|
1
|
Hall ED and Springer JE: Neuroprotection
and acute spinal cord injury: A reappraisal. NeuroRx. 1:80–100.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lewen A, Matz P and Chan PH: Free radical
pathways in CNS injury. J Neurotrauma. 17:871–890. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chinopoulos C and Adam-Vizi V:
Mitochondrial Ca2+ sequestration and precipitation
revisited. FEBS J. 277:3637–3651. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Pivovarova NB and Andrews SB:
Calcium-dependent mitochondrial function and dysfunction in
neurons. FEBS J. 277:3622–3636. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Szabo I and Zoratti M: The mitochondrial
megachannel is the permeability transition pore. J Bioenerg
Biomembr. 24:111–117. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zoratti M and Szabó I: The mitochondrial
permeability transition. Biochim Biophys Acta. 1241:139–176. 1995.
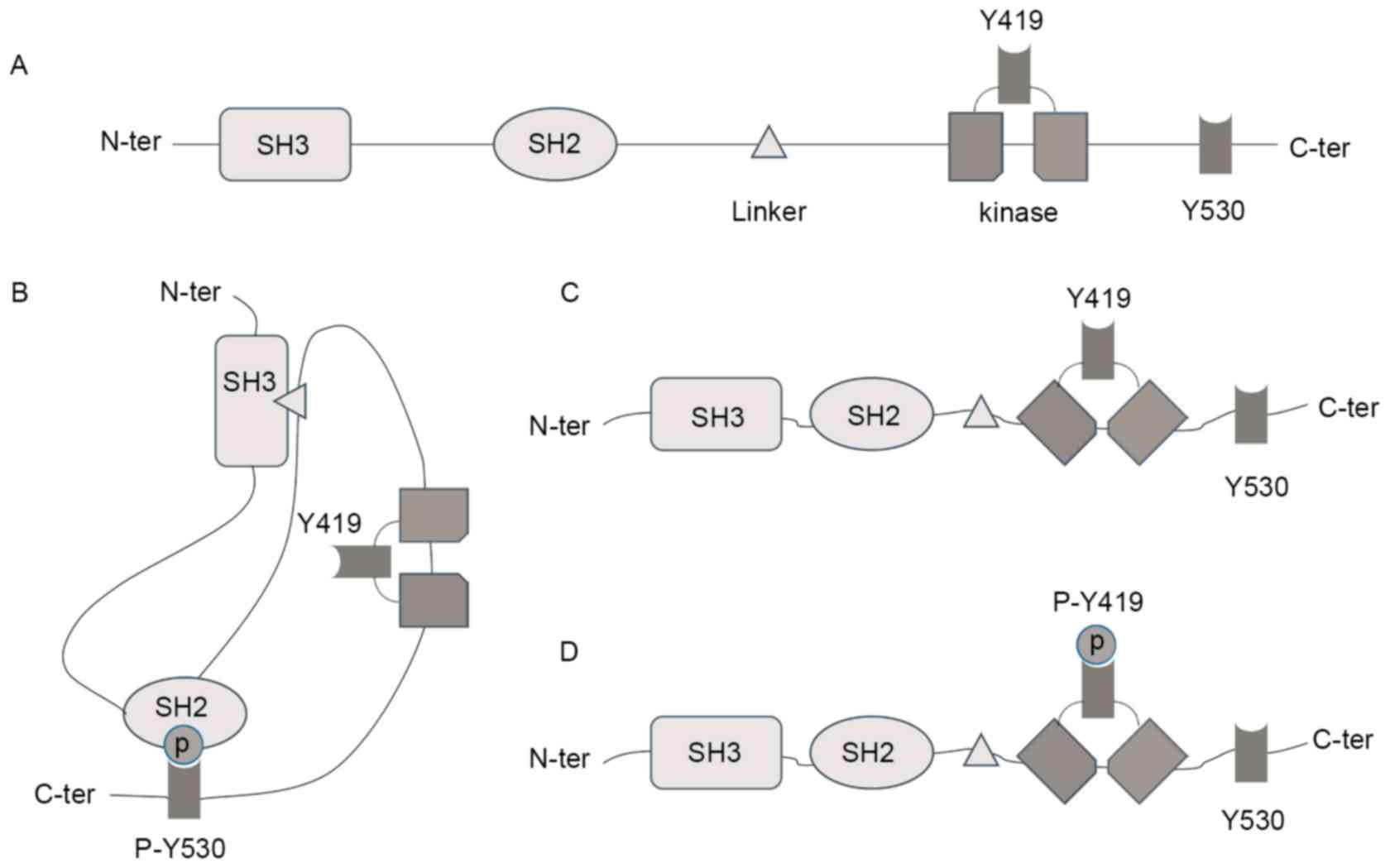
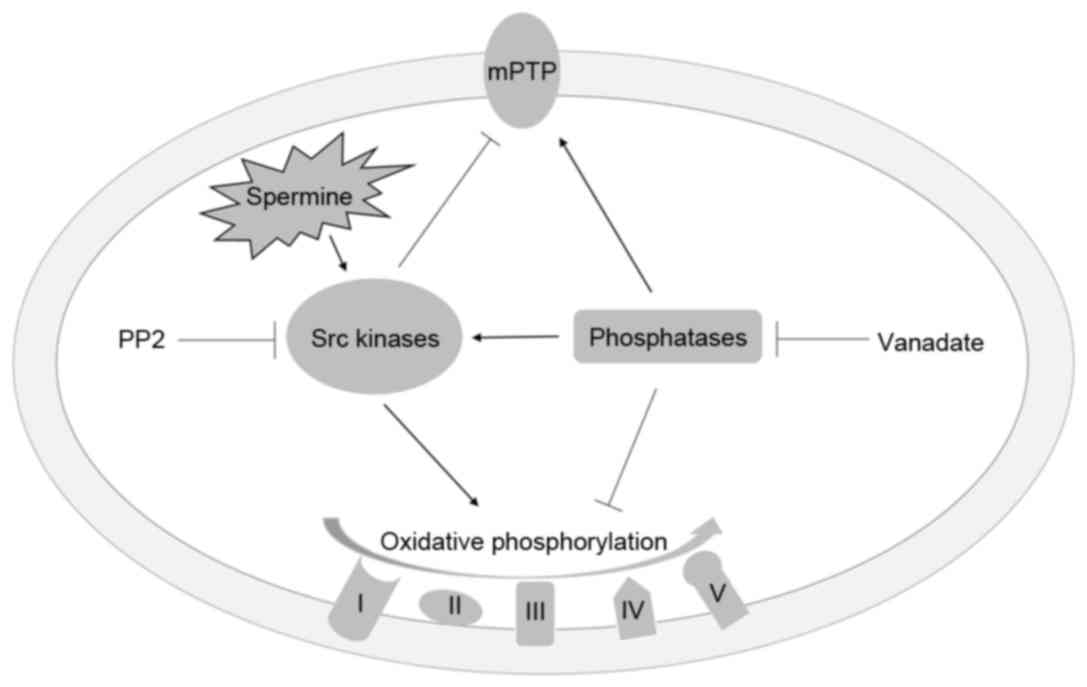
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sesso A, Marques MM, Monteiro MM,
Schumacher RI, Colquhoun A, Belizário J, Konno SN, Felix TB,
Botelho LA, Santos VZ, et al: Morphology of mitochondrial
permeability transition: morphometric volumetry in apoptotic cells.
Anat Rec A Discov Mol Cell Evol Biol. 281:1337–1351. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Christofferson DE and Yuan J: Necroptosis
as an alternative form of programmed cell death. Curr Opin Cell
Biol. 22:263–268. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Duprez L, Wirawan E, Vanden Berghe T and
Vandenabeele P: Major cell death pathways at a glance. Microbes
Infect. 11:1050–1062. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
McEwen ML, Sullivan PG, Rabchevsky AG and
Springer JE: Targeting mitochondrial function for the treatment of
acute spinal cord injury. Neurotherapeutics. 8:168–179. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Dalla Via L, Di Noto V, Siliprandi D and
Toninello A: Spermine binding to liver mitochondria. Biochim
Biophys Acta. 1284:247–252. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Siliprandi D, Toninello A and Dalla Via L:
Bidirectional transport of spermine in rat liver mitochondria.
Biochim Biophys Acta. 1102:62–66. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Pezzato E, Battaglia V, Brunati AM,
Agostinelli E and Toninello A: Ca2+ -independent effects
of spermine on pyruvate dehydrogenase complex activity in energized
rat liver mitochondria incubated in the absence of exogenous
Ca2+ and Mg2+. Amino Acids. 36:449–456. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Agostinelli E, Tempera G, Molinari A,
Battaglia V, Toninello A and Arancia G: The physiological role of
biogenic amines redox reactions in mitochondria. New perspectives
in cancer therapy. Amino Acids. 33:175–187. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sava IG, Battaglia V, Rossi CA, Salvi M
and Toninello A: Free radical scavenging action of the natural
polyamine spermine in rat liver mitochondria. Free Radic Biol Med.
41:1272–1281. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cesaro L and Salvi M: Mitochondrial
tyrosine phosphoproteome: New insights from an up-to-date analysis.
Biofactors. 36:437–450. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lewandrowski U, Sickmann A, Cesaro L,
Brunati AM, Toninello A and Salvi M: Identification of new tyrosine
phosphorylated proteins in rat brain mitochondria. FEBS Lett.
582:1104–1110. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Pantic B, Trevisan E, Citta A, Rigobello
MP, Marin O, Bernardi P, Salvatori S and Rasola A: Myotonic
dystrophy protein kinase (DMPK) prevents ROS-induced cell death by
assembling a hexokinase II-Src complex on the mitochondrial
surface. Cell Death Dis. 4:e8582013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Battaglia V, Tibaldi E, Grancara S, Zonta
F, Brunati AM, Martinis P, Bragadin M, Grillo MA, Tempera G,
Agostinelli E and Toninello A: Effect of peroxides on spermine
transport in rat brain and liver mitochondria. Amino Acids.
42:741–749. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Battaglia V, Grancara S, Satriano J,
Saccoccio S, Agostinelli E and Toninello A: Agmatine prevents the
Ca(2+)-dependent induction of permeability transition in rat brain
mitochondria. Amino Acids. 38:431–437. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sullivan PG, Dubé C, Dorenbos K, Steward O
and Baram TZ: Mitochondrial uncoupling protein-2 protects the
immature brain from excitotoxic neuronal death. Ann Neurol.
53:711–717. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kristal BS, Park BK and Yu BP:
4-Hydroxyhexenal is a potent inducer of the mitochondrial
permeability transition. J Biol Chem. 271:6033–6038. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Salvi M, Battaglia V, Mancon M, Colombatto
S, Cravanzola C, Calheiros R, Marques MP, Grillo MA and Toninello
A: Agmatine is transported into liver mitochondria by a specific
electrophoretic mechanism. Biochem J. 396:337–345. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Grancara S, Battaglia V, Martinis P,
Viceconte N, Agostinelli E, Toninello A and Deana R: Mitochondrial
oxidative stress induced by Ca2+ and monoamines:
Different behaviour of liver and brain mitochondria in undergoing
permeability transition. Amino Acids. 42:751–759. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bonaiuto E, Grancara S, Martinis P,
Stringaro A, Colone M, Agostinelli E, Macone A, Stevanato R,
Vianello F, Toninello A and Di Paolo ML: A novel enzyme with
spermine oxidase properties in bovine liver mitochondria:
Identification and kinetic characterization. Free Radic Biol Med.
81:88–99. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Salvi M and Toninello A: Effects of
polyamines on mitochondrial Ca(2+) transport. Biochim Biophys Acta.
1661:113–124. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lapidus RG and Sokolove PM: Spermine
inhibition of the permeability transition of isolated rat liver
mitochondria: An investigation of mechanism. Arch Biochem Biophys.
306:246–253. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lapidus RG and Sokolove PM: The
mitochondrial permeability transition. Interactions of spermine,
ADP and inorganic phosphate. J Biol Chem. 269:18931–18936.
1994.PubMed/NCBI
|
|
29
|
Zhao YJ, Xu CQ, Zhang WH, Zhang L, Bian
SL, Huang Q, Sun HL, Li QF, Zhang YQ, Tian Y, et al: Role of
polyamines in myocardial ischemia/reperfusion injury and their
interactions with nitric oxide. Eur J Pharmacol. 562:236–246. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Choi YH and Park HY: Anti-inflammatory
effects of spermidine in lipopolysaccharide-stimulated BV2
microglial cells. J Biomed Sci. 19:312012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wei C, Li HZ, Wang YH, Peng X, Shao HJ, Li
HX, Bai SZ, Lu XX, Wu LY, Wang R and Xu CQ: Exogenous spermine
inhibits the proliferation of human pulmonary artery smooth muscle
cells caused by chemically-induced hypoxia via the suppression of
the ERK1/2- and PI3K/AKT-associated pathways. Int J Mol Med.
37:39–46. 2016.PubMed/NCBI
|
|
32
|
Dalla Via L, Mammi S, Uriarte E, Santana
L, Lampronti I, Gambari R and Gia O: New furan side tetracyclic
allopsoralen derivatives: synthesis and photobiological evaluation.
J Med Chem. 49:4317–4326. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Grancara S, Zonta F, Ohkubo S, Brunati AM,
Agostinelli E and Toninello A: Pathophysiological implications of
mitochondrial oxidative stress mediated by mitochondriotropic
agents and polyamines: The role of tyrosine phosphorylation. Amino
Acids. 47:869–883. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Tahin QS, Blum M and Carafoli E: The fatty
acid composition of subcellular membranes of rat liver, heart, and
brain: Diet-induced modifications. Eur J Biochem. 121:5–13. 1981.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Augereau O, Claverol S, Boudes N, Basurko
MJ, Bonneu M, Rossignol R, Mazat JP, Gineste C, Hernandez A,
Ivarsson N, Cheng AJ, Naess K, Wibom R, Lesko N, Bruhn H, Wedell A,
Freyer C, et al: Cyclophilin D, a target for counteracting skeletal
muscle dysfunction in mitochondrial myopathy. Hum Mol Genet.
24:6580–6587. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Crompton M: On the involvement of
mitochondrial intermembrane junctional complexes in apoptosis. Curr
Med Chem. 10:1473–1484. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Distler AM, Kerner J and Hoppel CL:
Post-translational modifications of rat liver mitochondrial outer
membrane proteins identified by mass spectrometry. Biochim Biophys
Acta. 1774:628–636. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Gineste C, Hernandez A, Ivarsson N, et al:
Cyclophilin D, a target for counteracting skeletal muscle
dysfunction in mitochondrial myopathy. Hum Mol Genet. 24:6580–6587.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Roskoski R Jr: Src kinase regulation by
phosphorylation and dephosphorylation. Biochem Biophys Res Commun.
331:1–14. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Salvi M, Brunati AM and Toninello A:
Tyrosine phosphorylation in mitochondria: a new frontier in
mitochondrial signaling. Free Radic Biol Med. 38:1267–1277. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Hebert-Chatelain E: Src kinases are
important regulators of mitochondrial functions. Int J Biochem Cell
Biol. 45:90–98. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Arachiche A, Augereau O, Decossas M, et
al: Localization of PTP-1B, SHP-2, and Src exclusively in rat brain
mitochondria and functional consequences. J Biol Chem.
283:24406–24411. 2008. View Article : Google Scholar : PubMed/NCBI
|