Introduction
The cause of the progressive depletion of (cluster
of differentiation) CD4+ T cells in human
immunodeficiency virus (HIV)-infected people is one of the most
fundamental and controversial issues in HIV/acquired
immunodeficiency syndrome (AIDS) research. Multiple mechanisms have
been proposed to explain this depletion, including direct HIV
cytopathicity (1),
antigen-specific cytotoxic lymphocyte (CTL)-mediated lysis
(2), suppressed thymic function
(3–5) and chronic immune activation (6–8)
resulting in increased rates of apoptosis, and cell turnover which
consumes naïve and memory CD4+ T cell-pools. Previous
studies have suggested that lymph node (LN) fibrosis following HIV
infection is an important mechanism underlying the pathogenesis of
HIV/AIDS. As an important member of lymphatic tissues, LNs are
pivotal in the maintenance of immune homeostasis as well as the
survival, proliferation and differentiation of lymphocytes
(9–13). Immune activation and inflammation
subsequent to HIV infection may cause the deposition of excess
collagen in the LNs, which inhibits the interaction of lymphocytes
with antigen presenting cells and cytokines, and compromises
lymphocyte survival as well as the immune response.
Fibrotic diseases can occur in various organs,
including the lung and liver (14). Although multiple mechanisms may
contribute to the fibrogenic process, TGF-β1 is considered to serve
a central role in inducing fibroblasts to synthesize collagen
(15). As a multifunctional and
potent regulatory cytokine, TGF-β1 can be expressed on diverse cell
types, including T cells, B cells, macrophages, natural killer
cells and dendritic cells (16–19).
In the present study, the mechanism underlying collagen deposition
in the LNs following HIV infection was investigated by measuring
TGF-β1 expression in the lymphocytes of asymptomatic HIV carriers
and AIDS patients. The results of the present study have expanded
the understanding of the pathogenesis of AIDS.
Materials and methods
Patients
The present study recruited patients who were
diagnosed with HIV infection and had received LN biopsy at the
Fourth People's Hospital of Nanning (Nanning, China) and the 302
Military Hospital of China (Beijing, China) between October 2009
and March 2015. A total of 32 patients were included in this study
(neck, n=18; armpit, n=12; groin, n=2). The patients were divided
into asymptomatic HIV carriers (n=10; HIV infection for >6
months and CD4+T cells >200/mm3) and AIDS
patients (n=22; HIV infection for >6 months and CD4+T
cells <200/mm3). HIV infection was caused by
heterosexual sex (n=19), homosexual sex (n=8) or intravenous
infusion (n=5). Patients with opportunistic infection were
excluded. None of the patients received anti-viral therapy. In
addition, superficial LNs were also collected from subjects without
HIV infection (n=10) between February 2010 and December 2014 in the
302 Military Hospital of China (Beijing, China). Reactive
hyperplasia was diagnosed in these subjects by pathological
examination.
Informed consent was obtained prior to the present
study and the study protocol was approved by the Ethics Committee
of The Fourth People's Hospital of Nanning (Nanning, China), and
the Ethics Committee of 302 Military Hospital of China (Beijing,
China). The clinical characteristics of study subjects are
illustrated in Table I.
 | Table I.Characteristics of the subjects
recruited in the present study. |
Table I.
Characteristics of the subjects
recruited in the present study.
| Characteristic | Control | Asymptomatic HIV
infection | AIDS |
|---|
| Sex |
|
|
|
|
Male | 8 | 7 | 15 |
|
Female | 2 | 3 | 7 |
| Age (years) | 37.65±8.63 | 34.42±7.58 | 40.33±9.41 |
| CD4+
cells (cells/mm3) | N/A | 288.54±21.01 | 104.42±33.15 |
| HIV load
(Log10 IU/ml) | N/A |
4.03±1.12 |
5.21±1.46 |
| Anti-viral
therapy | N/A | No | No |
| Concomitant
infection | No | No | No |
Processing of LNs
Superficial LNs were collected surgically and
divided into 2 parts: One part was cut into blocks and ground using
a 100-mesh (pore size: 150-µm), filtered, and then transferred into
Hanks solution containing 2% fetal calf serum (FCS; Gibco; Thermo
Fisher Scientific, Inc., Waltham, MA, USA). The filter was washed
with Hanks solution containing 2% FCS. The resultant solution was
collected and centrifuged at 700 × g for 15 min at room temperature
(RT). The supernatant was removed and the pellet was collected and
re-suspended in 10 ml phosphate-buffered saline (PBS). This
suspension was then layered onto Ficoll-Hypaque solution and
lymphocytes were centrifuged at 400 × g for 35 min at RT. The
remaining LNs were fixed in 4% paraformaldehyde for 48 h at RT and
embedded in paraffin at a thickness of 4 µm.
Immunohistochemistry
Immunohistochemistry was performed with anti-TGF-β1
antibody (1:50; cat. no. ab190503; Abcam, Cambridge, UK),
anti-forkhead box p3 (Foxp3) antibody (1:200; cat. no. ab4728;
Abcam) and anti-CD8 antibody (1:250; cat. no. sc-7188; Santa Cruz
Biotechnology, Inc., Dallas, TX, USA) using the streptomycin
avidin-peroxidase (SP) method. Sections were first deparaffinized
and hydrated. Sections were subsequently boiled, maintained at
sub-boiling temperature for 15 min, washed twice in deionized
H2O and cooled for 30 min at RT. Sections were incubated
with 0.3–1.0% H2O2 in methanol to inactivate
endogenous peroxidase. Specimens were blocked in 5% FCS (Gibco;
Thermo Fisher Scientific, Inc.) for 30 min at RT, and incubated
with the appropriate antibody at 4°C overnight. Sections were then
incubated with horseradish peroxidase (HRP)-conjugated goat
anti-rabbit (cat. no. ab205718; 1:20,000; Abcam) or goat anti-mouse
(cat. no. ab205719; 1:10,000; Abcam) secondary antibodies and
aminoethyl carbazole (AEC) was used for visualization for 5 min at
RT. The slides were observed with a light microscope
(magnification, ×200; Olympus CX31; Olympus Corporation, Tokyo,
Japan). Positive cells were stained red. The double enzyme-double
substrate method was used for double staining. Cells positive for
5-bromo-4-chloro-3-indolyl phosphate (BCIP)/nitroblue-tetrazolium
(NBT) were blue and those positive for AEC were red. In the
negative control group, the primary antibody was replaced with PBS
in the HIV-infected patients. Sections from non-HIV infected
subjects also served as controls.
Flow cytometry
Antibodies against CD3-phycoerytherin (PE; 1:20;
cat. no. 555340; BD Pharmingen, San Diego, CA, USA),
CD3-Allophycocyanin (APC; 1:20; cat. no. 555342, BD Pharmingen),
CD4-Peridinin Chlorophyll (Percp; 1:20; cat. no. 550631; BD
Biosciences, San Jose, CA, USA), CD8-Percp (1:20; cat. no. 347314;
BD Biosciences), CD38-fluorescein isothiocyanate (FITC; 1:20; cat.
no. 555459; BD Pharmingen) and CD127-PE (1:20; cat. no. 557938; BD
Pharmingen), programmed cell death protein 1 (PD-1)-PE (1:40; cat.
no. 329906; BioLegend, Inc., San Diego, CA, USA), Foxp3-FITC (1:40;
cat. no. 11-4776-42; eBioscience; Thermo Fisher Scientific, Inc.)
and TGF-β1-APC (1:20; cat. no. IC240A; R&D Systems, Inc.,
Minneapolis, MN, USA) were used for flow cytometry. Lymphocytes
from LNs were suspended in PBS, followed by cell counting. The cell
density was >2.5×105/ml. Cell suspensions (200 µl)
were transferred into a tube and Human TruStain FcX blocking
solution (cat. no. 422302, BioLegend, Inc.) was applied to reduce
non-specific staining at RT for 7 min. Samples were subsequently
incubated with the above antibodies at RT for 30 min in the dark.
Following the addition of 1 ml PBS, samples were centrifuged at 350
× g at RT for 5 min. The pellet resuspended in 1 ml PBS and
centrifuged again at 350 × g at RT for 5 min. Cells were
resuspended in 200 µl PBS and subjected to flow cytometry (FACS
Aria; BD Biosciences, Franklin Lakes, NJ, USA). Detection of
regulatory T cells (Tregs) was done using anti-CD3, anti-CD4 and
anti-Foxp3 antibodies. Cell membranes were permeabilized and the
samples were incubated with anti-Foxp3 antibody and subjected to
flow cytometry.
Sorting and stimulation of
CD8+T cells
Two LNs were collected from non-HIV-infected
subjects and lymphocytes were extracted as described above.
CD8+ T cells were sorted with CD8 MicroBeads according
to the manufacturer's protocol (Miltenyi Biotec GmbH, Bergisch
Gladbach, Germany). Flow cytometry with an antibody against
CD8-Percp (1:20; cat. no. 347314; BD Biosciences) demonstrated that
the purity was >97%. Lymphocytes were stimulated with
phorbol-12-myristate 13-acetate (PMA; cat. no. ab120297, Abcam) at
50 µg/ml at 37°C for 6 h. The above two LNs were fixed and embedded
as described in the LN processing section. Paraffin sections were
subsequently stained with hematoxylin and eosin at RT (hematoxylin
for 10 min; eosin for 2 min) and observed under a light microscope
(magnification, ×200; Olympus CX31; Olympus Corporation) for the
pathological diagnosis of reactive hyperplasia.
Detection of collagens secreted by human lymphatic
fibroblasts. Human lymphatic fibroblasts (cat. no. 2530; ScienCell
Research Laboratories, Inc., Carlsbad, CA, USA) were cultured in
fibroblast medium (cat. no. 2301; ScienCell Research Laboratories,
Inc.). The effect of different concentrations of TGF-β1 on collagen
secretion from fibroblasts was investigated. Fibroblasts were
seeded in 12-well plates at a density of 2.5×105/well
and incubated at 37°C for 12 h. Cells were subsequently incubated
for 24 h with or without TGF-β1 (cat. no. 240-B, R&D Systems,
Inc.; 0.25, 0.5, 1 and 5 ng/ml). Cells were then digested with
trypsin and harvested for detection of type I collagen by western
blot analysis. The effect of stimulated CD8+ T cells on
collagen secretion by fibroblasts was also investigated.
Fibroblasts were seeded in 6-well plates at a density of
2.5×105 cells/well, incubated for 12 h at 37°C and then
co-cultured with CD8+ T cells from LNs of non-HIV
infected individuals. These cells were divided into 4 groups: The
fibroblasts group (group a), co-culture group of nonstimulated
CD8+ T cells + fibroblasts (group b), co-culture group
of PMA stimulated CD8+ T cells + fibroblasts (group c)
and co-culture group of PMA stimulated CD8+ T cells +
fibroblasts + TGF-β antagonist (2 µg/ml; cat. no. MAB1835, R&D
Systems, Inc.; group d). The ratio of fibroblasts to
CD8+ T cells was 1:10. Cultures were incubated for 24 h
at 37°C and the presence of type I collagen was detected using
immunofluorescence staining and western blot analysis.
Western blot analysis
Cells were digested and centrifuged at 350 × g for 5
min at 4°C. The pellet was washed with PBS and cells were lysed
with 100–200 µl radioimmunoprecipitation assay lysis and extraction
buffer (cat. no. 89900, Thermo Fisher Scientific, Inc.) at 4°C
overnight. Lysates were centrifuged at 20,000 × g for 15 min at 4°C
and the supernatant was collected for detection of type I collagen
using a routine western blot analysis as previously described
(20). Anti-type I collagen
antibody was purchased from Abcam (1:500; cat. no. ab190503) and
anti-β-actin antibody from Santa Cruz Biotechnology, Inc (1:200;
cat. no. sc-130065). Rabbit anti-mouse IgG antibody conjugated to
HRP was used as secondary antibody (1:2,000; cat. no. ab6728;
Abcam). Relative band density was analyzed with Image J 1.4.3.67
software (National Institutes of Health, Bethesda, MD, USA).
Immunofluorescence staining
Fibroblasts were harvested from 6-well plates and
washed thrice with PBS. The cells were fixed in 4% paraformaldehyde
for 10 min at RT and permeabilized with 0.2% Triton X-10. Cells
were then blocked with 5% bovine serum albumin (BSA; Amresco, LLC,
Solon, OH, USA) at RT for 30 min, followed by incubation with
anti-type I collagen antibody (Abcam) at 4°C overnight. Following
washing thrice with PBS, the cells were incubated with
FITC-conjugated secondary antibody (1:400; cat. no. ab97022; Abcam)
for 30 min in the dark. Representative images were captured under a
fluorescence microscope (magnification, ×400; Nikon 80i; Nikon
Corporation, Tokyo, Japan).
Statistical analysis
Data from flow cytometry were analyzed with
FlowJo5.7.2 software (FlowJo LLC, Ashland, OR, USA). Statistical
analysis was performed with SPSS version 17.0 (SPSS Inc., Chicago,
IL, USA). Data with normal distribution and homogeneity of variance
were expressed as the mean ± standard deviation. Comparisons were
done with one-way analysis of variance followed by Bonferroni's
procedure. P<0.05 was considered to indicate a statistically
significant difference. Western blot analysis and
immunofluorescence experiments were repeated three times. Flow
cytometry and immunohistochemistry were performed once for every
sample.
Results
TGF-β1-expressing cells and their
distribution
To elucidate the mechanism of fibrosis in LNs of
HIV-infected individuals, TGF-β1 expression and distribution in LNs
were detected with immunohistochemistry. The results of the present
study demonstrated that TGF-β1 was expressed in a large number of
cells in the LNs of the control group, as well as in asymptomatic
HIV carriers and AIDS patients. However, the border of
TGF-β1-positive cells was not sharp in asymptomatic HIV carriers
and AIDS patients compared with the control, which could be
attributed to disorganization of LNs following HIV infection
(Fig. 1).
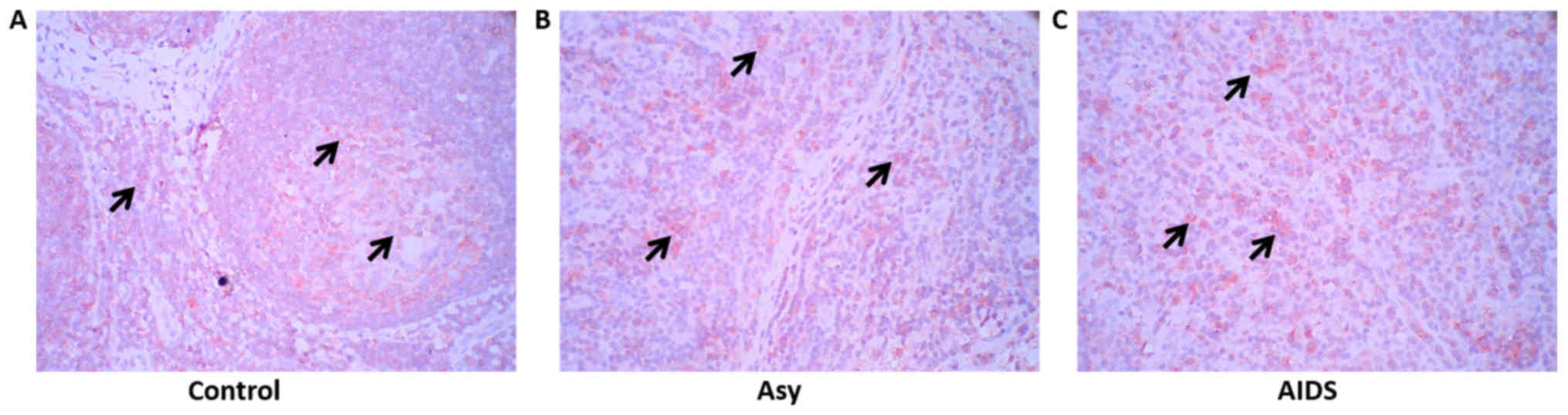 | Figure 1.TGF-β1 expression in the peripheral
superficial LNs of the Asy, AIDS and control subjects. (A) Non-HIV
infection subjects (n=10). (B) Asy (n=10). (C) AIDS patients
(n=22). Red, indicated by arrows, represents TGF-β1-positive cells
(cell membrane expression; aminoethyl carbazole). A large number of
TGF-β1-positive cells are observed in the LNs of the three groups,
but the distribution border of TGF-β1-positive cells was not sharp
in the asymptomatic HIV carriers and in AIDS patients.
Magnification, ×400. AIDs, acquired immunodeficiency syndrome; Asy,
asymptomatic HIV carriers; HIV, human immunodeficiency virus; LNs,
lymph nodes; TGF-β1, transforming growth factor-β1. |
Treg cells and TGF-β1-positive Treg
cells in LNs
The role of Treg cells in LN fibrosis of chronic HIV
infection remains unclear (21).
In the present study, the frequencies of Treg cells in the
lymphocytes from LNs and TGF-β1 expression in these Treg cells were
investigated by flow cytometry (Fig.
2). The results demonstrated that there was no significant
difference in the frequency of Treg cells between asymptomatic HIV
carriers and controls (8.635±1.159% vs. 8.169±1.388%; P>0.05),
but the frequency of TGF-β1-positive Treg cells was significantly
higher in asymptomatic HIV carriers compared with the controls
(18.487±1.571% vs. 9.875±2.183%, P<0.001; Fig. 2A, B, D and E). Patients with AIDS
exhibited a significantly lower proportion of Treg cells
(4.319±1.249%) compared with the other two groups (P<0.001;
Fig. 2A and D). In addition,
although AIDS patients had a significantly lower proportion of
TGF-β1+ Treg cells compared with asymptomatic HIV
carriers (7.691±2.321%; P<0.001), the level of
TGF-β1+ Treg cells were similar to the control group
(P=0.072; Fig. 2B and E).
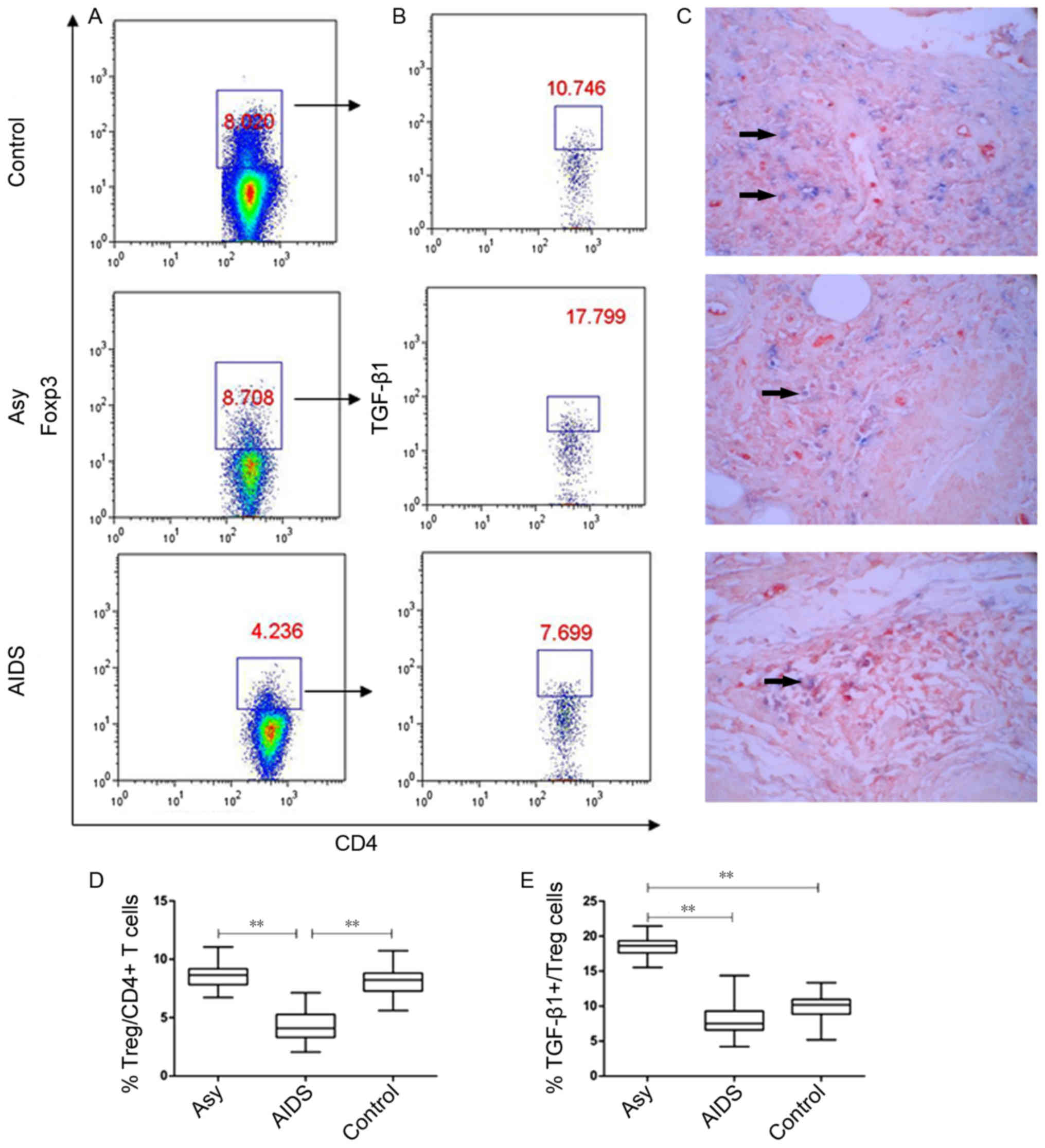 | Figure 2.Proportion of Treg cells and
TGF-β1-positive Treg cells in the peripheral superficial LNs of the
Asy, AIDS and control groups. (A) Representative prevalence of Treg
cells from individual subjects in the three studied groups. Foxp3,
an X chromosome-encoded fork head transcription factor family
member is indispensable for the differentiation of Treg cells. Treg
cells were gated from Foxp3+ subsets of
CD3+CD4+ T cells. (B) Representative
prevalence of TGF-β1-positive Treg cells from individual subjects
in three studied groups. (C) Double staining of Foxp3 and TGF-β1
from individual subjects in the three studied groups. Blue, Foxp3
(nucleus expression; BCIP/NBT, magnification, ×400); Red, TGF-β1
(cell membrane expression; aminoethyl carbazole; ×400). Double
positive cells for Foxp3 and TGF-β1 are indicated by arrows. (D)
Statistical analysis of the frequency of
CD3+CD4+Foxp3+ Treg and (E)
CD3+CD4+Foxp3+TGF-β1+
Tregs in peripheral superficial LNs of patients with AIDS, asy and
controls. Data in D and E are expressed as box plots, in which the
horizontal lines illustrate the 25, 50, 75th percentiles. Vertical
lines represent the 10 and 90th percentiles. Tissue samples, n=10
Control; n=10 Asy; n=22 AIDS. **P<0.001. AIDS, acquired
immunodeficiency syndrome; Asy, asymptomatic HIV carriers;
BCIP/NBT, 5-bromo-4-chloro-3-indolyl
phosphate/nitroblue-tetrazolium; CD, cluster of differentiation;
Foxp3, forkhead box P3; LNs, lymph nodes; TGF-β1, transforming
growth factor-β1; Treg, regulatory T cell. |
Furthermore, the results of the expression of TGF-β1
and Foxp3, which were detected with double staining of
immunohistochemistry in situ, demonstrated that Foxp3 and
TGF-β1 double positive cells accounted for only a small fraction of
TGF-β1 positive cells, indicating that most TGF-β1-positive cells
were not Treg cells (Fig. 2C).
CD8+T cells exhibit high
TGF-β1 expression in peripheral LNs
The above results implied that other TGF-β-positive
cells could serve specific roles in LN fibrosis following HIV
infection. B cells and T cells are localized in different areas in
the LNs. Therefore, TGF-β1 expression in the T cells of the LNs was
determined by flow cytometry. The results demonstrated that the
asymptomatic HIV carriers had the highest proportion of T cells
with high TGF-β1 expression (12.26±2.07%), followed by the AIDS
group (7.588±1.89%) and the control group (2.609±1.044%) and the
difference between all groups was statistically significant
(P<0.001; Fig. 3). In the
asymptomatic HIV carriers and AIDS group, the cells were mainly
CD8+T cells and accounted for 83.41±8.761% and
82.76±6.076%, respectively, which was not significantly different
(P>0.05). In the control group, the main type of cells were
CD4+ T cells, with only 33.1±4.928% of cells being
CD8+ T cells. The asymptomatic HIV carriers and the AIDS
group exhibited a significantly increased proportion of
CD8+ T cells with high TGF-β1 expression compared with
the control group (P<0.001; Fig.
3). Double staining for CD8 and TGF-β1 was performed to confirm
that CD8+ T cells expressed TGF-β1. The results
demonstrated that a number of CD8+ T cells (red) were
positive for TGF-β1 (blue) in the peripheral superficial LNs of
HIV-infected individuals (Fig.
3E).
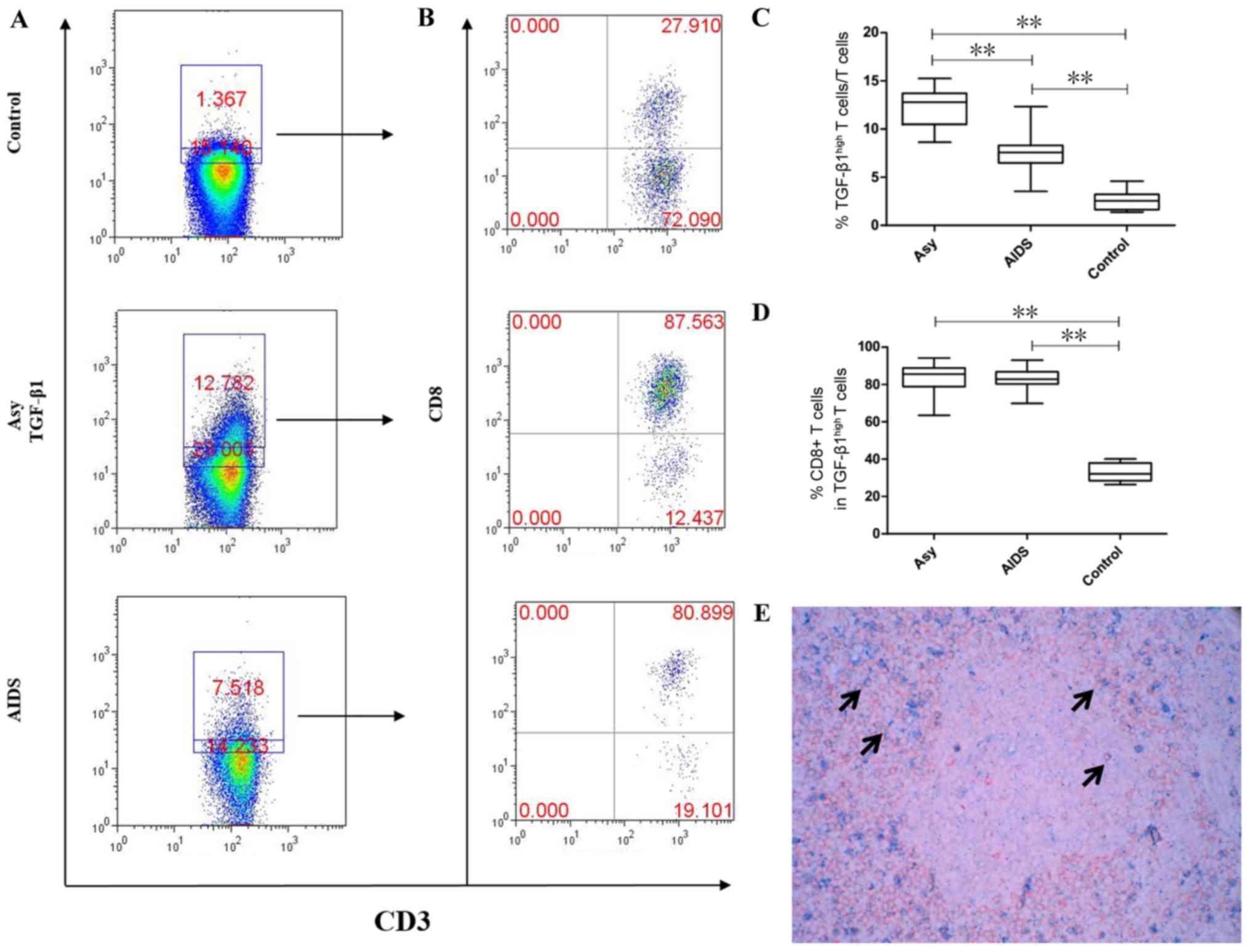 | Figure 3.Proportions of T cells with high
TGF-β1 expression in the peripheral superficial LNs of the Asy,
AIDS and control groups. (A) Representative prevalence of T cells
with high TGF-β1 expression from individual subjects in the three
studied groups. (B) Representative prevalence of TGF-β1-highly
expressing CD8+ T cells from individual subjects in the
three studied groups. (C) Statistical analysis demonstrates the
frequency of T cells with high TGF-β1 expression is the highest in
peripheral superficial LNs of Asy, followed by patients with AIDS
and lowest in controls. The difference between the three groups was
statistically significant. (D) Statistical analysis demonstrates
that CD8+ T cells are the main subset which highly
expressed TGF-β1 in Asy and AIDS patients and is significantly
higher than in controls. (E) Double staining of CD8 and TGF-β1 in
the peripheral superficial LNs of an asymptomatic HIV carrier.
Double positive cells of CD8 and TGF-β1 are indicated by arrows.
Blue, TGF-β1 (cell membrane expression; BCIP/NBT); Red, CD8 (cell
membrane expression; aminoethyl carbazole). Magnification, ×400.
Data in C and D are expressed as box plots, in which the horizontal
lines illustrate the 25, 50, 75th percentiles. Vertical lines
represent the 10 and 90th percentiles. Tissue samples, n=10
Control; n=10 Asy; n=22 AIDS. **P<0.001. AIDS, acquired
immunodeficiency syndrome; Asy, asymptomatic HIV carriers;
BCIP/NBT, 5-bromo-4-chloro-3-indolyl
phosphate/nitroblue-tetrazolium; CD, cluster of differentiation;
LNs, lymph nodes; TGF-β1, transforming growth factor-β1. |
Association between CD8+T
cells with high TGF-β1 expression and the secretion of type I
collagen in human lymphatic fibroblasts
In order to investigate the role of CD8+
T cells with high TGF-β1 expression in LN fibrosis, human lymphatic
fibroblasts were treated with different concentrations of TGF-β1
for 24 h and expression of type I collagen was detected by western
blot analysis. The results demonstrated that TGF-β1 upregulated the
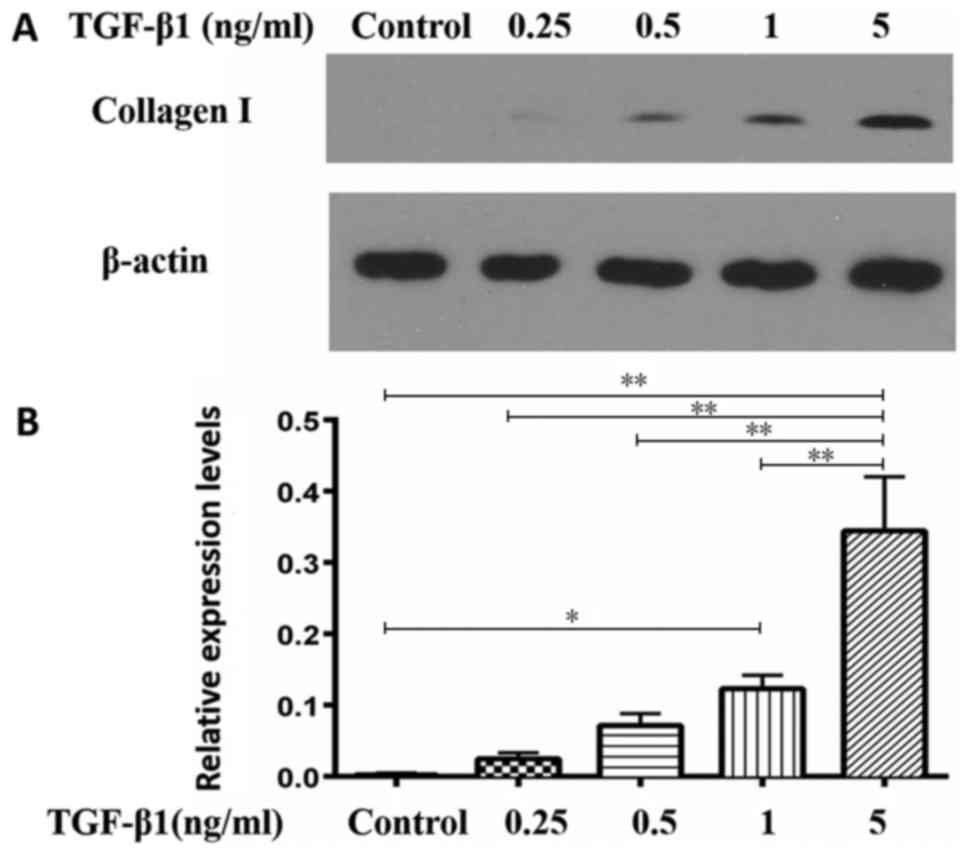
expression of type I collagen in a dose-dependent manner (Fig. 4). Relative expression levels of
type I collagen respectively were 0.0033±0.0018, 0.0247±0.0085,
0.7167±0.0165, 0.123±0.0187 and 0.3443±0.076 in the control, 0.25,
0.5, 1 and 5 ng/ml groups, respectively. The difference between 5
ng/ml group and the other four groups was statistically significant
(P<0.01).
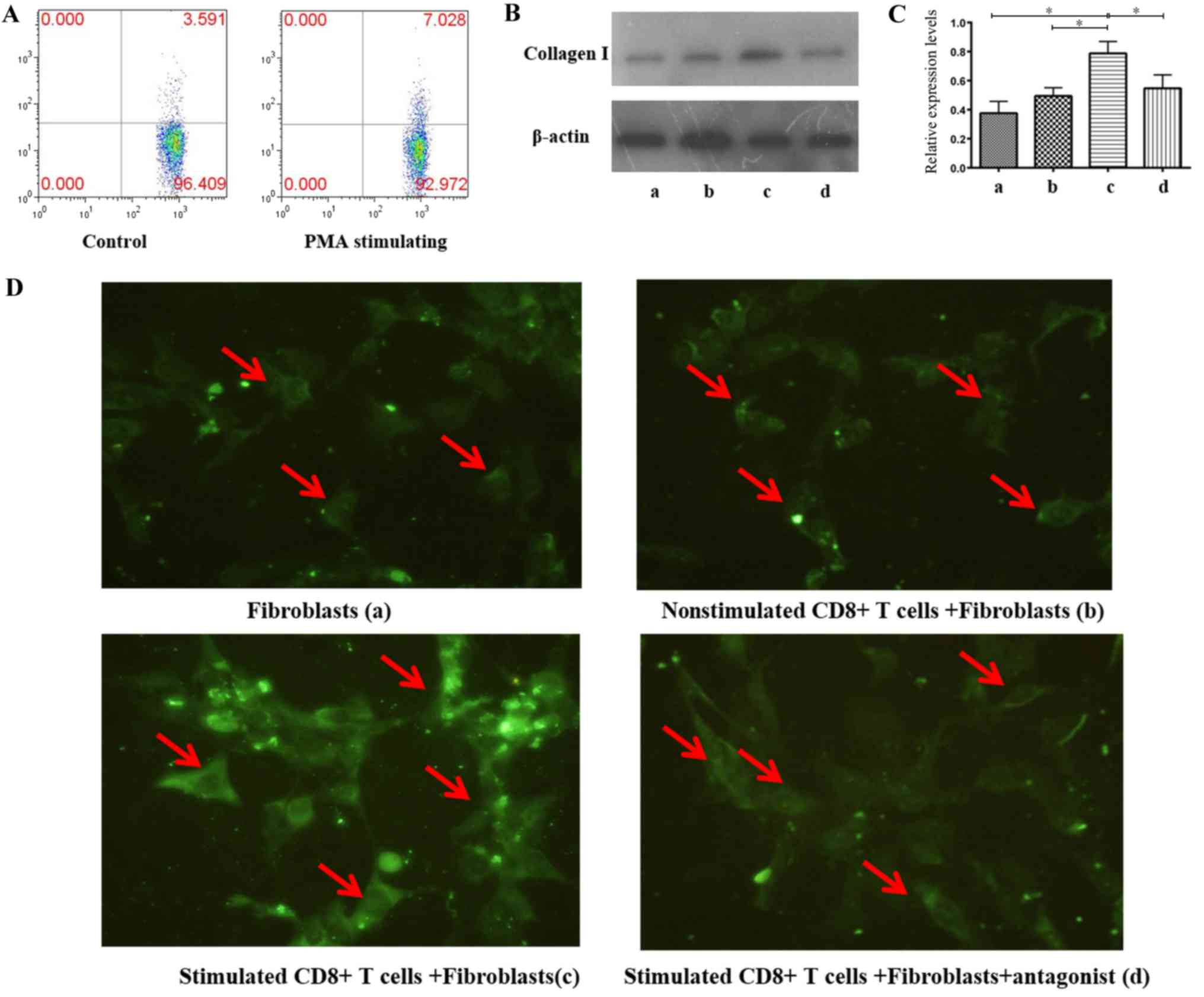
Subsequently, CD8+ T cells from LNs of
non-HIV infected subjects were separated, stimulated to express
TGF-β1 at a high level; flow cytometry demonstrated that the
CD8+ T cell TGF-β1 expression increased by 1-fold
following stimulation, compared with no stimulation (Fig. 5A). Cells were subsequently
co-cultured with human lymphatic fibroblasts to investigate the
effect of CD8+ T cells with high TGF-β1 expression in LN
fibrosis. Immunofluorescence staining and western blot analysis
were conducted to detect type I collagen expression according to
grouping described in the Materials and methods section. The
results demonstrated a significant increase in type I collagen
expression in the co-culture group with stimulated CD8+
T cells (0.7878±0.0814) compared with three other groups
(P<0.05); secretion of type I collagen was significantly lower
in the fibroblasts group (0.3761±0.0812) and the co-cultured group
of non-stimulated CD8+ T cells + fibroblasts
(0.4945±0.057). The antagonist significantly inhibited type I
collagen secretion of fibroblasts which were co-cultured with
CD8+ T cells with high TGF-β1 expression (0.5473±0.0919;
Fig. 5B-D). There was no
statistically significant difference between groups a, b and d. In
addition, the result of immunofluorescence staining according to
fluorescence intensity also implied that type I collagen expression
in the co-culture group with stimulated CD8+ T cells was
highest (Fig. 5D; group c).
Expression of CD38, PD-1 and CD127 in
CD8+ T cells of LNs from HIV-infected individuals
To evaluate the influence of HIV infection on
CD8+ T cells, the expression levels of CD127, CD38 and
PD-1 were measured in CD8+ T cells, which were obtained
from the LNs of HIV-infected individuals. The results demonstrated
that the proportions of CD38-, PD-1- and CD127-positive cells were
6.13±2.711, 10.242±4.603 and 73.444±9.34%, respectively in the
control group, 20.103±6.466, 27.387±9.304 and 55.576±9.073%,
respectively in the asymptomatic HIV carriers, and 24.219±7.016,
21.774±5.729 and 46.289±9.518% respectively in patients with AIDS
(Fig. 6). Compared with the
control group, the proportions of CD38+ cells and
PD-1+ cells were significantly increased, while the
proportion of CD127-positive cells was significantly decreased in
the asymptomatic HIV carriers and the AIDS patients (all
P<0.01). There were no significant differences in the
proportions of CD127-, CD38- or PD-1-positive cells between the
AIDS patients and asymptomatic HIV carriers (Fig. 6).
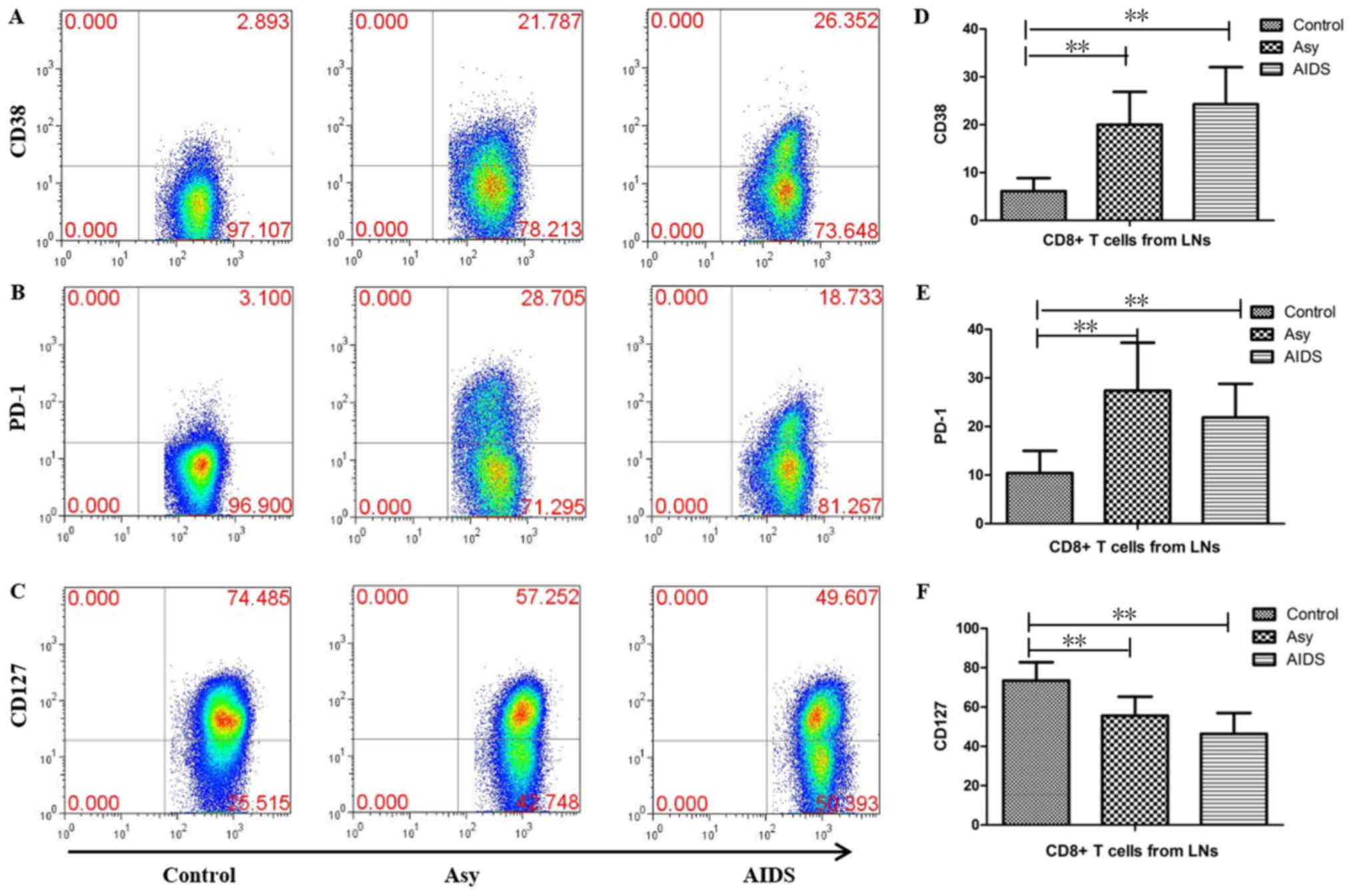 | Figure 6.Expression of CD38, PD-1 and CD127 in
CD8+ T cells in peripheral superficial LNs from
HIV-infected individuals and controls. Representative prevalence of
(A) CD38-, (B) PD-1- and (C) CD127-positive CD8+ T cells
which were isolated from LNs of the three studied groups.
Statistical analysis demonstrates that the frequencies of (D)
CD38+CD8+ T cells and (E)
PD-1+CD8+ T cells in peripheral superficial
LNs of asymptomatic HIV carriers and AIDS patients were
significantly higher than controls, but the frequency of (F)
CD127+CD8+ T cells was significantly lower
than controls. There were no significant differences in the
frequency of CD127-, CD38- or PD-1-positive CD8+ T cells
between the AIDS patients and asymptomatic HIV carriers. The data
are presented as the mean ± standard deviation. control tissue
samples, n=10; Asy, n=10; AIDs, n=22. **P<0.01. AIDs, acquired
immunodeficiency syndrome; Asy, asymptomatic HIV carriers. CD,
cluster of differentiation; HIV, human immunodeficiency virus; LNs,
lymph nodes; PD-1, programmed cell death protein 1. |
Discussion
The normal structure of LNs is crucial for the
survival, development and immune response of T cells, especially
naïve T cells. Previous studies demonstrated that LN fibrosis is an
important mechanism underlying the reduction in CD4+ T
cells following HIV infection. The severity of LN fibrosis in
HIV-infected individuals is negatively associated with the number
of CD4+ T cells and the recovery of peripheral
CD4+ T cells following anti-viral therapy (22–30).
LN fibrosis may be a cause of CD4+ T cell reduction as
collagen deposition disrupts the internal environment of the LNs,
inhibits the intercellular interaction leading to an abnormal
immune response of T cells and reduces the contact of T cells with
cytokines including interleukin (IL)-7, which are necessary for
survival.
TGF-β has been demonstrated to be necessary for
maintaining immune tolerance, inhibiting inflammation and mediating
fibrosis of tissues or organs (15,31–33).
TGF-β1 is the most important member of the TGF-β family (16). Previous studies have demonstrated
that TGF-β1+ Treg cells may induce the deposition of
collagens in LNs during the acute phase of Simian immunodeficiency
virus infection (21). Therefore,
the frequency of Treg cells and TGF-β1+ Treg cells were
analyzed in the peripheral superficial LNs of asymptomatic HIV
carriers, AIDS patients and non-HIV infected subjects. The results
of the present study demonstrated that the proportion of
TGF-β1+ Treg cells in the LNs of asymptomatic HIV
carriers was increased compared with the control group, although
the proportion of Treg cells was comparable between the
asymptomatic HIV carriers and control groups. Despite the
comparable proportion of TGF-β1+ Treg cells between the
AIDS and control group, the frequency of Treg cells was decreased
in the AIDS group compared with the control group, which may be
ascribed to the infection and consumption of Treg cells as
CD4+ cells in the AIDS stage. The results of the present
study indicated that TGF-β1+ Treg cells serve a dominant
role in LN fibrosis of asymptomatic HIV carrier in chronic HIV
infection.
In addition to Treg cells, TGF-β1 is also expressed
on other immune cells. The results of the immunohistochemistry
demonstrated that a large number of cells expressed TGF-β1 in the
LNs of non-HIV-infected subjects as well as HIV-infected subjects.
Since it remains unclear why LN fibrosis is only present in
HIV-infected patients and whether other cells expressing TGF-β1 are
involved in LN fibrosis following HIV infection, flow cytometry was
performed to detect TGF-β1 expression in T cells. The results
demonstrated that a fraction of T cells in LNs exhibited high
TGF-β1 expression, the proportions of which were respectively, a
6-fold and 3-fold higher in asymptomatic HIV carriers and AIDS
patients compared with the control group. In asymptomatic HIV
carriers and AIDS patients, these high TGF-β1 expressed cells were
mainly CD8+ T cells and accounted for >80% of the T
cells, with no significant difference between both groups. In
non-HIV-infected subjects, these cells were mainly CD4+
T cells, while CD8+ T cells accounted for ~30% of the T
cells.
TGF-β serves multiple roles in inflammation and
immune regulation (34–36). Different concentrations of TGF-β
have been demonstrated to activate distinct signaling pathways
(37). The results of the
co-culture experiment in the present study demonstrated that
stimulated CD8+ T cells induced human lymphatic
fibroblasts to secrete a large amount of collagen I. The dose
response data of the present study also demonstrated that the
secretion capacity of collagen I in fibroblasts is associated with
TGF-β1 concentrations. These results suggested that
TGF-β1+ T cells in LNs are unable to induce fibrosis
under normal conditions as most of them exhibit low TGF-β1
expression.
Persistent, chronic immune activation and
inflammation are important immunological characteristics of LNs
following HIV infection (38).
CD127, which is a receptor of IL-7 expressed on naïve and memory
CD8+ T cells, serves an important role in the
maintenance of homeostasis of naïve and memory CD8+ T
cells. CD127 expression was illustrated to be downregulated in
effector CD8+ T cells (39). PD-1 is expressed in activated T
cells, B cells and myeloid cells. Notably, CD8+ T cells
with functional deficits exhibit high PD-1 expression, which is
thought to compromise the functions of CD8+ T cells
(proliferation, secretion of cytokines and killing activity),
including in chronic HIV infection (40,41).
The results of the present study demonstrated a significant
increase in the proportions of CD38-positive cells and
PD-1-positive cells, and a significant decrease in CD127-positive
cells among CD8+ T cells in LNs from HIV-infected
individuals. These data suggested that CD8+ T cells are
activated in the LNs of HIV-infected individuals and demonstrate
compromised functions. CD8+ T cells from LNs, which are
stimulated in vitro demonstrated increased TGF-β1
expression, implying that the increase of CD8+ T cells
that express high levels of TGF-β1 in LNs of HIV infected patients
could be also associated with immune activation and
inflammation.
As cytotoxic T cells, CD8+ T cells are
the major cells defending the host against HIV infection. In
addition, they are also regarded as serving an important role in
other immune processes. Previous studies have demonstrated that a
subset of CXCR5+CD8+ T cells can localize in
B-cell follicles and act as regulatory cells suppressing follicular
T helper cells to help B cells and maintaining immune tolerance
(42). Certain autoimmune
diseases, which possess a normal quantity and function of
CD4+ Treg cells may be a result of abnormal
CD8+ regulatory T cells (43–46).
A previous study demonstrated another subset of
CXCR5+CD8+ T cells, which can also settle in
B-cell follicles and serves a crucial role in the control of
chronic viral infection (47,48).
In addition, a previous study also revealed that IL-13-producing
CD8+ T cells aggregate in the skin of patients with
systemic sclerosis, especially in early stages of inflammation and
may induce the secretion of a large amount of extracellular matrix
by fibroblasts in the epidermis, which regulates skin fibrosis
(49). These results indicate that
CD8+ T cells have multiple functions, in addition to
clearing infection and are able to regulate the immune response and
inflammatory reaction.
Fibrosis may occur in multiple organs and tissues
and cause corresponding diseases in these organs. To date, the
pathogenesis of fibrosis remains poorly understood. The results
demonstrated that LN fibrosis may be mediated by an increase in
CD8+ T cells that express a high level of TGF-β1 in the
LNs following HIV-infection. This also implies that the increase in
TGF-β1-highly expressing CD8+ T cells is associated to
inflammation and activation of immune cells in LNs following HIV
infection. The results of the present study provided evidence to
understand the mechanism underlying LN fibrosis following HIV
infection. CD8+ T cells serve multiple roles in the
immune response and inhibition of immune activation can delay or
inhibit LN fibrosis following HIV infection.
Acknowledgements
Not applicable.
Funding
The present study was supported by grants from the
National Natural Science Foundation of China (grant no. 81772185)
and the 12th Five Year Research Project of People's Liberation Army
(grant no. CWS11J160).
Availability of data and materials
The analyzed data sets generated during the study
are available from the corresponding author on reasonable
request.
Authors' contributions
LH, PYM and XZZ conceptualized the study design. LH,
JND, WX and HBW performed the experiments. LH, WX, WMN, LS and DW
analyzed the data. JND, HBW, WMN, FYW and MZ recruited subjects and
collected specimens. LH, WX, PYM and XZZ wrote the paper.
Ethics approval and consent to
participate
Informed consent was obtained prior to the present
study and the study protocol was approved by the Ethics Committee
of The Fourth People's Hospital of Nanning, and the Ethics
Committee of 302 Military Hospital of China.
Consent for publication
Not applicable.
Competing interests
The authors declare they have no competing
interests.
References
|
1
|
Gandhi RT, Chen BK, Straus SE, Dale JK,
Lenardo MJ and Baltimore D: HIV-1 directly kills CD4+ T cells by a
Fas-independent mechanism. J Exp Med. 187:1113–1122. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zinkernagel RM: Are HIV-specific CTL
responses salutary or pathogenic? Curr Opin Immunol. 7:462–470.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
McCune JM: The dynamics of CD4+ T-cell
depletion in HIV disease. Nature. 410:974–979. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Dion ML, Poulin JF, Bordi R, Sylvestre M,
Corsini R, Kettaf N, Dalloul A, Boulassel MR, Debré P, Routy JP, et
al: HIV infection rapidly induces and maintains a substantial
suppression of thymocyte proliferation. Immunity. 21:757–768. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
McCune JM, Loftus R, Schmidt DK, Carroll
P, Webster D, Swor-Yim LB, Francis IR, Gross BH and Grant RM: High
prevalence of thymic tissue in adults with human immunodeficiency
virus-1 infection. J Clin Invest. 101:2301–2308. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Brenchley JM, Price DA and Douek DC: HIV
disease: Fallout from a mucosal catastrophe? Nat Immunol.
7:235–239. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Grossman Z, Meier-Schellersheim M, Paul WE
and Picker LJ: Pathogenesis of HIV infection: What the virus spares
is as important as what it destroys. Nat Med. 12:289–295. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Derdeyn CA and Silvestri G: Viral and host
factors in the pathogenesis of HIV infection. Curr Opin Immunol.
17:366–373. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Drayton DL, Liao S, Mounzer RH and Ruddle
NH: Lymphoid organ development: From ontogeny to neogenesis. Nat
Immunol. 7:344–353. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Angeli V, Ginhoux F, Llodrà J, Quemeneur
L, Frenette PS, Skobe M, Jessberger R, Merad M and Randolph GJ: B
cell-driven lymphangiogenesis in inflamed lymph nodes enhances
dendritic cell mobilization. Immunity. 24:203–215. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bajénoff M, Egen JG, Koo LY, Laugier JP,
Brau F, Glaichenhaus N and Germain RN: Stromal cell networks
regulate lymphocyte entry, migration, and territoriality in lymph
nodes. Immunity. 25:989–1001. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Allan RS, Waithman J, Bedoui S, Jones CM,
Villadangos JA, Zhan Y, Lew AM, Shortman K, Heath WR and Carbone
FR: Migratory dendritic cells transfer antigen to a lymph
node-resident dendritic cell population for efficient CTL priming.
Immunity. 25:153–162. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Link A, Vogt TK, Favre S, Britschgi MR,
Acha-Orbea H, Hinz B, Cyster JG and Luther SA: Fibroblastic
reticular cells in lymph nodes regulate the homeostasis of naive T
cells. Nat Immunol. 8:1255–1265. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
14
|
Urban ML, Manenti L and Vaglio A:
Fibrosis-a common pathway to organ injury and failure. N Engl J
Med. 373:95–96. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Denton CP and Abraham DJ: Transforming
growth factor-beta and connective tissue growth factor: Key
cytokines in scleroderma pathogenesis. Curr Opin Rheumatol.
13:505–511. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li MO, Wan YY, Sanjabi S, Robertson AK and
Flavell RA: Transforming growth factor-beta regulation of immune
responses. Ann Rev Immunol. 24:99–146. 2006. View Article : Google Scholar
|
|
17
|
Kehrl JH, Roberts AB, Wakefield LM,
Jakowlew S, Sporn MB and Fauci AS: Transforming growth factor beta
is an important immunomodulatory protein for human B lymphocytes. J
Immunol. 137:3855–3860. 1986.PubMed/NCBI
|
|
18
|
Esplugues E, Sancho D, Vega-Ramos J,
Martínez C, Syrbe U, Hamann A, Engel P, Sánchez-Madrid F and
Lauzurica P: Enhanced antitumor immunity in mice deficient in CD69.
J Exp Med. 197:1093–1106. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gray JD, Hirokawa M, Ohtsuka K and Horwitz
DA: Generation of an inhibitory circuit involving CD8+ T cells,
IL-2, and NK cell-derived TGF-beta: Contrasting effects of anti-CD2
and anti-CD3. J Immunol. 160:2248–2254. 1998.PubMed/NCBI
|
|
20
|
Zhang W, He T, Wang Q, Li X, Wei J, Hou X,
Zhang B, Huang L and Wang L: Interleukin-1 receptor-associated
kinase-2 genetic variant rs708035 increases NF-κB activity through
promoting TRAF6 ubiquitination. J Biol Chem. 289:12507–12519. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Estes JD, Wietgrefe S, Schacker T,
Southern P, Beilman G, Reilly C, Milush JM, Lifson JD, Sodora DL,
Carlis JV and Haase AT: Simian immunodeficiency virus-induced
lymphatic tissue fibrosis is mediated by transforming growth factor
beta 1-positive regulatory T cells and begins in early infection. J
Infect Dis. 195:551–561. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Schacker TW, Nguyen PL, Beilman GJ,
Wolinsky S, Larson M, Reilly C and Haase AT: Collagen deposition in
HIV-1 infected lymphatic tissues and T cell homeostasis. J Clin
Invest. 110:1133–1139. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Diaz A, Alós L, León A, Mozos A, Caballero
M, Martinez A, Plana M, Gallart T, Gil C, Leal M, et al: Factors
associated with collagen deposition in lymphoid tissue in long-term
treated HIV-infected patients. AIDS. 24:2029–2039. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Diaz A, García F, Mozos A, Caballero M,
León A, Martinez A, Gil C, Plana M, Gallart T, Gatell JM and Alós
L: Lymphoid tissue collagen deposition in HIV-infected patients
correlates with the imbalance between matrix metalloproteinases and
their inhibitors. J Infect Dis. 203:810–813. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Estes JD: Role of collagen deposition in
lymphatic tissues and immune reconstruction during HIV-1 and SIV
infections. Current HIV/AIDS Rep. 6:29–35. 2009. View Article : Google Scholar
|
|
26
|
Schacker TW, Brenchley JM, Beilman GJ,
Reilly C, Pambuccian SE, Taylor J, Skarda D, Larson M, Douek DC and
Haase AT: Lymphatic tissue fibrosis is associated with reduced
numbers of naive CD4+ T cells in human immunodeficiency virus type
1 infection. Clin Vaccine Immunol. 13:556–560. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Nies-Kraske E, Schacker TW, Condoluci D,
Orenstein J, Brenchley J, Fox C, Daucher M, Dewar R, Urban E, Hill
B, et al: Evaluation of the pathogenesis of decreasing CD4(+) T
cell counts in human immunodeficiency virus type 1-infected
patients receiving successfully suppressive antiretroviral therapy.
J Infect Dis. 199:1648–1656. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zeng M, Smith AJ, Wietgrefe SW, Southern
PJ, Schacker TW, Reilly CS, Estes JD, Burton GF, Silvestri G,
Lifson JD, et al: Cumulative mechanisms of lymphoid tissue fibrosis
and T cell depletion in HIV-1 and SIV infections. J Clin Invest.
121:998–1008. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zeng M, Southern PJ, Reilly CS, Beilman
GJ, Chipman JG, Schacker TW and Haase AT: Lymphoid tissue damage in
HIV-1 infection depletes naive T cells and limits T cell
reconstitution after antiretroviral therapy. PLoS Pathog.
8:e10024372012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Estes JD, Haase AT and Schacker TW: The
role of collagen deposition in depleting CD4+ T cells and limiting
reconstitution in HIV-1 and SIV infections through damage to the
secondary lymphoid organ niche. Semin Immunol. 20:181–186. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Shull MM, Ormsby I, Kier AB, Pawlowski S,
Diebold RJ, Yin M, Allen R, Sidman C, Proetzel G, Calvin D, et al:
Targeted disruption of the mouse transforming growth factor-beta 1
gene results in multifocal inflammatory disease. Nature.
359:693–699. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kulkarni AB, Huh CG, Becker D, Geiser A,
Lyght M, Flanders KC, Roberts AB, Sporn MB, Ward JM and Karlsson S:
Transforming growth factor beta 1 null mutation in mice causes
excessive inflammatory response and early death. Proc Natl Acad Sci
USA. 90:770–774. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Furuya Y, Furuya AK, Roberts S, Sanfilippo
AM, Salmon SL and Metzger DW: Prevention of influenza virus-induced
immunopathology by TGF-β produced during allergic asthma. PLoS
Pathog. 11:e10051802015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Tang B, Böttinger EP, Jakowlew SB, Bagnall
KM, Mariano J, Anver MR, Letterio JJ and Wakefield LM: Transforming
growth factor-beta1 is a new form of tumor suppressor with true
haploid insufficiency. Nat Med. 4:802–807. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ziv E, Cauley J, Morin PA, Saiz R and
Browner WS: Association between the T29->C polymorphism in the
transforming growth factor beta1 gene and breast cancer among
elderly white women: The Study of Osteoporotic Fractures. JAMA.
285:2859–2863. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kushiyama Y, Fukuda R, Suetsugu H,
Kazumori H, Ishihara S, Adachi K and Kinoshita Y: Site-dependent
production of transforming growth factor beta1 in colonic mucosa:
Its possible role in tumorigenesis of the colon. J Lab Clin Med.
136:201–208. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Oh SP, Seki T, Goss KA, Imamura T, Yi Y,
Donahoe PK, Li L, Miyazono K, ten Dijke P, Kim S and Li E: Activin
receptor-like kinase 1 modulates transforming growth factor-beta 1
signaling in the regulation of angiogenesis. Proc Natl Acad Sci
USA. 97:2626–2631. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Estes JD: Pathobiology of
HIV/SIV-associated changes in secondary lymphoid tissues. Immunol
Rev. 254:65–77. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Lang KS, Recher M, Navarini AA, Harris NL,
Löhning M, Junt T, Probst HC, Hengartner H and Zinkernagel RM:
Inverse correlation between IL-7 receptor expression and CD8 T cell
exhaustion during persistent antigen stimulation. Eur J Immunol.
35:738–745. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Agata Y, Kawasaki A, Nishimura H, Ishida
Y, Tsubata T, Yagita H and Honjo T: Expression of the PD-1 antigen
on the surface of stimulated mouse T and B lymphocytes. Int
Immunol. 8:765–772. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Velu V, Titanji K, Zhu B, Husain S,
Pladevega A, Lai L, Vanderford TH, Chennareddi L, Silvestri G,
Freeman GJ, et al: Enhancing SIV-specific immunity in vivo by PD-1
blockade. Nature. 458:206–210. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Kim HJ, Verbinnen B, Tang X, Lu L and
Cantor H: Inhibition of follicular T-helper cells by CD8(+)
regulatory T cells is essential for self tolerance. Nature.
467:328–332. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Kim HJ, Barnitz RA, Kreslavsky T, Brown
FD, Moffett H, Lemieux ME, Kaygusuz Y, Meissner T, Holderried TA,
Chan S, et al: Stable inhibitory activity of regulatory T cells
requires the transcription factor Helios. Science. 350:334–339.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Kim HJ and Cantor H: Regulation of
self-tolerance by Qa-1-restricted CD8(+) regulatory T cells. Semin
Immunol. 23:446–452. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Lu L, Kim HJ, Werneck MB and Cantor H:
Regulation of CD8+ regulatory T cells: Interruption of the
NKG2A-Qa-1 interaction allows robust suppressive activity and
resolution of autoimmune disease. Proc Natl Acad Sci USA.
105:19420–19425. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Jiang H, Canfield SM, Gallagher MP, Jiang
HH, Jiang Y, Zheng Z and Chess L: HLA-E-restricted regulatory
CD8(+) T cells are involved in development and control of human
autoimmune type 1 diabetes. J Clin Invest. 120:3641–3650. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
He R, Hou S, Liu C, Zhang A, Bai Q, Han M,
Yang Y, Wei G, Shen T, Yang X, et al: Follicular CXCR5-expressing
CD8(+) T cells curtail chronic viral infection. Nature.
537:412–428. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Mylvaganam GH, Rios D, Abdelaal HM, Iyer
S, Tharp G, Mavigner M, Hicks S, Chahroudi A, Ahmed R, Bosinger SE,
et al: Dynamics of SIV-specific CXCR5+ CD8 T cells during chronic
SIV infection. Proc Natl Acad Sci USA. 114:1976–1981. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Fuschiotti P, Larregina AT, Ho J,
Feghali-Bostwick C and Medsger TA Jr: Interleukin-13-producing CD8+
T cells mediate dermal fibrosis in patients with systemic
sclerosis. Arthritis Rheum. 65:236–246. 2013. View Article : Google Scholar : PubMed/NCBI
|