Introduction
Colorectal cancer (CRC) is one of the leading causes
of death in the world, and is the third most common cancer in the
United States (1). Recent US data
on CRC incidence are alarming, with an estimated 103,170 cases,
including 51,690 deaths, in 2012 (2), despite the increased use of early
detection techniques. Therefore, it is essential to develop more
aggressive prevention strategies as well as novel agents for CRC
treatment. In recent years, natural products have received great
attention as potential agents for cancer prevention or therapy
owing to their health benefits and appreciably reduced toxicity and
side effects, the major known limitations of most current
chemotherapeutic agents (3).
Natural compounds that block or suppress the proliferation of tumor
cells and/or induce apoptosis are deemed to have potential as
antitumor agents (4). Plumbagin
(5-hydroxy-2-methyl-1, 4-naphthoquinone) occurs naturally in the
medicinal herb Plumbago zeylinica, which has been safely
used for centuries in Indian Ayurvedic and Oriental medicine for
the treatment of various ailments (5–9).
Plumbagin has attracted a great deal of research interest due to
its known pharmacological effects as an anti-bacterial,
hypolipidaemic, anti-atherosclerotic, Leishmanicidal and anticancer
compound (10–16). In addition Plumbagin has been shown
to be a potent radiosensitizer (17–19).
In the present study, we explored the possible anticancer activity
of Plumbagin on HCT116 colon cancer cells by analyzing its effects
on cell cycle regulation, the expression of apoptosis-related
signaling molecules and the formation of reactive oxygen species
(ROS). Our data showed that Plumbagin had a dual effect on HCT116
cells: it caused G1 phase arrest by downregulating the expression
of cyclin B1, cyclin D1 and NF-κB, and simultaneously promoted
apoptosis by upregulating effectors of the mitochondrial pathway
and inducing ROS formation. The magnitude of the apoptotic effect
of Plumbagin was greater when cells were kept in suspension,
whereas cell cycle effects predominated when treatments were
performed on attached cells. As it was previously reported that
Plumbagin does not show any toxicity on normal colon cells
(20), our data suggest that, on
the basis of predominant killing of cancer cells, Plumbagin should
be considered as a promising drug for the treatment for colon
cancer.
Materials and methods
Reagents
Plumbagin and DMSO, with purity >97%, were
purchased from Sigma-Aldrich (St. Louis, MO, USA). A 100 mM stock
solution of Plumbagin was prepared in DMSO, stored as small
aliquots at −20°C, and then diluted as needed into cell culture
medium. Penicillin-streptomycin solution, RPMI-1640 medium and
fetal bovine serum (FBS) were obtained from CellGro (Manassas, VA,
USA). Antibodies against AKT, BIM, PARP1, NF-κB, cyclin B1, BCL2,
cleaved PARP1, caspase 3, cleaved caspase 3, caspase 9 and cleaved
caspase 9 were purchased from Cell Signaling Technology (Beverly,
MA, USA). The antibody to cyclin D1 was obtained from Santa Cruz
Biotechnology (Santa Cruz, CA, USA); the FAK and Src antibodies
were from BD Bioscience (San Jose, CA, USA), and the antibodies
against p53 and p21WAF1/CIP1 were obtained from
Epitomics (Burlingame, CA, USA) and EMD Millipore (Billerica, MA,
USA), respectively. Ethidium homodimer was obtained from
Sigma-Aldrich.
Cell culture
The human colon cancer cell line HCT116 was obtained
from the American Type Culture Collection (Manassas, VA, USA). All
experiments were performed within three passages of cells cultured
in RPMI-1640 supplemented with 10% FBS and 1% of a solution of
penicillin (100 U/ml) and streptomycin (100 mg/ml). Cultures were
incubated at 37°C, in an air atmosphere with 5% CO2 and
85% humidity.
Cytotoxicity assays
The sensitivity of HCT116 cells to Plumbagin was
determined by using the CellTiter-Glo® luminescent cell
viability assay in its 96-well format (Promega, Madison, WI, USA).
Cells (8×104) were exposed to different concentrations
(≤10 μM) of Plumbagin for 24 h. Cells treated with DMSO served as
control. After Plumbagin treatment, the CellTiter-Glo reagent (200
μl) was added to the culture medium in each well to induce cell
lysis. After 10 min at room temperature (RT), the luminescence was
recorded in a Berthold Microlumat Plus LV 96V luminometer from the
Genomics and Epigenomics Shared Resource of the Lombardi
Comprehensive Cancer Center (LCCC). Percentage of residual cell
viability was determined by the ratio luminescence of treated
cells/luminescence of control cells.
Flow cytometry analysis
For cell cycle analysis, cells were harvested 24 h
after exposure to Plumbagin, washed once in phosphate-buffered
saline (PBS), fixed in 75% ethanol, resuspended in PBS containing
20 μg/ml propidium iodide (EMD Millipore), and incubated 30 min at
37°C before being analyzed using a FACScan instrument (BD
Bioscience), at the LCCC Flow Cytometry and Cell Sorting Shared
Resource.
Ethidium bromide staining
The possibility that Plumbagin treatment may induce
apoptosis was evaluated by staining with ethidium homodimer
(EthD-1), a cell viability indicator with high affinity for DNA
that emits strong red fluorescence only in its DNA-bound state.
EthD-1 is impermeant to healthy cells, but will stain cells
undergoing apoptosis. HCT116 cells (8×104) exposed to
Plumbagin for 24 h as described above were stained with EthD-1 at
37°C for 1 h. The presence of red EthD-1 fluorescence was monitored
by fluorescence microscopy.
Detection of changes in mitochondrial
transmembrane potential
The possible disruption of the mitochondrial
potential in HCT116 cells by Plumbagin treatment was monitored
using the MitoCapture™ Apoptosis Detection kit (BioVision,
Milpitas, CA, USA) following the manufacturer’s instructions. This
fluorescence-based assay detects the disruption or total loss of
the mitochondrial transmembrane potential as one of the earliest
intracellular events that occur following stimulation of apoptotic
pathways. Cells were imaged immediately using an IX71 fluorescence
microscope at the LCCC Microscopy and Imaging Shared Resource.
Excitation was induced at either 478 or 507 nm, and emission (Em)
was monitored with FITC (Em, 512–535) and rhodamine filters (Em,
540–560 nm).
Western immunoblot analysis
Cells were lysed with RIPA buffer (50 mM Tris-HCl pH
7.4, 150 mM NaCl, 0.1% SDS, 1% deoxycholic acid, 1% NP-40, 1 mM
EDTA) supplemented with a protease inhibitor cocktail (Roche
Diagnostics, Indianapolis, IN, USA). Protein concentrations were
determined using the BCA assay (Pierce/Thermo Fisher, Rockford, IL,
USA) and a colorimetric plate reader. Extracts were electrophoresed
on 4–20% gradient or 10% Tris-glycine SDS-polyacrylamide gels (Life
Technologies, Grand Island, NY, USA), and the resolved polypeptides
were transferred to polyvinylidene fluoride membranes. Transfers
were performed at 25 V for 2 h at RT or 10 V overnight at 4°C.
Non-specific binding was blocked by incubation with a solution of
5% skim milk in PBS containing 0.1% Tween-20 (PBS-T) at RT. After
blocking, membranes were incubated with primary antibody and,
following washes with PBS-T, were then incubated with horseradish
peroxidase (HRP)-conjugated secondary antibodies, followed by
visualization using the ECL (Pierce/Thermo Fisher)
chemiluminescence detection system. Antibodies were prepared at the
appropriate dilutions in blocking solution. For re-probing,
membranes were incubated for 30 min at 50°C in stripping buffer (2%
SDS, 62.5 mM Tris (pH 6.7) and 100 mM β-mercaptoethanol), rinsed
thoroughly, and used again as described above.
Measurement of reactive oxygen
species
Intracellular ROS levels were determined using
dihydrodichloro-fluorescein diacetate (H2DCF-DA), which
is ultimately converted by oxidation into DCF, a fluorescent
compound, in the presence of ROS. Cells (1×106) treated
with Plumbagin as described above were incubated at 37°C for 30 min
with 10 μM H2DCF-DA, washed, resuspended in PBS, and
immediately analyzed for fluorescence intensity with a fluorescence
multi-well plate reader with excitation and emission wavelengths of
485 and 530 nm.
Statistical analysis
Unless otherwise indicated, data are given as the
mean ± standard error of the mean (SEM). All data were analyzed
using a two-tailed paired Student’s t-test or one-way ANOVA, and
values were considered to be statistically significant at
p≤0.05.
Results
Most cancer patients succumb to the consequences of
metastatic cancer progression rather than as a result of their
primary tumors (21). Cells
dissociate from the primary solid tumors, enter the lymphatic and
blood circulation, and disseminate through the body, ultimately
homing in distant organs and forming colonies of metastatic cells
(22). However, because normal
cells and most tumor cells are known to die by anoikis when
detached from an extracellular matrix (23,24),
it is suggested that in the initial phases of their transient
unattached state circulating tumor cells (CTCs) may in fact be
stressed and, consequently, they might be more susceptible to the
action of natural compounds with anticancer activity (25). In order to test any possible
differential effect of Plumbagin on attached or unattached CRC
cells, we utilized two treatment conditions: HCT116 cells were
exposed to Plumbagin either i) at the seeding time, when they will
remain unattached for several hours, or ii) 24 h after the cultures
had been established, when all cells were already attached to the
substrate.
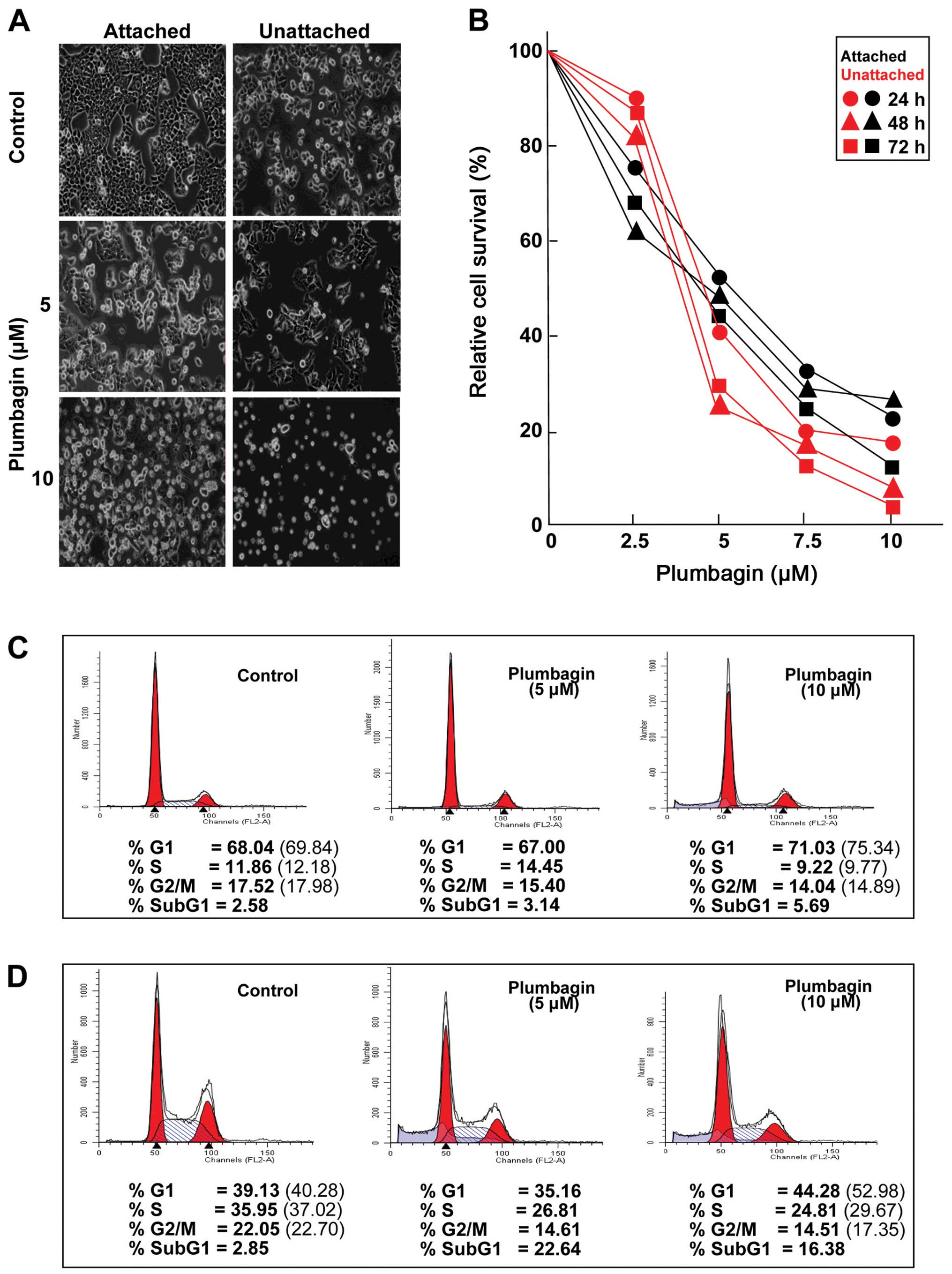
Plumbagin decreases the viability of CRC
cells in a dose-dependent manner
Regardless of the timing of addition, Plumbagin
exposure had substantial effects on both attached and unattached
HCT116 cells, which became obvious during morphological observation
by phase-contrast microscopy (Fig.
1A) as well as in experiments designed to determine the
viability of cultures treated with increasing drug concentrations
(Fig. 1B). Plumbagin treatments
for ≤72 h resulted in dose-dependent decreases in the viability of
both attached and unattached cells, although unattached cells
appeared to be somewhat more susceptible to the drug than the
attached cells (Fig. 1B). On the
basis of these results, two Plumbagin concentrations (5 and 10 μM)
were chosen for further experiments. Cell cycle analyses were
carried out to determine whether the observed effects on cell
viability may be related to alterations in cell proliferation. The
fact that Plumbagin exposure caused G1 arrest in cultures of either
attached (Fig. 1C) or unattached
(Fig. 1D) cells became apparent as
early as 24 h after treatment initiation, even when the lowest (5
μM) concentration was used. Relative to vehicle-treated cultures,
when considering only actively cycling cells, it became clear that
exposure to 10 μM Plumbagin increased the percentage of attached
cells (Fig. 1C) in G1 (7.9%), with
concomitant decreases in the proportion of cells in the S (19.8%)
and G2/M (17.2%) phases. The cell cycle effects of Plumbagin (10
μM) on unattached cells (Fig. 1D)
were even more pronounced, showing a 31.5% increase in G1 cells,
paralleled by decreases in the S (20.1%) and G2/M (23.6%) cell
populations. In addition, comparisons of the relative proportions
of cells detectable in the sub-G1 populations of attached (Fig. 1C) and unattached (Fig. 1D) cells in control and Plumbagin
treated (10 μM) cultures revealed 2.24- and 6.19-fold increases,
respectively, suggesting that cell death may also play a role in
reducing the viability of HCT116 cells after drug exposure.
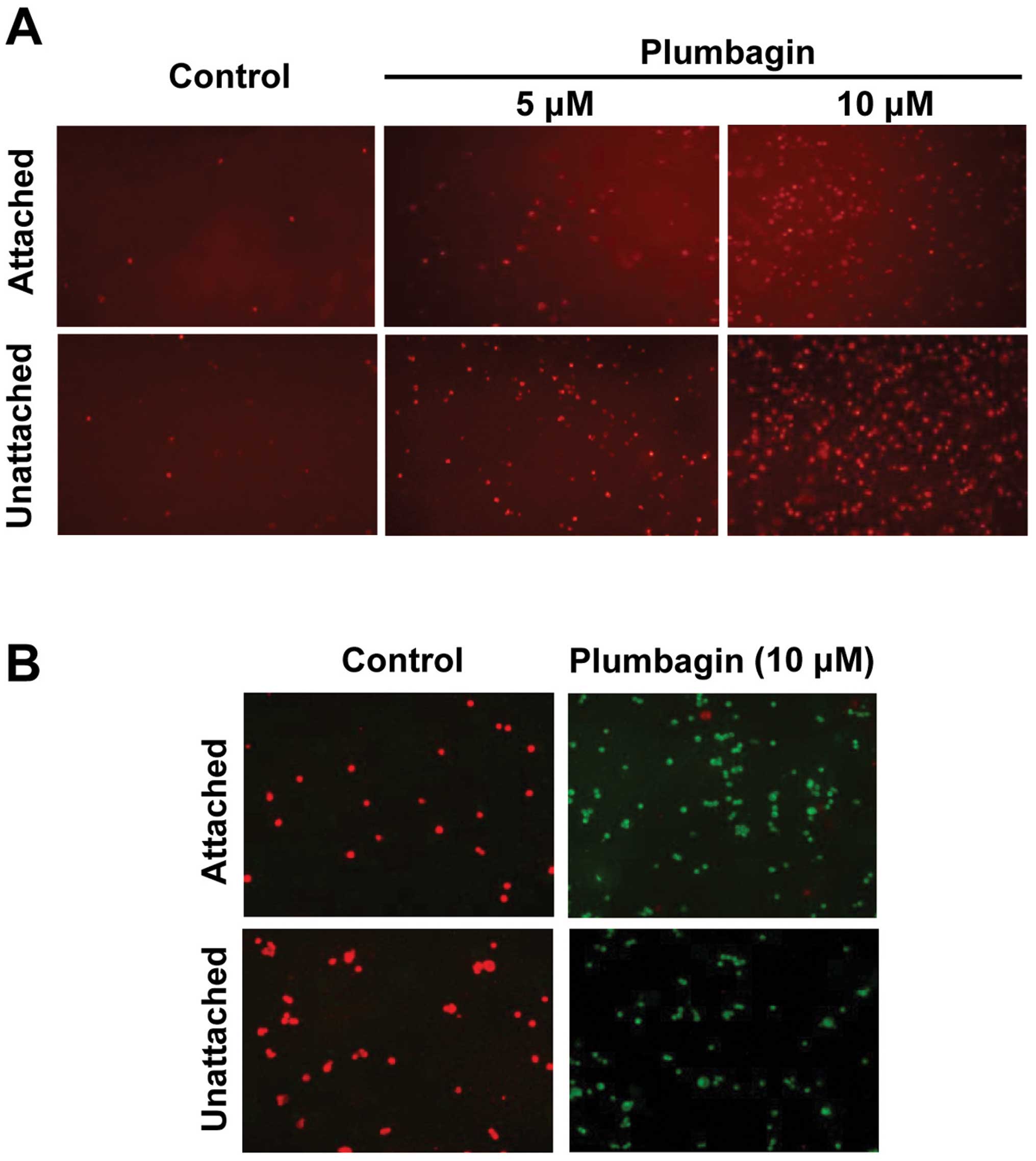
Plumbagin treatment promotes apoptotic
death of CRC cells
To explore whether Plumbagin was causing the extent
of cell death observed during cell cycle analyses (Fig. 1C and D) by promoting apoptosis,
control and drug-treated HCT116 cells were subjected to ethidium
bromide staining 24 h after treatment initiation, using the
ethidium homodimer (EthD-1). This reagent is a cell impermeant
viability indicator, which is a high-affinity nucleic acid stain
that is weakly fluorescent until bound to DNA, when it emits
intense red fluorescence. Results (Fig. 2A) showed a dose-related increase in
fluorescence intensity that, in agreement with the relative
proportion of sub-G1 populations after similar treatments (Fig. 1C and D), was clearly more
pronounced in the case of Plumbagin-exposed unattached HCT116
cells. These findings demonstrated that Plumbagin exposure induced
apoptosis of HCT116 cells, and that unattached cells were more
susceptible to the drug treatment. To test the possible
mitochondrial involvement in the apoptotic mechanism of Plumbagin
action, control and drug-treated HCT116 cells were monitored using
the MitoCapture™ system. In healthy cells, this cationic dye
accumulates in mitochondria, yielding a bright red fluorescence,
whereas in cells undergoing apoptosis mediated by alterations in
the mitochondrial transmembrane potential it cannot aggregate in
the mitochondria and remains in the cytoplasm giving off green
fluorescence.
Results (Fig. 2B)
showed that, while untreated cells consistently yielded red
fluorescence only, almost no red-stained cells were detectable in
cultures treated with 10 μM Plumbagin. The fact that the same
pattern was observed in experiments using attached and unattached
cells indicated that in both cases the apoptotic process triggered
by Plumbagin exposure was mediated by alterations in the
mitochondrial transmembrane potential induced by the drug.
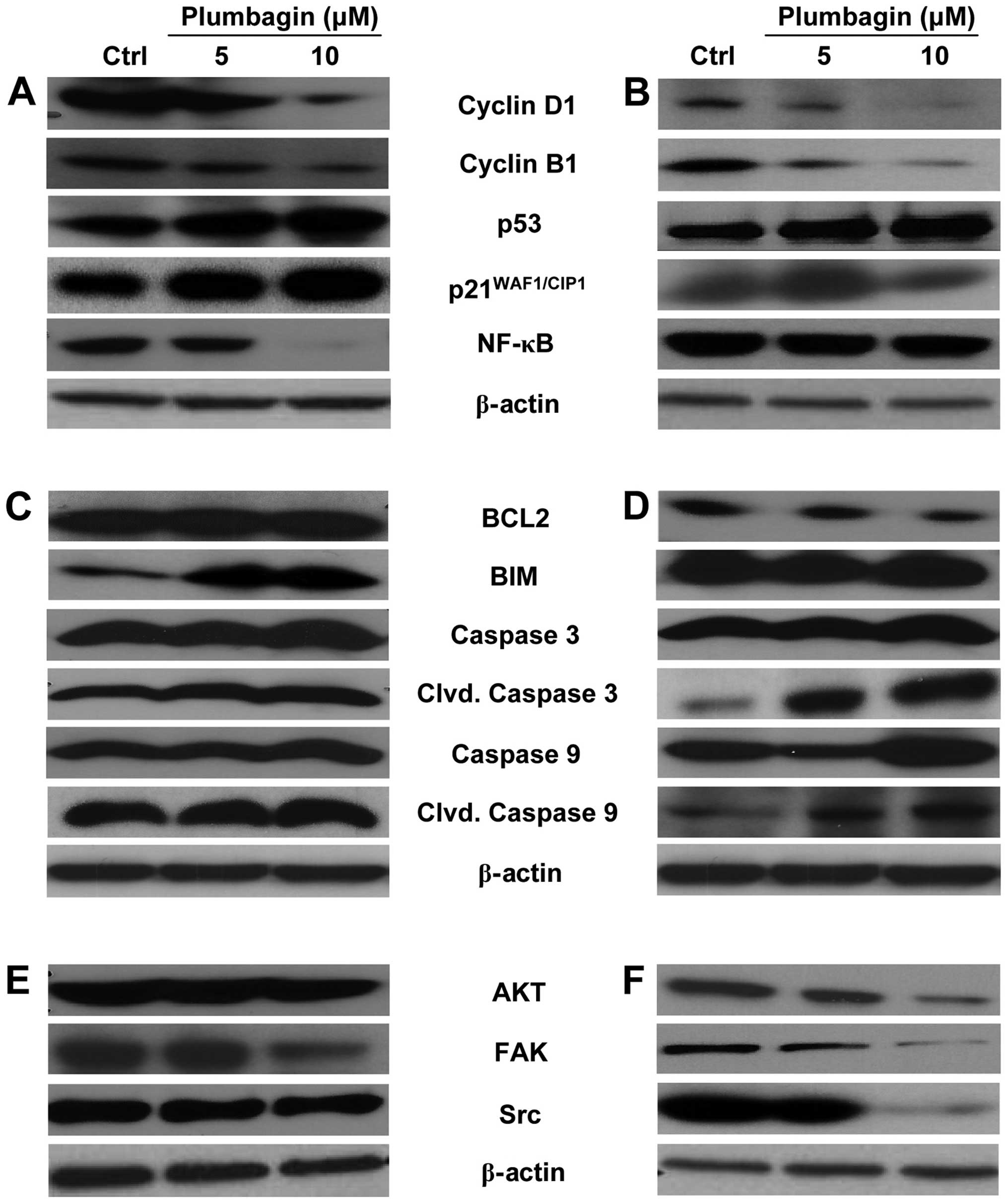
Plumbagin alters the expression of the
cell cycle, apoptosis and proliferation markers in CRC cells
Western immunoblot analyses were performed on total
cell extracts prepared from control and Plumbagin-treated cells, to
detect possible changes in the expression of specific gene products
that could be associated with the anti-proliferative and
apoptosis-inducing activity of the drug, and to identify possible
differences between the action of Plumbagin on attached and
unattached cells. As shown in Fig.
3, most Plumbagin-induced protein expression changes were
similar between attached (Fig. 3A, C
and E) and unattached (Fig. 3B, D
and F) HCT116 cells, relative to their respective untreated
controls. The differences observed were mainly related to the
intensity of the stimulatory or inhibitory effects than to the
nature of the protein products involved. With regard to cell
cycle-related markers, Plumbagin decreased the expression of cyclin
D1 and cyclin B1 and increased the expression of p53 and
p21WAF1/CIP1 in a dose-dependent manner to similar
extents in attached (Fig. 3A) and
unattached (Fig. 3B) cells.
However, the expression of NF-κB was almost completely abrogated in
attached HCT116 cells exposed to 10 μM Plumbagin (Fig. 3A), whereas it was essentially
unaffected by the same treatment in unattached cells (Fig. 3B). With regard to
apoptosis-associated markers, as expected from the more pronounced
pro-apoptotic activity of Plumbagin on unattached cells (Figs. 1 and 2), the expression of the anti-apoptotic
protein BCL2 was reduced by drug treatment in the unattached cells,
whereas it was essentially not modified by drug exposure in
attached cultures. In addition, the increases in cleaved caspase 3
and cleaved caspase 9 caused by Plumbagin treatment were more
pronounced in unattached cells (Fig.
3D) than in the attached cultures (Fig. 3C), although the total caspase 3 and
caspase 9 contents were not altered in any case. It seemed rather
interesting that the expression of the anti-apoptotic protein Bim
was increased by drug treatment in attached cells (Fig. 3C), while it was essentially
unchanged in unattached cells even after exposure to the highest
(10 μM) Plumbagin concentration (Fig.
3D). The expression of other proliferation-associated proteins
such as AKT, FAK and Src were markedly decreased by Plumbagin
treatment in unattached HCT116 cells, in a dose-dependent manner
(Fig. 3F), whereas unchanged AKT
expression levels and only changes of lower magnitude in the
expression of FAK and Src were detectable in the drug-treated,
attached cells (Fig. 3E).
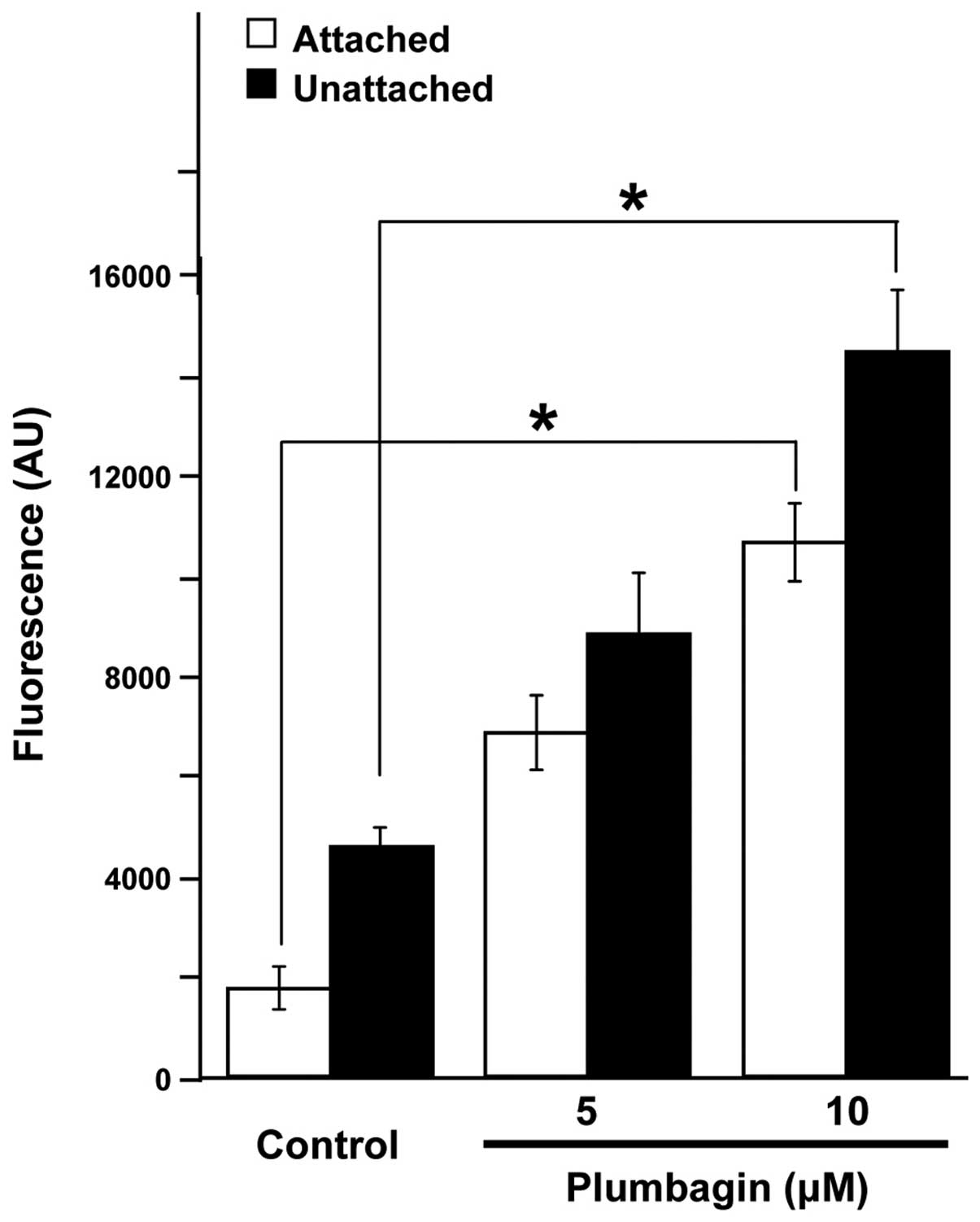
Plumbagin induces the production of ROS
by CRC cells
ROS have been suggested as possible triggers and/or
effectors of apoptosis (26,27).
To test whether the actions observed in CRC cells after Plumbagin
exposure could be mediated by production of the superoxide anion,
we determined the levels of intracellular ROS in control and
Plumbagin-treated HCT116 cells using H2DCF-DA, a
fluorescent dye which diffuses through cell membranes and in the
presence of ROS is hydrolyzed by intracellular esterases to DCF,
which is highly fluorescent. Results (Fig. 4) showed that Plumbagin exposure
increased the intracellular levels of ROS in both attached and
unattached HCT116 cells, in a dose-dependent manner, reaching
levels of production that were statistically significant
(p<0.01) in both experimental scenarios when treatments were
carried out by exposing the cells to 10 μM Plumbagin.
Discussion
Natural products are considered as one of the most
important sources of promising leads for the development of novel
anticancer chemotherapeutics. Of special interest are those of
plant origin, the so-called ‘nutraceuticals’ (28), that by being part of the human diet
have already demonstrated low toxicity levels. Studies of these
compounds, particularly those that may have the ability to
stimulate tumor cell apoptosis (4,29)
may lead to the discovery of new anticancer drugs among the large
pool of plant secondary metabolites, which provide a great variety
of chemical structures (30). Our
studies have focused on the effects of Plumbagin, a quinone derived
from plant secondary metabolites, on colon cancer cells that derive
from the tissue type most likely to be the immediate target of
Plumbagin and its metabolites after ingestion (31).
Although the potential anticancer activity of
Plumbagin has been tested on various human tumor types (32–37),
and despite the fact that studies in a rat model for
azoxymethane-induced intestinal carcinogenesis identified Plumbagin
as a promising chemopreventive agent (13), few studies on its effect on human
colon cancer have been reported (38,39).
Our results clearly demonstrate that Plumbagin has a potent action
against HCT116 colon cancer cells regardless of whether attached or
unattached cultures were exposed to the drug. In general, the
overall survival effects and G1 cell cycle arrest were quite
similar between attached and unattached cells (Fig. 1B–D), although there were also some
obvious differences. The extent of cell death caused by Plumbagin
on unattached cells was 3–4-fold higher than in drug-treated
attached cultures, as reflected by the proportion of sub-G1 cells
detectable in each case (Fig. 1C and
D). Nevertheless, the observed cell cycle and apoptosis effects
of Plumbagin were consistent with the changes detected, at the
protein level, in the expression of genes relevant to cell cycle
control, cell death response and the regulation of cell
proliferation (Fig. 3). In
general, the nature of the effects of Plumbagin on protein
expression detected in our case agreed with those reported earlier
on other colon cancer cell lines (38,39).
However, because we used lower concentrations of Plumbagin (≤10
μM), compared with the concentrations used in those studies (≤75
μM), it seems that HCT116 cells are more sensitive than HT-29 and
HCT15 cells. In addition, within our own experimental system there
was a high level of coherence among the various results obtained:
i) the cell cycle arrest observed was consistent with the decreased
levels of cyclins D1 and B1, together with the increased levels of
p53 and p21WAF1/CIP1; ii) the induction of apoptosis
through the involvement of mitochondrial pathways suggested by
fluorescence microscopy after specific cellular stains (Fig. 2) was consistent with increased
levels of cleaved-caspase 3 and cleaved-caspase 9; and iii) the
decreased expression of proliferation marker proteins was also
consistent with the global decrease in the size of the
proliferating cell population.
In general, the magnitude of the observed effects
was the main difference between the action of Plumbagin on attached
and unattached cells. However, it appears that there is a clear
difference in the mechanism by which the drug induces apoptosis
under the different experimental conditions: unaltered BCL2
expression (40) and increased BIM
expression (41) in the attached
cells, in contrast to decreased BCL2 and unaltered BIM expression
in the unattached cells. The fact that these differences were
observed with both Plumbagin concentrations used suggests that
another apoptosis-regulatory protein may be differentially affected
by the drug and contribute to the ultimate decision between the two
pro-apoptotic mechanisms.
Another important point of contrast, not only
between attached and unattached cells in our system, but also in
relation to the published literature on the anticancer action of
Plumbagin is the effect on NF-κB expression. Some reports indicated
that Plumbagin treatment increased NF-κB (38), whereas others described not only
that NF-κB was decreased by drug treatment (36,42),
but even that Plumbagin exposure suppressed the NF-κB increasing
effect of ionizing radiation (43). In our experimental system, NF-κB
remained essentially unchanged by Plumbagin treatment in unattached
cells (Fig. 3B), whereas it was
reduced to nearly undetectable levels by drug treatment in attached
cells (Fig. 3A). At present, there
is no obvious explanation for this discrepancy. Additional
experiment will be required to answer this question. What seems to
be universally consistent among data reported in the literature
(14,44) as well as in our own system
(Fig. 4) is the ability of
Plumbagin to promote the production of ROS, which agree with its
known pro-oxidant nature (45).
The levels of ROS production by Plumbagin-treated cells agree well
with the overall extent of survival inhibition caused by the drug
under attached and unattached culture conditions.
Overall, our data indicate that Plumbagin treatment
had a dual effect on HCT116 cells: the induction of ROS formation,
which promoted apoptosis via the mitochondrial cell death pathway,
and the simultaneous induction of cell cycle arrest at the G1 phase
with associated increases in the levels of p53 and
p21WAF1/CIP1. The fact that these effects were observed
both in attached cells as well as in cells maintained under
unattached conditions strongly suggests that Plumbagin treatment
may be effective not only on cells in primary solid tumors but also
on cells that may have dissociated from the primary tumors and are
transiently suspended in biological fluids on their way to homing
in distant tissues where they will establish metastatic colonies.
Therefore, it seems evident that Plumbagin is a promising
anticancer drug with potential therapeutic uses for the treatment
of CRC patients.
Acknowledgements
The authors express their gratitude to University
grant commission of India, Sanction number F.No. 37-265/2009 (SR)
13-1-2010, New Delhi, for providing grant to B.E. This study was
supported in part by grant R01-CA134727 from the US National Cancer
Institute to V.N., and by the Tissue Culture, Flow Cytometry,
Microscopy and Imaging, and Genomics and Epigenomics Shared
Resources of the Lombardi Comprehensive Cancer Center, which are
funded through USPHS grant P30-CA-51008.
References
|
1
|
Arber N and Levin B: Chemoprevention of
colorectal cancer; ready for routine use? Recent results. Cancer
Res. 166:213–230. 2005.PubMed/NCBI
|
|
2
|
Siegel R, Naishadham D and Jamal A: Cancer
statistics. CA Cancer J Clin. 62:10–29. 2012.
|
|
3
|
Mehta RG, Munillo G, Naithani R and
Xinjian P: Cancer chemoprevention by natural products: how far have
we come. Pharm Res. 27:950–961. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Frankfurt OS and Krishnan A:
Apoptosis-based drug screening and detection of selective toxicity
to cancer cells. Anticancer Drugs. 14:555–561. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wang YC and Huang TL: Screening of
anti-Helicobacter pylori herbs deriving from Taiwanese folk
medicinal plants. FEMS Immunol Med Microbiol. 43:51–59. 2005.
|
|
6
|
Wang YC and Huang TL: Anti-Helicobacter
pylori activity of Plumbago zeylanica L. FEMS Immunol
Med Microbiol. 43:407–412. 2005.
|
|
7
|
Dai Y, Hou L, Cheng YP and But PP:
Inhibition of immediate allergic reactions by ethanol extract from
Plumbago zeylanica stems. Biol Pharm Bull. 27:429–432. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chan-Bacab MJ and Pena-Rodriguez LM: Plant
natural products with leishmanicidal activity. Nat Prod Rep.
18:674–688. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
9
|
Krishnaswamy M and Purushothaman KK:
Plumbagin: a study of its anticancer, antibacterial and antifungal
properties. Indian J Exp Biol. 18:876–877. 1980.PubMed/NCBI
|
|
10
|
Sharma I, Gusain D and Dixit VP:
Hypolipidaemic and anti-atherosclerotic effects of plumbagin in
rabbits. Indian J Physiol Pharmacol. 35:59–63. 1991.
|
|
11
|
Parimala R and Sachdanandem P: Effect of
plumbagin on some glucose metabolizing enzymes studied in rats in
experimental hepatoma. Mol Cell Biochem. 125:10–14. 1996.
|
|
12
|
Naresh RA, Udupa N and Devi PU: Niosomal
plumbagin with reduced toxicity and improved anticancer activity in
BALB/C mice. J Pharmacol. 48:1128–1132. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sugie S, Okamoto K, Rahman KM, et al:
Inhibitory effects of plumbagin and juglone on azoxymethane-induced
intestinal carcinogenesis in rats. Cancer Lett. 127:177–183.
1998.PubMed/NCBI
|
|
14
|
Srinivas P, Gopinath G, Banerji A, Dinakar
A and Srinivas G: Plumbagin induces reactive oxygen species, which
mediate apoptosis in human cervical cancer cells. Mol Carcinog.
40:201–211. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kuo PL, Hsu YL and Cho CY: Plumbagin
induces G2-M arrest and autophagy by inhibiting the AKT/mammalian
target of rapamycin pathway in breast cancer cells. Mol Cancer
Ther. 5:3209–3221. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hsu YL, Cho CY, Kuo PL, Huang YT and Lin
CC: Plumbagin induces apoptosis and cell cycle arrest in A549 cells
through p53 accumulation via c-Jun NH2- terminal kinase-mediated
phosphorylation at serine 15 in vitro and in vivo. J Pharmacol Exp
Ther. 318:484–494. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Devi PU, Rao BS and Solomon FE: Effect of
plumbagin on the radiation induced cytogenetic and cell cycle
changes in mouse Ehrlich ascites carcinoma in vivo. Indian J Exp
Biol. 36:891–895. 1998.PubMed/NCBI
|
|
18
|
Prasad VS, Devi PU, Rao BS and Kamath R:
Radiosensitizing effect of Plumbagin on mouse melanoma cells grown
in vitro. Indian J Biol. 34:857–858. 1996.PubMed/NCBI
|
|
19
|
Ganasoundari A, Zare SM and Devi PU:
Modification of bone marrow radiosensitivity by medicinal plant
extracts. Br J Radiol. 70:599–602. 1996. View Article : Google Scholar
|
|
20
|
Subramaniya BR, Srinivas G, Sadullah SSM,
et al: Apoptosis inducing effect of Plumbagin on colonic cancer
cells depends on expression of COX-2. PLoS One. 6:1–11. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Seyfried TN and Huysentruyt LC: On the
origin of cancer metastasis. Crit Rev Oncog. 18:43–73. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Nguyen DX, Bos PD and Massagué J:
Metastasis: from dissemination to organ-specific colonization. Nat
Rev Cancer. 9:274–284. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Liotta LA and Kohn E: Anoikis: cancer and
the homeless cell. Nature. 430:973–974. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Konstantopoulos K and Thomas SN: Cancer
cells in transit: the vascular interactions of tumor cells. Annu
Rev Biomed Eng. 11:177–202. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jiang YL and Liu ZP: Natural products as
anti-invasive and anti-metastatic agents. Curr Med Chem.
18:808–829. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Simon HU, Haj-Yehia A and Levi-Schaffer F:
Role of reactive oxygen species (ROS) in apoptosis induction.
Apoptosis. 5:415–418. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Circu ML and Aw TY: Reactive oxygen
species, cellular redox systems and apoptosis. Free Radic Biol Med.
48:749–762. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gupta SC, Kim JH, Prasad S and Aggarwal
BB: Regulation of survival, proliferation, invasion, angiogenesis,
and metastasis of tumor cells through modulation of inflammatory
pathways by nutraceuticals. Cancer Metastasis Rev. 29:405–434.
2010. View Article : Google Scholar
|
|
29
|
Kaufmann SH and Earnshaw WC: Induction of
apoptosis by cancer chemotherapy. Exp Cell Res. 256:42–49. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lu JJ, Bao JL, Wu GS, et al: Quinones
derived from plant secondary metabolites as anti-cancer agents.
Anticancer Agents Med Chem. 13:456–463. 2013.PubMed/NCBI
|
|
31
|
Padhye S, Dandawate P, Yusufi M, Ahmad A
and Sarkar FH: Perspectives on medicinal properties of Plumbagin
and its analogs. Med Res Rev. 32:1131–1158. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Li J, Shen L, Lu FR, et al: Plumbagin
inhibits cell growth and potentiates apoptosis in human gastric
cancer cells in vitro through the NF-κB signaling pathway. Acta
Pharmacol Sin. 33:242–249. 2012.PubMed/NCBI
|
|
33
|
Hafeez BB, Jamal MS, Fischer JW, Mustafa A
and Verma AK: Plumbagin, a plant derived natural agent inhibits the
growth of pancreatic cancer cells in vitro and in vivo via
targeting EGFR, Stat3 and NF-κB signaling pathways. Int J Cancer.
131:2175–2186. 2012.PubMed/NCBI
|
|
34
|
Sinha S, Pal K, Elkhanany A, et al:
Plumbagin inhibits tumorigenesis and angiogenesis of ovarian cancer
cells in vivo. Int J Cancer. 132:1201–1212. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Abedinpour P, Baron VT, Chrastina A, Welsh
J and Borgström P: The combination of Plumbagin with androgen
withdrawal causes profound regression of prostate tumors in vivo.
Prostate. 73:489–499. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Xu TP, Shen H, Liu LX and Shu YQ:
Plumbagin from Plumbago zeylanica L induces apoptosis in
human non-small cell lung cancer cell lines through NF-κB
inactivation. Asian Pac J Cancer Prev. 14:2325–2331. 2013.
|
|
37
|
Sagar S, Esau L, Moosa B, Khashab NM,
Bajic VB and Kaur M: Cytotoxicity and apoptosis induced by a
Plumbagin derivative in estrogen positive MCF-7 breast cancer
cells. Anticancer Agents Med Chem. 14:170–180. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Subramaniya BR, Srinivasan G, Sadullah SS,
et al: Apoptosis inducing effect of plumbagin on colonic cancer
cells depends on expression of COX-2. PLoS One. 6:e186952011.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Chen MB, Zhang Y, Wei MX, et al:
Activation of AMP-activated protein kinase (AMPK) mediates
plumbagin-induced apoptosis and growth inhibition in cultured human
colon cancer cells. Cell Signal. 25:1993–2002. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wu Y, Mehew JW, Heckman CA, Arcinas M and
Boxer LM: Negative regulation of bcl-2 expression by p53 in
hematopoietic cells. Oncogene. 20:240–251. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Mérino D, Giam M, Hughes PD, et al: The
role of BH3-only protein Bim extends beyond inhibiting Bcl-2-like
prosurvival proteins. J Cell Biol. 186:355–362. 2009.PubMed/NCBI
|
|
42
|
Sandur SK, Ichikawa H, Sethi G, Ahn KS and
Aggarwal BB: Plumbagin (5-hydroxy-2-methyl-1,4-naphthoquinone)
suppresses NF-κB activation and NF-κB-regulated gene products
through modulation of p65 and IκBα kinase activation, leading to
potentiation of apoptosis induced by cytokine and chemotherapeutic
agents. J Biol Chem. 281:17023–17033. 2006.PubMed/NCBI
|
|
43
|
Nambiar D, Rajamani P and Singh RP:
Effects of phytochemicals on ionization radiation-mediated
carcinogenesis and cancer therapy. Mutat Res. 728:139–157. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Powolny AA and Singh SV: Plumbagin-induced
apoptosis in human prostate cancer cells is associated with
modulation of cellular redox status and generation of reactive
oxygen species. Pharm Res. 25:2171–2180. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Checker R, Sharma D, Santosh SK, et al:
Plumbagin inhibits proliferative and inflammatory response of T
cells independent of ROS generation but by modulating intracellular
thiols. J Cell Biochem. 110:1082–1093. 2011. View Article : Google Scholar
|