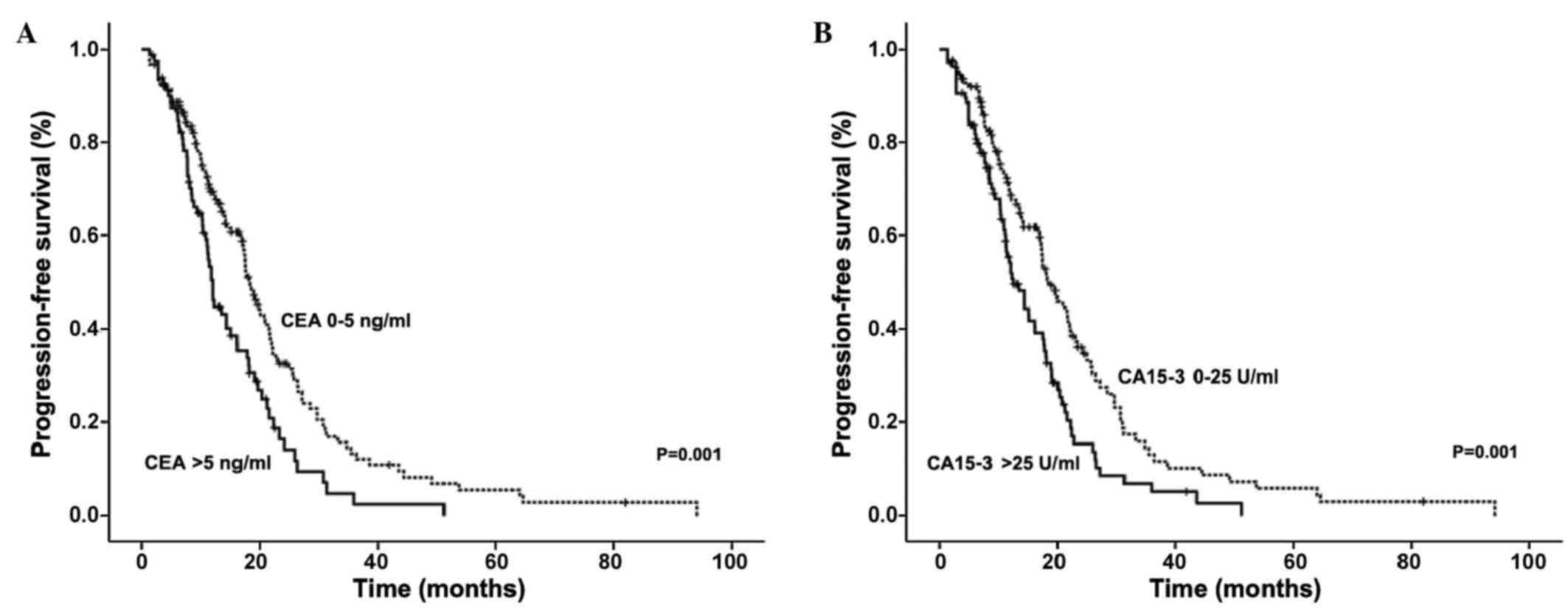
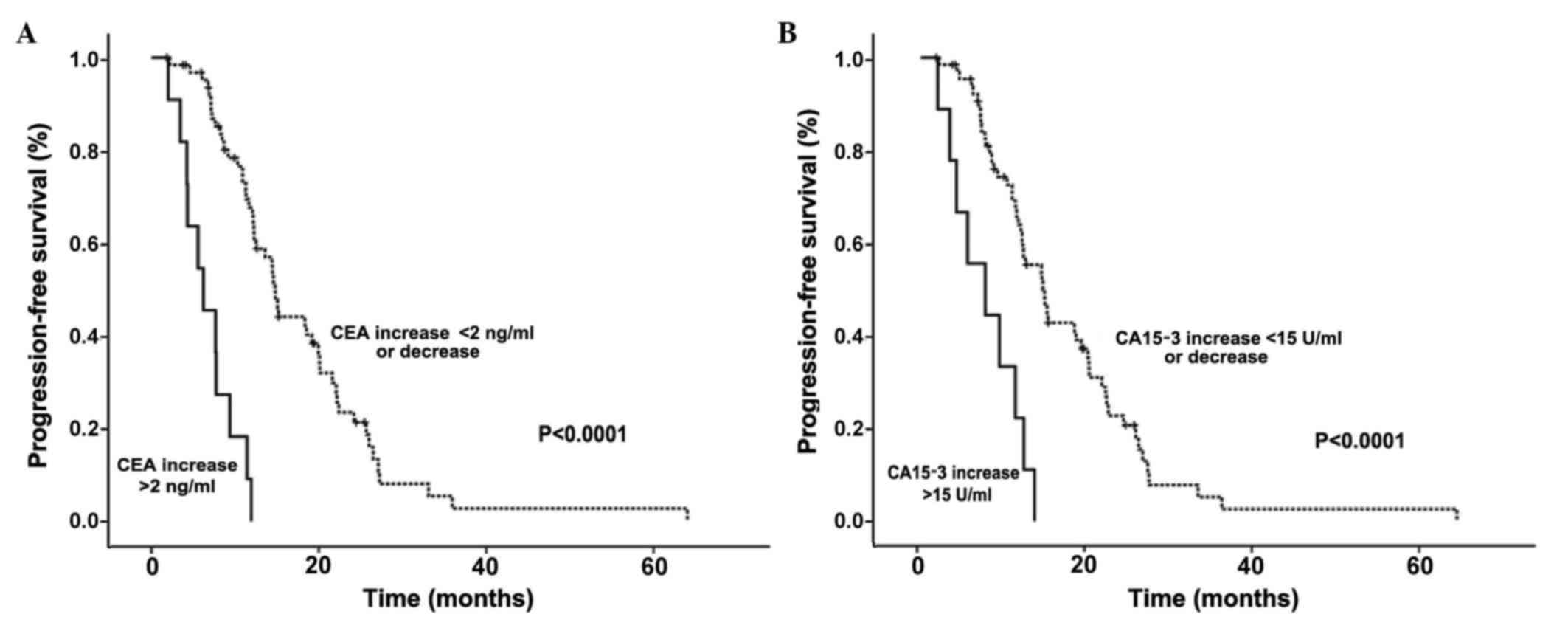
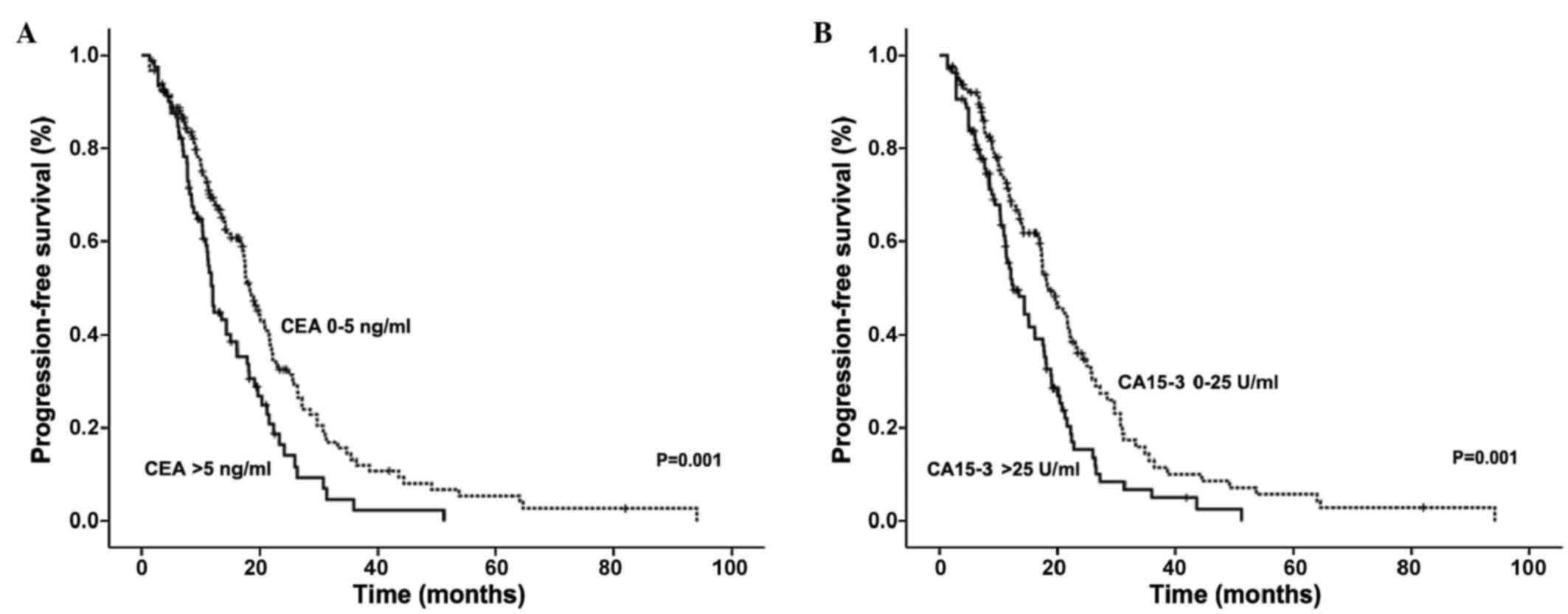
|
1
|
Siegel R, DeSantis C, Virgo K, Stein K,
Mariotto A, Smith T, Cooper D, Gansler T, Lerro C, Fedewa S, et al:
Cancer treatment and survivorship statistics, 2012. CA Cancer J
Clin. 62:220–241. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Esteva FJ, Valero V, Pusztai L,
Boehnke-Michaud L, Buzdar AU and Hortobagyi GN: Chemotherapy of
metastatic breast cancer: What to expect in 2001 and beyond.
Oncologist. 6:133–146. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Robertson JF, Jaeger W, Syzmendera JJ,
Selby C, Coleman R, Howell A, Winstanley J, Jonssen PE, Bombardieri
E, Sainsbury JR, et al: European Group for Serum Tumour Markers in
Breast Cancer: The objective measurement of remission and
progression in metastatic breast cancer by use of serum tumour
markers. Eur J Cancer. 35:47–53. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Watanabe H, Okada M, Kaji Y, Satouchi M,
Sato Y, Yamabe Y, Onaya H, Endo M, Sone M and Arai Y: New response
evaluation criteria in solid tumours-revised RECIST guideline
(version 1.1). Gan To Kagaku Ryoho. 36:2495–2501. 2009.(In
Japanese). PubMed/NCBI
|
|
5
|
Clamp A, Danson S, Nguyen H, Cole D and
Clemons M: Assessment of therapeutic response in patients with
metastatic bone disease. Lancet Oncol. 5:607–616. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Stafford SE, Gralow JR, Schubert EK, Rinn
KJ, Dunnwald LK, Livingston RB and Mankoff DA: Use of serial FDG
PET to measure the response of bone-dominant breast cancer to
therapy. Acad Radiol. 9:913–921. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Costa L, Demers LM, Gouveia-Oliveira A,
Schaller J, Costa EB, de Moura MC and Lipton A: Prospective
evaluation of the peptide-bound collagen type I cross-links
N-telopeptide and C-telopeptide in predicting bone metastases
status. J Clin Oncol. 20:850–856. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lipton A, Demers L, Curley E, Chinchilli
V, Gaydos L, Hortobagyi G, Theriault R, Clemens D, Costa L, Seaman
J, et al: Markers of bone resorption in patients treated with
pamidronate. Eur J Cancer. 34:2021–2026. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Massacesi C, Rocchi MB, Marcucci F, Pilone
A, Galeazzi M and Bonsignori M: Serum tumor markers may precede
instrumental response to chemotherapy in patients with metastatic
cancer. Int J Biol Markers. 18:295–300. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Molina R, Auge JM, Farrus B, Zanón G,
Pahisa J, Muñoz M, Torne A, Filella X, Escudero JM, Fernandez P, et
al: Prospective evaluation of carcinoembryonic antigen (CEA) and
carbohydrate antigen 15.3 (CA 15.3) in patients with primary
locoregional breast cancer. Clin Chem. 56:1148–1157. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Duffy MJ: Serum tumor markers in breast
cancer: Are they of clinical value? Clin Chem. 52:345–351. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zheng H and Luo RC: Diagnostic value of
combined detection of TPS, CA153 and CEA in breast cancer. Di Yi
Jun Yi Da Xue Xue Bao. 25(1293–1294): 12982005.(In Chinese).
|
|
13
|
Ebeling FG, Stieber P, Untch M, Nagel D,
Konecny GE, Schmitt UM, Fateh-Moghadam A and Seidel D: Serum CEA
and CA 15-3 as prognostic factors in primary breast cancer. Br J
Cancer. 86:1217–1222. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Marić P, Ozretić P, Levanat S, Oresković
S, Antunac K and Beketić-Oreskovic L: Tumor markers in breast
cancer - evaluation of their clinical usefulness. Coll Antropol.
35:241–247. 2011.
|
|
15
|
Agrawal AK, Jelen M, Rudnicki J,
Grzebieniak Z, Zyśko D, Kielan W, Słonina J and Marek G: The
importance of preoperative elevated serum levels of CEA and CA15-3
in patients with breast cancer in predicting its histological type.
Folia Histochem Cytobiol. 48:26–29. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Parker C: Active surveillance: Towards a
new paradigm in the management of early prostate cancer. Lancet
Oncol. 5:101–106. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Di Gioia D, Heinemann V, Nagel D, Untch M,
Kahlert S, Bauerfeind I, Koehnke T and Stieber P: Kinetics of CEA
and CA15-3 correlate with treatment response in patients undergoing
chemotherapy for metastatic breast cancer (MBC). Tumour Biol.
32:777–785. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mariani L, Miceli R, Michilin S and Gion
M: Serial determination of CEA and CA 15.3 in breast cancer
follow-up: An assessment of their diagnostic accuracy for the
detection of tumour recurrences. Biomarkers. 14:130–136. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Harris L, Fritsche H, Mennel R, Norton L,
Ravdin P, Taube S, Somerfield MR, Hayes DF and Bast RC Jr: American
Society of Clinical Oncology: American Society of Clinical Oncology
2007 update of recommendations for the use of tumor markers in
breast cancer. J Clin Oncol. 25:5287–5312. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Williams MR, Turkes A, Pearson D,
Griffiths K and Blamey RW: An objective biochemical assessment of
therapeutic response in metastatic breast cancer: A study with
external review of clinical data. Br J Cancer. 61:126–132. 1990.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Robertson JF, Pearson D, Price MR, Selby
C, Blamey RW and Howell A: Objective measurement of therapeutic
response in breast cancer using tumour markers. Br J Cancer.
64:757–763. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dixon AR, Jackson L, Chan SY, Badley RA
and Blamey RW: Continuous chemotherapy in responsive metastatic
breast cancer: A role for tumour markers? Br J Cancer. 68:181–185.
1993. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kurebayashi J, Nishimura R, Tanaka K,
Kohno N, Kurosumi M, Moriya T, Ogawa Y and Taguchi T: Significance
of serum tumor markers in monitoring advanced breast cancer
patients treated with systemic therapy: A prospective study. Breast
Cancer. 11:389–395. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hashimoto K, Yonemori K, Katsumata N,
Shimizu C, Hirakawa A, Hirata T, Kouno T, Tamura K, Ando M and
Fujiwara Y: Prediction of progressive disease using tumor markers
in metastatic breast cancer patients without target lesions in
first-line chemotherapy. Ann Oncol. 21:2195–2200. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sölétormos G, Nielsen D, Schiøler V,
Mouridsen H and Dombernowsky P: Monitoring different stages of
breast cancer using tumour markers CA 15-3, CEA and TPA. Eur J
Cancer. 40:481–486. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Schroeder JA, Thompson MC, Gardner MM and
Gendler SJ: Transgenic MUC1 interacts with epidermal growth factor
receptor and correlates with mitogen-activated protein kinase
activation in the mouse mammary gland. J Biol Chem.
276:13057–13064. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tanaka M, Kitajima Y, Sato S and Miyazaki
K: Combined evaluation of mucin antigen and E-cadherin expression
may help select patients with gastric cancer suitable for minimally
invasive therapy. Br J Surg. 90:95–101. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Rahn JJ, Dabbagh L, Pasdar M and Hugh JC:
The importance of MUC1 cellular localization in patients with
breast carcinoma: An immunohistologic study of 71 patients and
review of the literature. Cancer. 91:1973–1982. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Cheng JP, Yan Y, Wang XY, Lu YL, Yuan YH,
Jia J and Ren J: MUC1-positive circulating tumor cells and MUC1
protein predict chemotherapeutic efficacy in the treatment of
metastatic breast cancer. Chin J Cancer. 30:54–61. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Sölétormos G, Nielsen D, Schiøler V,
Skovsgaard T and Dombernowsky P: Tumor markers cancer antigen 15.3,
carcinoembryonic antigen, and tissue polypeptide antigen for
monitoring metastatic breast cancer during first-line chemotherapy
and follow-up. Clin Chem. 42:564–575. 1996.PubMed/NCBI
|
|
31
|
Cheung KL, Graves CR and Robertson JF:
Tumour marker measurements in the diagnosis and monitoring of
breast cancer. Cancer Treat Rev. 26:91–102. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Tondini C, Hayes DF, Gelman R, Henderson
IC and Kufe DW: Comparison of CA15-3 and carcinoembryonic antigen
in monitoring the clinical course of patients with metastatic
breast cancer. Cancer Res. 48:4107–4112. 1988.PubMed/NCBI
|
|
33
|
Lee JS, Park S, Park JM, Cho JH, Kim SI
and Park BW: Elevated levels of serum tumor markers CA 15-3 and CEA
are prognostic factors for diagnosis of metastatic breast cancers.
Breast Cancer Res Treat. 141:477–484. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yerushalmi R, Tyldesley S, Kennecke H,
Speers C, Woods R, Knight B and Gelmon KA: Tumor markers in
metastatic breast cancer subtypes: Frequency of elevation and
correlation with outcome. Ann Oncol. 23:338–345. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Bidard FC, Hajage D, Bachelot T, Delaloge
S, Brain E, Campone M, Cottu P, Beuzeboc P, Rolland E, Mathiot C,
et al: Assessment of circulating tumor cells and serum markers for
progression-free survival prediction in metastatic breast cancer: A
prospective observational study. Breast Cancer Res. 14:R292012.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Nishimura R, Nagao K, Miyayama H, Matsuda
M, Baba K, Matsuoka Y and Yamashita H: Elevated serum CA15-3 levels
correlate with positive estrogen receptor and initial favorable
outcome in patients who died from recurrent breast cancer. Breast
Cancer. 10:220–227. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Sørlie T, Perou CM, Tibshirani R, Aas T,
Geisler S, Johnsen H, Hastie T, Eisen MB, van de Rijn M, Jeffrey
SS, et al: Gene expression patterns of breast carcinomas
distinguish tumor subclasses with clinical implications. Proc Natl
Acad Sci USA. 98:pp. 10869–10874. 2001, View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Nakshatri H, Srour EF and Badve S: Breast
cancer stem cells and intrinsic subtypes: Controversies rage on.
Curr Stem Cell Res Ther. 4:50–60. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Molina R, Augé JM, Escudero JM, Filella X,
Zanon G, Pahisa J, Farrus B, Muñoz M and Velasco M: Evaluation of
tumor markers (HER-2/neu oncoprotein, CEA, and CA 15.3) in patients
with locoregional breast cancer: Prognostic value. Tumour Biol.
31:171–180. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Pedersen AC, Sorensen PD, Jacobsen EH,
Madsen JS and Brandslund I: Sensitivity of CA 15-3, CEA and serum
HER2 in the early detection of recurrence of breast cancer. Clin
Chem Lab Med. 51:1511–1519. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Bao H, Yu D, Wang J, Qiu T, Yang J and
Wang L: Predictive value of serum anti-p53 antibodies,
carcino-embryonic antigen, carbohydrate antigen 15-3, estrogen
receptor, progesterone receptor and human epidermal growth factor
receptor-2 in taxane-based and anthracycline-based neoadjuvant
chemotherapy in locally advanced breast cancer patients. Anticancer
Drugs. 19:317–323. 2008. View Article : Google Scholar : PubMed/NCBI
|