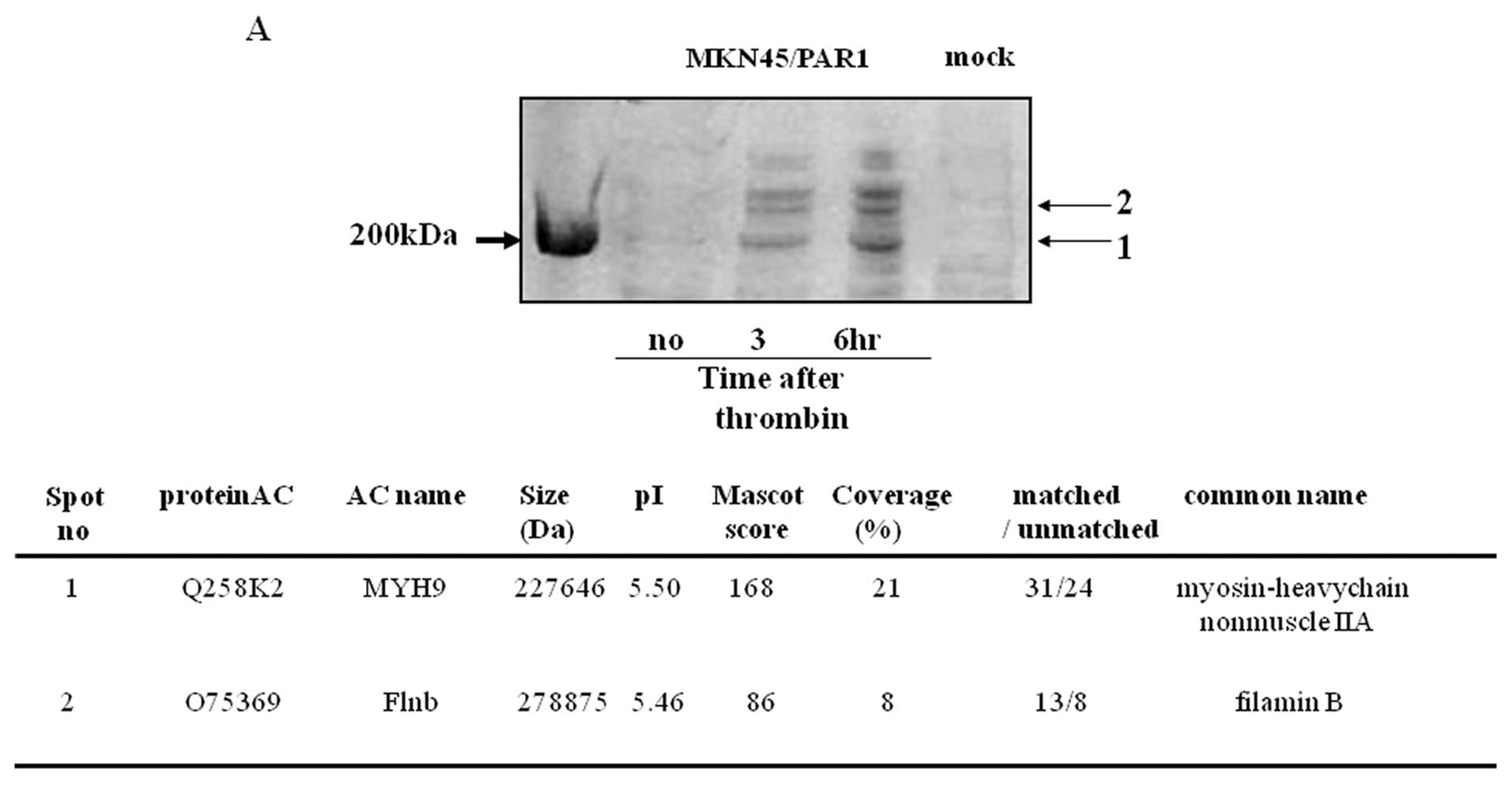
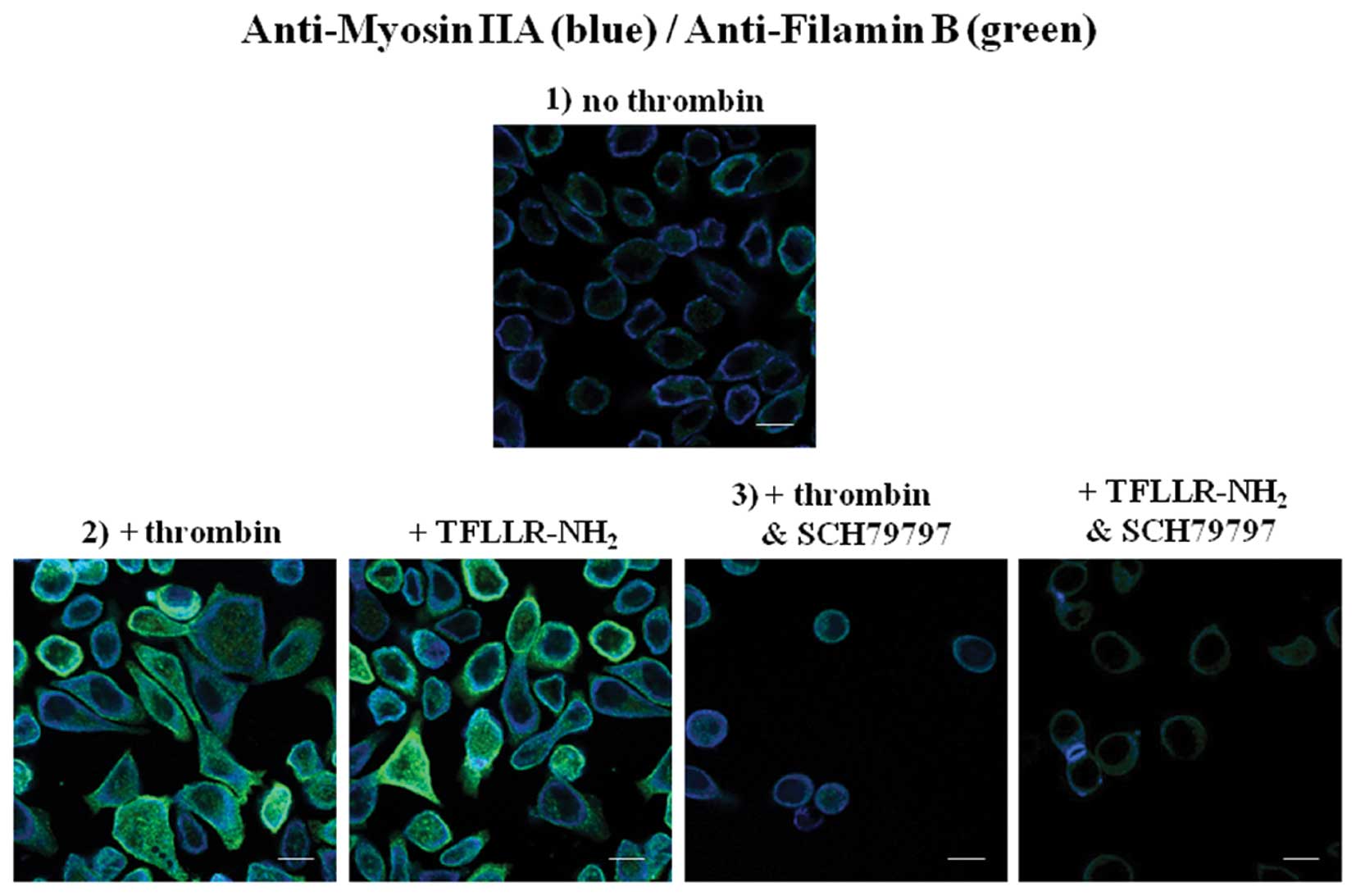
|
1.
|
Kurschat P and Mauch C: Mechanisms of
metastasis. Clin Exp Dermatol. 25:482–489. 2000.
|
|
2.
|
Rofstad EK: Microenviroment-induced cancer
metastasis. Int J Radiat Biol. 76:589–605. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Bhujwalla ZM, Artemov D, Ballesteros P,
Cerdan S, Gillies RJ, et al: Combined vascular and extracellular pH
imaging of solid tumors. NMR Biomed. 15:114–119. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Gatenby RA and Gawlinski ET: Mathematical
models of tumour invasion mediated by transformation-induced
alteration of microenvironmental pH. Novartis Found Symp.
240:85–96. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Xu L, Fukumura D and Jain RK: Acidic
extracellular pH induces vascular endothelial growth factor (VEGF)
in human glioblastoma cells via ERK1/2 MAPK signaling pathway:
mechanism of low pH-induced VEGF. J Biol Chem. 277:11368–11374.
2002. View Article : Google Scholar
|
|
6.
|
Lauffenburger DA and Horwitz AF: Cell
migration: a physically integrated molecular process. Cell.
84:359–369. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Horwitz AR and Parsons JT: Cell migration
- movin’ on. Science. 286:1102–1103. 1999.
|
|
8.
|
Ballestrem C, Wehrle-Haller B, Hinz B and
Imhof BA: Actin-dependent lamellipodia formation and
microtubule-dependent tail retraction control-directed cell
migration. Mol Biol Cell. 11:2999–3012. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Prins M and Otten HMM: Thrombosis and
cancer. A short history of Trousseau’s syndrome. Thrombosis and
Cancer. Lugassy G, Falanga A, Kakkar A and Rickles F: Taylor &
Francis; London: pp. 1–10. 2004
|
|
10.
|
Walz DA and Fenton JW: The role of
thrombin in tumor cell metastasis. Invasion Metastasis. 14:303–308.
1994.PubMed/NCBI
|
|
11.
|
Nierodzik ML and Karpatkin S: Thrombin
induces tumor growth, metastasis, and angiogenesis: Evidence for a
thrombin-regulated dormant tumor phenotype. Cancer Cell.
10:355–362. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Coughlin SR: Protease-activated receptors
in hemostasis, thrombosis and vascular biology. J Thromb Haemost.
3:1800–1814. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Hollenberg MD and Compton SJ:
International Union of Pharmacology. XXVIII. Proteinase-activated
receptors. Pharmacol Rev. 54:203–217. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Steinhoff M, Buddenkotte J, Shpacovitch V,
et al: Proteinase-activated receptors: transducers of
proteinase-mediated signalong in inflamation and immune response.
Endocr Rev. 26:1–43. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Ramachandran R and Hollenberg MD:
Proteinases and signaling: pathophysiological and therapeutic
implications via PARs and more. Br J Pharmacol. 153(Suppl 1):
S263–S282. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16.
|
Nystedt S, Emilsson K, Wahlestedt C and
Sundelin J: Molecular cloning of a potential proteinase activated
receptor. Proc Natl Acad Sci USA. 91:9208–9212. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Chambers RC, Dabbagh K, McAnulty RJ, Gray
AJ, Blanc-Brude OP and Laurent GJ: Thrombin stimulates fibroblast
procollagen production via proteolytic activation of
protease-activated receptor 1. Biochem J. 333:121–127.
1998.PubMed/NCBI
|
|
18.
|
Dawes KE, Gray AJ and Laurent GJ: Thrombin
stimulates fibroblast chemotaxis and replication. Eur J Cell Biol.
61:126–130. 1993.PubMed/NCBI
|
|
19.
|
Boire A, Covic L, Agarwal A, Jacques S,
Sherifi S and Kuliopulos A: PAR1 is a matrix metalloproteinase-1
receptor that promotes invasion and tumorigenesis of breast cancer
cells. Cell. 120:303–313. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Even-Ram SC, Uziely B, Cohen P,
Grisaru-Granovsky S, Maoz M, et al: Thrombin receptor
overexpression in malignant and physiological invasion processes.
Nat Med. 4:909–914. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Even-Ram SC, Maoz M and Pokroy E: Tumor
cell invasion is promoted by activation of protease activated
receptor-1 in cooperation with the alpha beta 5 integrin. J Biol
Chem. 276:10952–10962. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
22.
|
Van Aelst L and D’Souza-Schorey C: Rho
GTPases and signaling networks. Genes Dev. 11:2295–2322.
1997.PubMed/NCBI
|
|
23.
|
Nobes CD and Hall A: Rho, rac, and cdc42
GTPases regulate the assembly of multimolecular focal complexes
associated with actin stress fibers, lamellipodia, and filopodia.
Cell. 81:53–62. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
24.
|
Seasholtz TM, Majumdar M, Kaplan DD and
Brown JH: Rho and Rho kinase mediate thrombin-stimulated vascular
smooth muscle cell DNA synthesis and migration. Circ Res.
84:1186–1193. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
25.
|
Zohn IE, Symons M, Chrzanowska-Wodnicka M,
Westwick JK and Der CJ: Mas oncogene signaling and transformation
require the small GTP-binding protein Rac. Mol Cell Biol.
18:1225–1235. 1998.PubMed/NCBI
|
|
26.
|
Ridley AJ and Hall A: The small
GTP-binding protein rho regulates the assembly of focal adhesions
and actin stress fibers in response to growth factors. Cell.
70:389–399. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
27.
|
Ridley AJ, Paterson HF, Johnston CL,
Diekmann D and Hall A: The small GTP-binding protein rac regulates
growth factor-induced membrane ruffling. Cell. 70:401–410. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
28.
|
Fujimoto D, Hirono Y, Goi T, Katayama K,
Matsukawa S and Yamaguchi A: The activation of proteinase-activated
receptor-1 (PAR1) mediates gastric cancer cell proliferation and
invasion. BMC Cancer. 10:4432010. View Article : Google Scholar : PubMed/NCBI
|
|
29.
|
Ahn HS, Foster C, Boykow G, Stamford A,
Manna M and Graziano M: Inhibition of cellular action of thrombin
by N3-cyclopropyl-7-[[4-(1-methylethyl)phenyl]methyl]-7H-pyrrolo[3,
2-f]quinazoline-1,3-diamine (SCH79797), a nonpeptide thrombin
receptor antagonist. Biochem Pharmacol. 60:1425–1434.
2000.PubMed/NCBI
|
|
30.
|
Dery O, Corvera CU, Steinhoff M and
Bunnett NW: Proteinase-activated receptors: novel mechanisms of
signaling by serine proteases. Am J Physiol Cell Physiol.
274:C1429–C1452. 1998.PubMed/NCBI
|
|
31.
|
Van Obberghen-Schilling E, Vouret-Craviari
V, Chen YH, Grall D, Chambard JC and Pouyssegur J: Thrombin and its
receptor in growth control. Ann NY Acad Sci. 766:431–441.
1995.PubMed/NCBI
|
|
32.
|
Seasholtz TM, Majumdar M and Brown JH: Rho
as a mediator of G protein-coupled receptor signaling. Mol
Pharmacol. 55:949–956. 1999.PubMed/NCBI
|
|
33.
|
Kozasa T, Jiang X, Hart MJ, et al: p115
RhoGEF, a GTPase activating protein for Galpha12 and Galpha13.
Science. 280:2109–2111. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
34.
|
Hart MJ, Jiang X, Kozasa T, et al: Direct
stimulation of the guanine nucleotide exchange activity of p115
RhoGEF by Galpha13. Science. 280:2112–2114. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
35.
|
Bresnick AR: Mechanisms of nonmuscle
myosin-II regulation. Curr Opin Cell Biol. 11:26–33. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
36.
|
Warrick HM and Spudich JA: Myosin
structure and function in cell motility. Annu Rev Cell Biol.
3:379–421. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
37.
|
Giannone G, Dubin-Thaler BJ, Rossier O, et
al: Lamellipodial actin mechanically links myosin activity with
adhesion-site formation. Cell. 128:561–575. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
38.
|
Wu D, Asiedu M and Wei Q:
Myosin-interacting guanine exchange factor (MyoGEF) regulates the
invasion activity of MDA-MB-231 breast cancer cells through
activation of RhoA and RhoC. Oncogene. 28:2219–2230. 2009.
View Article : Google Scholar
|
|
39.
|
Robertson SP: Filamin A: phenotypic
diversity. Curr Opin Genet Dev. 15:301–307. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
40.
|
Weihing RR: The filamins: properties and
functions. Can J Biochem Cell Biol. 63:397–413. 1985.PubMed/NCBI
|
|
41.
|
Cunningham CC: Actin structural proteins
in cell motility. Cancer Metastasis Rev. 11:69–77. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
42.
|
Takafuta T, Saeki M, Fujimoto TT, Fujimura
K and Shapiro SS: A new member of the LIM protein family binds to
filamin B and localizes at stress fibers. J Biol Chem.
278:12175–12181. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
43.
|
van der Flier A, Kuikman I, Kramer D, et
al: Different splice variants of filamin-B affect myogenesis,
subcellular distribution, and determine binding to integrin [beta]
subunits. J Cell Biol. 156:361–376. 2002.PubMed/NCBI
|
|
44.
|
Baldassarre M, Razinia Z, Burande CF,
Lamsoul I, Lutz PG and Calderwood DA: Filamins regulate cell
spreading and initiation of cell migration. PLoS One. 4:e78302009.
View Article : Google Scholar : PubMed/NCBI
|
|
45.
|
Hollenberg MD, Mokashi M, Leblond L and
DiMaio J: Synergistic actions of a thrombin-derived synthetic
peptide and a thrombin receptor-activating peptide in stimulating
fibroblast mitogenesis. J Cell Physiol. 169:491–496. 1996.
View Article : Google Scholar
|
|
46.
|
Badgwell DB, Lu Z, Le K, et al: The
tumor-suppressor gene ARHI (DIRAS3) suppresses ovarian cancer cell
migration through inhibition of Stat3 and FAK/Rho signaling
pathway. Oncogene. 31:68–79. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
47.
|
Betson M, Lozano E, Zhang J and Braga VM:
Rac activation upon cell-cell contact formation is dependent on
signaling from the epidermal growth factor receptor. J Biol Chem.
277:36962–36969. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
48.
|
Macfarlane SR, Seatter MJ, Kanke T, Hunter
GD and Plevin R: Proteinase-activated receptors. Pharmacol Rev.
53:245–282. 2001.PubMed/NCBI
|
|
49.
|
Bogatcheva NV, Garcia JG and Verin AD:
Molecular mechanisms of thrombin-induced endothelial cell
permeability. Biochemistry (Mosc). 67:75–84. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
50.
|
Garcia JG, Pavalko FM and Patterson CE:
Regulation of endothelial cell gap formation and barrier
dysfunction: role of myosin light chain phosphorylation. J Cell
Physiol. 163:510–522. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
51.
|
Buhl AM, Johnson NL, Dhanasekaran N and
Johnson GL: G alpha 12 and G alpha 13 stimulate Rho-dependent
stress fiber formation and focal adhesion assembly. J Biol Chem.
270:24631–24634. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
52.
|
Majumdar M, Seasholtz TM, Buckmaster C,
Toksoz D and Brown JH: A rho exchange factor mediates thrombin and
Gα(12)-induced cytoskeletal responses. J Biol Chem.
274:26815–26821. 1999.PubMed/NCBI
|
|
53.
|
Amano M, Ito M, Kimura K, et al:
Phosphorylation and activation of myosin by Rho-associated kinase
(Rho-kinase). J Biol Chem. 271:20246–20249. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
54.
|
Kimura K, Ito M, Amano M, et al:
Regulation of myosin phosphatase by Rho-associated kinase
(Rho-kinase). Science. 273:245–248. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
55.
|
Kraynov VS, Chamberlain C, Bokoch GM,
Achwartz MA, Slabough S and Hahn KM: Localized Rac activation
dynamics visualized in living cells. Science. 290:333–337. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
56.
|
Small JV, Stradal T, Vignal E and Rottner
K: The lamellipodium: where motility begins. Trends Cell Biol.
12:112–120. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
57.
|
Miki H, Suetsugu S and Takenawa T: WAVE, a
novel WASP-family protein involved in actin reorganization induced
by Rac. EMBO J. 17:6932–6941. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
58.
|
Yamazaki D, Suetsugu S, Miki H, et al:
WAVE2 is required for directed cell migration and cardiovascular
development. Nature. 424:452–456. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
59.
|
Welch MD and Mullins RD: Cellular control
of actin nucleation. Annu Rev Cell Dev Biol. 18:247–288. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
60.
|
Pollard TD and Borisy GG: Cellular
motility driven by assembly and disassembly of actin filaments.
Cell. 112:453–465. 2003. View Article : Google Scholar : PubMed/NCBI
|