Introduction
Lung cancer is one of the most common tumors, every
year over one million people die from this cancer (1). Currently, the management of non-small
cell lung cancer is ineffective and insufficient, which is why it
is necessary to search for new drugs that will replace or assist
those currently used (2).
Ciprofloxacin is an antibiotic that belongs to the
fluoroquinolone class (3). This
group of antibiotics has a broad spectrum (4,5).
Ciprofloxacin also inhibits topoisomerase II in eukaryotes,
including mammalian cells (6,7).
Previously we showed that ciprofloxacin can act
against lung cancer cells using trypan blue and MTT assays
(8). These methods give little
information on this drug and its influence on lung cancer cells
in vitro(8–11). In the current study, we aimed to
present cell viability in a long-term continuous culture system
with analysis of the subcellular architecture and the cell cycle.
To show the possible difference the highly resistant lines B16 and
C6 were chosen for the experiments.
Materials and methods
Cell lines
A549 (human non-small cell lung cancer) and C6 (rat
glioblastoma) cell lines were purchased by the American Type
Culture Collection (Manassas, VA). The B16 (mouse melanoma) cell
line was established from B16 tumors excised from C57BL/6J mouse.
A549, B16 and C6 cell lines were cultured in DMEM/HAM’S F-12 medium
containing 10% of fetal bovine serum (FBS), supplemented with 5
μg/ml amphotericin B, 100 μg/ml streptomycin and 100
U/ml penicillin. All cell lines were grown in 25-cm2
Nunc T-flasks at 36°C and 5% CO2.
Lethal Concentration calculation
Lethal concentration (LC) values were calculated
using trypan blue assay. The results are presented in Table I(8).
 | Table ILC values for cell lines: A549, B16
and C6. |
Table I
LC values for cell lines: A549, B16
and C6.
| | LC values
(μg/ml)
|
|---|
| Cell line | Time of incubation
with ciprofloxacin (h) | LC10 | LC50 | LC90 |
|---|
| A549 | 24 | 27.7 | 133.3 | 593.2 |
| 48 | 18.2 | 102.1 | 389.5 |
| B16 | 24 | 23.8 | 409.5 | NA |
| 48 | 12.0 | 81.0 | NA |
| C6 | 24 | NA | 5915.0 | NA |
| 48 | 572.0 | 1092.0 | NA |
Proliferation assay
The influence of ciprofloxacin on different cancer
cell lines was established using the Real-Time Cell Analyzer
(RTCA)-DP that belongs to the xCELLigence system (Roche Applied
Science). This enables monitoring cellular events in real time
without the incorporation of levels. Cells (n=6250) were seeded on
each well of E-Plate 16 (Roche Applied Science). Cells were
incubated in normal cell culture medium for 24 h. In the next step
ciprofloxacin in concentrations corresponding to LC values
(Table I) was added. LC values for
ciprofloxacin were previously established (8). Cells were incubated with
ciprofloxacin for 24 and 48 h. The viability of cells has been
expressed by means of cell index (CI). Cell index values are the
results of the increase in electrode impedance on E-Plate by the
increase in cell number attached to the electrodes. Cell index
values can be used to monitor the viability, number, morphology and
adhesion degree in a number of cell-based assays. A medium without
a cell culture served as the background. The results are presented
in two ways: as an effect of cell index and after normalization of
these results. Normalization allows reference of the obtained
results of ciprofloxacin cytotoxicity to the control and the
background. For each well, the normalized cell index is calculated
as the cell index at a given point divided by the cell index at the
normalization time point. Thus, the normalized cell index for all
wells must equal 1 at the normalization time point (for example
when a tested compound was added). Due to normalization we can
determine the mode of action of the tested drug, as cytotoxic or
cytostatic. The experiments were carried out in triplicate. Figure
curves are presented as mean values of three experiments (Figs. 1 and 2).
Fluorescent staining of F-actin
Cells were seeded on cover slips in 24-well plates
(density, 1×104 cells/well). After 24 h of incubation
cells were treated with ciprofloxacin for 48 h in doses according
to LC values (Table I). Then cells
were washed in PBS and fixed in paraformaldehyde (15 min, 37°C).
After washing in PBS (3×5 min), cells were incubated with 0.1
Triton X-100 (Serva Electrophoresis, Heidelberg, Germany) in HBSS
(5 min), washed in PBS (3×5 min) and labeled with phalloidine
conjugated with Alexa 488 [dilution 1:40, 20 min, room temperature
(RT), darkroom] (Invitrogen Life Technologies, Carlsbad, CA). Cover
slips were washed 3×5 min in PBS before counterstaining for DNA
with DAPI (100 ng/ml, 10 min, RT, darkroom) (Sigma). Finally cells
were washed in distilled water before mounting in Aqua-Poly/Mount
(Polysciences, Inc.) Stained cells were analyzed using NIS-Elements
4.0 software in Eclipse E800 fluorescence microscope (Nikon).
Cell cycle analysis
Cells were seeded on 24-well plates at a density of
5×104 cells/well and exposed to ciprofloxacin at
concentrations corresponding to LC values (Table I). After 24 h of incubation cells
were washed twice with PBS solution, detached from wells with
trypsin and then centrifuged for 5 min at 300 x g at 4°C. In the
next stage 500 μl of hypotonic, DNA coloring solution of
PI/Triton X-100 were added. The solution consisted of 50
μg/ml PI, 0.1 mg/ml RNAase and 0.05% Triton X-100. Cells
were incubated for 30 min in the dark at room temperature. Then, in
order to stop the reaction, the tubes with reaction mixture were
placed on ice and then transferred to a flow cytometer equipped
with System IITM Software, Version 1.0. (Coulter Electronics,
Krefeld, Germany). The results obtained after 24 h in LC50 for C6
cells were not analyzed in cytometer because of high ciprofloxacin
concentration and small number of cells obtained after incubation.
The experiments were carried out in triplicate.
Results
Proliferation assay
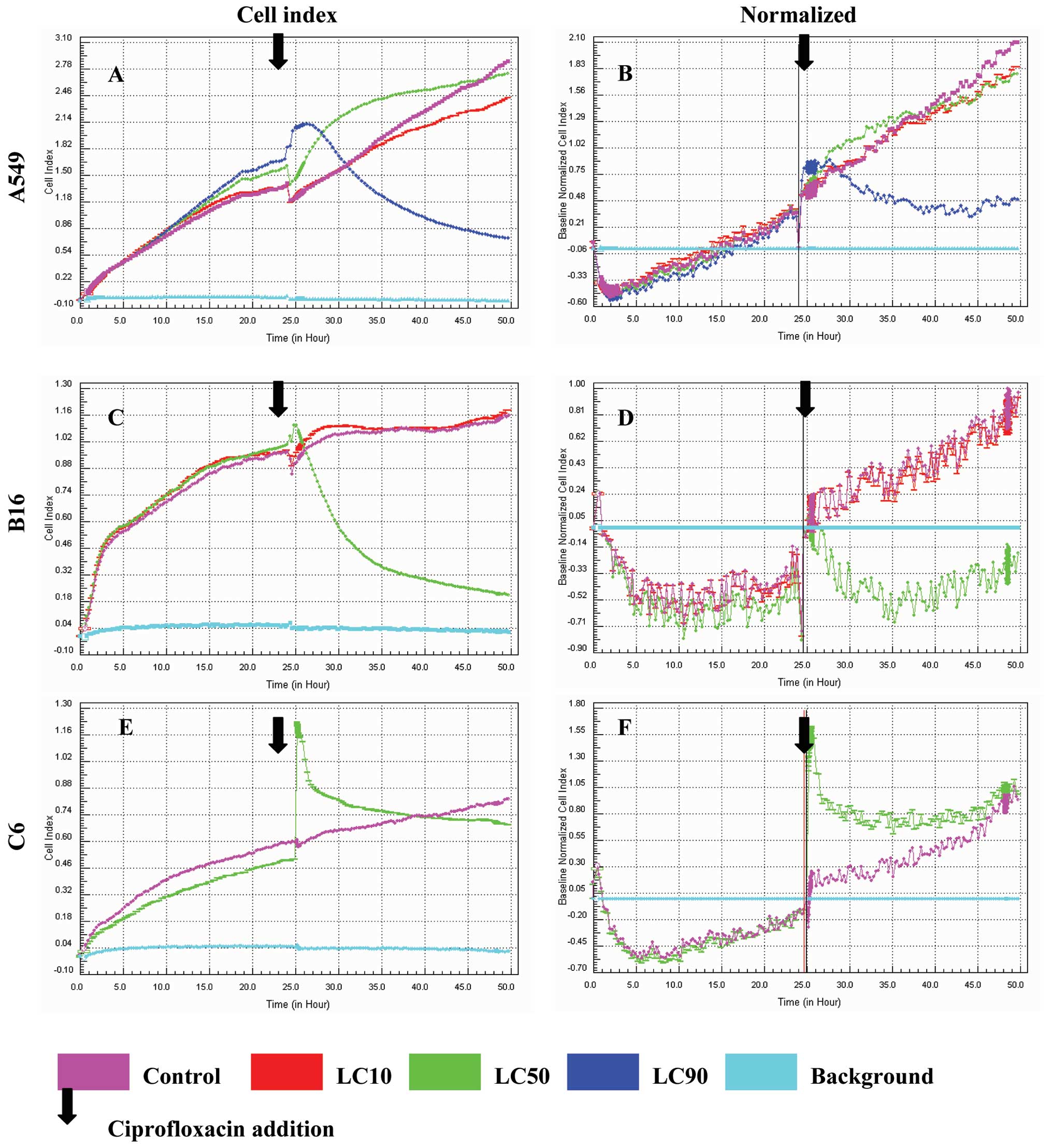
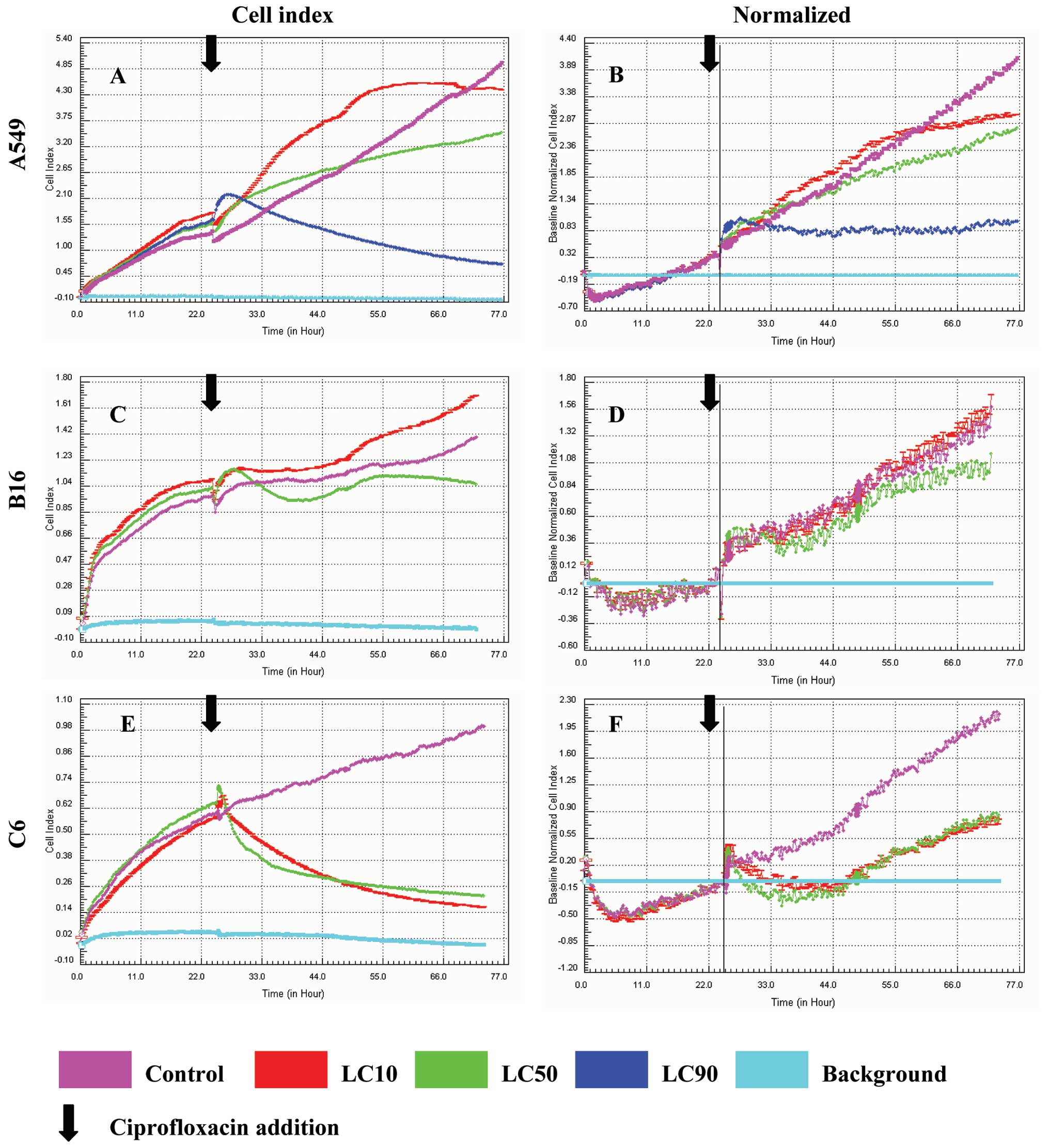
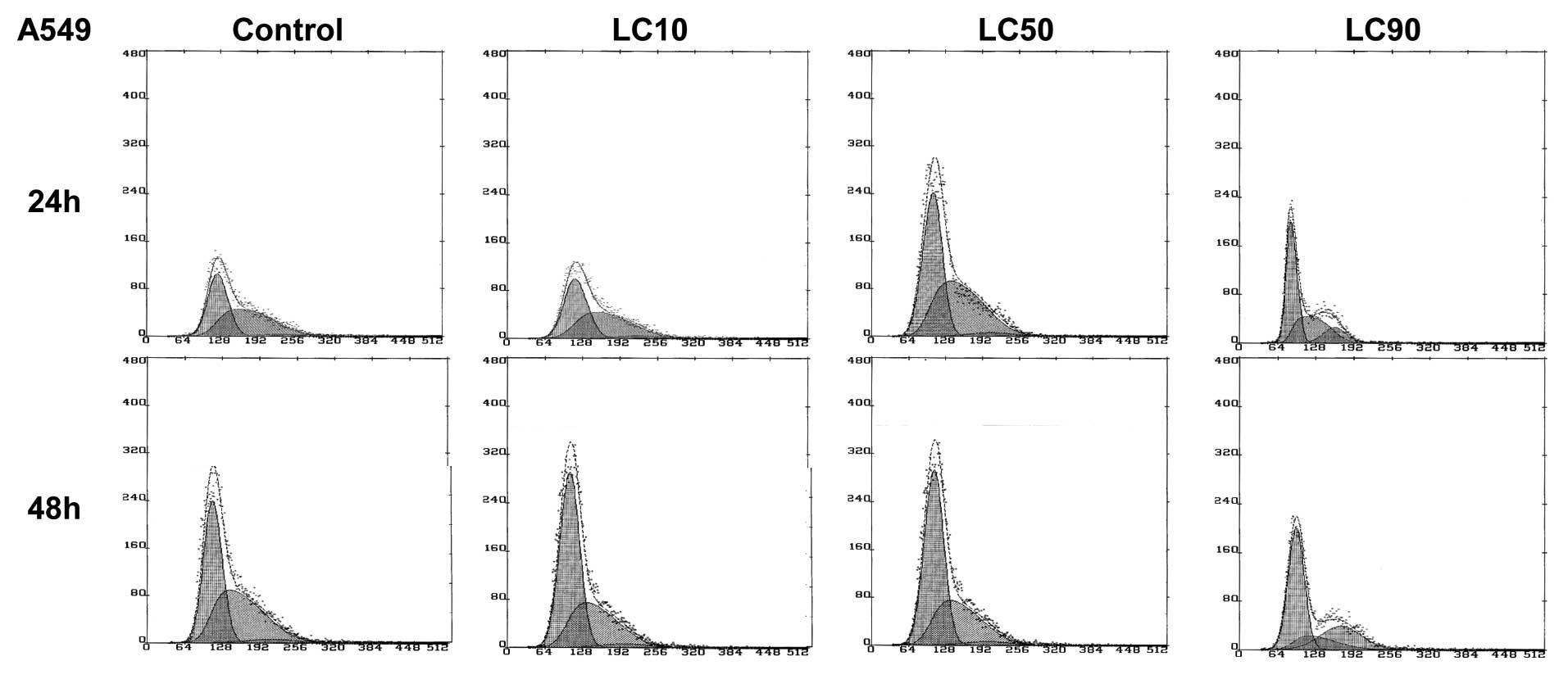
Proliferation assays are presented in Figs. 1 and 2, after 24 and 48 h respectively.
Ciprofloxacin was most effective against the A549 lung cancer cell
line. We observed a decrease in viability at concentrations
corresponding to LC10, LC50 and LC90. The graphs obtained after
cell index normalization showed that ciprofloxacin acted as a
cytostatic but not cytotoxic compound in the case of A549 cells
after 24 and 48 h in LC90 (Figs.
1B and 2B). In the
concentration corresponding to LC10 and LC50 a cytostatic effect on
A549 cells was not observed (Figs.
1B and 2B).
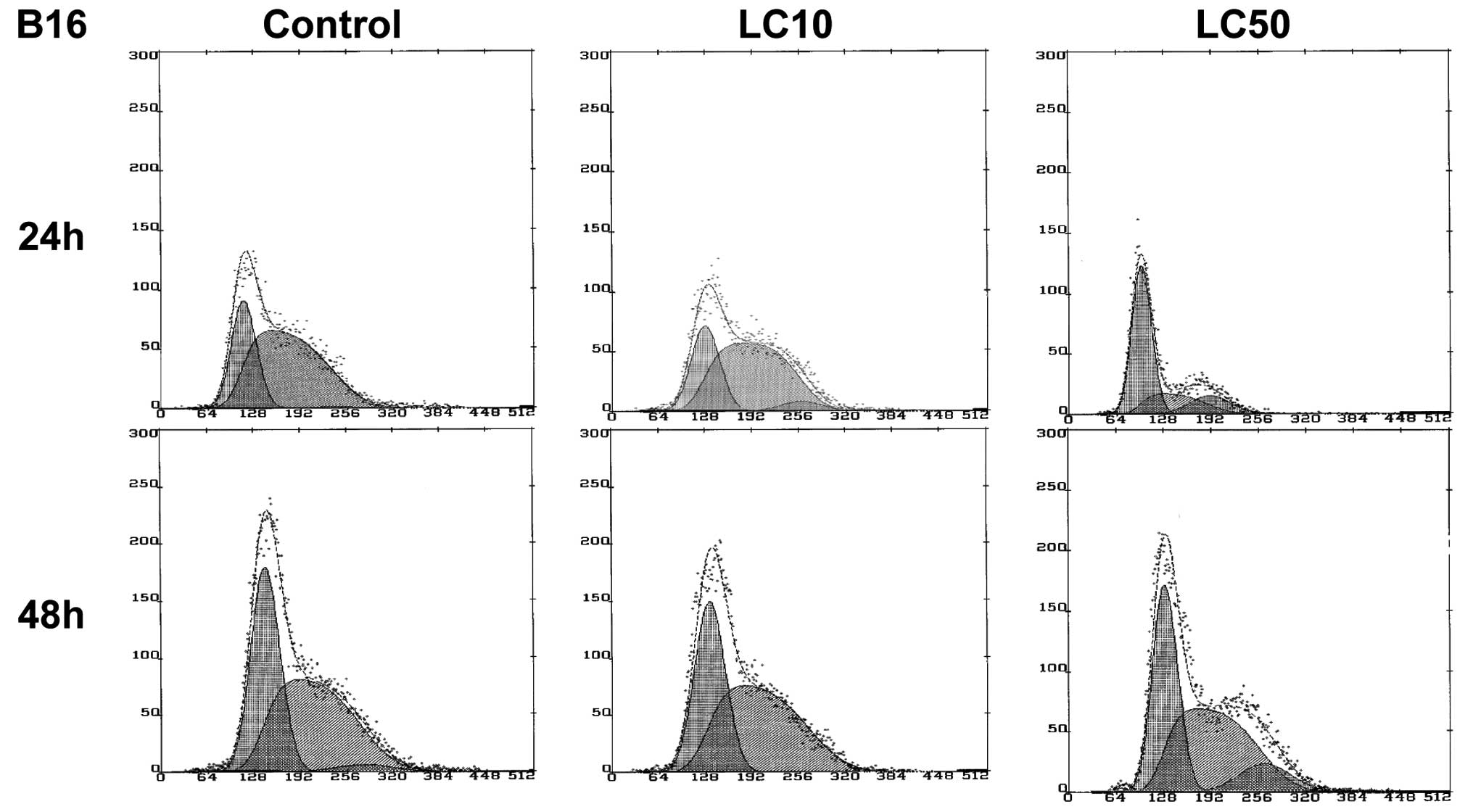
No significant decrease of the viability of B16
cells was observed except for LC50 after 24 h, but after
normalization a cytostatic effect was not achieved (Fig. 1C). A cytostatic effect was not
achieved either after 24 and 48 h for all concentrations in the
case of the C6 glioblastoma cell line after result normalization
(Figs. 1F and 2F).
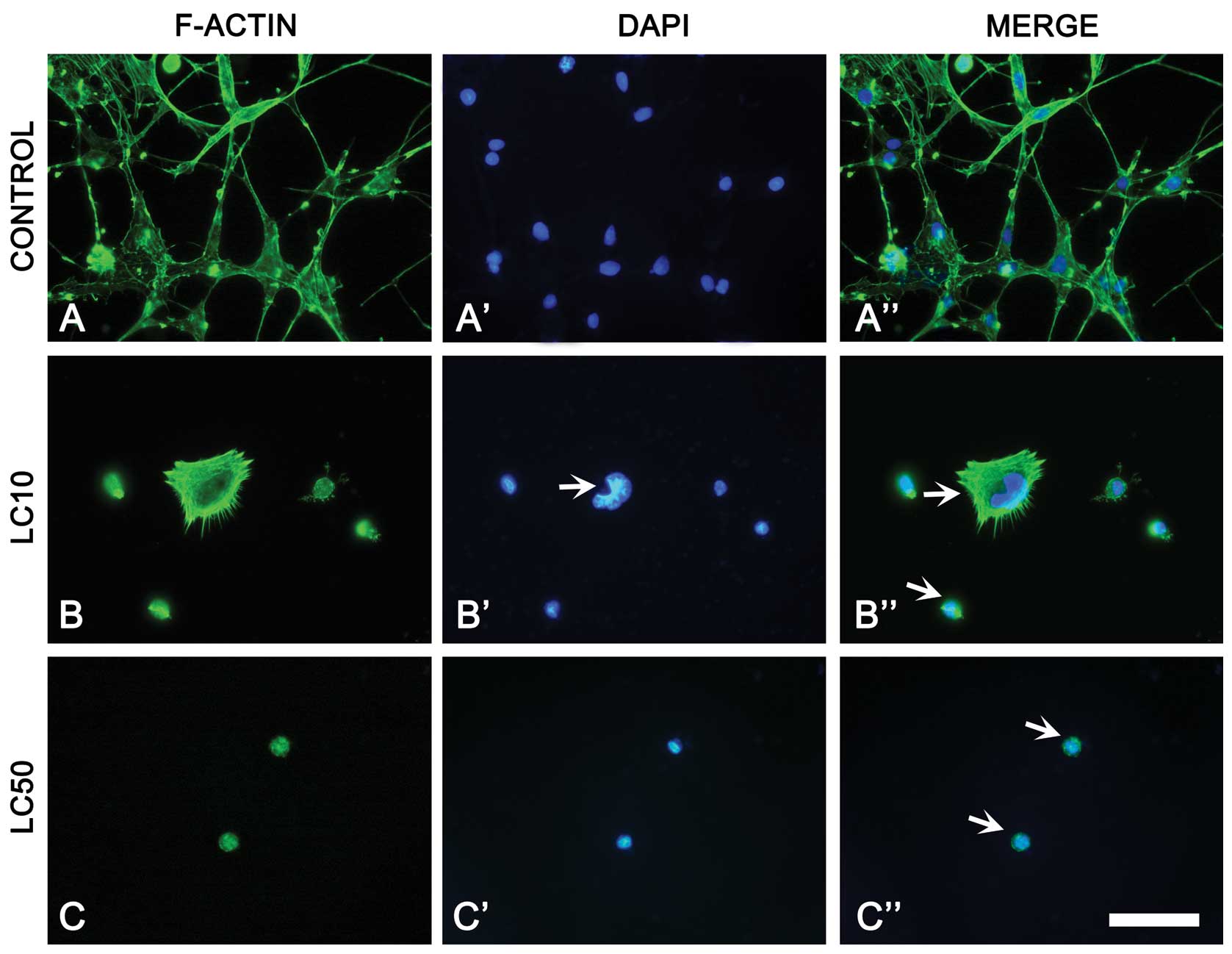
Fluorescent staining of F-actin
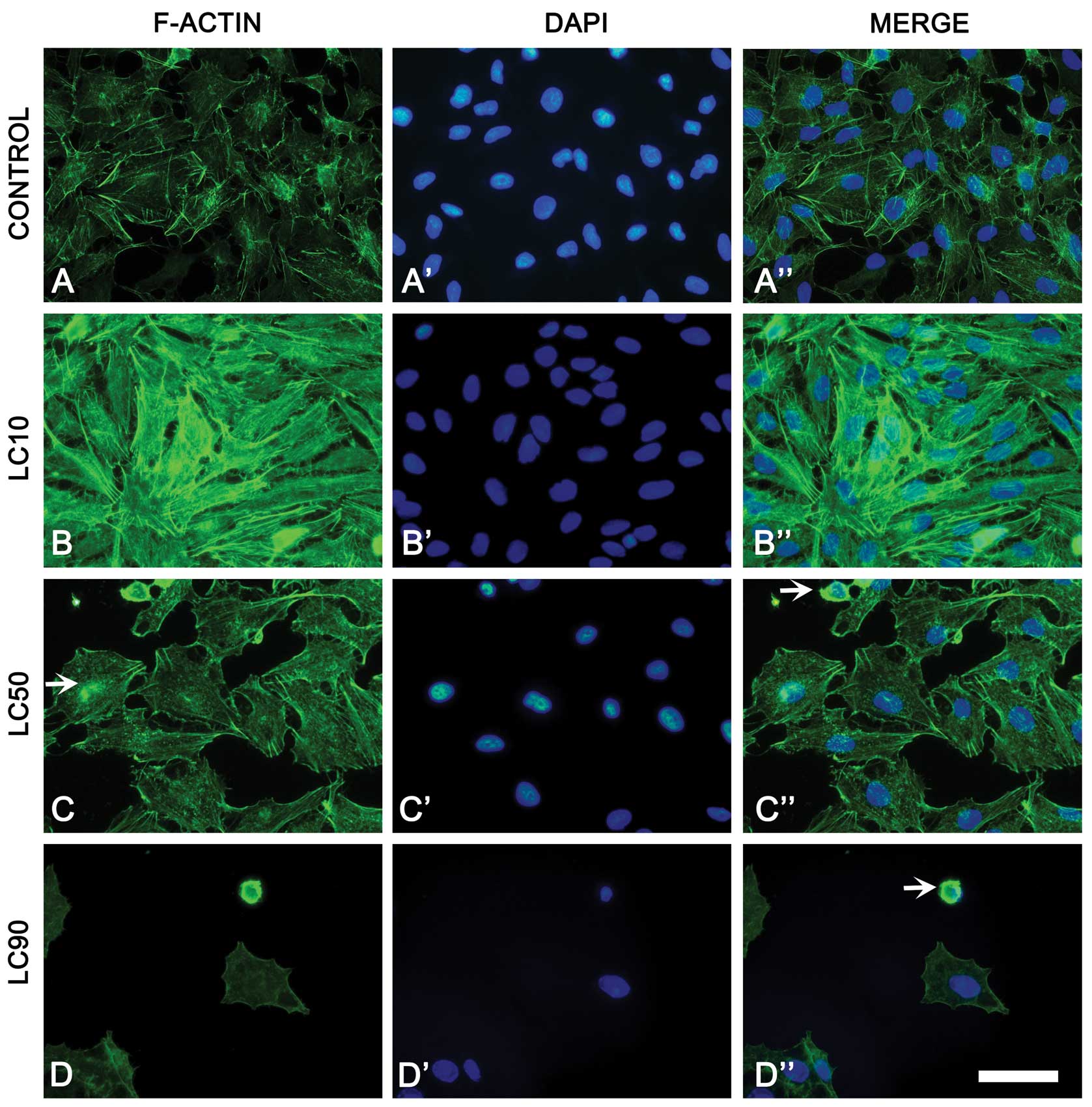
Changes in cytoskeletal organization were observed
in non-small cell lung cancer cells treated with ciprofloxacin.
LC10 caused increase of F-actin fluorescence. However,
ciprofloxacin treatment (LC50, LC90) induced changes of F-actin
distribution (decreased number of actin fibers), leading to cell
shrinkage. In LC50, actin aggregates were observed in the
cytoplasm. Furthermore, rounded cells with condensed actin in LC50
and LC90 were present. In the highest dose of ciprofloxacin no
actin fibers were observed in A549 cells (Fig. 3). A few apoptotic cells with cell
blebbing were also observed in highest concentration of
ciprofoxacin (data not shown).
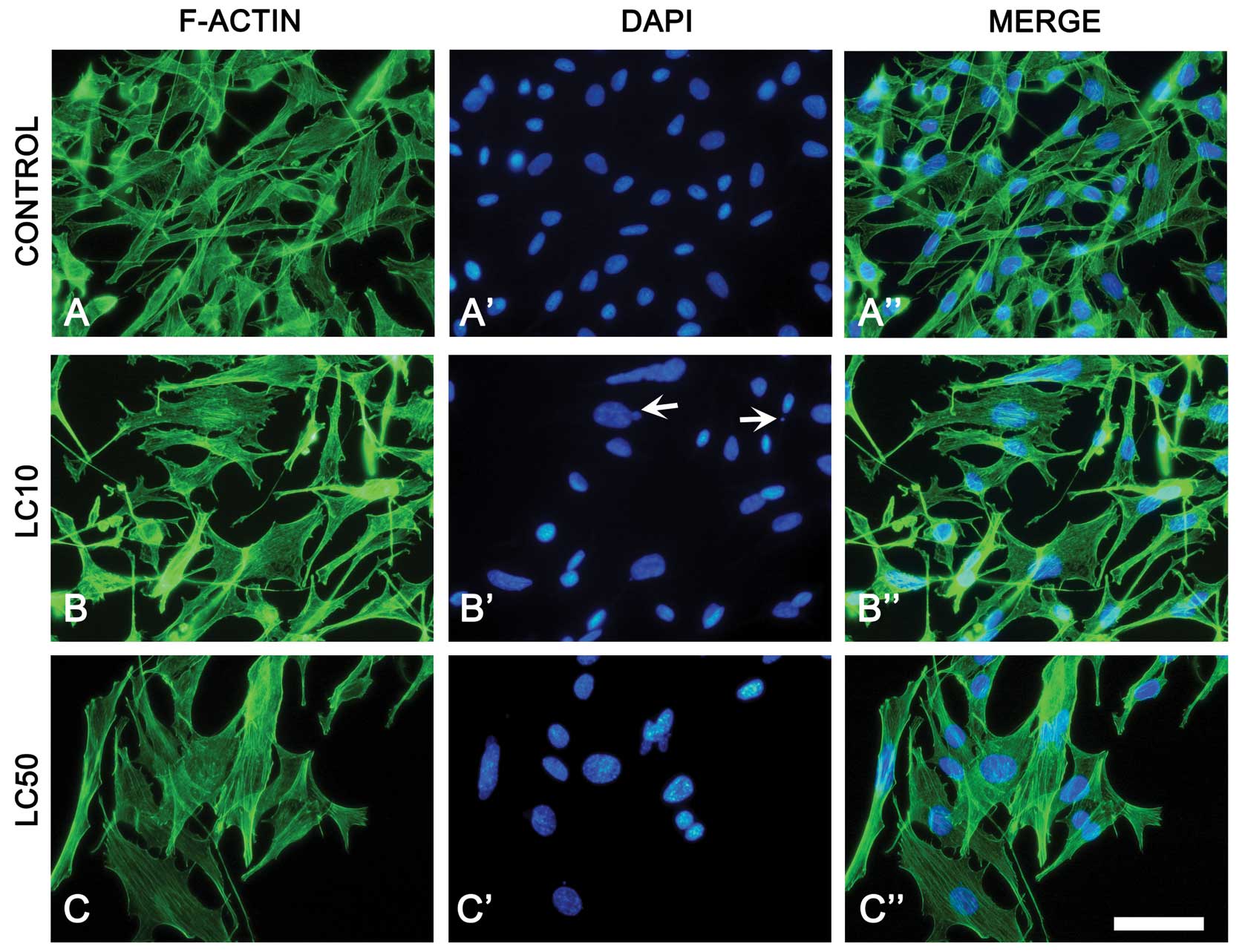
The B16 cell line treated with ciprofoxacin showed
more extensive cytoskeletal organization that resulted in cell
enlargement especially in LC50. DAPI staining revealed alternation
in size (increase) and shape of the nucleus after incubation with
the drug, but apoptotic bleb formation was also observed (Fig. 4).
Fluorescent microscopy showed elongated shape and
some intercellular junction in the C6 cell line. Actin fibers were
visible. Ciprofloxacin treatment caused loss of intracellular
connection and cell shrinkage (LC10, LC50). Shrunken and rounded
cells with condensed actin were observed. However larger cells with
irregular nuclei shape were also observed (Fig. 5).
Cell cycle analysis
The A549 cell cycle control study showed that after
a short incubation time (24 h), a large number of cells was in the
S phase, even up to half of the population. This reflected a high
prolifaration rate at the beginning. This number decreased with the
incubation time, which was associated with the proliferation rate
inhibition after 48 h as a result of contact inhibition. This
effect was also observed for LC10 and LC50. In the highest
concentration (LC90) G2/M accumulation was observed, which
indicated the arrest of the cell cycle in this phase (Fig. 6).
In case of the B16 cell line, the LC90 value could
not be established. In the control group, after a short incubation
time (24 h) melanoma cells proliferated intensively (67% of cells
in S phase), after a longer incubation time (48 h) up to 52% of
cells were in the S phase. Similar results were observed in LC10,
indicating that this concentration does not affect B16 cells. In
LC50, a higher amount of cells in the G2/M phase was observed
indicating G2/M block after 48 h (Fig.
7).
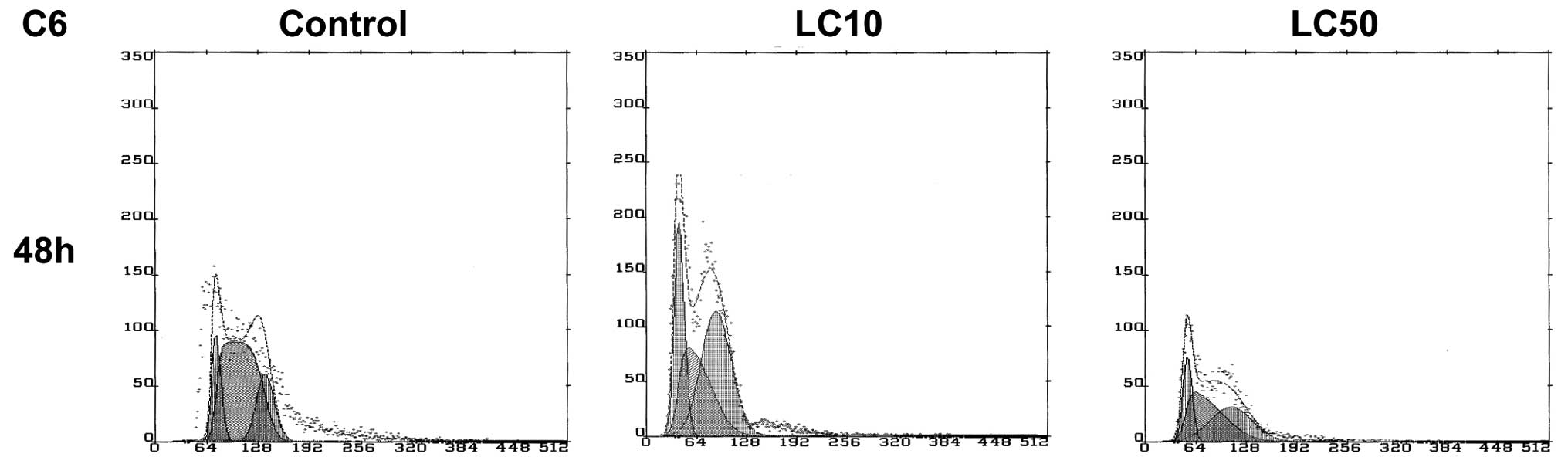
The results of the cell cycle analysis for C6
glioblastoma cells were significantly different from two previously
described cell lines. In control, cells still proliferated
intensively after 48 h. With the increasing ciprofloxacin
concentration we observed a decrease in the number of cells in the
S phase, (from 57-49% in LC50) with predominance of G1/G0 and G2/M
phases. The cell cycle was not established for 24 h due to
impossible to established values of LC10 and LC90 (Table I) (8). The LC50 concentration led to
intensive proliferation and unpredictable behavior of the cell
cycle of C6 cells incubated with ciprofloxacin (Figs. 1E and 8).
Discussion
xCELLigence system is a relatively new method. This
system measured cell viability in real time. Apoptosis occurs only
during a short period of time, often within hours, that is why
continuous viability monitoring is a useful tool that can show how
the examined drug acts against cells. This system is able to
distinguish the cytotoxic effect from the inhibition of
proliferation. The continuous monitoring of cell viability by the
xCELLigence system makes it possible to distinguish different
perturbations of cell viability, such as senescence, cell toxicity
(cell death) and reduced proliferation (cell cycle arrest)
(12). Thus, this system can help
to predict biological in vivo effect exerted by the tested
drug.
Ciprofloxacin exerted cytostatic effect against A549
cells (lung cancer), but not against B16 (melanoma) and C6 cells
(glioblastoma). The most effective action of ciprofloxacin was
directed against lung cancer cells. Only in the case of the A549
cell line, all LC values (10, 50, 90) could be calculated and
examined based on our previous work (8). The cell index based on the experiment
performed on the xCELLigence system indicates better toxic
effectiveness in relation to A549 cells than B16 and C6 cells. A
significant decrease in the viability in concentration
corresponding to LC50 after a 48 h incubation was observed for A549
cells (Fig. 2A). Results obtained
after F-actin staining confirm the results obtained by the
xCELLigence system. We observed a decrease in the cell index
(xCELLigence) (Fig. 2A) and the
cell number (fluorescence microscopy) (Fig. 3D) with increasing concentration of
drug. DAPI staining showed that one of the possible A549 cell death
mechanisms is apoptosis, single cells with apoptotic blebs were
visible in LC90. Most cells had very condensed F-actin and
chromatin, cells without stress fibers were also observed (Fig. 3). The results obtained from cell
cycle analysis showed that ciprofloxacin influenced the inhibition
of proliferation and the accumulation of cells in G2/M phase
(Fig. 6). G2/M accumulation can
potentially lead to the induction of cell death after ciprofloxacin
treatment, but it has to be proven. The supposed mechanism of
action leads to G2/M accumulation is a topoisomerase II inhibition
(13,14). This means that ciprofloxacin can be
a potential topoisomerase II inhibitor for A549 cells.
In the case of the B16 cell line, LC90 could not be
established. Lack of cytostatic effect on B16 cells induced by
ciprofloxacin was recorded in LC50. In LC10, the viability of cells
was observed to be even higher than in control (Table I, Figs. 1C and 2C). This was probably due to the
predominant activation of prosurvival pathways over the apoptotic
pathways in lower concentrations of ciprofloxacin (15). This phenomenon was described as
hormesis (16,17). This phenomenon was accompanied by
changes in F-actin distribution. Results obtained in fluorescence
microscopy indicate that B16 cells reduced in number and increased
in size with increasing ciprofloxacin concentration (Fig. 4). This may explain the lack of
cytostatic effect after a 48 h incubation, because this system
measures the level of well surface coverage and not the cell
number. DAPI staining showed nuclei blebs which can indicate that
ciprofloxacin can induce apoptosis in the B16 cell line (Fig. 4B′). G2/M accumulation was also
observed after 48 h in LC50 (Fig.
7), confirming that ciprofloxacin inhibited topoisomerase II
(13). Thus, the mode of action is
topoisomerase II inhibition.
The weakest action of ciprofloxacin was observed in
the case of the rat glioblastoma cell line. In this case, the LC90
value was impossible to establish. The calculated LC values were
the highest compared to the other tested cell lines, A549 and B16.
No cytostatic effect was observed after ciprofloxacin addition.
After F-actin staining, cell shrinkage and F-actin condensation was
observed. Cells lost their long cytoplasmatic lammelopodia which
reduced their attachement to the well surface (Fig. 5). These results are the
confirmation of the cell viability test (Fig. 2E). The changes in the cell cycle
after incubation with ciprofloxacin were unspecific (Fig. 8). All the above observations lead
to a conclusion that C6 cells were highly resistant to
ciprofloxacin treatment.
Aranha et al(18) analyzed the influence of
ciprofloxacin on the cell cycle against the HTB9 bladder cancer
cell line. They showed that after a 72- and 96 h incubation with
this drug at a concentration of 300 μg/ml, cells accumulated
in the S and G2/M phase (10% more cells in the S phase and 28–34%
in the G2/M phase as compared to control). We have shown that a
shorter incubation time could exert a cytostatic effect in G2/M
accumulation in A549 lung cancer cells.
The influence of this antibiotic on a different
NSCLC line, NCI-H460, was examined by Mondal et al(19). They showed that a lethal
concentration value (LC50) after a 48 h ciprofloxacin treatment was
as low as 19.5 μg/ml (in our study 102.1 μg/ml),
which confirmed the effectiveness of ciprofloxacin action against
lung cancer.
Chemotherapeutic agents can act in two ways: inhibit
cell growth (cytostatic) or kill cells (cytotoxic) (7). Toxic ciprofloxacin action against
cancer cells depends on topoisomerase II inhibition. We also
observed G2/M inhibition and showed its properties as a cytostatic
agent.
The anticancer effect against lung cancer can be
enhanced due to the ability of ciprofloxacin to accumulate in
higher concentrations in lung tissue than in serum after an
intravenous (i.v.) and oral administration. The excellent
penetration of ciprofloxacin from the blood into the lung
parenchyma was confirmed soon after an oral drug delivering. The
maximum oral dosage of ciprofloxacin prescribed to patients is 750
mg twice a day. Drug dose after an oral administration reaches 4.3
μg/ml concentration in the serum. The concentration of
ciprofloxacin after an oral administration is up to 7 times higher
in the lung tissue than in the serum (20). This indicates that the maximum
concentration of ciprofloxacin after an oral dose of one 750 mg
tablet can reach 30.1 μg/ml. After a long-term therapy this
concentration can be even higher. In some cases a patient can take
1 g oral dose of ciprofloxacin twice a day for 20 months (21). Ciprofloxacin penetrates into the
lung parenchyma also after an intravenous administration. After a
high-dose treatment (800 mg every 12 h for 8 days, i.v.)
ciprofloxacin can reach a serum concentration of 13 μg/ml
(potential concentration in the lung 91 μg/ml) (22). Such a high-dose of the drug did not
cause any side effects and do not cause the induction of bacterial
resistance. The minimal inhibitory concentration of ciprofloxacin
for most bacteria is 2 μg/g (5). After an administration of 300 mg i.v.
ciprofloxacin can reach a concentration of 7.3 μg/g.
Ciprofloxacin can be administered intrapulmonarily. Administration
of 200 μg/kg of ciprofloxacin can reach 40 and 20
μg/ml values in alveolar macrophages and epithelial lining
fluid, respectively. This method allows administration of lower
doses of ciprofloxacin compared to the oral route (23). LC50 after 48 h (100 μg/ml)
can be theoretically achieved in vivo in lung tissue
(Table I). This indicates that
ciprofloxacin can be added to conventional NSCLC chemotherapy as an
adjuvant drug with a different mechanism of action.
Ciprofloxacin seemed to be a good candidate for
experimental therapy for lung cancer and an antibiotic of choice
for the treatment of lung cancer with concomitant inflammation.
References
|
1.
|
SS HechtTobacco smoke carcinogens and lung
cancerJ Natl Cancer
Inst9111941210199910.1093/jnci/91.14.119410413421
|
|
2.
|
R RosellA VergnenegreB LiuBiomarkers in
lung oncologyPulm Pharmacol
Ther23508514201010.1016/j.pupt.2010.05.00320471486
|
|
3.
|
JT LouieCiprofloxacin: an oral quinolone
for the treatment of infections with gram-negatine pathogens.
Committee on Antimicrobial Agents. Canadian Infectious Disease
SocietyCMAJ15066967619948313286
|
|
4.
|
N SipsasC KosmasP ZiakasComparison of two
oral regiments for the outpatient treatment of low-risk cancer
patients with chemotherapy-induced neutropenia and fever:
Ciprofloxacin plus cefuroxime axetil versus ciprofloxacin plus
amoxicillin/clavulanateScand J Infect
Dis39786791200710.1080/00365540701367769
|
|
5.
|
S HaraguchiM HiokiK YamashitaCiprofloxacin
penetration into the pulmonary parenchyma in Japanese patientsSurg
Today37282284200710.1007/s00595-006-3393-417387558
|
|
6.
|
A DalhoffImmunomodulatory activities of
fluoroquinolonesInfection33S55S70200510.1007/s15010-005-8209-8
|
|
7.
|
K DrlicaMechanism of fluoroquinolone
actionCurr Opin
Microbiol2504508199910.1016/S1369-5274(99)00008-910508721
|
|
8.
|
T KloskowskiN GurtowskaM NowakR
JoachimiakA BajekJ OlkowskaT DrewaThe influence of ciprofloxacin on
A549, HepG2, A375.S2, B16 and C6 cell lines in vitroActa Pol
Pharm68859865201110.5506/APhysPolB.42.85922125950
|
|
9.
|
T KloskowskiN GurtowskaT DrewaDoes
ciprofloxacin have an obverse and a reverse?Pulm Pharmacol
Ther23373375201010.1016/j.pupt.2010.02.00520211752
|
|
10.
|
N GurtowskaT KloskowskiT
DrewaCiprofloxacin criteria in antimicrobial prophylaxis and
bladder cancer recurrenceMed Sci Monit16218223201020885364
|
|
11.
|
T KloskowskiJ OlkowskaA NazlicaT DrewaThe
influence of ciprofloxacin on hamster ovarian cancer cell line CHO
AA8Acta Pol Pharm67345349201020635529
|
|
12.
|
N KeX WangX XuYA AbassiThe xCELLigence
system for real-time and label-free monitoring of cell
viabilityMethods Mol
Biol7403343201110.1007/978-1-61779-108-6_621468966
|
|
13.
|
RE GonzalezCU LimK ColeCH BianchiniGP
SchoolsBE DavisI WadaIB RoninsonEV BroudeEffects of conditional
depletion of topoisomerase II on cell cycle progression in
mammalian cellsCell
Cycle1035053514201110.4161/cc.10.20.1777822067657
|
|
14.
|
P BenesL KnopfovaF TrckaInhibition of
topoisomerase IIα: Novel function of wedelolactoneCancer
Lett30329382011
|
|
15.
|
R SrivastavaApoptosis, Cell Signaling, and
Human DiseasesHumana PressTotowa, NJ2007
|
|
16.
|
EJ CalabreseLA BaldwinHormesis: U-shaped
dose responses and their centrality in toxicologyTrends in
Pharmacol Sci22285291200110.1016/S0165-6147(00)01719-311395156
|
|
17.
|
A GürbayF HincaA FavierCytotoxic effect of
ciprofloxacin in primary culture of rat astrocytes and protection
by vitamin EToxicology2295461200717098346
|
|
18.
|
O AranhaDP WoodFU SarkarCiprofloxacin
mediated cell growth inhibition, S/G2-M cell cycle arrest, and
apoptosis in human transitional cell carcinoma of the bladder cell
lineClin Cancer Res6891900200010741713
|
|
19.
|
ER MondalSK DasP MukherjeeComparative
evaluation of antiproliferative activity and induction of apoptosis
by some fluoroquinolones with a human non-small cell lung cancer
cell line in cultureAsian Pac J Cancer Prev5196204200415244525
|
|
20.
|
R RohwedderT BerganE CarusoSB
ThorsteinssonH Della TorreH SchollPenetration of ciprofloxacin and
metabolites into human lung, bronchial and pleural tissue after 250
and 500 mg oral
ciprofloxacinChemotherapy37229238199110.1159/0002388601790720
|
|
21.
|
IS KourbetiDE AlegakisS MarakiG
SamonisImpact of prolonged treatment with high-dose ciprofloxacin
on human gut flora: a case reportJ Med Case
Rep4111201010.1186/1752-1947-4-11120409330
|
|
22.
|
TR UtrupEW MuellerDP HealyRA CallcutJD
PetersonWE HurfordHigh-dose ciprofloxacin for serious gram-negative
infection in an obese, critically III patient receiving continuous
venovenous hemodiafiltrationAnn
Pharmacother4416601664201010.1345/aph.1P234
|
|
23.
|
S ChonoT TaninoT SekiK
MorimotoPharmacokinetic and pharmacodynamic efficacy of
intrapulmonary administration of ciprofloxacin for the treatment of
respiratory infectionsDrug Metab
Pharmacokinet228895200710.2133/dmpk.22.8817495415
|