Introduction
Gastric carcinoma remains one of the most common and
lethal types of malignancy worldwide (1). Although patients receive radical surgery
and chemotherapy, recurrence and metastasis remain a challenge for
this type of malignancy (2).
Therefore, the identification of novel therapeutic targets and
prognostic factors for patients with gastric cancer is
required.
Kinases represent a group of promising therapeutic
targets for cancers. Identification of novel specific kinase
targets and prognostic factors may not only expand upon the options
of treatments for patients with gastric cancer, but also provide
classifiers to discriminate subgroups that may benefit from kinase
inhibitors (3).
The microtubule-associated serine/threonine kinase
like (Mastl) gene encodes a kinase that promotes mitotic
progression and cell cycle re-entry following DNA damage (4,5). A
previous study demonstrated that Mastl was commonly overexpressed
in a number of types of cancer, including breast and oral cancer
(6). Furthermore, Mastl upregulation
was associated with recurrence following initial cancer therapy and
decreased patient survival in a number of cancer types (6,7). These
previous studies indicated that Mastl may be a novel therapeutic
target for cancer. In spite of this, the expression of Mastl in
patients with gastric cancer has not been investigated and limited
information is known about the mechanisms of Mastl with respect to
tumor progression. In the present study, it was hypothesized that
Mastl may induce epithelial to mesenchymal transition (EMT) and
consequently promote tumor migration and metastasis. Therefore, the
present study aimed to investigate the expression level of Mastl
and EMT-associated proteins in a cohort of patients with gastric
cancer, and to evaluate the associated clinical significance and
correlation.
Materials and methods
Patients and tissue specimens
The present study was approved by the Ethics
Committee of Shandong Cancer Hospital and Institute (Jinan, China).
A cohort of 152 tumor node metastasis (TNM) (8) stage II/III patients with gastric cancer
(84 male, 68 female; median age 52 years, range 28–75 years), who
underwent gastrectomy with lymph node dissection between July 2006
and December 2010, were included in this retrospective study. TNM
staging, histology and margins were classified or defined according
to the Chinese guidelines for diagnosis and treatment of gastric
cancer (2011 edition) (8). The
following exclusion criteria were predefined: i) Patients with
R1/R2 margins (as defined by the aforementioned guidelines); ii)
patients who succumbed during the peri-operative period; and iii)
patients receiving preoperative chemotherapy or radiotherapy.
Finally, a total of 126 patients with advanced disease (where
metastasis or recurrence occurred during the follow-up period)
received postoperative platinum-based chemotherapy with or without
radiotherapy were included in the present study following the
exclusion of 26 cases. All patients were followed up subsequent to
surgery. Examinations for recurrence or metastasis were performed
using serum tumor marker assays, including carcinoembryonic
antigen, and diagnostic imaging (computed tomography,
ultrasonography or magnetic resonance imaging) every 3 months for
the initial 2 postoperative years and subsequently every 6 months.
The median follow-up time was 60 months (range, 1–111 months).
Patients who experienced distant metastases or recurrence during
follow-up received oxaliplatin (85 mg/m2, every 2
weeks), docetaxel (75 mg/m2, every 3 weeks) or
irinotecan (150–180 mg/m2, every 2 weeks) single agent
chemotherapy, or treatment in combination with cetuximab (500
mg/m2, every 2 weeks) or bevacizumab (5 mg/kg, every 2
weeks) regimens (8).
Immunohistochemistry (IHC)
Immunohistochemical staining was performed to
determine the protein expression of Mastl, Vimentin and epithelial
(E-)cadherin in 126 gastric cancer tissues. Briefly, 4-µm 10%
formalin-fixed (1 day at room temperature) paraffin embedded
sections were dewaxed in xylene and antigen was retrieved by
heating the sections (95°C for 30 min) in 10 mmol/l citrate
solution (pH 6.0) for 30 min. After blocking the endogenous
peroxidase activity, using 3% H2O2 (10 min at
room temperature), anti-Mastl (#15739-1-AP, ProteinTech Group,
Inc., Chicago, IL, USA), anti-Vimentin (#10366-1-AP, ProteinTech
Group, Inc.) and anti-E-Cadherin (#20874-1-AP, ProteinTech Group,
Inc.) (all dilution at 1:100) antibodies were incubated with the
sections overnight at 4°C. Subsequently, sections were washed and
treated with biotinylated horseradish peroxidase-labeled anti
rabbit secondary antibody (#sc-2357, Santa Cruz Biotechnology,
Inc., Dallas, TX, USA, 1:100) at 37°C for 30 min. Diaminobenzene
was used to detect chromogen and hematoxylin was used as the
nuclear counterstain at room temperature for 1 min, and staining
was observed under light microscope (magnification, ×40).
Non-specific rabbit immunoglobulin G (#sc-2027, at 1:100 dilution,
Santa Cruz Biotechnology, Inc.) was used to replace the primary
antibodies in immunohistostaining as negative controls.
Evaluation of immunohistochemical
staining
The immunostaining was scored by two independent
senior pathologists who were unaware of the patients' clinical
information. For Mastl staining, an immunoreactive score (IRS) was
derived by addition of the intensity score (0, negative to weak; 1,
positive; and 2, strongly positive) and the density score (0, 0–25%
of tumor area stained; 1, 26–50; 2, 51–75; and 3, 76–100%) for
statistical analysis with respect to each patient. An IRS score of
≥4 was defined as high expression and <4 was defined as low
expression, as described previously (9). The membranous E-cadherin and cytoplasmic
vimentin expression was defined according to previously published
guidelines (10). The slides were
graded as follows: Grade 1, 0–25% staining; grade 2, 26–50%
staining; and grade 3, >50% staining. According to previous
criteria (11), patients with
vimentin or E-cadherin expression of greater than or equal to grade
1 were classified as exhibiting positive EMT status, while the
other patients were defined as the wild-type group.
Statistical analysis
The χ2 test or Fisher's exact test was
used to evaluate the association between Mastl expression, the
clinicopathological variables and EMT status. Kaplan-Meier
estimator curves were plotted using a log-rank test for univariable
analysis of overall survival and differences between the groups
were evaluated using the log-rank test. Multivariable Cox's
proportional hazards regression models were used to assess the
prognostic significance of Mastl expression and of several accepted
prognostic factors. Statistical analysis was performed using the
GraphPad Prism 5.0 software package (GraphPad Software, Inc., San
Diego, USA). P<0.05 was considered to indicate a statistically
significant difference.
Results
Table I lists the
clinicopathological characteristics of the 126 patients with
gastric cancer patients included in the present study. First, the
Mastl expression level in gastric cancer and adjacent normal
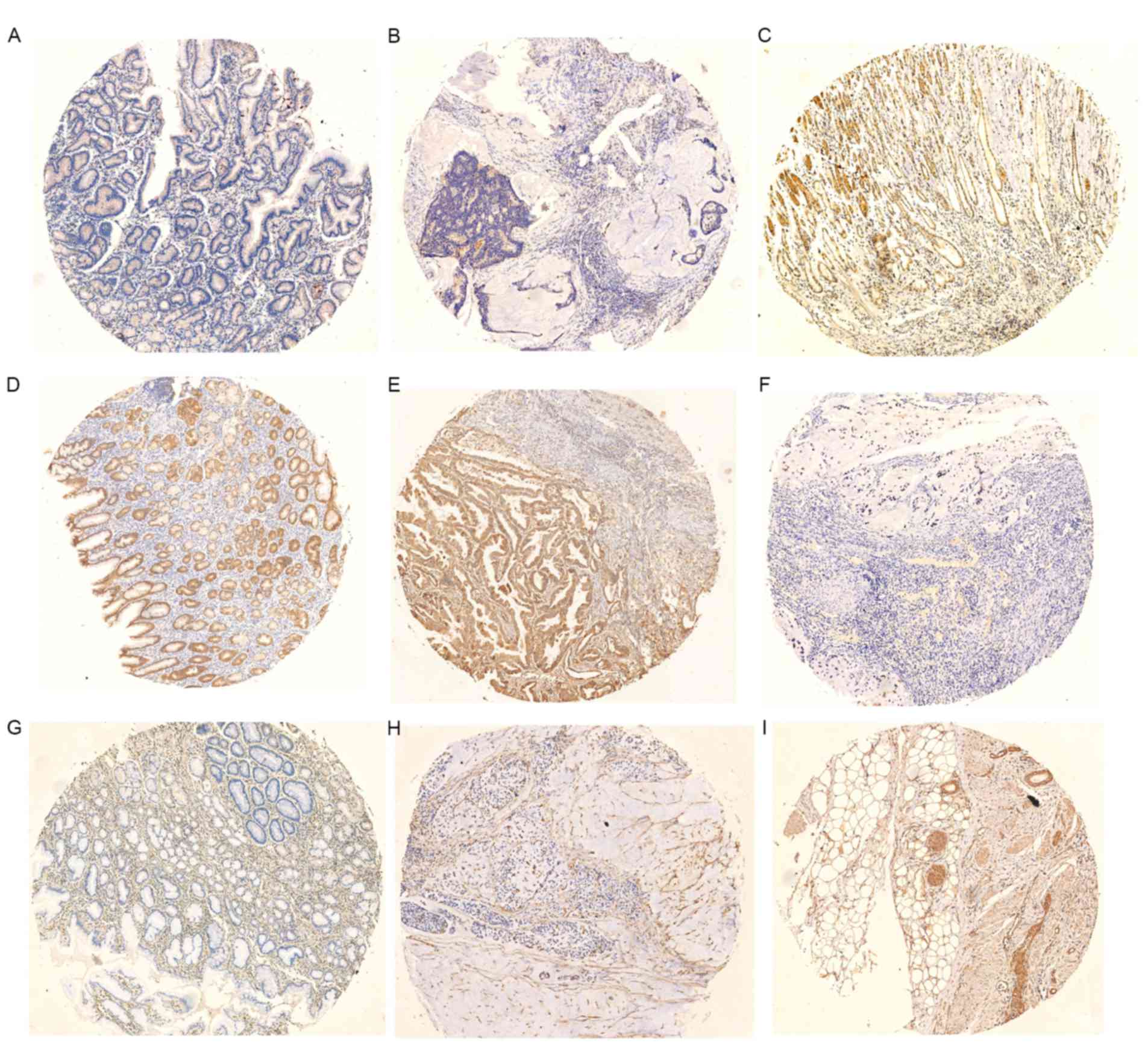
gastric tissues was analyzed using IHC. The representative images
of immunohistochemical staining are presented in Fig. 1. Mastl immunoreactivity was primarily
within the membranes and cytoplasm of the gastric cancer cells. The
majority of the stroma or normal gastric cells exhibited negative
staining, in spite of sporadic positive staining on these cells.
According to the IHC analysis, the patients were separated into two
groups: High Mastl expression (IRS≥4) and low Mastl expression
(IRS<4). Furthermore, 54/126 (42.9%) cases exhibited high
expression of Mastl in the gastric cancer tissues.
 | Table I.Association between Mastl expression
and clinicopathological features. |
Table I.
Association between Mastl expression
and clinicopathological features.
|
| Mastl expression |
|
|---|
|
|
|
|
|---|
| Prognostic
factor | Low | High | P-value |
|---|
| Age, years |
|
| 0.634 |
|
<60 | 20 | 18 |
|
| ≥60 | 52 | 36 |
|
| Sex |
|
| 0.938 |
| Male | 42 | 31 |
|
|
Female | 30 | 23 |
|
| Pathology type |
|
| 0.362 |
|
Differentiated | 52 | 34 |
|
|
Undifferentiated | 20 | 20 |
|
| Tumor
sizea |
|
| 0.501 |
| T2–3 | 51 | 42 |
|
| T4 | 21 | 12 |
|
| Lymph
nodea |
|
| 0.013 |
| N0 | 42 | 21 |
|
| N1 | 27 | 23 |
|
| N2 | 3 | 10 |
|
| Recurrence |
|
| 0.016 |
|
Negative | 58 | 32 |
|
|
Positive | 14 | 22 |
|
| EMT status |
|
| 0.029 |
|
Wild-type | 60 | 35 |
|
|
Undergoing EMT | 12 | 19 |
|
The association between Mastl protein expression and
the clinicopathological characteristics of gastric cancer was
subsequently determined. This analysis revealed that increased
expression of Mastl was positively associated with lymph node
metastasis (P=0.013) and tumor recurrence (P=0.016). However, no
association between Mastl expression and other clinicopathological
features was observed (Table I).
As seen in Fig. 1,
Positive E-cadherin staining was observed in normal and part of the
malignant gastric epithelium, and vimentin staining was observed in
all the stroma and part of the cancerous epithelium. On the basis
of the IHC results of Vimentin and E-cadherin and the
aforementioned previously published criteria (11), the patients were separated into two
groups: EMT type and wild-type. A total of 31/126 (24.6%) cases
were defined as the EMT type in the gastric cancer cohort.
Increased Mastl expression was identified to be significantly
associated with EMT status (P=0.029).
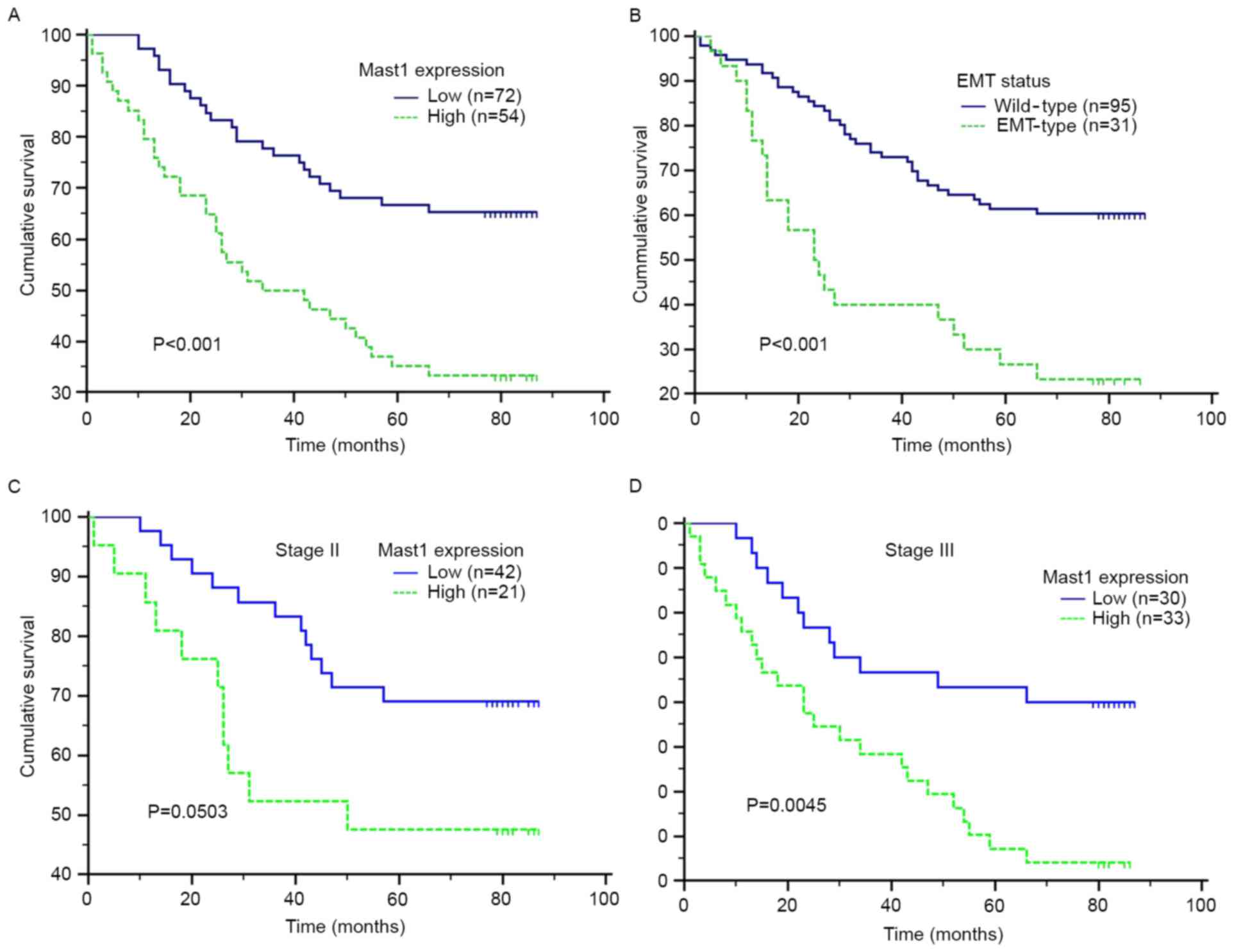
The patients with high Mastl expression exhibited a
significantly decreased overall survival (OS) time compared with
that of the patients with low Mastl expression [hazard ratio (HR),
2.591; 95% confidence interval (CI), 1.535–4.374; log-rank test,
P<0.001] (Fig. 2; Table II). Furthermore, a survival analysis
was performed according to the level of Mastl expression in subsets
of patients with gastric cancer at different clinical stages. The
results demonstrated that high expression of Mastl was a prognostic
factor for the patients with stage III disease (HR, 2.590; 95% CI,
1.355–4.953; log-rank test, P=0.0045) (Fig. 2). However, Mastl expression levels
failed to retain prognostic significance for the patients at stage
II (HR, 2.1810; 95% CI, 0.8943–5.3190; log-rank test, P=0.0503)
(Fig. 2).
 | Table II.Prognostic factors on univariable and
multivariable Cox proportional hazards regression models for
overall survival in stage II and III colorectal cancer
patients. |
Table II.
Prognostic factors on univariable and
multivariable Cox proportional hazards regression models for
overall survival in stage II and III colorectal cancer
patients.
|
| Univariable | Multivariable |
|---|
|
|
|
|
|---|
| Prognostic
factor | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| Age, years |
|
|
|
|
|
|
| <60
vs. ≥60 | 1.557 | 0.908–2.671 | 0.141 |
|
|
|
| Sex |
|
|
|
|
|
|
| Male vs.
female | 0.756 | 0.455–1.249 | 0.281 |
|
|
|
| Pathology type |
|
|
|
|
|
|
|
Undifferentiated vs.
differentiated | 1.638 | 0.930–2.882 | 0.058 |
|
|
|
| Tumor size |
|
|
|
|
|
|
| T4 vs.
T2-3 | 1.299 | 0.724–2.332 | 0.347 |
|
|
|
| Lymph node |
|
|
|
|
| 0.011 |
| Positive
vs. negative | 1.826 | 1.104–3.022 | 0.019 | 1.681 | 1.130–2.501 |
|
| Mastl expression |
|
|
|
|
| 0.010 |
| High vs.
low | 2.591 | 1.535–4.374 | <0.001 | 2.006 | 1.176–3.420 |
|
| EMT status |
|
|
|
|
| <0.001 |
|
Undergoing EMT vs.
wild-type | 2.893 | 1.490–5.617 | <0.001 | 2.605 | 1.533–4.426 |
|
Clinical characteristics were included in the
univariate analysis to assess their impact on the survival of
patients with gastric cancer; positive lymph node metastasis,
underlying EMT status and high Mastl expression were identified to
be prognostic factors, but were not associated with each other. On
multivariable analysis adjusted for established clinical prognostic
factors, high Mastl expression was a prognostic factor of OS time
(HR, 2.006; 95% CI, 1.176–3.420; P=0.010), as was the presence of
lymph node metastasis (HR, 1.681; 95% CI, 1.130–2.501; P=0.011) and
EMT status (HR, 2.605; 95% CI, 1.533–4.426; P<0.001).
Discussion
The clinical significance of Mastl expression has
been investigated only in oral and breast cancer (6). To the best of our knowledge, the present
study was the first to clinically evaluate the expression of Mast1
and its significance in gastric cancer. The results of the present
study identified that Mastl protein was upregulated in the gastric
cancer tissues, relative to the normal adjacent non-cancerous
tissues, which was consistent with the results of a previous study
of oral cancer tissues and healthy controls (6). Furthermore, analysis of gastric cancer
tissues revealed a significant association between Mastl expression
with aggressive characteristics and poor patient survival, which is
consistent with the results of a previous study of breast cancer
(7). Therefore, the present study
revealed Mastl as a candidate oncogene and a potential prognostic
factor in cancer, and Mastl-positive gastric cancer may require
intensive treatments.
Mastl-knockdown in recurrent tumor cells may
re-sensitize their response to chemotherapy and radiotherapy in
vitro and in vivo, indicating that Mastl is a novel
therapeutic target for tumor recurrence (6). In vitro studies have indicated a
number of possible mechanisms for this effect, including Mastl
acting as a cyclin-dependent kinase dephosphorylation inhibitor and
a regulator of the DNA damage response (12,5).
However, the association between Mastl and tumor recurrence remains
unknown. In the present study, it was validated that, in gastric
cancer samples, Mastl was associated with EMT status. Previous
studies have identified that EMT is involved in tumor recurrence
and the resistance to initial cancer therapy (13,14), and
the results of the present study support this.
Polo-like kinase 1 (Plk1), aurora kinases and Mastl
represent a group of promising serine/threonine kinase targets for
cancer treatment. Inhibitors of Plk1 and aurora kinases are
currently under clinical development for cancer therapy (15,16). On
the basis of the biological and the clinical significance of Mastl
in the present and previous studies, it has been hypothesized that
Mastl may sensitize the response to initial cancer therapy in
gastric cancer, thus decreasing the incidence of tumor recurrence.
Additionally, potential Mast1 inhibitors deserve future
investigation.
To the best of our knowledge, the results of the
present study were the first to indicate that Mastl expression is
associated with EMT status, tumor recurrence and poor survival in
gastric cancer. The expression of Mastl, as determined using IHC,
may serve as an independent predictor of clinical outcome for
patients with gastric cancer. The results of the present study
suggested that Mastl may serve as a promising therapeutic target
for gastric cancer. Additional studies are required to investigate
the clinical potential of Mastl inhibition for gastric cancer
therapy.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2016. CA Cancer J Clin. 66:7–30. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yang L: Incidence and mortality of gastric
cancer in China. World J Gastroenterol. 12:17–20. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Fleuren ED, Zhang L, Wu J and Daly RJ: The
kinome ‘at large’ in cancer. Nat Rev Cancer. 16:83–98. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Álvarez-Fernández M, Sánchez-Martínez R,
Sanz-Castillo B, Gan PP, Sanz-Flores M, Trakala M, Ruiz-Torres M,
Lorca T, Castro A and Malumbres M: Greatwall is essential to
prevent mitotic collapse after nuclear envelope breakdown in
mammals. Proc Natl Acad Sci USA. 110:pp. 17374–17379. 2013,
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Peng A, Yamamoto TM, Goldberg ML and
Maller JL: A novel role for greatwall kinase in recovery from DNA
damage. Cell Cycle. 9:4364–4369. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang L, Luong VQ, Giannini PJ and Peng A:
Mastl kinase, a promising therapeutic target, promotes cancer
recurrence. Oncotarget. 5:11479–11489. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Dahlhaus M, Burkovski A, Hertwig F, Mussel
C, Volland R, Fischer M, Debatin KM, Kestler HA and Beltinger C:
Boolean modeling identifies Greatwall/MASTL as an important
regulator in the AURKA network of neuroblastoma. Cancer Lett.
371:79–89. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ji J: Chinese guidelines for diagnosis and
treatment of gastric cancer (2011 edition). Transl Gastrointest
Cancer. 1:103–114. 2012.
|
|
9
|
Remmele W and Stegner HE: Recommendation
for uniform definition of an immunoreactive score (IRS) for
immunohistochemical estrogen receptor detection (ER-ICA) in breast
cancer tissue. Pathologe. 8:138–140. 1987.(In German). PubMed/NCBI
|
|
10
|
Yamada S, Fuchs BC, Fujii T, Shimoyama Y,
Sugimoto H, Nomoto S, Takeda S, Tanabe KK, Kodera Y and Nakao A:
Epithelial-to-mesenchymal transition predicts prognosis of
pancreatic cancer. Surgery. 154:946–954. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yang MH, Hsu DS, Wang HW, Wang HJ, Lan HY,
Yang WH, Huang CH, Kao SY, Tzeng CH, Tai SK, et al: Bmi1 is
essential in Twist1-induced epithelial-mesenchymal transition. Nat
Cell Biol. 12:982–992. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Manchado E, Guillamot M, de Cárcer G,
Eguren M, Trickey M, García-Higuera I, Moreno S, Yamano H, Cañamero
M and Malumbres M: Targeting mitotic exit leads to tumor regression
in vivo: Modulation by Cdk1, Mastl, and the PP2A/B55α,δ
phosphatase. Cancer Cell. 18:641–654. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Smith BN and Bhowmick NA: Role of EMT in
metastasis and therapy resistance. J Clin Med. 5:pii: E17. 2016.
View Article : Google Scholar
|
|
14
|
Mitra A, Mishra L and Li S: EMT, CTCs and
CSCs in tumor relapse and drug-resistance. Oncotarget.
6:10697–10711. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Frost A, Mross K, Steinbild S, Hedbom S,
Unger C, Kaiser R, Trommeshauser D and Munzert G: Phase i study of
the Plk1 inhibitor BI 2536 administered intravenously on three
consecutive days in advanced solid tumours. Curr Oncol. 19:e28–e35.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Traynor AM, Hewitt M, Liu G, Flaherty KT,
Clark J, Freedman SJ, Scott BB, Leighton AM, Watson PA, Zhao B, et
al: Phase I dose escalation study of MK-0457, a novel Aurora kinase
inhibitor, in adult patients with advanced solid tumors. Cancer
Chemother Pharmacol. 67:305–314. 2011. View Article : Google Scholar : PubMed/NCBI
|