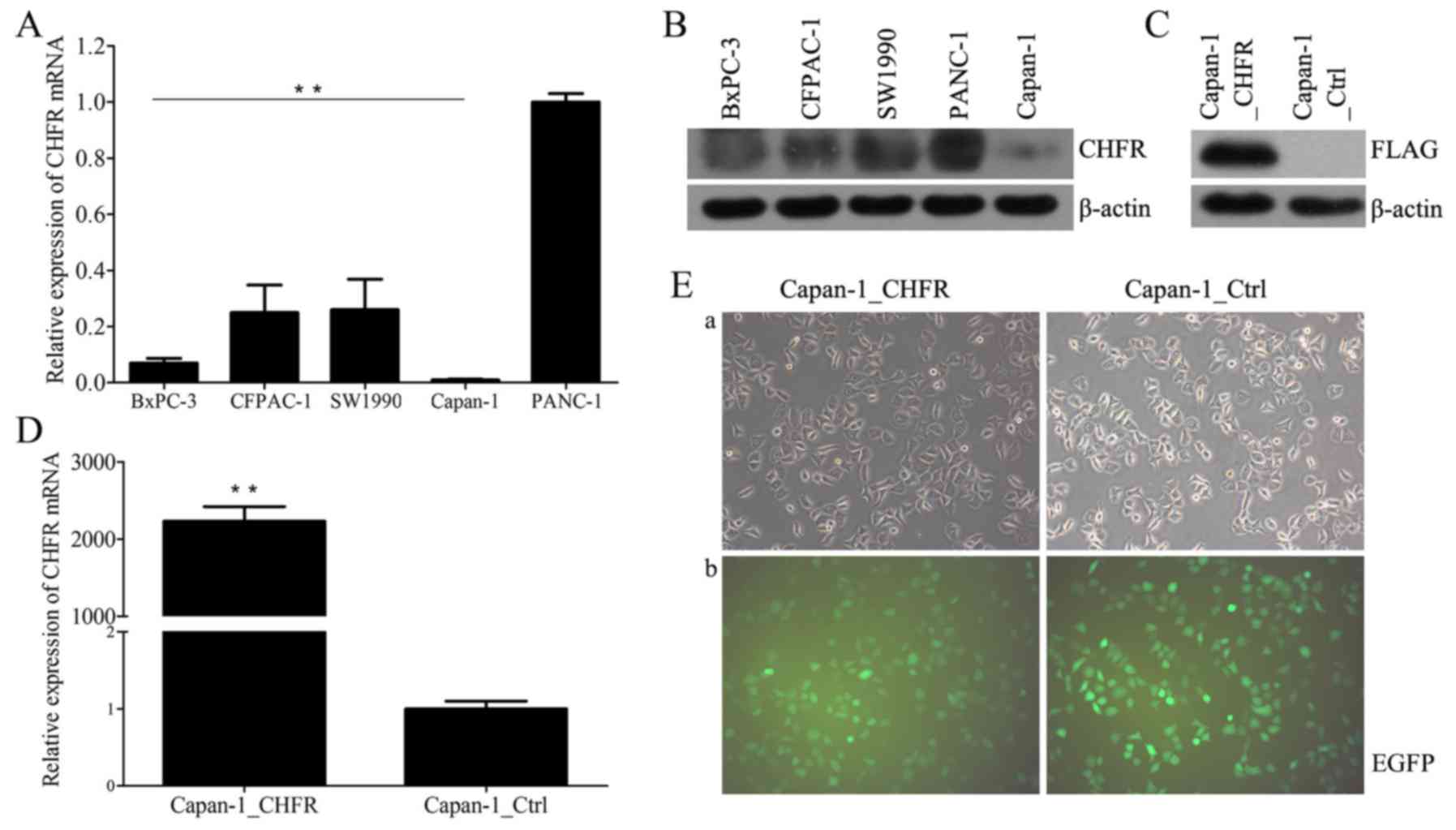
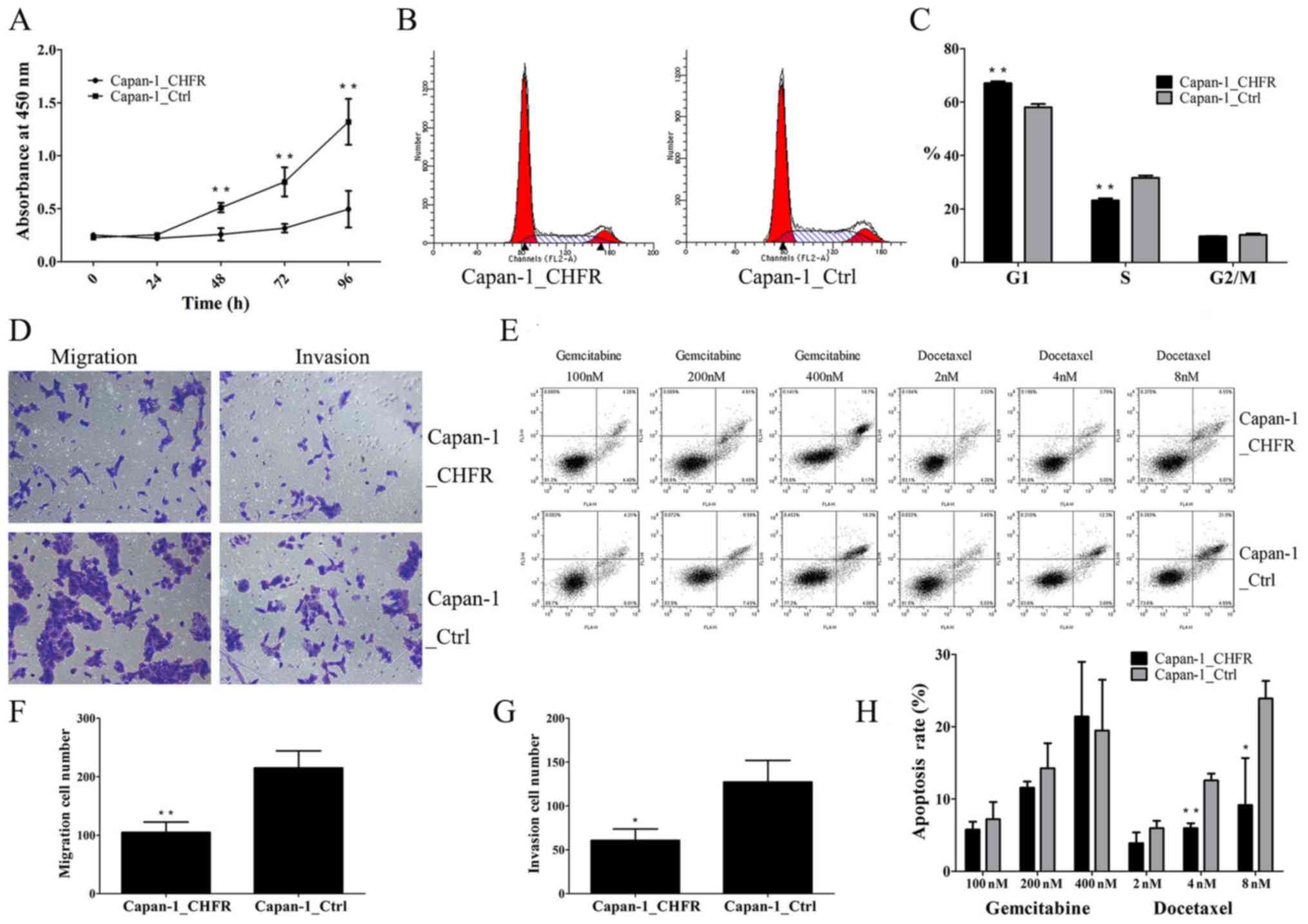
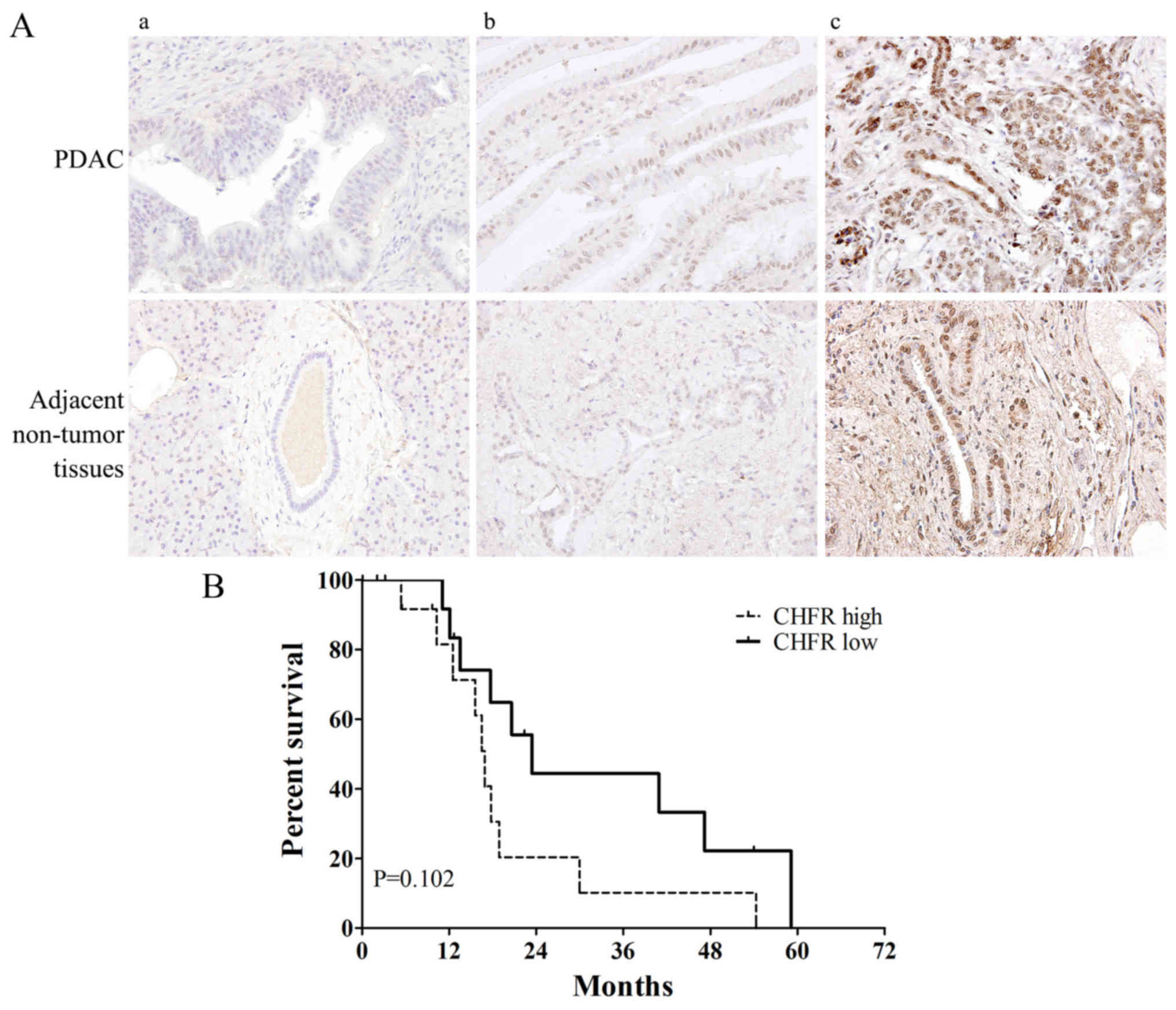
|
1
|
Ilic M and Ilic I: Epidemiology of
pancreatic cancer: World. J Gastroenterol. 22:9694–9705. 2016.
|
|
2
|
Pannala R, Leirness JB, Bamlet WR, Basu A,
Petersen GM and Chari ST: Prevalence and clinical profile of
pancreatic cancer-associated diabetes mellitus. Gastroenterology.
134:981–987. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Greenhalf W, Grocock C, Harcus M and
Neoptolemos J: Screening of high-risk families for pancreatic
cancer. Pancreatology. 9:215–222. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Shin EJ and Canto MI: Pancreatic cancer
screening. Gastroenterol Clin North Am. 41:143–157. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Scolnick DM and Halazonetis TD: Chfr
defines a mitotic stress checkpoint that delays entry into
metaphase. Nature. 406:430–435. 2000. View
Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hofmann K and Bucher P: The FHA domain: A
putative nuclear signalling domain found in protein kinases and
transcription factors. Trends Biochem Sci. 20:347–349. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lovering R, Hanson IM, Borden KL, Martin
S, O'Reilly NJ, Evan GI, Rahman D, Pappin DJ, Trowsdale J and
Freemont PS: Identification and preliminary characterization of a
protein motif related to the zinc finger. Proc Natl Acad Sci USA.
90:pp. 2112–2116. 1993, View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Borden KL, Boddy MN, Lally J, O'Reilly NJ,
Martin S, Howe K, Solomon E and Freemont PS: The solution structure
of the RING finger domain from the acute promyelocytic leukaemia
proto-oncoprotein PML. EMBO J. 14:1532–1541. 1995.PubMed/NCBI
|
|
9
|
Durocher D and Jackson SP: The FHA domain.
FEBS Lett. 513:58–66. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yu X, Minter-Dykhouse K, Malureanu L, Zhao
WM, Zhang D, Merkle CJ, Ward IM, Saya H, Fang G, van Deursen J and
Chen J: Chfr is required for tumor suppression and aurora a
regulation. Nat Genet. 37:401–406. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Oh YM, Kwon YE, Kim JM, Bae SJ, Lee BK,
Yoo SJ, Chung CH, Deshaies RJ and Seol JH: Chfr is linked to tumour
metastasis through the downregulation of HDAC1. Nat Cell Biol.
11:295–302. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bertholon J, Wang Q, Falette N, Verny C,
Auclair J, Chassot C, Navarro C, Saurin JC and Puisieux A: Chfr
inactivation is not associated to chromosomal instability in colon
cancers. Oncogene. 22:8956–8960. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yoshida K, Hamai Y, Suzuki T, Sanada Y,
Oue N and Yasui W: DNA methylation of CHFR is not a predictor of
the response to docetaxel and paclitaxel in advanced and recurrent
gastric cancer. Anticancer Res. 26:49–54. 2006.PubMed/NCBI
|
|
14
|
Yanokura M, Banno K, Kawaguchi M, Hirao N,
Hirasawa A, Susumu N, Tsukazaki K and Aoki D: Relationship of
aberrant DNA hypermethylation of CHFR with sensitivity to taxanes
in endometrial cancer. Oncol Rep. 17:41–48. 2007.PubMed/NCBI
|
|
15
|
Pillai RN, Brodie SA, Sica GL, Shaojin Y,
Li G, Nickleach DC, Yuan L, Varma VA, Bonta D, Herman JG, et al:
CHFR protein expression predicts outcomes to taxane-based first
line therapy in metastatic NSCLC. Clin Cancer Res. 19:1603–1611.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ketefian S: Ethical considerations in
research. Focus on vulnerable groups. Invest Educ Enferm.
33:164–172. 2015.PubMed/NCBI
|
|
18
|
Kobayashi C, Oda Y, Takahira T, Izumi T,
Kawaguchi K, Yamamoto H, Tamiya S, Yamada T, Iwamoto Y and
Tsuneyoshi M: Aberrant expression of CHFR in malignant peripheral
nerve sheath tumors. Mod Pathol. 19:524–532. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chin CF and Yeong FM: Safeguarding entry
into mitosis: The antephase checkpoint. Mol Cell Biol. 30:22–32.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bothos J, Summers MK, Venere M, Scolnick
DM and Halazonetis TD: The Chfr mitotic checkpoint protein
functions with Ubc13-Mms2 to form Lys63-linked polyubiquitin
chains. Oncogene. 22:7101–7107. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chaturvedi P, Sudakin V, Bobiak ML, Fisher
PW, Mattern MR, Jablonski SA, Hurle MR, Zhu Y, Yen TJ and Zhou BB:
Chfr regulates a mitotic stress pathway through its RING-finger
domain with ubiquitin ligase activity. Cancer Res. 62:1797–1801.
2002.PubMed/NCBI
|
|
22
|
Brooks L III, Heimsath EG Jr, Loring GL
and Brenner C: FHA-RING ubiquitin ligases in cell division cycle
control. Cell Mol Life Sci. 65:3458–3466. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Summers MK, Bothos J and Halazonetis TD:
The CHFR mitotic checkpoint protein delays cell cycle progression
by excluding Cyclin B1 from the nucleus. Oncogene. 24:2589–2598.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ogi K, Toyota M, Mita H, Satoh A, Kashima
L, Sasaki Y, Suzuki H, Akino K, Nishikawa N, Noguchi M, et al:
Small interfering RNA-induced CHFR silencing sensitizes oral
squamous cell cancer cells to microtubule inhibitors. Cancer Biol
Ther. 4:773–780. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bieganowski P, Shilinski K, Tsichlis PN
and Brenner C: Cdc123 and checkpoint forkhead associated with RING
proteins control the cell cycle by controlling eIF2gamma abundance.
J Biol Chem. 279:44656–44666. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kashima L, Toyota M, Mita H, Suzuki H,
Idogawa M, Ogi K, Sasaki Y and Tokino T: CHFR, a potential tumor
suppressor, downregulates interleukin-8 through the inhibition of
NF-kappaB. Oncogene. 28:2643–2653. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lee KH, Bae SH, Lee JL, Hyun MS, Kim SH,
Song SK and Kim HS: Relationship between urokinase-type plasminogen
receptor, interleukin-8 gene expression and clinicopathological
features in gastric cancer. Oncology. 66:210–217. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kang D, Chen J, Wong J and Fang G: The
checkpoint protein Chfr is a ligase that ubiquitinates Plk1 and
inhibits Cdc2 at the G2 to M transition. J Cell Biol. 156:249–259.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Hagting A, Jackman M, Simpson K and Pines
J: Translocation of cyclin B1 to the nucleus at prophase requires a
phosphorylation-dependent nuclear import signal. Curr Biol.
9:680–689. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hirota T, Kunitoku N, Sasayama T, Marumoto
T, Zhang D, Nitta M, Hatakeyama K and Saya H: Aurora A and an
interacting activator, the LIM protein Ajuba, are required for
mitotic commitment in human cells. Cell. 114:585–598. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Matsusaka T and Pines J: Chfr acts with
the p38 stress kinases to block entry to mitosis in mammalian
cells. J Cell Biol. 166:507–516. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Erson AE and Petty EM: CHFR-associated
early G2/M checkpoint defects in breast cancer cells. Mol Carcinog.
39:26–33. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
33
|
Awasthi N, Zhang C, Schwarz AM, Hinz S,
Schwarz MA and Schwarz RE: Enhancement of nab-paclitaxel antitumor
activity through addition of multitargeting antiangiogenic agents
in experimental pancreatic cancer. Mol Cancer Ther. 13:1032–1043.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Toyota M, Sasaki Y, Satoh A, Ogi K,
Kikuchi T, Suzuki H, Mita H, Tanaka N, Itoh F, Issa JP, et al:
Epigenetic inactivation of CHFR in human tumors. Proc Natl Acad Sci
USA. 100:pp. 7818–7823. 2003, View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gebauer F, Wicklein D, Horst J, Sundermann
P, Maar H, Streichert T, Tachezy M, Izbicki JR, Bockhorn M and
Schumacher U: Carcinoembryonic antigen-related cell adhesion
molecules (CEACAM) 1, 5 and 6 as biomarkers in pancreatic cancer.
PLoS One. 9:e1130232014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Privette LM, González ME, Ding L, Kleer CG
and Petty EM: Altered expression of the early mitotic checkpoint
protein, CHFR, in breast cancers: Implications for tumor
suppression. Cancer Res. 67:6064–6074. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Soutto M, Peng D, Razvi M, Ruemmele P,
Hartmann A, Roessner A, Schneider-Stock R and El-Rifai W:
Epigenetic and genetic silencing of CHFR in esophageal
adenocarcinomas. Cancer. 116:4033–4042. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Mariatos G, Bothos J, Zacharatos P,
Summers MK, Scolnick DM, Kittas C, Halazonetis TD and Gorgoulis VG:
Inactivating mutations targeting the chfr mitotic checkpoint gene
in human lung cancer. Cancer Res. 63:7185–7189. 2003.PubMed/NCBI
|
|
39
|
Cleven AH, Derks S, Draht MX, Smits KM,
Melotte V, Van Neste L, Tournier B, Jooste V, Chapusot C,
Weijenberg MP, et al: CHFR promoter methylation indicates poor
prognosis in stage II microsatellite stable colorectal cancer. Clin
Cancer Res. 20:3261–3271. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Gao L, Liu F, Zhang H, Sun J and Ma Y:
CHFR hypermethylation, a frequent event in acute myeloid leukemia,
is independently associated with an adverse outcome. Genes
Chromosomes Cancer. 55:158–168. 2016. View Article : Google Scholar : PubMed/NCBI
|