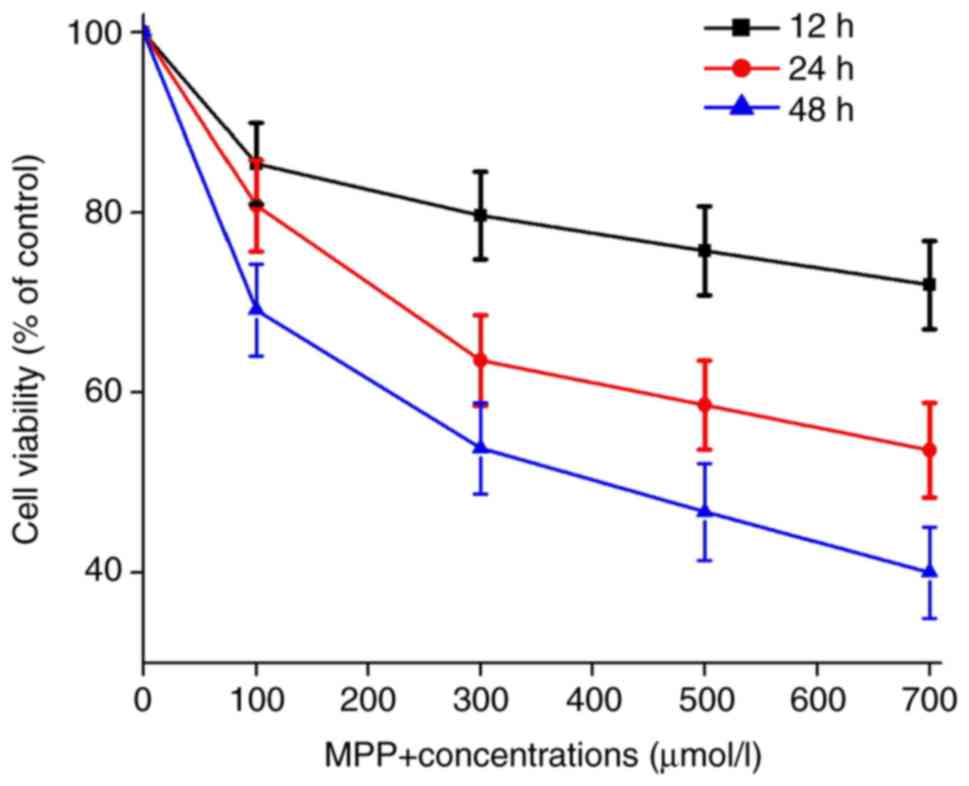
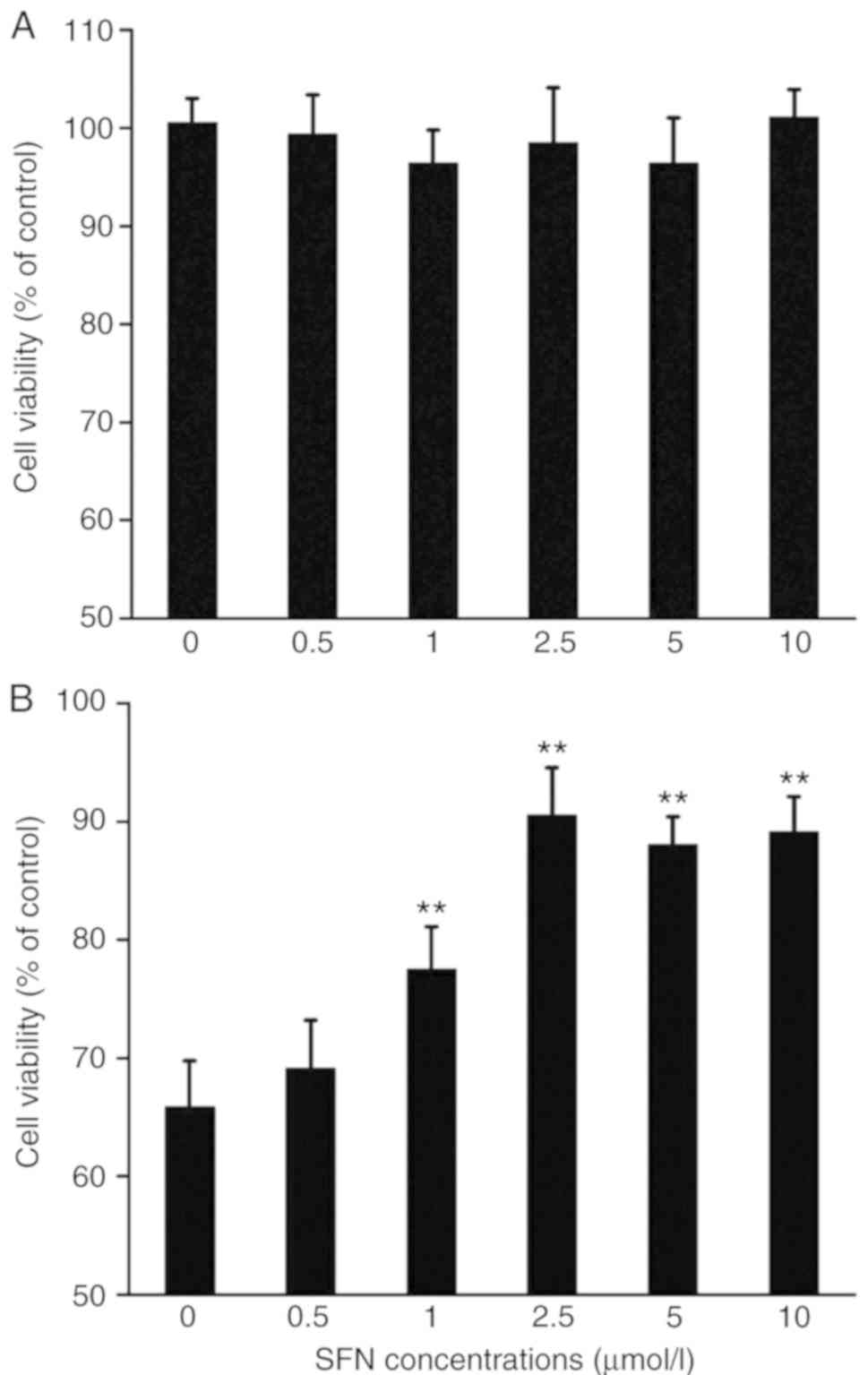
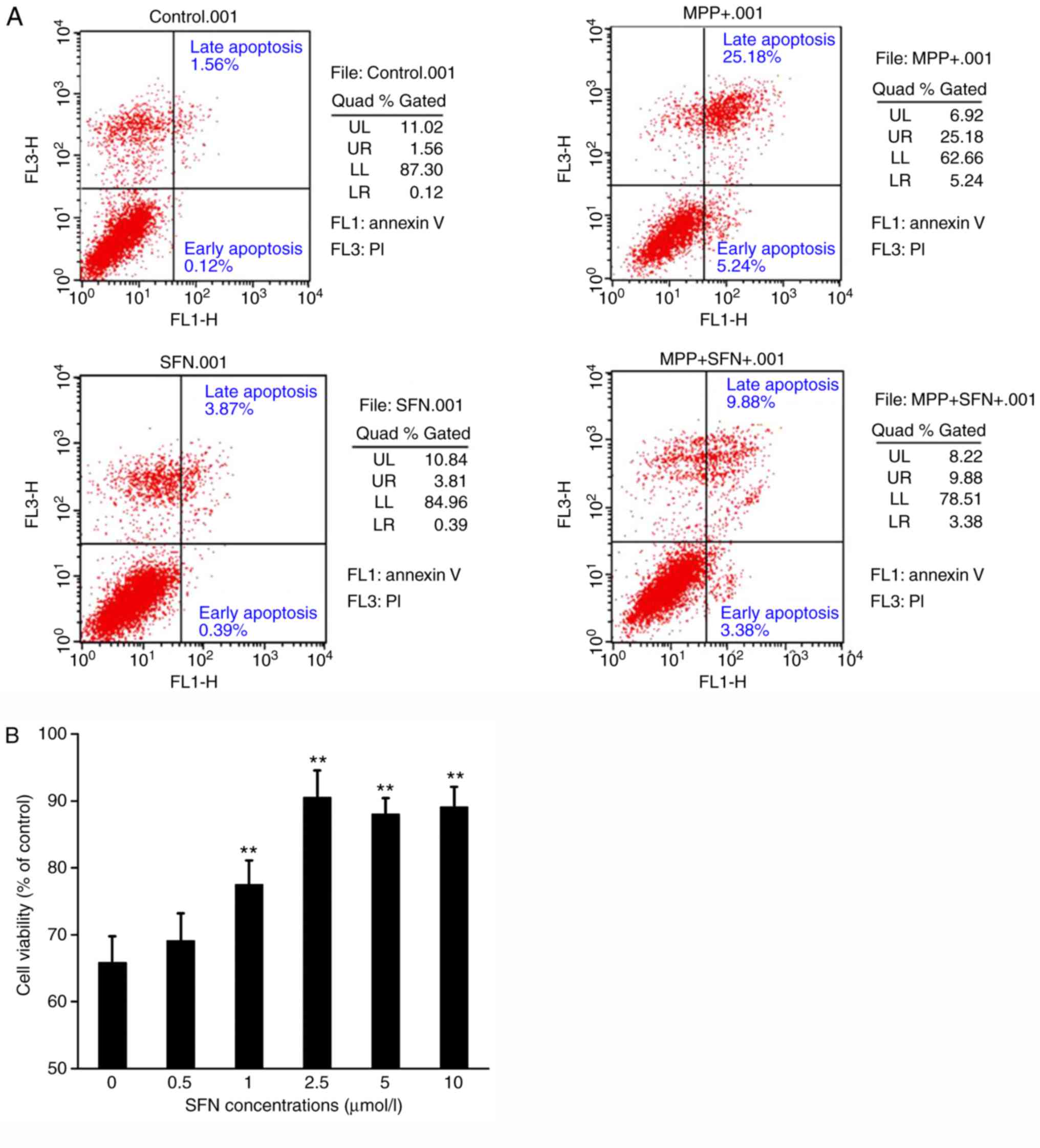
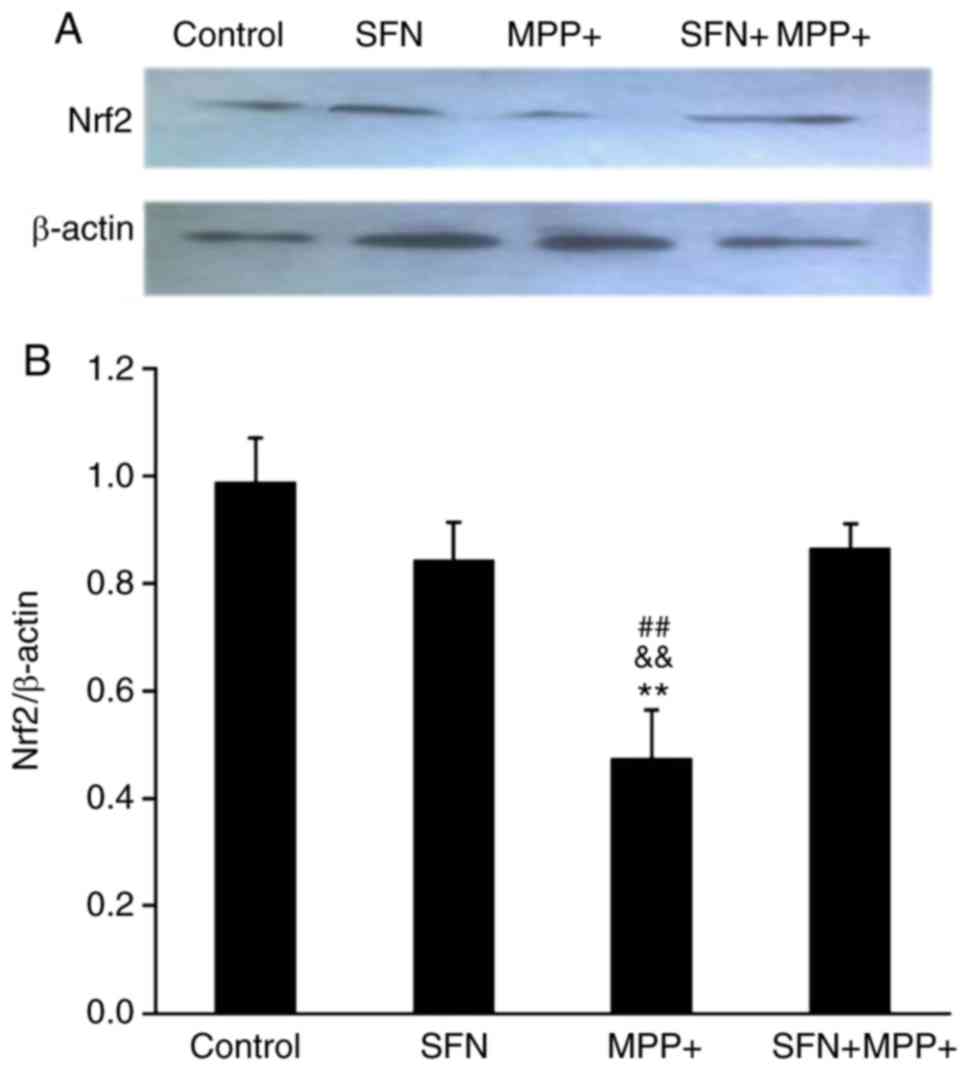
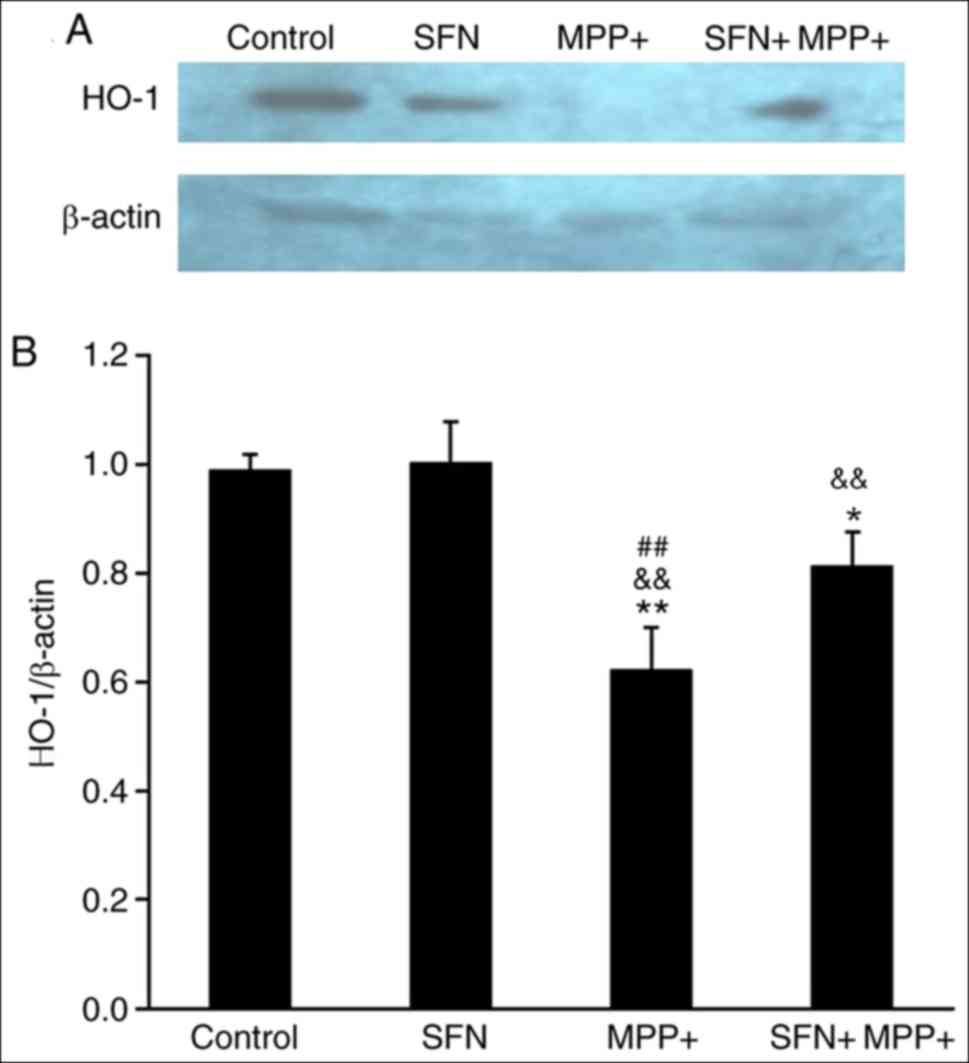
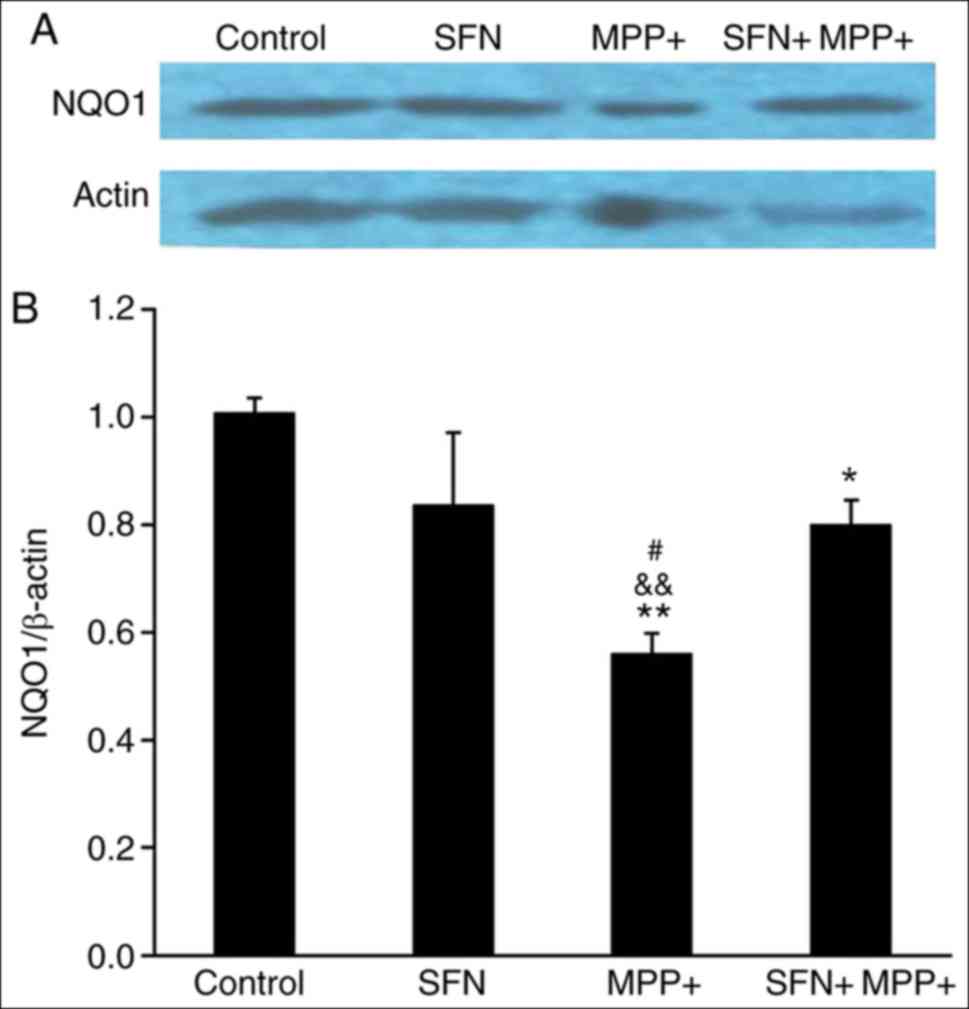
|
1
|
Dauer W and Przedborski S: Parkinson's
disease: Mechanisms and models. Neuron. 39:889–909. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Tolleson CM and Fang JY: Advances in the
mechanisms of Parkinson's disease. Discov Med. 15:61–66.
2013.PubMed/NCBI
|
|
3
|
Coyle JT and Puttfarcken P: Oxidative
stress, glutamate, and neurodegenerative disorders. Science.
262:689–695. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhou C, Huang Y and Przedborski S:
Oxidative stress in Parkinson's disease: A mechanism of pathogenic
and therapeutic significance. Ann N Y Acad Sci. 1147:93–104. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liu Z, Zhou T, Ziegler AC, Dimitrion P and
Zuo L: Oxidative stress in neurodegenerative diseases: From
molecular mechanisms to clinical applications. Oxid Med Cell
Longev. 2017:25259672017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lerin C, Rodgers JT, Kalume DE, Kim SH,
Pandey A and Puigserver P: GCN5 acetyltransferase complex controls
glucose metabolism through transcriptional repression of
PGC-1alpha. Cell Metab. 3:429–438. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Jha N, Kumar MJ, Boonplueang R and
Andersen JK: Glutathione decreases in dopaminergic PC12 cells
interfere with the ubiquitin protein degradation pathway: Relevance
for Parkinson's disease? J Neurochem. 80:555–561. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang JJ, Zhang T, Niu DB, Wang K, Li KR,
Xue B and Wang XM: Doxycycline-regulated co-expression of GDNF and
TH in PC12 cells. Neurosci Lett. 401:142–145. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Liu Y, Liu L, Ying XX, Wei WJ, Han C, Liu
Y, Han CH, Leng AJ, Ma JY and Liu J: Dried rehmannia root protects
against glutamate-induced cytotoxity to PC12 cells through energy
metabolism-related pathways. Neural Regen Res. 12:1338–1346. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Soane L, Li Dai W, Fiskum G and Bambrick
LL: Sulforaphane protects immature hippocampal neurons against
death caused by exposure to hemin or to oxygen and glucose
deprivation. J Neurosci Res. 88:1355–1363. 2010.PubMed/NCBI
|
|
11
|
Xu C, Huang MT, Shen G, Yuan X, Lin W,
Khor TO, Conney AH and Kong AN: Inhibition of
7,12-dimethylbenz(a)anthracene-induced skin tumorigenesis in
C57BL/6 mice by sulforaphane is mediated by nuclear factor
E2-related factor 2. Cancer Res. 66:8293–8296. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fisher ML, Ciavattone N, Grun D, Adhikary
G and Eckert RL: Sulforaphane reduces YAP/ΔNp63α signaling to
reduce cancer stem cell survival and tumor formation. Oncotarget.
8:73407–73418. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kubo E, Chhunchha B, Singh P, Sasaki H and
Singh DP: Sulforaphane reactivates cellular antioxidant defense by
inducing Nrf2/ARE/Prdx6 activity during aging and oxidative stress.
Sci Rep. 7:141302017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Thejass P and Kuttan G: Immunomodulatory
activity of sulforaphane, a naturally occurring isothiocyanate from
broccoli (Brassica oleracea). Phytomedicine. 14:538–545. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Greaney AJ, Maier NK, Leppla SH and
Moayeri M: Sulforaphane inhibits multiple inflammasomes through an
Nrf2-independent mechanism. J Leukoc Biol. 99:189–199. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Dwivedi S, Rajasekar N, Hanif K, Nath C
and Shukla R: Sulforaphane ameliorates okadaic acid-induced memory
impairment in rats by activating the Nrf2/HO-1 antioxidant pathway.
Mol Neurobiol. 53:5310–5323. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Jang M and Cho IH: Sulforaphane
ameliorates 3-nitropropionic acid-induced striatal toxicity by
activating the keap1-Nrf2-ARE pathway and inhibiting the MAPKs and
NF-κB pathways. Mol Neurobiol. 53:2619–2635. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Minelli A, Conte C, Cacciatore I,
Cornacchia C and Pinnen F: Molecular mechanism underlying the
cerebral effect of Gly-Pro-Glu tripeptide bound to L-dopa in a
Parkinson's animal model. Amino Acids. 43:1359–1367. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen PC, Vargas MR, Pani AK, Smeyne RJ,
Johnson DA, Kan YW and Johnson JA: Nrf2-mediated neuroprotection in
the MPTP mouse model of Parkinson's disease: Critical role for the
astrocyte. Proc Natl Acad Sci USA. 106:2933–2938. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yin XP, Chen ZY, Zhou J, Wu D and Bao B:
Mechanisms underlying the perifocal neuroprotective effect of the
Nrf2-ARE signaling pathway after intracranial hemorrhage. Drug Des
Devel Ther. 9:5973–5986. 2015.PubMed/NCBI
|
|
21
|
Yin XP, Wu D, Zhou J, Chen ZY, Bao B and
Xie L: Heme oxygenase 1 plays role of neuron-protection by
regulating Nrf2-ARE signaling post intracerebral hemorrhage. Int J
Clin Exp Pathol. 8:10156–10163. 2015.PubMed/NCBI
|
|
22
|
Chen G, Fang Q, Zhang J, Zhou D and Wang
Z: Role of the Nrf2-ARE pathway in early brain injury after
experimental subarachnoid hemorrhage. J Neurosci Res. 89:515–523.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Rostamian Delavar M, Baghi M, Safaeinejad
Z, Kiani-Esfahani A, Ghaedi K and Nasr-Esfahani MH: Differential
expression of miR-34a, miR-141, and miR-9 in MPP+-treated
differentiated PC12 cells as a model of Parkinson's disease. Gene.
662:54–65. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Cheng W, Chen W, Wang P and Chu J: Asiatic
acid protects differentiated PC12 cells from
Aβ25-35-induced apoptosis and tau hyperphosphorylation
via regulating PI3K/Akt/GSK-3β signaling. Life Sci. 208:96–101.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yin X, Zhang X, Wang W, Chang L, Jiang Y
and Zhang S: Perihematoma damage at different time points in
experimental intracerebral hemorrhage. J Huazhong Univ Sci
Technolog Med Sci. 26:59–62. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yin M, Chen Z, Ouyang Y, Zhang H, Wan Z,
Wang H, Wu W and Yin X: Thrombin-induced, TNFR-dependent miR-181c
downregulation promotes MLL1 and NF-κB target gene expression in
human microglia. J Neuroinflammation. 14:1322017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kajimura Y, Aoki T, Kuramochi K, Kobayashi
S, Sugawara F, Watanabe N and Arai T: Neoechinulin A protects PC12
cells against MPP+-induced cytotoxicity. J Antibiot (Tokyo).
61:330–333. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lu XL, Lin YH, Wu Q, Su FJ, Ye CH, Shi L,
He BX, Huang FW, Pei Z and Yao XL: Paeonolum protects against
MPP(+)-induced neurotoxicity in zebrafish and PC12 cells. BMC
Complement Altern Med. 15:1372015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Houghton CA, Fassett RG and Coombes JS:
Sulforaphane and other nutrigenomic Nrf2 activators: Can the
clinician's expectation be matched by the reality? Oxid Med Cell
Longev. 2016:78571862016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Calkins MJ, Johnson DA, Townsend JA,
Vargas MR, Dowell JA, Williamson TP, Kraft AD, Lee JM, Li J and
Johnson JA: The Nrf2/ARE pathway as a potential therapeutic target
in neurodegenerative disease. Antioxid Redox Signal. 11:497–508.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Gilgun-Sherki Y, Rosenbaum Z, Melamed E
and Offen D: Antioxidant therapy in acute central nervous system
injury: Current state. Pharmacol Rev. 54:271–284. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hartley A, Stone JM, Heron C, Cooper JM
and Schapira AH: Complex I inhibitors induce dose-dependent
apoptosis in PC12 cells: Relevance to Parkinson's disease. J
Neurochem. 63:1987–1990. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhang L, Huang L, Li X, Liu C, Sun X, Wu
L, Li T, Yang H and Chen J: Potential molecular mechanisms
mediating the protective effects of tetrahydroxystilbene glucoside
on MPP+-induced PC12 cell apoptosis. Mol Cell Biochem.
436:203–213. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ye S, Koon HK, Fan W, Xu Y, Wei W, Xu C
and Cai J: Effect of a traditional chinese herbal medicine
formulation on cell survival and apoptosis of
MPP+-treated MES 23.5 dopaminergic cells. Parkinsons
Dis. 2017:47642122017.PubMed/NCBI
|
|
35
|
Zhang Z, Li C, Shang L, Zhang Y, Zou R,
Zhan Y and Bi B: Sulforaphane induces apoptosis and inhibits
invasion in U251MG glioblastoma cells. Springerplus. 5:2352016.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Żuryń A, Litwiniec A, Safiejko-Mroczka B,
Klimaszewska- Wiśniewska A, Gagat M, Krajewski A, Gackowska L and
Grzanka D: The effect of sulforaphane on the cell cycle, apoptosis
and expression of cyclin D1 and p21 in the A549 non-small cell lung
cancer cell line. Int J Oncol. 48:2521–2533. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Pledgie-Tracy A, Sobolewski MD and
Davidson NE: Sulforaphane induces cell type-specific apoptosis in
human breast cancer cell lines. Mol Cancer Ther. 6:1013–1021. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
An YW, Jhang KA, Woo SY, Kang JL and Chong
YH: Sulforaphane exerts its anti-inflammatory effect against
amyloid-β peptide via STAT-1 dephosphorylation and activation of
Nrf2/HO-1 cascade in human THP-1 macrophages. Neurobiol Aging.
38:1–10. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Pearson BL, Simon JM, McCoy ES, Salazar G,
Fragola G and Zylka MJ: Identification of chemicals that mimic
transcriptional changes associated with autism, brain aging and
neurodegeneration. Nat Commun. 7:111732016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Chirumbolo S and Bjørklund G: Sulforaphane
and 5-fluorouracil synergistically inducing autophagy in breast
cancer: A possible role for the Nrf2-Keap1-ARE signaling? Food Chem
Toxicol. 2018. View Article : Google Scholar
|
|
41
|
Wang F, Wang W, Li J, Zhang J, Wang X and
Wang M: Sulforaphane reverses gefitinib tolerance in human lung
cancer cells via modulation of sonic hedgehog signaling. Oncol
Lett. 15:109–114. 2018.PubMed/NCBI
|
|
42
|
Ramirez CN, Li W, Zhang C, Wu R, Su S,
Wang C, Gao L, Yin R and Kong AN: In vitro-in vivo dose response of
ursolic acid, sulforaphane, PEITC, and curcumin in cancer
prevention. AAPS J. 20:192017. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Evans PC: The influence of sulforaphane on
vascular health and its relevance to nutritional approaches to
prevent cardiovascular disease. EPMA J. 2:9–14. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Nagata N, Xu L, Kohno S, Ushida Y, Aoki Y,
Umeda R, Fuke N, Zhuge F, Ni Y, Nagashimada M, et al: Glucoraphanin
ameliorates obesity and insulin resistance through adipose tissue
browning and reduction of metabolic endotoxemia in mice. Diabetes.
66:1222–1236. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Brown RH, Reynolds C, Brooker A, Talalay P
and Fahey JW: Sulforaphane improves the bronchoprotective response
in asthmatics through Nrf2-mediated gene pathways. Respir Res.
16:1062015. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Xin Y, Bai Y, Jiang X, Zhou S, Wang Y,
Wintergerst KA, Cui T, Ji H, Tan Y and Cai L: Sulforaphane prevents
angiotensin II-induced cardiomyopathy by activation of Nrf2 via
stimulating the Akt/GSK-3ß/Fyn pathway. Redox Biol. 15:405–417.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Stepniewski J, Pacholczak T, Skrzypczyk A,
Ciesla M, Szade A, Szade K, Bidanel R, Langrzyk A, Grochowski R,
Vandermeeren F, et al: Heme oxygenase-1 affects generation and
spontaneous cardiac differentiation of induced pluripotent stem
cells. IUBMB Life. 70:129–142. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Dinkova-Kostova AT, Fahey JW, Kostov RV
and Kensler TW: KEAP1 and done? targeting the NRF2 pathway with
sulforaphane. Trends Food Sci Technol. 69:257–269. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Izumi Y, Kataoka H, Inose Y, Akaike A,
Koyama Y and Kume T: Neuroprotective effect of an Nrf2-ARE
activator identified from a chemical library on dopaminergic
neurons. Eur J Pharmacol. 818:470–479. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Cho JH, Kim YW and Keum YS: Sulforaphane
suppresses LPS-induced or TPA-induced downregulation of PDCD4 in
RAW 264.7 cells. Phytother Res. 28:1606–1611. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Ma Q: Role of nrf2 in oxidative stress and
toxicity. Annu Rev Pharmacol Toxicol. 53:401–426. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Cho HY, Jedlicka AE, Reddy SP, Kensler TW,
Yamamoto M, Zhang LY and Kleeberger SR: Role of NRF2 in protection
against hyperoxic lung injury in mice. Am J Respir Cell Mol Biol.
26:175–182. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Mastrangelo L, Cassidy A, Mulholland F,
Wang W and Bao Y: Serotonin receptors, novel targets of
sulforaphane identified by proteomic analysis in Caco-2 cells.
Cancer Res. 68:5487–5491. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Lee S and Kim J, Seo SG, Choi BR, Han JS,
Lee KW and Kim J: Sulforaphane alleviates scopolamine-induced
memory impairment in mice. Pharmacol Res. 85:23–32. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Chinaglia G, Alvarez FJ, Probst A and
Palacios JM: Mesostriatal and mesolimbic dopamine uptake binding
sites are reduced in Parkinson's disease and progressive
supranuclear palsy: A quantitative autoradiographic study using
[3H]mazindol. Neuroscience. 49:317–327. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Leenders KL, Salmon EP, Tyrrell P, Perani
D, Brooks DJ, Sager H, Jones T, Marsden CD and Frackowiak RS: The
nigrostriatal dopaminergic system assessed in vivo by positron
emission tomography in healthy volunteer subjects and patients with
Parkinson's disease. Arch Neurol. 47:1290–1298. 1990. View Article : Google Scholar : PubMed/NCBI
|