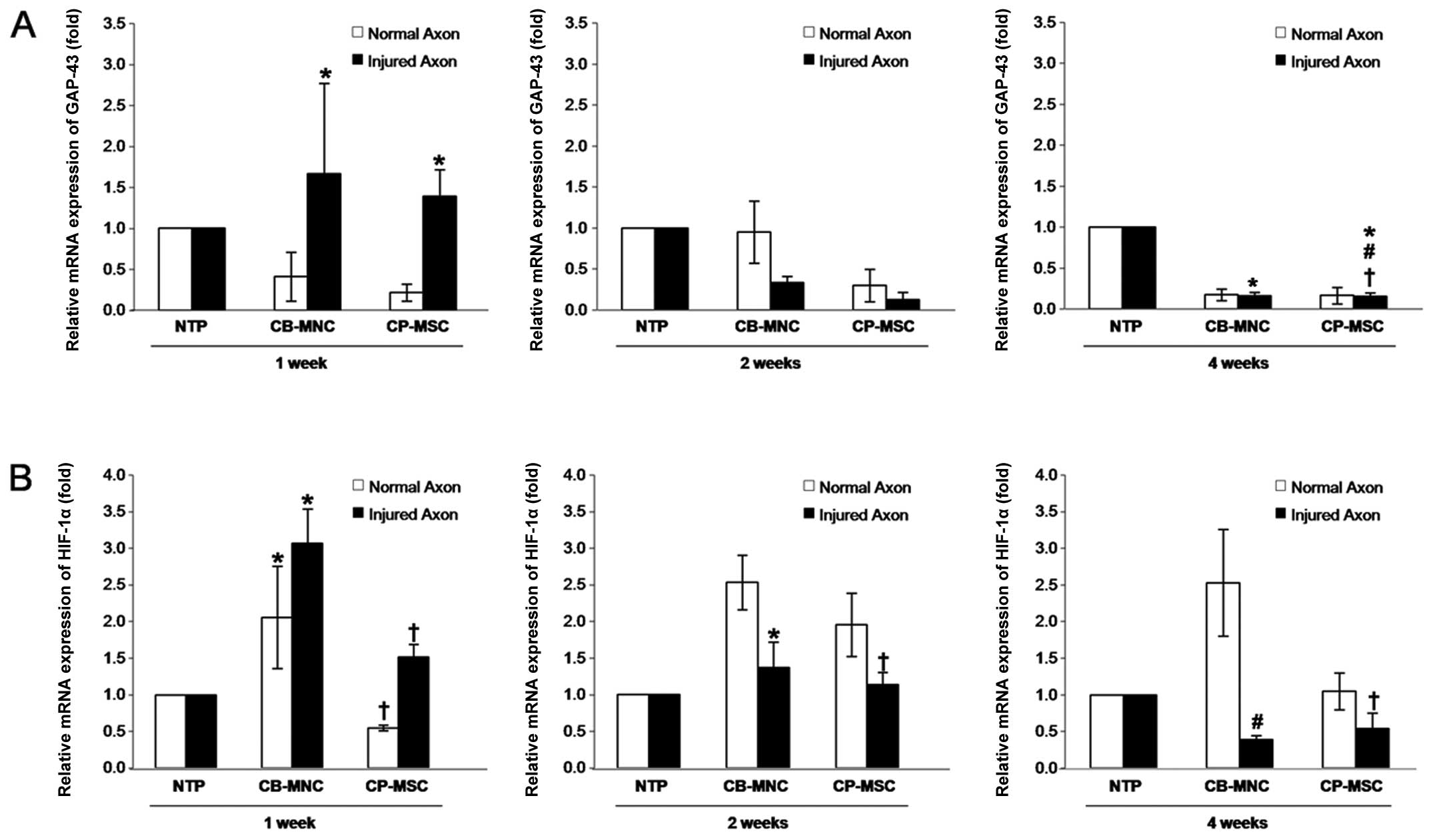 | Figure 4Expression and activity analyses of
growth-associated protein-43 (GAP-43) and hypoxia-inducible
factor-1α (HIF-1α) in the optic nerves from a rat model of optic
nerve crush (ONC) injury following umbilical cord blood mononuclear
cell (CB-MNC) or chorionic plate-derived MSC (CP-MSC)
transplantation. Equal amounts of RNA were pooled from individual
animals in the following groups and analyzed by RT-qPCR: the
control non-transplanted group (NTP) at 1 week (n=5), 2 weeks (n=5)
and 4 weeks (n=5), the CB-MNC-transplanted group (CB-MNC) at 1 week
(n=5), the CB-MNC-transplanted group at 2 weeks (n=5), the
CB-MNC-transplanted group at 4 weeks (n=5), as well as the
CP-MSC-transplanted group (CP-MSC) at 1 week (n=5), the
CP-MSC-transplanted group at 2 weeks (n=5), and the
CP-MSC-transplanted group at 4 weeks (n=5). (A) Determination of
the GAP-43 mRNA level using RT-qPCR. No significant
differences were observed between the normal side and the ONC
injured side in the rats in the NTP group at 1 week post-injury;
significant differences were observed between the NTP and
CP-MSC-transplanted groups in the ONC injured side at 1 week
(*p<0.05), and between the CB-MNC-transplanted group
or CP-MSC-transplanted group and NTP groups in the ONC injured side
at 4 weeks post-transplantation (*,#p<0.05). (B) The
mRNA expression of HIF-1α was determined. Significant
differences were observed between the NTP and CB-MNC-transplanted
groups at 1 week (*p<0.05), between the
CB-MNC-transplanted group and NTP group in the ONC injured side,
and the CB-MNC- and CP-MSC-transplanted groups at 2 weeks
post-transplantation (*,✝p<0.05), and. between the
normal side and ONC injured side in the CB-MSC- and
CP-MSC-transplanted groups at 4 weeks post-transplantation
(*,✝p<0.05). Data are expressed as the means ± SD of
triplicate experiments. |
CB-MNC transplantation upregulates HIF-1α
expression
As HIF-1α is a known trigger factor for adaptation
and survival-related signal transduction (27), we investigated whether CB-MNCs or
CP-MSCs induce its expression and activation. We found that in the
CB-MNC-transplanted group, HIF-1α expression was increased
significantly compared with that in the NTP group rats at 1 week
post-transplantation (p<0.05). Moreover, CB-MNC or CP-MSC
transplantation markedly enhanced HIF-1α expression in the normal
and ONC injured sides until 2 weeks post-ONC injury (p<0.05).
However, HIF-1α expression was reduced again in the ONC injured
side compared to the normal side in the CP-MSC- and
CB-MNC-transplanted groups at 2 weeks post-transplantation
(p<0.05; Fig. 4B). HIF-1α
expression also decreased in the ONC injured side in both the
CP-MSC- and CB-MNC-transplanted groups at 4 weeks
post-transplantation (p<0.05; Fig.
4B) These findings suggest that transplantation procedures may
induce HIF-1α expression, and the transplanted stem cells
contribute to recovery of HIF-1α expression and possibly, neuronal
survival in the injured optic nerve.
Histological examination of optic nerve
tissue from rats with ONC injury following CP-MSC or CB-MNC
transplantation
Sequential histological findings in the rat model of
ONC injury following the injection of CB-MNCs and CP-MSCs using
hematoxylin and eosin staining revealed subacute inflammation
around the compression area compared with the controls in the optic
nerve tissue 2 weeks post-CB-MNC injection. At 2 weeks post-CP-MSC
injection, subacute inflammation was observed in the optic nerve
tissue around the compression line, compared with that observed in
the controls (Fig. 5A).
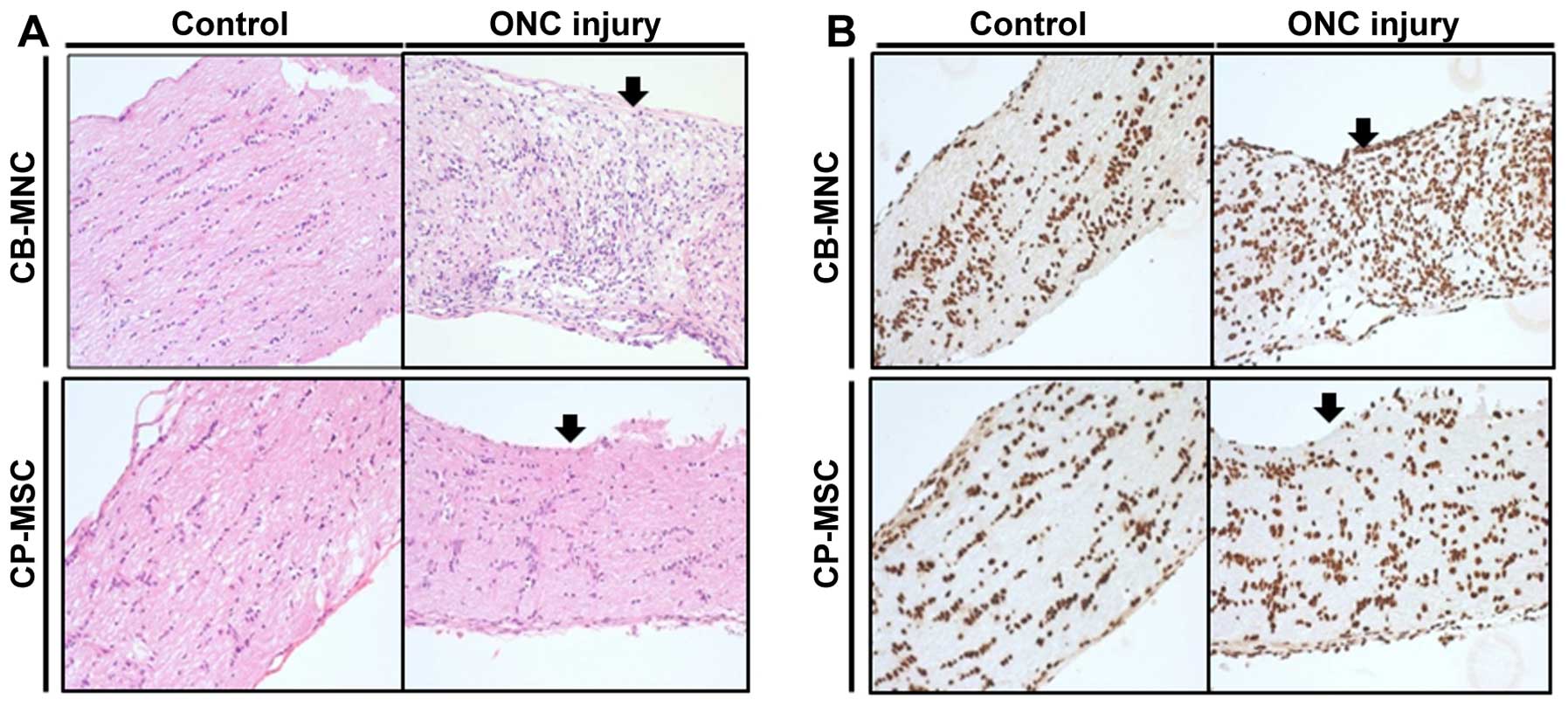 | Figure 5(A) Sequential histological findings
using hematoxylin and eosin staining of optic nerve samples from a
rat model of optic nerve compression after injection of umbilical
cord blood mononuclear cells (CB-MNCs) or chorionic plate-derived
MSCs (CP-MSCs). At 2 weeks post-CB-MNC injection, subacute
inflammation was observed in the optic nerve around the compression
area (arrow), compared with the controls. At 2 weeks post-CP-MSC
injection, subacute inflammation was observed in the optic nerve
around the compression line (arrow), compared with the control. (B)
Immunohistochemical findings in the optic nerve after injection
with CB-MNCs or CP-MSCs using 5-µm-thick sections cut from
formalin-fixed, paraffin wax-embedded tissue and the anti-human
nuclei and chromosomes, histone H1 protein antibody (mouse
monoclonal, clone 1415-1, 1:200 dilution). The BenchMark XT was
used as the visualization system together with heat-induced epitope
retrieval (CC1 solution) and the ultraView Universal DAB Detection
kit. At 2 weeks post-CB-MNC injection, the presence of surviving
human MNCs was hardly identified immunohistologically due to strong
cross reactivity (dark brown nuclei around the compression area
(arrow) in the optic nerve compared with the control). Similarly,
at 2 weeks post-CP-MSC injection, the presence of surviving human
MNCs was hardly identified immunohistologically due to strong cross
reactivity (dark brown nuclei around the compression area (arrow)
in the optic nerve compared with the control). All images are ×400
magnification. Control, non-transplanted group; CB-MNC,
CB-MNC-transplanted group; CP-MSC, CP-MSC-transplanted group. |
Immunohistochemical findings from the optic nerve
tissue 5-µm-thick) sections following the injection of
CB-MNCs or CP-MSCs revealed limitations in detecting the presence
of the surviving human MNCs, as it was difficult to identify human
nuclei and chromosomes, stained as dark brown nuclei, around the
compression area, compared with the control optic nerve, at 2 weeks
post-CB-MNC injection. At 2 weeks post-CP-MSC injection, it was
also very difficult to identify dark brown nuclei around the
compression area compared with the control optic nerve tissue
(Fig. 5B).
RNA analysis of optic nerve tissue from
rats with ONC injury post-CP-MSC or -CB-MNC injection by
RT-PCR
To determine whether the regeneration of the optic
nerve was induced by the injection of human cells or by natural
rescue, we selected the human-specific genes, ERMN and
LRAT, which are expressed specifically in the human retinal
pigment epithelium (RPE) (28).
These two genes differ in sequence between humans and mice. The
expression of these two genes was expected to demonstrate stem cell
differentiation and recovery of the damaged optic nerves. In
addition, we determined the expression of the another human
specific gene, SRGAP2, which is involved in neuronal
migration and differentiation (29,30). To confirm that stem cells are
still present in axons following the injection of the CP-MSCs, mRNA
expression was evaluated using human specific primers. To assess
mRNA expression, firstly, we isolated the optic nerves and
homogenized these samples using TRIzol reagent. The RNA was
extracted and subjected to cDNA synthesis. We then performed PCR
using primers specific to ERMN, LRAT and
SRGAP2. Also, human gene expression was confirmed in the
kidney tissue obtained from the rats in the NTP group as a negative
control and from the rats in the CP-MSC-transplanted group as a
positive control (Fig. 6A). A
human RNA signal was detected 1 week after the CP-MSC injection.
These findings indicated that recovery from ONC injury was promoted
by the injection of human stem cells. Western blot analysis was
performed to measure the SRGAP2, NF, ERMN and OCM protein in the
samples from CP-MSC-injected rats, indicating that recovery from
ONC injury was promoted by human stem cell injection after 1 week
(Fig. 6B). The results revealed
that the transplantation of the CP-MSCs resulted in a reduction in
ONC injury and damaged nerve function in the rats, suggesting that
transplanted CP-MSCs may provide protection against traumatic
injury to the optic nerve.
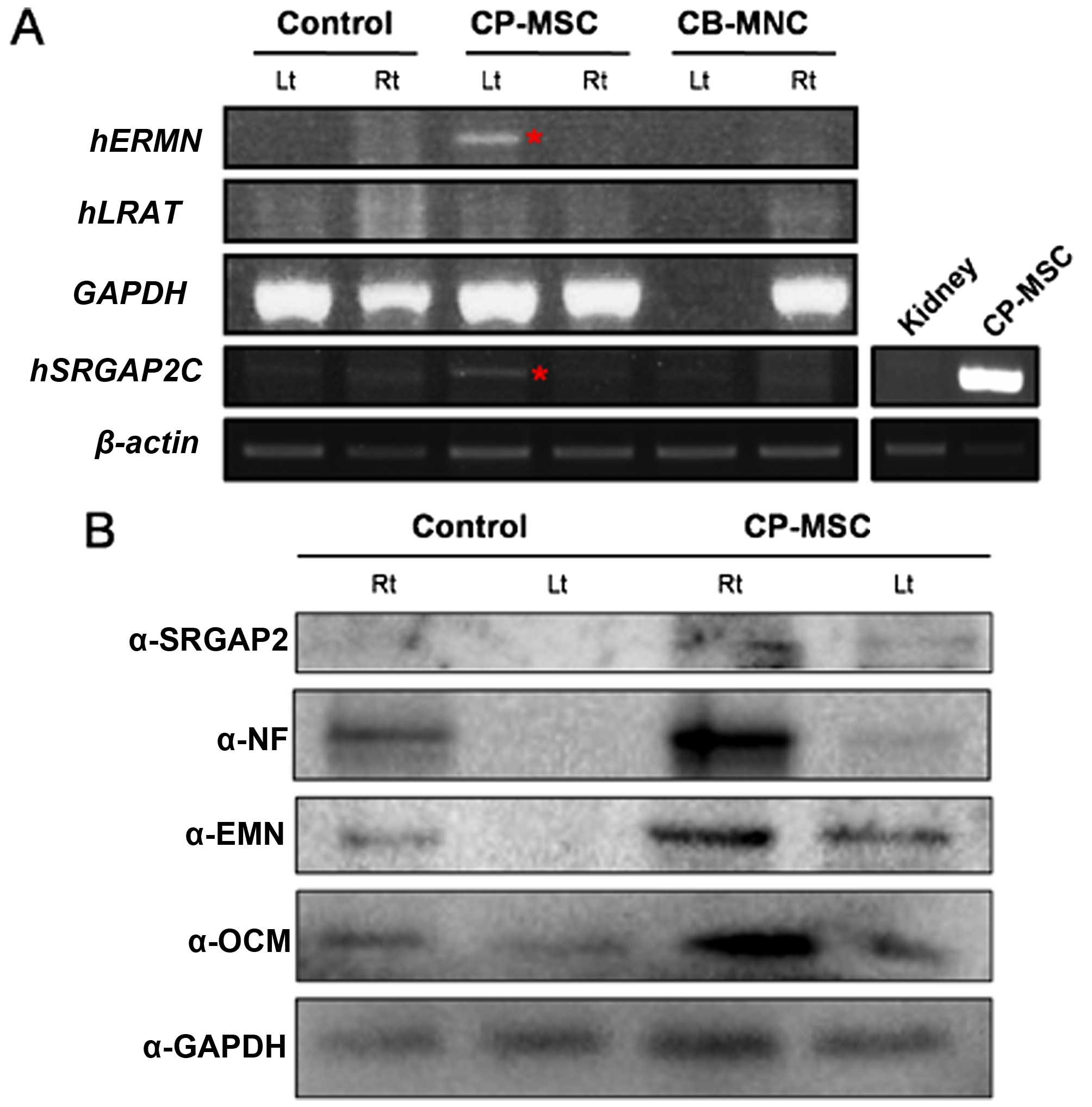 | Figure 6(A) RT-PCR analysis of human RNA in
the optic nerves from a rat model of optic nerve crush (ONC) injury
after umbilical cord blood mononuclear cell (CB-MNC) or chorionic
plate-derived MSC (CP-MSC) transplantation. Equal amounts of RNA
were pooled from individual animals of the following groups:
non-transplanted group (NTP) at 1 week post-injury,
CB-MNC-transplanted (CB-MNC) or CP-MSC-transplanted group (CP-MSC)
at 1 week, and CB-MNC-transplanted or CP-MSC-transplanted group at
2 weeks post-transplantation. A human RNA signal was detected 1
week post-CP-MSC injection (red asterisk). Actin was used as a
loading control. Kidney samples from the rats in the NTP group and
CP-MSC-transplatned group were used as the negative and positive
controls, respectively (side panel, bottom right). (B) SRGAP2, NF,
ERMN, and OCM levels were measured by western blot analysis using
pooled protein samples from individual animals of the NTP group and
CP-MSC-transplanted group at 2 weeks post-transplantation. GAPDH
was used as loading control. Rt, right eye; Lt, left eye-optic
nerve compression side; ERMN, ERM-like protein; LRAT,
lecithin-retinol acyltransferase; SRGAP2, SLIT-ROBO Rho GTPase
activating protein 2. |
Discussion
The mature optic nerve is not able to regenerate
when injured, leaving victims of traumatic nerve damage or diseases
with irreversible vision loss. Research is currently focused on
developing new treatment strategies to replenish damaged
photoreceptors and retinal ganglion cells (RGCs), and to replace
lost cells through transplants. The neuroprotective and
regenerative potential of stem and progenitor cells from a variety
of sources has been explored in models of retinal disease and
ganglion cell loss (1).
In a previous study (31), the optic nerve regeneration
process after an experimental axotomy performed on goldfish was
very long in duration; it took approximately half a year for visual
function to be fully recovered through synaptic refinement. The
first period was a preparation period, within 5–6 days after the
nerve lesion. The second period was an axonal elongation period,
within 1–5 weeks after the nerve lesion. The third period involved
synaptic connection and refinement in the tectum, within 1.5-4
months after the nerve lesion. The fourth period was a period of
visual function recovery, within 5–6 months after the nerve lesion
(31).
In this study, we counted the axons of the optic
nerve observed in digital images obtained at ×40 magnification
using a light microscope and performed histological examinations of
axonal density using ImageJ software. The loss of RGC axons in the
optic nerves of glaucomatous eyes was previously assessed using an
established semiquantitative optic nerve grading scheme with
modfications under a light microscope (32,33). The results of a previous study
validate the use of both fixed-pattern sampling and targeted
sampling for the estimation of axonal damage; however, it does not
indicate that the latter method is superior for detection of axon
loss in animals with minor damage (34). Recently, it was demonstrated that
the results of the semi-automated method for counting axons in TEM
images strongly correlated with those of conventional counting
methods and exhibited excellent reproducibility (34,35).
As previously demonstrated (36), the changes in GAP-43 expression
observed in the fish retina during optic nerve regeneration reflect
the state of visual function following nerve injury. GAP-43 is an
exceptional molecule, which has short peak (7–20 days) and long
plateau (50–120 days) phases in regrowing optic axons. However, the
location of GAP-43 at the presynaptic terminal, which is the
appropriate site for synaptic refinement of regrowing optic axons
in the tectum, has not been considered. However, GAP-43 is a well
known marker protein of growing axons during development and
regeneration (36).
HIF-1 is a dimeric transcriptional complex that has
been recognized primarily for its roles in the maintenance of
oxygen and energy homoeostasis. HIF-1 functions as a master
regulator of cellular and systemic homeostatic responses to hypoxia
by activating the transcription of several genes, including those
involved in energy metabolism, angiogenesis, apoptosis and those
whose protein products increase oxygen delivery or facilitate
metabolic adaptation to hypoxia. HIF-1 thus plays an essential role
in embryonic vascularization, tumor angiogenesis and the
pathophysiology of ischemic disease (37). The α-subunit of HIF-1 is
O2-labile and is degraded by proteasomes following
prolyl hydroxylation and ubiquitination in normoxic cells (38). It has also been shown that HIF-1α
is involved in immune reactions (38). Immunomodulatory peptides,
including interleukin-1 (IL-1) and tumor necrosis factor-α (TNF-α)
stimulate HIF-1-dependent gene expression even in normoxic cells.
Both the hypoxia- and cytokine-induced activation of HIF-1α
increases the transcription of several genes encoding proteins that
promote blood flow and inflammation, including vascular endothelial
growth factor (VEGF), hemeoxygenase-1, endothelial and inducible
nitric oxide synthetase (NOS) and cyclooxygenase-2 (COX-2)
(38). Thus, the pharmacological
activation of the HIF-1 complex may be desirable in ischemic and
inflammatory disorders. In this study, an increase in HIF-1α
expression was observed following the intra-arterial injection of
CB-MNCs or CP-MSCs at 1–2 weeks post-ONC injury. An increase in
HIF-1α expression was observed in the optic nerve injured sides
resulting by the engrafted stem cells CP-MSCs at 2 weeks
post-injury. Thus, stem cell supply may be beneficial for optic
nerve axons to survive or recover from perineural vascular rupture
and injury, as shown by the histological examination of the optic
nerve.
The differentiation of seeded bone marrow-derived
stem cells into mature vascular cells has previously been reported
(39). Our results support the
concept of the stem cell-mediated vascular regeneration and
recovery from ONC injury. As we did identify small populations of
hematopoietic progenitor cells used for seeding in the optic nerve,
we detected human-derived stem cells in the perineural tissues from
1 week post-implantation. Our data suggest that CP-MSCs or CB-MNCs
contribute to functional mature vascular regeneration through an
inflammation-mediated process of vascular remodeling that is
expedited by the secretion of CP-MSCs 1 week
post-transplantation.
A direct mechanism involving implanted blood stem
cells may still be involved in the recovery from neural or ischemic
injury (40). A paracrine role
for implanted blood stem cells in optic nerve recovery from injury
has been demonstrated in studies reporting the rare occurrence of
the transdifferentiation of various stem cells in vivo
(1,2). Studies examining the role of BM-MSCs
in cell therapies for neurological or ischemic diseases have
demonstrated that these cells do not differentiate into mature
endothelial cells or regenerating tissues but, instead, function by
releasing multiple cytokines that induce therapeutic angiogenesis,
arteriogenesis and/or cytoprotection (41,42).
Early data on transdifferentiation were likely
attributed to misinterpretations of cell fusion (43–46), explaining the results of
fluorescent membrane labeling techniques to track seeded stem
cells. As discussed elsewhere, injected mononuclear cells,
including monocytes/macrophages, are very adept at phagocytosis and
cell fusion (45). To the best of
our knowledge, murine cells have never been previously shown to
express human antigens in vivo; however, there was strong
cross reactivity observed between human cells and rat cells in our
immunohistochemical staining experiment.
Additionally, our rat model enabled us to validate
our immunohistochemical findings further using species-specific
RT-qPCR. Seeded stem cells do not need to be incorporated directly
into the vascular neotissue of the optic nerve. A certain type of
cord blood stem cell (CB-SC) is multipotent and displays embryonic
and hematopoietic characteristics. In previous studies, phenotypic
characterization demonstrated that CB-SCs display embryonic cell
markers [e.g., the transcription factors, octamer-binding
transcription factor 4 (OCT-4) and Nanog, stage-specific embryonic
antigen (SSEA)-3 and SSEA-4] and leukocyte common antigen CD45, but
are negative for blood cell lineage markers (e.g., CD1a, CD3, CD4,
CD8, CD11b, CD11c, CD13, CD14, CD19, CD20, CD34, CD41a, CD41b,
CD83, CD90, CD105 and CD133) (8).
Additionally, CB-SCs display very low
immunogenicity, as indicated by very low expression levels of major
histocompatibility complex (MHC) antigens and failure to stimulate
the proliferation of allogeneic lymphocytes (47).
It is generally recognized that no specific markers
are readily available to identify non-hematopoietic stem cells, and
CB-MNCs do not express hematopoietic stem cell markers, such as
CD45 or CD14. However, CB-MNCs do display a surface marker profile
similar to that of BM-MSCs. CB-MNCs express the stromal markers,
CD9, CD10, CD29, CD44, CD73, CD90, CD166, as well as CD34, which is
a cell-surface marker also expressed by endothelial cells. CD34(+)
and total cell numbers also affect the outcomes in that greater
cell numbers are associated with a 'better' response (9,10).
The high potential of human placenta-derived stem
cells for proliferation, differentiation and self-renewal has been
considered; moreover, these cells are readily available and are
easily procured without the need for invasive procedures (22). Among several types of PD-MSCs,
CP-MSCs have been shown to differentiate into a variety of cell
types and to possess effective anti-inflammatory properties
(20,21).
In terms of a novel therapeutic mechanism,
intravenously infused CB-MNCs may act as remote bioreactors that
eventually increase systemic anti-inflammatory or anti-ischemic
cytokine production following sequestration into the lung
microvasculature and parenchyma, and entrapment in the spleen by
the direct stimulation of pulmonary macrophages and spleen-naïve T
cells, as previously reported (11). The potential of infused stem cells
to trigger autophagy to enhance regeneration and/or provide a
therapeutic effect as cellular survival promoters is of interest
(23,48). A previous study focused on the
direct mechanisms of CP-MSC therapeutic effectiveness, as well as
on an indirect paracrine mechanism (40,49). In this study, we provide direct
evidence of the effects of CP-MSCs through incorporation into the
optic nerves and increased axonal density, matched with enhanced
neuronal regeneration markers and HIF.
To date, no reported study has indicated an
effective cell therapy application method in acute optic nerve
injury, to the best of our knowledge. A previous study on systemic
CB-MNC administration for brain injury revealed no remaining cells
1 month after therapy, even with the continuous administration of
cyclosporine (50). Thus,
proximal injection of stem cells into the optic nerve may be the
optimal route to maximize the collection of implanted stem cells
around the optic nerve and minimize systemic spreading. However, we
considered the application method for the high survival rate of
stem cells after the systemic circulation, which provided the
chance to enter the ophthalmic artery branching to pial vessels of
optic nerves. Between the two methods of systemic injection, such
as intravenously or intra-arterially, the majority of the stem
cells injected intravenously are sequestered into the lungs and
hardly enter to the arterial circulation to the ophthalmic artery.
Among the proximal arteries branching to the ophthalmic artery, the
internal carotid artery is relatively easily accessable and thick
enough to engage the 26 gauge needle of injection.
Erythropoietin and granulocyte colony-stimulating
factors have neuroprotective and neural-repair properties,
particularly in ischemic brain injury (17). As certain studies have
demonstrated the neuroprotective effects of cyclosporine and the
activation of the AKT pathway, similar to erythropoietin, and that
the combination of the two exerts even more potent neuroprotective
effects, additional medications may also enhance the efficacy of
CB-MNC transplantation (11,19); however, neurotoxicity has been
reported after long-term use of cyclosporine (51). As our study demonstrated, CB-MNCs
and CP-MSCs indeed exerted protective effects against optic nerve
injury in rats undergoing intra-arterial transplantation. However,
the translation of these findings into clinical therapies has been
achieved to a limited extent. Thus, further studies are warranted
to delineate the long-term effects of CB-MNC and CP-MSC
transplantation, to examine the differences in efficacy, injection
time and stem cell numbers between the two cell types, and to fully
elucidate the mechanisms of action of CB-MNCs and CP-MSCs, and thus
to develop easier and safer methods of stem cell
transplantation.
In conclusion, regenerative medicine using human
stem cells is a novel and promising research field, which has
potential for use in the treatment of various intractable diseases
and damaged organs, including difficult-to-treat acute optic nerve
injuries. Taken together, the findings of the present study
demonstrate that CP-MSC or CB-MNC transplantation using a rat model
of ONC injury ameliorates optic nerve damage and increases axon
survival through incorporation in the optic nerve, as well as an
increase in the HIF-1α and GAP-43 expression levels.
Acknowledgments
The present study was supported by the National
Research Foundation of Korea (NRF) grant funded by the Korean
government (MSIP) (no. NRF-2014R1A2A2A01002617).
References
|
1
|
Zhao T, Li Y, Tang L, Li Y, Fan F and
Jiang B: Protective effects of human umbilical cord blood stem cell
intravitreal transplantation against optic nerve injury in rats.
Graefes Arch Clin Exp Ophthalmol. 249:1021–1028. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Junyi L, Na L and Yan J: Mesenchymal stem
cells secrete brain-derived neurotrophic factor and promote retinal
ganglion cell survival after traumatic optic neuropathy. J
Craniofac Surg. 26:548–552. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bull ND, Irvine KA, Franklin RJ and Martin
KR: Transplanted oligodendrocyte precursor cells reduce
neurodegeneration in a model of glaucoma. Invest Ophthalmol Vis
Sci. 50:4244–4253. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Aoki H, Hara A, Niwa M, Motohashi T,
Suzuki T and Kunisada T: Transplantation of cells from eye-like
structures differentiated from embryonic stem cells in vitro and in
vivo regeneration of retinal ganglion-like cells. Graefes Arch Clin
Exp Ophthalmol. 246:255–265. 2008. View Article : Google Scholar
|
|
5
|
Bull ND, Limb GA and Martin KR: Human
Müller stem cell (MIO-M1) transplantation in a rat model of
glaucoma: survival, differentiation, and integration. Invest
Ophthalmol Vis Sci. 49:3449–3456. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Levkovitch-Verbin H, Sadan O, Vander S,
Rosner M, Barhum Y, Melamed E, Offen D and Melamed S: Intravitreal
injections of neurotrophic factors secreting mesenchymal stem cells
are neuroprotective in rat eyes following optic nerve transection.
Invest Ophthalmol Vis Sci. 51:6394–6400. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Charalambous P, Hurst LA and Thanos S:
Engrafted chicken neural tube-derived stem cells support the innate
propensity for axonal regeneration within the rat optic nerve.
Invest Ophthalmol Vis Sci. 49:3513–3524. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Canque B, Camus S, Dalloul A, Kahn E,
Yagello M, Dezutter-Dambuyant C, Schmitt D, Schmitt C and Gluckman
JC: Characterization of dendritic cell differentiation pathways
from cord blood CD34(+)CD7(+)CD45RA(+) hematopoietic progenitor
cells. Blood. 96:3748–3756. 2000.PubMed/NCBI
|
|
9
|
Kao CH, Chen SH, Chio CC and Lin MT: Human
umbilical cord blood-derived CD34+ cells may attenuate
spinal cord injury by stimulating vascular endothelial and
neurotrophic factors. Shock. 29:49–55. 2008.
|
|
10
|
Lee MW, Jang IK, Yoo KH, Sung KW and Koo
HH: Stem and progenitor cells in human umbilical cord blood. Int J
Hematol. 92:45–51. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Min K, Song J, Kang JY, Ko J, Ryu JS, Kang
MS, Jang SJ, Kim SH, Oh D, Kim MK, et al: Umbilical cord blood
therapy potentiated with erythropoietin for children with cerebral
palsy: a double-blind, randomized, placebo-controlled trial. Stem
Cells. 31:581–591. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Newcomb JD, Sanberg PR, Klasko SK and
Willing AE: Umbilical cord blood research: current and future
perspectives. Cell Transplant. 16:151–158. 2007.PubMed/NCBI
|
|
13
|
Lu D, Sanberg PR, Mahmood A, Li Y, Wang L,
Sanchez-Ramos J and Chopp M: Intravenous administration of human
umbilical cord blood reduces neurological deficit in the rat after
traumatic brain injury. Cell Transplant. 11:275–281.
2002.PubMed/NCBI
|
|
14
|
Meier C, Middelanis J, Wasielewski B,
Neuhoff S, Roth-Haerer A, Gantert M, Dinse HR, Dermietzel R and
Jensen A: Spastic paresis after perinatal brain damage in rats is
reduced by human cord blood mononuclear cells. Pediatr Res.
59:244–249. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nan Z, Grande A, Sanberg CD, Sanberg PR
and Low WC: Infusion of human umbilical cord blood ameliorates
neurologic deficits in rats with hemorrhagic brain injury. Ann N Y
Acad Sci. 1049:84–96. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nikolic WV, Hou H, Town T, Zhu Y, Giunta
B, Sanberg CD, Zeng J, Luo D, Ehrhart J, Mori T, et al:
Peripherally administered human umbilical cord blood cells reduce
parenchymal and vascular beta-amyloid deposits in Alzheimer mice.
Stem Cells Dev. 17:423–439. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Papadopoulos KI, Low SS, Aw TC and
Chantarojanasiri T: Safety and feasibility of autologous umbilical
cord blood transfusion in 2 toddlers with cerebral palsy and the
role of low dose granulocyte-colony stimulating factor injections.
Restor Neurol Neurosci. 29:17–22. 2011.PubMed/NCBI
|
|
18
|
Sun J, Allison J, McLaughlin C, Sledge L,
Waters-Pick B, Wease S and Kurtzberg J: Differences in quality
between privately and publicly banked umbilical cord blood units: a
pilot study of autologous cord blood infusion in children with
acquired neurologic disorders. Transfusion. 50:1980–1987. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Vendrame M, Cassady J, Newcomb J, Butler
T, Pennypacker KR, Zigova T, Sanberg CD, Sanberg PR and Willing AE:
Infusion of human umbilical cord blood cells in a rat model of
stroke dose-dependently rescues behavioral deficits and reduces
infarct volume. Stroke. 35:2390–2395. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kim MJ, Shin KS, Jeon JH, Lee DR, Shim SH,
Kim JK, Cha DH, Yoon TK and Kim GJ: Human chorionic-plate-derived
mesenchymal stem cells and Wharton's jelly-derived mesenchymal stem
cells: a comparative analysis of their potential as
placenta-derived stem cells. Cell Tissue Res. 346:53–64. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lee JM, Jung J, Lee HJ, Jeong SJ, Cho KJ,
Hwang SG and Kim GJ: Comparison of immunomodulatory effects of
placenta mesenchymal stem cells with bone marrow and adipose
mesenchymal stem cells. Int Immunopharmacol. 13:219–224. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Parolini O, Alviano F, Bagnara GP, Bilic
G, Bühring HJ, Evangelista M, Hennerbichler S, Liu B, Magatti M,
Mao N, et al: Concise review: isolation and characterization of
cells from human term placenta: outcome of the first international
Workshop on Placenta Derived Stem Cells. Stem Cells. 26:300–311.
2008. View Article : Google Scholar
|
|
23
|
Jung J, Choi JH, Lee Y, Park JW, Oh IH,
Hwang SG, Kim KS and Kim GJ: Human placenta-derived mesenchymal
stem cells promote hepatic regeneration in CCl4-injured
rat liver model via increased autophagic mechanism. Stem Cells.
31:1584–1596. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lee MJ, Jung J, Na KH, Moon JS, Lee HJ,
Kim JH, Kim GI, Kwon SW, Hwang SG and Kim GJ: Anti-fibrotic effect
of chorionic plate-derived mesenchymal stem cells isolated from
human placenta in a rat model of CCl4-injured liver:
potential application to the treatment of hepatic diseases. J Cell
Biochem. 111:1453–1463. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Koriyama Y, Homma K, Sugitani K, Higuchi
Y, Matsukawa T, Murayama D and Kato S: Upregulation of IGF-I in the
goldfish retinal ganglion cells during the early stage of optic
nerve regeneration. Neurochem Int. 50:749–756. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Benowitz LI and Yin Y: Optic nerve
regeneration. Arch Ophthalmol. 128:1059–1064. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tajima T, Goda N, Fujiki N, Hishiki T,
Nishiyama Y, Senoo-Matsuda N, Shimazu M, Soga T, Yoshimura Y,
Johnson RS and Suematsu M: HIF-1alpha is necessary to support
gluconeogenesis during liver regeneration. Biochem Biophys Res
Commun. 387:789–794. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Booij JC, ten Brink JB, Swagemakers SM,
Verkerk AJ, Essing AH, van der Spek PJ and Bergen AA: A new
strategy to identify and annotate human RPE-specific gene
expression. PLoS One. 5:e93412010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Guerrier S, Coutinho-Budd J, Sassa T,
Gresset A, Jordan NV, Chen K, Jin WL, Frost A and Polleux F: The
F-BAR domain of srGAP2 induces membrane protrusions required for
neuronal migration and morphogenesis. Cell. 138:990–1004. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Dennis MY, Nuttle X, Sudmant PH, Antonacci
F, Graves TA, Nefedov M, Rosenfeld JA, Sajjadian S, Malig M,
Kotkiewicz H, et al: Evolution of human-specific neural SRGAP2
genes by incomplete segmental duplication. Cell. 149:912–922. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kato S, Devadas M, Okada K, Shimada Y,
Ohkawa M, Muramoto K, Takizawa N and Matsukawa T: Fast and slow
recovery phases of goldfish behavior after transection of the optic
nerve revealed by a computer image processing system. Neuroscience.
93:907–914. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Johnson TV, Bull ND, Hunt DP, Marina N,
Tomarev SI and Martin KR: Neuroprotective effects of intravitreal
mesenchymal stem cell transplantation in experimental glaucoma.
Invest Ophthalmol Vis Sci. 51:2051–2059. 2010. View Article : Google Scholar :
|
|
33
|
Ebneter A, Casson RJ, Wood JP and Chidlow
G: Estimation of axon counts in a rat model of glaucoma: comparison
of fixed-pattern sampling with targeted sampling. Clin Experiment
Ophthalmol. 40:626–633. 2012. View Article : Google Scholar
|
|
34
|
Kim CY, Rho S, Lee NE, Lee CK and Sung YJ:
Semi-automated counting method of axons in transmission electron
microscopic images. Vet Ophthalmol. 19:29–37. 2016. View Article : Google Scholar
|
|
35
|
Rho S, Park I, Seong GJ, Lee N, Lee CK,
Hong S and Kim CY: Chronic ocular hypertensive rat model using
microbead injection: comparison of polyurethane,
polymethylmethacrylate, silica and polystyene microbeads. Curr Eye
Res. 39:917–927. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kaneda M, Nagashima M, Mawatari K, Nunome
T, Muramoto K, Sugitani K and Kato S: Growth-associated protein43
(GAP43) is a biochemical marker for the whole period of fish optic
nerve regeneration. Adv Exp Med Biol. 664:97–104. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Vollmer S, Kappler V, Kaczor J, Flügel D,
Rolvering C, Kato N, Kietzmann T, Behrmann I and Haan C:
Hypoxia-inducible factor 1alpha is up-regulated by oncostatin M and
participates in oncostatin M signaling. Hepatology. 50:253–260.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Hellwig-Bürgel T, Stiehl DP, Wagner AE,
Metzen E and Jelkmann W: Review: hypoxia-inducible factor-1
(HIF-1): a novel transcription factor in immune reactions. J
Interferon Cytokine Res. 25:297–310. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kinnaird T, Stabile E, Burnett MS, Shou M,
Lee CW, Barr S, Fuchs S and Epstein SE: Local delivery of
marrow-derived stromal cells augments collateral perfusion through
paracrine mechanisms. Circulation. 109:1543–1549. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Gnecchi M, He H, Liang OD, Melo LG,
Morello F, Mu H, Noiseux N, Zhang L, Pratt RE, Ingwall JS and Dzau
VJ: Paracrine action accounts for marked protection of ischemic
heart by Akt-modified mesenchymal stem cells. Nat Med. 11:367–368.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kinnaird T, Stabile E, Burnett MS, Lee CW,
Barr S, Fuchs S and Epstein SE: Marrow-derived stromal cells
express genes encoding a broad spectrum of arteriogenic cytokines
and promote in vitro and in vivo arteriogenesis through paracrine
mechanisms. Circ Res. 94:678–685. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
O'Neill TJ IV, Wamhoff BR, Owens GK and
Skalak TC: Mobilization of bone marrow-derived cells enhances the
angiogenic response to hypoxia without transdifferentiation into
endothelial cells. Circ Res. 97:1027–1035. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Terada N, Hamazaki T, Oka M, Hoki M,
Mastalerz DM, Nakano Y, Meyer EM, Morel L, Petersen BE and Scott
EW: Bone marrow cells adopt the phenotype of other cells by
spontaneous cell fusion. Nature. 416:542–545. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Uemura R, Xu M, Ahmad N and Ashraf M: Bone
marrow stem cells prevent left ventricular remodeling of ischemic
heart through paracrine signaling. Circ Res. 98:1414–1421. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Alvarez-Dolado M, Pardal R, Garcia-Verdugo
JM, Fike JR, Lee HO, Pfeffer K, Lois C, Morrison SJ and
Alvarez-Buylla A: Fusion of bone-marrow-derived cells with Purkinje
neurons, cardiomyocytes and hepatocytes. Nature. 425:968–973. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Nygren JM, Jovinge S, Breitbach M, Säwén
P, Röll W, Hescheler J, Taneera J, Fleischmann BK and Jacobsen SE:
Bone marrow-derived hematopoietic cells generate cardiomyocytes at
a low frequency through cell fusion, but not transdifferentiation.
Nat Med. 10:494–501. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
47
|
Li C, Zhang W, Jiang X and Mao N:
Human-placenta-derived mesenchymal stem cells inhibit proliferation
and function of allogeneic immune cells. Cell Tissue Res.
330:437–446. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Sanchez CG, Penfornis P, Oskowitz AZ,
Boonjindasup AG, Cai DZ, Dhule SS, Rowan BG, Kelekar A, Krause DS
and Pochampally RR: Activation of autophagy in mesenchymal stem
cells provides tumor stromal support. Carcinogenesis. 32:964–972.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Lee Y, Jung J, Cho KJ, Lee SK, Park JW, Oh
IH and Kim GJ: Increased SCF/c-kit by hypoxia promotes autophagy of
human placental chorionic plate-derived mesenchymal stem cells via
regulating the phosphorylation of mTOR. J Cell Biochem. 114:79–88.
2013. View Article : Google Scholar
|
|
50
|
Gornicka-Pawlak el B, Janowski M, Habich
A, Jablonska A, Drela K, Kozlowska H, Lukomska B, Sypecka J and
Domanska-Janik K: Systemic treatment of focal brain injury in the
rat by human umbilical cord blood cells being at different level of
neural commitment. Acta Neurobiol Exp (Wars). 71:46–64. 2011.
|
|
51
|
Gijtenbeek JM, van den Bent MJ and Vecht
CJ: Cyclosporine neurotoxicity: A review. J Neurol. 246:339–346.
1999. View Article : Google Scholar : PubMed/NCBI
|