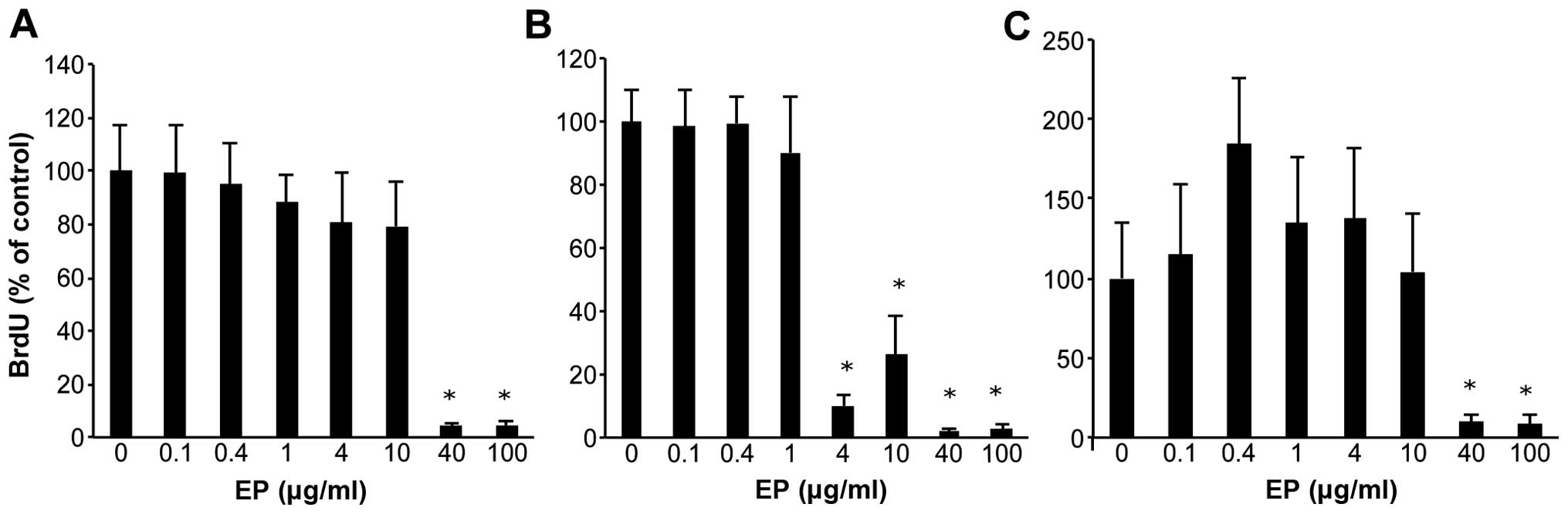
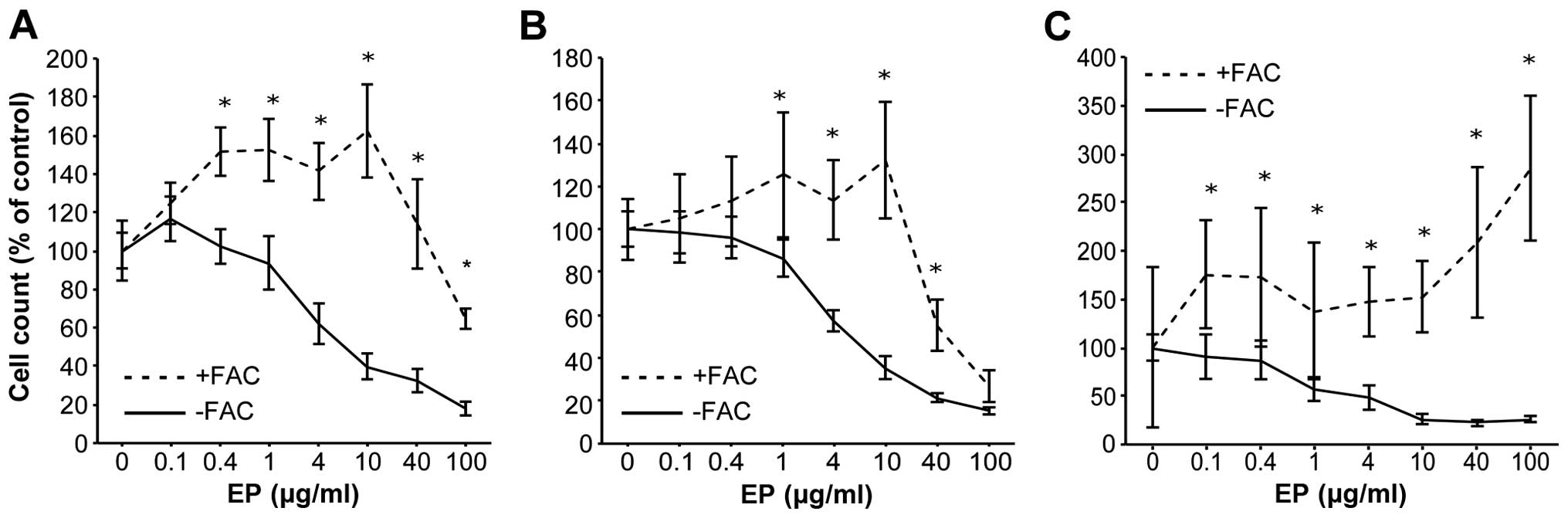
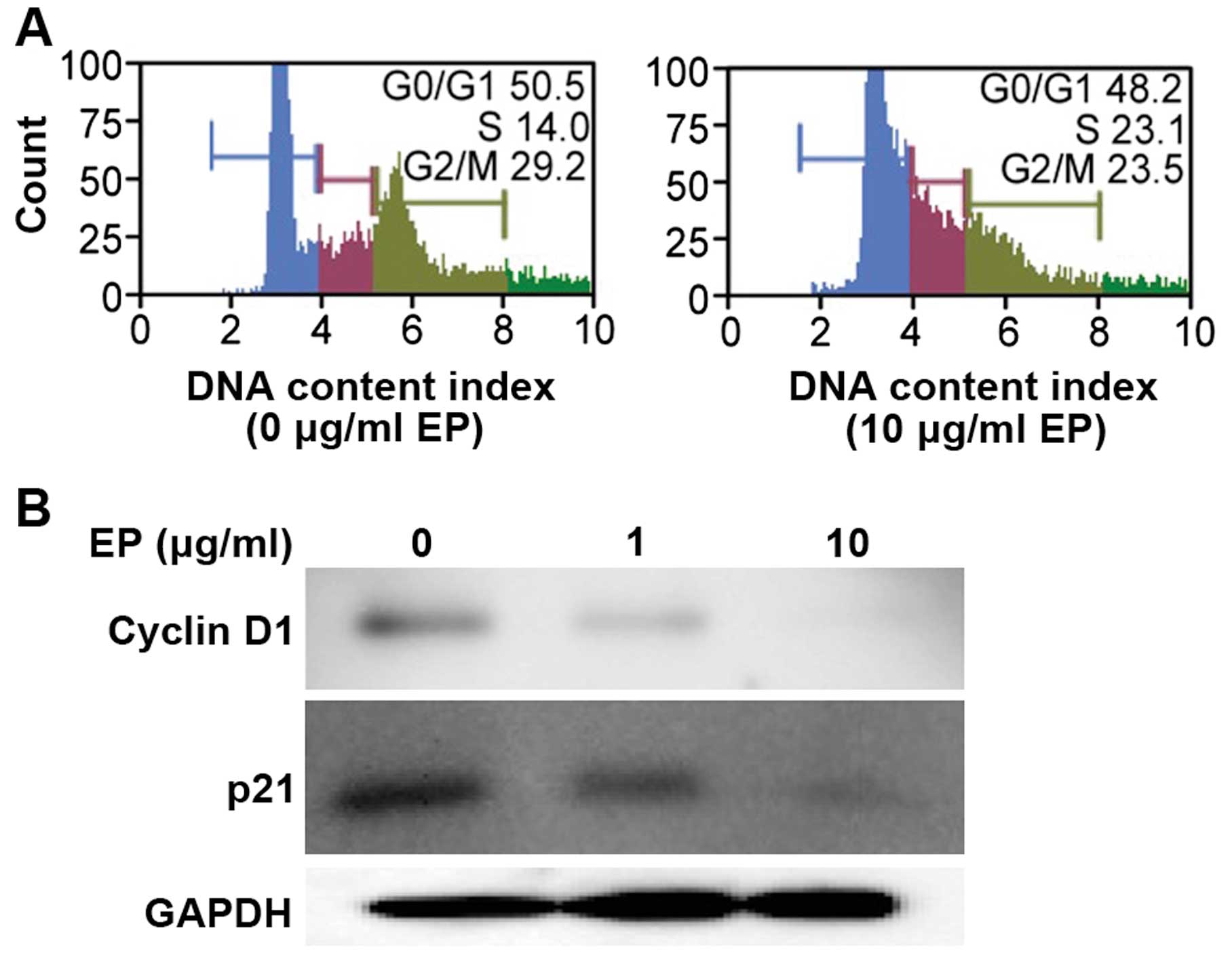
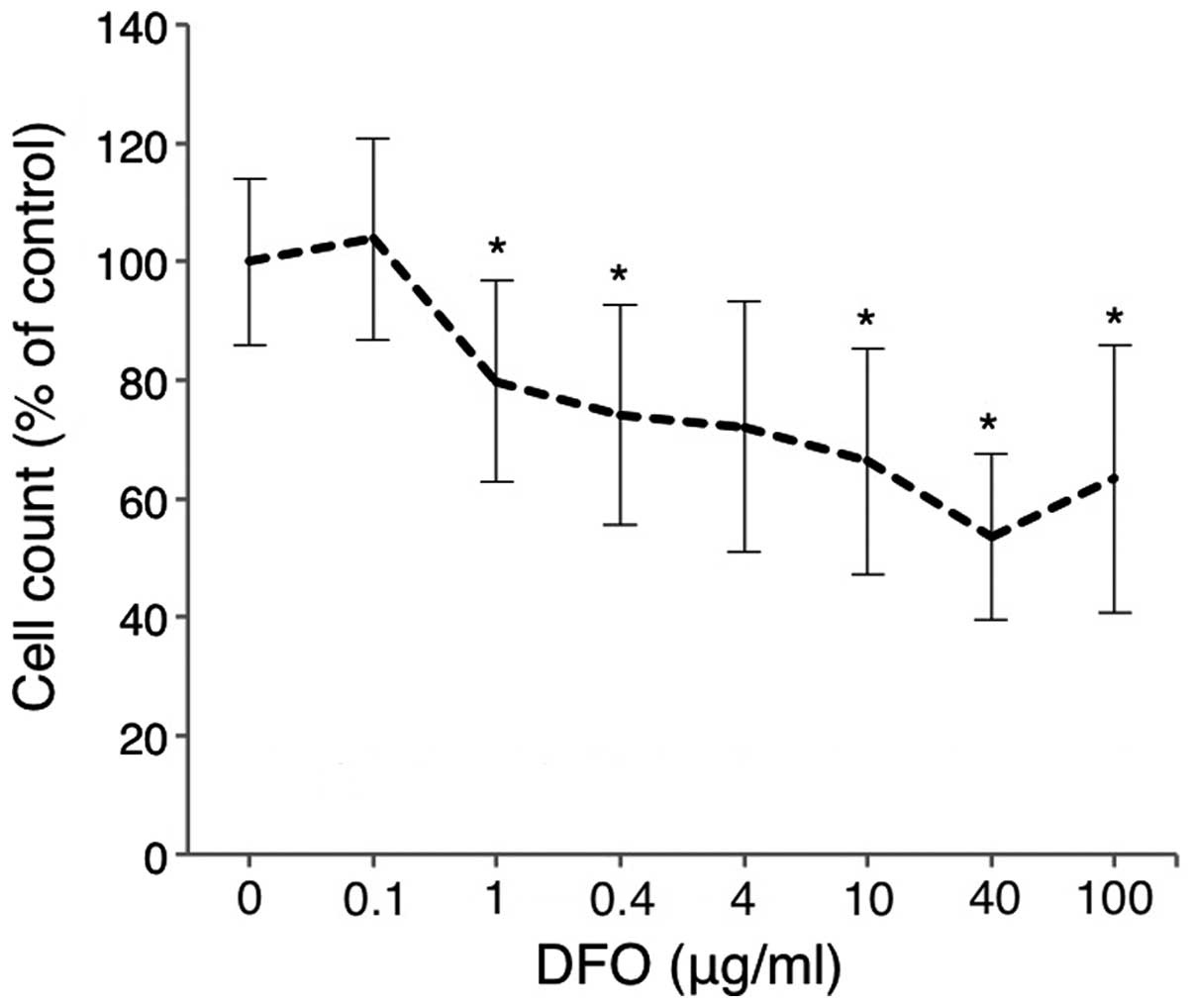
|
1
|
El-Serag HB: Hepatocellular carcinoma. N
Engl J Med. 365:1118–1127. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Montalto G, Cervello M, Giannitrapani L,
Dantona F, Terranova A and Castagnetta LA: Epidemiology, risk
factors, and natural history of hepatocellular carcinoma. Ann N Y
Acad Sci. 963:13–20. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Abou-Alfa GK, Schwartz L, Ricci S, Amadori
D, Santoro A, Figer A, De Greve J, Douillard JY, Lathia C, Schwartz
B, et al: Phase II study of sorafenib in patients with advanced
hepatocellular carcinoma. J Clin Oncol. 24:4293–4300. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Llovet JM, Ricci S, Mazzaferro V, Hilgard
P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A,
et al; SHARP Investigators Study Group. Sorafenib in advanced
hepatocellular carcinoma. N Engl J Med. 359:378–390. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Villanueva A and Llovet JM: Targeted
therapies for hepatocellular carcinoma. Gastroenterology.
140:1410–1426. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wolber E-M and Jelkmann W: Thrombopoietin:
The novel hepatic hormone. News Physiol Sci. 17:6–10.
2002.PubMed/NCBI
|
|
7
|
de Sauvage FJ, Carver-Moore K, Luoh SM,
Ryan A, Dowd M, Eaton DL and Moore MW: Physiological regulation of
early and late stages of megakaryocytopoiesis by thrombopoietin. J
Exp Med. 183:651–656. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kaushansky K and Drachman JG: The
molecular and cellular biology of thrombopoietin: The primary
regulator of platelet production. Oncogene. 21:3359–3367. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Deutsch VR and Tomer A: Megakaryocyte
development and platelet production. Br J Haematol. 134:453–466.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kaushansky K, Broudy VC, Lin N, Jorgensen
MJ, McCarty J, Fox N, Zucker-Franklin D and Lofton-Day C:
Thrombopoietin, the Mp1 ligand, is essential for full megakaryocyte
development. Proc Natl Acad Sci USA. 92:3234–3238. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bussel JB, Cheng G, Saleh MN, Psaila B,
Kovaleva L, Meddeb B, Kloczko J, Hassani H, Mayer B, Stone NL, et
al: Eltrombopag for the treatment of chronic idiopathic
thrombocytopenic purpura. N Engl J Med. 357:2237–2247. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
McHutchison JG, Dusheiko G, Shiffman ML,
Rodriguez-Torres M, Sigal S, Bourliere M, Berg T, Gordon SC,
Campbell FM, Theodore D, et al; TPL102357 Study Group. Eltrombopag
for thrombocytopenia in patients with cirrhosis associated with
hepatitis C. N Engl J Med. 357:2227–2236. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Will B, Kawahara M, Luciano JP, Bruns I,
Parekh S, Erickson-Miller CL, Aivado MA, Verma A and Steidl U:
Effect of the nonpeptide thrombopoietin receptor agonist
Eltrombopag on bone marrow cells from patients with acute myeloid
leukemia and myelodysplastic syndrome. Blood. 114:3899–3908. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Erickson-Miller CL, Kirchner J, Aivado M,
May R, Payne P and Chadderton A: Reduced proliferation of
non-megakaryocytic acute myelogenous leukemia and other leukemia
and lymphoma cell lines in response to eltrombopag. Leuk Res.
34:1224–1231. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Roth M, Will B, Simkin G, Narayanagari S,
Barreyro L, Bartholdy B, Tamari R, Mitsiades CS, Verma A and Steidl
U: Eltrombopag inhibits the proliferation of leukemia cells via
reduction of intracellular iron and induction of differentiation.
Blood. 120:386–394. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Erickson-Miller CL, Pillarisetti K,
Kirchner J, Figueroa DJ, Ottesen L, Martin AM, Liu Y, Kamel YM and
Messam C: Low or undetectable TPO receptor expression in malignant
tissue and cell lines derived from breast, lung, and ovarian
tumors. BMC Cancer. 12:4052012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kawaguchi T, Komori A, Seike M, Fujiyama
S, Watanabe H, Tanaka M, Sakisaka S, Nakamuta M, Sasaki Y, Oketani
M, et al: Efficacy and safety of eltrombopag in Japanese patients
with chronic liver disease and thrombocytopenia: A randomized,
open-label, phase II study. J Gastroenterol. 47:1342–1351. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Matthys G, Park JW, McGuire S, Wire MB,
Bowen C, Williams D, Jenkins J and Peng B: Clinical
pharmacokinetics, platelet response, and safety of eltrombopag at
supratherapeutic doses of up to 200 mg once daily in healthy
volunteers. J Clin Pharmacol. 51:301–308. 2011. View Article : Google Scholar
|
|
19
|
Chou T-C: Drug combination studies and
their synergy quantification using the Chou-Talalay method. Cancer
Res. 70:440–446. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Nurtjahja-Tjendraputra E, Fu D, Phang JM
and Richardson DR: Iron chelation regulates cyclin D1 expression
via the proteasome: A link to iron deficiency-mediated growth
suppression. Blood. 109:4045–4054. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Cardier JE and Dempsey J: Thrombopoietin
and its receptor, c-mpl, are constitutively expressed by mouse
liver endothelial cells: Evidence of thrombopoietin as a growth
factor for liver endothelial cells. Blood. 91:923–929.
1998.PubMed/NCBI
|
|
22
|
Schmelzer E, Deiwick A, Bruns H, Fiegel HC
and Bader A: Thrombopoietin is a growth factor for rat hepatic
progenitors. Eur J Gastroenterol Hepatol. 20:209–216. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Nozaki R, Murata S, Nowatari T, Maruyama
T, Ikeda N, Kawasaki T, Fukunaga K and Ohkohchi N: Effects of
thrombopoietin on growth of hepatocellular carcinoma: Is
thrombopoietin therapy for liver disease safe or not? Hepatol Res.
43:610–620. 2013. View Article : Google Scholar
|
|
24
|
Erickson-Miller CL, DeLorme E, Tian SS,
Hopson CB, Stark K, Giampa L, Valoret EI, Duffy KJ, Luengo JL,
Rosen J, et al: Discovery and characterization of a selective,
nonpeptidyl thrombopoietin receptor agonist. Exp Hematol. 33:85–93.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Erickson-Miller C, Delorme E, Giampa L,
Hopson C, Valoret E, Tian SS, Miller SG, Keenan R, Rosen J, Dillon
S, et al: Biological activity and selectivity for Tpo receptor of
the orally bioavailable, small molecule Tpo receptor agonist,
SB-497115. Blood (ASH Annual Meeting Abstracts). 104:29122004.
|
|
26
|
Kalota A, Brennan K, Erickson-Miller CL,
Danet G, Carroll M and Gewirtz AM: Effects of SB559457, a novel
small molecule thrombopoietin receptor (TpoR) agonist, on human
hematopoietic cell growth and differentiation. Blood (ASH Annual
Meeting Abstracts). 104:29132004.
|
|
27
|
Safonov IG, Heerding DA, Keenan RM, Price
AT, Erickson-Miller CL, Hopson CB, Levin JL, Lord KA and Tapley PM:
New benzimidazoles as thrombopoietin receptor agonists. Bioorg Med
Chem Lett. 16:1212–1216. 2006. View Article : Google Scholar
|
|
28
|
Luengo JI, Duffy KJ, Shaw AN, Delorme E,
Wiggall KJ, Giampa L, Liu N, Smith H, Tian SS, Miller SG, et al:
Discovery of SB-497115, a small-molecule thrombopoietin (TPO)
receptor agonist for the treatment of thrombocytopenia. Blood (ASH
Annual Meeting Abstracts). 104:29102004.
|
|
29
|
Peeters K, Stassen J-M, Collen D, Van Geet
C and Freson K: Emerging treatments for thrombocytopenia:
Increasing platelet production. Drug Discov Today. 13:798–806.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Duffy KJ, Shaw AN, Delorme E, Dillon SB,
Erickson-Miller C, Giampa L, Huang Y, Keenan RM, Lamb P, Liu N, et
al: Identification of a pharmacophore for thrombopoietic activity
of small, non-peptidyl molecules. 1. Discovery and optimization of
salicylaldehyde thiosemicarbazone thrombopoietin mimics. J Med
Chem. 45:3573–3575. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Duffy KJ, Price AT, Delorme E, Dillon SB,
Duquenne C, Erickson-Miller C, Giampa L, Huang Y, Keenan RM, Lamb
P, et al: Identification of a pharmacophore for thrombopoietic
activity of small, non-peptidyl molecules. 2. Rational design of
naphtho[1,2-d]imidazole thrombopoietin mimics. J Med Chem.
45:3576–3578. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Williams DD, Peng B, Bailey CK, Wire MB,
Deng Y, Park JW, Collins DA, Kapsi SG and Jenkins JM: Effects of
food and antacids on the pharmacokinetics of eltrombopag in healthy
adult subjects: Two single-dose, open-label, randomized-sequence,
crossover studies. Clin Ther. 31:764–776. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hershko C: Control of disease by selective
iron depletion: A novel therapeutic strategy utilizing iron
chelators. Baillieres Clin Haematol. 7:965–1000. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Buss JL, Greene BT, Turner J, Torti FM and
Torti SV: Iron chelators in cancer chemotherapy. Curr Top Med Chem.
4:1623–1635. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Andrews NC: Disorders of iron metabolism.
N Engl J Med. 341:1986–1995. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Lucas JJ, Szepesi A, Domenico J, Takase K,
Tordai A, Terada N and Gelfand EW: Effects of iron-depletion on
cell cycle progression in normal human T lymphocytes: Selective
inhibition of the appearance of the cyclin A-associated component
of the p33cdk2 kinase. Blood. 86:2268–2280. 1995.PubMed/NCBI
|
|
37
|
Brodie C, Siriwardana G, Lucas J,
Schleicher R, Terada N, Szepesi A, Gelfand E and Seligman P:
Neuroblastoma sensitivity to growth inhibition by deferrioxamine:
Evidence for a block in G1 phase of the cell cycle. Cancer Res.
53:3968–3975. 1993.PubMed/NCBI
|
|
38
|
Kwok JC and Richardson DR: The iron
metabolism of neoplastic cells: Alterations that facilitate
proliferation? Crit Rev Oncol Hematol. 42:65–78. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Le NTV and Richardson DR: The role of iron
in cell cycle progression and the proliferation of neoplastic
cells. Biochim Biophys Acta. 1603:31–46. 2002.PubMed/NCBI
|
|
40
|
Kalinowski DS and Richardson DR: The
evolution of iron chelators for the treatment of iron overload
disease and cancer. Pharmacol Rev. 57:547–583. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Larrick JW and Cresswell P: Modulation of
cell surface iron transferrin receptors by cellular density and
state of activation. J Supramol Struct. 11:579–586. 1979.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Richardson DR and Baker E: The uptake of
iron and transferrin by the human malignant melanoma cell. Biochim
Biophys Acta. 1053:1–12. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Richardson DR and Baker E: The effect of
desferrioxamine and ferric ammonium citrate on the uptake of iron
by the membrane iron-binding component of human melanoma cells.
Biochim Biophys Acta. 1103:275–280. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Elford HL, Freese M, Passamani E and
Morris HP: Ribonucleotide reductase and cell proliferation. I.
Variations of ribonucleotide reductase activity with tumor growth
rate in a series of rat hepatomas. J Biol Chem. 245:5228–5233.
1970.PubMed/NCBI
|
|
45
|
Takeda E and Weber G: Role of
ribonucleotide reductase in expression in the neoplastic program.
Life Sci. 28:1007–1014. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Witt L, Yap T and Blakley RL: Regulation
of ribonucleotide reductase activity and its possible exploitation
in chemotherapy. Adv Enzyme Regul. 17:157–171. 1978. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Donfrancesco A, Deb G, Dominici C, Pileggi
D, Castello MA and Helson L: Effects of a single course of
deferoxamine in neuroblastoma patients. Cancer Res. 50:4929–4930.
1990.PubMed/NCBI
|
|
48
|
Estrov Z, Tawa A, Wang XH, Dubé ID, Sulh
H, Cohen A, Gelfand EW and Freedman MH: In vitro and in vivo
effects of deferoxamine in neonatal acute leukemia. Blood.
69:757–761. 1987.PubMed/NCBI
|
|
49
|
Kaplinsky C, Estrov Z, Freedman MH,
Gelfand EW and Cohen A: Effect of deferoxamine on DNA synthesis,
DNA repair, cell proliferation, and differentiation of HL-60 cells.
Leukemia. 1:437–441. 1987.PubMed/NCBI
|
|
50
|
Blatt J, Taylor SR and Stitely S:
Mechanism of antineuroblastoma activity of deferoxamine in vitro. J
Lab Clin Med. 112:433–436. 1988.PubMed/NCBI
|
|
51
|
Blatt J and Stitely S: Antineuroblastoma
activity of desferoxamine in human cell lines. Cancer Res.
47:1749–1750. 1987.PubMed/NCBI
|
|
52
|
Gao J and Richardson DR: The potential of
iron chelators of the pyridoxal isonicotinoyl hydrazone class as
effective antiproliferative agents, IV: The mechanisms involved in
inhibiting cell-cycle progression. Blood. 98:842–850. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Le NTV and Richardson DR: Iron chelators
with high antiproliferative activity up-regulate the expression of
a growth inhibitory and metastasis suppressor gene: A link between
iron metabolism and proliferation. Blood. 104:2967–2975. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Fu D and Richardson DR: Iron chelation and
regulation of the cell cycle: 2 mechanisms of posttranscriptional
regulation of the universal cyclin-dependent kinase inhibitor
p21CIP1/WAF1 by iron depletion. Blood. 110:752–761. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Keberle H: The Biochemistry of
desferrioxamine and its relation to iron metabolism. Ann NY Acad
Sci. 119:758–768. 1964. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Whitnall M, Howard J, Ponka P and
Richardson DR: A class of iron chelators with a wide spectrum of
potent antitumor activity that overcomes resistance to
chemotherapeutics. Proc Natl Acad Sci USA. 103:14901–14906. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Rakba N, Loyer P, Gilot D, Delcros JG,
Glaise D, Baret P, Pierre JL, Brissot P and Lescoat G:
Antiproliferative and apoptotic effects of O-Trensox, a new
synthetic iron chelator, on differentiated human hepatoma cell
lines. Carcinogenesis. 21:943–951. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Rao VA, Klein SR, Agama KK, Toyoda E,
Adachi N, Pommier Y and Shacter EB: The iron chelator Dp44mT causes
DNA damage and selective inhibition of topoisomerase IIalpha in
breast cancer cells. Cancer Res. 69:948–957. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Gangaidzo IT and Gordeuk VR:
Hepatocellular carcinoma and African iron overload. Gut.
37:727–730. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Ba Q, Hao M, Huang H, Hou J, Ge S, Zhang
Z, Yin J, Chu R, Jiang H, Wang F, et al: Iron deprivation
suppresses hepatocellular carcinoma growth in experimental studies.
Clin Cancer Res. 17:7625–7633. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Yamasaki T, Terai S and Sakaida I:
Deferoxamine for advanced hepatocellular carcinoma. N Engl J Med.
365:576–578. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Tanaka H, Fujita N, Sugimoto R, Urawa N,
Horiike S, Kobayashi Y, Iwasa M, Ma N, Kawanishi S, Watanabe S, et
al: Hepatic oxidative DNA damage is associated with increased risk
for hepatocellular carcinoma in chronic hepatitis C. Br J Cancer.
98:580–586. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Kato J, Kobune M, Nakamura T, Kuroiwa G,
Takada K, Takimoto R, Sato Y, Fujikawa K, Takahashi M, Takayama T,
et al: Normalization of elevated hepatic
8-hydroxy-2′-deoxyguanosine levels in chronic hepatitis C patients
by phlebotomy and low iron diet. Cancer Res. 61:8697–8702.
2001.PubMed/NCBI
|
|
64
|
Louandre C, Ezzoukhry Z, Godin C, Barbare
JC, Mazière JC, Chauffert B and Galmiche A: Iron-dependent cell
death of hepatocellular carcinoma cells exposed to sorafenib. Int J
Cancer. 133:1732–1742. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Musto P, D'Auria F, Pietrantuono G,
Bringhen S, Morabito F, Di Raimondo F, Pozzi S, Sacchi S, Boccadoro
M and Palumbo A; Gruppo Italiano Malatte Ematologiche dell'Adulto
Multiple Myeloma Working Party; Italian Myeloma Network and Gruppo
Italiano Studio Linfomi. Role of thalidomide in previously
untreated patients with multiple myeloma. Expert Rev Anticancer
Ther. 8:1569–1580. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Mark TM, Reid W, Niesvizky R, Gergis U,
Pearse R, Mayer S, Greenberg J, Coleman M, Van Besien K and Shore
T: A phase 1 study of bendamustine and melphalan conditioning for
autologous stem cell transplantation in multiple myeloma. Biol
Blood Marrow Transplant. 19:831–837. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Sleigh SH and Barton DCL: Repurposing
strategies for therapeutics. Pharmaceut Med. 24:151–159. 2010.
|
|
68
|
Erickson-Miller CL, Delorme E, Tian S-S,
Hopson CB, Landis AJ, Valoret EI, Sellers TS, Rosen J, Miller SG,
Luengo JI, et al: Preclinical activity of eltrombopag (SB-497115),
an oral, nonpeptide thrombopoietin receptor agonist. Stem Cells.
27:424–430. 2009. View Article : Google Scholar :
|
|
69
|
Kawaguchi T, Nakano M, Satani M, Sumie S,
Yamada S, Amano K, Kuromatsu R and Sata M: Usefulness of short-term
eltrombopag treatment as a supportive treatment in hepatocellular
carcinoma patients with cirrhosis and severe thrombocytopenia: A
report of two cases. Oncol Lett. 7:2130–2134. 2014.PubMed/NCBI
|