Introduction
Allergic asthma is a chronic respiratory disease,
the prevalence of which is increasing rapidly worldwide (1). The characteristic pathophysiological
features of asthma include chronic eosinophilic inflammation of the
airways, airway hyper-responsiveness (AHR) and eventual progression
to airway remodeling (1). Nitric
oxide (NO) is derived from inducible NO synthase (iNOS), and has a
crucial role in airway inflammation (2). The development of allergic asthma is
associated with overexpression of iNOS in the airways, which
results in an asthmatic response. Previous studies have
demonstrated that iNOS expression is associated with the induced
activation of T-helper (Th)2 lymphocytes, which results in the
pathophysiological alterations that occur in asthma (2,3). Th2
lymphocytes produce various Th2 cytokines that have important roles
in the development of allergic asthma (3). Th2 cytokines include interleukin
(IL)-4, IL-5 and IL-13, and induce secretion of allergen-specific
immunoglobulin (Ig) E, chemokines and eosinophils, as well as mucus
production. Chemokines, such as eotaxin, are essential for the
delivery of eosinophils to the airways (4). Matrix metalloproteinase 9 (MMP-9) is
also associated with the development of allergic asthma. MMP-9
belongs to a family of extracellular proteases that are responsible
for the degradation of the extracellular matrix during tissue
remodeling (5). MMP-9 in
particular appears to have an important role in chronic airway
inflammation and remodeling in asthma, and was shown to be
upregulated in a murine asthma model in association with an
accumulation of inflammatory cells (6). The suppression of NO production and
MMP-9 expression is an important therapeutic target for asthma.
Zingiber mioga (Thunb.) Roscoe (ZM) is a
medicinal herb belonging to the Mioga family. ZM is globally used
to treat inflammatory diseases, including stomatitis (7). In addition to their extensive use as
spices, numerous types of ginger have been used in traditional
Oriental medicine in order to ameliorate symptoms, such as
inflammation, rheumatic disorders and gastrointestinal discomforts
(7). Previous studies have
demonstrated that ZM exhibits anti-inflammatory, anticancer and
antioxidant effects in vitro (7,8). Kim
et al (8) previously showed
that ZM was able to inhibit the production of inflammatory
mediators in lipopolysaccharide (LPS)-stimulated macrophage cells.
However, to the best of our knowledge, no study has investigated
the protective effects of ZM on airway inflammation in an
OVA-induced allergic asthma model.
The present study aimed to investigate the
inhibitory potential of ZM on airway inflammation in asthma, using
a murine model of OVA-induced asthma and LPS-stimulated RAW264.7
cells. In order to investigate the possible mechanism underlying
the effects of ZM, the abundance of pro-inflammatory proteins in
lung tissue was examined, as well as the expression of mRNA in
RAW264.7 cells.
Materials and methods
Cell culture and cell viability
The RAW264.7 murine macrophage cells (American Type
Tissue Collection, Manassas, VA, USA) were maintained in Dulbecco's
modified Eagle's medium (DMEM; Gibco Life Technologies, Grand
Island, NY, USA), supplemented with 10% heat-inactivated fetal
bovine serum (FBS; Gibco Life Technologies) and antibiotics (100
U/ml penicillin, 100 µg/ml streptomycin, Gibco Life
Technologies) at 37°C in an incubator containing 5% CO2
and 95% air. The cell viability of cells treated with ZM was
measured using a
3-(4,5-dimethylthiazol-2-yl)-2,5-di-phenyltetrazolium bromide (MTT;
Amresco, LLC, Solon, OH, USA) assay. The RAW264.7 cells were
cultured in 96-well plates at a density of 1×104
cells/well. The ZM extract was obtained from The Plant Extract Bank
at the Korea Research Institute of Bioscience and Biotechnology
(PB2176.2; Chungbuk, Korea). The ZM extract was added to each
individual well at a concentration of 20, 40, 80 and 100
µg/ml, and then incubated for 24 h. The control cells were
treated with an equivalent volume of phosphate-buffered saline
(PBS; pH 7.4). MTT solution (10 µl) was added to each well,
and the plates were incubated for 4 h at 37°C. After 4 h, 100
µl dimethyl sulfoxide (Samchun Pure Chemical Co., Ltd.,
Daejeon, Korea) was added to each well to solubilize the formazan
crystals. The optical density was measured at 570 nm using a
microplate reader (VersaMax; Molecular Devices, Sunnyvale, CA,
USA).
Determination of NO production
The cells (5×104 cells/well) were seeded
in 96-well plates in FBS-free DMEM, and were treated with various
concentrations of ZM (10, 20 and 40 µg/ml) for 1 h.
Subsequently, the cells were incubated in the presence of LPS (0.5
µg/ml) for 24 h. The nitrite accumulation in the culture
medium was measured using Griess reagent (Promega Corporation,
Madison, WI, USA). The absorbance was then measured at 540 nm using
a microplate reader (iMark Microplate absorbance reader; Bio-Rad
Laboratories, Inc. Hercules, CA, USA).
Determination of pro-inflammatory
cytokines
Cells were treated as described above, and the
concentration of tumor necrosis factor-α (TNF-α) was quantified in
the culture medium, using a competitive enzyme-linked immunosorbent
assay (ELISA) kit (BD Biosciences, San Jose, CA, USA), according to
the manufacturer's instructions. The absorbance was measured at 450
nm in a microplate reader (Bio-Rad Laboratories, Inc.). The
absolute concentrations were obtained by running standard curves on
the identical ELISA plates.
Reverse transcriptase-polymerase chain
reaction (RT-PCR)
Total RNA was extracted from the cells using
TRIzol® reagent (Life Technologies, Carlsbad, CA, USA).
For RT-PCR, single-strand cDNA (Qiagen, Hilden, Germany) was
synthesized from 2 µg total RNA using a commercial PCR kit
(166-2600EDU; Bio-Rad Laboratories Inc.) and a T100 thermal cycler
(186–1096; Bio-Rad Laboratories Inc.). The primer sequences used in
the PCR were as follows: Forward: 5′-CAAGAGTTTGACCAGAGGACC-3′, and
reverse: 5′-TGGAACCACTCGTACTTGGGA-3′ for iNOS, forward:
5′-AAGCACATGCAGAATGAGTACCG-3′ and reverse:
5′-GTGGGACAGCTTCTGGTCGAT-3′ for MMP-9 and forward:
5′-CGCTCATTGCCGATAGTGAT-3′ and reverse: 5′-TGTTTGAGACCTTCAACACC-3′
for β-actin (Bioneer, Daejeon, Korea). The following cycle
conditions were set for each PCR reaction: iNOS, forward, 95°C for
5 min, then 94°C for 30 sec, 58°C for 30 sec and 72°C for 45 sec
for 32 cycles, and a final extension step at 72°C for 5 min; MMP-9
and β-actin, forward, 94°C for 5min, then 94°C for 30 sec, 50°C for
30 sec and 72°C for 45 sec for 30 cycles, and a final extension
step at 72°C for 10 min. The PCR products were then subjected to
1.5% agarose gel electrophoresis and were stained with 5
µg/ml ethidium bromide (Bio-Rad Laboratories Inc.). Images
were captured using an Olympus C4000 zoom camera system (Olympus
America Inc., Center Valley, PA, USA)
Mice
Specific pathogen-free female BALB/c mice (19–21 g,
6–8 weeks-old) were obtained from Koatech Co. (Pyeongtaek, Korea).
The 28 mice were housed under standard conditions (temperature,
22±2°C; humidity, 55±5%; 12 h-light/dark cycle) with food and
water. All of the experimental procedures were approved by the
Institutional Animal Care and Use Committee of the Korea Research
Institute of Bioscience and Biotechnology.
Induction of asthma in BA LB/c mice and
drug administration
The mice were divided into four groups (n=7): Normal
control, PBS sensitization/challenge; OVA, OVA
sensitization/challenge + PBS; Mon, OVA sensitization/challenge +
montelukast (30 mg/kg; Sigma-Aldrich, St. Louis, MO, USA); ZM, OVA
sensitization/challenge + ZM (30 mg/kg). Airway inflammation was
induced by an OVA (Sigma-Aldrich) challenge. Each mouse was
immunized by an intraperitoneal injection of 20 µg OVA,
emulsified with 2 mg aluminum hydroxide (Thermo Fisher Scientific,
Waltham, MA, USA) in 200 µl PBS (pH 7.4), on days 0 and 14.
On days 21, 22 and 23 the mice were exposed, by inhalation, to 1%
(w/v) OVA aerosolized solution using an ultrasonic nebulizer
(NE-U12; Omron Corp., Tokyo, Japan) for 1 h. The ZM groups of
asthma-induced mice were treated orally with 30 mg/kg ZM, or with
an oral injection of 30 mg/kg montelukast 1 h prior to the OVA
challenge. Montelukast is a cysteinyl leukotriene (cys-LT)-1
receptor agonist, and was used as a positive control in the present
study. The control mice were treated orally with an equivalent
volume of PBS.
Measurement of AHR
A total of 24 hours after the final aerosol
challenge, AHR was measured indirectly using single-chamber whole
body plethysmography (Allmedicus, Seoul, Korea) following exposure
to aerosolized PBS, followed by aerosols containing increasing
concentrations of methacholine (12.5, 25 and 50 mg/ml;
Sigma-Aldrich) in PBS, for 3 min at each exposure level.
Collection of bronchoalveolar lavage
fluid (BALF) and serum
For collection of BALF, the mice were sacrificed 24
h after AHR measurement with an intraperitoneal injection of
phenobarbital (50 mg/kg; Hanlim Pharm., Co., Ltd., Seoul, Korea),
and tracheotomy was performed as described previously (9). The lung was lavaged three times, and
each time, fluid was withdrawn via the trachea with 0.5 ml PBS
(total volume, 1.5 ml). The number of inflammatory cells was
determined by counting the cells in ≥5 squares of a hemocytometer
(Marienfeld, Lauda-Königshofen, Germany), following exclusion of
the dead cells by Trypan blue (Sigma-Aldrich) staining. The
differential cell count of the BALF was performed using
Diff-Quik® staining reagent (B4132–1A; IMEB Inc., San
Marcos, CA, USA), according to the manufacturer's instructions. The
blood was collected from the inferior vena cava and centrifuged at
200 x g for 10 min at 4°C. The supernatant was then stored at -70°C
until further use.
Measurement of immunoglobulin (Ig)E in
serum and cytokines in BALF
The Th2 cytokines (IL-4, IL-5, IL-13) and eotaxin in
BALF were measured using Mouse IL-4, IL-5 and IL-13 Quantikine
ELISA kits (R&D Systems, Minneapolis, MN, USA), according to
the manufacturer's instructions. The total IgE and OVA-specific IgE
serum levels were measured by ELISA. Briefly, microtiter plates
were coated with anti-IgE antibodies (anti-mouse IgE; 10 g/ml; AbD
Serotec, Oxford, UK) in PBS-Tween 20, and incubated with BALF or
plasma samples. The plates were then washed four times with washing
solution (contained in ELISA kits), and 200 µl
o-phenylenediamine dihydrochloride (Sigma-Aldrich) was added to
each well. The plates were incubated for 10 min in the dark and the
absorbance was measured at 450 nm using the iMark microplate
absorbance reader.
Immunoblotting
The mouse lung tissue was homogenized (1/10 w/v) in
Tissue Lysis/Extraction reagent (Sigma-Aldrich) that contained a
protease inhibitor cocktail (Sigma-Aldrich) using a homogenizer
(IKA, Guangzhou, China). In order to extract the nuclear protein,
the lung tissue was homogenized using an extraction kit (Thermo
Fisher Scientific). The protein concentrations of the samples were
determined using Bradford reagent (Bio-Rad Laboratories, Inc.).
Equal quantities of total protein (30 µg) were separated by
10% SDS-polyacrylamide gel electrophoresis and transferred onto a
polyvinylidene fluoride membrane (IPVH00010; Merck Millipore,
Damstadt, Germany). The membranes were then incubated with blocking
solution (5% non-fat milk), followed by an overnight incubation at
4°C with the appropriate primary antibodies. The following primary
antibodies and dilutions were used: Rabbit monoclonal anti-β-actin
(1:2,000 dilution; cat. no. P60709; Cell Signaling Technologies,
Inc., Danvers, MA, USA); rabbit polyclonal anti-iNOS (1:1,000
dilution; cat. no. ADI-KAS-NO001-F; Enzo Life Sciences Inc.,
Farmingdale, NY, USA) and rabbit polyclonal anti-MMP-9 (1:1,000
dilution; cat. no. ab38898; Cell Signaling Technologies, Inc.). The
blots were washed three times with tris-buffered saline containing
Tween-20 (TBST; Biosesang, Seongnam, Korea) and then incubated with
a 1:10,000 dilution of horseradish peroxidase (HRP)-conjugated
secondary antibody (cat. no. 211–542–171; Jackson ImmunoResearch
Laboratories, Inc., West Grove, PA, USA) for 30 min at room
temperature. The blots were washed a further three times with TBST,
and then developed using an Enhanced Chemiluminescence kit (Thermo
Fisher Scientific).
Lung tissue histopathology
The lung tissue was fixed in 4% (v/v)
paraformaldehyde, embedded in paraffin, and cut into 4-µm
sections, prior to staining with hematoxylin and eosin solution
(Sigma-Aldrich) and periodic acid-Schiff (IMEB Inc.), in order to
detect the inflammation and mucus production of the lung sections,
respectively. Tissue sections were mounted and coverslipped.
Pathological changes to the lung tissue were examined using a
microscope (Leica DM1000; Leica Microsystems, Wetzlar,
Germany).
Statistical analysis
Data are expressed as the mean ± standard deviation.
The statistical significance of the results were determined by
analysis of variance, followed by a multiple comparison test with
Dunnet's adjustment. Statistical analyses were performed using
GraphPad InStat v 3.0 (GraphPad, La Jolla, CA, USA). P<0.05 was
considered to indicate a statistically significant difference.
Results
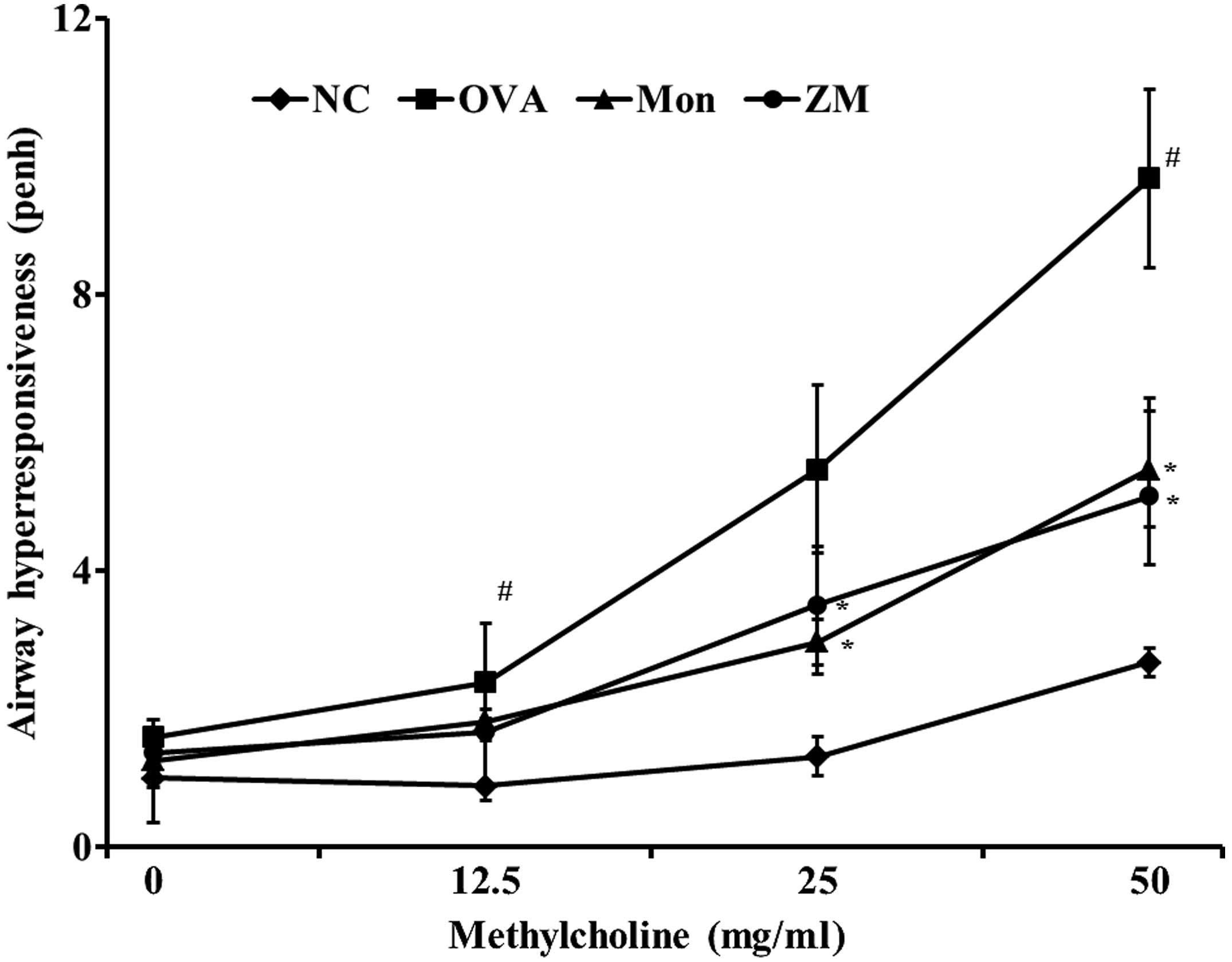
ZM reduces OVA-induced AHR
The OVA-induced mice exhibited a significant
increase in AHR with increasing concentrations of methylcholine
exposure, compared with the normal control mice. The
montelukast-treated mice exhibited significantly reduced AHR,
compared with the OVA-induced mice. The ZM-treated mice also
exhibited decreased AHR at methylcholine concentrations of 25 and
50 mg/ml, compared with the OVA-challenged mice. These reductions
were similar to the results observed in the montelukast-treated
mice (Fig. 1).
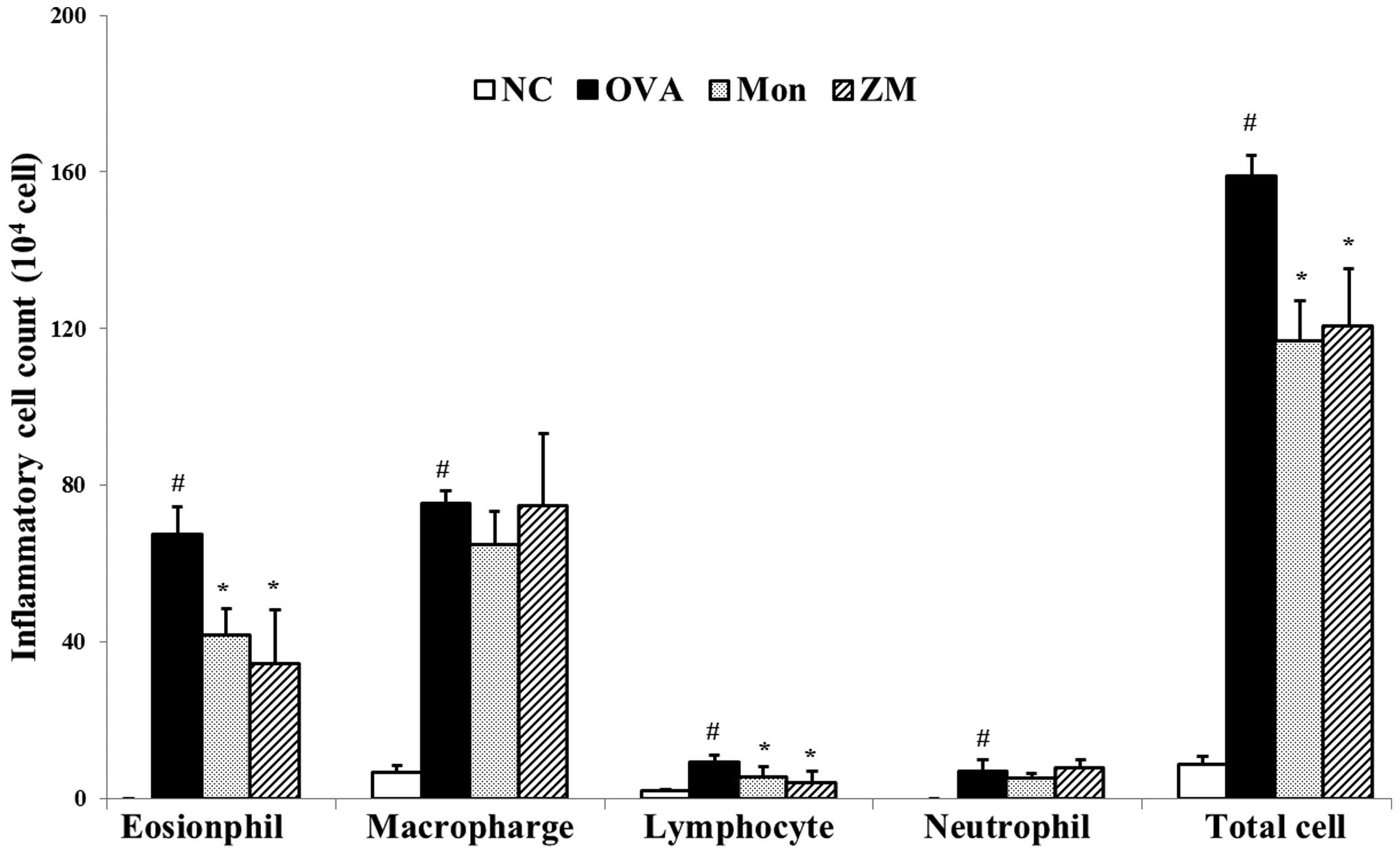
ZM decreases the number of inflammatory
cells in BALF
Significantly increased numbers of inflammatory
cells, particularly eosinophils, were observed in the BALF samples
from OVA-induced mice compared with samples from the normal
controls. BALF samples from the ZM-treated mice exhibited a
markedly decreased number of inflammatory cells, including
eosinophils, macrophages, lymphocytes and neutrophils, compared
with samples from the OVA-challenged mice (Fig. 2).
ZM reduces the levels of proinflammatory
cytokines in BALF, and levels of IgE in serum
The levels of IL-4, IL-5, IL-13 and eotaxin were
significantly increased in the BALF from OVA-challenged mice,
compared with that from the normal controls. However,
montelukast-treated mice had significantly reduced levels of these
cytokines, as compared with the OVA-challenged mice. The levels of
these cytokines were also significantly decreased in the BALF of
the ZM-treated mice, compared with that of the OVA-challenged mice
(Fig. 3A and B). The total IgE and
OVA-specific IgE serum levels in the OVA-challenged mice were
significantly elevated, compared with levels in the normal
controls. Conversely, montelukast-treated mice had markedly
decreased levels of total and OVA-specific IgE, as compared with
the OVA-challenged mice. The ZM-treated mice also exhibited a
significant reduction in the total IgE and OVA-specific IgE serum
levels, compared with those in the OVA-challenged mice (Fig. 3C and D).
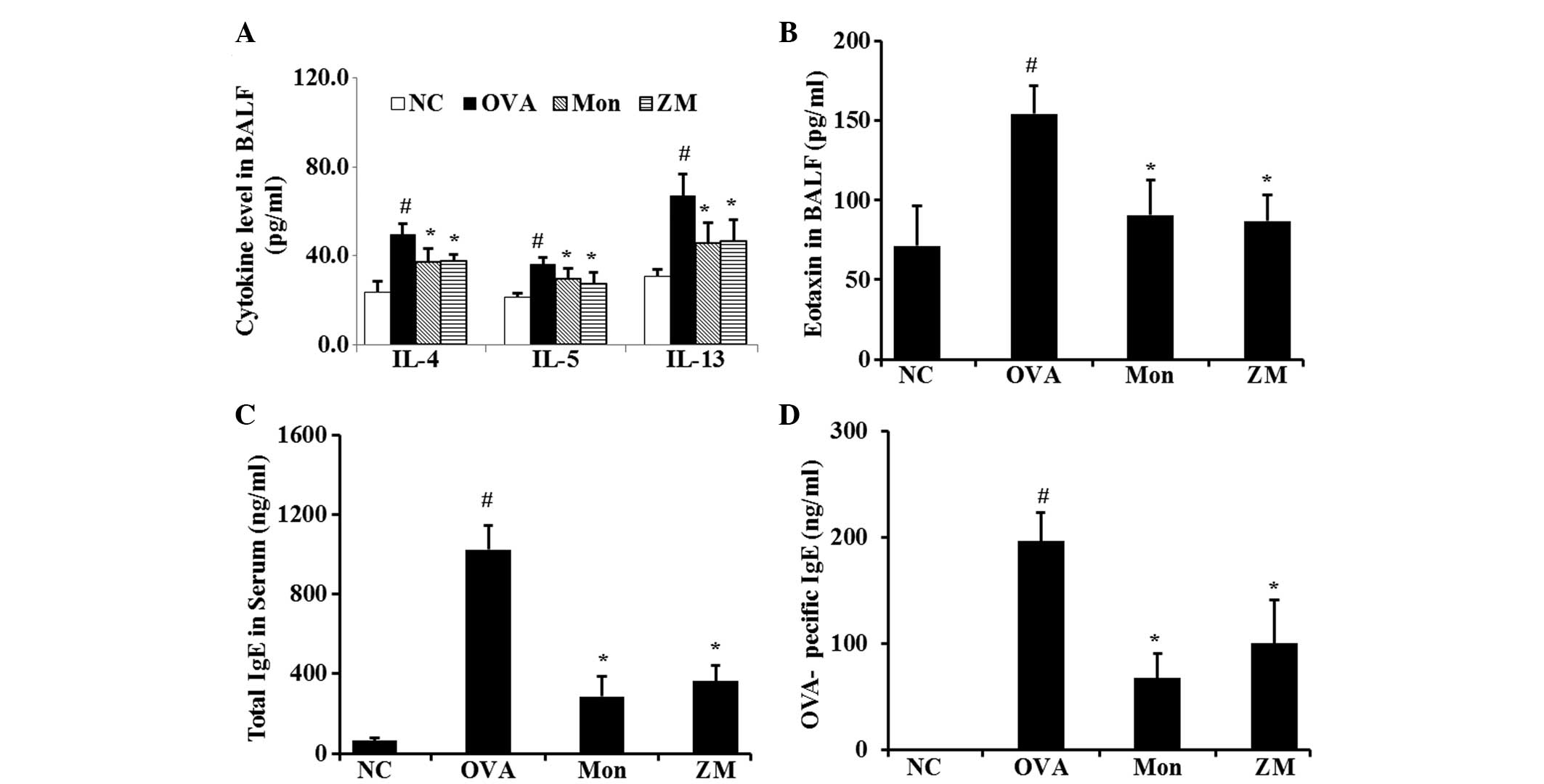 | Figure 3ZM inhibited the levels of
pro-inflammatory cytokines, eotaxin and IgE. Levels of (A)
pro-inflammatory cytokines in BALF, (B) chemokine in BALF, (C)
total IgE in serum and (D) OVA-specific IgE in serum, as determined
by ELISA. Data are expressed as the mean ± standard deviation
(n=7/group). #P<0.05, compared with NC and
*P<0.05, compared with OVA. NC, normal control mice;
OVA, ovalbumin-challenged mice; Mon, montelukast (30 mg/kg) +
OVA-challenged mice; ZM, Zingiber mioga (Thunb.) Roscoe (30
mg/kg) + OVA-challenged mice; BALF, bronchoalveolar lavage fluid;
IL, interleukin; IgE, immunoglobulin E; NC, normal control. |
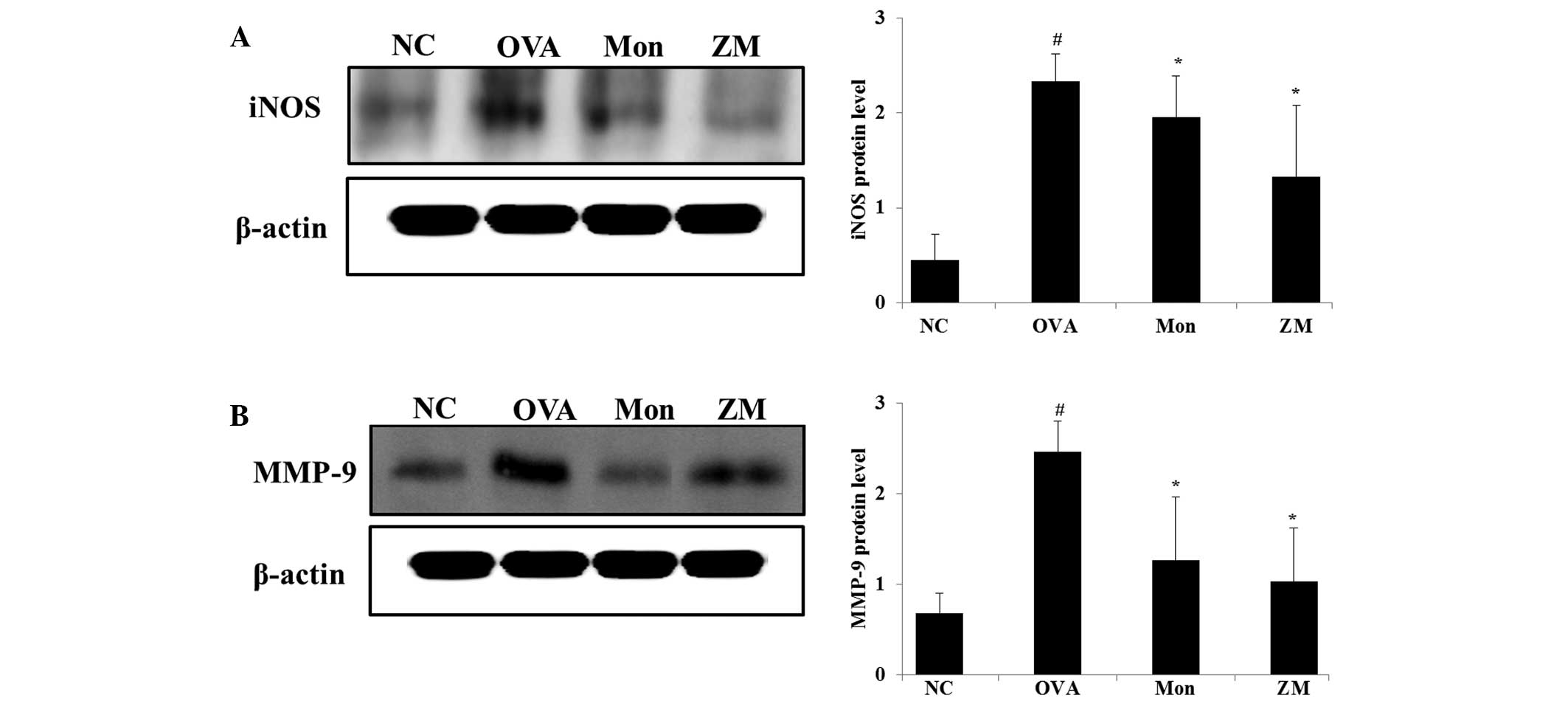
ZM inhibits protein expression levels of
iNOS and MMP-9 in the lung tissue of OVA-challenged mice
OVA-challenged mice exhibited significantly
increased protein expression levels of iNOS in the lung tissue,
compared with the normal controls. However, montelukast-treated
mice exihibited significantly reduced iNOS expression in lung
tissue, as compared with the OVA-challenged mice. In addition, in
the ZM-treated mice, a marked reduction in the expression levels of
iNOS were detected, as compared with the OVA-challenged mice
(Fig. 4A). In the OVA-challenged
mice, MMP-9 expression was increased, compared with the normal
controls, whereas they were significantly decreased in the
montelukast- and ZM-treated mice, compared with the OVA-challenged
mice (Fig. 4B).
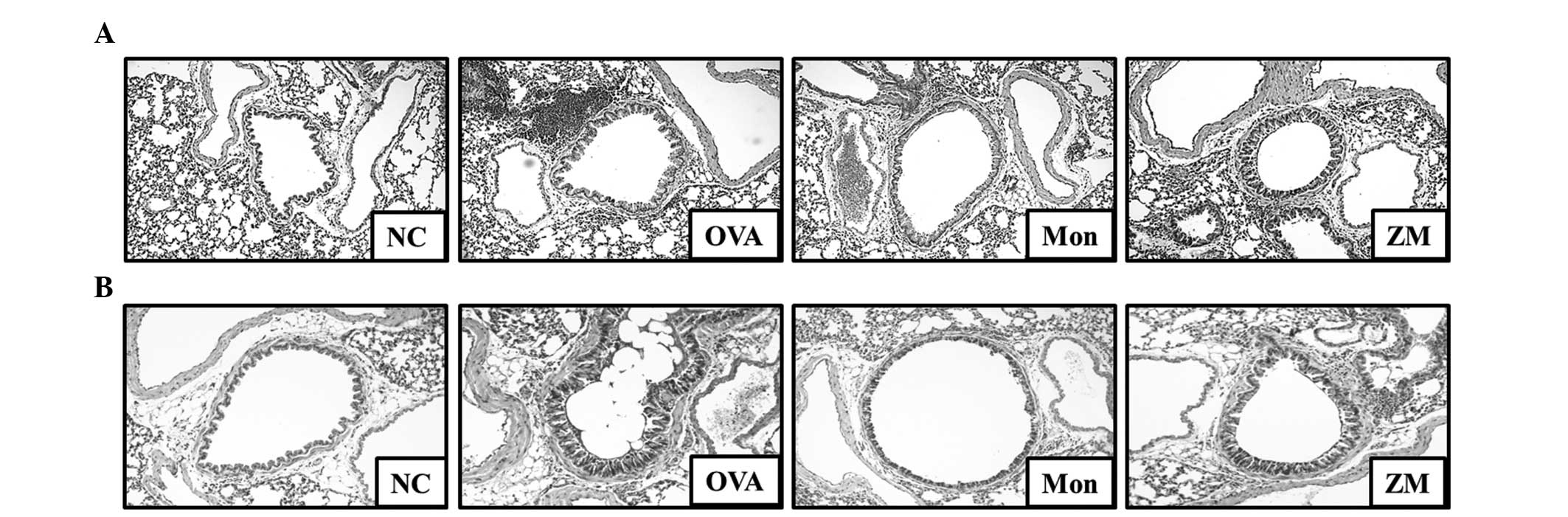
ZM decreases inflammatory cell
infiltration and mucus production
Lung tissue from the OVA-challenged mice exhibited
infiltration of inflammatory cells into the peribronchial and
perivascular lesions, and mucus hypersecretion in airway epithelial
cells (Fig. 5A and B). However,
lung tissue from montelukast-treated mice exhibited a reduction in
inflammatory cell infiltration and mucus secretion. In addition,
lung tissue from ZM-treated mice exhibited significantly lower
inflammatory cell infiltration and mucus secretion, compared with
the OVA-challenged mice.
ZM reduces NO production and TNF-α in
LPS-stimulated cells
In the present study, nontoxic concentrations of ZM
(20, 40, 60 and 80 µg/ml; Fig.
6A) were tested for their ability to inhibit LPS-stimulated NO
production. The LPS-stimulated cells exhibited increased cellular
NO levels, compared with non-stimulated cells. By contrast, the
production of NO was inhibited in the cells pre-treated with ZM, in
a concentration-dependent manner (Fig.
6B).
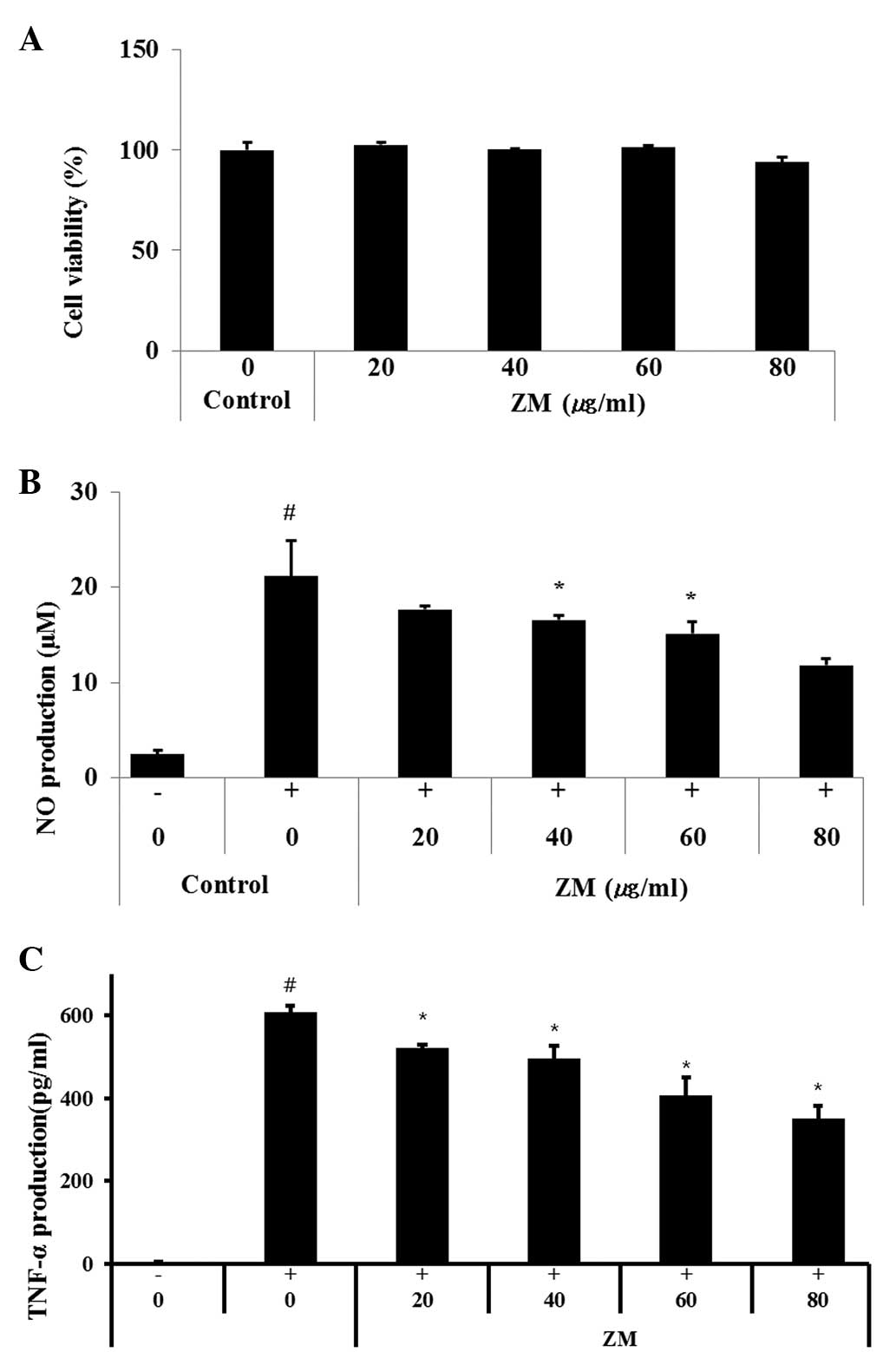 | Figure 6RAW264.7 murine macrophage cells were
treated with 20, 40, 60 or 80 µg/ml of ZM for 24 h. (A) Cell
survival rates were determined using an MTT assay. ZM inhibited the
production of (B) NO and (C) TNF-α in the LPS stimulated RAW264.7
cells. Cells were treated with ZM (20, 40, 60 or 80 µg/ml)
for 1 h and with LPS (0.5 µg/ml) for 24 h. Data are
expressed as the mean ± standard deviation. #P<0.05,
compared with control cells and *P<0.05, compared
with LPS-treated cells. ZM, Zingiber mioga (Thunb.)
Roscoe; LPS, lipopolysaccharide; NO, nitric oxide; TNF-α,
tumor necrosis factor-α. |
As shown in Fig.
6C, the LPS-stimulated cells exhibited markedly increased
levels of TNF-α, compared with the normal controls, whereas the
ZM-treated cells exhibited a significant concentration-dependent
decrease in the levels of TNF-α, compared with the LPS-stimulated
cells.
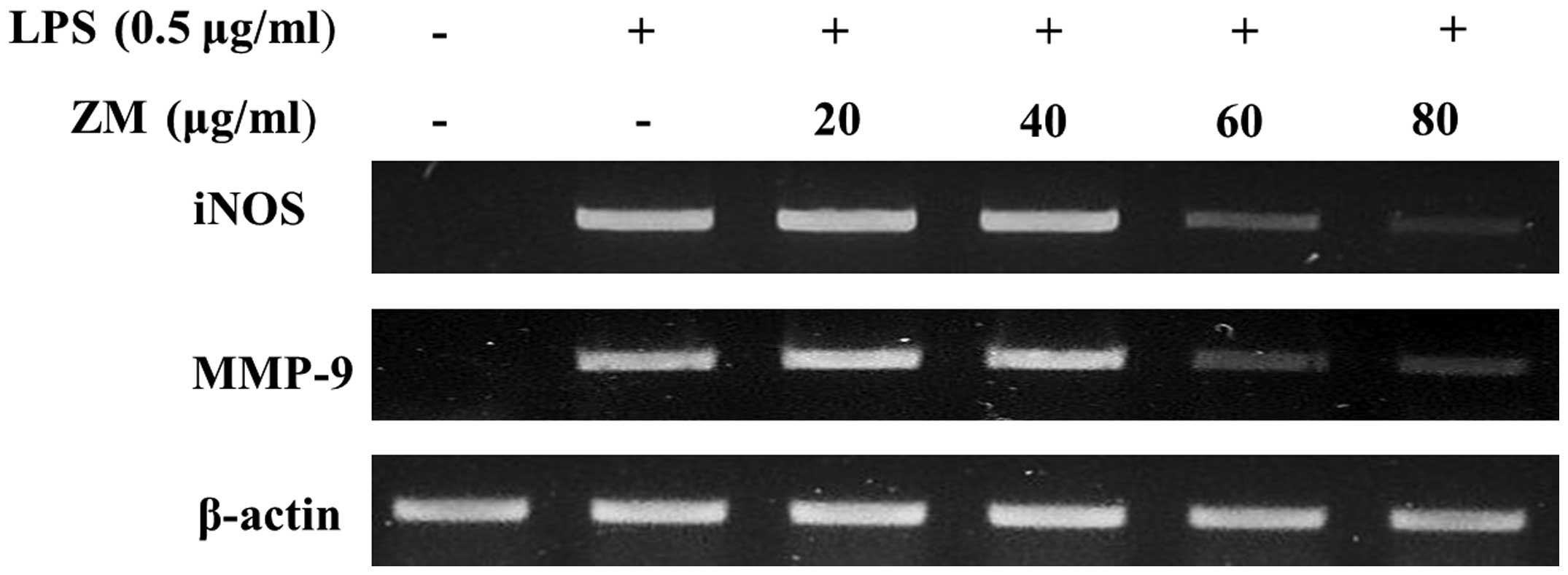
ZM inhibits iNOS and MMP-9 mRNA
expression in LPS-stimulated cells
The LPS-stimulated cells exhibited a significant
increase in mRNA expression levels of iNOS, TNF-α and MMP-9,
compared with the control group. The LPS-induced mRNA expression
levels of iNOS and MMP-9 were significantly reducted in the
ZM-treated cells (Fig. 7).
Discussion
The present study aimed to evaluate the
anti-inflammatory effects of ZM in an OVA-induced allergic asthma
murine model, and in LPS-stimulated RAW264.7 cells. ZM decreased
the number of inflammatory cells, the Th2 cytokine levels and the
levels of eotaxin in BALF from mice treated with OVA. In addition,
mice treated with ZM produced significantly reduced serum levels of
total and OVA-specific IgE, compared with the OVA-challenged mice.
ZM also decreased the protein expression levels of iNOS and MMP-9
in the lung tissue, as well as reducing AHR, airway inflammation
and mucus production. Furthermore, ZM inhibited the production of
NO and TNF-α in the LPS-stimulated RAW264.7 cells, and reduced the
mRNA expression levels of iNOS, TNF-α and MMP-9.
Allergic asthma is a chronic inflammatory airway
disease that is characterized by persistent AHR, Th2 lymphocyte
infiltration and plasma exudation. Allergic asthma is associated
with an elevated serum IgE level, eosinophilia and mucus production
in the airways (10,11). NO is a particularly important
inflammatory mediator of allergic asthma. NO is a gas molecule
synthesized by NOS, which is associated with inflammatory
responses. It has previously been suggested that iNOS is induced by
inflammatory cytokines, such as TNF-α, and results in the
production of much larger amounts of NO (12). The overproduction of NO underlies
pathophysiology of airway inflammation. NO contributes to the
infiltration of inflammatory cells, such as neutrophils, monocytes
and eosinophils, which produce IL-4, IL-5 and IL-13 Th2 cytokines
(6,13). IL-4 promotes B-cell isotype
switching and the upregulation of IgE, whereas IL-5 induces the
infiltration of eosinophils into the airways (14). IL-13 is also a potent stimulator of
airway remodeling, since it induces lung and alveolar enlargement,
as well as mucus metaplasia and AHR (15,16).
In the present study, Th2 cytokines, including IL-4, IL-5, and
IL-13, were markedly decreased in the ZM-treated mice. The
ZM-treated mice also exhibited a significant reduction in the
number of inflammatory cells, which was accompanied by decreased
levels of total and OVA-specific IgE, and eotaxin. Treatment with
ZM reduced airway inflammation and mucus production alongside
inhibition of iNOS expression in the lung tissue. These findings
indicated that ZM may effectively inhibit the development of
allergic asthma induced by an OVA challenge. It was hypothesized
that this may be associated with the suppression of iNOS
expression. In addition, ZM reduced NO and TNF-α production, and
iNOS levels in the LPS-stimulated RAW264.7 cells in a
concentration-dependent manner.
MMPs are a family of extracellular proteases that
are responsible for the degradation of the extracellular matrix
during tissue remodeling. Within the MMP family, the MMP-9
gelatinases are specific to the denatured collagens and collagen-IV
of the basement membrane (17).
MMP-9 is controlled by the inflammatory mediator NO, as well as
cytokines and chemokines, and is produced by various types of
cells, including inflammatory, endothelial and epithelial cells
(18). MMP-9 is associated with
airway remodeling and inflammatory cell infiltration into the
airways. Previous studies have demonstrated that increased
expression levels of MMP-9 in patients with asthma are associated
with an increase in the number of inflammatory cells (19,20).
Inhibition of MMP-9 has been shown to significantly reduce the
production of Th2 cytokines and IgE, in addition to the expression
levels of adhesion molecules, which results in the alleviation of
asthmatic responses, such as airway inflammation, mucus
overproduction, and AHR (21).
Numerous studies have explored novel therapeutic materials focusing
on the suppression of MMP-9 expression in asthma (6,14,17).
The present study evaluated the protein expression levels of MMP-9
in lung tissue. ZM-treated mice exhibited reduced MMP-9 expression
in lung tissue, compared with the OVA-challenged mice. These
findings indicated that ZM may reduce asthmatic responses by
downregulating the expression of MMP-9 in lung tissue.
In conclusion, ZM effectively suppressed
inflammatory responses in the OVA-challenged mice and
LPS-stimulated RAW264.7 cells. Based on these results, the
anti-inflammatory effects of ZM were hypothesized to be associated
with the suppression of iNOS and MMP-9 expression. The findings
suggested that ZM may be a potent therapeutic agent in inflammatory
diseases, such as allergic asthma.
Acknowledgments
The present study was supported by a grant from the
KRIBB research Initiative program (grant. no. KGM 1221521) of the
Republic of Korea.
References
|
1
|
Shalaby KH, Allard-Coutu A, O'Sullivan MJ,
Nakada E, Qureshi ST, Day BJ and Martin JG: Inhaled birch pollen
extract induces airway hyperresponsiveness via oxidative stress but
independently of pollen-intrinsic NADPH oxidase activity, or the
TLR-4-TRIF pathway. J Immunol. 191:922–933. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Prado CM, Martins MA and Tibério IF:
Nitric oxide in asthma physiopathology. ISRN Allergy.
2011:8325602011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lee JH, Shon JH, Ryu SY, Hong CS, Moon KD
and Park JW: A novel human anti-VCAM-1 monoclonal antibody
ameliorates airway inflammation and remodelling. J Cell Mol Med.
17:1271–1281. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Deng Y, Guan M, Xie X, Yang X, Xiang H, Li
H, Zou L, Wei J, Wang D and Deng X: Geniposide inhibits airway
inflammation and hyperresponsiveness in a mouse model of asthma.
Int Immunopharmacol. 17:561–567. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lee MY, Shin IS, Lim HS and Shin HK: A
water extract of Samchulkunbi-tang attenuates airway inflammation
by inhibiting inos and MMP-9 activities in an ovalbumin-induced
murine asthma model. BMC Complement Altern Med. 12:2572012.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Shin IS, Lee MY, Lim HS, Ha H, Seo CS, Kim
JC and Shin HK: An extract of Crataegus pinnatifida fruit
attenuates airway inflammation by modulation of matrix
metalloproteinase-9 in ovalbumin induced asthma. PLoS One.
7:e457342012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Miyoshi N, Nakamura Y, Ueda Y, Abe M,
Ozawa Y, Uchida K and Osawa T: Dietary ginger constituents,
galanals A and B, are potent apoptosis inducers in Human T lymphoma
Jurkat cells. Cancer Lett. 199:113–119. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kim HW, Murakami A, Abe M, Ozawa Y,
Morimitsu Y, Williams MV and Ohigashi H: Suppressive effects of
mioga ginger and ginger constituents on reactive oxygen and
nitrogen species generation, and the expression of inducible
pro-inflammatory genes in macrophages. Antioxid Redox Signal.
7:1621–1629. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Shin IS, Park JW, Shin NR, Jeon CM, Kwon
OK, Lee MY, Kim HS, Kim JC, Oh SR and Ahn KS: Melatonin inhibits
MUC5AC production via suppression of MAPK signaling in human airway
epithelial cells. J Pineal Res. 56:398–407. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ra J, Lee S, Kim HJ, Jang YP, Ahn H and
Kim J: Bambusae Caulis in Taeniam extract reduces ovalbumin-induced
airway inflammation and T helper 2 responses in mice. J
Ethnopharmacol. 128:241–247. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Nader MA, El-Awady MS, Shalaby AA and
El-Agamy DS: Sitagliptin exerts anti-inflammatory and anti-allergic
effects in ovalbumin-induced murine model of allergic airway
disease. Naunyn Schmiedebergs Arch Pharmacol. 385:909–919. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Koarai A, Ichinose M, Sugiura H, Tomaki M,
Watanabe M, Yamagata S, Komaki Y, Shirato K and Hattori T: iNOS
depletion completely diminishes reactive nitrogen-species formation
after an allergic response. Eur Respir J. 20:609–616. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Schmudde I, Laumonnier Y and Köhl J:
Anaphylatoxins coordinate innate and adaptive immune responses in
allergic asthma. Semin Immunol. 25:2–11. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Shin IS, Hong J, Jeon CM, Shin NR, Kwon
OK, Kim HS, Kim JC, Oh SR and Ahn KS: Diallyl-disulfide, an
organosulfur compound of garlic, attenuates airway inflammation via
activation of the Nrf-2/HO-1 pathway and NF-kappaB suppression.
Food Chem Toxicol. 62:506–513. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Simoes DC, Xanthou G, Petrochilou K,
Panoutsakopoulou V, Roussos C and Gratziou C: Osteopontin
deficiency protects against airway remodeling and
hyperresponsiveness in chronic asthma. Am J Respir Crit Care Med.
179:894–902. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Shen JJ, Chiang MS, Kuo Ml, Leu YL, Hwang
TL, Liou CJ and Huang WC: Partially purified extract and viscolin
from Viscum coloratum attenuate airway inflammation and eosinophil
infiltration in ovalbumin-sensitized mice. J Ethnopharmacol.
135:646–653. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hsu CH, Hu CM, Lu KH, Yang SF, Tsai CH, Ko
CL, Sun HL and Lue KH: Effect of selective cysteinyl leukotriene
receptor antagonists on airway inflammation and matrix
metalloproteinase expression in a mouse asthma model. Pediatr
Neonatol. 53:235–244. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Oh YC, Cho WK, Jeong YH, Im GY, Kim A,
Hwang YH, Kim T, Song KH and Ma JY: A novel herbal medicine KIOM-MA
exerts an anti-inflammatory effect in LPS-stimulated RAW 264.7
macrophage cells. Evid Based Complement Alternat Med.
2012:4623832012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gueders MM, Foidart JM, Noel A and Cataldo
DD: Matrix metalloproteinases (MMPs) and tissue inhibitors of MMPs
in the respiratory tract: Potential implications in asthma and
other lung diseases. Eur J Pharmacol. 533:133–144. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ventura I, Vega A, Chacón P, Chamorro C,
Aroca R, Gómez E, Bellido V, Peunte Y, Blanca M and Monteseirin J:
Neutrophils from allergic asthmatic patients produce and release
metalloproteinase-9 upon direct exposure to allergens. Allergy.
69:898–905. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lee KS, Jin SM, Kim HJ and Lee YC: Matrix
metalloproteinase inhibitor regulates inflammatory cell migration
by reducing ICAM-1 and VCAM-1 expression in a murine model of
toluene diisocyanate-induced asthma. J Allergy Clin Immunol.
111:1278–1284. 2003. View Article : Google Scholar : PubMed/NCBI
|