Introduction
Animal models have been used in front-line
preclinical studies for predicting efficacy and possible toxicities
of anticancer drugs in cancer patients (1,2).
Current tumor models used for drug evaluation generally consist of
implantation into immunodeficient mice of xenografts generated from
well-established human cancer cell lines that have already adapted
to in vitro growth. These models have been used extensively
for decades for rapid screening of anticancer drug efficacy
(3,4).
In recent years, xenografts derived from engrafting
fresh surgical specimens directly into immunodeficient mice have
enabled the development of more relevant in vivo models for
human tumors (5). Such
patient-derived xenograft (PDX) models, established by direct
transfer of tumor tissue, retain similar morphology, architecture,
and molecular signatures as the original cancers and thus should be
used for rapid screening of potential therapeutics (6,7).
Whereas the conventional xenograft models using cell lines provide
only a monoclonal mass of tumor cells, PDX models recapitulate not
only interactions from the host microenvironments but also the
cancerous heterogeneity including the cancer stem cells (5,6,8).
Results from these investigations support the use of direct
transfer xenografts as a reliable strategy to anticipate clinical
findings, provide direction for optimizing personalized treatment
in advanced cancers, and suggest novel treatment opportunities in
patients with no other therapeutic options (9). The advantages of PDX models in
preserving cancer stem cells and the clinical information of the
donor patient (so-called ‘cancer xenopatient’) may allow for
accelerated cancer research by simulating the situation in cancer
patients more closely (6,7).
However, the establishment of direct xenografts is
still technically difficult (1,10,11).
Recently, a new immunodeficient animal model,
NOD/Shi-scid/IL-2Rγnull (NOG) mice, derived from the
NOD/SCID mouse with a common gamma chain, has been introduced. In
addition to lacking functional T and B lymphocytes, the NOG mouse
has multifunctional defects in natural killer cell activity,
macrophage function, complement activity, and dendritic cell
function (12). NOG mice were
reported to be the most appropriate immunodeficient host animal for
direct xenografting of fresh tumor tissue (5).
In the present study, we investigated the efficient
establishment of PDX using NOG mice with clinical factors of
xenotransplantation. We also discuss herein the application of this
newly developed system for not only reliable preclinical studies of
new anticancer drugs but also personalized anti-cancer
therapies.
Materials and methods
Tumor tissues for transplantation
The 116 surgically removed fresh tumor tissues for
transplantation were obtained at Kanagawa Cancer Center (Yokohama,
Kanagawa, Japan) and Kawasaki Municipal Hospital (Kawasaki,
Kanagawa, Japan) with the patients’ written informed consent for
the study. The study was performed in collaboration with Keihin
Coastal Area Life Innovation Comprehensive Special Zones for
International Competitiveness Development (Japan) from 2011 to
2012. The ethics committees independently approved the study
(authorization number: 176 at Kanagawa Cancer Center, 23–410 at
Kawasaki Municipal Hospital). The entire list of engrafted tumors
with the patient profiles is shown in Table I.
 | Table IThe entire list of patients from which
the engrafted tumors were taken and the fate of the xenografts. |
Table I
The entire list of patients from which
the engrafted tumors were taken and the fate of the xenografts.
| No. | Age | Gender | Original tumor
site | Pathology |
Primary/Metastasis | Tumor type | Result |
|---|
| 1 | 43 | M | Lung | Adenosquamous
carcinoma | Brain metastasis | Epithelial | Established |
| 2 | 60 | M | Lung | Adenocarcinoma | Brain metastasis | Epithelial | Established |
| 3 | 69 | M | Lung | Adenocarcinoma | Brain metastasis | Epithelial | Failedc |
| 4 | 35 | F | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 5 | 51 | F | Breast | Ductal
carcinoma | Brain
metastasis | Epithelial | Established |
| 6 | 62 | M | Prostate | Adenocarcinoma | Primary | Epithelial | Failed |
| 7 | 65 | M | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 8 | 76 | F | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 9 | 60 | F | Lung | Adenocarcinoma | Brain
metastasis | Epithelial | Failedc |
| 10 | 66 | F | Lung | Adenocarcinoma | Brain
metastasis | Epithelial | Established |
| 11 | 74 | F | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 12 | 58 | M | Nerve | MPNST | Primary | Mesenchymal | Established |
| 13 | 28 | M | Bone | Ewing/PNET | Brain
metastasis | Mesenchymal | Established |
| 14 | 58 | M | Thyroid | Papillary
carcinoma | Brain
metastasis | Epithelial | Failed |
| 15 | 76 | M | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Failedc |
| 16 | 65 | F | Breast | Ductal
carcinoma | Brain
metastasis | Epithelial | Failed |
| 17 | 69 | F | Large
intestine | Tubular
adenocarcinoma | Brain
metastasis | Epithelial | Established |
| 18 | 71 | F | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 19 | 74 | M | Esophagus | Squamous cell
carcinoma | Brain
metastasis | Epithelial | Established |
| 20 | 68 | M | Kidney | Renal cell
carcinoma | Brain
metastasis | Epithelial | Established |
| 21 | 81 | F | Lung | Adenocarcinoma | Brain
metastasis | Epithelial | Failedc |
| 22 | 80 | M | Small
intestine | GIST | Primary | Mesenchymal | Failed |
| 23 | 55 | M | Prostate | Adenocarcinoma | Primary | Epithelial | Failed |
| 24 | 65 | F | Breast | Ductal
carcinoma | Brain
metastasis | Epithelial | Failed |
| 25 | 51 | M | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 26 | 66 | M | Pancreas | Ductal
carcinoma | Primary | Epithelial | Failed |
| 27 | 40 | F | Brain | Glioblastoma | Primary | Mesenchymal | Failed |
| 28 | 61 | M | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 29 | 43 | M | Brain | Glioblastoma | Primary | Mesenchymal | Failed |
| 30 | 60 | M | Stomach | GIST | Peritoneal
metastasis | Mesenchymal | Established |
| 31 | 77 | M | Stomach | Tubular
adenocarcinoma | Peritoneal
metastasis | Epithelial | Established |
| 32 | 46 | M | Brain | Astrocytoma | Primary | Mesenchymal | Failed |
| 33 | 61 | F | Duodenum | Tubular
adenocarcinoma | Primary | Epithelial | Failed |
| 34 | 65 | F | Breast | Ductal
carcinoma | Brain
metastasis | Epithelial | Failed |
| 35 | 64 | M | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 36 | 69 | F | Lung | Squamous cell
carcinoma | Brain
metastasis | Epithelial | Established |
| 37 | 69 | F | Uterus body | Adenocarcinoma | Brain
metastasis | Epithelial | Established |
| 38a | 58 | F | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 39 | 70 | F | Large
intestine | Tubular
adenocarcinoma | Brain
metastasis | Epithelial | Established |
| 40 | 47 | M | Primary
unknown | Adenocarcinoma | Brain
metastasis | Epithelial | Established |
| 41 | 71 | F | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 42 | 71 | F | Uterus body | Adenocarcinoma | Brain
metastasis | Epithelial | Failedc |
| 43 | 55 | F | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 44 | 52 | M | Lung | Small cell
carcinoma | Brain
metastasis | Epithelial | Established |
| 45 | 68 | M | Prostate | Adenocarcinoma | Primary | Epithelial | Failed |
| 46 | 73 | M | Lung | Adenocarcinoma | Brain
metastasis | Epithelial | Failed |
| 47 | 73 | M | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 48 | 55 | F | Breast | Ductal
carcinoma | Brain
metastasis | Epithelial | Failed |
| 49 | 68 | M | Stomach | Tubular
adenocarcinoma | Brain
metastasis | Epithelial | Failedc |
| 50 | 52 | M | Large
intestine | GIST | Primary | Mesenchymal | Failed |
| 51 | 53 | F | Nerve | MPNST | Primary | Mesenchymal | Failed |
| 52 | 64 | M | Large
intestine | Tubular
adenocarcinoma | Brain
metastasis | Epithelial | Failed |
| 53 | 62 | F | Pancreas | Ductal
carcinoma | Primary | Epithelial | Established |
| 54b | 70 | M | Pancreas | Ductal
carcinoma | Lymph node
metastasis | Epithelial | Failed |
| 55b | 70 | M | Pancreas | Ductal
carcinoma | Lymph node
metastasis | Epithelial | Established |
| 56b | 70 | M | Pancreas | Ductal
carcinoma | Lymph node
metastasis | Epithelial | Established |
| 57 | 35 | F | Stomach | GIST | Primary | Mesenchymal | Failed |
| 58 | 64 | M | Lung | Large cell
carcinoma | Brain
metastasis | Epithelial | Established |
| 59 | 74 | F | Pancreas | Anaplastic
carcinoma | Primary | Epithelial | Failed |
| 60 | 71 | F | Pancreas | Ductal
carcinoma | Primary | Epithelial | Established |
| 61 | 74 | F | Pancreas | Ductal
carcinoma | Primary | Epithelial | Failed |
| 62 | 70 | M | Kidney | Transitional cell
carcinoma | Brain
metastasis | Epithelial | Failed |
| 63 | 53 | F | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Failed |
| 64 | 85 | F | Stomach | GIST | Primary | Mesenchymal | Failed |
| 65 | 67 | M | Kidney | Renal cell
carcinoma | Brain
metastasis | Epithelial | Established |
| 66 | 82 | M | Lung | Adenocarcinoma | Brain
metastasis | Epithelial | Established |
| 67 | 61 | M | Kidney | Renal cell
carcinoma | Peritoneal
metastasis | Epithelial | Established |
| 68 | 70 | F | Stomach | GIST | Primary | Mesenchymal | Failed |
| 69 | 64 | F | Brain | Glioblastoma | Primary | Mesenchymal | Failed |
| 70 | 49 | M | Pancreas | Ductal
carcinoma | Primary | Epithelial | Failed |
| 71 | 61 | F | Lung | Adenocarcinoma | Brain
metastasis | Epithelial | Established |
| 72 | 70 | F | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Failedc |
| 73 | 72 | F | Large
intestine | GIST | Primary | Mesenchymal | Failed |
| 74 | 69 | F | Stomach | GIST | Primary | Mesenchymal | Failed |
| 75 | 67 | F | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 76 | 79 | M | Brain | Glioblastoma | Primary | Mesenchymal | Failed |
| 77 | 60 | M | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 78 | 63 | F | Gallbladder | Pleomorphic
carcinoma | Brain
metastasis | Epithelial | Failedc |
| 79 | 37 | F | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Failed |
| 80 | 70 | M | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 81 | 68 | F | Stomach | GIST | Primary | Mesenchymal | Failed |
| 82 | 63 | M | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Failed |
| 83 | 70 | F | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Failed |
| 84 | 60 | F | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Failed |
| 85 | 56 | M | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 86 | 58 | F | Breast | Ductal
carcinoma | Brain
metastasis | Epithelial | Established |
| 87 | 16 | F | Stomach | GIST | Primary | Mesenchymal | Failed |
| 88 | 62 | M | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Failed |
| 89 | 71 | M | Pancreas | Ductal
carcinoma | Primary | Epithelial | Established |
| 90 | 65 | F | Kidney | Renal cell
carcinoma | Skin
metastasis | Epithelial | Failed |
| 91 | 51 | M | Pancreas | Ductal
carcinoma | Primary | Epithelial | Failed |
| 92 | 51 | F | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Failed |
| 93 | 75 | M | Lung | Adenocarcinoma | Brain
metastasis | Epithelial | Failed |
| 94 | 80 | M | Pancreas | Ductal
carcinoma | Primary | Epithelial | Failed |
| 95 | 77 | M | Kidney | Renal cell
carcinoma | Skin
metastasis | Epithelial | Established |
| 96 | 68 | M | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 97 | 63 | M | Pancreas | Ductal
carcinoma | Primary | Epithelial | Failed |
| 98 | 61 | F | Pancreas | Ductal
carcinoma | Primary | Epithelial | Failed |
| 99 | 67 | F | Pancreas | Ductal
carcinoma | Primary | Epithelial | Established |
| 100 | 61 | M | Lung | Adenocarcinoma | Brain
metastasis | Epithelial | Established |
| 101 | 71 | M | Stomach | Tubular
adenocarcinoma | Brain
metastasis | Epithelial | Established |
| 102a | 59 | F | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 103 | 63 | M | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 104 | 61 | F | Thyroid | Follicular
carcinoma | Brain
metastasis | Epithelial | Established |
| 105 | 71 | M | Pancreas | Ductal
carcinoma | Primary | Epithelial | Established |
| 106 | 46 | M | Large
intestine | Mucinous
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 107 | 66 | M | Pancreas | Ductal
carcinoma | Primary | Epithelial | Established |
| 108 | 80 | F | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 109 | 65 | M | Pancreas | Ductal
carcinoma | Primary | Epithelial | Established |
| 110 | 81 | F | Thyroid | Anaplastic
carcinoma | Primary | Epithelial | Established |
| 111 | 71 | M | Pancreas | Ductal
carcinoma | Primary | Epithelial | Established |
| 112 | 54 | F | Breast | Ductal
carcinoma | Brain
metastasis | Epithelial | Failed |
| 113 | 62 | M | Large
intestine | Tubular
adenocarcinoma | Liver
metastasis | Epithelial | Established |
| 114 | 68 | M | Lung | Adenocarcinoma | Brain
metastasis | Epithelial | Established |
| 115 | 63 | M | Large
intestine | Tubular
adenocarcinoma | Brain
metastasis | Epithelial | Established |
| 116 | 78 | M | Kidney | Renal cell
carcinoma | Brain
metastasis | Epithelial | Failed |
Animals
NOG mice, between 6 and 12 weeks of age, were used
in this study. The mice were obtained from the Central Institute
for Experimental Animals (CIEA; Kanagawa, Japan) (12). All animals were housed in plastic
cages (136×208×115 mm) within a vinyl isolator system (1150×500×500
mm) in a pathogen-free state, at a temperature of 22±1°C with
45±10% humidity, and a 12 h light/12 h dark cycle. All experiments
involving laboratory animals were performed in accordance with the
care and use guidelines of the CIEA, according to our previous
studies (13–15). These guidelines meet the generally
accepted international criteria on humane treatment that spare the
animal needless pain and suffering, and require confirmation that
the experiments conducted are of actual scientific benefit to
humankind.
Procedures for the establishment of PDX
models by serial engraftment
Fresh tumor tissues were divided into three pieces
under sterile conditions. One piece of each tissue specimen was
immediately placed in Dulbecco’s modified minimal essential medium
without antibiotics and without fetal bovine serum, and stored at
4°C until engrafting. Another piece was cryopreserved for molecular
biological examination, and the last piece was fixed in 4%
formaldehyde for histopathological examination. The piece for
engraftment was further divided into small pieces (~8–64
mm3) using sterilized surgical scissors. A small
incision was made in the leg of each mouse and a transplant needle
was inserted until the tip reached the dorsal subcutaneous area.
Approximately 10 pieces of tumor tissue were inoculated into the
dorsal subcutis via the needle. After the engrafted mass expanded
to over quadruple its size, the xenograft tumor was harvested and
directly re-transplanted for expansion in later serial generations
using the same procedure. After the tumor tissue had been passaged
three times or more and histopathological examination confirmed the
PDX to be a growing human tumor, we considered the PDX line as
‘established’. The established PDX tissue was divided into small
pieces, completely submerged in cryopreservation medium
(Cellbanker®1, Zenoaq, Fukushima, Japan), and then
stored in liquid nitrogen. The frozen tissues were later thawed and
used for experiments including re-transplantation and expansion.
Mice that did not develop tumor mass over six months after
engraftment were sacrificed as ‘failed’, and this was confirmed
histopathologically.
Morphological examination of the primary
engrafts and the PDX descendants
For morphological analyses, sample tissues were
formalin-fixed, paraffin-embedded (FFPE), sliced into 4-μm
sections, and subjected to standard hematoxylin and eosin (H&E)
staining or immunohistochemistry (IHC). IHC was performed using the
Bond Polymer Refine Detection system (Leica Microsystems, Tokyo,
Japan) according to the manufacturer’s instructions. Nuclei were
counterstained with hematoxylin. Primary antibodies used for IHC
were: monoclonal anti-HLA class 1-A, B, C (Hokudo, Sapporo, Japan),
rabbit polyclonal anti-c-kit (Nichirei Biosciences, Tokyo, Japan);
monoclonal anti-CD34, clone NU-4A1 (Nichirei Biosciences);
monoclonal antileukocyte common antigen, clone PD7/26, 2B11 (CD111,
Nichirei Biosciences); HER2 (Hercep Test™, Dako, Japan), monoclonal
anti-estrogen receptor (ER), clone 1D5 (Nichirei Biosciences); and
monoclonal antiprogesterone receptor (PgR), clone A9621A (Nichirei
Biosciences). Chromogenic in situ hybridization (ISH) for
Epstein-Barr virus (EBV)-encoded RNA (EBER) was performed using the
EBER 1 DNP probe (Ventana/Roche, Tuscon, AZ, USA) and the ISH iView
blue plus detection kit (Ventana/Roche) according to the provider’s
instructions.
Genetic examination of xenograft tumors
in NOG mice
The exon 11 deletion mutation in the KIT gene
in the 3rd generation xenograft of the gastrointestinal stromal
tumor (GIST) was investigated as previously described (16). Briefly, DNA was extracted from the
FFPE thin sections of the xenograft tumor and amplified by
polymerase chain reaction (PCR) with primers:
5′-gactgagacaataattattaaaag-3′ (forward) and
5′-acccaaaaaggtgacatggaaagc-3′ (reverse). PCR products were then
directly sequenced using the PCR primers and the Sanger’s method
with Genetic Analyzer 3100 (Applied Biosystems/Hitachi, Japan). For
EWS-FLI1 fusion mRNA detection, total RNA was extracted from the
3rd generation xenograft of the Ewing sarcoma/primitive
neuroectodermal tumor (PNET), reverse transcribed to cDNA, and
PCR-amplified with primers: EWS-exon 8
(5′-tcctacagccaagctccaagtc-3′) and the FLI1 exon 9
(5′-gtgatacagctggcgttggc-3′). The obtained product was directly
sequenced as described for the KIT analysis.
Statistical analysis
Statistical comparisons of data sets were performed
by a two-sample t-test. The Chi-square test or the two-sided
Fisher’s probability exact test was applied for comparisons between
group frequencies. These analyses were performed using JMP version
11 software (SAS Institute Inc., Cary, NC, USA). P-values of
<0.05 were considered significant.
Results
Efficacy of PDX line establishment in NOG
mice
In total, 116 surgically removed tumor tissues were
engrafted in NOG mice (Tables I
and II). The group of patients
who provided tumors for this study comprised 63 men and 53 women,
with a mean age of 63 years. Thirty-seven tumors were obtained from
primary sites and 79 tumors were from metastases. Ninety-eight
tumors were epithelial (carcinomas) and 18 tumors were
nonepithelial (sarcomas). Tumor specimens were engrafted on the day
of surgery or 1–6 days after the surgical removal (owing to sample
transport and public holidays). The primary organ site of the
transplant together with the difference between the primary tumor
or metastasis and the fate (established or failed) are summarized
in Table III.
 | Table IISummary of the engrafted tumors and
the fate of xenografts. |
Table II
Summary of the engrafted tumors and
the fate of xenografts.
| Engrafted tumor
information | Established | Failed (LPL) | Total |
|---|
|
|
|
|
|---|
| Organ site | Type | Primary | Metastasis | Primary | Metastasis | Primary | Metastasis |
|---|
|
Gastrointestinal |
| Esophagus | Epithelial | 0 | 1 | 0 | 0 | 0 | 1 |
| Stomach | Epithelial | 0 | 2 | 0 | 1 (1) | 0 | 3 |
| Mesenchymal | 0 | 1 | 6 | 0 | 6 | 1 |
| Small
intestine | Epithelial | 0 | 0 | 1 | 0 | 1 | 0 |
| Mesenchymal | 0 | 0 | 1 | 0 | 1 | 0 |
| Large
intestine | Epithelial | 0 | 25 | 0 | 10 (2) | 0 | 35 |
| Mesenchymal | 0 | 0 | 2 | 0 | 2 | 0 |
| Other
digestive |
| Pancreas | Epithelial | 8 | 2 | 8 | 1 | 16 | 3 |
| Gallbladder | Epithelial | 0 | 0 | 0 | 1 (1) | 0 | 1 |
| Respiratory |
| Lung | Epithelial | 0 | 10 | 0 | 5 (3) | 0 | 15 |
| Breast and female
genital |
| Breast | Epithelial | 0 | 2 | 0 | 5 | 0 | 7 |
| Uterus | Epithelial | 0 | 1 | 0 | 1 (1) | 0 | 2 |
| Urologic |
| Kidney | Epithelial | 0 | 4 | 0 | 3 | 0 | 7 |
| Neurologic |
| Brain | Mesenchymal | 0 | 0 | 5 | 0 | 5 | 0 |
| Nerve | Mesenchymal | 1 | 0 | 1 | 0 | 2 | 0 |
| Others |
| Bone | Mesenchymal | 0 | 1 | 0 | 0 | 0 | 1 |
| Thyroid | Epithelial | 1 | 1 | 0 | 1 | 1 | 2 |
| Prostate | Epithelial | 0 | 0 | 3 | 0 | 3 | 0 |
| Primary
unknown | Epithelial | 0 | 1 | 0 | 0 | 0 | 1 |
 | Table IIIComparison of the establishment rate
of xenograft lines. |
Table III
Comparison of the establishment rate
of xenograft lines.
| Established | Failed | Total | % | P-value |
|---|
| Original tumor
sites | | | | | 0.29a |
|
Gastrointestinal | 29 | 21 | 50 | 58 | |
| Other
digestive | 10 | 10 | 20 | 50 | |
| Respiratory | 10 | 5 | 15 | 67 | |
| Breast and
female | 3 | 6 | 9 | 33 | |
| genital | | | | | |
| Urological | 4 | 3 | 7 | 57 | |
| Neurological | 1 | 6 | 7 | 14 | |
| Others | 4 | 4 | 8 | 50 | |
| Total | 61 | 55 | 116 | 53 | |
| Tumor type | | | | | <0.001b |
| Carcinomas | 58 | 40 | 98 | 59 | |
| Sarcomas | 3 | 15 | 18 | 17 | |
| Tumor site | | | | | <0.001b |
| Primary | 10 | 27 | 37 | 27 | |
| Metastasis | 51 | 28 | 79 | 65 | |
| Time to
engraftmentc | | | | | 0.49b |
| Early | 47 | 46 | 93 | 51 | |
| Delayed | 14 | 9 | 23 | 61 | |
PDX lines were considered established when they were
passaged three times or more and histopathological examination
confirmed their human origin and their morphological similarity to
the corresponding engrafted tumor. Of the 116 tumors engrafted, 61
were established as PDX lines, a success rate of 53%. On comparing
the established cases with the failed cases, no significant
differences were observed in age or gender. The average age of
patients in established cases was 64 years, compared with 63 years
in the failed cases and there was no statistically significant
difference (P=0.53, t-test). In the established cases, 38 cases
were from male patients (60%) and 23 were from female patients
(43%) cases and there was also no statistically significant
difference (P=0.09, Fisher’s probability exact test). High
establishment rates of PDX lines were observed in tumors of the
respiratory system (67%), gastrointestinal tumors (58%), and
urological tumors (57%). None of the primary prostatic tumors or
brain tumors yielded PDX lines in multiple trials.
The establishment rate among the primary organ sites
of engrafts was different; however, there was no statistical
significance (P=0.29, Chi-square test). Fifty-eight PDX lines of
carcinomas (59%) and three of sarcomas (17%) were established, and
the establishment rate was significantly higher for carcinomas
(P<0.001, Fisher’s probability exact test). Metastatic tumors
yielded PDX lines more effectively than tumors from primary sites
(65% and 27%, respectively; P<0.001, Fisher’s probability exact
test). Tumors engrafted into NOG mice two or more days after
surgical removal showed a higher establishment rate (61%) than
those engrafted on the day of surgery or the next day (51%), but
there was no statistically significant difference (P=0.49, Fisher’s
probability exact test).
Preservation of the original tumor
characteristics in the PDX of NOG mice
The morphological characteristics of the
transplanted tumors, as examined by H&E-staining, were well
maintained in the corresponding xenograft tumors both cellularly
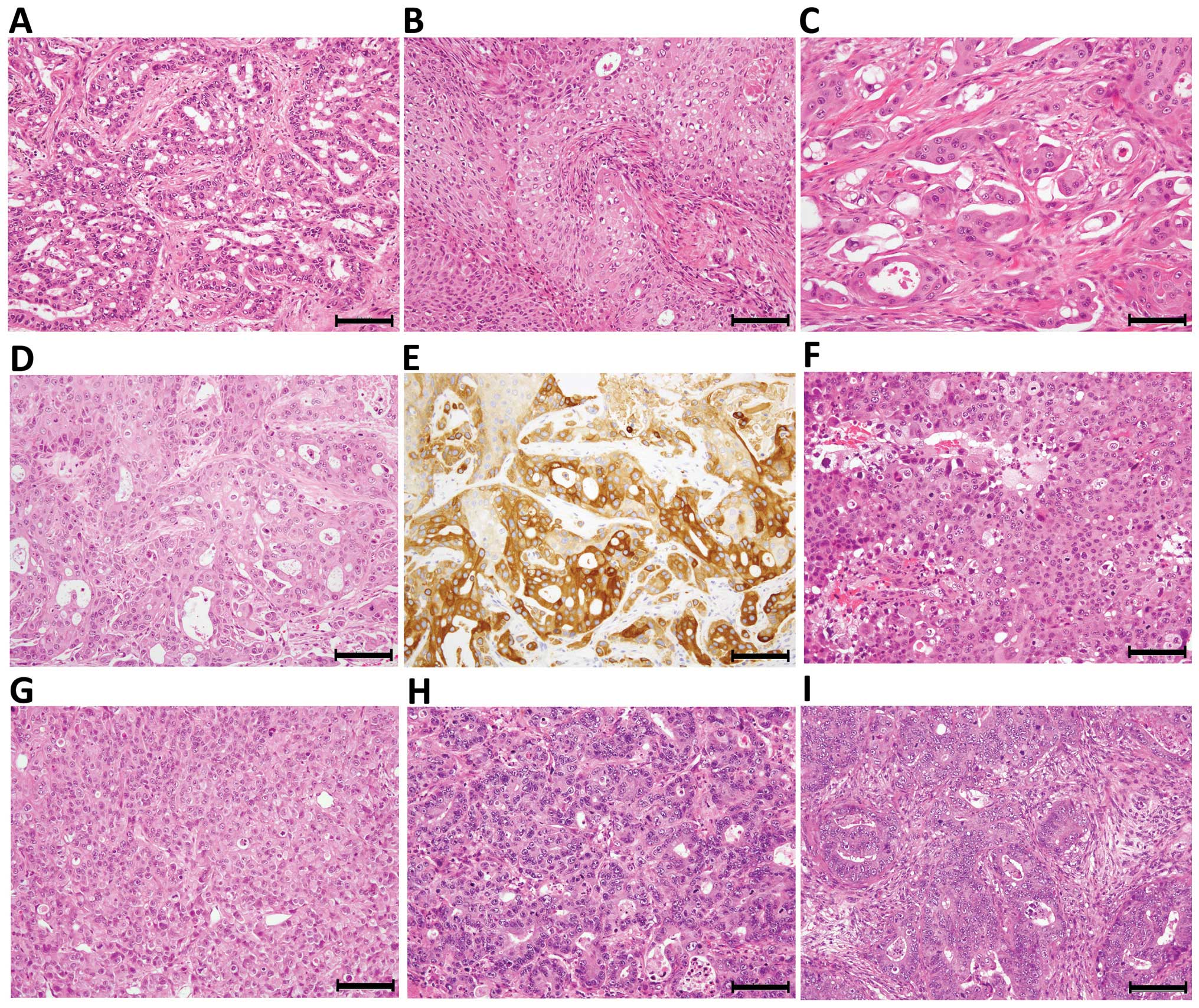
and structurally. One representative case of the PDX line derived
from a brain metastasis of an adenosquamous carcinoma of the lung
is presented in Fig. 1. The
primary tumor of the lung consisted of an adenocarcinoma component
and a less abundant squamous carcinoma component, and the
transitional pattern between them was rare (Fig. 1A and B). In contrast, the brain
metastasis tumor that was engrafted in NOG mice showed a
histological structure of an admixture of adenocarcinoma and
squamous carcinoma components, and the tumor characteristics were
well preserved in the xenograft tumors through the 1st to the 5th
generations (Fig. 1C and D). All
of the components of the PDX tumor, except for the interstitium,
were confirmed as having human origin by IHC for HLA class I
(Fig. 1E). Similarly, a brain
metastasis of a poorly differentiated adenocarcinoma of the lung
was established as a PDX tumor of a poorly differentiated
adenocarcinoma (Fig. 1F and G),
whereas a liver metastasis of a moderately differentiated
adenocarcinoma of the colon with a cribriform pattern was
established as a PDX tumor with a similar histology, indicating
that differentiation capacity was generally preserved (Fig. 1H and I).
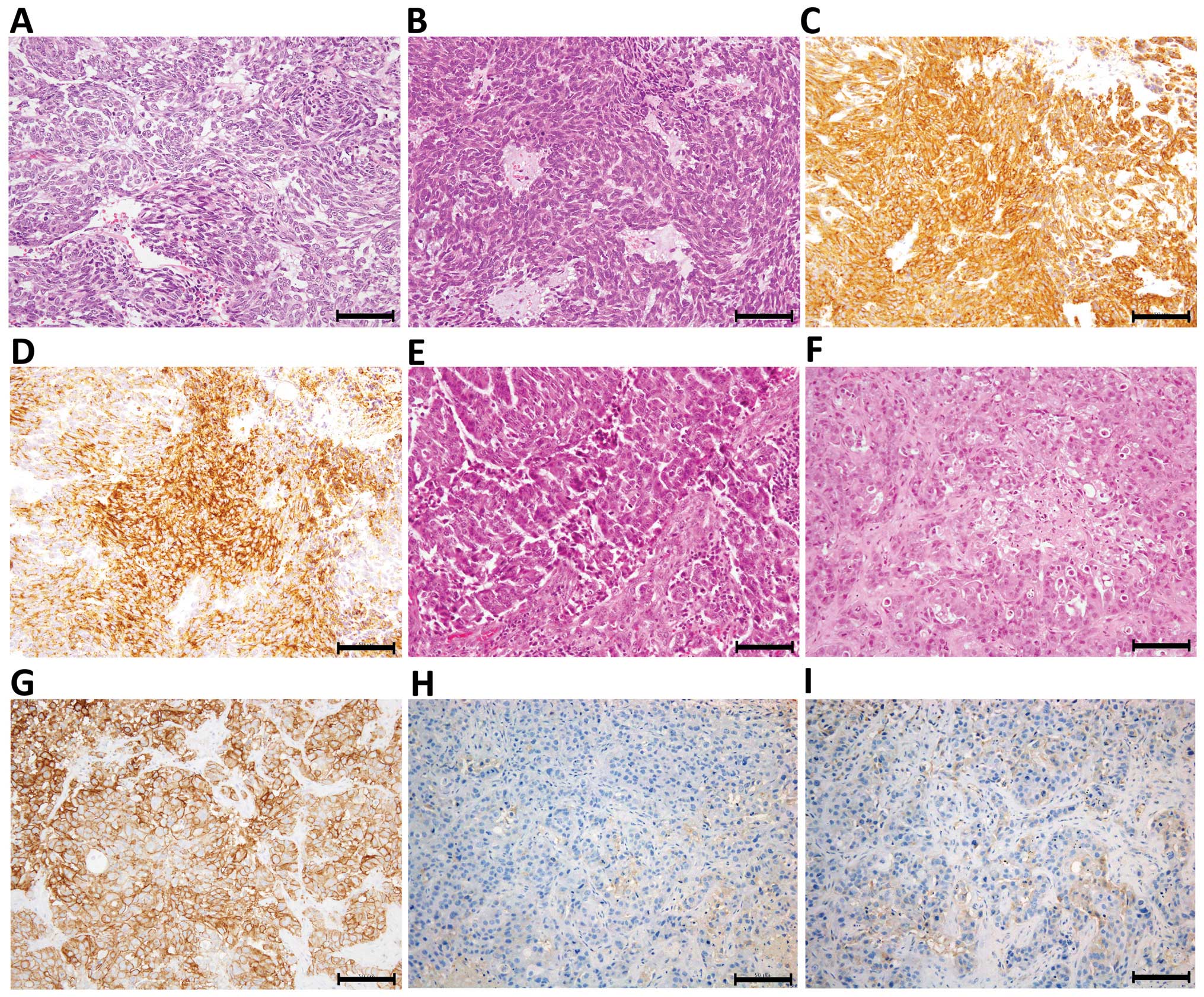
Protein expression, as examined by IHC, was also
well maintained in PDX tumors. Only one PDX line was successfully
established from 10 trials of GIST engraftment (Table I). The established line was derived
from a recurrent metastasis after imatinib methylate treatment. The
3rd generation xenograft was examined and revealed to be strongly
positive for c-kit (a proto-oncogene) and CD34 (Fig. 2A–D). A PDX line established from a
brain metastasis of a HER2-positive breast cancer showed strong
membranous positivity (3+) for HER2, and weak dispersed
positivity for ER and PgR, and therefore shared the same
characteristics as the engrafted tumor (Fig. 2E–I).
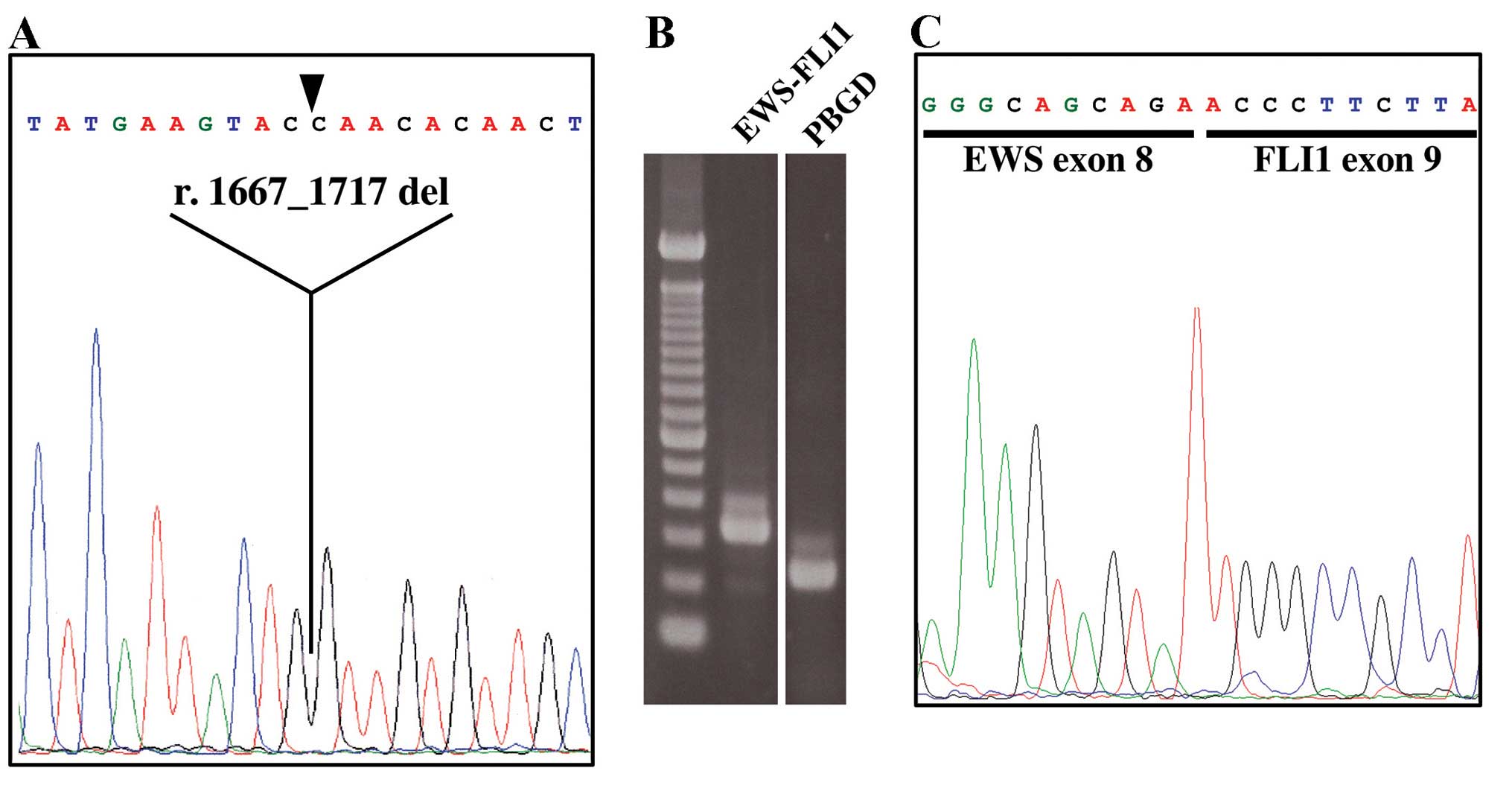
As expected, genetic alterations were conserved in
the PDX tumors of NOG mice. The engrafted GIST tumor contained a
51-nucleotide deletion in exon 11 of KIT (r.1667_1717 del/p.
Q556_D572 del) and DNA extracted from the 3rd generation xenograft
was found to contain an identical mutation (Fig. 3A). Furthermore, the Ewing/PNET
sarcoma xenograft tumor established from a brain metastasis
contained the EWS-FLI1 fusion mRNA just as the original engraft
(Fig. 3B and C).
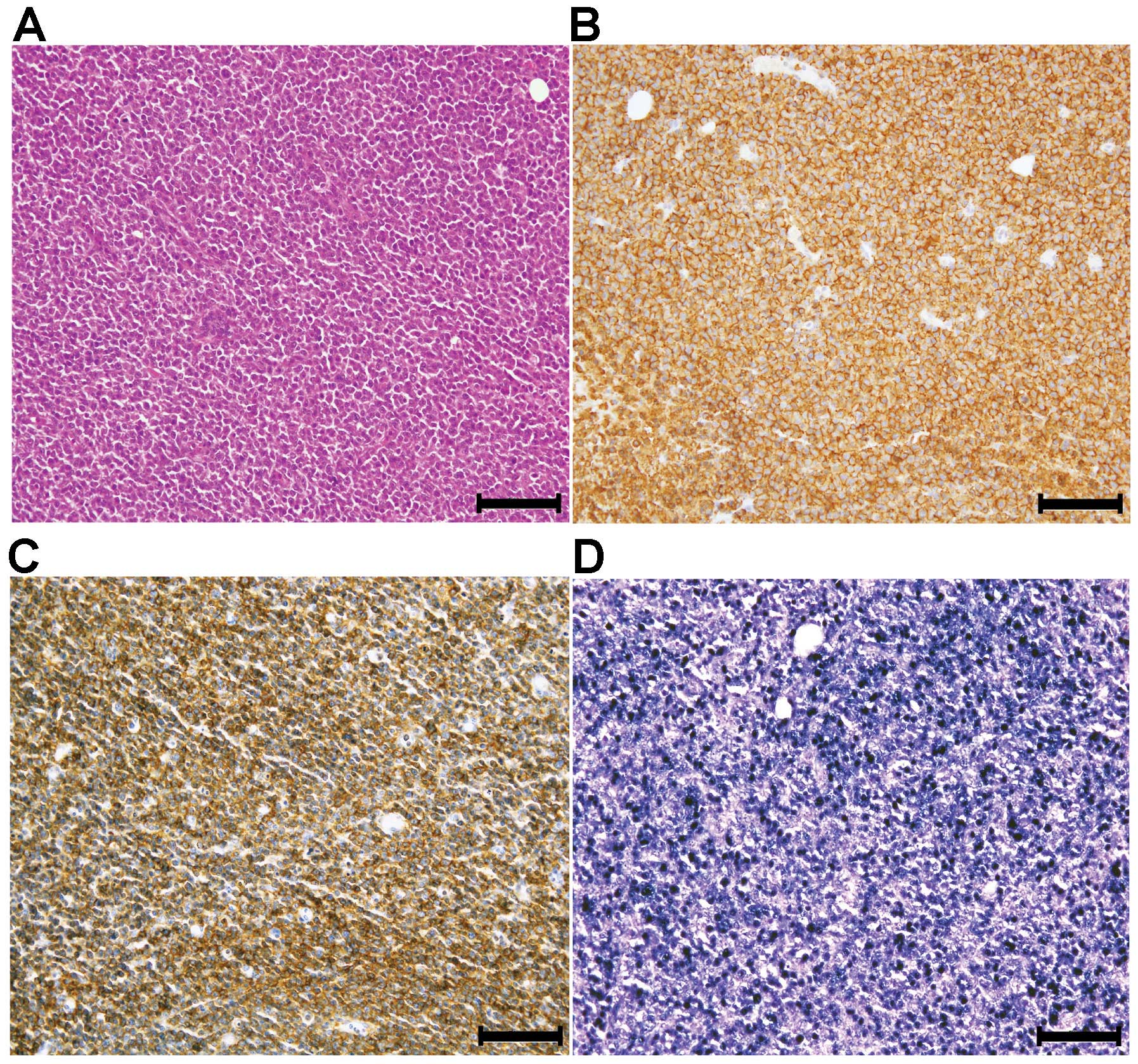
Lymphoproliferative lesion (LPL) in NOG
mice
In eight cases (7% of all engraftments),
transplantable xenograft tumors composed of large monotonous
nonepithelial cells were observed whose morphology differed from
that of the original tumor (Fig.
4A). This phenomenon was observed only in epithelial tumor
engraftments (Tables I and
II). The monotonous cells were
HLA class I positive, demonstrating their human origin, and were
also positive for leukocyte common antigen (CD111) by IHC and EBER
by ISH, indicating the possibility that they were EBV
infection-associated LPLs (Fig.
4B–D).
Discussion
In this study, we aimed to establish a PDX line in
NOG mice that preserved the original characteristics of the
engrafted tumor. In a previous PDX trial in NOG mice, which
included more than 300 surgically removed tumors, the establishment
rate of the xenograft line was 16% (41 of 259 engrafts) for primary
tumors, 31% (5 of 16 engrafts) for distant metastasis sites, and
16% (8 of 51 engrafts) for lymph node metastases (5). In this study, we achieved higher
establishment rates both for primary tumors (27%) and metastatic
tumors (65%). However, the constitution of the tumors used for PDX
and those in the primary organ site, the ratios of primary tumors
to metastatic tumors, and the numbers of each case all differed
between our study and the previous study, making comparisons
difficult. For example, the previous study in NOG mice used 57
primary breast cancers and obtained only three PDX lines (5%),
whereas primary tumors of the breast were not included in our study
(5). In fact, the establishment
rate may depend on the organ site from which the engraft is
taken.
Colorectal tumors showed relatively higher
establishment rates than tumors from other sites in nude or SCID
mice (10,11); this was also the case in the
previous NOG study (17/48 engraftments, 35%) (5). In the present study, establishment
rates were found to differ between sites (e.g., 58% for
gastrointestinal tumors compared with 14% for urological tumors)
but this difference was not statistically significant. The
establishment rates of metastatic tumors were significantly higher
than those of primary tumors, a finding that was consistent with
previous studies (5,17,18).
In our study, carcinomas showed a significantly higher
establishment rate (59%) than nonepithelial tumors (17%); however,
a limited number of nonepithelial tumors were tested and a larger
sample size would be needed to confirm this difference.
In the present study, only subcutaneous
transplantations were performed. The transplantation site has been
reported to have an influence on xenograft growth (5,19–21).
Considering what is known about tumor cells and microenvironmental
biology, heterotopic subcutaneous tumor models seem to have some
shortcomings compared with orthotopic transplantation, especially
in the establishment rate and preservation of the original tumor
characteristics (21). However,
our subcutaneous models revealed well-preserved characteristics of
the original engrafts in morphology, protein expression and gene
alterations. Although further studies are needed to clarify whether
this was because of the highly immunodeficient nature of NOG mice,
the establishment of PDX using manageable subcutaneous
transplantations is convenient when compared with skillful
orthotopic transplantation.
Unexpectedly, in the present study, we experienced
no significant difference in the establishment rate of PDX lines
between the tumors engrafted early (on the day of surgery or the
next day) and the tumors engrafted after 2 days. To the best of our
knowledge, no previous studies have investigated this issue. One
could speculate that the so-called cancer stem cells responsible
for tumorigenicity in mice might be resistance to the severe stress
induced by removal from the patients. Although further
investigation is needed, this information might help oncology
researchers to improve and simplify PDX line establishment,
particularly in light of our findings that subcutaneous
transplantation is not inferior to orthotopic transplantation with
regard to preserving the original engraft characteristics.
The high occurrence of LPL was the most problematic
aspect of the establishment of NOG mice-PDX, which arose because of
the severely immunodeficient nature of the animal model. In eight
cases (7% of all engraftments), the engrafted tumors were replaced
by LPL until the 3rd generation of xenografts. The LPLs were
demonstrated to be EBV infection-associated, as has been previously
reported (20,22,23).
Fujii and colleagues also reported that EBV-infected B cells
originating from the donor were distributed systemically within the
NOG mouse (23). LPLs were
transplantable, and difficult to distinguish from the proper
xenografts in terms of gross appearance. It is therefore important
that histology of xenografts is checked before transplantation into
new mice. However, the frequency of LPLs is acceptable when
considering the merits of NOG mice. Replacement of the engrafted
tumors by LPL accounted for 15% of the failed cases in PDX line
establishment, indicating that the major cause of failed cases is
therefore likely to be the nature of the xenografts.
Owing to progression in the field of oncology, the
demand for relevant human tumor models is increasing. In
vivo models play a vital role in the extrapolation of data to
human patients, especially in the development of anticancer agents.
Evidence that both tumor differentiation and tumor structure were
highly conserved between the original surgical specimen and the PDX
tumor confirms the suitability of our mouse model for the study of
tumor biology. Applications of this model, not only for more common
tumors, but also for uncommon tumors, such as sarcomas or pediatric
tumors, will provide researchers with reliable comparative
preclinical data that may contribute to the development of novel
cancer therapies. The rapid and efficient establishment of PDX
linking with clinical information may lead in the future to the
development of personalized anticancer therapies by simulating
various treatments in individual PDX mice, so-called cancer
xenopatients.
Acknowledgements
The authors thank Ms. Aki Namiki, Miyuki Kon, Takayo
Shimamura, and Manami Ino at the Kanagawa Cancer Center for their
assistance in tissue preparation and obtaining informed consent
from patients involved in the study. This work was partly supported
by JSPS Grant-in-Aid for Scientific Research (C), numbers 25430098
and 26430097.
References
|
1
|
Morton CL and Houghton PJ: Establishment
of human tumor xenografts in immunodeficient mice. Nat Protoc.
2:247–250. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jin K, Li G, Cui B, Zhang J, Lan H, Han N,
Xie B, Cao F, He K, Wang H, et al: Assessment of a novel VEGF
targeted agent using patient-derived tumor tissue xenograft models
of colon carcinoma with lymphatic and hepatic metastases. PLoS One.
6:e283842011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Johnson JI, Decker S, Zaharevitz D,
Rubinstein LV, Venditti JM, Schepartz S, Kalyandrug S, Christian M,
Arbuck S, Hollingshead M, et al: Relationships between drug
activity in NCI preclinical in vitro and in vivo models and early
clinical trials. Br J Cancer. 84:1424–1431. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sausville EA, Burger AM, Becher OJ and
Holland EC: Contributions of human tumor xenografts to anticancer
drug development. Cancer Res. 66:3351–3354. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Fujii E, Suzuki M, Matsubara K, Watanabe
M, Chen YJ, Adachi K, Ohnishi Y, Tanigawa M, Tsuchiya M and Tamaoki
N: Establishment and characterization of in vivo human tumor models
in the NOD/SCID/gamma(c)(null) mouse. Pathol Int. 58:559–567. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bertotti A, Migliardi G, Galimi F, Sassi
F, Torti D, Isella C, Corà D, Di Nicolantonio F, Buscarino M, Petti
C, et al: A molecularly annotated platform of patient-derived
xenografts (‘xenopatients’) identifies HER2 as an effective
therapeutic target in cetuximab-resistant colorectal cancer. Cancer
Discov. 1:508–523. 2011. View Article : Google Scholar
|
|
7
|
Galimi F, Torti D, Sassi F, Isella C, Corà
D, Gastaldi S, Ribero D, Muratore A, Massucco P, Siatis D, et al:
Genetic and expression analysis of MET, MACC1, and HGF in
metastatic colorectal cancer: Response to met inhibition in patient
xenografts and pathologic correlations. Clin Cancer Res.
17:3146–3156. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kobayashi S, Yamada-Okabe H, Suzuki M,
Natori O, Kato A, Matsubara K, Jau Chen Y, Yamazaki M, Funahashi S,
Yoshida K, et al: LGR5-positive colon cancer stem cells
inter-convert with drug-resistant LGR5-negative cells and are
capable of tumor reconstitution. Stem Cells. 30:2631–2644. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Scott CL, Becker MA, Haluska P and Samimi
G: Patient-derived xenograft models to improve targeted therapy in
epithelial ovarian cancer treatment. Front Oncol. 3:2952013.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tokunaga T, Nakamura M, Oshika Y, Ohnishi
Y and Ueyama Y: Is xenotransplantability of human colon cancers in
SCID mice affected by angiogenic factors? J Natl Cancer Inst.
90:400–401. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Dangles-Marie V, Pocard M, Richon S,
Weiswald LB, Assayag F, Saulnier P, Judde JG, Janneau JL, Auger N,
Validire P, et al: Establishment of human colon cancer cell lines
from fresh tumors versus xenografts: Comparison of success rate and
cell line features. Cancer Res. 67:398–407. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ito M, Hiramatsu H, Kobayashi K, Suzue K,
Kawahata M, Hioki K, Ueyama Y, Koyanagi Y, Sugamura K, Tsuji K, et
al: NOD/SCID/gamma(c)(null) mouse: An excellent recipient mouse
model for engraftment of human cells. Blood. 100:3175–3182. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Suemizu H, Monnai M, Ohnishi Y, Ito M,
Tamaoki N and Nakamura M: Identification of a key molecular
regulator of liver metastasis in human pancreatic carcinoma using a
novel quantitative model of metastasis in NOD/SCID/gammacnull (NOG)
mice. Int J Oncol. 31:741–751. 2007.PubMed/NCBI
|
|
14
|
Chijiwa T, Abe Y, Ikoma N, Yamazaki H,
Tsukamoto H, Suemizu H, Kawai K, Wakui M, Nishime C, Matsumoto H,
et al: Thrombospondin 2 inhibits metastasis of human malignant
melanoma through microenvironment-modification in
NOD/SCID/gammaCnull (NOG) mice. Int J Oncol. 34:5–13. 2009.
|
|
15
|
Kubo A, Ohmura M, Wakui M, Harada T,
Kajihara S, Ogawa K, Suemizu H, Nakamura M, Setou M and Suematsu M:
Semi-quantitative analyses of metabolic systems of human colon
cancer metastatic xenografts in livers of superimmunodeficient NOG
mice. Anal Bioanal Chem. 400:1895–1904. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cho H, Watanabe T, Aoyama T, Hayashi T,
Yamada T, Ogata T, Yoshikawa T, Tsuburaya A, Sekiguchi H, Nakamura
Y, et al: Small bud of probable gastrointestinal stromal tumor
within a laparoscopically-resected gastric schwannoma. Int J Clin
Oncol. 17:294–298. 2012. View Article : Google Scholar
|
|
17
|
Yoshida T, Kinoshita H, Segawa T, Nakamura
E, Inoue T, Shimizu Y, Kamoto T and Ogawa O: Antiandrogen
bicalutamide promotes tumor growth in a novel androgen-dependent
prostate cancer xenograft model derived from a bicalutamide-treated
patient. Cancer Res. 65:9611–9616. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Marangoni E, Vincent-Salomon A, Auger N,
Degeorges A, Assayag F, de Cremoux P, de Plater L, Guyader C, De
Pinieux G, Judde JG, et al: A new model of patient tumor-derived
breast cancer xenografts for preclinical assays. Clin Cancer Res.
13:3989–3998. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Mueller BM and Reisfeld RA: Potential of
the scid mouse as a host for human tumors. Cancer Metastasis Rev.
10:193–200. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bankert RB, Hess SD and Egilmez NK: SCID
mouse models to study human cancer pathogenesis and approaches to
therapy: Potential, limitations, and future directions. Front
Biosci. 7:c44–c62. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
21
|
Bibby MC: Orthotopic models of cancer for
preclinical drug evaluation: Advantages and disadvantages. Eur J
Cancer. 40:852–857. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Itoh T, Shiota M, Takanashi M, Hojo I,
Satoh H, Matsuzawa A, Moriyama T, Watanabe T, Hirai K and Mori S:
Engraftment of human non-Hodgkin lymphomas in mice with severe
combined immunodeficiency. Cancer. 72:2686–2694. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Fujii E, Kato A, Chen YJ, Matsubara K,
Ohnishi Y and Suzuki M: Characterization of EBV-related
lymphoproliferative lesions arising in donor lymphocytes of
transplanted human tumor tissues in the NOG mouse. Exp Anim.
63:289–296. 2014. View Article : Google Scholar : PubMed/NCBI
|