Introduction
Prostate cancer is the sixth leading cause of
cancer-related death in men in the world (1) and the second in the USA (2). The number of men diagnosed with
prostate cancer is increasing every year, especially in the
developing country. Although most of the patients survive the
initial stage of prostate cancer with androgen ablation therapy,
with tumor progression, it may gradually metastasize to other
organs, which lead to poor prognosis and quick death.
Epithelial-mesenchymal transition (EMT) is now
viewed as a crucial process during tumor progression and metastasis
(3). EMT is characterized by
remarkable cell morphology change from epithelial round phenotype
to mesenchymal spindle-shaped phenotype. The process involves
downregulation of cell adhesion/junction proteins, such as
E-cadherin and Zonula occludens (4,5), as
well as upregulation of mesenchymal and ECM degradative proteins
such as Vimentin, N-cadherin and MMPs (6,7),
which results in loss of cell-cell adhesion and increased migratory
capacity that contribute to cancer metastasis (8). Many factors promote cancer cell
invasion and metastasis by inducing EMT, such as transforming
growth factor (TGF) β, platelet derived growth factor (PDGF) D and
fibroblast growth factor (FGF) (4,9,10).
However, the role of TGF-α in the process of EMT and the mechanism
remain to be elucidated.
TGF-α and its receptor epidermal growth factor
receptor (EGFR) are crucial oncogenes in the development of
different cancers (11,12), including prostate cancer (13). Compared with benign prostate
epithelium, TGF-α mRNA and protein levels are increased in
carcinoma cells (13,14). Interestingly, studies have shown
that compared with the early-stage localized tumors, TGF-α
expression is more abundant when prostate cancer metastasizes
(13,15–17).
However, a reason for the strong correlation between TGF-α
expression and high-invasive capacity has not been elucidated. Here
we report TGF-α was able to induce EMT in prostate cancer.
MicroRNAs (miRNAs) are small non-coding RNAs that
can sequence-specifically bind to the 3′-untranslated regions of
target mRNAs resulting in mRNA destruction or translational
inhibition (18). Growing evidence
suggests that miRNAs are important regulators of EMT. Upregulation
of miR-9 provokes the development of EMT (19). Downregulation of miR-200
contributes to PDGF-D induced EMT. However, the involvement of
miRNAs in TGF-α-induced EMT has not been elucidated.
In this study, we showed that TGF-α, frequently
upregulated in advanced prostate cancer, is able to incuce EMT in
DU145 cells. Downregulation of miR-124 is ubiquitous in TGF-α
treated cancer cells and contributes to TGF-α-induced EMT by
targeting Slug. This study indicates that miR-124 may be a
promising therapeutic candidate for prostate cancer metastasis.
Materials and methods
Cell culture
DU145, HepG2 and Hu7 cell lines were obtained from
the American Type Culture Collection (ATCC, Manassas, VA, USA).
MKN-45, MDA-MB-231, HCT15 and PA1 cell lines were purchased from
Cell Resource Center, IBMS, CAMS/PUMC (China). Cells were cultured
in RPMI-1640 medium containing 10% FBS and 1%
penicillin/streptomycin, and maintained under the conditions of
37°C with 5% CO and 95% air.
Tansfection of miRNAs
DU145 cells were transiently transfected with
miR-124 mimics or miR-124 inhibitors or negative control (RiboBio
Co., Ltd., Guangzhou, China), using Lipofectamine 2000 reagent
(Invitrogen), according to the manufacturer’s instructions.
Briefly, cells were trypsinized and seeded in 60-mm dishes 24 h
before transfection. The Opti-MEM serum-free media was used to
dissolve the transfection mixture. After 24 h of transfection, the
media were removed and RPMI media containing 10% FBS and 1%
penicillin/streptomycin was added to the cells.
Western blot analysis
The procedure of western blot analysis was described
previously (20). In brief, cells
were washed by PBS several times and harvested using RIPA lysis
buffer with protease inhibitor. Protein concentration was
determined using BCA reagent (Beyotime Institute of Biotechnology,
Nanjing, China) according to the manufacturer’s protocols. Equal
amounts of proteins (60 μg) were electrophoresed in 12% SDS-PAGE
gels and transferred to the nitrocellulose membranes. After being
blocked with 5% milk for 2 h at room temperature, the membranes
were incubated with primary antibodies for E-cadherin, Vimentin,
Slug and Tubulin (Cell Signaling Technology, USA) overnight at 4°C.
Tubulin was used as an internal control. The bands were detected
with ECL reagents and analysed by Quantity One software.
RNA extraction and real-time RT-PCR
Total RNA was extracted using TRIzol reagent (Gibco,
Invitrogen, Carlsbad, CA, USA). Then 1 μg of extracted RNA was used
for reverse transcription and the obtained cDNA was used in
real-time PCR. The specific RT-PCR primers for miR-124 and U6 were
purchased from Ribo (RiboBio Co., Ltd.).
Immunofluorescence staining
We performed immunofluorescence staining on DU145
cells to determine Vimentin expression. Firstly, cells were fixed
with 4% paraformaldehyde for 15 min followed by permeabilized with
0.1% Triton X-100 for 10 min. Then cells were blocked with 10% goat
serum (in PBS) blocking solution for 1 h followed by incubation
with Vimentin antibodies at a dilution of 1:100 at 4°C overnight.
At the end of the incubation, cells were washed and incubated with
FITC-conjugated secondary antibody for 1 h. PI was added to the
cells for another 10 min. Cells were observed and images were
captured by fluorescence microscopy.
Luciferase reporter assay
Luciferase reporter assay was done as previously
described (21). Briefly, cells
were seeded in a 96-well plate and transfected with plasmids.
Twenty-four hours after transfection, cells were harvested and
Firefly and Renilla luciferase activities were measured using
Luciferase Reporter Gene assay kit of Beyotime (China). Renilla
luciferase was used as an internal control and the RLU (relative
light unit) ratio of Firefly luciferase relative to Renilla
luciferase was calculated.
Wound healing assay
DU145 cells were seeded in 6-well plates to achieve
90% confluence. Twenty-four hours after transfection, a vertical
wound was created using a 200-μl pipette tip. Then the cells were
washed with PBS for three times and medium without serum was added
into the wells. After 24-h incubation, the wound was observed and
random fields in each well were selected for imaging. The images
were analysed by ImageJ and the distance of wound closure was used
to estimate the migration ability.
Migration and invasion assay
Cell migration and invasive ability was evaluated
using 24-well transwell plates. For migration assay, 24 h after
transfection, 5×104 cells per well were seeded into the
top chamber and maintained in serum-free medium. Medium (600 μl)
containing 10% FBS was added into the bottom chamber. After
incubated for 24 h at 37°C, cells that migrated through the pore
polycarbonate membrane were fixed with methanol and stained with
Giemsa. Then the migrated cells were observed and images were
captured using microscopy. For invasion assay, before cell seeding,
the Matrigel were diluted in serum-free medium at the concentration
of 1 mg/ml. Diluted Matrigel (100 μl) per well were added into the
top chamber and incubated for 4 h at 37°C. The following procedures
were the same as in the migration assay.
Statistical analysis
The experiments in this study were repeated at least
three times and values were expressed as mean ± SEM. The results
were analyzed by One-way ANOVA accompanied with Turkey
multiple-comparisions test (GraphPad Prism version 5.0). Student’s
t-test was used for the comparison between two groups. P<0.05
was considered as statistically significant.
Results
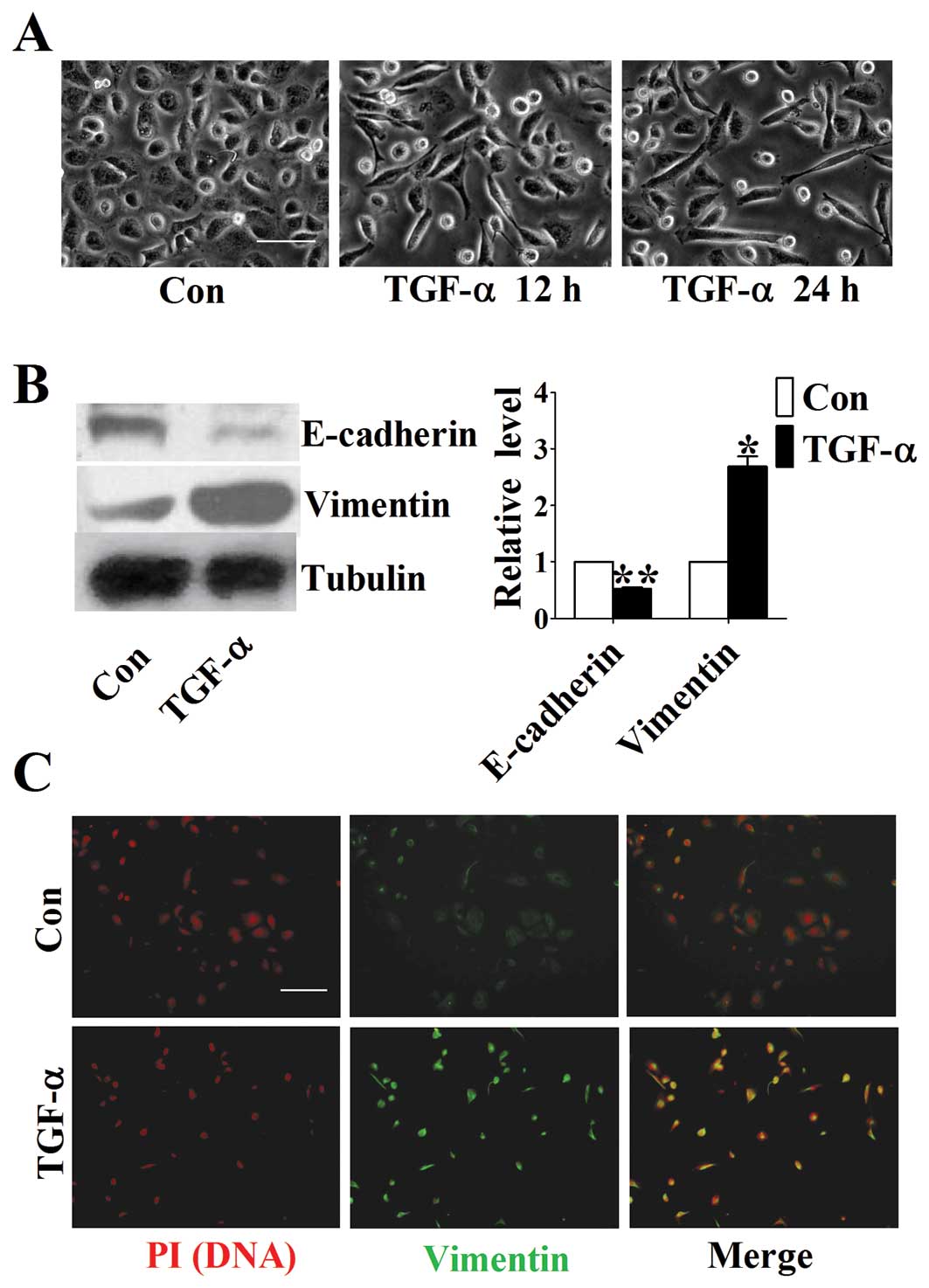
TGF-α induces changes in morphology and
EMT related markers of DU145 cells
To identify the possible effect of TGF-α on the
progression of prostate cancer, we treated DU145 cells with 50
ng/ml TGF-α. At 0, 12 and 24 h after TGF-α treatment, images of the
morphology of cells were taken by microscopy. Intrestingly, we
found that TGF-α induced a remarkable morphological change from the
round and cobblestone form to the long fusiform shape (Fig. 1A), reminiscent of EMT, an important
process in cancer development. To further confirm whether TGF-α
could induce EMT in DU145 cells, we assessed the expression levels
of EMT related markers using western blot analysis. We found that
the expression of E-cadherin, an epithelial marker, was
significantly decreased after TGF-α treatment, whereas Vimentin, a
mesenchymal marker, was increased concomitantly (Fig. 1B). Similar results were obtained
from immunofluorescent staining (Fig.
1C). These data suggest that TGF-α is able to induce EMT in
DU145 cells.
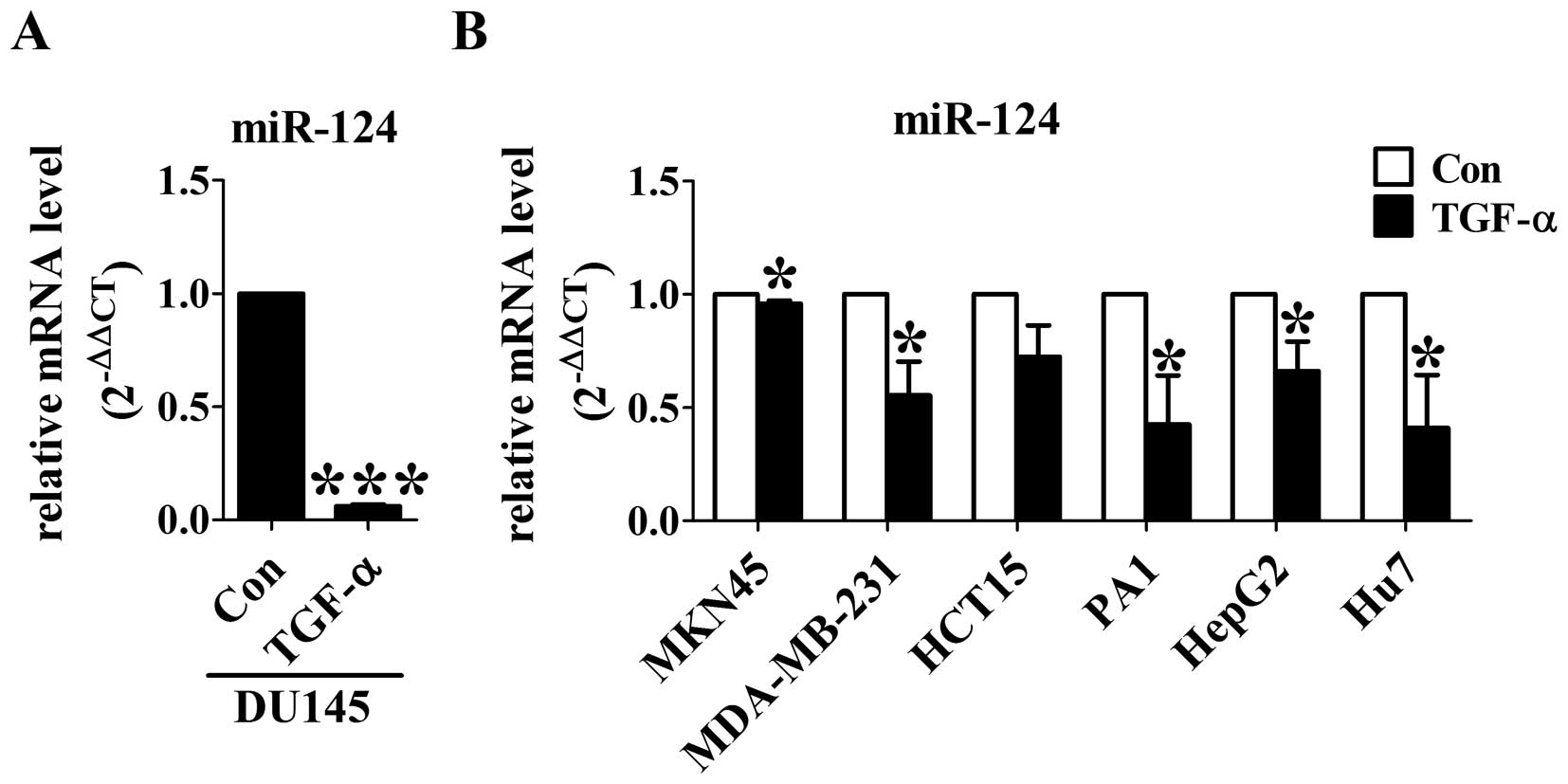
Downregulation of miR-124 is ubiquitous
in TGF-α treated cancer cells
To test whether miR-124 could contribute to
TGF-α-induced EMT, we first detected miR-124 levels using real-time
RT-PCR in DU145 cells. After TGF-α treatment for 24 h, the
expression of miR-124 was significantly decreased compared with
control group (Fig. 2A). To
further confirm the effect of TGF-α on miR-124 expression, TGF-α
was applied to gastric cancer MKN-45 cells, breast cancer
MDA-MB-231 cells, colon cancer HCT15 cells, ovarian teratocarcinoma
PA1 cells, liver cancer HepG2 cells and Hu7 cells. As expected, the
levels of miR-124 was significantly decreased by TGF-α in all of
the six different cancer cell lines (Fig. 2B). These results indicate that
miR-124 may function as a key downstream effector of TGF-α in
cancer cells.
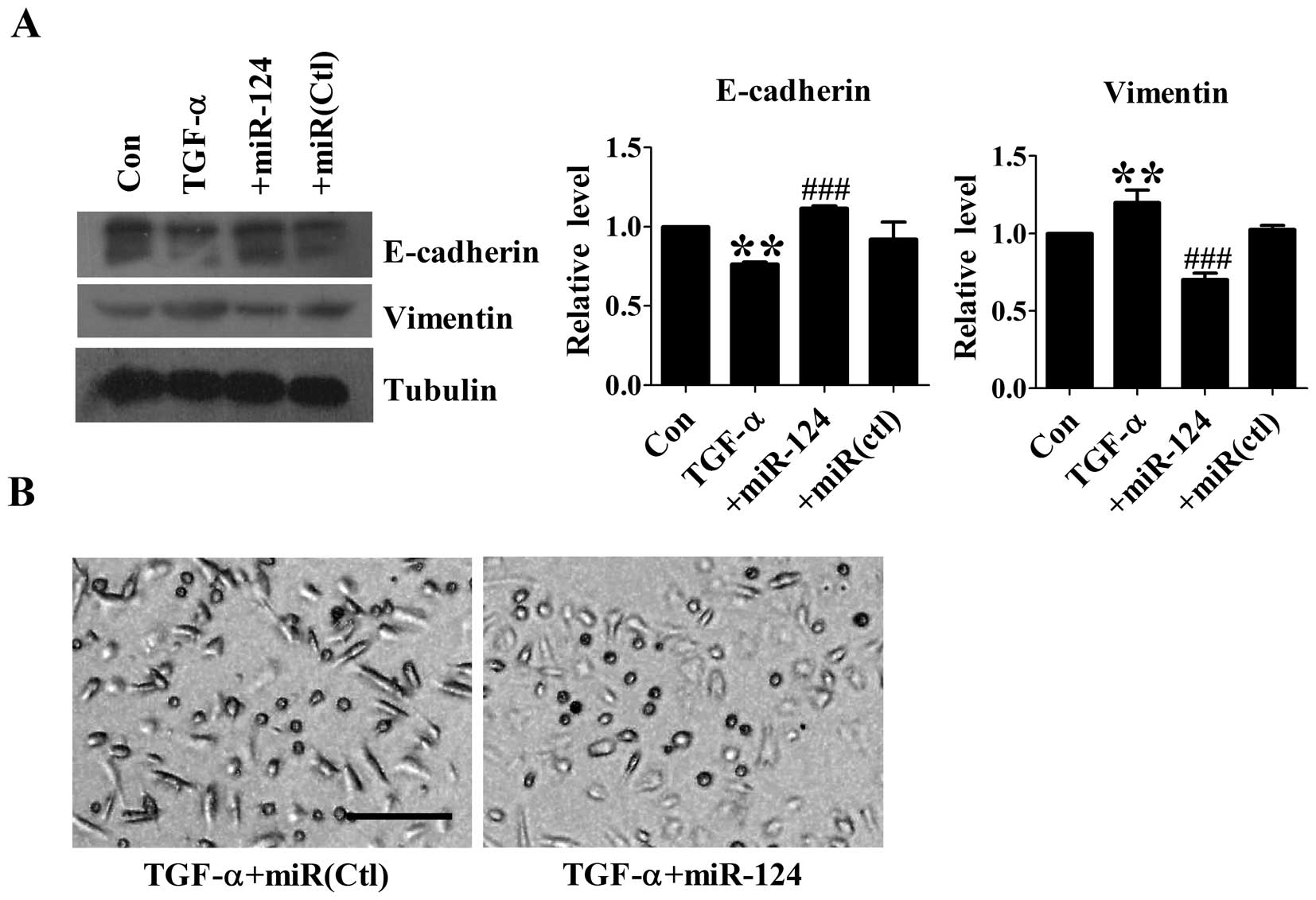
Overexpression of miR-124 inhibits TGF-α
induced-EMT in DU145 cells
The effect of miR-124 on TGF-α-induced EMT was
evaluated by western blot analysis. Treatment with 50 ng/ml TGF-α
on DU145 cells led to a significant EMT change as indicated by
E-cadherin and Vimentin protein levels. Overexpression of miR-124
inhibited TGF-α-induced downregulation of E-cadherin and
upregulation of Vimentin, whereas transfection with microRNA
negative control had no effect on these proteins (Fig. 3A). In addition, we found that TGF-α
treated DU145 cells with miR-124 overexpression displayed
round-like morphology, indicating acquisition of
mesenchymal-epithelial transition (MET) phenotype (Fig. 3B). The above results indicate that
overexpression of miR-124 is able to reverse TGF-α-induced EMT.
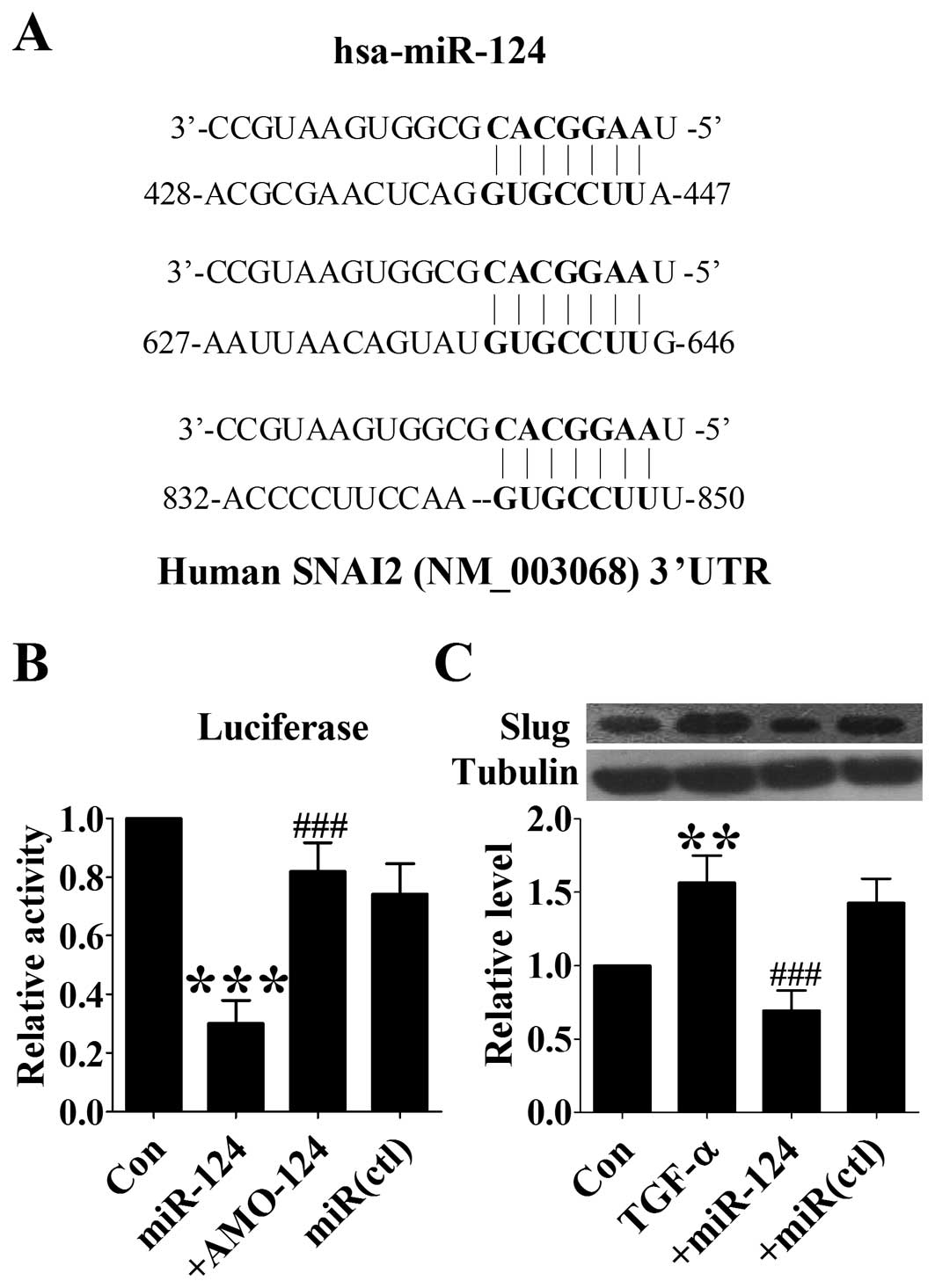
Involvement of Slug in miR-124-mediated
suppression of TGF-α-induced EMT
Slug was reported to regulate expression of
E-cadherin and the EMT process of cancer cells. Computational
analysis using the miRNA TargetScan predicted that Slug might be a
target of miR-124. The 3′-UTR of Slug mRNA contains 3 binding sites
for miR-124 in humans (Fig. 4A). A
previous study has proved that Slug could be regulated by miR-124.
In this study, we sought to test whether or not miR-124 could
inhibit TGF-α-induced EMT via regulating Slug. First we confirmed
the regulation of Slug by miR-124 using luciferase assay. The
results showed that miR-124 markedly suppressed the luciferase
activity of Slug 3′-UTR luciferase vector (Fig. 4B). In DU145 cells, TGF-α treatment
induced significant upregulation of Slug expression as indicated by
western blot analysis, whereas overexpression of miR-124 abolished
this effect (Fig. 4C). These data
suggest that miR-124 may inhibit TGF-α-induced EMT by regulating
Slug.
Overexpression of miR-124 inhibites
migration and invasion of TGF-α treated DU145 cells
EMT is a major process leading to increased
migration and invasive ability in cancer cells. The migration
ability of DU145 cells was first assessed by wound healing. The
migration rate of TGF-α-treated cells significantly increased
compared with control group, while overexpression of miR-124
obviously attenuated cell movement (Fig. 5A). Moreover, migration assay was
also used to evaluate the migration ability. We found that
tranfection of miR-124 markedly reduced the amount of migrated
cells induced by TGF-α (Fig. 5B).
Additionally, miR-124 overexpression resulted in a significant
repression of the invasion ability as evidenced by the invasion
assay (Fig. 5B). These results
clearly indicate that re-expression of miR-124 suppresses migration
and invasion of TGF-α treated DU145 cells.
Discussion
The epidermal growth factor receptor (EGFR) and two
of its ligands, epidermal growth factor (EGF) and TGF-α are
frequently overexpressed and play a central role in the development
of prostate cancer. Studies have reported that the progression of
prostate cancer from androgen-dependence to androgen-independence
involves a ‘switch’ in the synthesis from EGF to TGF-α. In the
localized prostate cancer, EGF is more abundant than TGF-α. When
the disease develops to advanced, metastatic stages, the expression
of TGF-α increases and EGF decreases, indicating that TGF-α may
play a more important role in cancer progression and metastasis. It
is now recognized that carcinoma cells undergo a process called EMT
to acquire migratory properties. Therefore, in this study, we
determined if TGF-α is able to induce EMT in prostate cancer. Our
data showed that treatment of DU145 cells with TGF-α induced a
dramatic cell morphological alteration that is similar to EMT.
Moreover, western blot results showed the expression of E-cadherin
was downregulated and the expression of Vimentin was upregulated,
which are consistent with the characteristics of EMT.
MicroRNAs are thought to play important roles in a
wide variety of biological processes. Aberrant expression of miRNAs
is often involved in the pathogenesis of human diseases, such as
cardiovascular disease (22),
infection (23) and tumor
(24). Genomic profiling and
expression studies found a global downregulation of miRNAs in many
cancers. The expression of miR-124 was found to be downregulated in
various tumors, including breast cancer (25), gastric cancer (26) and prostate cancer (27), indicating miR-124 may function as a
negative regulator of cancer progression. A previous study reported
that miR-124 inhibited proliferation of prostate cancer cells by
targeting the androgen receptor (27). In this study, we found that the
expression of miR-124 was reduced in TGF-α treated DU145 cells. To
further validate the TGF-α-induced downregulation of miR-124 was
not specific in prostate cancer cells, we treated different cell
lines, including gastric cancer MKN-45 cells, breast cancer
MDA-MB-231 cells, colon cancer HCT15 cells, ovarian teratocarcinoma
PA1 cells, liver cancer HepG2 cells and Hu7 cells with TGF-α.
Treatment of the above-mentioned cell lines with TGF-α all led to a
decrease in the expression of miR-124. These results imply the
possible role of miR-124 as a downstream effector of TGF-α in
cancer cells.
Recent studies have reported that microRNAs,
especially miR-200 family, acted as a key regulator of EMT. MiR-200
family and miR-205 were significantly suppressed in cells
undergoing EMT and forced expression of miR-200 family was able to
initiate MET, the reversed process of EMT, via downregulating
transcription factors ZEB1 and SIP1 (28). Besides miR-200 family, miR-9 led to
EMT-like tranformation and increased cell invasive ability by
directly targeting E-cadherin (19). Mir-30a was found to target Snail, a
transcription repressor of E-cadherin, leading to EMT induction
(29). Interestingly, microRNA
could also affect the process of EMT by regulating expression of
other microRNAs. As an example the miR-103/107 family, which
directly targeted Dicer, resulting in decreased miR-200 levels and
EMT occurrence (30). A previous
study by Liang et al showed that miR-124 could regulate the
process of EMT in breast cancer MDA-MB-231 cells (31). Since in this study, we have proven
that TGF-α was able to induce EMT and reduce miR-124 expression in
DU145 cells, it is tempting to speculate that miR-124 may mediate
TGF-α-induced EMT. Our results showed that transfection of DU145
cells with miR-124 before TGF-α treatment significantly increased
the expression of epithelial marker E-cadherin and decreased the
expression of mesenchymal marker Vimentin, suggesting that the
reduction miR-124 contributed to TGF-α-induced EMT and
re-expression of miR-124 could lead to the reversal of
TGF-α-induced EMT.
Several transcription factors have been validated as
pivotal inducers of EMT, including ZEB1 (32), ZEB2 (33), Twist (34), Snail (35), and Slug (36). Slug is a zinc-finger transcription
factor known to repress endogenous E-cadherin expression by binding
to E-box elements within the promoter. Upregulation of Slug has
been shown to play an important role in the induction of EMT
(37). Studies have confirmed the
regulation of Slug by miR-124 using luciferase reporting assay
(31,38). Liang et al (30) have proven that miR-124 could
regulate EMT by targeting Slug in breast cancer cells. However,
whether or not Slug could mediate the inhibitory effect of miR-124
in TGF-α-induced EMT is unknown. In this study, we found that Slug
was significantly increased in response to TGF-α treatment and
transfection of miR-124 inhibited Slug expression, suggesting Slug
is a downstream effector of miR-124 in TGF-α-induced EMT.
Cancer cells that undergo EMT often have an
increased migratory and invasive capacity (39), and the overexpression of miR-124
led to the reversal of TGF-α-induced EMT. In this study, our data
indicated that transfection of DU145 cells with miR-124
significantly inhibited cell migration and invasion induced by
TGF-α, indicating overexpression of miR-124 led to a less invasive
cell phenotype, which was consistent with MET characteristics.
In conclusion, we found that TGF-α is able to induce
EMT in DU145 cells, which may provide a reason for the EGFR ligand
switch in the late stage of prostate cancer. Downregulation of
miR-124 contributes to TGF-α-induced EMT and re-expression of
miR-124 could reverse this process. In addition, miR-124
overexpression reduces cell migratory and invasive ability in TGF-α
treated cells, the underlying mechanism involves the downregulation
of Slug. These results provide evidence that miR-124 may be a
potential therapeutic target for advanced prostate cancer.
Acknowledgements
This study was supported by grants from the National
Youth Natural Science Foundation of China (no. 81101942).
References
|
1
|
Baade PD, Youlden DR and Krnjacki LJ:
International epidemiology of prostate cancer: geographical
distribution and secular trends. Mol Nutr Food Res. 53:171–184.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Siegel R, Ward E, Brawley O and Jemal A:
Cancer statistics, 2011: the impact of eliminating socioeconomic
and racial disparities on premature cancer deaths. CA Cancer J
Clin. 61:212–236. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Thiery JP, Acloque H, Huang RY and Nieto
MA: Epithelial-mesenchymal transitions in development and disease.
Cell. 139:871–890. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Strutz F, Zeisberg M, Ziyadeh FN, et al:
Role of basic fibroblast growth factor-2 in epithelial-mesenchymal
transformation. Kidney Int. 61:1714–1728. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ikenouchi J, Matsuda M, Furuse M and
Tsukita S: Regulation of tight junctions during the
epithelium-mesenchyme transition: direct repression of the gene
expression of claudins/occludin by Snail. J Cell Sci.
116:1959–1967. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jorda M, Olmeda D, Vinyals A, et al:
Upregulation of MMP-9 in MDCK epithelial cell line in response to
expression of the Snail transcription factor. J Cell Sci.
118:3371–3385. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Miyoshi A, Kitajima Y, Sumi K, et al:
Snail and SIP1 increase cancer invasion by upregulating MMP family
in hepatocellular carcinoma cells. Br J Cancer. 90:1265–1273. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kovacic JC, Mercader N, Torres M, Boehm M
and Fuster V: Epithelial-to-mesenchymal and
endothelial-to-mesenchymal transition: from cardiovascular
development to disease. Circulation. 125:1795–1808. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kong D, Wang Z, Sarkar SH, et al:
Platelet-derived growth factor-D overexpression contributes to
epithelial-mesenchymal transition of PC3 prostate cancer cells.
Stem Cells. 26:1425–1435. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Valcourt U, Kowanetz M, Niimi H, Heldin CH
and Moustakas A: TGF-beta and the Smad signaling pathway support
transcriptomic reprogramming during epithelial-mesenchymal cell
transition. Mol Biol Cell. 16:1987–2002. 2005. View Article : Google Scholar
|
|
11
|
Todaro GJ, Fryling C and De Larco JE:
Transforming growth factors produced by certain human tumor cells:
polypeptides that interact with epidermal growth factor receptors.
Proc Natl Acad Sci USA. 77:5258–5262. 1980. View Article : Google Scholar
|
|
12
|
Lippman ME: The development of biological
therapies for breast cancer. Science. 259:631–632. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ching KZ, Ramsey E, Pettigrew N, D’Cunha
R, Jason M and Dodd JG: Expression of mRNA for epidermal growth
factor, transforming growth factor-alpha and their receptor in
human prostate tissue and cell lines. Mol Cell Biochem.
126:151–158. 1993. View Article : Google Scholar
|
|
14
|
Glynne-Jones E, Goddard L and Harper ME:
Comparative analysis of mRNA and protein expression for epidermal
growth factor receptor and ligands relative to the proliferative
index in human prostate tissue. Hum Pathol. 27:688–694. 1996.
View Article : Google Scholar
|
|
15
|
Seth D, Shaw K, Jazayeri J and Leedman PJ:
Complex post-transcriptional regulation of EGF-receptor expression
by EGF and TGF-alpha in human prostate cancer cells. Br J Cancer.
80:657–669. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
DeHaan AM, Wolters NM, Keller ET and
Ignatoski KM: EGFR ligand switch in late stage prostate cancer
contributes to changes in cell signaling and bone remodeling.
Prostate. 69:528–537. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Scher HI, Sarkis A, Reuter V, et al:
Changing pattern of expression of the epidermal growth factor
receptor and transforming growth factor alpha in the progression of
prostatic neoplasms. Clin Cancer Res. 1:545–550. 1995.PubMed/NCBI
|
|
18
|
Bartel DP: MicroRNAs: genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ma L, Young J, Prabhala H, et al: miR-9, a
MYC/MYCN-activated microRNA, regulates E-cadherin and cancer
metastasis. Nat Cell Biol. 12:247–256. 2010.PubMed/NCBI
|
|
20
|
Lin F, Lin P, Zhao D, et al: Sox2 targets
cyclinE, p27 and survivin to regulate androgen-independent human
prostate cancer cell proliferation and apoptosis. Cell Prolif.
45:207–216. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhou F, Zhang L, Gong K, et al: LEF-1
activates the transcription of E2F1. Biochem Biophys Res Commun.
365:149–153. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhou S, Liu Y, Prater K, Zheng Y and Cai
L: Roles of microRNAs in pressure overload- and ischemia-related
myocardial remodeling. Life Sci. 93:855–862. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chang Y, Dou Y, Bao H, et al: Multiple
microRNAs targeted to internal ribosome entry site against
foot-and-mouth disease virus infection in vitro and in vivo. Virol
J. 11:12014. View Article : Google Scholar
|
|
24
|
Luo Q, Wei C, Li X, et al: MicroRNA-195–5p
is a potential diagnostic and therapeutic target for breast cancer.
Oncol Rep. 31:1096–1102. 2014.
|
|
25
|
Lv XB, Jiao Y, Qing Y, et al: miR-124
suppresses multiple steps of breast cancer metastasis by targeting
a cohort of pro-metastatic genes in vitro. Chin J Cancer.
30:821–830. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Xia J, Wu Z, Yu C, et al: miR-124 inhibits
cell proliferation in gastric cancer through down-regulation of
SPHK1. J Pathol. 227:470–480. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Shi XB, Xue L, Ma AH, et al: Tumor
suppressive miR-124 targets androgen receptor and inhibits
proliferation of prostate cancer cells. Oncogene. 32:4130–4138.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gregory PA, Bert AG, Paterson EL, et al:
The miR-200 family and miR-205 regulate epithelial to mesenchymal
transition by targeting ZEB1 and SIP1. Nat Cell Biol. 10:593–601.
2008. View
Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kumarswamy R, Mudduluru G, Ceppi P, et al:
MicroRNA-30a inhibits epithelial-to-mesenchymal transition by
targeting Snai1 and is downregulated in non-small cell lung cancer.
Int J Cancer. 130:2044–2053. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Martello G, Rosato A, Ferrari F, et al: A
MicroRNA targeting dicer for metastasis control. Cell.
141:1195–1207. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Liang YJ, Wang QY, Zhou CX, et al: MiR-124
targets Slug to regulate epithelial-mesenchymal transition and
metastasis of breast cancer. Carcinogenesis. 34:713–722. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Eger A, Aigner K, Sonderegger S, et al:
DeltaEF1 is a transcriptional repressor of E-cadherin and regulates
epithelial plasticity in breast cancer cells. Oncogene.
24:2375–2385. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Comijn J, Berx G, Vermassen P, et al: The
two-handed E box binding zinc finger protein SIP1 downregulates
E-cadherin and induces invasion. Mol Cell. 7:1267–1278. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yang J, Mani SA, Donaher JL, et al: Twist,
a master regulator of morphogenesis, plays an essential role in
tumor metastasis. Cell. 117:927–939. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Cano A, Perez-Moreno MA, Rodrigo I, et al:
The transcription factor snail controls epithelial-mesenchymal
transitions by repressing E-cadherin expression. Nat Cell Biol.
2:76–83. 2000. View
Article : Google Scholar : PubMed/NCBI
|
|
36
|
Hajra KM, Chen DY and Fearon ER: The SLUG
zinc-finger protein represses E-cadherin in breast cancer. Cancer
Res. 62:1613–1618. 2002.PubMed/NCBI
|
|
37
|
Wen FC, Chang TW, Tseng YL, Lee JC and
Chang MC: hRAD9 functions as a tumor suppressor by inducing
p21-dependent senescence and suppressing epithelial-mesenchymal
transition through inhibition of Slug transcription.
Carcinogenesis. Feb 5–2014.(Epub ahead of print).
|
|
38
|
Xia H, Cheung WK, Ng SS, et al: Loss of
brain-enriched miR-124 microRNA enhances stem-like traits and
invasiveness of glioma cells. J Biol Chem. 287:9962–9971. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Tamagawa S, Beder LB, Hotomi M, et al:
Role of miR-200c/miR-141 in the regulation of
epithelial-mesenchymal transition and migration in head and neck
squamous cell carcinoma. Int J Mol Med. 33:879–886. 2014.PubMed/NCBI
|