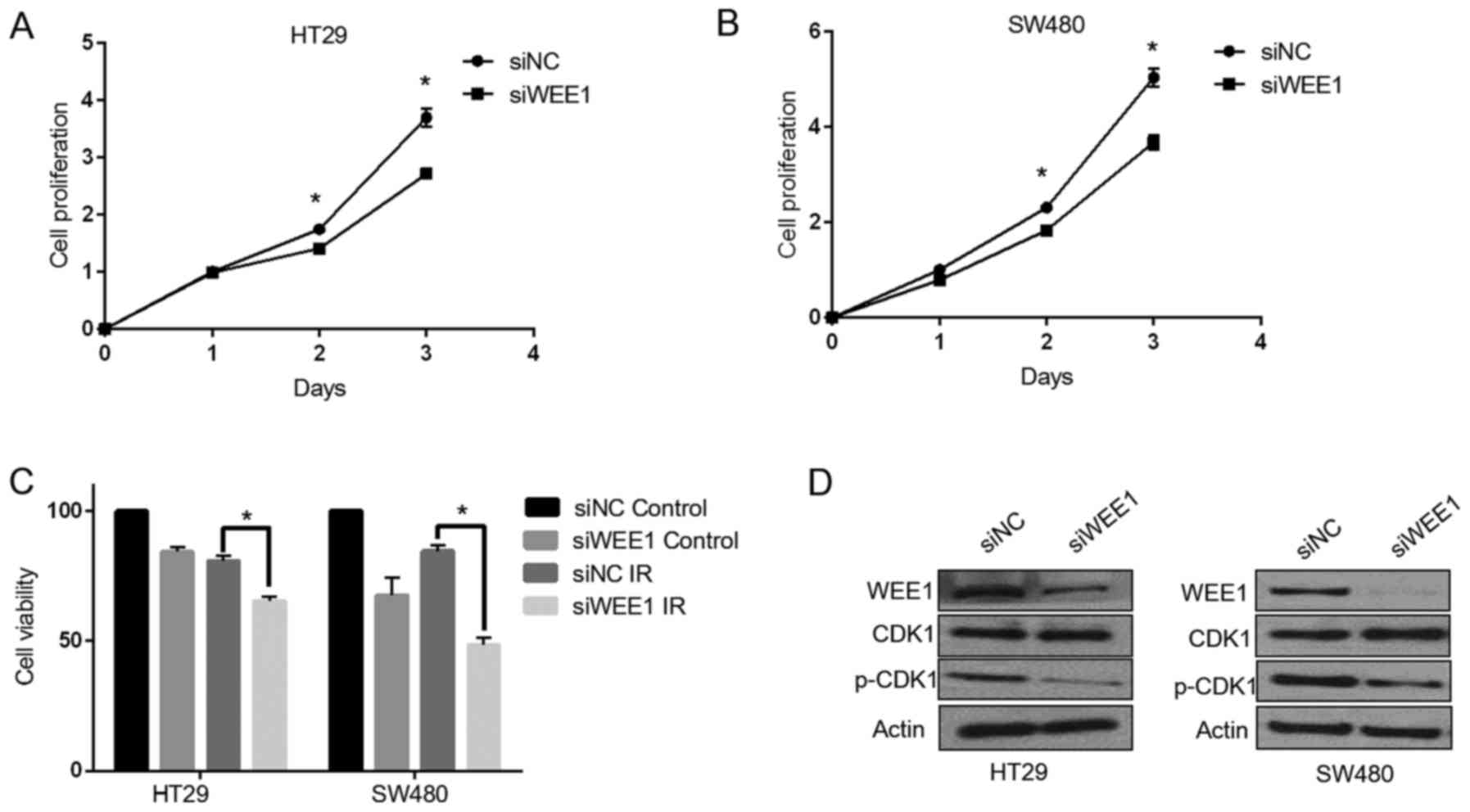
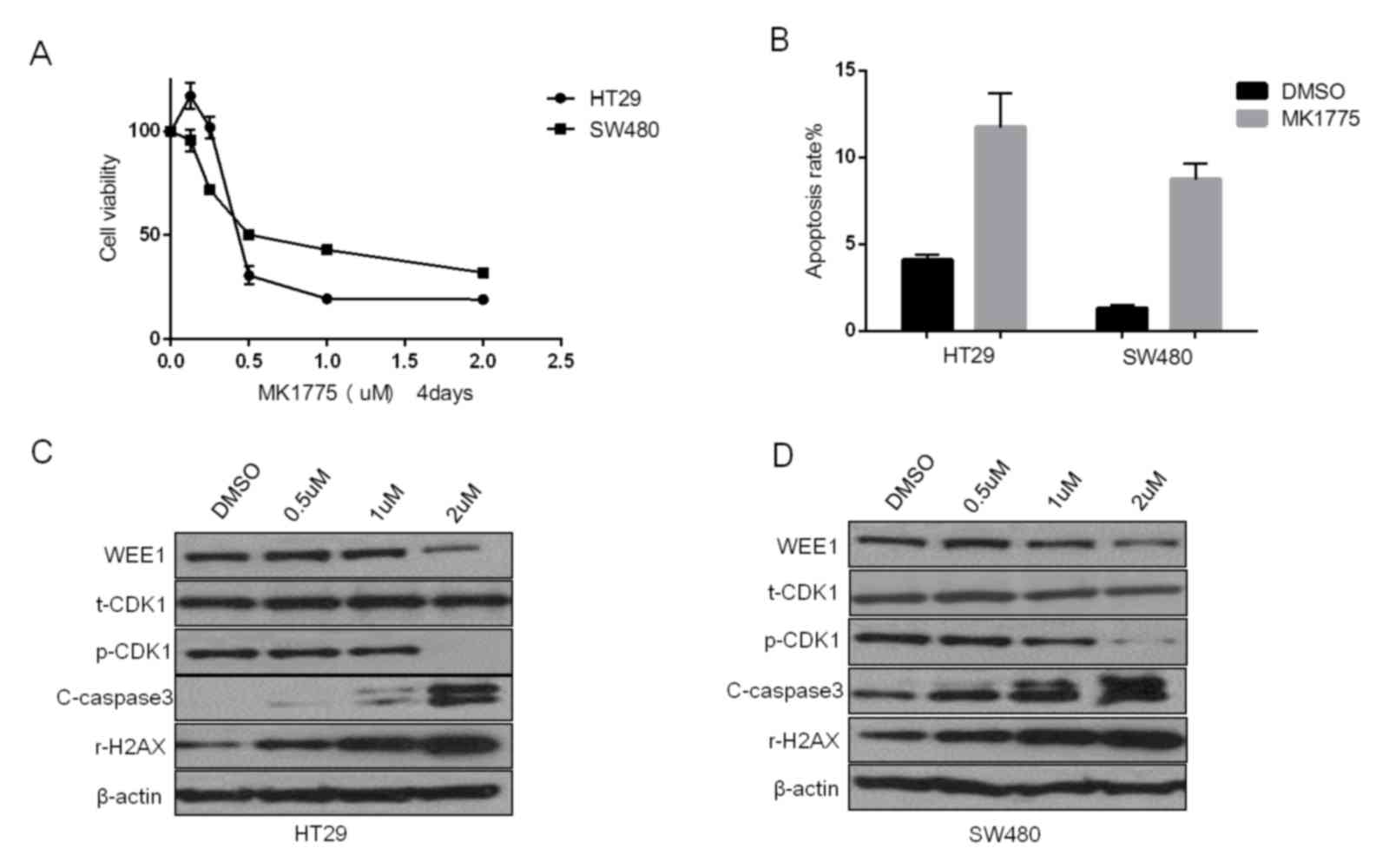
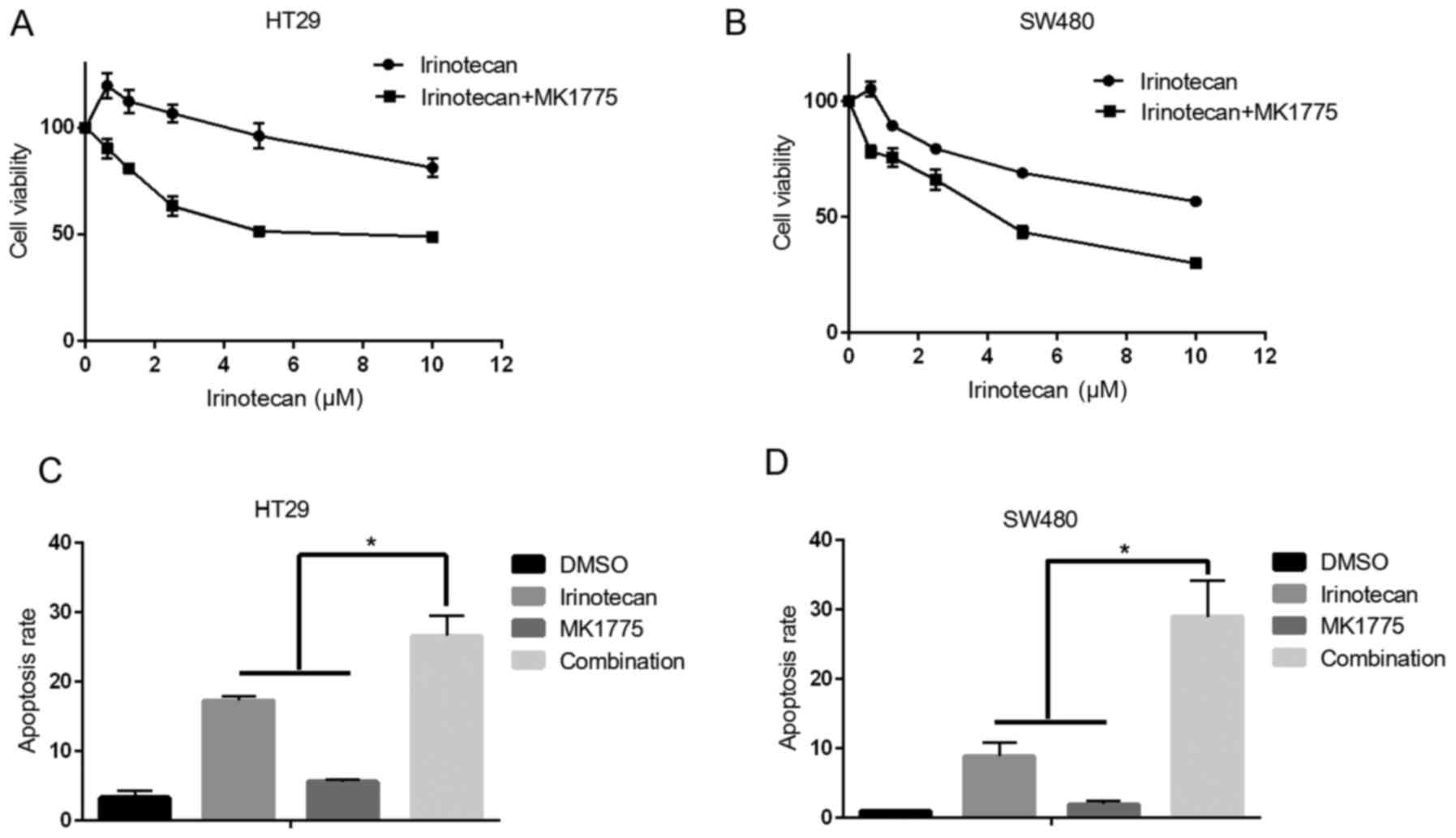
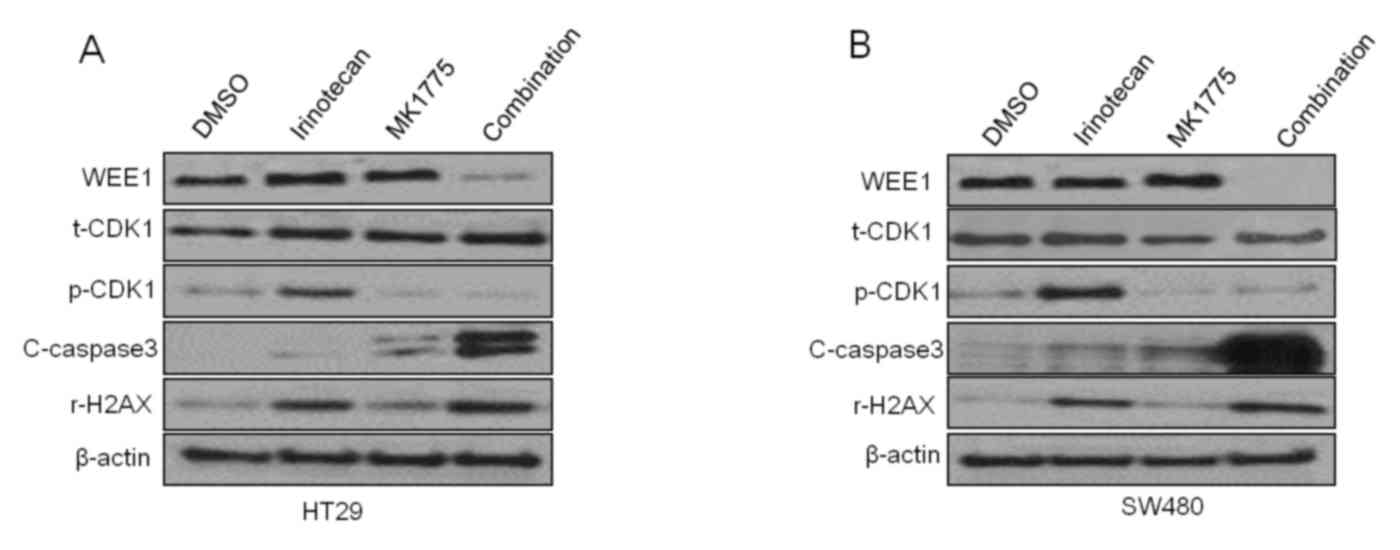
|
1
|
Miller KD, Siegel RL, Lin CC, Mariotto AB,
Kramer JL, Rowland JH, Stein KD, Alteri R and Jemal A: Cancer
treatment and survivorship statistics, 2016. CA Cancer J Clin.
66:271–289. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zhou Y, Abel GA, Hamilton W,
Pritchard-Jones K, Gross CP, Walter FM, Renzi C, Johnson S, McPhail
S, Elliss-Brookes L and Lyratzopoulos G: Diagnosis of cancer as an
emergency: A critical review of current evidence. Nat Rev Clin
Oncol. 14:45–56. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kolligs FT: Diagnostics and epidemiology
of colorectal cancer. Visc Med. 32:158–164. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Moriarity A, O'Sullivan J, Kennedy J,
Mehigan B and McCormick P: Current targeted therapies in the
treatment of advanced colorectal cancer: A review. Ther Adv Med
Oncol. 8:276–293. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kastan MB and Bartek J: Cell-cycle
checkpoints and cancer. Nature. 432:316–323. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Santo L, Siu KT and Raje N: Targeting
cyclin-dependent kinases and cell cycle progression in human
cancers. Semin Oncol. 42:788–800. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Benada J and Macurek L: Targeting the
checkpoint to kill cancer cells. Biomolecules. 5:1912–1937. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Russell P and Nurse P: Negative regulation
of mitosis by Wee1+, a gene encoding a protein kinase homolog.
Cell. 49:559–567. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gould KL and Nurse P: Tyrosine
phosphorylation of the fission yeast cdc2+ protein kinase regulates
entry into mitosis. Nature. 342:39–45. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Leary A, Auguste A and Mesnage S: DNA
damage response as a therapeutic target in gynecological cancers.
Curr Opin Oncol. 28:404–411. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Magnussen GI, Holm R, Emilsen E, Rosnes
AK, Slipicevic A and Flørenes VA: High expression of Wee1 is
associated with poor disease-free survival in malignant melanoma:
Potential for targeted therapy. PLoS One. 7:e382542012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yoshida T, Tanaka S, Mogi A, Shitara Y and
Kuwano H: The clinical significance of Cyclin B1 and Wee1
expression in non-small-cell lung cancer. Ann Oncol. 15:252–256.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kim HY, Cho Y, Kang H, Yim YS, Kim SJ,
Song J and Chun KH: Targeting the WEE1 kinase as a molecular
targeted therapy for gastric cancer. Oncotarget. 7:49902–49916.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hirai H, Iwasawa Y, Okada M, Arai T,
Nishibata T, Kobayashi M, Kimura T, Kaneko N, Ohtani J, Yamanaka K,
et al: Small-molecule inhibition of Wee1 kinase by MK-1775
selectively sensitizes p53-deficient tumor cells to DNA-damaging
agents. Mol Cancer Ther. 8:2992–3000. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Osman AA, Monroe MM, Ortega Alves MV,
Patel AA, Katsonis P, Fitzgerald AL, Neskey DM, Frederick MJ, Woo
SH, Caulin C, et al: Wee-1 kinase inhibition overcomes cisplatin
resistance associated with high-risk TP53 mutations in head and
neck cancer through mitotic arrest followed by senescence. Mol
Cancer Ther. 14:608–619. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Egeland EV, Flatmark K, Nesland JM,
Flørenes VA, Mælandsmo GM and Boye K: Expression and clinical
significance of Wee1 in colorectal cancer. Tumour Biol.
37:12133–12140. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hirai H, Arai T, Okada M, Nishibata T,
Kobayashi M, Sakai N, Imagaki K, Ohtani J, Sakai T, Yoshizumi T, et
al: MK-1775, a small molecule Wee1 inhibitor, enhances anti-tumor
efficacy of various DNA-damaging agents, including 5-fluorouracil.
Cancer Biol Ther. 9:514–522. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cuneo KC, Morgan MA, Davis MA, Parcels LA,
Parcels J, Karnak D, Ryan C, Liu N, Maybaum J and Lawrence TS: Wee1
kinase inhibitor AZD1775 radiosensitizes hepatocellular carcinoma
regardless of TP53 mutational status through induction of
replication stress. Int J Radiat Oncol Biol Phys. 95:782–790. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Leijen S, van Geel RM, Sonke GS, de Jong
D, Rosenberg EH, Marchetti S, Pluim D, van Werkhoven E, Rose S, Lee
MA, et al: Phase II study of WEE1 inhibitor AZD1775 plus
carboplatin in patients with TP-53 mutated ovarian cancer
refractory or resistant to first-line therapy with 3 months. J Clin
Oncol. 34:4354–4361. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wulaningsih W, Wardhana A, Watkins J,
Yoshuantari N, Repana D and Van Hemelrijck M: Irinotecan
chemotherapy combined with fluoropyrimidines versus irinotecan
alone for overall survival and progression-free survival in
patients with advanced and/or metastatic colorectal cancer.
Cochrane Database Syst Rev. 2:CD0085932016.PubMed/NCBI
|
|
21
|
Beetham KL and Tolmach LJ: The action of
caffeine on X-irradiated hela cells. V. Identity of the sector of
cells that expresses potentially lethal damage in G1 and G2. Radiat
Res. 91:199–211. 1982. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dixon H and Norbury CJ: Therapeutic
exploitation of checkpoint defects in cancer cells lacking p53
function. Cell Cycle. 1:362–368. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Van Linden AA, Baturin D, Ford JB, Fosmire
SP, Gardner L, Korch C, Reigan P and Porter CC: Inhibition of wee1
sensitizes cancer cells to antimetabolite chemotherapeutics in
vitro and in vivo, independent of p53 functionality. Mol Cancer
Ther. 12:2675–2684. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Guertin AD, Li J, Liu Y, Hurd MS, Schuller
AG, Long B, Hirsch HA, Feldman I, Benita Y, Toniatti C, et al:
Preclinical evaluation of the WEE1 inhibitor MK-1775 as
single-agent anticancer therapy. Mol Cancer Ther. 12:1442–1452.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kreahling JM, Gemmer JY, Reed D, Letson D,
Bui M and Altiok S: MK1775, a selective Wee1 inhibitor, shows
single-agent antitumor activity against sarcoma cells. Mol Cancer
Ther. 11:174–182. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Fischer von Weikersthal L, Schalhorn A,
Stauch M, Quietzsch D, Maubach PA, Lambertz H, Oruzio D, Schlag R,
Weigang-Köhler K, Vehling-Kaiser U, et al: Phase III trial of
irinotecan plus infusional 5-fluorouracil/folinic acid versus
irinotecan plus oxaliplatin as first-line treatment of advanced
colorectal cancer. Eur J Cancer. 47:206–214. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Sun M, Zhang Q, Yang X, Qian SY and Guo B:
Vitamin D enhances the efficacy of irinotecan through
miR-627-mediated inhibition of intratumoral drug metabolism. Mol
Cancer Ther. 15:2086–2095. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Quackenbush KS, Bagby S, Tai WM,
Messersmith WA, Schreiber A, Greene J, Kim J, Wang G, Purkey A,
Pitts TM, et al: The novel tankyrase inhibitor (AZ1366) enhances
irinotecan activity in tumors that exhibit elevated tankyrase and
irinotecan resistance. Oncotarget. 7:28273–28285. 2016. View Article : Google Scholar : PubMed/NCBI
|