Introduction
Pancreatic cancer is the fourth most common cause of
cancer deaths in United States with a five-year survival rate of
less than 5%. According to American Cancer Society, in the US alone
it is estimated that 43,920 individuals will be diagnosed with and
37,390 of them will die of pancreatic cancer in 2012 (1). Pancreatic cancer arises from the
morphologically and genetically clearly defined precursor lesions
through a step-wise accumulation of genetic alterations. In
majority of the patients diagnosed with pancreatic cancer, symptoms
do not develop until it is either unresectable or metastatic,
rendering it difficult to cure (2). The low survival rate of patients
points towards an increased need for novel strategies to combat
pancreatic cancer. The concept of chemoprevention has recently
received significant attention as a novel strategy towards
preventing pancreatic cancer before it occurs (3). Some chemopreventive agents such as
COX-2 inhibitors, green and black tea derivatives, β-carotene,
vitamins, isothiocyanates, and farnesyl transferase inhibitors have
been reported as potential chemopreventive agents (4–7).
Aspirin (ASP), a well-known non-steroidal
anti-inflammatory drug (NSAID), has emerged as a promising
chemopreventive agent against various types of cancer. ASP is
capable of suppressing pancreatic cancer growth both in
vitro and in vivo(8).
Cyclooxygenase-2 (Cox-2) enzyme which plays a key role in
prostaglandin E2 (PGE2) synthesis is overexpressed in several
cancers including pancreatic cancer. ASP has been shown to inhibit
pancreatic carcinogenesis by the inhibition of Cox-2 enzymes
(6). Studies associated with the
use of ASP for pancreatic cancer chemoprevention have met with
mixed results thus far (5,7). Given these conflicting reports on the
use of ASP in pancreatic cancer, it reaffirms the need for further
study of this drug in pancreatic cancer chemoprevention.
Curcumin (CUR), a diferuloylmethane is a derivative
of spice turmeric (Curcuma longa). CUR has shown to have
pronounced chemopreventive, anti-inflammatory, anti-oxidative and
anti-carcinogenic activities in different cancer cell lines and
murine cancer models (9–11). CUR has been shown to suppress NF-κB
activation and NF-κB gene products (12) and can induce p53-dependent
apoptosis by induction of p53 in certain cancer cell lines
(13). In pancreatic cancer cell
lines, CUR acts by suppressing the activation of NF-κB through the
inhibition of IκBα protein (14).
Sulforaphane (SFN) is a naturally-occurring
sulfur-containing isothiocyanate found in cruciferous vegetables
such as broccoli, brussel sprouts, cauliflower and cabbage
(15). SFN has been shown to
reduce NF-κB activity and affect expression of NF-κB mediated genes
encoding adhesion molecules, inflammatory cytokines, growth factors
and anti-apoptotic factors (16).
SFN also modulates multiple targets, which regulate many cellular
activities including oxidative stress, apoptosis induction, cell
cycle arrest, angiogenesis and metastasis suppression (17).
There is an increasing interest to use a combination
of chemopreventive agents that differ in their mode of action and
target multiple pathways. This approach provides a means of
obtaining low-dose therapy and increased efficacy with less
toxicity (18,19). It is noteworthy that the phase III
clinical trial of difluoromethylornithine combination with sulindac
has shown greater chemopreventive efficacy (19), which pave the way for the use of
combinatorial regimens to achieve a synergistic effect. To date, no
group has investigated the combined therapeutic effects of low dose
mixtures of ASP, CUR and SFN on prevention of pancreatic cancer.
Since all of these agents have different mechanisms of action, an
anticipated synergistic effect in pancreatic cancer would provide
valuable information in assessing combination chemopreventive
regimens for eventual clinical use.
The method of delivery plays an important role in
improving the overall drug bioavailability. Novel modes of delivery
methods using microspheres and nanosphere technology are receiving
wide attention as these have shown superior delivery compared to
conventional dosage forms (20,21).
The strength of the drug delivery system is their ability to alter
the pharmacokinetics and biodistribution of the drug. Nanoparticles
have unusual properties that can be used to improve drug delivery.
Provided they are within the nanometer size range, there is an
increased uptake by the cells through enhanced permeation and
retention (EPR) effect thereby making it a potential tool to
diagnose and treat cancer. Nanosized drug delivery systems offer
several advantages over the conventional delivery system such as
controlled and sustained release of drugs, ability of the drug to
cross the mucosal barriers, decreased renal and hepatic clearance,
decreased immune recognition, increased half-lives of drugs due to
slow and controlled release from the nanoparticulate systems,
increased stability and solubility (22). All these advantages suggest the
emerging role of nanoparticles in cancer therapy and
chemoprevention demonstrating a need for further research in this
area.
A nanotechnology-based drug delivery system,
solid-lipid nanoparticles (SLN) has received considerable attention
in the last few years as a convenient method of delivering drugs
into the body in a controlled release manner (23). SLNs are commonly defined as solid
nanoscaled lipid matrices in size range of 50–1,000 nm typically
consisting of a solid lipid compound, surfactant and incorporated
active ingredients. Additionally, SLNs are biocompatible and act by
protecting incorporated compounds against chemical degradation.
However, the most important advantage of SLNs is that they increase
the bioavailability of lipophilic drugs administered by the oral
route (24). There is considerable
evidence that SLNs act by carrying most of the drugs through the
lymphatic system, and in part through the general blood circulation
thus avoiding first pass metabolism (25). This allows for administration of
lower doses with less chances of toxic effects, while maintaining
efficacy. It has been observed that the formulations exhibited
superior and better cytotoxicity profile compared to their
corresponding free drug (26).
Thus, the SLN delivery system due to the favorable physicochemical
characteristics, controlled release kinetics would be ideal for
delivery of lipophilic drugs like ASP, CUR and SFN.
Although many studies have been conducted to test
the chemopreventive efficacy of ASP, CUR and SFN, no studies have
been reported on the combined chemopreventive efficacy of these SLN
encapsulated drugs. We recently demonstrated significant
chemoprevention of colon cancer in rats using an orally
administered drug loaded polylactide-co-glycolide (PLGA) polymer in
a nanotechnology-based targeted delivery system (26). Here, we are proposing the use of a
novel solid-lipid nanoparticle (SLN) technology for the oral
delivery of combinations of chemopreventive agents for pancreatic
cancer. Thus, in the present study we formulate the above mentioned
chemopreventive agents into SLNs and further evaluate their
combined chemopreventive efficacy in two different human pancreatic
cancer cells, MIA PaCa-2 and Panc-1.
Materials and methods
Materials
For the cell culture assays and solid lipid
nanoparticle (SLN) formulations, the drugs ASP, CUR and SFN were
obtained from LKT Laboratories (St. Paul, MN). Dimethyl sulfoxide
(DMSO) was obtained from Sigma Chemicals (St. Louis, MO). Stearic
acid (lipid) and Poloxamer 188 (emulsion stabilizer) was obtained
from Spectrum Chemicals (Garden, CA). Dichloromethane (DCM) was
obtained from Fisher Scientific (Houston, TX).
Human cell lines
Human pancreatic cancer cell lines MIA PaCa-2 and
Panc-1 were obtained from ATCC (Rockville, MD). Cells were
maintained in Dulbecco’s modified Eagle’s medium (DMEM) containing
10% fetal bovine serum (FBS) obtained from ATCC. Cells were
cultured at 37°C in a humidified atmosphere of 5% CO2
and 95% air.
Preparation of solid lipid nanoparticles
(SLNs)
ASP and CUR SLNs were prepared using a hot melt
oil-in-water (o/w) emulsion technique. Stearic acid was used as the
solid lipid to make the nanoparticle formulations. Briefly, 1 mg of
stearic acid was melted by heating in a water bath at 70–80°C. The
drug (100 mg) was suspended in 3 ml of DCM. The suspended drug
solution was then added to the melted stearic acid and heated until
all DCM was evaporated. The water phase consisted of 1% poloxamer
solution which was heated to the same temperature as that of the
oil phase. The oil phase was then added to the poloxamer solution
and the mixture was further sonicated for 5 min using an
ultra-sonicator (Branson, Los Angeles, CA) to create an o/w
emulsion. The emulsion so formed was then cooled and washed with
water to remove excess free drug from the particle surface. SLNs
were freeze-dried in a freeze dryer (Labconco, Kansas City, MO) and
subjected to particle sizing and encapsulation efficiency
determination.
Particle size measurement
The particle sizes of the formulated SLNs were
measured using Nicomp submicron particle size analyzer Model 370
(New York, USA). Briefly, 5 mg of the SLN formulation was suspended
in 10 ml of phosphate saline buffer (PBS, pH 7.4) and was sonicated
for 2 min. Particle size was measured using 1 ml of the
suspension.
Encapsulation efficiency (%)
determination
Encapsulation efficiency (E.E) was determined by
dissolving 10 mg of the SLN formulation in DCM which dissolves the
stearic acid and releases the drug entrapped within the lipid. DCM
was evaporated under a current of inert air for 1 h. Evaporation of
DCM left a residue of the drug and lipid sticking to the bottom of
the test tube. Drug was separated from the lipid by dissolving it
in 5 ml of acetonitrile. The drug was allowed to dissolve freely
for about 30 min in a bath sonicator after which it was filtered
through a 0.45 μm filter. The resulting solution was further
diluted to 20 ml by adding acetonitrile. A total of 1 ml of the
resulting mixture was analyzed using Shimadzu LC-20 binary HPLC
system (Columbia, MD). A total of 20 μl of naproxen was used
as an internal standard. E.E was calculated using the following
expression: E.E (%) = amount (mg) of drug per HPLC
method/theoretical yield (mg) ×100.
Determination of drug release from
SLNs
The in vitro release profile of ASP and CUR
from SLNs was determined using a dialysis bag method. A hydrophilic
dialysis membrane pouch (MWCO 12 kDa) served as the donor
compartment. SLNs containing 100 mg of the drug were suspended in 5
ml of PBS and placed inside the membrane pouch. Subsequently, the
pouch was lowered into a container with 100 ml PBS solution
containing 1% sodium lauryl sulfate (SLS) serving as the receptor
medium for the study. At fixed time intervals, 5 ml receptor medium
was withdrawn and replaced with 5 ml fresh medium. Samples were
analyzed using HPLC. All the samples were carried out in
triplicates.
Drug encapsulated SLN stability
studies
Storage stability studies were conducted on ASP and
CUR SLN formulations over a period of 3 months. Samples were stored
at three different temperatures 4°C, 24°C and 37°C in closed glass
vials. Particle size and encapsulation efficiencies were determined
as indicators of storage stability of the prepared SLNs. All the
studies were conducted in triplicate.
Cell viability (MTS) assay
The cell viability assay was performed according to
manufacturer’s protocol included with the Promega CellTitre 96
Aqueous MTS reagent (Madison, WI). Briefly, 2.5×103
cells of MIA PaCa-2 cells and 4×103 cells of Panc-1 were
seeded in 96-well plates and incubated for 24 h. Test compounds ASP
SLNs, CUR SLNs and free SFN alone and in combination at a
concentration of 25 μM, 2.5 μM and 5 μM
respectively, were added and incubated for a period of 72 h. On the
last day of the incubation period, the growth medium was removed
followed by addition of 100 μl mixture consisting of 20% MTS
and 1% of phenazine methosulfate (PMS) to the serum-free culture
medium and incubated for 2 h at 37°C. MTS is bioreduced by cells
into formazan which can be measured at 490 nm. Thus, the quantity
of formazan product measured by the amount of 490 nm absorbance is
directly proportional to the number of living cells in culture.
IC50 values were determined using Prism software (San
Diego, CA). All the samples were performed in triplicates. Each
experiment was performed at least thrice.
Annexin V-PI apoptosis assay
The assay was performed according to manufacturer’s
protocol included with the Annexin V-fluorescein isothiocyanate
(FITC) Vybrant Apoptosis assay kit #3 ( Invitrogen, Green Island,
NY). Briefly, 3×105 MIA PaCa-2 and Panc-1 cells were
seeded in 6-well plates and incubated for a period of 24 h. Test
compounds ASP SLNs, CUR SLNs and free SFN alone and in combination
at a concentration of 25 μM, 2.5 μM and 5 μM,
respectively, were added and incubated for a period of 48 h. After
the incubation period, cells were washed twice with cold PBS
(phosphate buffered saline) and then resuspended in 1X Annexin
binding buffer such that the cell density was equivalent to
1×106 cells/ml. A total of 100 μl of this cell
suspension was then subjected to 5 μl of FITC Annexin V and
1 μl of the 100 μg/ml PI followed by incubation in
the dark for 15 min. After the incubation period, 400 μl of
1X annexin binding buffer was added to the cells followed by gentle
vortexing. The samples were kept on ice and were analyzed using
Beckman Coulter Cytomics FC500 (Brea, CA). The fluorescence
emission was measured at 530 nm (e.g. FL1) and >575 nm (e.g.
FL3).
Statistical analysis
Results were expressed as mean ± SEM. A one-way
ANOVA followed by Tukey’s post hoc analysis using Graph pad prism
software (La Jolla, CA) was done to analyze and compare the
results. A probability value of ≤0.05% was considered
significant.
Results
Particle sizing of drug encapsulated
SLNs
Nicomp volume weight was used as the standard
measure for poly distribution type particle sizing. The SLNs of ASP
and CUR were retained within the nanometer range (Table I). The particle size of ASP SLNs
(150 nm) was lower than that of CUR (249 nm). All the SLNs showed
optimal particle size with low variability.
 | Table IParticle size and encapsulation
efficiency of drug-loaded solid lipid nanoparticles. |
Table I
Particle size and encapsulation
efficiency of drug-loaded solid lipid nanoparticles.
| Drug | Particle size
(nm) | Encapsulation
efficiency (%) |
|---|
| Aspirin | 150±63 | 85±5.2 |
| Curcumin | 249±65 | 69±3.0 |
Encapsulation efficiency (%)
The SLNs exhibited 85 and 69% encapsulation
efficiency of the ASP and CUR, respectively, within the lipid
(Table I). The encapsulation
efficiency was higher for ASP in comparison to CUR.
Drug release from the SLNs
The drug release studies were performed using
membrane compartmental analysis. The release of the drug from
nanoparticles prepared using stearic acid as lipid was conducted
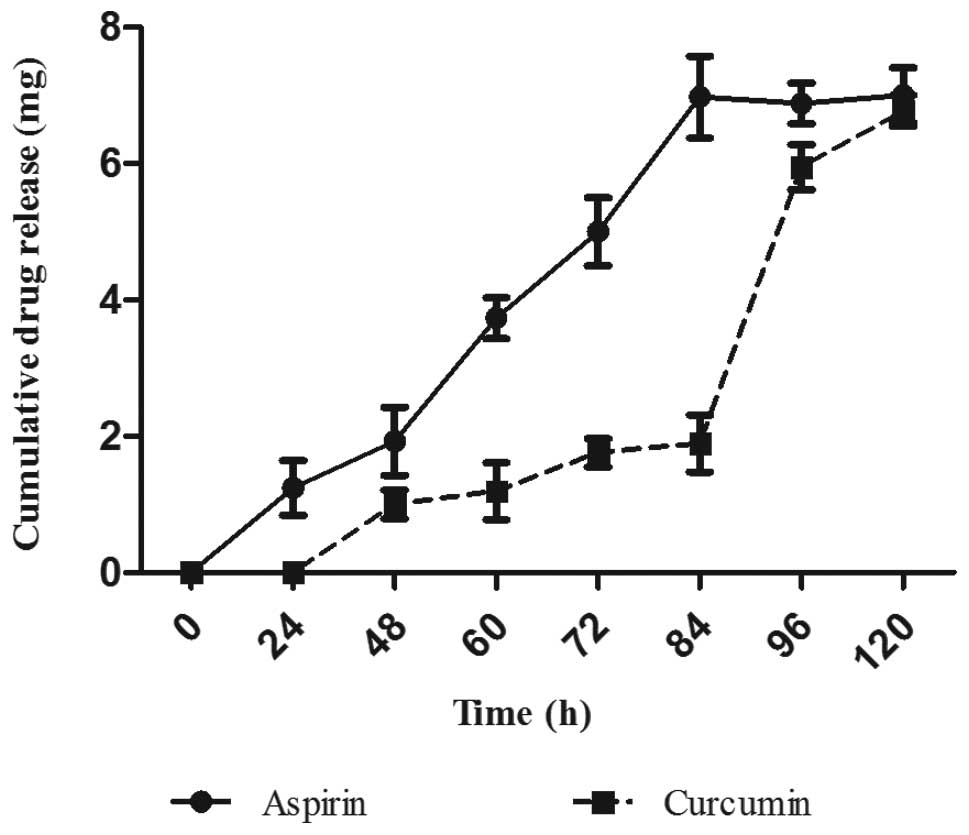
over a period of 5 days. As shown in Fig. 1, the release of ASP was faster
compared to CUR. Release of CUR was not observed until a period of
24 h. A cumulative drug release of approximately 7 mg of ASP was
observed within 90 h of the study showing faster release pattern in
comparison to the curcumin SLNs. However, in comparison to ASP
SLNs, CUR SLNs showed a slower drug release time profile releasing
approximately 6.5 mg of the drug entrapped. Both the formulations
exhibited slow sustained release of the drug.
Drug encapsulated SLN stability
studies
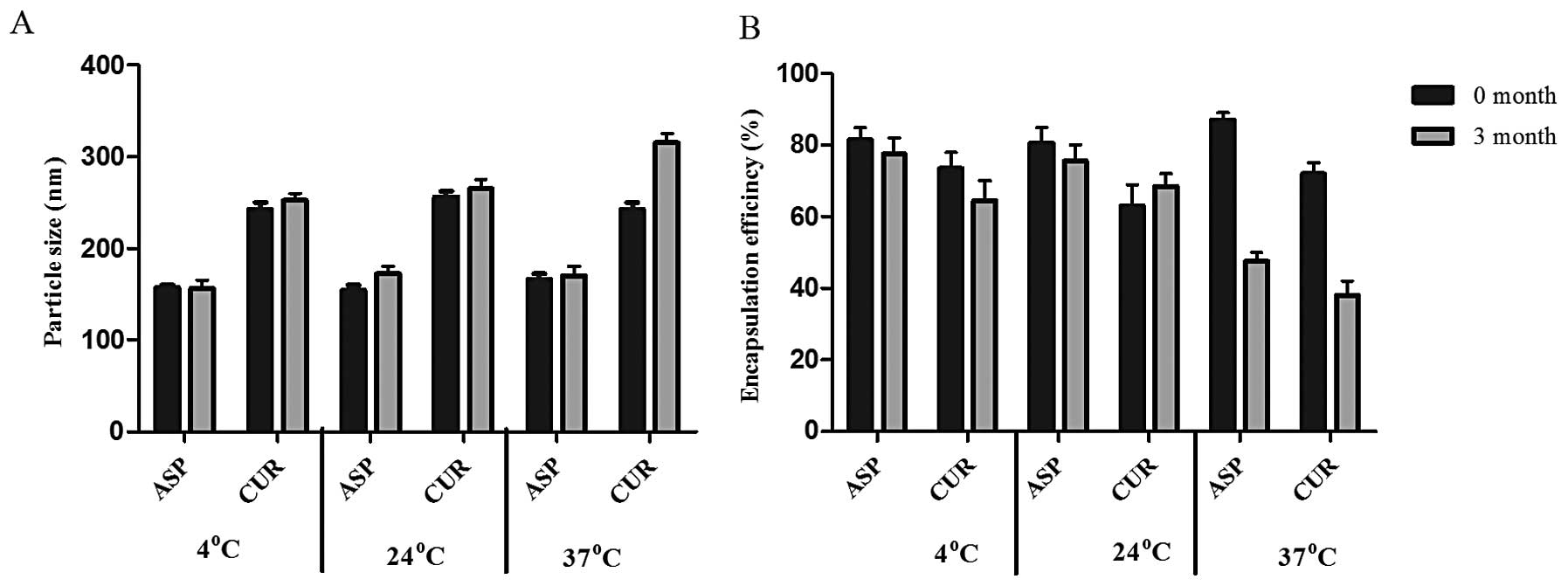
i) Particle size (nm). Particle size of ASP
SLNs measured at the start of the study showed a size range of
160±32 nm. After a three month duration, where samples were stored
at three different temperatures (4°C, 24°C and 37°C) the particle
sizes were 165, 164 and 180 nm, respectively. For CUR SLNs, the
size range at the start of the storage study was 250±40 nm. At the
end of 3 months study, the particle sizes were 260, 255 and 325 nm,
respectively. Thus both ASP and CUR SLNs exhibited stability at
lower temperatures (4°C and 24°C). However, some agglomeration was
evident at 37°C which demonstrated an increase in particle size at
that temperature (Fig. 2A). ii)
Encapsulation efficiency (%). ASP SLNs showed an initial
encapsulation efficiency (E.E) of 85% at the start of the storage
stability test. After 3 months at different temperatures, the E.E
was found to be 82% (4°C), 80% (24°C) and 45% (37°C). The
encapsulation efficiency of CUR SLNs was demonstrated to be 69% but
after 3 months at different temperatures showed 70% (4°C), 65%
(24°C) and 34% (37°C) encapsulation (Fig. 2B). Thus, the encapsulation seemed
to be affected at higher temperatures. Results of these studies
demonstrated that storage at higher temperatures of 37°C could
result in particle size and encapsulation changes which would have
a direct adverse impact on the drug release characteristics from
the SLNs used in the study.
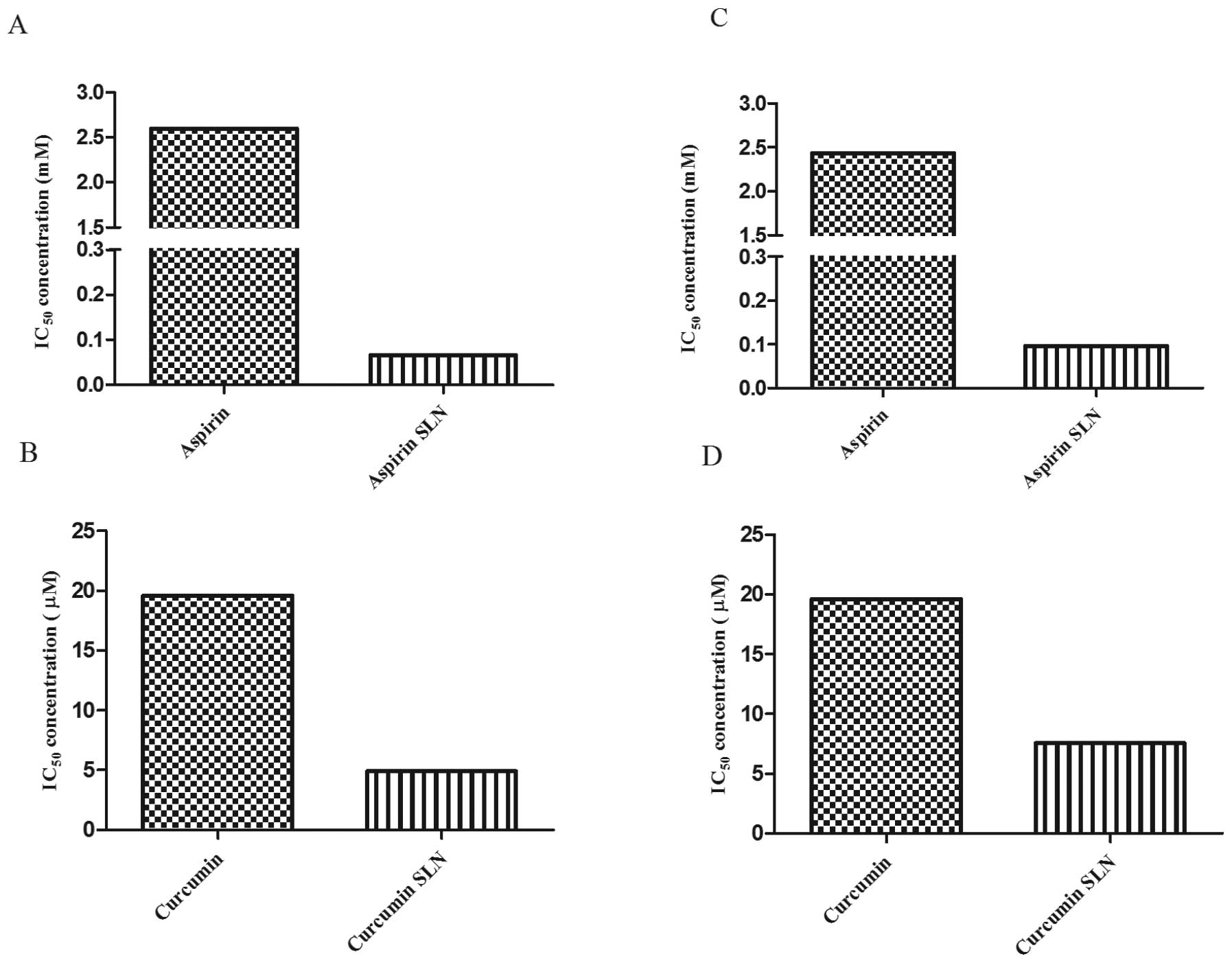
IC50 comparisons between free
drugs and drug encapsulated SLNs
In order to study the effects of SLNs, the
inhibitory concentrations (IC50) values of the free drug
and the drug encapsulated SLNs were compared. It was observed that
the SLNs exhibited lower IC50 values in comparison to
the free drug. In case of MIA PaCa-2 cells, free ASP exhibited
IC50 value of 2.6 mM. However, in case of drug
encapsulated SLNs, the IC50 value was significantly
reduced to 66.08 μM, showing approximately 38-fold reduction
compared to the free form of the drug (Fig. 3A). Whereas, free CUR exhibited the
IC50 value of 19.6 μM whereas CUR SLNs exhibited
IC50 value of 4.93 μM with ∼3-fold reductions
from its free form (Fig. 3B).
Similarly for Panc-1 cells, the IC50 values obtained for
free ASP and ASP SLNs were 2.4 mM and 99.11 μM, respectively
(Fig. 3C). Whereas, the
IC50 values of CUR and CUR SLNs obtained were 19.6
μM and 7.569 μM, respectively (Fig. 3D). Thus, our results demonstrate
that the drugs when encapsulated in SLNs exhibited cytotoxicity at
lower concentrations compared to free forms of the drugs.
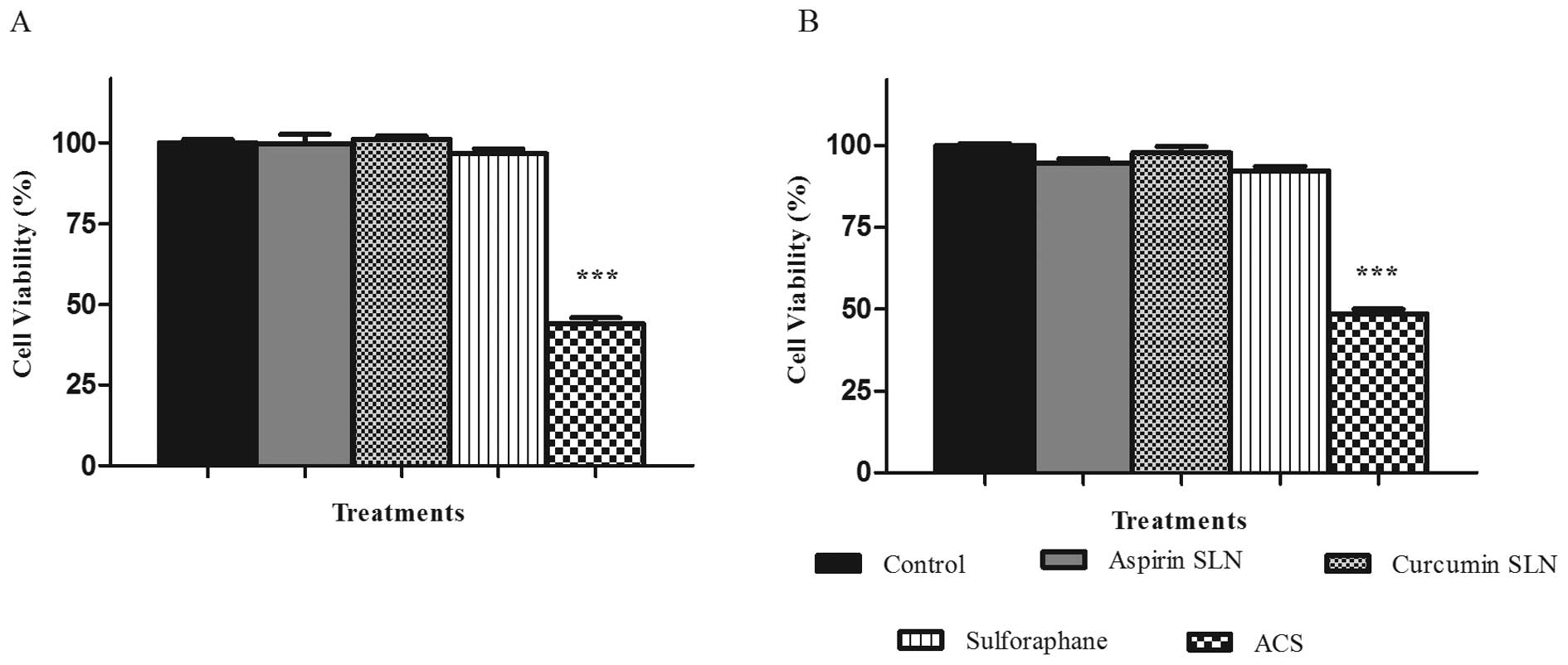
Novel combined chemopreventive regimen of
ASP SLNs, CUR SLNs and free SFN show a synergistic effect on the
reduction of cell viability
MTS assay was performed in order to study the
combination effects of chemopreventive drug SLNs on the cell lines
MIA PaCa-2 and Panc-1. After determining the dose response curves
individually and obtaining the IC50 value for each of
them, ineffective drug concentrations were selected for ASP SLN (25
μM), CUR SLN (2.5 μM) showing minimal inhibitory
response on the cell lines. SFN was used in its unmodified form at
a concentration of 5 μM. When combined together as a
combination (ACS), the cell viability was reduced to 43.6% for MIA
PaCa-2 cell line (Fig. 4A) and
48.49% for Panc-1 cell line (Fig.
4B), respectively. This change was significant (p<0.0001) in
comparison to the reduction in cell viability of individual
concentrations of ∼10%. Thus, synergistic effects were observed
when combination of drugs was used. Dual combinations showed
reduction in cell viability of ∼20% hence demonstrating no
significant differences when compared to individual drug
concentrations (data not shown).
Combined chemopreventive regimen shows
increased apoptosis in human pancreatic cancer cells
Induction of apoptosis by ASP (25 μM), CUR
(2.5 μM) and SFN (5 μM) alone or the combination was
evaluated in MIA PaCa-2 and Panc-1 cells. The cells were exposed to
the drugs for 48 h, and induction of cell apoptosis was examined by
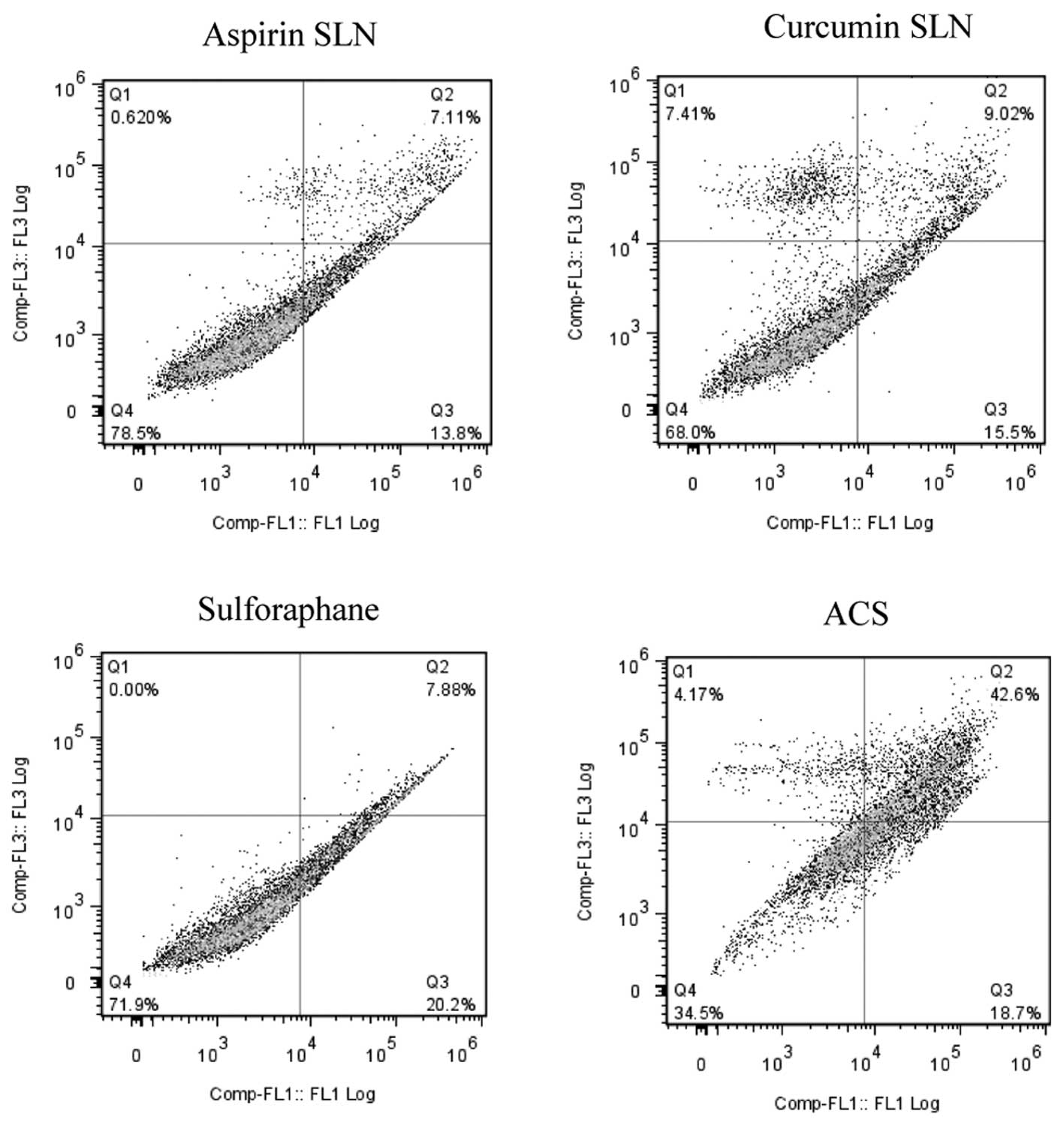
Annexin-V binding and PI staining assay. In case of MIA PaCa-2 cell
line as shown by the representative contour plots (Fig. 5), individual concentrations of SLN
chemopreventive agents showed low evidence of apoptosis in both the
cell lines; ASP SLNs exhibited 20.91%, CUR SLNs 24.52% and SFN
28.08% apoptotic cells. However, when the cells were exposed to a
combination (ACS), an apoptotic effect of 61.3% was observed.
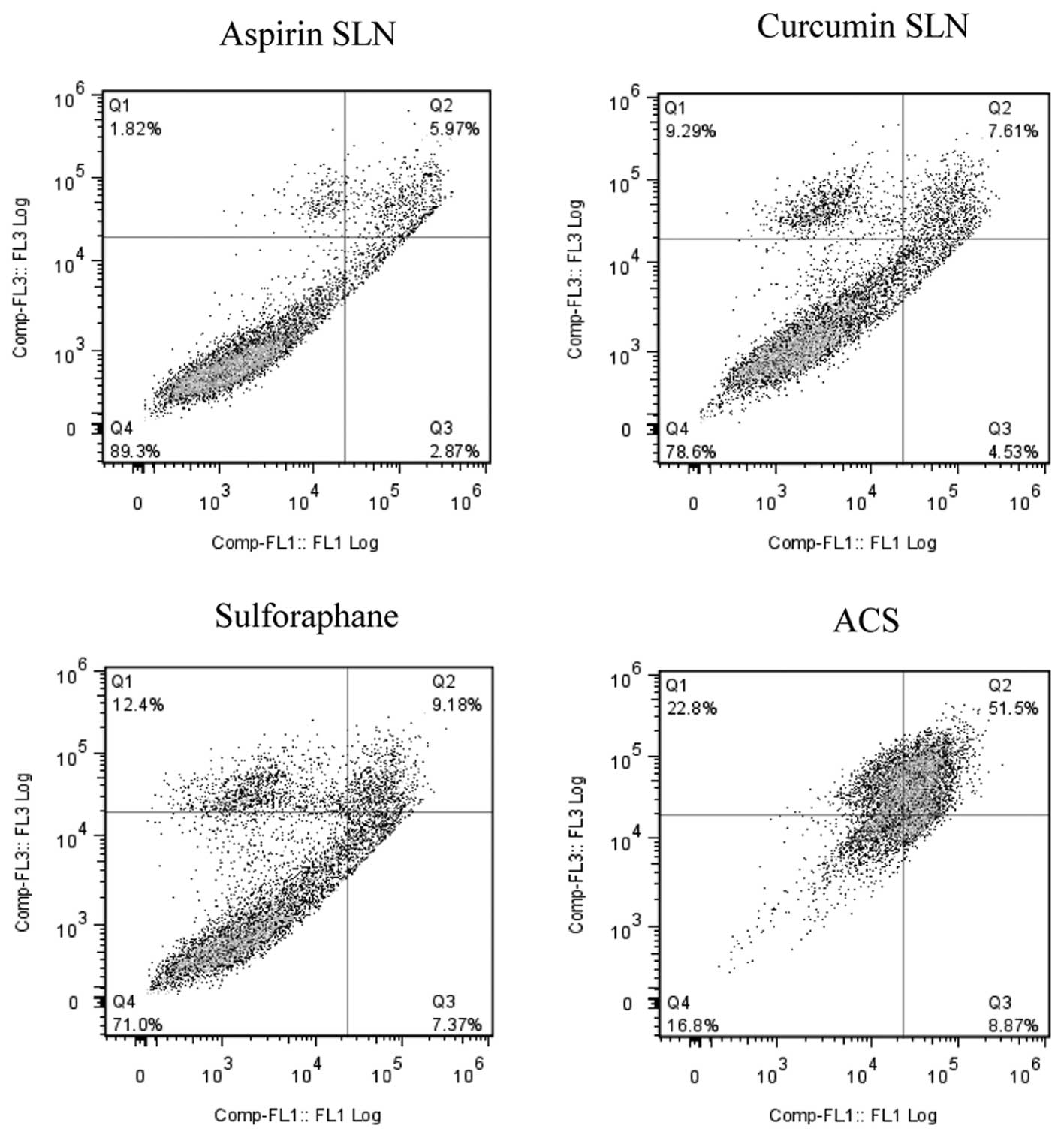
A similar effect was observed in the case of Panc-1
cells (Fig. 6) where individual
drug concentrations of ASP SLNs, CUR SLNs and SFN exhibited 8.84%,
12.14% and 16.55% of apoptotic cells, respectively. When the
combination was used, increase in apoptotic effect was observed
showing 60.37% apoptotic cells. Thus, the combination of SLN
chemopreventive agents was synergistically effective in inducing
apoptosis at low concentrations in both MIA PaCa-2 and Panc-1
cells.
Discussion
Pancreatic cancer ranks as the fourth most deadly
form of cancer in the United States with approximately 37,000
deaths each year (1). Early
diagnosis of this disease is difficult because it develops without
any early symptoms. Survival of patients with pancreatic cancer has
been <5% over 5 years which makes this disease of great concern
(2). Therapeutic outcomes with
pancreatic cancer are not useful for patients especially upon a
late diagnosis thus strategies to prevent this disease from
occurring have become an important area of research. Recently,
chemoprevention has emerged as an effective tool in the fight
against various types of cancers, including colon and pancreatic
cancer (19,27).
Our research has been focused on the administration
of low doses of ACS combination SLNs to study its chemopreventive
effects against pancreatic cancer cells MIA PaCa-2 and Panc-1.
Single agent administration at low concentrations has been
demonstrated to be ineffective, hence the hypothesis that two or
more chemopreventive agents when delivered at low concentrations
together, may exhibit an additive or synergistic effect against the
cancer cells was tested. This can be attributed to the
multi-factorial nature of carcinogenesis wherein cancer occurs as a
result of multiple cellular changes during a prolonged time period.
Of the various chemopreventive agents being studied, ASP, CUR and
SFN have been proven to be effective in the chemoprevention of
pancreatic cancer (2–4). Several in vivo and in
vitro studies have shown that NSAIDs like aspirin and celecoxib
have helped prevent the progression of pancreatic cancer. CUR and
SFN, both being effective and non-toxic in nature, have been
investigated for their chemopreventive actions (9,12,17).
Therefore using a multi-disciplinary approach, this study
investigated the synergistic effects of SLN combinations of
chemopreventive agents, namely, ASP in combination with CUR and
SFN.
Nevertheless, the clinical translation of these
agents has been significantly hampered due to various reasons
including poor oral bioavailability after administration.
Engineering an SLN drug delivery system for these agents offers a
means of increasing the bioavailability in the plasma and tissues
in comparison to the free form of these drugs, thereby ultimately
improving the therapeutic efficacy. Also, encapsulating the drug
within the lipid matrix allows for administration of lower doses of
drugs with less chance of toxic effects, while maintaining
efficacy. Furthermore, using combination of agents which differ in
their mode of action helps to obtain a desired preventive effect,
while minimizing the dose concentration and its toxic effects
(26). In terms of formulation
development, both ASP and CUR SLNs were found to have optimal
particle sizes, high encapsulation efficiency with good stability
at or below room temperature. The improved stability may be
explained by the use of organic solvent in the preparation process
which may have improved the hygroscopic nature of the lipid. The
high encapsulation can be attributed to the lipophilic nature of
both ASP and CUR. Also the particle sizing was found to be in
nanometer range suggesting a better chance of cellular uptake of
the drugs. The formulated SLNs are non-toxic because they are made
of physiological lipids such as stearic acid.
The effect of these agents was initially evaluated
by calculating the IC50 values and then by combining the
ineffective concentrations to exhibit an additive or synergistic
effect against the cancer cells proving to be more efficacious at
lower concentrations. A comparative study between the two forms of
the drugs i.e., the free form and SLN form was carried out. It was
observed that ASP and CUR SLNs IC50 concentrations
exhibited approximately 38- and 3-fold reductions, respectively in
comparison to the free form of the drug. Studies have been reported
where drug loaded SLNs have exhibited better cytotoxicity profile
in comparison to the free drug (28). This has been mainly attributed to
the smaller particle size of the nanoparticles which increases the
overall uptake of the drug. The surfactant used in the development
process determines the inhibitory effect on the cells. We used
Poloxamer 188 which has previously been shown to target cancerous
cells, due to differences in the membrane of these cells when
compared to the non-cancer cells. Poloxamer has not only been shown
to inhibit multiple drug resistance (MDR) proteins and other drug
efflux transporters on the surface of the cancer cells but also
shown to enhance protoapoptotic signaling and decrease
anti-apoptotic defense in MDR cells (29). The MTS assay on drug entrapped SLNs
was carried out using ASP (25 μM), CUR (2.5 μM) and
SFN (5 μM) as individual concentrations. Individually they
showed little or no decrease in the cell viability in the two cell
lines, but when combined, a significant reduction by 60% was
observed in MIA PaCa-2 and Panc-1 cells. In order to validate the
efficacy of the combination regimen, apoptosis assay was conducted
which determined the progression of a cancer cell from four
different phases after the addition of the drug; living cell, early
apoptotic cell, late apoptotic cell and necrotic cells. These
results seem to be consistent with our findings in the MTS assay.
The possible mechanisms involving the significant change for the
combination could be an additive effect of the COX-2 enzyme
inhibition, the regulation of the P53 suppressor pathway and by the
modulation of the Nrf2 pathway; however additional studies need to
be done to verify the findings.
From these results, we believe that chemoprevention
is an effective way to prevent pancreatic cancer especially as the
disease cannot be diagnosed at an early stage. Using a
multi-disciplinary approach, this study investigated the
synergistic effects of a combination of ASP and CUR SLN with free
SFN. We demonstrated for the first time that this SLN combination
showed a synergistic inhibition of cell viability and induce
apoptosis in human pancreatic cancer cell lines. However, further
in vivo studies have to be conducted to test the efficacy of
this SLN combination. In conclusion, the results obtained from
formulation studies and cell based assays clearly demonstrate the
scope of developing the combination drug encapsulated SLN
formulations to prevent pancreatic cancer.
Acknowledgements
This study was supported by a grant
from National Institutes of Health (1R03CA153812-01A1; SP).
References
|
1.
|
R SiegelD NaishadhamA JemalCancer
statistics, 2012CA Cancer J Clin621029201210.3322/caac.20138
|
|
2.
|
V FendrichChemoprevention of pancreatic
cancer-one step closerLangenbecks Arch
Surg397495505201210.1007/s00423-012-0916-x22350613
|
|
3.
|
CD LogsdonJL AbbruzzeseChemoprevention of
pancreatic cancer: ready for the clinic?Cancer Prev Res
(Phila)313751378201010.1158/1940-6207.CAPR-10-021621084259
|
|
4.
|
SS HusainIL SzaboAS TamawskiNSAID
inhibition of GI cancer growth: clinical implications and molecular
mechanisms of actionAm J
Gastroenterol97542553200210.1111/j.1572-0241.2002.05528.x11922545
|
|
5.
|
AT ChanEL GiovannucciES SchernhammerA
prospective study of aspirin use and the risk for colorectal
adenomaAnn Intern
Med140157166200410.7326/0003-4819-140-3-200402030-0000614757613
|
|
6.
|
A KokawaH KondoT GotodaIncreased
expression of cyclooxygenase-2 in human pancreatic neoplasms and
potential for chemoprevention by cyclooxygenase
inhibitorsCancer91333338200110.1002/1097-0142(20010115)91:2%3C333::AID-CNCR1006%3E3.0.CO;2-N11180079
|
|
7.
|
EJ JacobsCJ ConnellC RodriguezAV PatelEE
CalleMJ ThunAspirin use and pancreatic cancer mortality in a large
United States cohortJ Natl Cancer
Inst96524528200410.1093/jnci/djh08415069114
|
|
8.
|
SD StanSV SinghRE BrandChemoprevention
strategies for pancreatic cancerNat Rev Gastroenterol
Hepatol7347356201020440279
|
|
9.
|
ML KuoTS HuangJK LinCurcumin, an
antioxidant and anti-tumor promoter, induces apoptosis in human
leukemia cellsBiochim Biophys
Acta131795100199610.1016/S0925-4439(96)00032-48950193
|
|
10.
|
FH SarkarS BanerjeeY LiPancreatic cancer:
pathogenesis, prevention and treatmentToxicol Appl
Pharmacol224326336200710.1016/j.taap.2006.11.00717174370
|
|
11.
|
G SaT DasAnti cancer effects of curcumin:
cycle of life and deathCell
Div314200810.1186/1747-1028-3-1418834508
|
|
12.
|
L MoragodaR JaszewskiAP MajumdarCurcumin
induced modulation of cell cycle and apoptosis in gastric and colon
cancer cellsAnticancer Res21873878200111396178
|
|
13.
|
DW LiJP LiuYW MaoCalcium-activated
RAF/MEK/ERK signaling pathway mediates p53-dependent apoptosis and
is abrogated by alpha B-crystallin through inhibition of RAS
activationMol Biol
Cell1644374453200510.1091/mbc.E05-01-001016000378
|
|
14.
|
JA RomashkovaSS MakarovNF-kappaB is a
target of AKT in anti-apoptotic PDGF
signallingNature4018690199910.1038/4347410485711
|
|
15.
|
NV MatusheskiJA JuvikEH JefferyHeating
decreases epithiospecifier protein activity and increases
sulforaphane formation in
broccoliPhytochemistry6512731281200410.1016/j.phytochem.2004.04.01315184012
|
|
16.
|
G KallifatidisV RauschB
BaumannSulforaphane targets pancreatic tumour-initiating cells by
NF-kappaB-induced anti-apoptotic
signallingGut58949963200910.1136/gut.2008.14903918829980
|
|
17.
|
JW LampeSulforaphane: from chemoprevention
to pancreatic cancer
treatment?Gut58900902200910.1136/gut.2008.16669419520886
|
|
18.
|
CV RaoHL NewmarkBS ReddyChemopreventive
effect of farnesol and lanosterol on colon carcinogenesisCancer
Detect Prev26419425200210.1016/S0361-090X(02)00119-812507226
|
|
19.
|
P ZhouSW ChengR YangB WangJ LiuCombination
chemoprevention: future direction of colorectal cancer
preventionEur J Cancer
Prev21231240201210.1097/CEJ.0b013e32834dbbfd22456425
|
|
20.
|
A ChaudharyJ WangS PrabhuDevelopment and
validation of a high-performance liquid chromatography method for
the simultaneous determination of aspirin and folic acid from
nano-particulate systemsBiomed Chromatogr24919925201020033881
|
|
21.
|
S PrabhuLP TranGV BetageriEffect of
co-solvents on the controlled release of calcitonin polypeptide
from in situ biodegradable polymer implantsDrug
Deliv12393398200510.1080/1071754059096887316253955
|
|
22.
|
L Brannon-PeppasJO BlanchetteNanoparticle
and targeted systems for cancer therapyAdv Drug Deliv
Rev5616491659200410.1016/j.addr.2004.02.01415350294
|
|
23.
|
RH MüllerK MäderS GohlaSolid lipid
nanoparticles (SLN) for controlled drug delivery - a review of the
state of the artEur J Pharm Biopharm50161177200010840199
|
|
24.
|
M UnerG YenerImportance of solid lipid
nanoparticles (SLN) in various administration routes and future
perspectivesInt J Nanomed2289300200718019829
|
|
25.
|
H YuanJ ChenYZ DuFQ HuS ZengHL ZhaoStudies
on oral absorption of stearic acid SLN by a novel fluorometric
methodColloids Surf B
Biointerfaces58157164200710.1016/j.colsurfb.2007.03.00217446050
|
|
26.
|
A ChaudharyD SutariaY HuangJ WangS
PrabhuChemoprevention of colon cancer in a rat carcinogenesis model
using a novel nanotechnology-based combined treatment systemCancer
Prev Res (Phila)416551664201110.1158/1940-6207.CAPR-11-0129
|
|
27.
|
BS ReddyJ NayiniK TokumoJ RigottyE ZangG
KelloffChemoprevention of colon carcinogenesis by concurrent
administration of piroxicam, a nonsteroidal antiinflammatory drug
with D,L-alpha-difluoromethylornithine, an ornithine decarboxylase
inhibitor, in dietCancer Res50256225681990
|
|
28.
|
A MigliettaR CavalliC BoccaL GabrielMR
GascoCellular uptake and cytotoxicity of solid lipid nanospheres
(SLN) incorporating doxorubicin or paclitaxelInt J
Pharm2106167200010.1016/S0378-5173(00)00562-711163988
|
|
29.
|
F YanC ZhangY ZhengThe effect of poloxamer
188 on nanoparticle morphology, size, cancer cell uptake, and
cytotoxicityNanomedicine6170178201010.1016/j.nano.2009.05.00419447200
|