Introduction
The prevalence and mortality of colon cancer are the
highest among malignant tumors in Western countries and Japan
(1). Currently, resection of
metastatic foci and chemotherapy have been shown to be effective in
the treatment of liver and lung metastases and other hematogenous
metastases (2). In colon cancer
patients, however, the incidence of peritoneal dissemination is 7%
of initial colon cancers and 4–19% of recurrent colon cancers, but
no effective treatment modalities for this peritoneal dissemination
have been established, which is a major problem (3,4). At
present, cytoreduction and hyperthermic intraperitoneal
chemotherapy (HIPEC) are conducted for peritoneal dissemination,
and there are studies of improved outcomes and long-term survival
in some patients (5,6). Sugarbaker scored peritoneal
dissemination anatomically and with consideration of tumor
diameter, and investigated the contribution to outcome when
cytoreduction and HIPEC were conducted (7). Recently, Terence et al scored
clinical symptoms, extent of carcinomatosis, and tumor pathology in
patients who underwent HIPEC, and reported that the effect differs
depending on the score (8). As
there have been no reported molecular biological investigations on
the efficacy of HIPEC, we decided to search for useful
molecules.
Mucins have attracted attention as substances that
play a large role in the protective mechanisms of normal colonic
mucosa. Mucins are classified according to basic core protein type.
This core protein is abbreviated MUC, and 21 types of mucin have
been reported to date. Mucins are broadly classified into two
types: 1) Secretory mucins, which are secreted from epithelial
cells and are a main component of mucus in the traditional sense,
and these mucin molecules form gels; and 2) Membrane-bound mucins,
which bind to cell membranes. Mucin molecules have an extracellular
domain, a transmembrane domain and an intracellular domain, and
exist in a form that passes through the cell membrane (9,10).
Among these, the secretory type MUC2 is a major
mucin that is recognized to be expressed in the normal intestinal
tract, but expression of MUC5AC and MUC3 is also seen. The mucosal
layer of organs that contact the external world is protected
physically by mucins, and they are thought to be important
molecules in biological defense (11,12).
Mucins are also reported to be involved in carcinogenesis of the
intestinal tract, one of the causes is thought to be that the
intestinal mucosa is susceptible to chronic inflammation when
mucins are deficient (13,14). Thus, mucins secreted in normal
mucosa are thought to act to protect the body’s own cells from
external influences. Moreover, the expression of mucin family
proteins is not uniform in various malignant tumors, and expression
of MUC proteins is conjectured to serve certain function in cancer
cells (15,16). Thus, by enhancing expression in
cancer cells themselves, they are also thought to protect against
assault from anticancer agents or other substances from the
external world, contributing to anticancer agent resistance and
worsening prognosis. In the present study, therefore, we
investigated the effectiveness of HIPEC and whether expression of
mucin family proteins are involved.
Patients and methods
Patients
The subjects were 935 colon cancer patients who had
undergone resection for colorectal cancer at the First Department
of Surgery, University of Fukui, Japan, between 1994 and 2010
(Table I). Surgical specimens of
the peritoneal dissemination were obtained from 37 patients with
peritoneal metastases as the only distant metastasis of primary
colon cancer. HIPEC was performed in 22 patients with peritoneal
dissemination as the only synchronous distant metastasis in cases
of primary colon cancer at our hospital from 1994 to 2010. The
outcome was then compared with that of patients (15 cases) who did
not receive HIPEC. Cancers were reviewed and graded by two
pathologists using criteria recommended by the general rules of
clinical and pathological studies on cancer of colon, rectum and
anus for histological type, lymphatic invasion and venous invasion
(17). The research was performed
in accordance with the humane and ethical rules for human
experimentation that are stated in the Declaration of Helsinki.
 | Table IPatient characteristics. |
Table I
Patient characteristics.
| HIPEC cases | Non-HIPEC cases |
|---|
| No. of patients | 22 | 15 |
| Gender (M/F) | 10/22 | 8/7 |
| Age (years) | 54.1 (31–74) | 70.5 (47–81) |
| Differentiation |
| Well, mod | 17 | 6 |
| Others | 5 | 9 |
|
Synchronous/metachronous | 17/5 | 15/0 |
HIPEC procedure
This procedure allowed the abdominal cavity to be
extended widely enough to allow perfusate to spread throughout the
peritoneal cavity (18). Two
liters of saline contained 150 mg of cisplatin, 20 mg mitomycin C,
and 200 mg of etoposide. An additional 2 litres of the same
infusate were heated in a waterbath and pumped into circulation
between the abdomen and a reservoir at ~500 ml per minute. The
temperature at several points of the peritoneal surface was
maintained at appromimately 43°C by controlling the temperature of
the water bath and the speed of the pump. Abdominal temperatures
were measured at the serosal surface in the subphrenic space and
the cavity of Douglas, and the temperature of the infusate was also
measured in the inflow tube, outflow tube, and water bath. The
thermal dose (TD) obtained during the treatment was calculated
simultaneously during HIPEC and expressed in terms of equivalent
time at 43°C (19). HIPEC was
performed until the TD reached 40 min.
Immunohistostaining
The patients with peritoneal dissemination as the
only synchronous distant metastasis in cases of primary colon
cancer were analyzed for MUC1, 2, 3, 4, and 5AC protein expression.
Surgical specimens of the peritoneal dissemination prepared from
formalin-fixed, paraffin-embedded tissues were analyzed for protein
expression by the streptavidin-biotin peroxidase method (20). The expression was interpreted as
positive when the protein was expressed in >30% of the cancer
cells.
Antibody
The following antibodies were used: anti-human MUC1,
2, and MUC5 (Novocastra, UK), MUC3 (Santa Cruz Biotechnology, CA,
USA), MUC4 (Invitrogen, CA, USA).
Cell culture
The human colon cancer cell lines (SW620, colo205,
and LoVo) were cultured at 37°C in 5% CO2 in RPMI-1640
or DMEM medium containing 10% fetal bovine serum.
Total RNA extraction, RT-PCR
analysis
Total RNA was extracted from cells using Isogen
(Wako, Japan) (21). Single strand
cDNA prepared from 3 μg of total RNA using Moloney murine leukemia
virus reverse transcriptase (Gibco-BRL, MD, USA) with an oligo (dT)
primer-14 was used as the template for the polymerase chain
reaction (PCR). The primers for PCR to amplify MUC2 gene-coding
regions were: The 5′ primer, MUC2-AX: TGCCTGGCCCTGTCTTTG and the 3′
primer, MUC2-BX: CAGCTCCAGCATGAGTGC. Thirty cycles of denaturation
(94°C, 1 min), annealing (50°C, 0.75 min), and extension (72°C, 2
min) were carried out in a thermal cycler (PTC-100, programmable
thermal controller, NJ Research Inc., MA, USA). GAPDH amplification
was used as internal PCR control with
5′-GGGGAGCCAAAAGGGTCATCATCT-3′ as the sense primer and
5′-GACGCCTGCTTCACCACCTTCTTG-3′ as the antisense primer. Thirty
cycles of denaturation (94°C, 1 min), annealing (50°C, 1.5 min),
and extension (72°C, 2 min) were carried out in a thermal cycler.
PCR product (10 μl) was resolved by electrophoresis in 12%
acrylamide gel. The sequencing was performed on PCR products that
showed the bands in RT-PCR analysis. Sequence analysis showed the
presence of the MUC2 gene.
Transfection
The cells were cultured with RPMI-1640, 10% FBS and
1X penicillin/streptomycin at 37°C and 5% CO2 incubator.
MUC2-siRNA (Invitrogen) and scrambled (SCR)-siRNA (Invitrogen) were
purchased. Cells were transfected with 100 nM of MUC2-siRNA, or
SCR-siRNA with Lipofectamine 2000 reagent (Invitrogen) in
accordance with the manufacturer’s protocol. SCR-siRNA was used as
negative control.
Heat and anticancer agent treatment
The cells were plated onto a 96-well plate at
1×104 and incubated for 12 h. The cells were treated
with cisplatin of 100 μg/ml at 43°C (5% CO2 incubator)
for 24 h.
MTS analysis
To assess cell proliferation, Cell Titer 96 Aqueous
Non-radioactive Cell Proliferation Assay (Promega, Germany) was
used according to the manufacturer’s instructions. MTS solution (20
μl) was added to each well (96-well plate) and the plates were
incubated at 37°C for 1.5 h. The absorbance of the product
formazan, which is considered to be directly proportional to the
number of living cells in the culture, was measured at 490 nm using
a Microplate Reader (Molecular Devices, CA, USA).
Statistical considerations
Survival time was calculated using the Kaplan-Meier
method, and log-rank test was used to compare the curves of the
survival times.
Other characteristics of the two treatment arms were
compared using the chi-square test. Values of P<0.05 were
considered as statistically significant.
Results
Relationship between survival rate and
whether HIPEC was used in the patients with peritoneal
dissemination of colon cancer
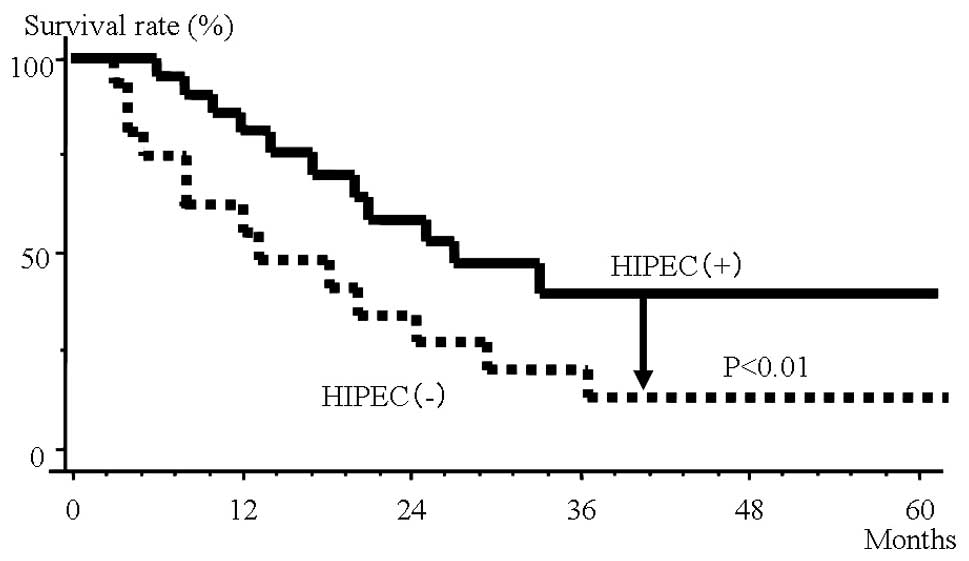
Fig. 1 shows the
survival rates in the HIPEC and non-HIPEC groups among all subjects
with peritoneal dissemination. In the non-HIPEC group, the median
survival time was 13 months and the 3-year survival rate was 15.6%,
whereas in the HIPEC group the median survival time was 24 months
and the 3-year survival rate was 39.2%. The HIPEC group thus had
significantly better outcomes.
Investigation of expression of mucin
proteins in dissemination foci of patients with peritoneal
dissemination of colon cancer
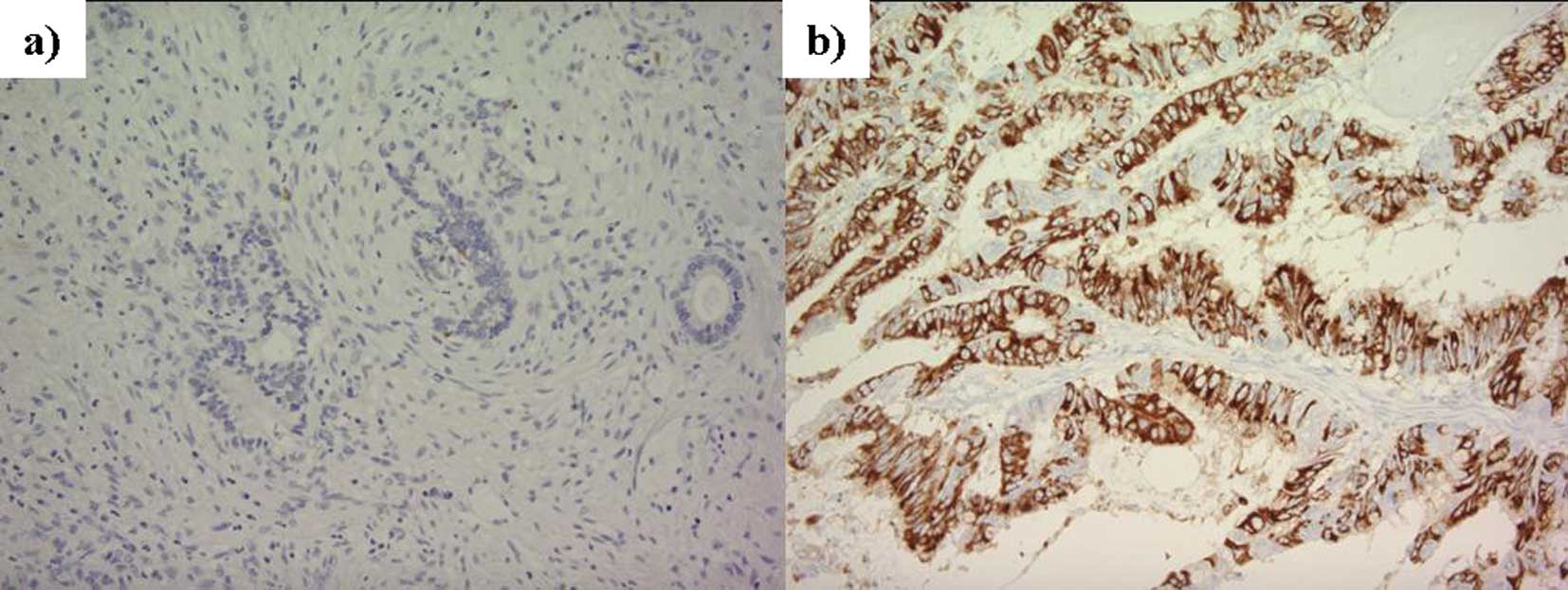
Fig. 2 shows images
of positive and negative immunohistochemical staining using
anti-MUC2 antibody in dissemination foci in patients with
peritoneal dissemination of colorectal cancer, and Fig. 3 shows their incidence. Of the 22
patients, MUC1 protein expression was seen in 19 patients (86.4%),
MUC2 in 10 (45.5%), MUC3 in 10 (45.5%), MUC4 in 15 (68.2%), and
MUC5AC was seen in 15 (68.2%).
Investigation of MUC2 protein expression
and outcome in colon cancer patients with peritoneal dissemination
who underwent HIPEC
Expression of MUC2 protein and outcome was
investigated in the patients with peritoneal dissemination of colon
cancer in the HIPEC group. In patients positive for MUC2
expression, the MST was 14 months and the 3-year survival rate was
0.0%, whereas in patients negative for MUC2 expression, the 3-year
survival rate was 61.1% (Fig. 3).
Patients negative for MUC2 expression thus had a significantly
better outcome. In the non-HIPEC group, no relationship was seen
between outcome and expression of MUC2 protein. There was no
significant difference between survival time and presence or
absence of MUC1, 3, 4 and 5AC expression (Fig. 4a–d).
Investigation of MUC2 mRNA expression in
colon cancer cell lines
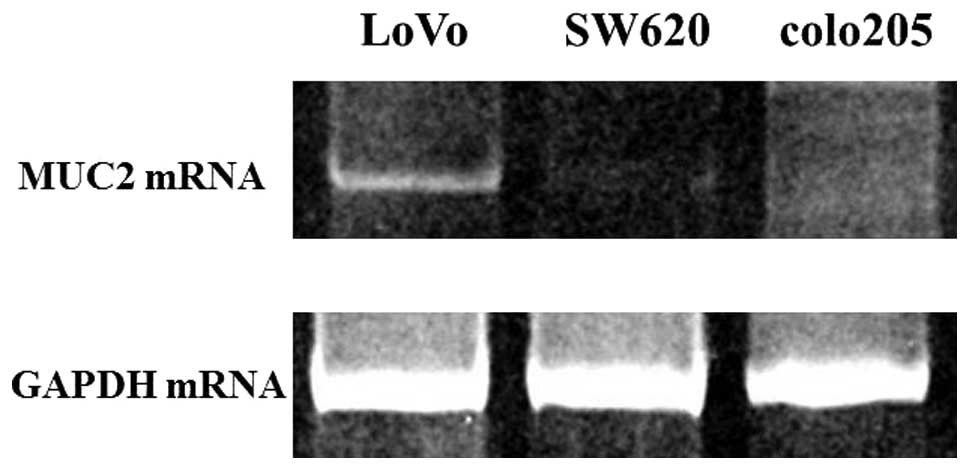
The results of an investigation of MUC2 mRNA
expression in three different colon cancer cell lines are shown in
Fig. 5. The highest expression of
MUC2 was seen in the LoVo cell line. No expression was observed in
SW620 and colo205.
Effects of MUC2-SiRNA introduction on
heat and anticancer agent
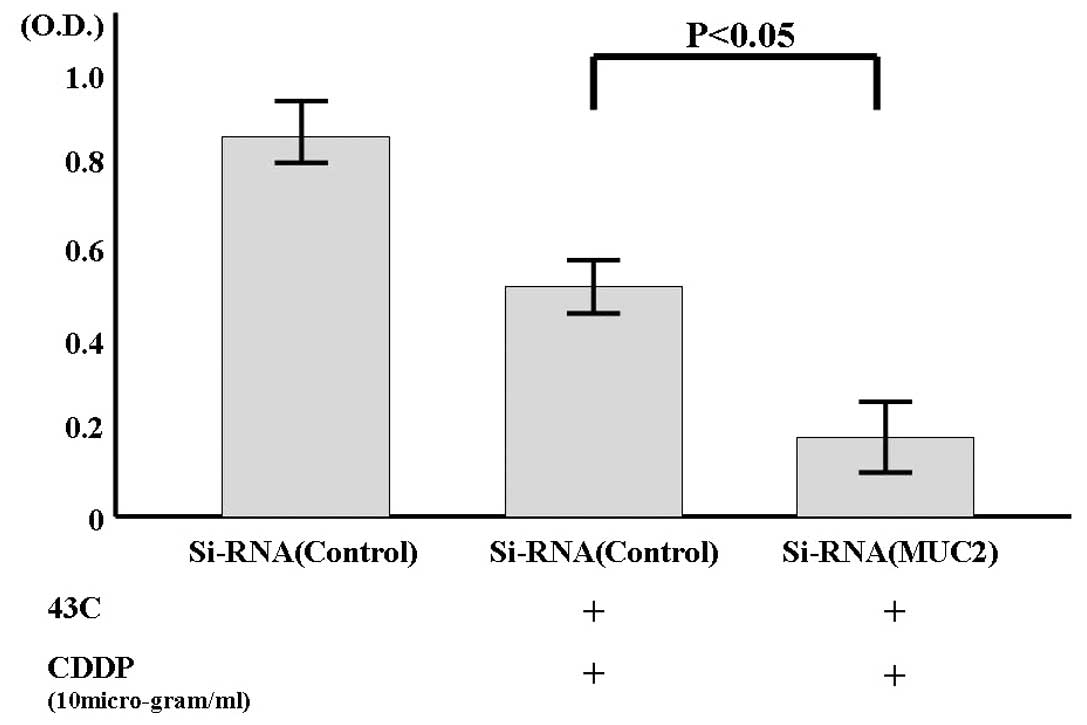
When SiRNA-MUC2 was introduced into the LoVo cell
line, which showed high MUC2 expression, the expression of MUC2
mRNA decreased as shown in Fig. 6.
The percentage of living cells was investigated after culturing
these cells for 1 day at 43°C in the presence of an anticancer
agent. When the percentage of living cells among the cells with
scrambled SiRNA (cultured at 37°C) was taken to be 100% (0.84O.D.),
the percentage in the cells with scrambled SiRNA was 63%
(0.53O.D.), and that in the cells with MUC2-SiRNA was 20%
(0.17O.D.).
Discussion
In recent years, with advances in anticancer agents
and molecularly targeted drugs for chemotherapy, improvements have
been seen in outcomes for unresectable colon cancer, particularly
hematogenous metastasis (2).
However, there are no reported large-scale trials showing clear
improvements in outcome for peritoneal dissemination, and there is
no established treatment. Cytoreduction and HIPEC are now conducted
in these patients, and reports of their efficacy are occasionally
seen (5,6). At our hospital, HIPEC has been
performed for colon cancer patients with peritoneal metastasis as
the only distant metastasis, and a significant effect has been seen
when compared with patients who did not receive HIPEC (5).
HIPEC also has mixed efficacy, being effective in
some cases and ineffective in others. If it were possible to judge
the cases in which it would be effective, it could reasonably be
considered to act in extending the lives of patients. Therefore,
the efficacy of HIPEC was investigated from a molecular biological
perspective, considering the importance of the properties of the
cancer cells themselves; that is, their gene/protein expression
state. We focused on mucin family proteins in healthy cell
membranes, which are thought to protect the cell from insults from
the outside. MUC1, 2, 3, 4 and 5A are thought to have a
particularly close relationship with colon tissue (12,22–24),
and from investigation of these proteins, it is thought that MUC2
expression is important in the effectiveness of HIPEC therapy.
MUC2 is a secretion type mucin, and in normal
mucosa, it is thought to cover the surface of soft mucosa and
provide a physically barrier, protecting the organism by constantly
washing off the mucosal surface (11). Thus, it may be that MUC2 proteins,
by being secreted on the surface of colon cancer cells, protect the
cancer cells at least partially from the effects of chemotherapy,
which is thought to be related to the effects of HIPEC in this
study.
We investigated the effects of HIPEC when RNAi was
used to block the activity of the MUC2 gene. When MUC2 gene
expression was inhibited, HIPEC was demonstrated experimentally to
have increased effectiveness, and the protection of MUC2 covering
the surface of cancer cells decreased. The degree to which cells
were affected by HIPEC was thought to have increased.
In the present study, the effectiveness of HIPEC was
seen to decrease when MUC2 protein expression was seen in colon
cancer cells and, conversely, to increase when MUC2 protein
expression was not seen. Thus, MUC2 expression may be useful as an
indicator in determining whether HIPEC is indicated.
References
|
1
|
Tominaga S and Oshima A: Cancer Mortality
and Morbidity statistics. Japan and the World-1999. Gann Monograph
on Cancer Research No. 47. Japan Scientific Societies Press; Tokyo,
Japan: 1999
|
|
2
|
Tournigand C, André T, Achille E, Lledo G,
Flesh M, Mery-Mignard D, Quinaux E, Couteau C, Buyse M, Ganem G,
Landi B, Colin P, Louvet C and de Gramont A: FOLFIRI followed by
FOLFOX6 or the reverse sequence in advanced colorectal cancer: a
randomized GERCOR study. J Clin Oncol. 22:229–237. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Jayne DG, Fook S, Loi C and Seow-Choen F:
Peritoneal carcinomatosis from colorectal cancer. Br J Surg.
89:1545–1550. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rampone B, Schiavone B, Martino A and
Confuorto G: Current role of hyperthermic intraperitoneal
chemotherapy in the treatment of peritoneal carcinomatosis from
colorectal cancer. World J Gastroenterol. 16:1299–1302. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Glehen O, Kwiatkowski F, Sugarbaker PH,
Elias D, Levine EA, De Simone M, Barone R, Yonemura Y, Cavaliere F,
Quenet F, Gutman M, Tentes AA, Lorimier G, Bernard JL, Bereder JM,
Porcheron J, Gomez-Portilla A, Shen P, Deraco M and Rat P:
Cytoreductive surgery combined with perioperative intraperitoneal
chemotherapy for the management of peritoneal carcinomatosis from
colorectal cancer: a multi-institutional study. J Clin Oncol.
22:3284–3292. 2004. View Article : Google Scholar
|
|
6
|
Shen P, Hawksworth J, Lovato J, Loggie BW,
Geisinger KR, Fleming RA and Levine EA: Cytoreductive surgery and
intraperitoneal hyperthermic chemotherapy with mitomycin C for
peritoneal carcinomatosis from nonappendiceal colorectal carcinoma.
Ann Surg Oncol. 11:178–186. 2004. View Article : Google Scholar
|
|
7
|
Sugarbaker PH: Intraperitoneal
chemotherapy and cytoreductive surgery for the prevention and
treatment of peritoneal carcinomatosis and sarcomatosis. Semin Surg
Oncol. 14:254–261. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chua TC, Morris DL and Esquivel J: Impact
of the peritoneal surface disease severity score on survival in
patients with colorectal cancer peritoneal carcinomatosis
undergoing complete cytoreduction and hyperthermic intraperitoneal
chemotherapy. Ann Surg Oncol. 17:1330–1336. 2010. View Article : Google Scholar
|
|
9
|
Moehle C, Ackermann N, Langmann T,
Aslanidis C, Kel A, Kel-Margoulis O, Schmitz-Madry A, Zahn A,
Stremmel W and Schmitz G: Aberrant intestinal expression and
allelic variants of mucin genes associated with inflammatory bowel
disease. J Mol Med. 84:1055–1066. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Itoh Y, Kamata-Sakurai M, Denda-Nagai K,
Nagai S, Tsuiji M, Ishii-Schrade K, Okada K, Goto A, Fukayama M and
Irimura T: Identification and expression of human
epiglycanin/MUC21: a novel transmembrane mucin. Glycobiology.
18:74–83. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Knowles MR and Boucher RC: Mucus clearance
as a primary innate defense mechanism for mammalian airways. J Clin
Invest. 109:571–577. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Takeyama K, Dabbagh K, Lee HM, Agusti C,
Lausier JA, Ueki IF, Grattan KM and Nadel JA: Epidermal growth
factor system regulates mucin production in airways. Proc Natl Acad
Sci USA. 96:3081–3086. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Van der Sluis M, De Koning BA, De Bruijn
AC, Velcich A, Meijerink JP, Van Goudoever JB, Büller HA, Dekker J,
Van Seuningen I, Renes IB and Einerhand AW: Muc2-deficient mice
spontaneously develop colitis, indicating that MUC2 is critical for
colonic protection. Gastroenterology. 131:117–129. 2006.PubMed/NCBI
|
|
14
|
Velcich A, Yang W, Heyer J, Fragale A,
Nicholas C, Viani S, Kucherlapati R, Lipkin M, Yang K and
Augenlicht L: Colorectal cancer in mice genetically deficient in
the mucin Muc2. Science. 295:1726–1729. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Allen A, Hutton DA and Pearson JP: The
MUC2 gene product: a human intestinal mucin. Int J Biochem Cell
Biol. 30:797–801. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Byrd JC and Bresalier RS: Mucins and mucin
binding proteins in colorectal cancer. Cancer Metastasis Rev.
23:77–99. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Japanese Society for Cancer of the Colon
and Rectum. Japanese Classification of Colorectal Carcinoma. 1st
English edition. Kanehara Publishers; Tokyo: 1997
|
|
18
|
Katayama K, Yamaguchi A, Murakami M,
Koneri K, Nagano H, Honda K, Hirono Y, Goi T, Iida A and Ito H:
Chemo-hyperthermic peritoneal perfusion (CHPP) for appendiceal
pseudomyxoma peritonei. Int J Clin Oncol. 14:120–124. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Dewey WC, Hopwood LE, Sapareto SA and
Gerweck LE: Cellular responses to combinations of hyperthermia and
radiation. Radiology. 123:463–474. 1977. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yamaguchi A, Urano T, Goi T, Saito M,
Takeuchi K, Hirose K, Nakagawara G, Shiku H and Furukawa K:
Expression of a CD44 variant containing exons 8 to 10 is a useful
independent factor for the prediction of prognosis in colorectal
cancer patients. J Clin Oncol. 14:1122–1127. 1996.PubMed/NCBI
|
|
21
|
Goi T, Yamaguchi A, Nakagawara G, Urano T,
Shiku H and Furukawa K: Reduced expression of deleted colorectal
carcinoma (DCC) protein in established colon cancers. Br J Cancer.
77:466–471. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gum JR Jr, Hicks JW, Toribara NW, Siddiki
B and Kim YS: Molecular cloning of human intestinal mucin (MUC2)
cDNA. Identification of the amino terminus and overall sequence
similarity to prepro-von Willebrand factor. J Bio Chem.
269:2440–2446. 1994.
|
|
23
|
Pratt WS, Crawley S, Hicks J, Ho J, Nash
M, Kim YS, Gum JR and Swallow DM: Multiple transcripts of MUC3:
evidence for two genes, MUC3A and MUC3B. Biochem Biophys Res
Commun. 275:916–923. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ho SB, Roberton AM, Shekels LL, Lyftogt
CT, Niehans GA and Toribata NW: Expression cloning of gastric mucin
complementary DNA and localization of mucin gene expression.
Gastroenterology. 109:735–747. 1995. View Article : Google Scholar : PubMed/NCBI
|