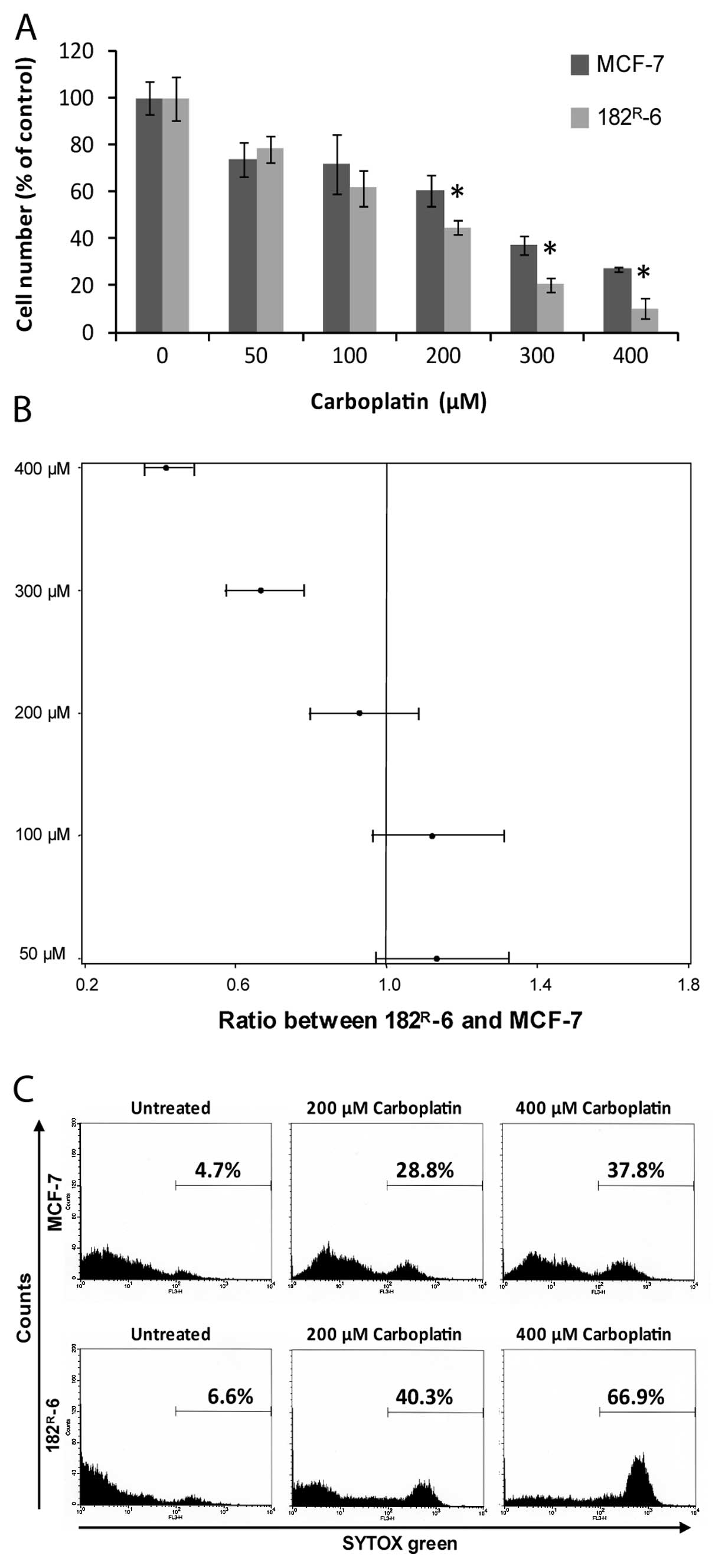
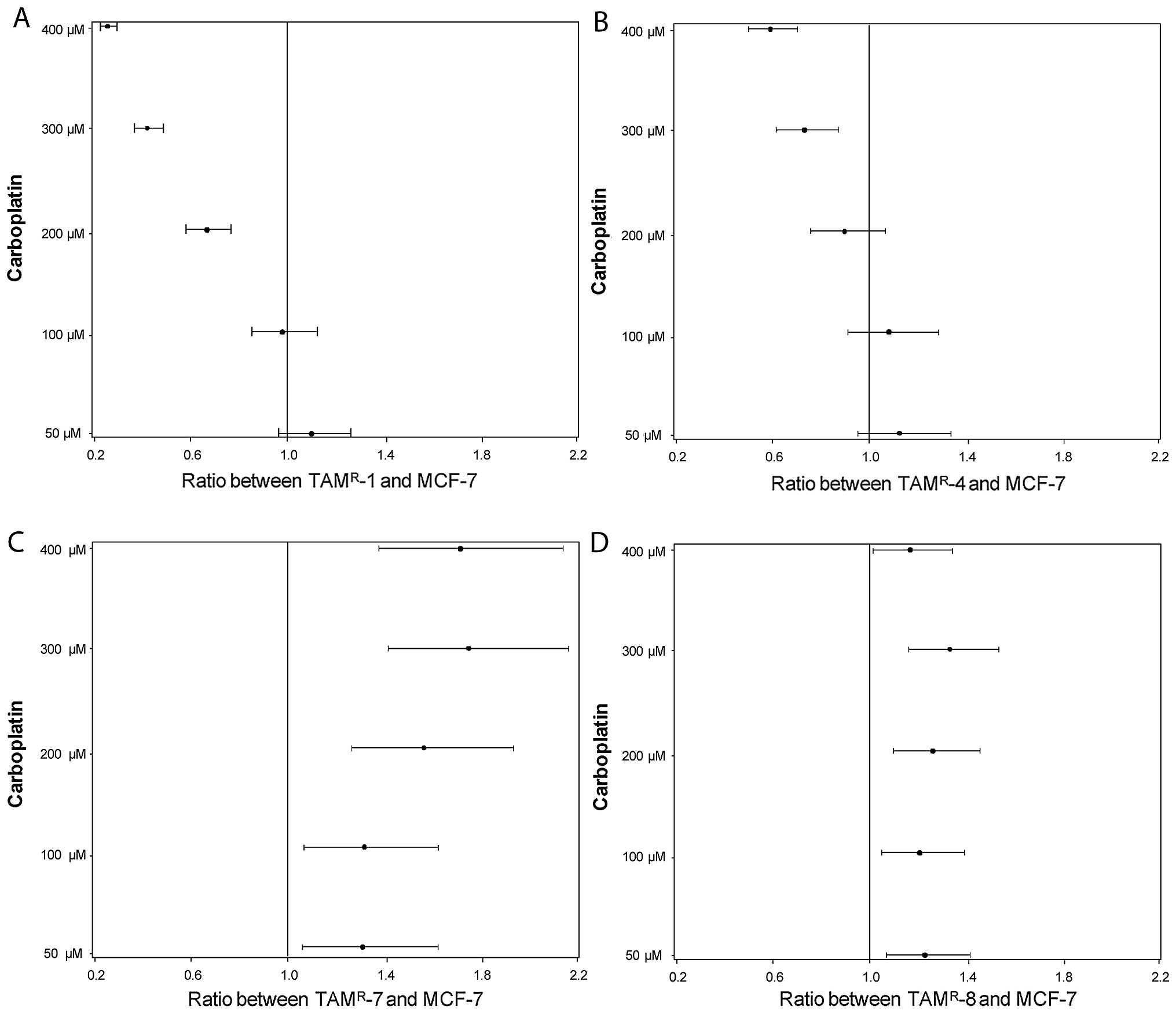
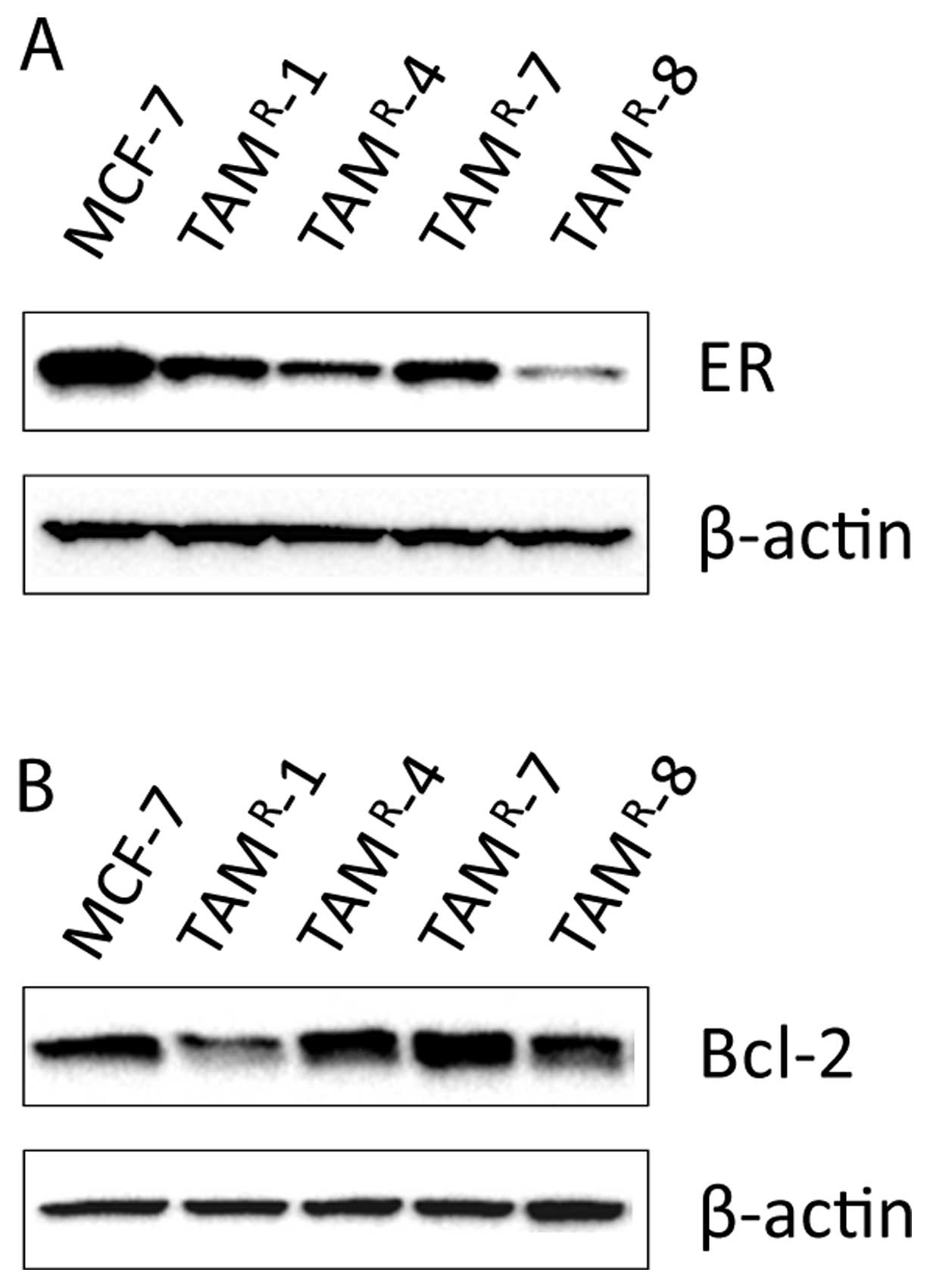
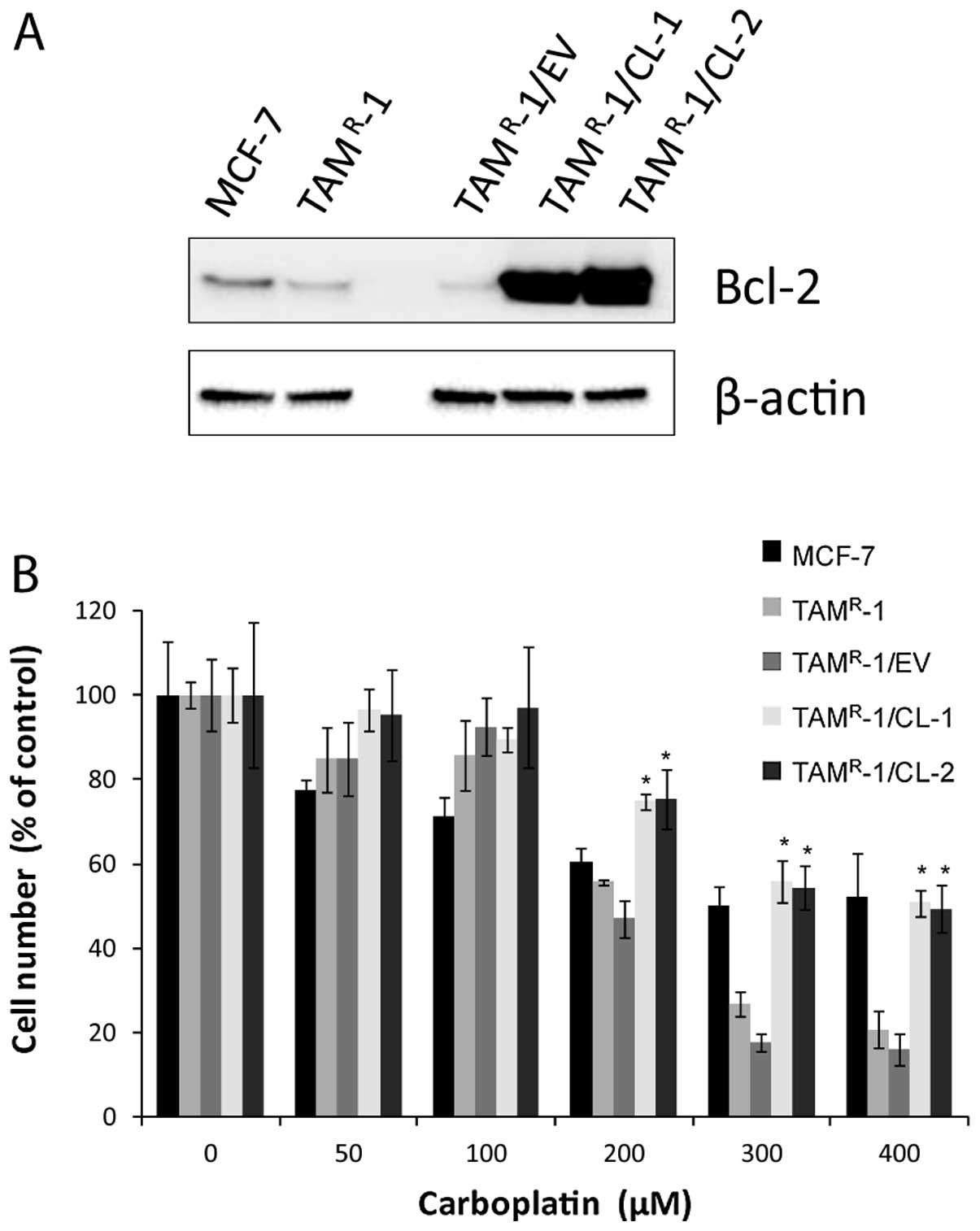
|
1.
|
Ali S and Coombes RC: Endocrine-responsive
breast cancer and strategies for combating resistance. Nat Rev
Cancer. 2:101–112. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
2.
|
Howell A, DeFriend DJ, Robertson JF,
Blamey RW, Anderson L, Anderson E, Sutcliffe FA and Walton P:
Pharmacokinetics, pharmacological and anti-tumour effects of the
specific anti-oestrogen ICI 182780 in women with advanced breast
cancer. Br J Cancer. 74:300–308. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Go RS and Adjei AA: Review of the
comparative pharmacology and clinical activity of cisplatin and
carboplatin. J Clin Oncol. 17:409–422. 1999.PubMed/NCBI
|
|
4.
|
Perez EA: Carboplatin in combination
therapy for metastatic breast cancer. Oncologist. 9:518–527. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Carrick S, Ghersi D, Wilcken N and Simes
J: Platinum containing regimens for metastatic breast cancer.
Cochrane Database Syst Rev. 3:CD0033742004.
|
|
6.
|
Martin M: Platinum compounds in the
treatment of advanced breast cancer. Clin Breast Cancer. 2:190–208.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Decatris MP, Sundar S and O’Byrne KJ:
Platinum-based chemotherapy in metastatic breast cancer: current
status. Cancer Treat Rev. 30:53–81. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Colbern GT, Hiller AJ, Musterer RS,
Working PK and Henderson IC: Antitumor activity of Herceptin in
combination with STEALTH liposomal cisplatin or nonliposomal
cisplatin in a HER2 positive human breast cancer model. J Inorg
Biochem. 77:117–120. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Pegram M, Hsu S, Lewis G, Pietras R, Beryt
M, Sliwkowski M, Coombs D, Baly D, Kabbinavar F and Slamon D:
Inhibitory effects of combinations of HER-2/neu antibody and
chemo-therapeutic agents used for treatment of human breast
cancers. Oncogene. 18:2241–2251. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
10.
|
Pegram MD, Lipton A, Hayes DF, Weber BL,
Baselga JM, Tripathy D, Baly D, Baughman SA, Twaddell T, Glaspy JA
and Slamon DJ: Phase II study of receptor-enhanced chemosensitivity
using recombinant humanized anti-p185HER2/neu monoclonal antibody
plus cisplatin in patients with HER2/neu-overex-pressing metastatic
breast cancer refractory to chemotherapy treatment. J Clin Oncol.
16:2659–2671. 1998.
|
|
11.
|
Slamon D, Eiermann W, Robert N, Pienkowski
T, Martin M, Press M, Mackey J, Glaspy J, Chan A, Pawlicki M,
Pinter T, Valero V, Liu MC, Sauter G, von Minckwitz G, Visco F, Bee
V, Buyse M, Bendahmane B, Tabah-Fisch I, Lindsay MA, Riva A and
Crown J; Breast Cancer International Research Group: Adjuvant
trastuzumab in HER2-positive breast cancer. N Engl J Med.
365:1273–1283. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Yde CW, Gyrd-Hansen M, Lykkesfeldt AE,
Issinger OG and Stenvang J: Breast cancer cells with acquired
antiestrogen resistance are sensitized to cisplatin-induced cell
death. Mol Cancer Ther. 6:1869–1876. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Siddik ZH: Cisplatin: mode of cytotoxic
action and molecular basis of resistance. Oncogene. 22:7265–7279.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Schorr K, Li M, Krajewski S, Reed JC and
Furth PA: Bcl-2 gene family and related proteins in mammary gland
involution and breast cancer. J Mammary Gland Biol Neoplasia.
4:153–164. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Yde CW and Issinger OG: Enhancing
cisplatin sensitivity in MCF-7 human breast cancer cells by
down-regulation of Bcl-2 and cyclin D1. Int J Oncol. 29:1397–1404.
2006.PubMed/NCBI
|
|
16.
|
Christensen GL, Jepsen JS, Fog CK,
Christensen IJ and Lykkesfeldt AE: Sequential versus combined
treatment of human breast cancer cells with antiestrogens and the
vitamin D analogue EB1089 and evaluation of predictive markers for
vitamin D treatment. Breast Cancer Res Treat. 85:53–63. 2004.
View Article : Google Scholar
|
|
17.
|
Larsen SS, Heiberg I and Lykkesfeldt AE:
Anti-oestrogen resistant human breast cancer cell lines are more
sensitive towards treatment with the vitamin D analogue EB1089 than
parent MCF-7 cells. Br J Cancer. 84:686–690. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Lykkesfeldt AE, Madsen MW and Briand P:
Altered expression of estrogen-regulated genes in a
tamoxifen-resistant and ICI 164,384 and ICI 182,780 sensitive human
breast cancer cell line, MCF-7/TAMR-1. Cancer Res. 54:1587–1595.
1994.PubMed/NCBI
|
|
19.
|
Lykkesfeldt AE, Larsen SS and Briand P:
Human breast cancer cell lines resistant to pure anti-estrogens are
sensitive to tamoxifen treatment. Int J Cancer. 61:529–534. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Madsen MW, Reiter BE, Larsen SS, Briand P
and Lykkesfeldt AE: Estrogen receptor messenger RNA splice variants
are not involved in antiestrogen resistance in sublines of MCF-7
human breast cancer cells. Cancer Res. 57:585–589. 1997.PubMed/NCBI
|
|
21.
|
Sekine I, Shimizu C, Nishio K, Saijo N and
Tamura T: A literature review of molecular markers predictive of
clinical response to cytotoxic chemotherapy in patients with breast
cancer. Int J Clin Oncol. 14:112–119. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22.
|
Lykkesfeldt AE and Sorensen EK: Effect of
estrogen and antiestrogens on cell proliferation and synthesis of
secreted proteins in the human breast cancer cell line MCF-7 and a
tamoxifen resistant variant subline, AL-1. Acta Oncol. 31:131–138.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Lykkesfeldt AE and Briand P: Indirect
mechanism of oestradiol stimulation of cell proliferation of human
breast cancer cell lines. Br J Cancer. 53:29–35. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
24.
|
Lundholt BK, Briand P and Lykkesfeldt AE:
Growth inhibition and growth stimulation by estradiol of estrogen
receptor transfected human breast epithelial cell lines involve
different pathways. Breast Cancer Res Treat. 67:199–214. 2001.
View Article : Google Scholar
|
|
25.
|
Frogne T, Benjaminsen RV, Sonne-Hansen K,
Sorensen BS, Nexo E, Laenkholm AV, Rasmussen LM, Riese DJ II, de
Cremoux P, Stenvang P and Lykkesfeldt AE: Activation of ErbB3, EGFR
and Erk is essential for growth of human breast cancer cell lines
with acquired resistance to fulvestrant. Breast Cancer Res Treat.
114:263–275. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26.
|
Krajewski S, Blomqvist C, Franssila K,
Krajewska M, Wasenius VM, Niskanen E, Nordling S and Reed JC:
Reduced expression of proapoptotic gene BAX is associated with poor
response rates to combination chemotherapy and shorter survival in
women with metastatic breast adenocarcinoma. Cancer Res.
55:4471–4478. 1995.
|
|
27.
|
Bottini A, Berruti A, Bersiga A, Brizzi
MP, Brunelli A, Gorzegno G, DiMarco B, Aguggini S, Bolsi G, Cirillo
F, Filippini L, Betri E, Bertoli G, Alquati P and Dogliotti L: p53
but not bcl-2 immunostaining is predictive of poor clinical
complete response to primary chemotherapy in breast cancer
patients. Clin Cancer Res. 6:2751–2758. 2000.PubMed/NCBI
|
|
28.
|
Miyashita T, Krajewski S, Krajewska M,
Wang HG, Lin HK, Liebermann DA, Hoffman B and Reed JC: Tumor
suppressor p53 is a regulator of bcl-2 and bax gene expression in
vitro and in vivo. Oncogene. 9:1799–1805. 1994.PubMed/NCBI
|
|
29.
|
Silvestrini R, Veneroni S, Daidone MG,
Benini E, Boracchi P, Mezzetti M, Di Fronzo G, Rilke F and Veronesi
U: The Bcl-2 protein: a prognostic indicator strongly related to
p53 protein in lymph node-negative breast cancer patients. J Natl
Cancer Inst. 86:499–504. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Benbrook DM and Masamha CP: The
pro-survival function of Akt kinase can be overridden or altered to
contribute to induction of apoptosis. Curr Cancer Drug Targets.
11:586–599. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31.
|
Frogne T, Jepsen JS, Larsen SS, Fog CK,
Brockdorff BL and Lykkesfeldt AE: Antiestrogen-resistant human
breast cancer cells require activated protein kinase B/Akt for
growth. Endocr Relat Cancer. 12:599–614. 2005. View Article : Google Scholar : PubMed/NCBI
|