Introduction
Breast cancer, the most prevalent cancer in women
worldwide, and the leading cause of cancer death in women, was
projected to claim the lives of approximately 39,500 women in the
US in 2011 (1,2). Though treatable in early stages, once
metastasis has occurred the survival rate is drastically reduced to
a median of 2–3 years and treatment focuses on palliative care
(3). Melanoma, another aggressive
cancer, also has no viable treatment once it metastasizes from the
skin to other areas of the body, such as lymph nodes, lungs, liver,
brain or bone. Since 90% of cancer deaths occur secondary to
metastasis, any successful anticancer treatment has to target this
stage of cancer development.
Critical events in tumor cell invasion include cell
attachment, proteolytic degradation of the extracellular matrix
(ECM) and migration through the disrupted matrix (4). Rath and Pauling proposed that the
most effective and universal approach to controlling cancer is
ensuring optimal synthesis and integrity of collagen, which is
dependent upon essential nutrients, such as vitamin C and lysine
(5). Ascorbic acid, needed for
synthesis and hydroxylation of collagen and optimal stability of
the ECM, is produced by most animals but not by humans (5). Humans must obtain ascorbate by diet
and cancer patients have been shown to have very low reserves of
vitamin C (6,7).
Our main objective was to determine the effect of
dietary ascorbate supplementation on the development of tumors in
mice unable to synthesize ascorbic acid, gulonolactone oxidase
(gulo) knockout (KO) mice using two models: challenging mice with
breast cancer 4T1 cells into the mammary pads and challenging with
injection of melanoma B16FO cancer cells intraperitoneally (IP).
The 4T1 mammary tumor carcinoma model was chosen for this study as
it has several characteristics that make it a suitable experimental
animal model for human mammary cancer growth (8,9). The
tumor cells are easily transplanted into the mammary gland so that
the primary tumor grows in the anatomically correct site. Second,
as in human breast cancer, 4T1 metastatic disease develops
spontaneously from the primary tumor. Also the progressive spread
of 4T1 metastases to the draining lymph nodes and other organs is
very similar to that of human mammary cancer. The B16FO model was
utilized since metastatic malignant melanoma cells, specifically
B16, cells are extremely aggressive and metastasize to secondary
areas of the body, such as lymph nodes, lungs, liver, brain or bone
and have been successfully utilized for experimental metastasis to
study the effectiveness of anticancer agents (10).
Materials and methods
Animals
Female Balb/C wild-type mice and mice heterozygous
for the sfx mutation, a deletion in the L-gulono-γ lactone oxidase
gene (gulo) were obtained from The Jackson Laboratory (Bar Harbor,
ME). The gulo mice were bred to develop a homozygous gulo KO colony
and were maintained on vitamin C fortified food and water.
Genotyping of litters was performed through Transnetyx (Cordova,
TN) and female gulo KO mice were selected from the homozygous
colony for the study. Gulo KO mice selected for the study were
approximately 40–42 weeks of age at the time of inoculation. In
addition, a group of wild mice 14 weeks of age at the time of
inoculation were used as a reference for the study. Mice were
acclimated for a week before treatments, housed in standard
separator cages with bedding on a 24-h light/dark schedule. All
animals were cared for in accordance with institutional guidelines
for the care and use of experimental animals.
Diet
Gulo KO mice were divided into two groups: group 1
and 2, and wild-type mice were allocated to group 3. Prior to
injection with melanoma or breast cancer cells, the groups of mice
were maintained for 4 weeks on the following diets. Group 1
(vitamin C deprived gulo KO mice) and group 3 (wild-type mice) were
maintained on a regular diet (Laboratory Rodent Diet 5001 from
Purina Mills, LLC/Test Diet) and distilled water. Group 2 (vitamin
C-supplemented gulo KO mice) were provided the regular diet
supplemented with 500 ppm L-ascorbyl-2-polyphosphate and distilled
water with 150 mg/l ascorbic acid, 0.01 mM EDTA. The
ascorbate-supplemented nutrient mix diet was milled and pressed by
Purina Mills, LLC.
Experimental design
Study 1
After mice had been on their respective diets for 4
weeks, 5×105 murine melanoma B16FO cells in 0.2 ml PBS
were injected intraperitoneally into each mouse. Mice were composed
of 3 groups: group 1, ascorbate-restricted gulo KO mice (n=6);
group 2, ascorbate-supplemented gulo KO mice (n=6); and group 3,
wild-type mice (n=6). Mice were continued on their respective diets
for another 2 weeks. The mice were then sacrificed, blood was drawn
for serum analysis and peritoneal cavities were exposed and
photographed. All procedures were conducted under protocols
approved by the Internal Animal Care and Use Committee (IACUC).
Study 2
After mice had been on their respective diets for 4
weeks, 5×105 4T1 breast cancer cells in 0.2 ml PBS were
injected into the mammary pad of mice. Mice were composed of 3
groups: group 1, ascorbate-restricted gulo KO mice (n=6), group 2,
ascorbate-supplemented gulo KO mice (n=6) and group 3, wild-type
mice (n=6). Mice were continued on their respective diets for
another 2 weeks after injection. The mice were then sacrificed,
blood was drawn for serum analysis and their tumors were measured,
excised, weighed, photographed and processed for histology and
immunohistochemistry. All procedures were conducted under protocols
approved by the IACUC.
Metastasis grading
Mice in all groups injected IP with B16FO melanoma
cells were evaluated for metastasis. They were graded based on the
following scale: 0, none (only primary tumor, no secondary); 1,
mild (sporadic and small metastases different from primary
inoculation site); 2, moderate (obvious disseminated multiple
secondary metastases); 3, severe (large, obvious disseminated
multiple secondary metastases); and 4, advanced (large, obvious
disseminated multiple secondary metastases with adjacent
destruction and boundary loss of organs).
Serum analysis
Serum was processed from whole blood and stored at
−80°C until analyzed. Cytokine analyses, including interleukin
(IL)-6 and vascular endothelial growth factor (VEGF) were run in
duplicates by Procarta® Cytokine Assay Service using
Procarta® Cytokine kit mouse 8-plex at Affymetrix
(Panomics, Santa Clara, CA). Serum ascorbate analysis was performed
using the Ferric Reducing Ascorbate (FRASC) Assay kit form
Biovision (Mountain View, CA).
Immunohistochemistry
Tumors were placed in a formalin cassette and sent
to IDEXX Pathology (Sacramento, CA, USA) and HistoTox Labs Inc.
(Boulder, CO, USA) for analyses. Formalin fixed samples of tumors
were trimmed, processed, blocked, sectioned and stained with
H&E and EVG stains, and evaluated microscopically by IDEXX
Pathology. Immunohistochemistry of tumor sections was conducted by
HistoTox Labs Inc. and included TUNEL stain, collagen I and IV,
matrix metalloproteinase (MMP)-2 and MMP-9, Ki67, fibronectin and
bcl-2.
Statistical analysis
The results are expressed as means ± SD, as
indicated in the results, for the groups. Data were analyzed by
independent sample t-test and Pearson’s correlation coefficient
using MedCalc Software (Mariakerke, Belgium).
Results
Effect of dietary ascorbate on
metastasis in mice injected with B16FO melanoma cells
Ascorbate-supplemented gulo KO mice demonstrated
profound and significant reduction in tumor metastasis than did the
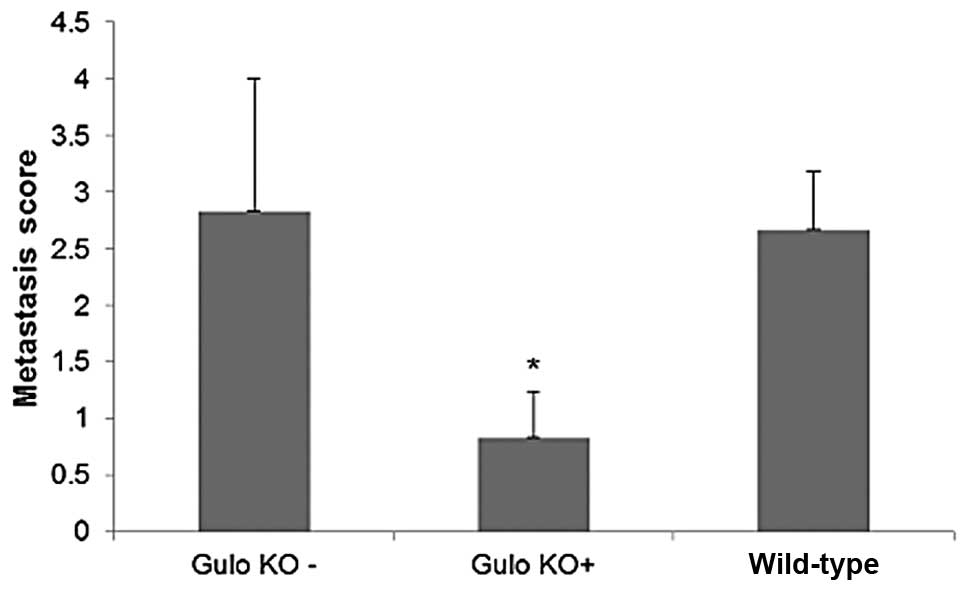
gulo KO mice on the control diet. The tumor grade was reduced by
71% (p=0.005) with ascorbate supplementation, as shown in Fig. 1. Intraperitoneal metastasis was
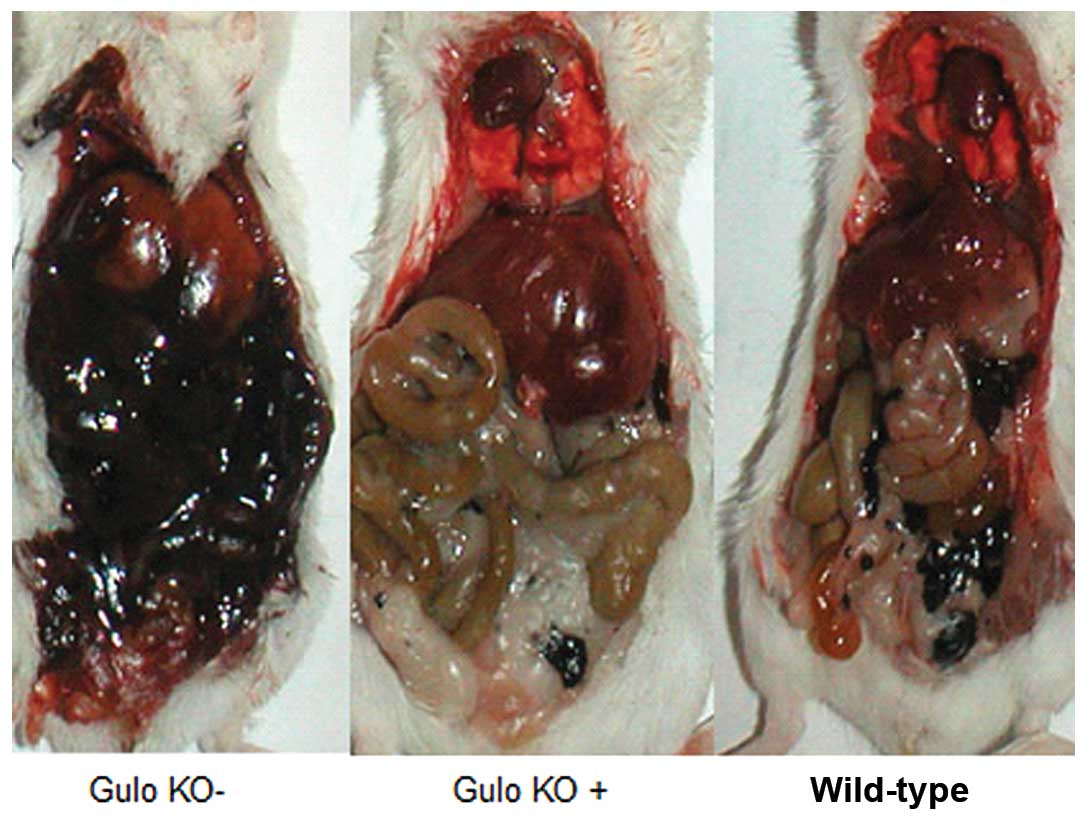
extensive in the ascorbate depleted gulo KO mice in contrast to the
ascorbate supplemented gulo KO mice and the wild-type mice, as
shown in Fig. 2. A significant
negative correlation (r=−0.693, p=0.026) was found between pooled
metastasis grade of gulo KO mice and serum ascorbate levels.
Effect of dietary ascorbate on tumor
growth in mice injected in the mammary pad with breast 4T1
cells
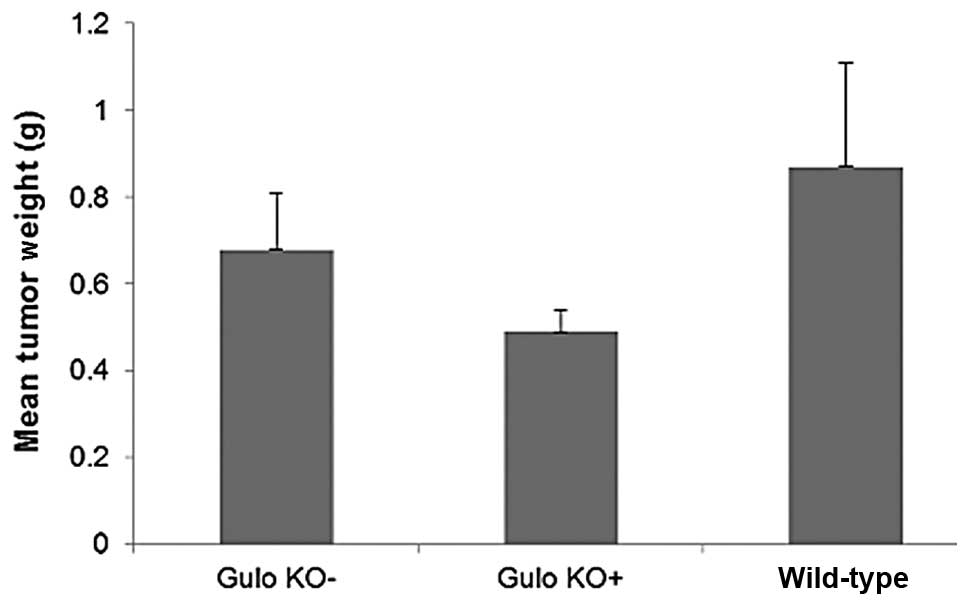
The mean tumor weight in ascorbate supplemented mice
(0.49±0.13 g) was reduced by 28%, compared to tumor weight in
ascorbate-deprived gulo KO mice (0.68±0.33 g), as shown in Fig. 3, but the difference did not reach
statistical significance. The mean tumor weight of wild-type mice
(0.87±0.35 g) was higher than that of the gulo KO mice. Mean tumor
dimension for scorbutic mice (137 mm2) was greater than
for supplemented (93.3 mm2) and wild-type (93.3
mm2) mice, but the difference did not reach statistical
significance.
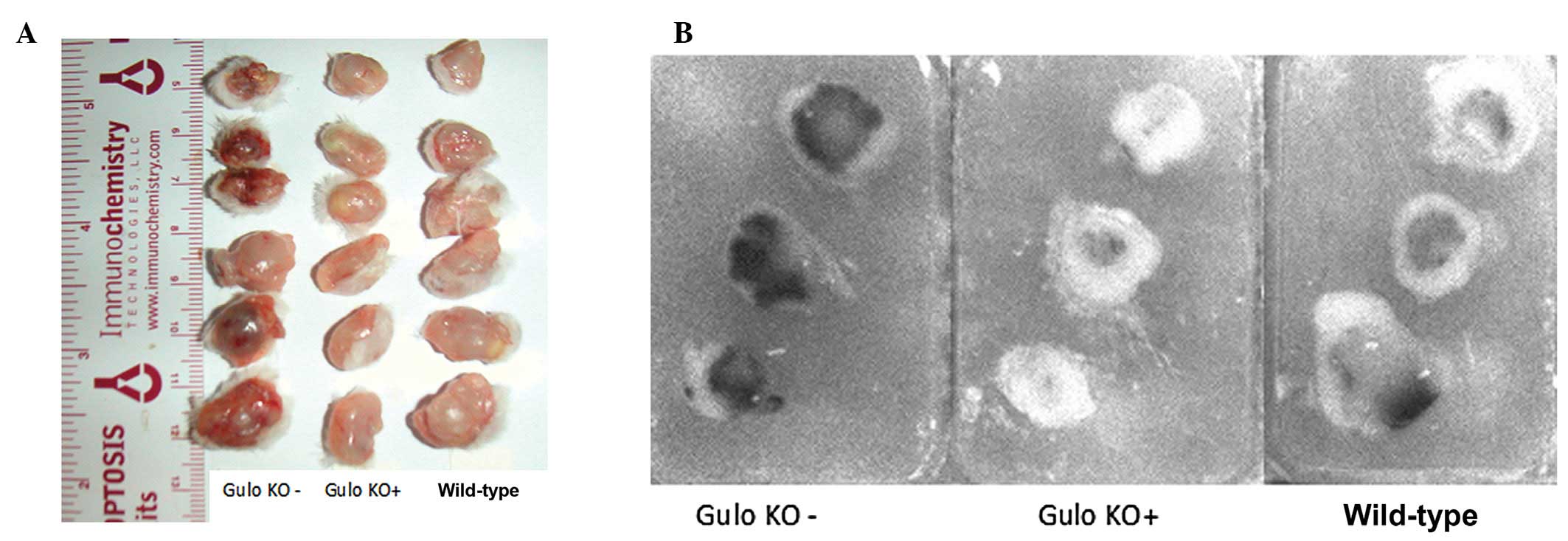
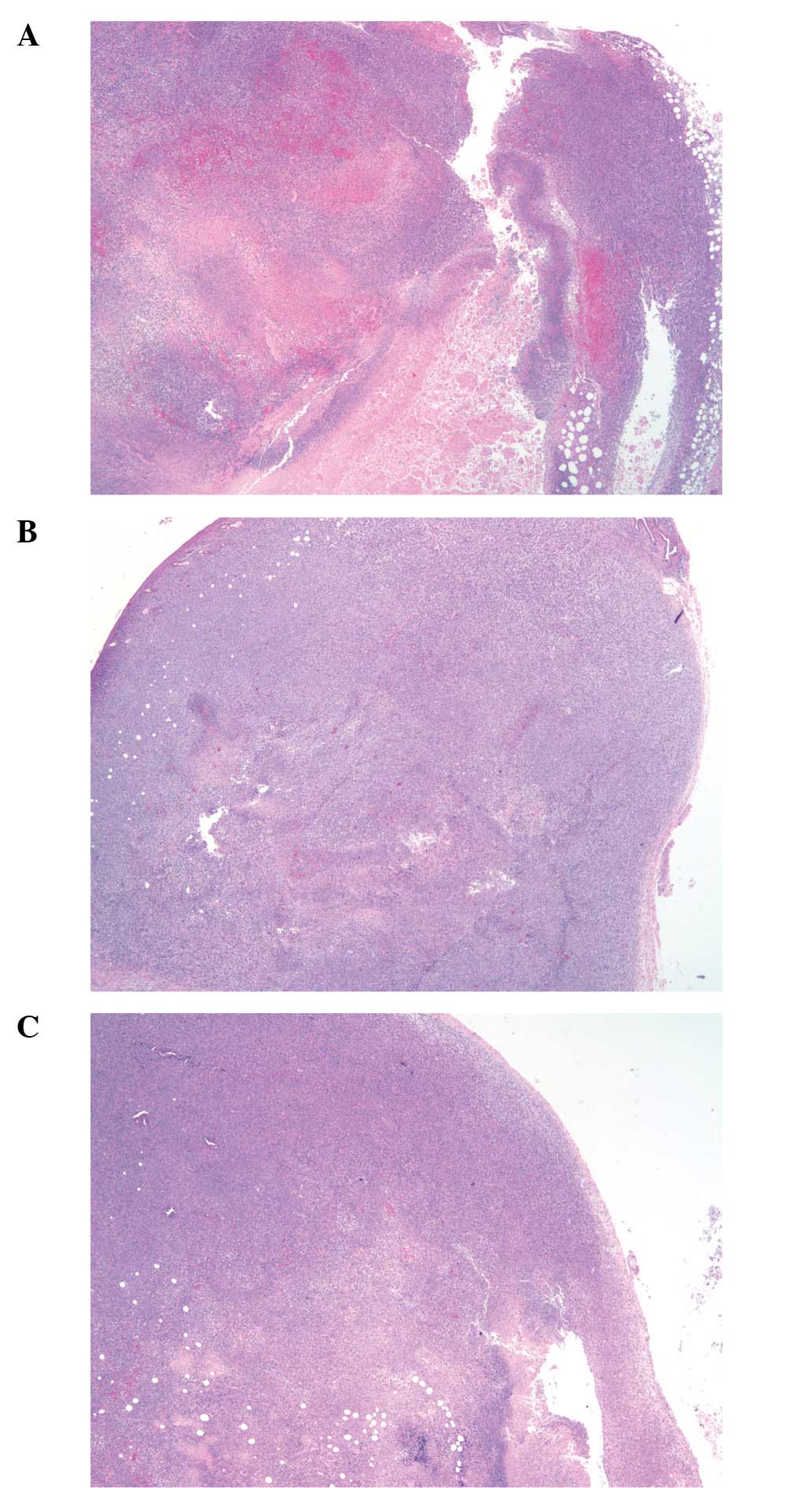
As shown in the gross tumor photographs and paraffin
sections of representative tumors from the gulo KO groups (Figs. 4 and 5), 4T1 tumors from the scorbutic groups
had large dark cores in contrast to the supplemented group tumors
which had smaller fainter colored cores. Tumors from wild-type mice
showed an intermediate appearance. The visual difference coincided
with increased core necrotic areas, which in some cases extended to
and breached through the tumor surface.
Effect of vitamin C supplementation on
tumor histology: 4T1 study: Immunohistochemistry
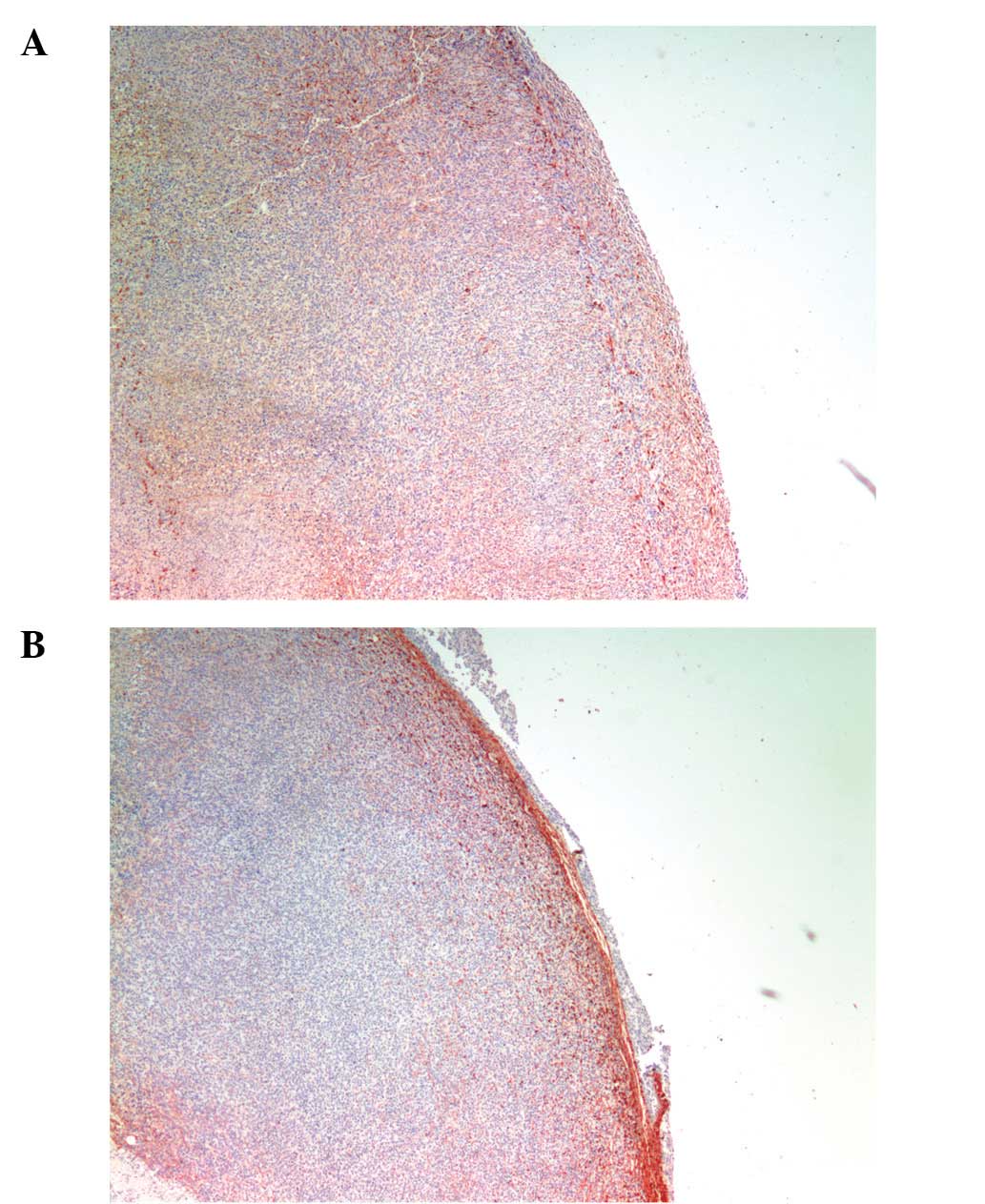
Collagen I and IV
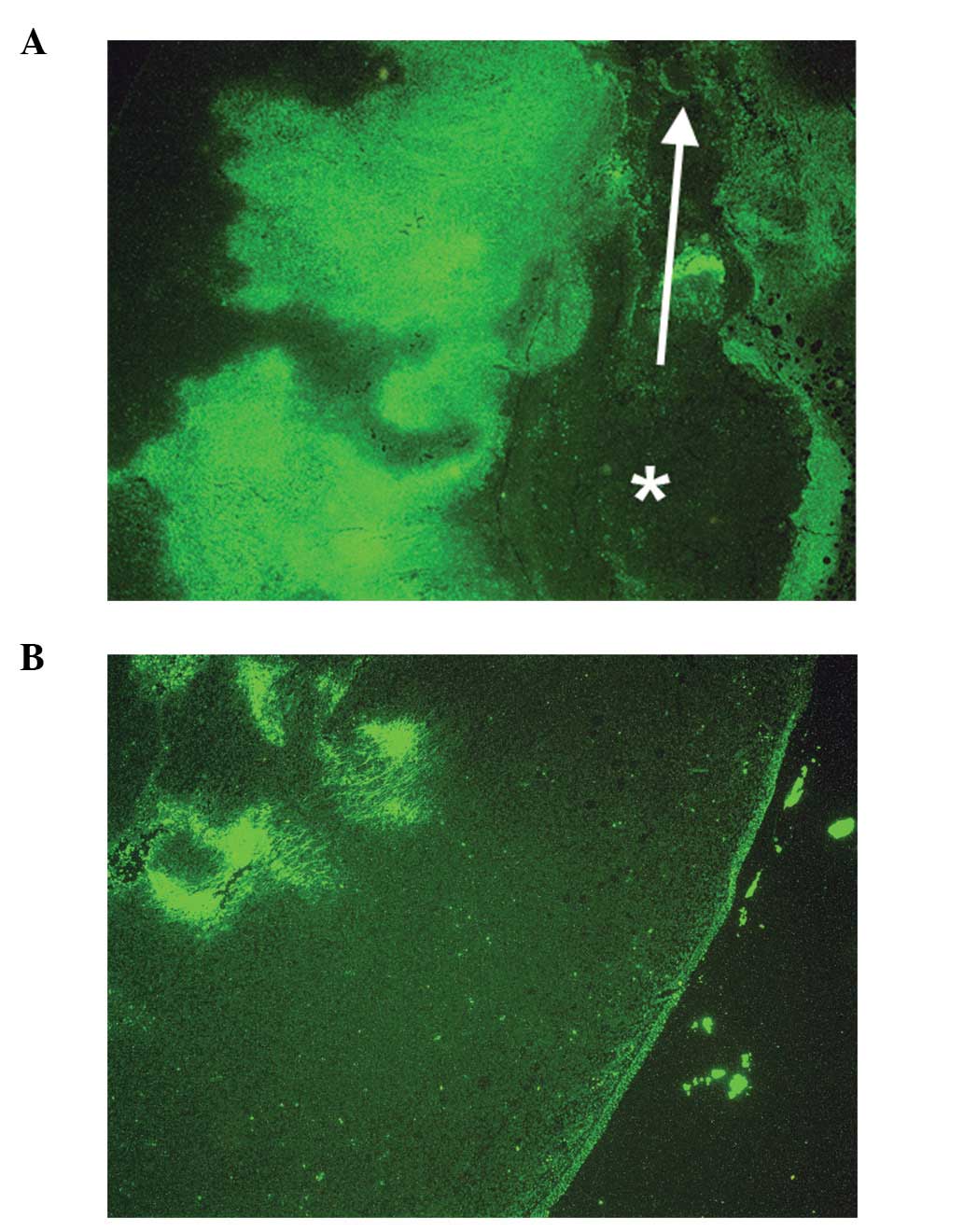
Tumors from ascorbate-supplemented mice (Figs. 6B and 7B) showed diffuse staining of collagen IV
around the core and a robust collagen I tumor capsule. Scorbutic
mice tumors (Figs. 6A and 7A) demonstrated a strong staining of
collagen IV around the core, within the tumor, and weaker,
disorganized, or missing collagen I tumor capsule. Scorbutic tumors
exhibited an ‘inside-out’ collagen IV expression pattern
internally, whereas vitamin C supplemented tumors were externally
encapsulated with a distinct, dense collagen I capsule.
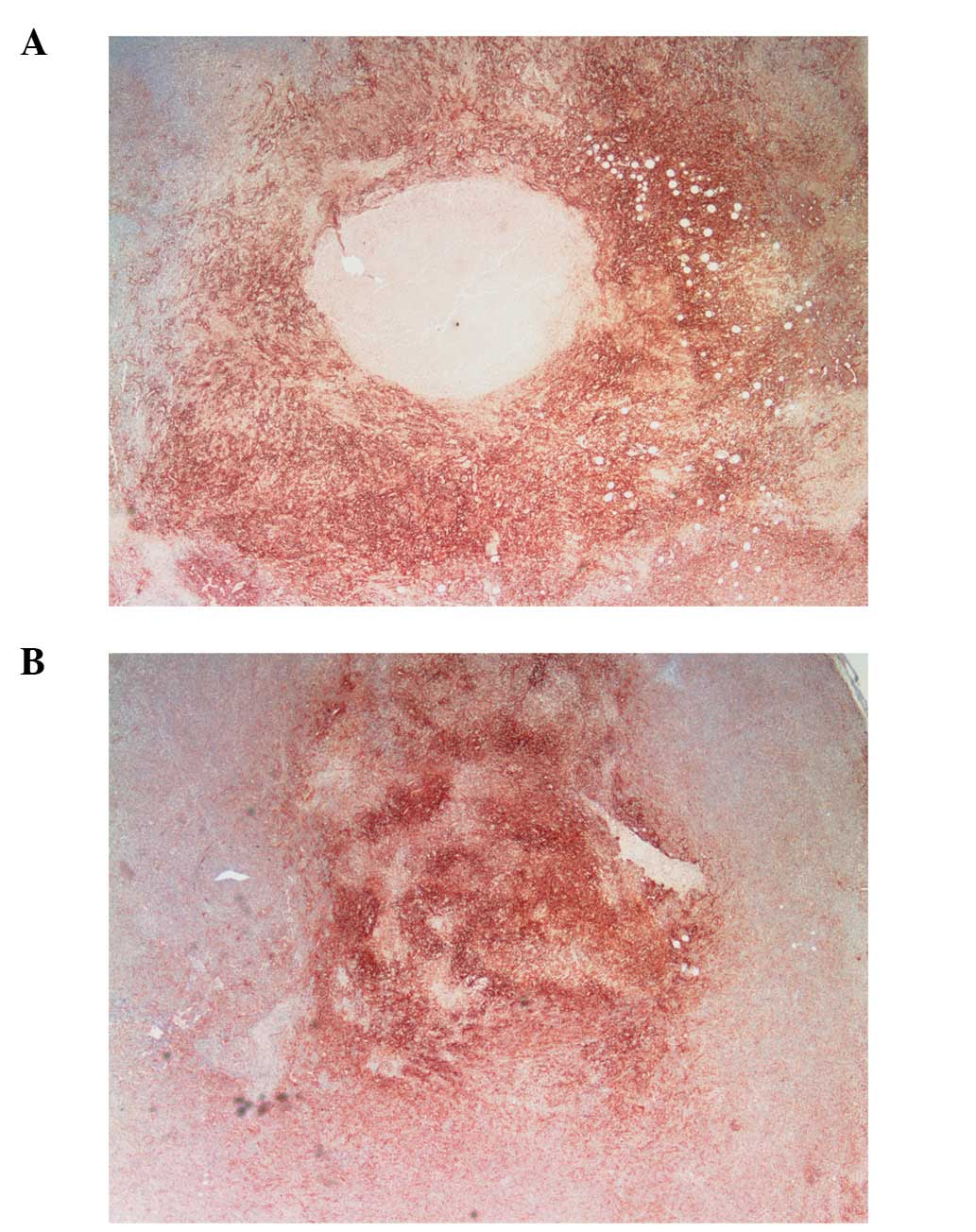
MMP-2 and MMP-9
No significant difference in MMP-2, which was
pervasive in both groups, was noted between scorbutic and
ascorbate-supplemented gulo KO mice, although MMP-2 staining
appeared to be less in the supplemented group (figure not shown).
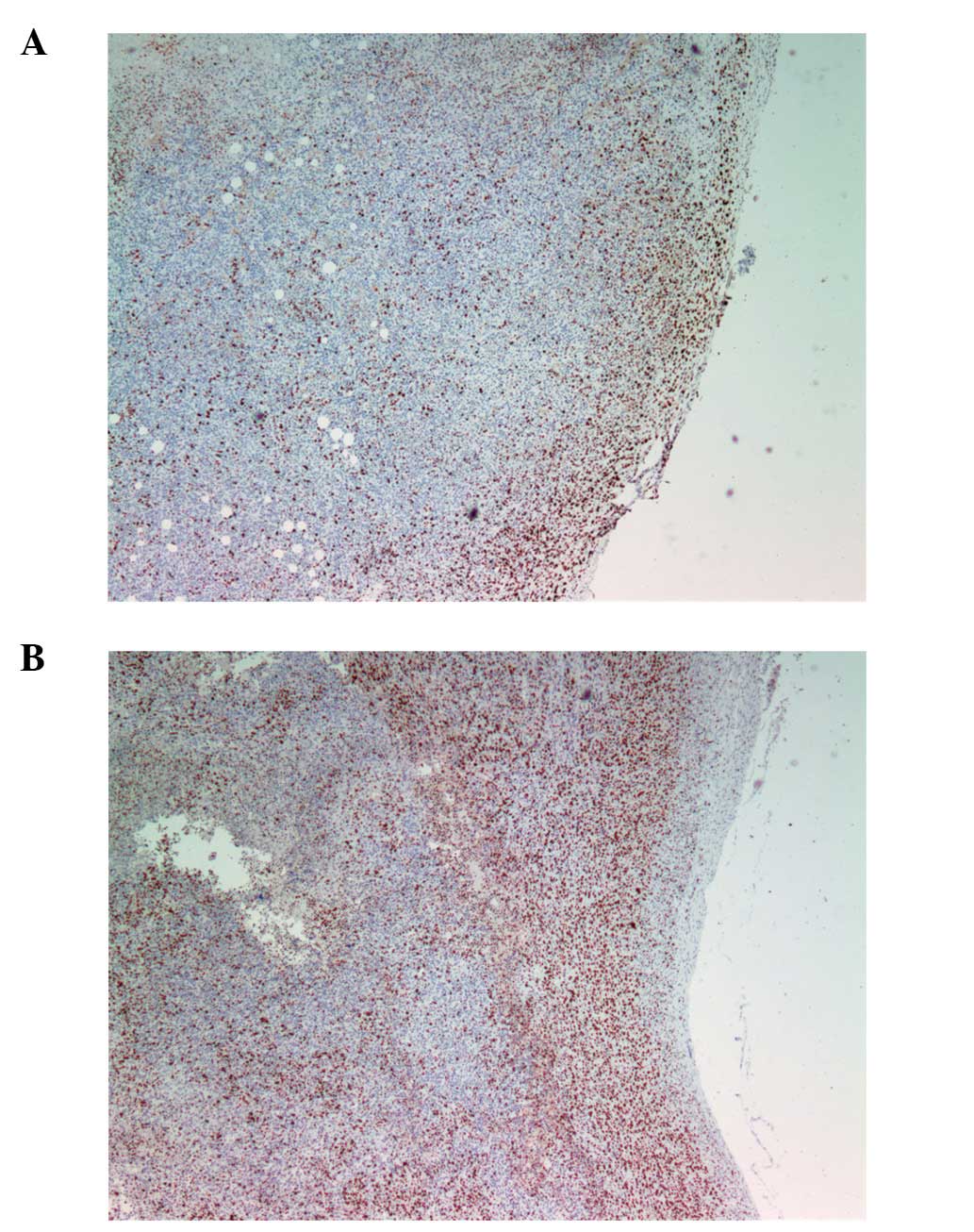
However, MMP-9 staining, which had a regional pattern directly
overlapping necrotic areas and surrounded by apoptotic areas,
differed significantly between the gulo KO groups. The scorbutic
group tumors (Fig. 8A)
characteristically showed an irregular pattern of necrosis in
tandem with MMP-9 expression that coincided with breaches in the
tumor, creating conduits and channels for viable tumor cells to
escape. In contrast, the supplemented tumors (Fig. 8B) exhibited more confined areas of
necrosis/apoptosis, with cancer cells in a more or less static,
organized spatial organization around the core with no breaches
from the core to the outside of the tumor.
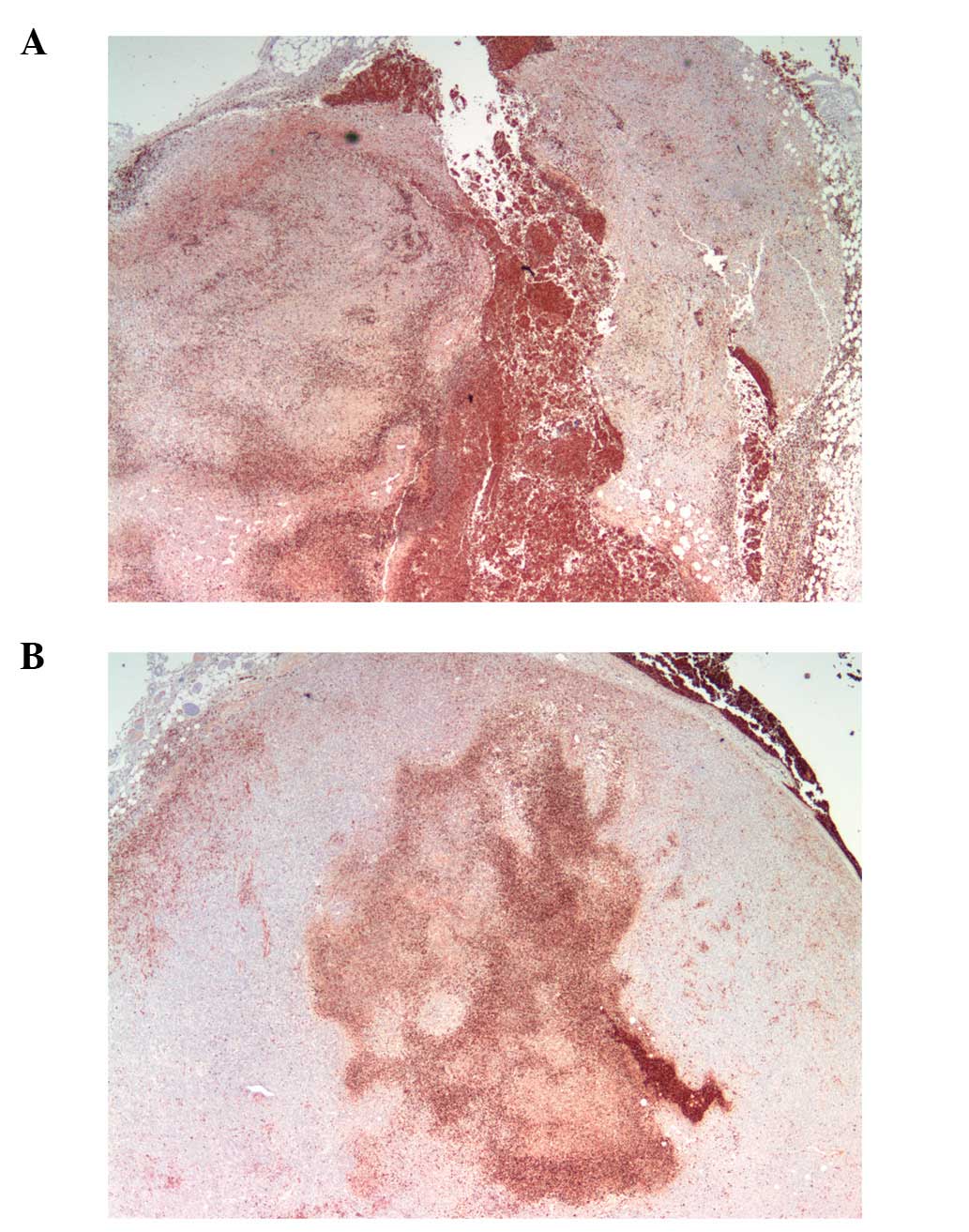
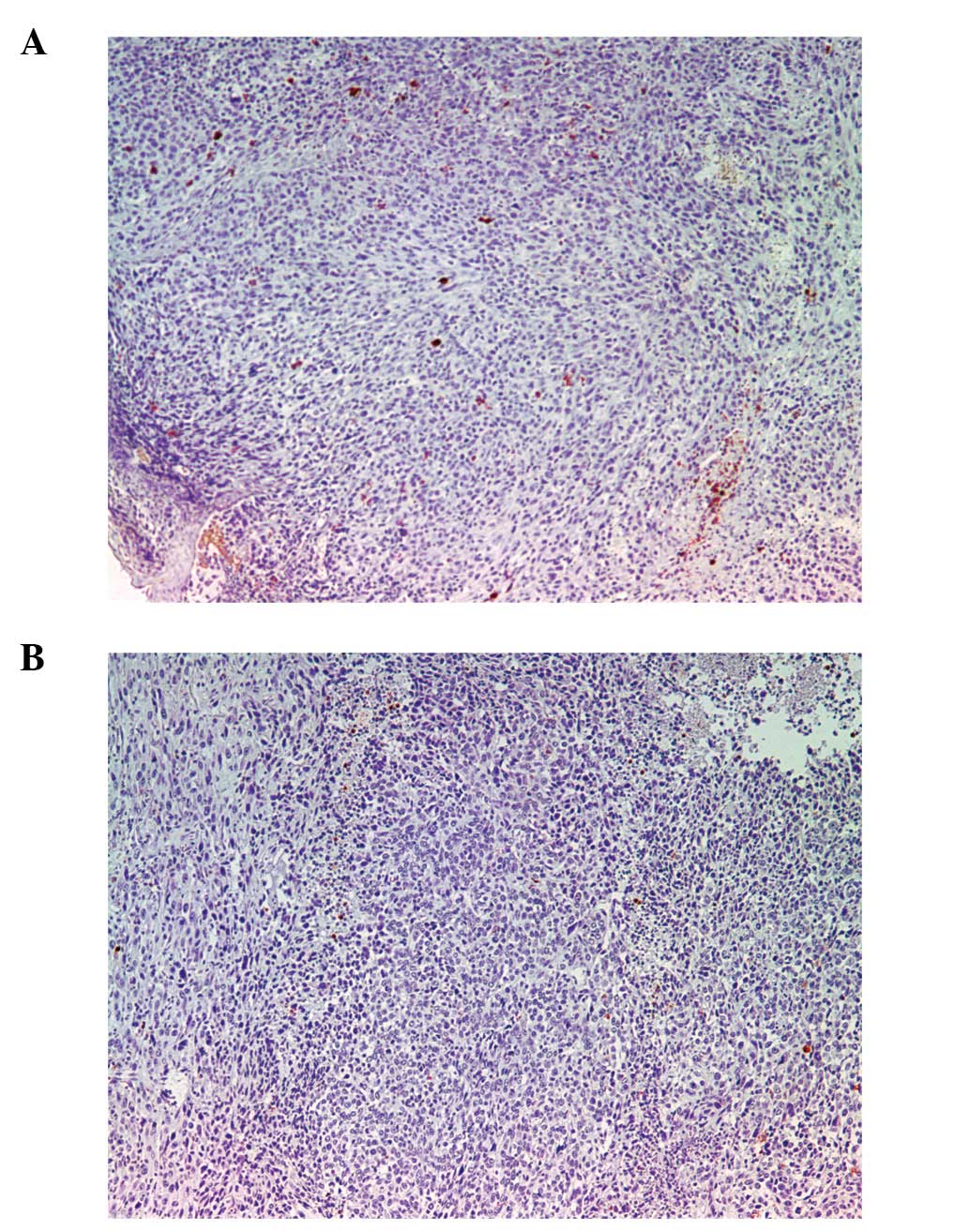
Apoptosis: TUNEL stain
Apoptotic areas and level of apoptosis surrounded
and coincided with MMP-9 area and intensity. Scorbutic group tumors
(Fig. 9A) showed a pattern of
apoptotic cells directly adjacent to MMP-9 staining, with necrotic
areas forming patterns of breaches/conduits in the tumor from the
core to the surface of the tumor. In contrast, apoptotic areas in
the supplemented group tumors (Fig.
9B) were more centralized and did not breach the surface of the
tumor.
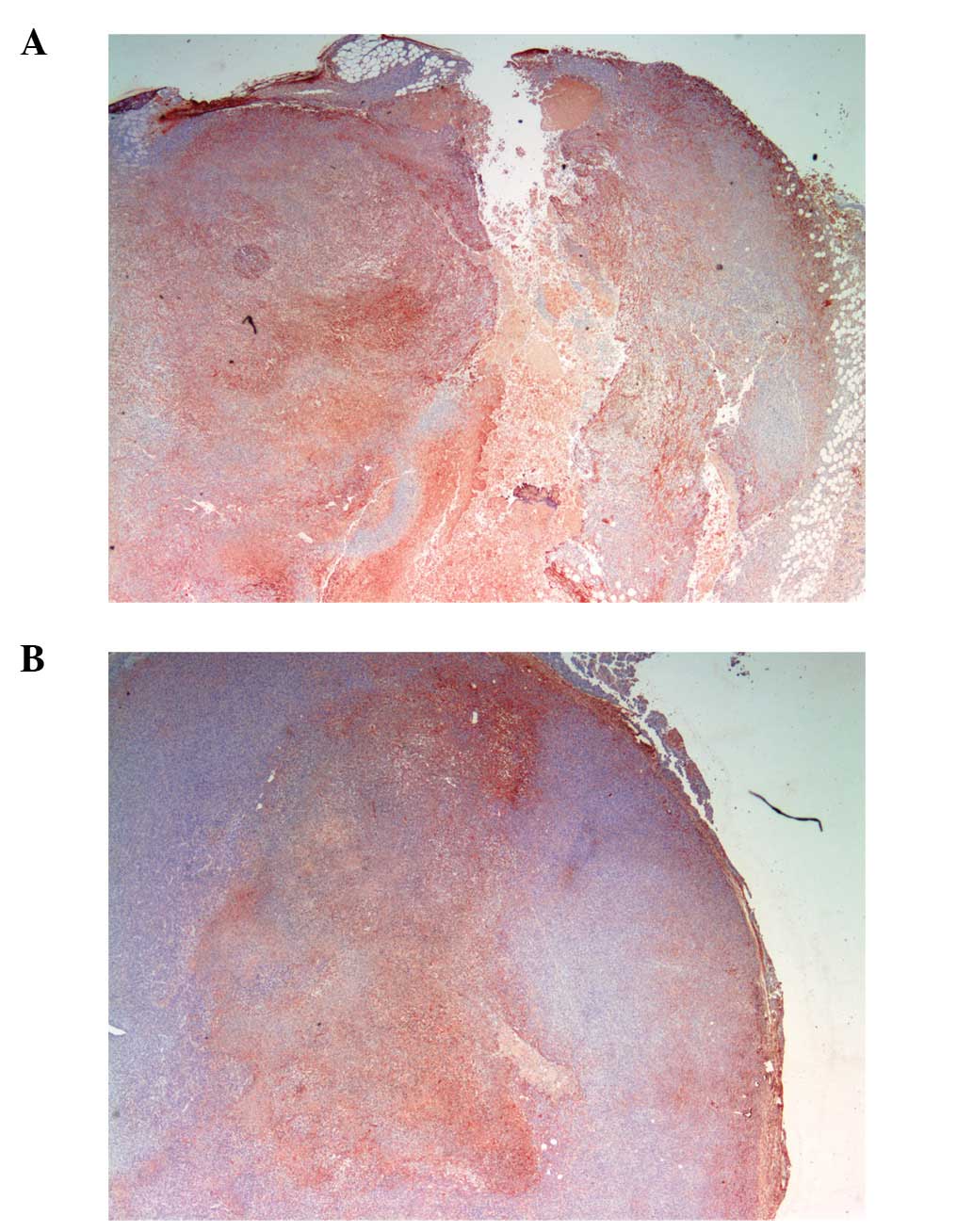
Proliferation: Ki67
The proliferation marker Ki67 showed similar
intensity and frequency of staining between the groups except that
proliferating cells were not confined by a capsule barrier in the
scorbutic group and extended to the surface of the tumor (Fig. 10A). In contrast, Ki67 cells were
confined and restrained by a capsule barrier in the ascorbate
supplemented group (Fig.
10B).
Bcl-2
More sporadic, punctate staining of Bcl-2, a
pro-survival, anti-apoptotic protein, was observed in ascorbate
supplemented and wild-type mice groups compared to scorbutic group
tumors, but the difference was not significant. Though present, the
low frequency of Bcl-2 expression would not be expected to
significantly contribute to tumor regression or progression. See
Fig. 11 for Bcl-2 levels in gulo
KO mice groups.
Fibronectin
Fibronectin staining in the scorbutic tumors was
more centralized in contrast to the more peripheral staining in the
ascorbate supplemented tumors, as shown in Fig. 12.
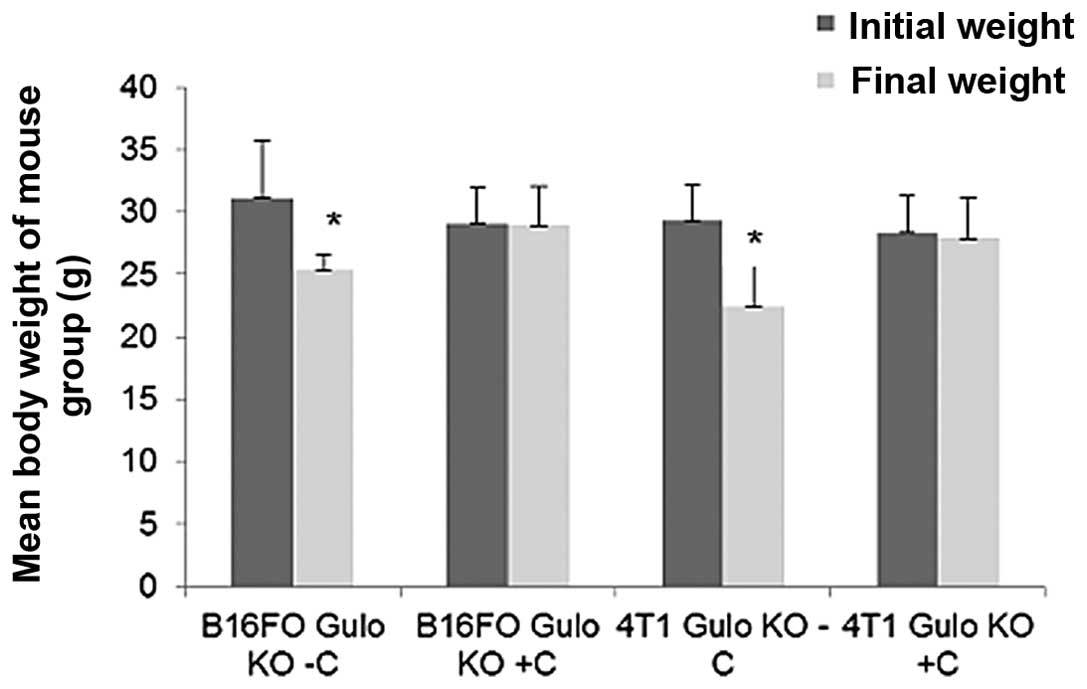
Effect of dietary ascorbate on weight
of mice
The mean weight of mice in each group did not
significantly differ at onset. However, the mean weight of
ascorbate restricted gulo KO mice significantly decreased with
time, showing a 19% (p=0.02) and 24% (p=0.003) decrease in weight
at 6.5 weeks post injection compared to the initial mean weight
prior to injection in the B16FO and 4T1 studies, respectively, as
shown in Fig. 13.
Ascorbate-supplemented gulo KO mice and wild-type mice maintained
their weight.
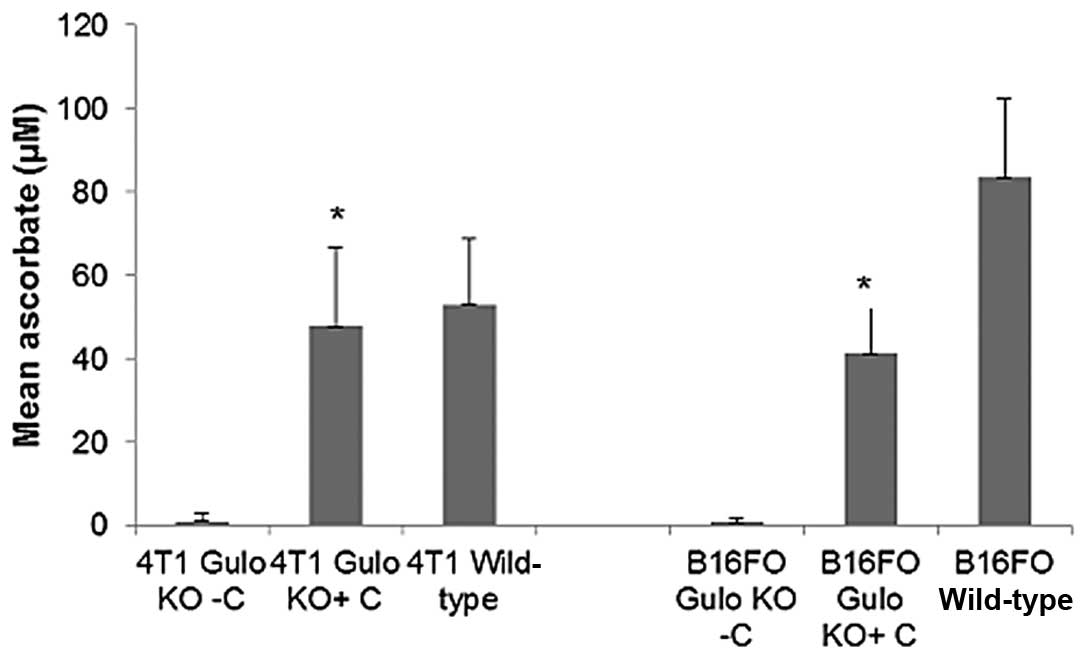
Effect of ascorbate supplementation on
serum ascorbate levels in gulo KO mice
As expected, ascorbate-supplemented gulo KO mice in
both studies were found to have profoundly higher ascorbate levels
than did the restricted gulo KO mice (Fig. 14). In the B16FO study, mean serum
ascorbate level in ascorbate restricted mice (0.80±1.2 μM)
was 2% (p<0.001) of the mean ascorbate level in supplemented
mice (41.2±11.6 μM). Wild-type mice showed significantly
higher ascorbate levels than either of the gulo KO mice (83.5±18.7
μM). The mean ascorbate level in ascorbate supplemented mice
was 49% of that in wild-type mice. In the 4T1 study, mean serum
ascorbate level in ascorbate restricted mice (1.1±2.01 μM)
was 2% (p<0.001) of the mean ascorbate level in supplemented
mice (47.9±18.9 μM). Wild-type mice showed slightly higher
ascorbate levels (53.1±15.7 μM) than the supplemented gulo
KO mice, but the difference did not reach statistical
significance.
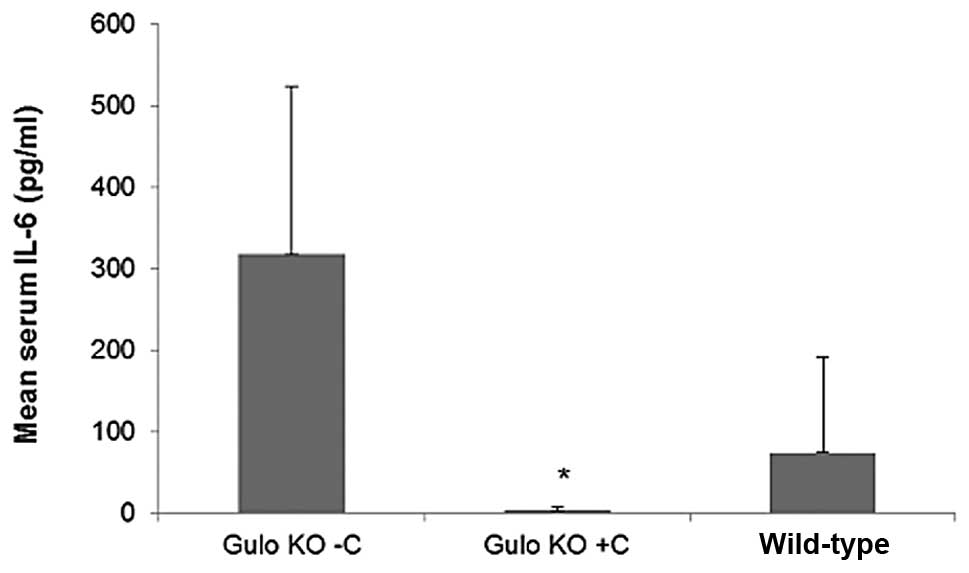
Effect of ascorbate supplementation on
IL-6
In both studies, serum inflammatory cytokine IL-6
was substantially higher in gulo KO mice deprived of ascorbate than
in gulo KO mice supplemented with ascorbate. In the B16FO IP
injection study, mean serum IL-6 level in the gulo KO scorbutic
mouse group was 317.8±205.4 pg/ml compared to 3.4±4.75 in the
ascorbate supplemented gulo KO group (99% decrease, p= 0.01), as
shown in Fig. 15. In regards to
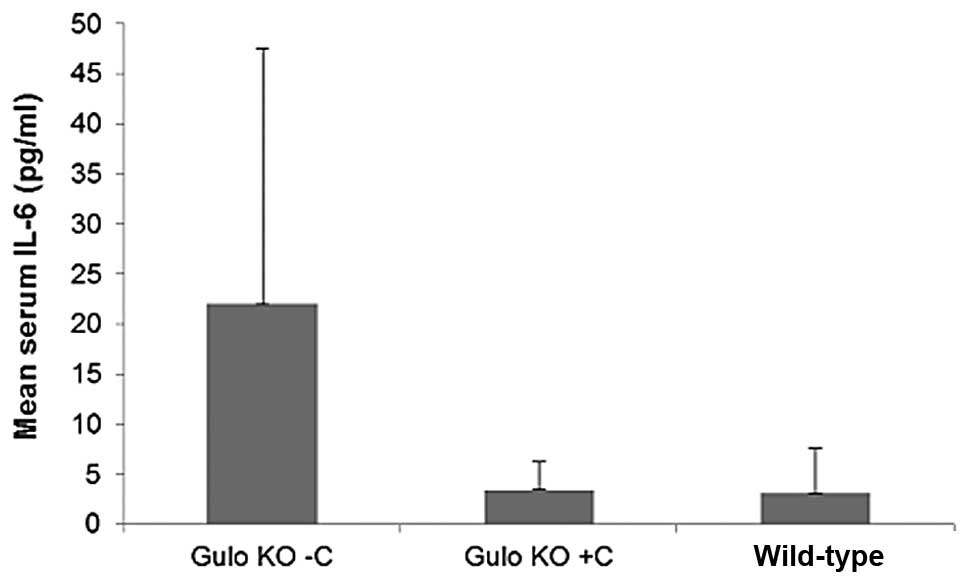
the 4T1 study, ascorbate supplementation resulted in an 85%
decrease from the level in the restricted gulo KO group, but the
difference did not reach statistical significance, as shown in
Fig. 16. Mean IL-6 levels per
mouse were 3.40±2.9 and 22.0±25.64 pg/ml in the ascorbate
supplemented and deprived gulo KO mice, respectively. A significant
negative correlation was found between pooled ascorbate and IL-6
serum levels in mice (coefficient r=−0.7211, p=0.0024 in the B16FO
study and r=−0.6062, p=0.0059 in the 4T1 study).
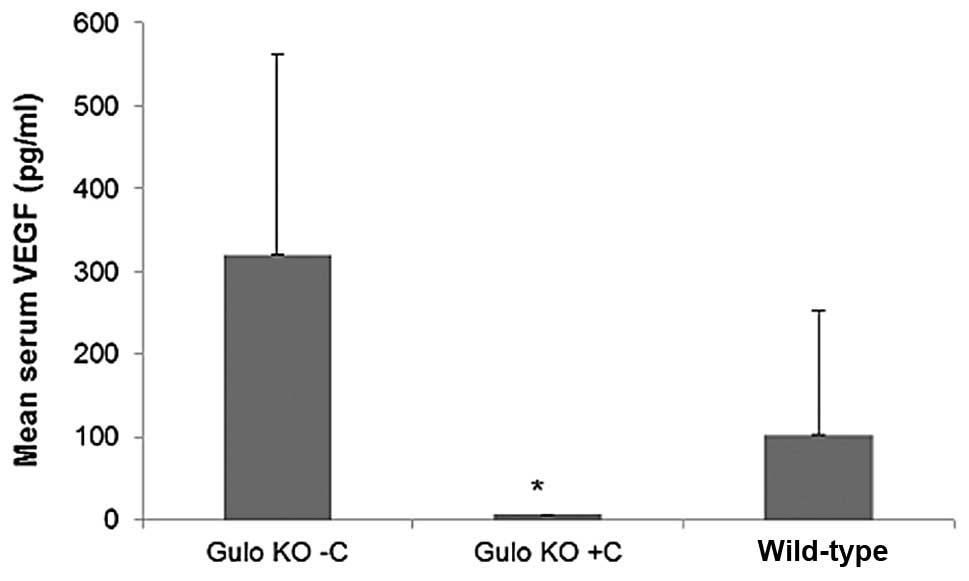
Effect of ascorbate supplementation on
VEGF
VEGF was substantially higher in gulo KO mice
deprived of ascorbate than in gulo KO mice supplemented with
ascorbate. In the B16FO IP injection study, mean serum VEGF level
in the gulo KO scorbutic mouse group was 321±241 pg/ml compared to
6.8±0.5 in the ascorbate supplemented gulo KO group (98% decrease,
p=0.02), as shown in Fig. 17.
Discussion
Numerous clinical studies have noted that cancer
patients exhibit abnormally low plasma ascorbate levels secondary
to the disease and/or treatment (6,7,11,12).
A significant correlation has been demonstrated between deficient
plasma ascorbate levels in cancer patients and decreased survival,
as well as increased expression of inflammatory factors (11). Tumor aggressiveness has also been
correlated to low ascorbate content (12). Ascorbate has been implicated in
host resistance to neoplasia, at both the stromal and systemic
levels.
Stromal resistance is dependent upon the host’s
ability to encapsulate the neoplastic cells by forming a
practically impenetrable barrier of dense fibrous tissue (5,13).
Highly invasive tumors are associated with a scanty, poorly defined
collagenous barrier. In contrast, collagenous barriers are more
defined in tumors of moderate rapidity of growth and very abundant
in slow-growing ‘contained’ atrophic scirrhous tumors (13). Degradation of adjacent matrix in
the vicinity of invading neoplastic cells is dependent upon
continual release of hyaluronidases, proteases and collagenases
from the invading cells (4,13,14).
The activity of matrix metalloproteinases (MMPs), especially MMP-9,
on the degradation of the extracellular matrix plays a critical
role in the formation of tumors and metastasis and high MMP-9
levels have been found to correlate with the aggressiveness of
cancers, as exemplified by breast cancer and melanoma (15–17).
In our study, ascorbate supplementation of gulo KO
mice injected IP with B16FO cells, demonstrated profound reduction
in metastasis compared to control. B16FO metastasis was reduced by
71% in supplemented mice compared to control mice. While metastasis
was not observed in the gulo KO mice that were injected with breast
cancer 4T1 cells into the mammary pads, a distinct difference was
observed in the size and tumor composition, especially the level of
protein expression and spatial expression of the proteins between
4T1 tumors harvested from ascorbate supplemented and restricted
mice. Metastasis of 4T1 cells was not expected to occur during the
term of this study since tumors were harvested 2 weeks after
introduction of cells while metastasis has been reported to not be
detectable until the second growth phase 5–6 weeks after injection
of cells (9).
In the 4T1 study, ascorbate supplementation of gulo
KO mice resulted in reduction in the mean tumor weight compared to
that in ascorbate-deprived gulo KO mice. Furthermore,
immunohistochemical staining of tumors confirmed that this
observation was not natural statistical variation inherent to in
vivo systems, but an authentic biochemical difference in
response to the presence or absence of ascorbate in the host
animal. Gulo KO mice deprived of ascorbate developed large tumors
with dark cores, showing more necrosis, and poorly defined borders.
In contrast, gulo KO mice supplemented with ascorbate hosted
smaller tumors with smaller, lighter cores, less necrosis and
enhanced collagen encapsulation, signifying less metastatic
potential. Apoptotic patterns from center to periphery of tumor in
the scorbutic gulo KO mice, in contrast to a uniform pattern in the
ascorbate supplemented gulo KO mice, suggests a more invasive and
metastatic tumor structure from which cancer cells could more
easily escape from the tumor mass of scorbutic mice into adjacent
circulation. Furthermore, the necrotic areas that extended from the
cores to breach the tumor surface in scorbutic mice were surrounded
with TUNEL apoptosis staining and directly coincided with MMP-9
staining areas. This implies that irregular, asymmetric apoptosis
and/or necrosis in tandem with MMP-9 expression create conduits and
channels through which viable tumor cells can escape. In contrast
to the irregular pattern of necrosis and breaches in the tumors
found in scorbutic mice, tumors from the supplemented mice showed
more confined areas of necrosis and apoptosis around the core with
no breaches from the core to the outside of the tumor.
Bonfil et al(18) in studying the role of intratumoral
necrosis in cell detachment and metastasis using non-metastatic
murine mammary adenocarcinma M3 and its metastatic variant MM3
reported similar patterns to what we observed in mammary 4T1 tumors
from ascorbate supplemented versus scorbutic gulo KO mice. The
histological studies of Bonfil et al revealed a central
necrosis limited by an uninterrupted peripheral ring of
well-preserved cells in M3, in contrast to alternated necrotic and
non-necrotic areas in MM3. They concluded that the distribution of
necrosis within the primary tumor was responsible in part for the
development of metastases (18).
Furthermore, in a subsequent study, they noted that tumor necrosis
was an important source of gelatinase/type IV collagenase, mainly
in its 92 kDa form, and thus played a major role in tumor invasion
(19).
In addition to promoting progression of cancer,
elevated pro-inflammatory cytokine levels have been associated with
a variety of pathologies, such as fatigue, depression and cachexia
(20–23). Levels of serum cytokines, such as
IL-6, IL-1β, IL-1α, IL-8, IL-12p40, IL-13, GM-CSF, monocyte
chemoattractant protein (MCP)-1, macrophage inflammatory protein
(MP)-1α, MP-1β, IFNα, tumor necrosis factor (TNF)-α, epidermal
growth factor, VEGF and TNF receptor II are reported to be
significantly higher in patients with breast cancer and with
resected high-risk melanoma than in healthy controls (24,25).
These factors promote cancer growth and stimulate angiogenesis,
which lead to cancer growth and metastasis in these and other
cancers (26,27). Clinical studies have shown elevated
IL-6 levels in breast cancer to be associated with poor breast
cancer prognosis (28–30) and to increase with tumor grade and
number of metastatic sites (29).
Similarly, significantly increased levels of serum IL-6 in
malignant metastatic melanoma patients have been found to be
correlated to the tumor burden in these patients (31–33).
Ascorbate supplemented gulo KO mice had profoundly reduced levels
of IL-6 compared to ascorbate-deprived gulo KO mice in both the
melanoma B16FO and breast cancer 4T1 studies. Furthermore, negative
correlations were found between IL-6 secretion and ascorbate levels
in both studies from pooled data. The pro-angiogenic factor VEGF,
which is critical for both primary tumor growth and metastasis, was
also found to be significantly higher in scorbutic mice challenged
with melanoma B16FO cells than in ascorbate-supplemented mice,
which was reflected in the higher metastasis grade of the scorbutic
group. Of interest, weight loss occurred in the gulo KO mice
deprived of ascorbate but not in those supplemented with ascorbate.
The weight loss observed in cancer patients, secondary to the
inflammation-associated cachexia, mirrors the weight loss observed
in the scorbutic mice with high IL-6 levels.
The comparative ascorbate serum levels in the gulo
KO groups in both studies support the claim that the beneficial
effects observed in the ascorbate-supplemented mice were due to
significantly increased serum ascorbate levels. Oral
supplementation of vitamin C to levels included in diets for
mammals that do not generate endogenous ascorbate, such as guinea
pigs, and in the drinking water did not achieve pharmacological
levels of ascorbate in the gulo KO mouse serum. Measured levels of
serum ascorbate from supplemented mice were within the normal
physiological range reported throughout the literature (34). Despite being 10-fold lower than
pharmacological or therapeutic concentrations, this normal
physiological level of serum ascorbate sustains a biochemical
environment less conducive to metastasis and growth of melanoma and
breast cancer, respectively. In contrast, the resultant reduced
host resistance and less effective response of scorbutic mice
encouraged metastasis and tumor growth to a degree that made normal
supplementation appear therapeutic in its activity. In both the
melanoma and breast cancer studies, ascorbate-depleted gulo KO mice
were found to have profoundly lower (2%) ascorbate levels than
found in ascorbate supplemented mice. Furthermore, a significant
negative correlation was found between pooled metastasis grade of
gulo KO mice and serum ascorbate levels in the B16FO study. A
previous mechanistic study showed that ascorbic acid reduced
leukemia HTLV-1 cell proliferation and induced apoptosis by the
modulation of p53, p21, Bcl-2 and Bax (35).
Mean ascorbate level was slightly higher in wild
mice compared to supplemented gulo KO mice in the 4T1 study and
significantly higher in the B16FO study. The higher ascorbate
levels in wild-type mice are probably due to the age difference
between gulo KO and wild-type mice. In addition, younger (3 months
old) Balb/c mice have been reported to grow significantly larger
tumors than older (9+ months old) mice (36). These findings would probably
explain the higher metastasis grade and larger tumor weight
observed with the wild-type mice compared to the supplemented gulo
KO mice. Though this was not the focus of the study, the use of
wild-type reference mice that were considerably younger (by 6
months) than the experimental set was a limitation. In a previous
study of B16FO growth/metastasis using wild-type and gulo KO groups
of mice both approximately 36–40 weeks of age, no statistical
difference was observed between tumor growth in wild-type and
supplemented gulo KO mice (37).
In conclusion, ascorbate supplementation resulted in
decreased metastasis elicited by melanoma IP challenge in gulo KO
mice and reduced tumor growth and enhanced encapsulation of tumors
resulting from breast cancer challenge. Tumor encapsulation is
critical in curbing invasion and metastasis of malignant cells. In
addition, ascorbate supplementation modulated inflammatory cytokine
secretion. Scurvy, which results from severe dietary lack of
ascorbate, exhibits generalized stromal changes identical to local
stromal changes observed in cancer in the immediate vicinity of
invading neoplastic cells (38).
Thus, ascorbate supplementation to cancer patients has been
proposed to reverse their scorbutic symptoms and treat the cancer
(13,39). The results of this study support
this proposal.
Acknowledgements
Special thanks to Earl Rainey for
animal colony maintenance. The research study was funded by Dr Rath
Health Foundation (Santa Clara, CA, USA), a non-profit
organization.
References
|
1.
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar
|
|
2.
|
Breastcancer.org: U.S. Breast Cancer
Statistics: http://www.breastcancer.org/symptoms/understand_bc/statistics.jsp
(accessed December 21, 2011).
|
|
3.
|
Ali SM, Harvey HA and Lipton A: Metastatic
breast cancer: overview of treatment. Clin Orthop. 414(Suppl):
132–137. 2003. View Article : Google Scholar
|
|
4.
|
Fidler IJ: Molecular biology of cancer:
invasion and metastasis. Cancer: Principles and Practice of
Oncology. De Vita VT, Hellman S and Rosenberg SA: 5th edition.
Lippincott-Raven; Philadelphia, PA: pp. 135–152. 1997
|
|
5.
|
Rath M and Pauling L: Plasmin-induced
proteolysis and the role of apoprotein(a), lysine and synthetic
analogs. Orthomol Med. 7:17–23. 1992.
|
|
6.
|
Nunez MC, De Apodaca O and Ruiz AL:
Ascorbic acid in the plasma and blood cells of women with breast
cancer. The effect of consumption of food with an elevated content
of this vitamin. Nutr Hosp. 10:368–372. 1995.PubMed/NCBI
|
|
7.
|
Anthony HM and Schorah CJ: Severe
hypovitaminosis C in lung-cancer patients: the utilization of
vitamin C in surgical repair and lymphocyte-related host
resistance. Br J Cancer. 46:354–367. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Pulaski BA and Ostrand-Rosenberg S: Mouse
4T1 breast tumor model. Curr Protoc Immunol Chapter. 20:Unit
20.2.2001. View Article : Google Scholar
|
|
9.
|
Tao K, Fang M, Alroy J and Sahagian GG:
Imagable 4T1 model for the study of late stage breast cancer. BMC
Cancer. 8:2282008. View Article : Google Scholar : PubMed/NCBI
|
|
10.
|
Hart IR and Fidler IJ: Role of organ
selectivity in the determination of metastatic patterns of B16
melanoma. Cancer Res. 40:2281–2297. 1980.PubMed/NCBI
|
|
11.
|
Mayland CR, Bennett MI and Allan K:
Vitamin C deficiency in cancer patients. Palliat Med. 19:17–20.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Kuiper C, Molenaar IG, Dachs GU, Currie
MJ, Sykes PH and Vissers MC: Low ascorbate levels are associated
with increased hypoxia-inducible factor-1 activity and an
aggressive tumor phenotype in endometrial cancer. Cancer Res.
70:5749–5758. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Cameron E, Pauling L and Leibovitz B:
Ascorbic acid and cancer: a review. Cancer Res. 39:663–681.
1979.
|
|
14.
|
Stetler-Stevenson WG, Hewitt R and
Corcoran M: Matrix metalloproteinases and tumor invasion from
correlation and causality to the clinic. Semin Cancer Biol.
7:147–154. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Bachmeier BE, Nerlich AG, Lichtinghagen R
and Sommerhoff CP: Matrix metalloproteinases (MMPs) in breast
cancer cell lines of different tumorigenicity. Anticancer Res.
6A:3821–3828. 2001.PubMed/NCBI
|
|
16.
|
Pellikainen JM, Ropponen KM, Kataja VV,
Kellokoski JK, Eskelinen MJ and Kosma VM: Expression of matrix
metalloproteinase (MMP)-2 and MMP-9 in breast cancer with a special
reference to activator protein-2, HER-2, and prognosis. Clin Cancer
Res. 10:7621–7628. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Nikkola J, Vihinen P, Vuorista MS,
Kellokumpu-Lehtinen P, Kähäri VM and Pyrhönen S: High serum levels
of matrix metalloproteinase-9 and matrix metallopreoteinase-1 are
associated with rapid progression in patients with metastatic
melanoma. Clin Cancer Res. 11:5158–5166. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Bonfil RD, Bustuabad OD, Ruggiero RA,
Meiss RP and Pasqualini CD: Tumor necrosis can facilitate the
appearance of metastases. Clin Exp Metastasis. 6:121–129. 1988.
View Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Bonfil RD, Medina PA, Gómez DE, Farías E,
Lazarowski A, Lucero Gritti MF, Meiss RP and Bustuabad OD:
Expression of gelatinase/type IV collagenase in tumor necrosis
correlates with cell detachment and tumor invasion. Clin Exp
Metastasis. 10:211–220. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Argilés JM, Busquets S, Toledo M and
López-Soriano FJ: The role of cytokines in cancer cachexia. Curr
Opin Support Palliat Care. 3:263–268. 2009.
|
|
21.
|
Deans C and Wigmore SJ: Systemic
inflammation, cachexia and prognosis in patients with cancer. Curr
Opin Clin Nutr Metab Care. 8:265–269. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22.
|
Musselman D, Miller A, Porter M, Manatunga
A, Gao F, Penna S, Pearce BD, Landry J, Glover S, McDaniel JS and
Nemeroff CB: Higher than normal plasma interleukin-6 concentrations
in cancer patients with depression: Preliminary findings. Am J
Psychiatry. 158:1252–1257. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Bower J, Ganz P, Desmond K, Rowland J,
Meyerowitz BE and Belin TR: Fatigue in breast cancer survivors:
occurrence, correlates and impact on quality of life. J Clin Oncol.
18:743–753. 2000.PubMed/NCBI
|
|
24.
|
Lyon DE, McCain NL, Walter J and Schubert
C: Cytokine comparisons between women with breast cancer and women
with a negative breast biopsy. Nurs Res. 57:51–58. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25.
|
Yurkovetsy ZR, Kirkwood JM, Edington HD,
Marragoni AM, Velikokhatnaya L, Winans MT, Gorelik E and Lokshin
AE: Multiplex analysis of serum cytokines in melanoma patients
treated with interferon-α2b. Clin Cancer Res. 13:2422–2428.
2007.
|
|
26.
|
Lazar-Molnar E, Hegyesi H, Toth S and
Falus A: Autocrine and paracrine regulation by cytokines and growth
factors in melanoma. Cytokine. 12:547–554. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
27.
|
Leek R and Harris A: Tumor-associated
macrophages in breast cancer. J Mammary Gland Biol Neoplasia.
7:177–189. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
28.
|
Bachelot T, Ray-Coquard I, Menetrier-Caux
C, Rashtkha M, Duc A and Blay JY: Prognostic value of serum levels
of interleukin 6 and of serum and plasma levels of vascular
endothelial growth factor in hormone-refractory metastatic breast
cancer patients. Br J Cancer. 88:1721–1726. 2003. View Article : Google Scholar
|
|
29.
|
Salgado R, Junius S, Benoy I, Van Dam P,
Vermeulen P, Van Marck E, Huget P and Dirix LY: Circulating
interleukin-6 predicts survival in patients with metastatic breast
cancer. Int J Cancer. 103:642–646. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Zhang GJ and Adachi I: Serum interleukin-6
levels correlate to tumor progression and prognosis in metastatic
breast carcinoma. Anticancer Res. 19:1427–1432. 1999.PubMed/NCBI
|
|
31.
|
Mouawad R, Benhammouda A, Rixe O, Antoine
EC, Borel C, Weil M, Khayat D and Soubrane C: Endogenous
interleukin 6 levels in patients with metastatic malignant
melanoma: correlation with tumor burden. Clin Cancer Res.
2:1405–1409. 1996.PubMed/NCBI
|
|
32.
|
Mouawad R, Rixe O, Meric JB, Khayat D and
Soubrane C: Serum interleukin 6 concentration as predictive factor
of time to progression in metastatic malignant melanoma patients
treated by biochemotherapy: a retrospective study. Cytokines Cell
Mol Ther. 7:151–156. 2002. View Article : Google Scholar
|
|
33.
|
Tawara K, Oxford JT and Jorcyk CL:
Clinical significance of interleukin (IL)-6 in cancer metastasis to
bone: potential of anti-IL-6 therapies. Cancer Manag Res.
3:177–189. 2011.PubMed/NCBI
|
|
34.
|
Maeda N, Hagihara H, Nakata Y, Hiller S,
Wilder J and Reddick R: Aortic wall damage in mice unable to
synthesize ascorbic acid. Proc Natl Acad Sci USA. 97:841–846. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
35.
|
Harakeh S, Diab-Assaf M, Khalife JC,
Abu-El-Ardat KA, Baydoun E, Niedzwiecki A, Marwan E, El-Sabban ME
and Rath M: Ascorbic acid induces apoptosis in adult T-cell
leukemia. Anticancer Res. 27:289–298. 2007.PubMed/NCBI
|
|
36.
|
Gravenkamp C, Sypniewska R, Gauntt S,
Tarango M, Price P and Reddick R: Behavior of metastatic and
nonmetastatic breast tumors in old mice. Exp Biol Med. 229:665–675.
2004.PubMed/NCBI
|
|
37.
|
Cha J, Roomi MW, Ivanov V, Kalinovsky T,
Niedzwiecki A and Rath M: Ascorbate depletion increases growth and
metastasis of melanoma cells in vitamin C deficient mice. Exp
Oncol. 33:226–230. 2011.PubMed/NCBI
|
|
38.
|
McCormick WJ: Cancer: a collagen disease,
secondary to nutritional deficiency? Arch Pediatr. 76:166–171.
1959.PubMed/NCBI
|
|
39.
|
Ichim TE, Minev B, Braciak T, Luna B,
Hunninghake R, Mikirova NA, Jackson JA, Gonzalez MJ,
Miranda-Massari JR, Alexandrescu DT, Dasunu CA, Bogin V, Ancans J,
Stvens RB, Markosian B, Koropatnick J, Chen CS and Riordan NH:
Intravenous ascorbic acid to prevent and treat cancer-associated
sepsis? J Transl Med. 9:25–38. 2011. View Article : Google Scholar : PubMed/NCBI
|