Introduction
Colorectal cancer is one of the most prevalent
malignant diseases, and is the fourth highest cancer-associated
mortality worldwide (1). Early
diagnosis has improved the clinical outcome of patients with
colorectal cancer, and conventional therapeutic strategies serve a
crucial role for advanced colorectal cancer, including surgery, and
chemoradiotherapy (2). Due to the
poor therapeutic efficiency of traditional cytotoxic drugs, the
prognosis of patients with advanced cancer remains poor (3). Therefore, novel targeted drugs and
more efficient therapeutic strategies are urgently needed to
improve the clinical outcome for patients with advanced colorectal
cancer (4).
Important mechanisms in response to DNA damage are
the cell cycle checkpoints that can prevent cells entering into
mitosis by arresting the cell in the G1 or G2 phase, which allows
enough time for DNA repair to maintain genomic integrity (5). The lack of a functional G1 arrest is
common in cancer cells due to dysfunction of p53, so cancer cells
are largely dependent on the G2 checkpoint to repair endogenous and
exogenous DNA damage; therefore, targeting the G2 checkpoint is a
promising strategy for cancer therapy (6,7),
especially in p53 mutant cancer cells. Wee1 is a nuclear kinase,
which can regulate the G2 checkpoint by activating phosphorylation
of cyclin dependent kinase 1 (CDK1) at the Tyr15 residue and
therefore delaying the entry into mitosis (8,9).
Wee1 overexpression occurs in a number of cancer types, including
ovarian cancer (10), melanoma
(11) and lung cancer (12), and indicates a poor outcome. In
light of this promising hypothesis, inhibitors targeting Wee1 have
been designed, and a number of clinical trials are underway. Of
these inhibitors, MK1775 is one of the most promising candidates,
which can potently and selectively inhibit Wee1 kinase in an
adenosine triphosphate-competitive manner, and a number of studies
have demonstrated that MK1775 can sensitize cells to the effect of
various chemotherapy drugs in solid tumor, including cisplatin,
5-fluorouracil (13–15).
In the field of colorectal cancer, the value of
targeting Wee1 is disputed. On one hand, certain studies have
reported that Wee1 has limited therapeutic value due to its poor
correlation with clinical biological characteristics and prognosis
(16); however, the Wee1 inhibitor
MK1775 could sensitize p53-deficient colonic cancer cell lines to
the anticancer effect of a number of DNA damaging drugs (17). However, other studies have argued
that this chemosensitization effect was not dependent on p53 status
(18). Due to this controversy,
further evaluation of the potential therapeutic value of Wee1 in
colonic cancer is needed. In the present study, the effect of Wee1
on cell proliferation and the response to ionizing radiation (IR)
treatment in HT29 and SW480 cells was investigated. The anticancer
effect of the Wee1 inhibitor MK11775 was analyzed, and whether
MK1775 can enhance the efficiency of DNA damage associated drug
irinotecan, which is one of the most important chemotherapy
reagents used in the clinical treatment of patients with colorectal
cancer with metastasis, was further examined.
Materials and methods
Cell culture
Two human colon cancer cells lines, HT29 and SW480
were purchased from the American Type Culture Collection (Manassas,
VA, USA) and cultured with RMPI-1640 (Gibco; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) with 10% fetal bovine serum
(Gibco; Thermo Fisher Scientific, Inc.) supplemented with 1%
penicillin and streptomycin (Gibco; Thermo Fisher Scientific,
Inc.). Cells were conventionally maintained in an incubator at 37°C
with 5% carbon dioxide and the culture medium was routinely changed
every three days.
Small interfering (si)RNA construction
and transfection
Wee1 and non-target control siRNA were purchased
from Sigma-Aldrich (Merck KGaA, Darmstadt, Germany), the Wee1 siRNA
sequence: 5′-AAUGAUUCCUGUGGUGAAGAC-3′. Non-target control siRNA
sequence: 5′-UAAGGCUAUGAAGAGAUAC-3′, the final concentration of the
siRNA for the experiments was 20 nM. Wee1 and non-target siRNA were
transient transfected into cells using Oligofectamine (Invitrogen;
Thermo Fisher Scientific, Inc.) for 24 or 48 h according to the
protocol from the manufacturer.
MTT assays
Cells were seeded into 96-wells cell culture plates
(3,000 cells/well) overnight, then analyzed under different
experimental conditions. A total of 20 µl MTT (Invitrogen; Thermo
Fisher Scientific, Inc.; 2 mg/ml) was added to each well, and cells
were and incubated for 4 h. Dimethyl sulfoxide (DMSO) was added to
dissolve the crystals, cells were agitated gently for another 5
min, and then the absorbance of each well was detected at 570 nM.
The MTT assay was used to analyze the effect of Wee1 on cell
proliferation, the response to treatment with IR and to examine the
anticancer effect of MK1775, irinotecan and combination therapy in
HT29 and SW480 cells.
Radiosensitivity assays
Cell radiosensitivity was detected using an MTT
assay. HT29 and SW480 cells were transfected with Wee1 siRNA for 48
h and the control groups were transfected with siRNA. Cells were
then seeded into a 96-well plate (500 cells/well). Cells were
cultured for another 24 h, then IR was administered (dosage, 4 Gy)
using an X-ray irradiator (RAD Source, LLC., Brentwood, TN, USA).
Cells were cultured for 4 days, then the cell viability in the
control and Wee1 knockdown groups with or without IR treatment were
detected by MTT assay.
Apoptosis assays
HT29 and SW480 cells were seeded into 6-well plates
(3×105 cells/well), to analyze the anticancer effect of
MK1775. Cells were treated with 1 µM MK1775 for 24 h, to
investigate whether MK1775 can sensitize cells to the effect of
irinotecan. Cells were divided into four groups: Control (DMSO),
MK1775 monotherapy, irinotecan monotherapy, and combination.
Following treatment for 24 h, cell were harvested, washed with cold
PBS twice, and Annexin-V (Invitrogen; Thermo Fisher Scientific,
Inc.) and propidium iodide (Sigma-Aldrich; Merck KGaA) were added.
Cells were then incubated for 10 min at room temperature in the
dark, and the apoptosis rate in each group was detected using flow
cytometry (Beckman Coulter, Inc., Brea, CA, USA and analysed by
Kaluza® software version 1.2 (Beckman Coulter,
Inc.).
Western blot analysis
Cells were harvested and lysed in a
radioimmunoprecipitation assay lysis buffer with protease and
phosphorylation inhibitor cocktails (GenDepot, Inc., Barker, TX,
USA). The protein concentration for each group was determined by
Bradford protein assay. A total of 30 µg protein for each sample
was loaded onto a gradient SDS-PAGE gel. Following gel
electrophoresis, the proteins were transferred to polyvinyl
difluoride membranes, then blocked with 5% skimmed milk
(Sigma-Aldrich; Merck KGaA) at room temperature for 1 h.
Subsequently, the membranes were washed with PBS with 0.1% Tween-20
(PBST) three times, then incubated overnight at 4°C with primary
antibodies: Anti-Wee1 (1:1,000; cat no. 4936), anti-phospho-CDK1
(Thr 14/Tyr15 (1:1,000; cat no. 4539), anti-cleaved-caspase 3
(1:1,000; cat no. 9661) and anti-r-histone H2AX (H2AX; 1:1,000; cat
no. 7631) were purchased from Cell Signaling Technology, Inc.,
(Danvers, MA, USA); anti-total CDK1 (1:500; cat no. sc-53219) was
purchased from Santa Cruz Biotechnology, Inc., (Dallas, TX, USA)
and anti-β-actin (1:100,000; cat no. A1978) was purchased from
Sigma-Aldrich (Merck KGaA). β-actin was used as a loading control.
Anti mouse or rabbit-horse radish peroxidase conjugated secondary
antibodies (cat nos. 32260 and 32230; Invitrogen; Thermo Fisher
Scientific, Inc.; 1:2,500 in PBST) were incubated with the
membranes at room temperature for 1 h, following washing with PBST
three times. The expression of target proteins was detected using
enhanced chemiluminescence solution (Bio-Rad Laboratories, Inc.,
Hercules, CA, USA).
Statistical analysis
All data were presented as the mean ± standard
deviation and each experiment was performed three times. The paired
Student's t-test was used to calculate the statistical value for
the difference between two groups, and multigroup comparisons were
performed using one way analysis of variance followed by the
Student-Newman-Keuls post hoc test using GraphPad Prism version 6.0
software (GraphPad Software, Inc., La Jolla, CA, USA). P<0.05
was considered to indicate a statistically significant
difference.
Results
Wee1 ablation could impair the
proliferative ability of the cell and sensitize p53 mutant colonic
cancer cell lines, HT29 and SW480 to the effect of IR
treatment
To examine the role of Wee1 in colonic cancer cells,
HT29 and SW480 cells were transfected with Wee1 siRNA for 48 h
(Fig. 1). Wee1 knockdown
efficiency was examined by western blotting, as exhibited in
Fig. 1D. Wee1 expression was
decreased in HT29 and SW480 cells, and it was also demonstrated
that phosphorylation at the Tyr15 residue of CDK1 was inhibited. An
MTT assay was used to examine cell proliferation and the
sensitivity of cells to treatment with IR; Wee1 knockdown was
demonstrated to significantly suppress the proliferation of HT29
(P<0.05; Fig. 1A) and SW480
(P<005; Fig. 1B) cells. The
effect of Wee1 silencing by siRNA on cell viability during
treatment with IR was investigated, and the sensitivity of cells to
IR treatment was increased compared with the untreated cells
(Fig. 1C).
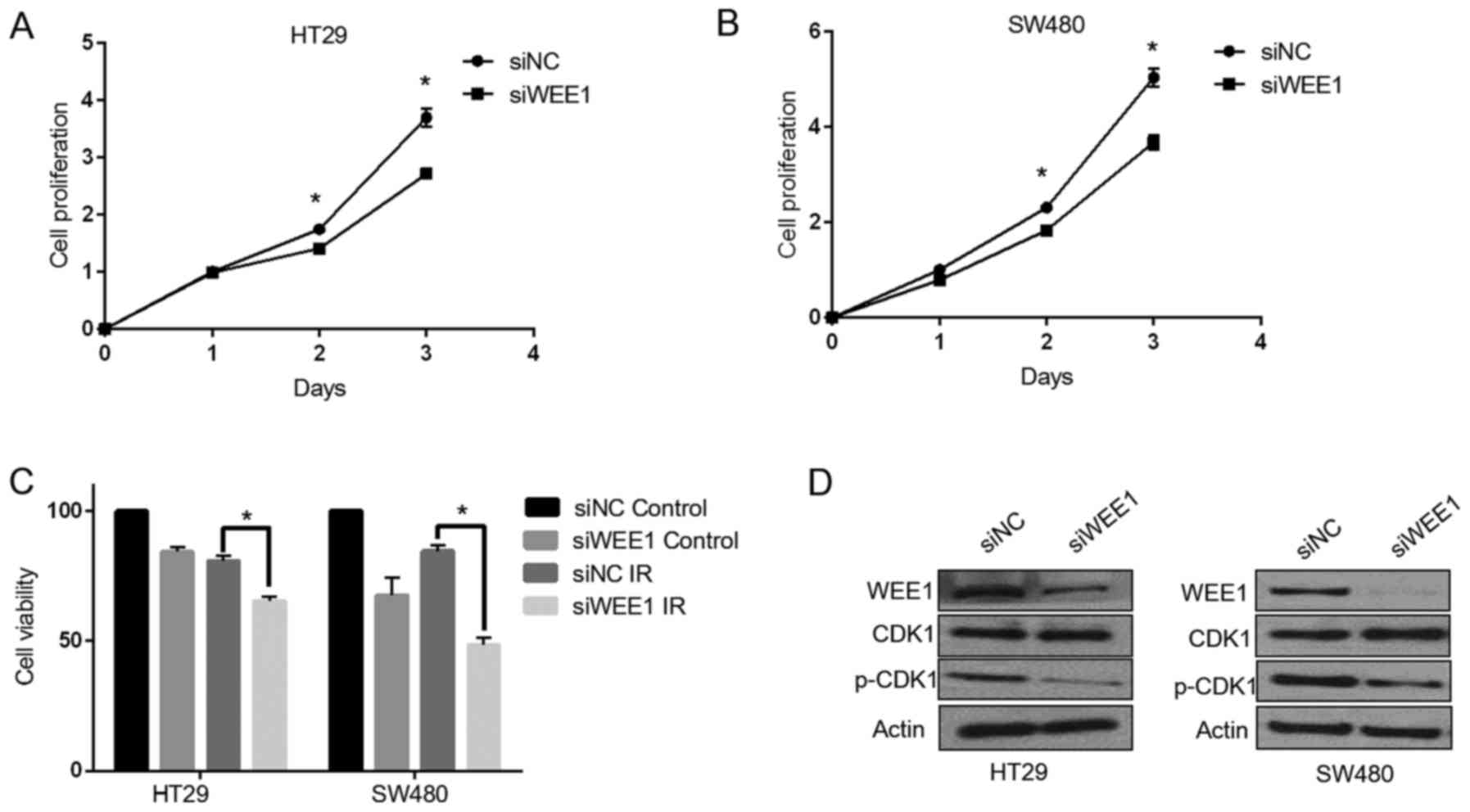 | Figure 1.Wee1 knockdown by siRNA reduces cell
proliferation and sensitizes colonic cancer cells to treatment with
IR. HT29 and SW480 cells were transfected with siNC and Wee1 siRNA.
Cell proliferation on days 1, 2 and 3 following transfection were
measured by MTT assay in (A) HT29 and (B) SW480 cells. (C) HT29 and
SW480 cells were exposed to IR treatment with a dosage of 4 Gy,
then cultured for another 4 days, and cell viability were detected
by MTT assays. (D) In HT29 and SW480 cells, the expression of Wee1,
total CDK1, p-CDK1 (Ty15) in the control and Wee1 knockdown groups
were detected by western blotting. Data are presented as the mean ±
SD. *P<0.05 vs. siNC. IR, ionizing radiation; si, small
interfering; CDK1, cyclin dependent kinase; NC, non-target control;
p, phosphorylated. |
Wee1 inhibitor MK1775 can
significantly induce DNA damage, leading to apoptosis and decreased
cell viability in HT29 and SW480 cells
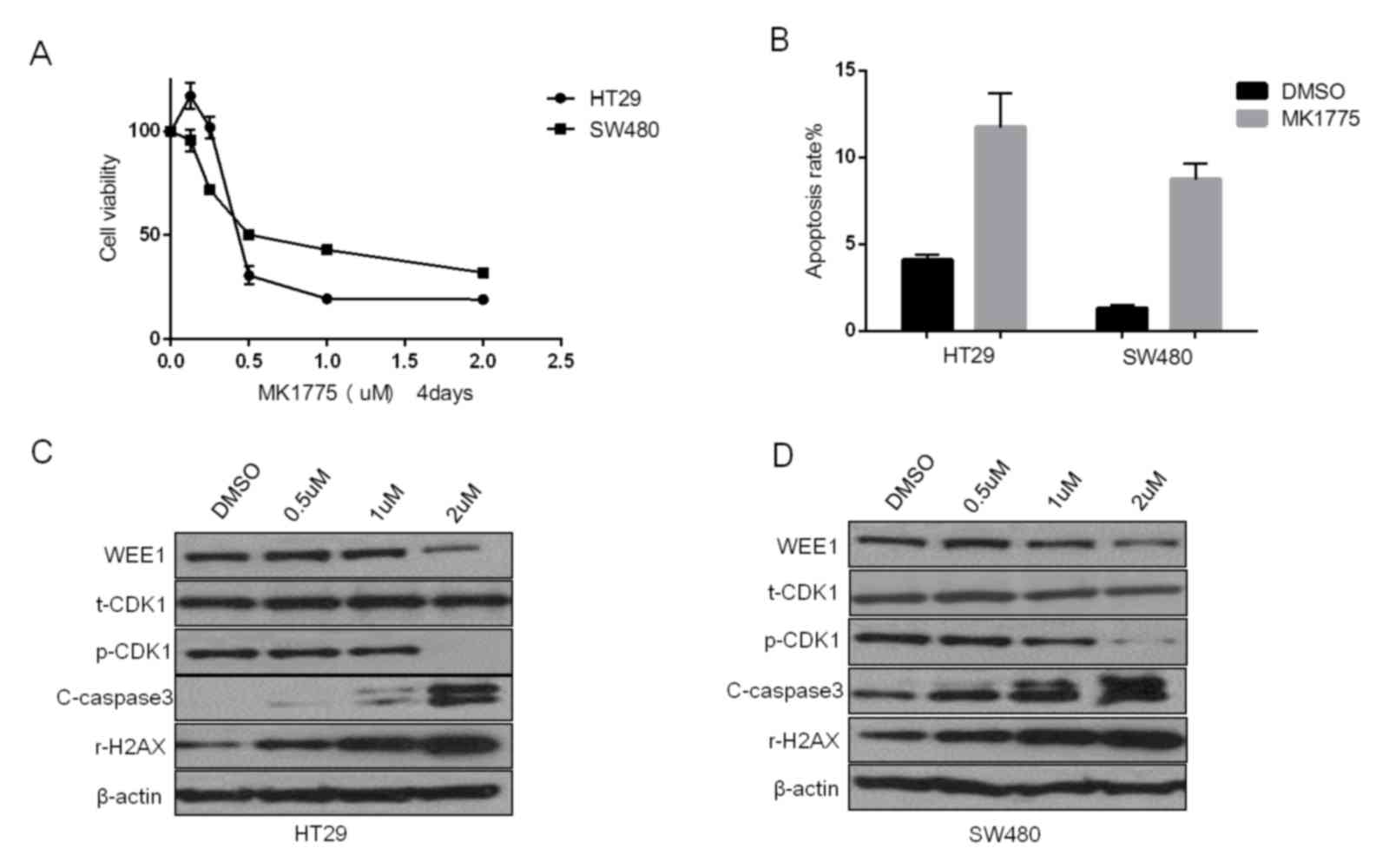
Nearly 40 clinical trials of the Wee1 inhibitor
MK1775 are now underway (19), but
its anticancer effect and the potential molecular alterations are
not well studied in colorectal cancer cells. From the results of
the MTT assay exhibited in Fig.
2A, MK1775 can decrease cell viability in HT29 and SW480 cells.
The IC50 following treatment for 4 days was 0.376±0.134
µM in HT29 and 0.739±0.214 µM in SW480 cells. The apoptosis assays
also demonstrated that MK1775 could induce cell apoptosis in these
two colonic cancer cell lines (Fig.
2B). Western blotting was used to detect the molecular
alterations induced by MK1775 in HT29 and SW480 cells (Fig. 2C and D, respectively). MK1775
decreased the level of Wee1 and the phosphorylation of CDK1, and it
also was observed that the DNA damage marker γ-H2AX and apoptosis
marker cleaved-caspase 3 increased in a dose-dependent manner
(Fig. 2C and D).
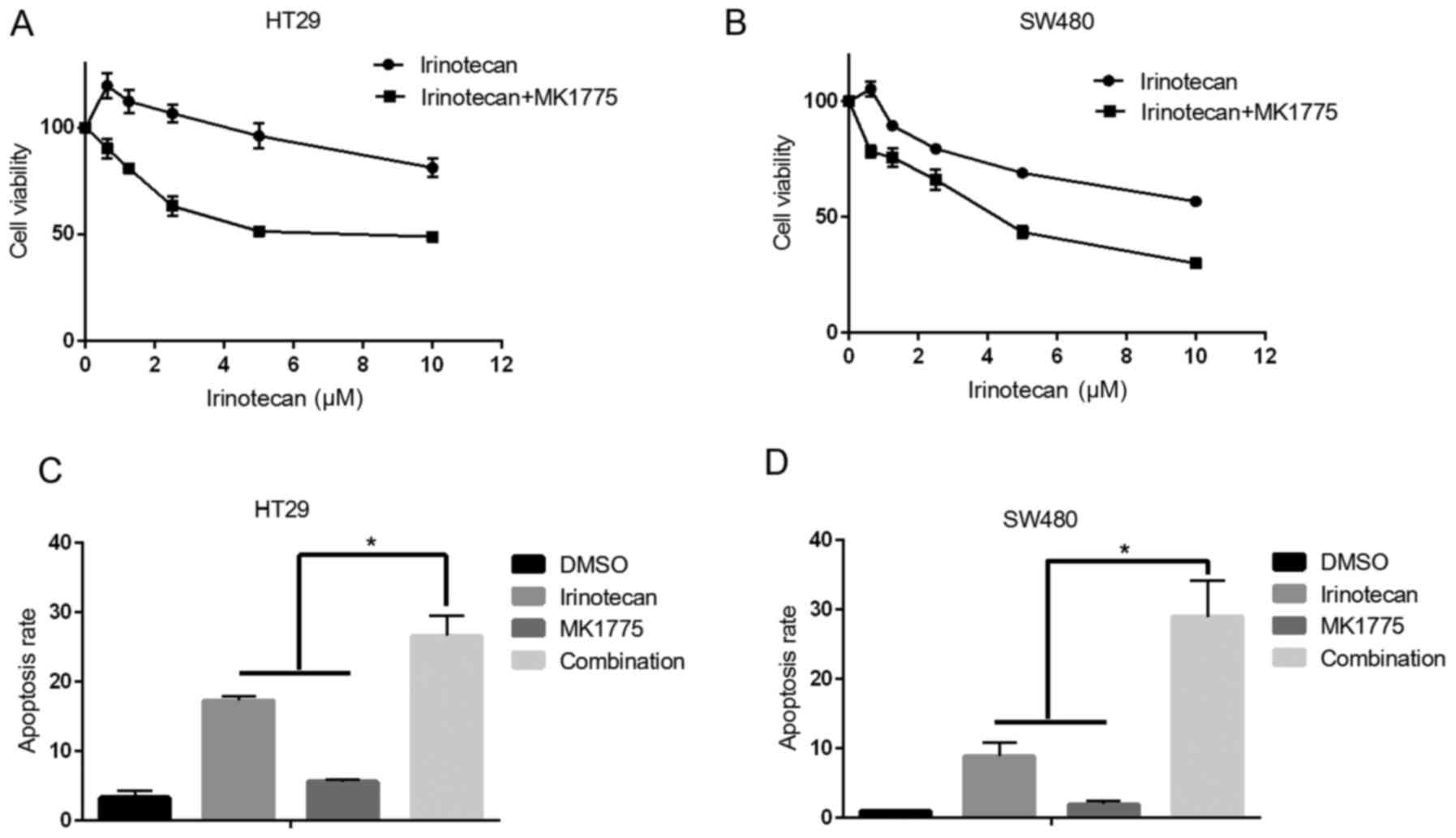
Wee1 inhibitor MK1775 could sensitize
p53 mutant colonic cancer cells to the effect of irinotecan
Although MK1775 demonstrated a potent anticancer
effect in colonic cancer cells, it remains a better choice to use
it in combination with other DNA damaging drugs for cancer
treatment, because combination strategy can decrease the effective
dosage for each drug, and it also can delay the development of drug
resistance. Irinotecan is one of most important chemotherapy drugs,
which is widely used for advanced colorectal cancer treatment
(20), especially for fluorouracil
resistant patients. In the present study, whether MK1775 could
enhance the therapeutic efficiency of irinotecan in HT29 and SW480
cells was investigated. The results revealed that irinotecan
combined with MK1775 decreased cell viability compared with
irinotecan treatment alone (Fig. 3A
and B). Additionally, combined treatment induced increased
levels of apoptosis, compared with treatment with one drug alone
(Fig. 3C and D).
Wee1 inhibitor MK1775 combined with
irinotecan could induce an increased level of DNA damage associated
protein γ-H2AX and apoptosis marker cleaved-caspase 3 in HT29 and
SW480 cells
Along with investigating the potency of MK1775 and
irinotecan combination in HT29 and SW480 cells, the molecular
mechanisms were also analyzed using western blotting. As presented
in Fig. 4A and B, irinotecan can
induce phosphorylation of CDK1, which may serve a role in aiding
cancer cells in escaping from the anticancer effect of irinotecan,
and MK1775 was also demonstrated to decreased the phosphorylation
of CDK1. And in the combination treatment groups, higher levels of
DNA damage associated protein γ-H2AX and apoptosis marker
cleaved-caspase 3 were observed in both HT29 and SW480 cells. The
results of the present study support the hypothesis that MK1775
could enhance the anticancer effect of irinotecan on p53 mutant
colonic cancer cells.
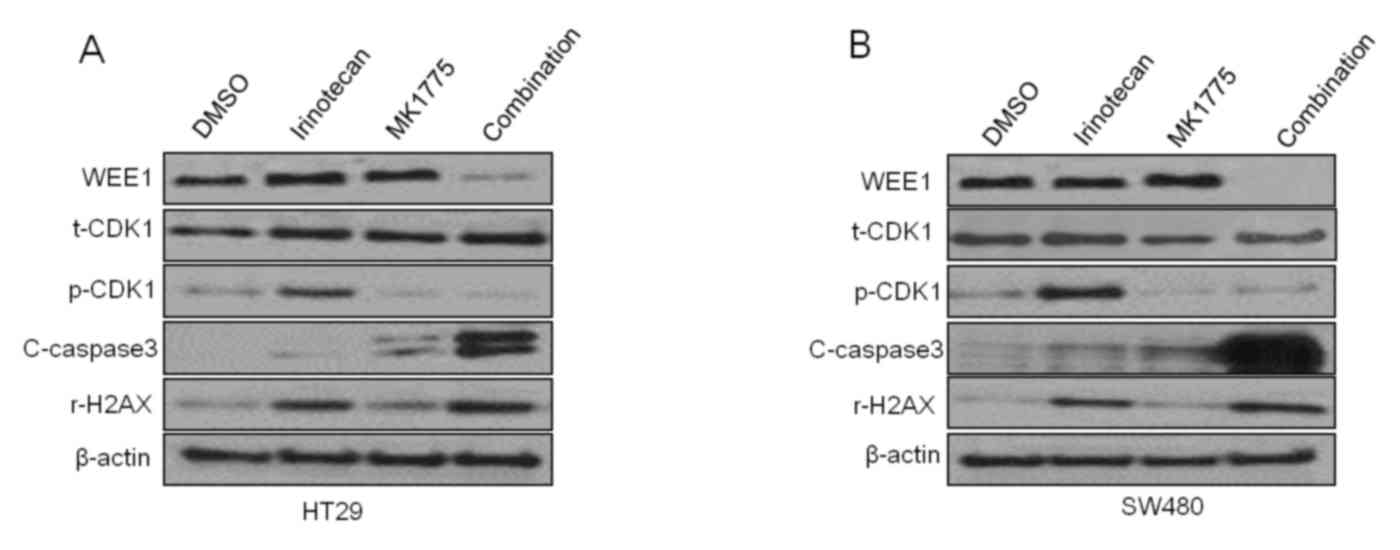 | Figure 4.Wee1 inhibitor MK1775 combined with
irinotecan can induce an increase in DNA damage associated protein
γ-H2AX and apoptosis marker c-caspase 3 in HT29 and SW480 cells.
Representative western blot images of protein expression levels of
Wee1, CDK1, p-CDK1, c-caspase 3, H2AX and the actin control to
treatment with irinotecan, MK1775, a combination of the two or DMSO
control in (A) HT29 and (B) SW480 cells. CDK1, cyclin dependent
kinase 1; p-, phosphorylated; H2AX, histone H2AX; c-, cleaved;
t-total; DMSO, dimethyl sulfoxide. |
Discussion
In normal cells, DNA damage usually is repaired by
arresting the cell at the G1 phase, but in tumor cells (20), due to the frequency of G1
checkpoint deficiencies, especially in p53 deregulated cells, the
G2 checkpoint serves a crucial role in DNA damage repair (21,22).
As a result of this, targeting the effectors, which are involved in
G2 checkpoints, is considered a potent strategy for cancer therapy.
Unlike other strategies, which aim to block cell cycle processes,
Wee1 inhibition can speed up cell cycle progression, promoting
increased entry into mitosis with unrepaired DNA damage, ultimately
leading to apoptosis and cell death. Therefore, it is reasonable
that Wee1 inhibition can potentiate the anticancer effect of a
number of conventional DNA damaging drugs. However, there remains
some controversy about the chemosensitization effect of Wee1
inhibition, with certain studies reporting that Wee1 inhibition can
only enhance the effect of DNA damage-associated agents and
radiotherapy in p53 deficient cancer cells (17,18),
but others argued that Wee1 inhibition demonstrated a
chemosensitization effect in both p53 wild and mutant cancer cells
(18,23).
The results of the present study demonstrated that
Wee1 knockdown can inhibit tumor cell proliferation in p53 mutant
colonic cancer cell lines HT29 and SW480. This may be due to the
attenuated capacity of the cell to repair endogenous DNA damage
from the loss of the G2 checkpoint by Wee1 inhibition, and Wee1
ablation may also increase the effect of treatment with IR. To
further investigate the potential clinical value of the results of
the present study, the anticancer effect of the Wee1 inhibitor
MK1775 in HT29 and SW480 cells was analyzed. Logically, Wee1
inhibition ought to have an improved anticancer effect in p53
mutant cells due to the lack of G1 arrest, and the data from the
present study demonstrated that MK1775 could significantly inhibit
cell proliferation and induce apoptosis in these two p53 mutant
colon cancer cell lines. The results of the present study were in
accordance with data published in previous studies in other cancer
types (13,24), and western blotting also
demonstrated that MK1775 could increase the DNA damage marker
γ-H2AX and apoptosis associated protein cleaved caspase 3 in
dose-dependent manner. Although it seems that MK1775 monotherapy
could achieve an anticancer effect in p53 mutant colonic cancer
cells, the dosage for treatment is still a challenge, as the
toxicity of agents targeting the cell cycle checkpoint is big
problem. Combination with other chemotherapy drugs is a better
choice as a first-line chemotherapy regime in metastatic colonic
cancer (25), and a number of
studies are seeking the proper strategy to increase the sensitivity
of cancer cells to the cytotoxicity effect (26–28).
In the present study MK1775 was demonstrated to increase the
sensitivity of cells to the effect of irinotecan both in HT29 and
SW480, and resulting in increased γ-H2AX and cleaved-caspase 3.
In conclusion, although negative results have been
reported regarding the association between Wee1 expression and
clinical outcome, Wee1 is still a valuable target in patients with
p53 mutant colorectal cancer. Wee1 inhibition can decrease cell
viability in colorectal cancer cell lines, furthermore, it was
demonstrated that the Wee1 inhibitor MK1775 is a potent anticancer
drugs and can induce DNA damage in colorectal cancer cells. In
addition, MK1775 has a chemosensitization effect on treatment with
irinotecan in colorectal cancer cells, which may help to increase
the efficiency of treatment with irinotecan, enabling improved
clinical outcomes in patients with advanced colorectal cancer.
Acknowledgements
The present study was supported by the National
Natural Science Foundation of China (grant no. 81572413), the
Natural Science Foundation of Hubei Province (grant no.
2015CFB378), the Research Fund of Public welfare in Health,
Industry Health and Family Planning Committee of China (grant no.
201402015).
References
|
1
|
Miller KD, Siegel RL, Lin CC, Mariotto AB,
Kramer JL, Rowland JH, Stein KD, Alteri R and Jemal A: Cancer
treatment and survivorship statistics, 2016. CA Cancer J Clin.
66:271–289. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zhou Y, Abel GA, Hamilton W,
Pritchard-Jones K, Gross CP, Walter FM, Renzi C, Johnson S, McPhail
S, Elliss-Brookes L and Lyratzopoulos G: Diagnosis of cancer as an
emergency: A critical review of current evidence. Nat Rev Clin
Oncol. 14:45–56. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kolligs FT: Diagnostics and epidemiology
of colorectal cancer. Visc Med. 32:158–164. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Moriarity A, O'Sullivan J, Kennedy J,
Mehigan B and McCormick P: Current targeted therapies in the
treatment of advanced colorectal cancer: A review. Ther Adv Med
Oncol. 8:276–293. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kastan MB and Bartek J: Cell-cycle
checkpoints and cancer. Nature. 432:316–323. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Santo L, Siu KT and Raje N: Targeting
cyclin-dependent kinases and cell cycle progression in human
cancers. Semin Oncol. 42:788–800. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Benada J and Macurek L: Targeting the
checkpoint to kill cancer cells. Biomolecules. 5:1912–1937. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Russell P and Nurse P: Negative regulation
of mitosis by Wee1+, a gene encoding a protein kinase homolog.
Cell. 49:559–567. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gould KL and Nurse P: Tyrosine
phosphorylation of the fission yeast cdc2+ protein kinase regulates
entry into mitosis. Nature. 342:39–45. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Leary A, Auguste A and Mesnage S: DNA
damage response as a therapeutic target in gynecological cancers.
Curr Opin Oncol. 28:404–411. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Magnussen GI, Holm R, Emilsen E, Rosnes
AK, Slipicevic A and Flørenes VA: High expression of Wee1 is
associated with poor disease-free survival in malignant melanoma:
Potential for targeted therapy. PLoS One. 7:e382542012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yoshida T, Tanaka S, Mogi A, Shitara Y and
Kuwano H: The clinical significance of Cyclin B1 and Wee1
expression in non-small-cell lung cancer. Ann Oncol. 15:252–256.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kim HY, Cho Y, Kang H, Yim YS, Kim SJ,
Song J and Chun KH: Targeting the WEE1 kinase as a molecular
targeted therapy for gastric cancer. Oncotarget. 7:49902–49916.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hirai H, Iwasawa Y, Okada M, Arai T,
Nishibata T, Kobayashi M, Kimura T, Kaneko N, Ohtani J, Yamanaka K,
et al: Small-molecule inhibition of Wee1 kinase by MK-1775
selectively sensitizes p53-deficient tumor cells to DNA-damaging
agents. Mol Cancer Ther. 8:2992–3000. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Osman AA, Monroe MM, Ortega Alves MV,
Patel AA, Katsonis P, Fitzgerald AL, Neskey DM, Frederick MJ, Woo
SH, Caulin C, et al: Wee-1 kinase inhibition overcomes cisplatin
resistance associated with high-risk TP53 mutations in head and
neck cancer through mitotic arrest followed by senescence. Mol
Cancer Ther. 14:608–619. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Egeland EV, Flatmark K, Nesland JM,
Flørenes VA, Mælandsmo GM and Boye K: Expression and clinical
significance of Wee1 in colorectal cancer. Tumour Biol.
37:12133–12140. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hirai H, Arai T, Okada M, Nishibata T,
Kobayashi M, Sakai N, Imagaki K, Ohtani J, Sakai T, Yoshizumi T, et
al: MK-1775, a small molecule Wee1 inhibitor, enhances anti-tumor
efficacy of various DNA-damaging agents, including 5-fluorouracil.
Cancer Biol Ther. 9:514–522. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cuneo KC, Morgan MA, Davis MA, Parcels LA,
Parcels J, Karnak D, Ryan C, Liu N, Maybaum J and Lawrence TS: Wee1
kinase inhibitor AZD1775 radiosensitizes hepatocellular carcinoma
regardless of TP53 mutational status through induction of
replication stress. Int J Radiat Oncol Biol Phys. 95:782–790. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Leijen S, van Geel RM, Sonke GS, de Jong
D, Rosenberg EH, Marchetti S, Pluim D, van Werkhoven E, Rose S, Lee
MA, et al: Phase II study of WEE1 inhibitor AZD1775 plus
carboplatin in patients with TP-53 mutated ovarian cancer
refractory or resistant to first-line therapy with 3 months. J Clin
Oncol. 34:4354–4361. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wulaningsih W, Wardhana A, Watkins J,
Yoshuantari N, Repana D and Van Hemelrijck M: Irinotecan
chemotherapy combined with fluoropyrimidines versus irinotecan
alone for overall survival and progression-free survival in
patients with advanced and/or metastatic colorectal cancer.
Cochrane Database Syst Rev. 2:CD0085932016.PubMed/NCBI
|
|
21
|
Beetham KL and Tolmach LJ: The action of
caffeine on X-irradiated hela cells. V. Identity of the sector of
cells that expresses potentially lethal damage in G1 and G2. Radiat
Res. 91:199–211. 1982. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dixon H and Norbury CJ: Therapeutic
exploitation of checkpoint defects in cancer cells lacking p53
function. Cell Cycle. 1:362–368. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Van Linden AA, Baturin D, Ford JB, Fosmire
SP, Gardner L, Korch C, Reigan P and Porter CC: Inhibition of wee1
sensitizes cancer cells to antimetabolite chemotherapeutics in
vitro and in vivo, independent of p53 functionality. Mol Cancer
Ther. 12:2675–2684. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Guertin AD, Li J, Liu Y, Hurd MS, Schuller
AG, Long B, Hirsch HA, Feldman I, Benita Y, Toniatti C, et al:
Preclinical evaluation of the WEE1 inhibitor MK-1775 as
single-agent anticancer therapy. Mol Cancer Ther. 12:1442–1452.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kreahling JM, Gemmer JY, Reed D, Letson D,
Bui M and Altiok S: MK1775, a selective Wee1 inhibitor, shows
single-agent antitumor activity against sarcoma cells. Mol Cancer
Ther. 11:174–182. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Fischer von Weikersthal L, Schalhorn A,
Stauch M, Quietzsch D, Maubach PA, Lambertz H, Oruzio D, Schlag R,
Weigang-Köhler K, Vehling-Kaiser U, et al: Phase III trial of
irinotecan plus infusional 5-fluorouracil/folinic acid versus
irinotecan plus oxaliplatin as first-line treatment of advanced
colorectal cancer. Eur J Cancer. 47:206–214. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Sun M, Zhang Q, Yang X, Qian SY and Guo B:
Vitamin D enhances the efficacy of irinotecan through
miR-627-mediated inhibition of intratumoral drug metabolism. Mol
Cancer Ther. 15:2086–2095. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Quackenbush KS, Bagby S, Tai WM,
Messersmith WA, Schreiber A, Greene J, Kim J, Wang G, Purkey A,
Pitts TM, et al: The novel tankyrase inhibitor (AZ1366) enhances
irinotecan activity in tumors that exhibit elevated tankyrase and
irinotecan resistance. Oncotarget. 7:28273–28285. 2016. View Article : Google Scholar : PubMed/NCBI
|