Introduction
Asparaginase is one of the major anticancer drugs
used in the treatment of acute lymphoblastic leukemia (ALL)
(1,2).
This drug hydrolyzes the amino acid asparagine to aspartic acid and
ammonia to deplete asparagine in the blood, inducing the malignant
lymphoid cells that depend on extracellular asparagine to enter
into apoptosis (2). The overall
survival (OS) rate is ~90% in pediatric patients with ALL and only
30% in adult patients with ALL (3,4). One of
the differences between pediatric and adult treatment regimens is
the intensive use of asparaginase in pediatric regimens.
Intensified use of asparaginase increases event-free survival (EFS)
for children with ALL by 10–15% (5–7).
Adolescent and adult patients receiving pediatric-based regimens
with greater use of asparaginase have also achieved better outcomes
(8–10). However, they are more prone to severe
grade 3–4 toxicities, including thrombosis, pancreatitis and
chemical hepatitis, compared with pediatric patients intensively
using asparaginase (11). Therefore,
the investigation of novel preparations of asparaginase is
essential.
Polyethylene glycol-conjugated asparaginase (PEG
asparaginase) is formed by the polyethylene glycosylation of the
Escherichia coli (E. coli)-derived enzyme, resulting
in a longer circulating half-life and lower immunogenicity, which
decreases the incidence of allergic reactions and anti-asparaginase
antibody formation (12,13). PEG asparaginase has been recommended
as a replacement for E. coli asparaginase in the treatment
of pediatric ALL due to its prolonged effect, similar safety
profile and convenience (14). In
addition, considering the decreased number of doses and clinic
visits, the total patient costs of PEG asparaginase are comparable
with those of native E. coli asparaginase (15). However, only a few clinical studies
have compared the efficacy and safety of PEG asparaginase with
native E. coli asparaginase in adolescent and adult patients
with ALL. The present study retrospectively compared the relative
efficacy and safety of PEG asparaginase and E. coli
asparaginase in adolescent and adult patients newly diagnosed with
ALL.
Materials and methods
Patients
A total of 122 patients who were ≥14 years old with
de novo ALL and had received treatment at the Department of
Hematology, Nanfang Hospital (Guangzhou, China) between January
2008 and July 2015 were included in this retrospective study,
including 46 patients in the PEG asparaginase group and 76 in the
E. coli asparaginase group. There were 76 males and 46
females, the median age of these patients was 27.4 years (ranged
from 14 to 62 years). ALL and central nervous system leukemia
(CNSL) cases were diagnosed based on the National Comprehensive
Cancer Network (NCCN) Clinical Practice Guidelines in Oncology:
Acute Lymphoblastic Leukemia (version 1, 2015) (16). Patients with liver dysfunction, renal
function damage or other organ dysfunctions prior to induction
therapy were excluded. Clinical data, including the patients'
characteristics, response to chemotherapy and adverse effects
experienced in association with asparaginase preparations, were
retrospectively analyzed. Ethical approval was obtained from the
Ethics Committee of Nanfang Hospital. Written informed consent was
obtained from all patients or their family members prior to
enrollment in the present study.
Treatment
Patients with newly diagnosed ALL in Nanfang
Hospital were administered a standard four-drug induction regimen
containing vincristine at 1.4 mg/m2/day, with a maximum
of 2 mg/day, or vindesine at 4 mg/day on days 1, 8, 15 and 22;
daunorubicin at 40 mg/m2/day or idarubicin at 8
mg/m2/day on days 1, 8, 15 and 22; E. coli
asparaginase at 6,000 IU/m2 once every 2 days from day
17 to 28 or PEG asparaginase at 2,500 IU/m2 on day 22;
and prednisone 1 mg/kg/day on days 1–28. Either E. coli
asparaginase or PEG asparaginase were used based on the patients'
approval. Philadelphia chromosome-positive patients with ALL
received tyrosine kinase inhibitors as long as the Philadelphia
chromosome was observed. Patients who went into CR received
consolidation chemotherapy for 2–4 commutative courses of the Hyper
CVAD-A/B regimen: Odd courses (1, 3, 5 and 7) of hyper-fractionated
cyclophosphamide (Cytoxan), doxorubicin (Adriamycin), vincristine
(Oncovin) and dexamethasone were given alternately with even
courses (2, 4, 6 and 8) of high-dose cytarabine and methotrexate.
Lumbar puncture and CNSL prophylaxis was provided during every
consolidation therapy. A total of 51 patients with
human-leukocyte-antigen-matched donors underwent an allogeneic bone
marrow transplant (allo-BMT) and 4 patients underwent an autologous
bone marrow transplant in CR1. A total of 7 relapsed patients and 2
patients with no response following continuous chemotherapy
underwent an allo-BMT.
Response criteria
The patients' responses to chemotherapy were
assessed on day 14±2 and at the end of induction chemotherapy. The
treatment efficacy was also evaluated according to the 2015 NCCN
Guidelines (16). Remission also
required normal marrow cellularity and elements. Follow-up was
performed for the patients in the clinic or by telephone, and the
median follow-up occurred at 15.4 months. Overall survival was
determined from ALL diagnosis to mortality or the final follow-up.
Relapse-free survival (RFS) was determined from the first CR to
relapse, mortality or the final follow-up.
Adverse events
All of the adverse events were graded according to
the National Cancer Institute Common Terminology Criteria for
Adverse Events, version 4.03 (17).
Coagulation disorder was defined as an activated partial
thromboplastin time prolonged by >10 sec over its upper limit of
normal (ULN, 31–43 sec), a thromboplastin time prolonged by >3
sec over its ULN (11–13 sec) or a fibrinogen level <1.5 g/l (2–4
g/l).
Statistical analysis
All of the evaluations were performed using SPSS
version 18.0 software (SPSS Inc., Chicago, IL, USA). The
descriptive statistical values included mean ± standard deviation,
median and range. Comparisons of the various groups were performed
using Student's t-test, Wilcoxon's rank sum test, χ2
test or Fisher's exact test. Comparisons of baseline clinical
variables across groups were made using the χ2 test or
Fisher's exact test for categorical data and constituent ratio;
Wilcoxon's rank sum test was applied for ranked data. Mean
comparisons between two groups were performed using Spearman's
rank. OS curves were estimated using the Kaplan-Meier method.
Comparisons of different survival curves were performed using
log-rank test. Two-sided P<0.05 was considered to indicate a
statistically significant difference.
Results
Patient characteristics
Of the 122 patients enrolled in the study, 46 were
treated with PEG asparaginase and 76 were treated with E.
coli asparaginase. Table I
displays the pretreatment characteristics of the study group. The
two groups were well balanced in terms of their pretreatment
characteristics, including age, initial white blood cell count,
bone marrow blast, extramedullary invasion, immunophenotype,
molecular biology aberration and chromosome characteristics
(Table I). Furthermore, 25 of the 46
patients in the PEG asparaginase group and 39 of the 76 patients in
the E. coli asparaginase group underwent hematopoietic stem
cell transplantation.
 | Table I.Baseline patient characteristics. |
Table I.
Baseline patient characteristics.
| Characteristic | PEG asparaginase
(n=46) | E. coli
asparaginase (n=76) | P-value |
|---|
| Total patients,
n | 46 | 76 |
|
| Median age (range),
years | 26 (14–62) | 23 (14–56) | 0.262 |
| Sex, n (%) |
|
|
|
|
Male | 30 (65.22) | 46 (60.53) | 0.604 |
|
Female | 16 (34.78) | 30 (39.47) |
|
| Median WBC (range),
×109/l | 22.61
(1.24–395.65) | 13.37
(0.54–435) | 0.146 |
| Hemoglobin,
g/l | 84.92±28.18 | 90.14±33.54 | 0.379 |
| Platelet (range),
×109/l | 40 (6–173) | 38.5 (4–289) | 0.996 |
| Bone marrow
lymphoblast % (range) | 87.45 (26–100) | 90 (24.5–99.5) | 0.574 |
| Extramedullary
invasion, n (%) | 22 (47.83) | 45 (59.21) | 0.221 |
| Immunophenotype, n
(%) |
|
| 0.745 |
|
B-ALL | 37 (80.43) | 64 (84.21) |
|
|
T-ALL | 7 (15.22) | 8 (10.53) |
|
|
Unknown | 2 (4.35) | 4 (5.26) |
|
| Molecular biology
aberration, n (%) |
|
|
|
|
BCR/ABL | 7 (15.22) | 19 (25.00) | 0.201 |
| P16
depletion | 10 (21.74) | 12 (15.79) | 0.407 |
| MLL
rearrangement | 3 (6.52) | 1 (1.32) | 0.150 |
| IGH
rearrangement | 3 (6.52) | 3 (3.95) | 0.671 |
| Chromosome, n
(%) |
|
| 0.705 |
| Ph | 6 (13.04) | 16 (21.05) |
|
| Complex
karyotype | 3 (6.52) | 4 (5.26) |
|
|
Hyperdiploidy | 1 (2.17) | 4 (5.26) |
|
|
Hypodiploidy | 1 (2.17) | 1 (1.32) |
|
|
Normal | 30 (65.22) | 40 (52.63) |
|
| No
mitotic phase | 5 (10.87) | 11 (14.47) |
|
| Transplantation, n
(%) | 25 (53.35) | 39 (51.32) | 0.745 |
Response to induction therapy
Overall, 113 (92.62%) patients achieved CR following
the induction of chemotherapy. As presented in Table II, 44 of the 46 patients in the PEG
asparaginase group (95.65%) and 69 of the 76 patients in the E.
coli asparaginase group (90.79%) achieved CR. Table II also presents the median times
taken to achieve CR; 30 days for the PEG asparaginase group and
29.5 days for the E. coli asparaginase group.
 | Table II.Response to induction therapy. |
Table II.
Response to induction therapy.
| Response | PEG asparaginase
(n=46) | E. coli
asparaginase (n=76) | P-value |
|---|
| CR, n (%) | 44 (95.65) | 69
(90.79) | 0.481 |
| Time to achieve CR
(range), days | 30 (12–94) | 29.50 (13–203) | 0.814 |
| CNSL in
consolidation phase, n (%) | 5
(10.87) | 21
(27.63) | 0.028 |
PEG asparaginase is superior to E.
coli asparaginase at preventing CNSL
During the consolidation phase, CNSL occurred in 5
of the 46 patients (10.87%) in the PEG asparaginase group and 21 of
the 76 patients (27.63%) in the E. coli asparaginase group.
The incidence of CNSL in the E. coli asparaginase group was
significantly higher compared with that in the PEG asparaginase
group (P=0.028; Table II),
suggesting that PEG asparaginase had a potential role in preventing
CNSL.
OS and RFS
As 1 patient in the PEG asparaginase group succumbed
to mortality due to severe pulmonary infection after achieving CR,
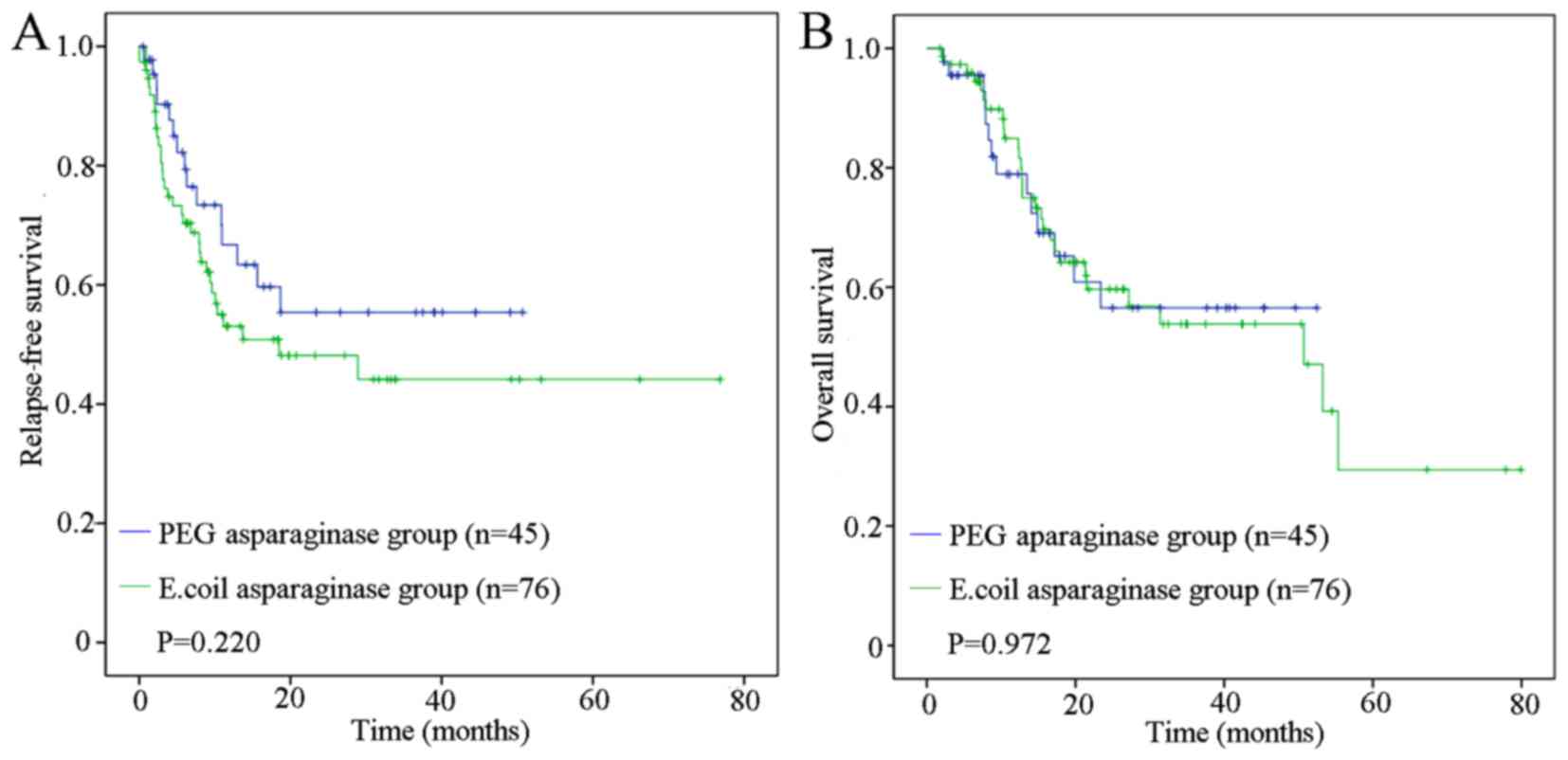
there were 45 patients to follow-up. As presented in Fig. 1A and B, at a median follow-up of 15.4
months, the median RFS time was 10.00 months (range, 0.5–50.70
months) for the PEG asparaginase group and 8.57 months (range,
0–76.83 months) for the E. coli asparaginase group (P=0.220;
Fig. 1A). In addition, the median OS
time for the PEG asparaginase group was 14.07 months (range,
2.27–52.47 months) and that for the E. coli asparaginase
group was 16.29 (range, 1.80–79.90) months (P=0.972; Fig. 1B). The present study observed no
significant difference in RFS and OS times between the two groups.
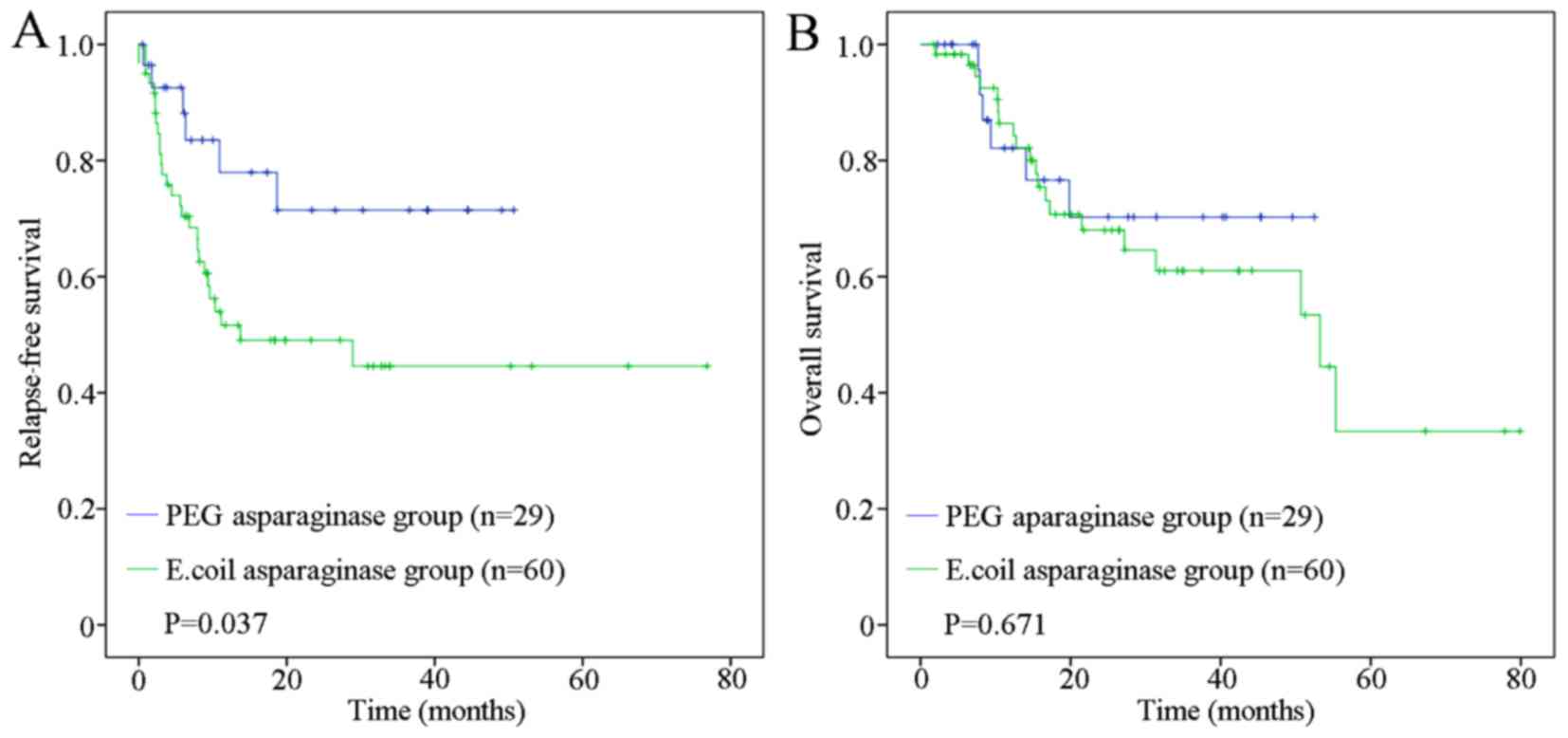
However, patients <35 years old in the PEG asparaginase group
obtained a longer median RFS time of 10.93 months (range, 0.5–50.70
months) compared with those in the E. coli asparaginase
group, who had a time of 8.97 months (range, 0–76.83 months)
(P=0.037; Fig. 2A); the median OS
time was not significantly different at 14.07 months (range,
2.27–52.47 months) for the PEG asparaginase group and 17.63 months
(1.80–79.90 months) for the E. coli asparaginase group
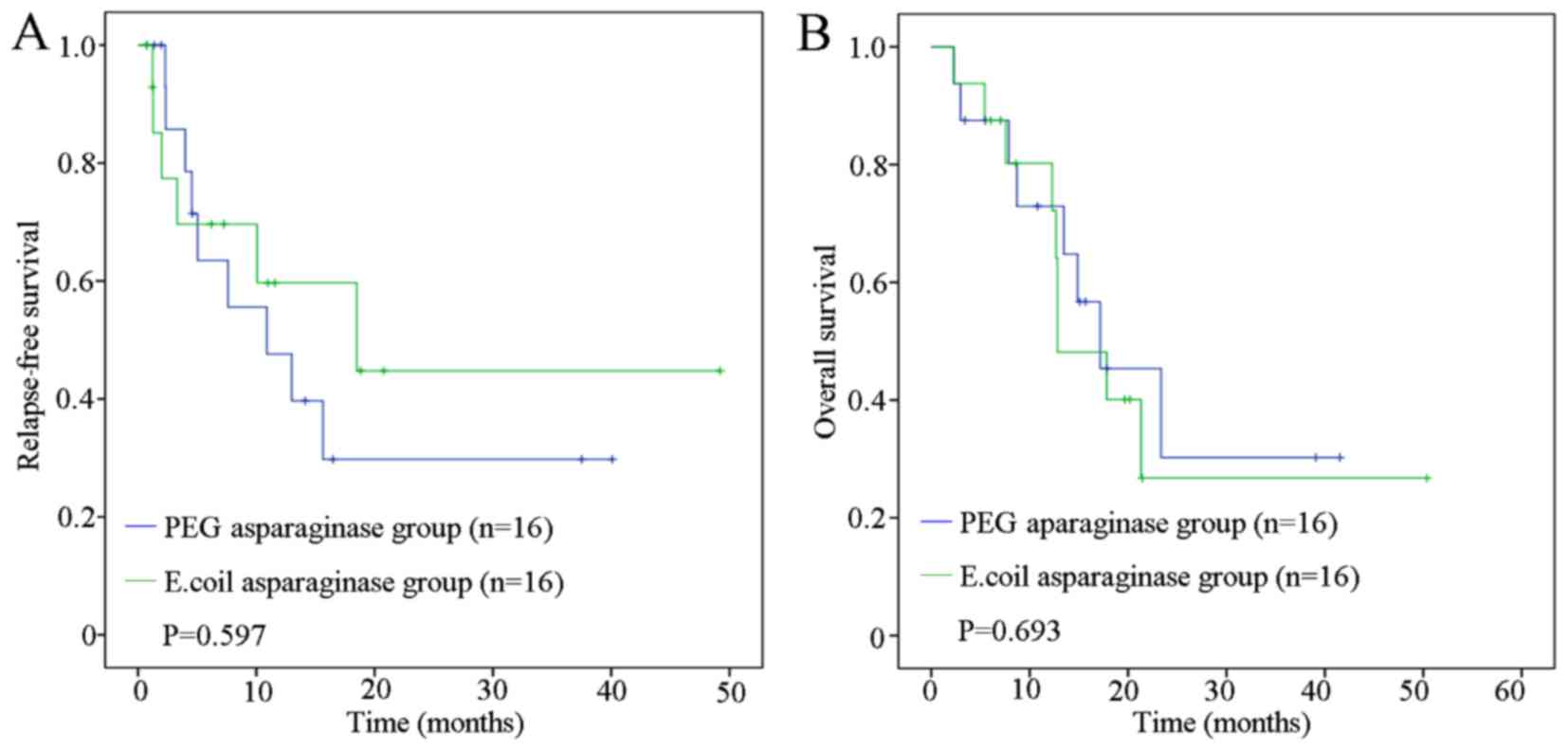
(P=0.671; Fig. 2B). For patients ≥35
years old, the PEG asparaginase and E. coli asparaginase
groups had a similar median RFS time of 6.32 months (range,
1.37–40.10 months) and 6.73 months (range, 0.7–49.20 months),
respectively (P=0.597), and a similar OS time of 14.17 months
(range, 2.27–41.53 months) and 12.77 months (range, 2.33–50.37
months), respectively (P=0.693), (Fig. 3A
and B).
Toxicity
Table III presents
the toxic events observed during the asparaginase-containing phases
of chemotherapy. Overall, the toxicities of asparaginase were
tolerable in the two groups. The incidences of allergy, grade
III–IV liver dysfunction, renal function damage and pancreatic
lesions were similar between the two groups. Patients in the PEG
asparaginase group demonstrated a longer duration of coagulation
dysfunction (9.80±5.51 vs. 6.80±4.21 days; P=0.002) compared with
those in the E. coli asparaginase group. However, the
present study observed no significant difference in bleeding events
between the two groups (8.70 vs. 3.95%; P=0.424). The grade of
myelosuppression between the two groups was similar for patients
who achieved CR; however, the duration of agranulocytosis was
significantly longer for the patients treated with PEG asparaginase
compared with those in the E. coli asparaginase group
(18.89±8.79 vs. 12.03±8.34 days; P<0.001). Consistent with the
difference in agranulocytosis duration, the patients who received
PEG asparaginase revealed a higher incidence of grade IV–V
infection compared with those who received E. coli
asparaginase (22.73 vs. 7.25%; P=0.018). However, this did not
increase the incidence of infection-associated mortality.
 | Table III.Adverse events during
asparaginase-containing treatment. |
Table III.
Adverse events during
asparaginase-containing treatment.
| Event | PEG asparaginase
(n=46) | E. coli
asparaginase (n=76) | P-value |
|---|
| Allergy, n (%) | 1 (2.17) | 0 (0.00) | 0.377 |
| Grade III–IV liver
dysfunction, n (%) | 10 (21.74) | 9 (11.84) | 0.144 |
| Renal function
damage, n (%) | 0 | 0 | – |
| Pancreatic lesion,
n (%) | 3 (6.52) | 4 (5.26) | 1.000 |
| Bleeding event, n
(%) | 4 (8.70) | 3 (3.95) | 0.424 |
| Maximal PT (range),
sec | 16.00
(10.50–49.00) | 15.00
(11.4–24.00) | 0.214 |
| Maximal APTT
(range), sec | 50.05
(26.50–100.70) | 45.85
(30.6–90.20) | 0.250 |
| Minimal Fbg
(range), g | 1.01
(0.15–2.36) | 1.00
(0.25–1.90) | 0.403 |
| Beginning of
coagulation dysfunction (range), days | 3.50 (1–14) | 4.00 (1–14) | 0.462 |
| Duration of
coagulation dysfunction, days | 9.80±5.51 | 6.80±4.21 | 0.002 |
| Grade of
granulocytopenia, n (%) |
|
| 0.298 |
| I | 1 (2.27) | 1 (1.45) |
|
| II | 0 (0) | 1 (1.45) |
|
|
III | 1 (2.27) | 5 (7.25) |
|
| IV | 42 (95.46) | 62 (89.85) |
|
| Duration of
agranulocytosis, days | 18.89±8.79 | 12.03±8.34 | <0.001 |
| Grade of
erythrocytopenia (%) |
|
| 0.068 |
| 0 | 1 (2.27) | 0 (0) |
|
| I | 0 (0) | 1 (1.45) |
|
| II | 3 (6.82) | 9 (13.04) |
|
|
III | 35 (79.55) | 58 (84.06) |
|
| IV | 5 (11.36) | 1 (1.45) |
|
| Grade of
thrombopenia (%) |
|
| 0.138 |
| 0 | 2 (4.55) | 6 (8.69) |
|
| I | 1 (2.27) | 2 (2.90) |
|
| II | 2 (4.55) | 4 (5.80) |
|
|
III | 5 (11.36) | 13 (18.84) |
|
| IV | 34 (77.27) | 44 (63.77) |
|
| Duration of
platelet level <20 g/l (range), days | 2 (0–34) | 1 (0–21) | 0.508 |
| Grade IV–V
infection, n (%)a | 10 (22.73) | 5 (7.25) | 0.018 |
|
Infection-associated mortality, n
(%)a | 1 (2.27) | 0 (0.00) | 0.389 |
Discussion
Asparaginase is an essential and routinely used
component in the frontline clinical treatment for ALL (1). However, administration of asparaginase
can be limited by the occurrence of anaphylaxis and formation of
antibodies to asparaginase. Studies have indicated that patients
with high-titer asparaginase antibodies demonstrate an increased
rate of clearance and degrade the activity of the agent, resulting
in poorer EFS (18,19). Patients who exhibit these reactions
are switched to another asparaginase product to ensure they are
exposed to a high enough dosage of asparaginase in accordance with
the treatment plan to obtain the optimal EFS. Due to its
polyethylene glycol covalent conjugation, PEG asparaginase has
decreased levels of renal excretion, a prolonged half-life,
improved bioavailability, decreased immunogenicity and a reduced
frequency of administration, making it an alternative to
L-asparaginase (12,20,21).
Previous clinical trials have revealed a more rapid lymphoblast
clearance, a lower titer of antibody and prolonged enzyme activity
in pediatric patients with ALL undergoing induction therapy with
PEG asparaginase compared with E. coli asparaginase
(22,23).
The present study compared the efficacy and safety
of PEG asparaginase with E. coli asparaginase in adolescent
and adult patients with ALL. The results demonstrated that the PEG
asparaginase and E. coli asparaginase groups had
satisfactory CR rates (95.65 vs. 90.79%) following induction
therapy. The median times taken to achieve CR were similar at 30
days in the PEG asparaginase group and 29.5 days in the E.
coli asparaginase group. When taking long-term efficacy into
consideration, the present study followed up on these patients and
identified no significant difference in median RFS time (10.00 vs.
8.57 months) or median OS time (14.07 vs. 16.29 months) between the
two groups. As age is an independent prognostic factor for patients
with ALL, the present study considered various age subgroups in
analyses and revealed that patients <35 years old in the PEG
asparaginase group experienced a longer median RFS time compared
with those in the E. coli asparaginase group (10.93 vs. 8.97
months; P=0.037), whereas no statistical significance was observed
in median RFS or OS times among patients ≥35 years old in the two
groups.
Furthermore, patients treated with E. coli
asparaginase demonstrated a significantly higher incidence of CNSL
compared with those treated with PEG asparaginase (27.63 vs.
10.87%; P=0.028) during the consolidation phase. This suggested
that PEG asparaginase served a potential role in preventing CNSL
compared with E. coli asparaginase. Patients with higher
cerebrospinal fluid (CSF) asparagine levels (>1 µmol/l) during
asparaginase treatment were more likely to experience isolated CNS
relapse (24). PEG asparaginase and
E. coli asparaginase may induce a decrease in the asparagine
level in the CSF (14,24). Asparaginase cannot cross the
blood-brain barrier or be detected in the CSF. However, it may
deplete the asparagine in the CSF if there is an adequate
expression level in the plasma (22,25).
Furthermore, the expression level of asparagine in the CSF was
inversely associated with the titer of the anti-asparaginase
antibody (22,24,26,27).
Hence, the present study hypothesized that the lower incidence of
CNSL in the PEG asparaginase group may have contributed to the
improved activity of the asparaginase. Numerous factors, including
pharmacological factors, asparaginase clearance and immunological
factors, such as antibody formation, influence the activity of
asparaginase. Firstly, the half-life of PEG asparaginase (~1 week)
is significantly longer compared with that of E. coli (~1.25
days). It is uncertain whether the efficacy of preventing CNSL is
associated with the half-life of asparaginase. Moghrabi et
al (7) previously compared the
efficacy of Erwinia asparaginase (t1/2 ~0.65 days) and
E. coli asparaginase (t1/2 ~1.24 days) and
revealed that pediatric patients with ALL treated with E.
coli asparaginase had fewer CNSL relapses (7). Secondly, patients treated with native
E. coli asparaginase have been shown to demonstrate a higher
titer of antibodies compared with those treated with PEG
asparaginase (14,23). Antibodies tend to diminish the
efficacy of asparaginase by neutralizing its activity (resulting in
faster asparaginase clearance) and high expression levels of
anti-asparaginase antibodies are associated with shorter durations
of asparagine depletion. Therefore, PEG asparaginase may strengthen
the anti-leukemia effect in the CNS due to its advantages of
extended effectiveness and lower immunogenicity.
Furthermore, a higher titer of anti-asparaginase
antibodies results in a more rapid clearance of the agent and
glucocorticoid. It has been reported that patients with ALL who
test positive for anti-asparaginase antibodies experience a more
rapid clearance of dexamethasone and a lower area under the curve
in the plasma (28). Furthermore,
multivariate analysis has demonstrated that a rapid clearance of
glucocorticoid and a positive anti-asparaginase antibody status are
independent risk factors in CNSL relapse (28). Due to its impact on hepatic synthesis,
asparaginase may decrease the synthesis of CYP3A cytochrome P450,
which is responsible for the metabolism of glucocorticoid (28). Based on these findings, patients who
develop anti-asparaginase antibodies are disadvantaged, as they
have less exposure to the two agents critical to ALL treatment. Due
to the small number of cases considered in the present study, the
difference in CNSL incidence may be associated with a sample error
rather than PEG asparaginase treatment. Therefore, a larger
population must be examined to verify the potential role of PEG
asparaginase in preventing CNSL.
The present study also evaluated the safety of the
two asparaginase preparations. Toxic events, including allergy,
grade III–IV liver dysfunction, renal function damage and
pancreatic lesions, were similar between the two groups. A
prolonged duration of coagulation dysfunction (9.80±5.51 vs.
6.80±4.21 days; P=0.002) was observed in the PEG asparaginase
group; however, it did not increase bleeding events (Table III). Patients treated with PEG
asparaginase also had longer durations of agranulocytosis
(18.89±8.79 vs. 12.03±8.34 days; P<0.01) and a higher grade IV–V
infection rate (22.73 vs. 7.25%; P=0.018) compared with those
treated with E. coli asparaginase. Nevertheless, there was
no statistical difference in infection-associated mortalities
between the two groups (Table III).
The present study suggested that this finding may be attributed to
the prolonged duration of agranulocytosis. The coagulation
dysfunction was attributable to the longer half-life and decreased
immunogenicity of the PEG asparaginase group. This revealed a more
evident effect on hepatic function, which changed the
pharmacokinetics of other chemotherapeutics and inhibited the
synthesis of coagulation factors (29).
The toxicities of asparaginase can be divided into
two main categories: Those associated with immunogenicity,
including hypersensitivity reactions (urticaria, rash, serum
sickness, bronchospasm and anaphylaxis) and those associated with
the inhibition of protein synthesis, including liver dysfunction,
pancreatitis and coagulation deficiency (14,30).
Patients with ALL may tolerate complications resulting from an
inhibition of protein synthesis under supportive treatment
(11). However, severe
hypersensitivity reactions, including life-threatening anaphylaxis,
require the urgent discontinuation of drug administration. Data
from previous studies have demonstrated that the incidence of
hypersensitivity reactions was 10% for PEG asparaginase patients
and varied from 13–30% for E. coli asparaginase patients
(22). The hypersensitivity reactions
were relative to the formation of anti-asparaginase antibodies. In
addition, ‘silent inactivation’ occurs when patients develop
antibodies and experience no clinical reaction. Patients in these
conditions may respond more poorly to the agent. The immunogenicity
of asparaginase remains an issue complicating its clinical use.
Studies have implied that the use of native E. coli
asparaginase in induction induces a high rate and intensification
of hypersensitivity (30%) to PEG asparaginase among pediatric
patients with ALL (31). Thus, PEG
asparaginase should be used up front in induction rather than in
consolidation. Given the immunogenicity of asparaginase, it is
strongly recommended that the anti-asparaginase antibodies should
be monitored. Detecting the status and expression level of
patients' antibodies is more important for silent inactivation
patients and serves a critical role in preventing the further use
of inactive asparaginase preparations (18,19).
In conclusion, PEG asparaginase was revealed to have
comparable CR rates, and OS and RFS times with those of E.
coli asparaginase in adolescent and adult patients with ALL.
More importantly, PEG asparaginase revealed the potential to
improve RFS times among patients <35 years old and prevent CNSL
development. Although it had more of an effect on coagulation
dysfunction and agranulocytosis, it did not increase
toxicity-associated mortalities. When considering its convenience
and improved enzyme activity, PEG asparaginase is a candidate for
first-line treatment of adolescent and adult ALL. As the present
study was a retrospective observation, a larger prospective
clinical trial is required to further confirm these findings.
Acknowledgements
The present study was supported by the 2014 General
Program of National Natural Science For Youth Foundation of China
(grant no. 81400104). Some preliminary analysis for this study have
been published previously in the Journal of Third Military Medical
University (Vol. 38, No. 7, Apr. 15, 2016). This article is the
subsequent one, which excludes the cases from the former sample,
including those who died following induction therapy, those lost to
follow-up and those not strictly treated according to the standard
dosage or treatment cycle. Furthermore, new cases were included and
an insight into the patients’ subgroup follow-up analysis was
included in this study.
Glossary
Abbreviations
Abbreviations:
|
PEG asparaginase
|
polyethylene glycol-conjugated
asparaginase
|
|
E. coli asparaginase
|
Escherichia coli
asparaginase
|
|
ALL
|
acute lymphoblastic leukemia
|
|
CR
|
complete remission
|
|
OS
|
overall survival
|
|
RFS
|
relapse-free survival
|
|
EFS
|
event-free survival
|
|
CNSL
|
central nervous system leukemia
|
|
NCCN
|
National Comprehensive Cancer
Network
|
|
allo-BMT
|
allogeneic bone marrow transplant
|
|
ULN
|
upper limit of normal
|
|
CSF
|
cerebrospinal fluid
|
References
|
1
|
Kawedia JD and Rytting ME: Asparaginase in
Acute Lymphoblastic Leukemia. Clin Lymph Myelom Leukem. 14
Suppl:S14–S17. 2014. View Article : Google Scholar
|
|
2
|
Shrivastava A, Khan AA, Khurshid M, Kalam
MA, Jain SK and Singhal PK: Recent developments in L-asparaginase
discovery and its potential as anticancer agent. Crit Rev Oncol
Hematol. 100:1–10. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Marinescu C, Vlădăreanu AM and Mihai F:
Acute lymphocytic leukemia in adults. Pathologic features and
prognosis. Rom J Intern Med. 53:31–36. 2015.PubMed/NCBI
|
|
4
|
Cooper SL and Brown PA: Treatment of
pediatric acute lymphoblastic leukemia. Pediatr Clin North Am.
62:61–73. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Amylon MD, Shuster J, Pullen J, Berard C,
Link MP, Wharam M, Katz J, Yu A, Laver J, Ravindranath Y, et al:
Intensive high-dose asparaginase consolidation improves survival
for pediatric patients with T cell acute lymphoblastic leukemia and
advanced stage lymphoblastic lymphoma: A Pediatric Oncology Group
study. Leukemia. 13:335–342. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Silverman LB, Gelber RD, Dalton VK,
Asselin BL, Barr RD, Clavell LA, Hurwitz CA, Moghrabi A, Samson Y,
Schorin MA, et al: Improved outcome for children with acute
lymphoblastic leukemia: Results of Dana-Farber Consortium Protocol
91–01. Blood. 97:1211–1218. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Moghrabi A, Levy DE, Asselin B, Barr R,
Clavell L, Hurwitz C, Samson Y, Schorin M, Dalton VK, Lipshultz SE,
et al: Results of the Dana-Farber Cancer Institute ALL Consortium
Protocol 95–01 for children with acute lymphoblastic leukemia.
Blood. 109:896–904. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Huguet F, Leguay T, Raffoux E, Thomas X,
Beldjord K, Delabesse E, Chevallier P, Buzyn A, Delannoy A,
Chalandon Y, et al: Pediatric-inspired therapy in adults with
Philadelphia chromosome-negative acute lymphoblastic leukemia: The
GRAALL-2003 study. J Clin Oncol. 27:911–938. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Stock W, La M, Sanford B, Bloomfield CD,
Vardiman JW, Gaynon P, Larson RA and Nachman J; Children's Cancer
Group, : Cancer and Leukemia Group B studies: What determines the
outcomes for adolescents and young adults with acute lymphoblastic
leukemia treated on cooperative group protocols? A comparison of
Children's Cancer Group and Cancer and Leukemia Group B studies.
Blood. 112:1646–1654. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Salzer WL, Devidas M, Shuster JJ, Wang C,
Chauvenet A, Asselin BL, Camitta BM and Kurtzberg J; Children's
Oncology Group, : Intensified PEG-L-asparaginase and
antimetabolite-based therapy for treatment of higher risk
precursor-B acute lymphoblastic leukemia: A report from the
Children's Oncology Group. J Pediatr Hematol Oncol. 29:369–375.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Stock W, Douer D, DeAngelo DJ, Arellano M,
Advani A, Damon L, Kovacsovics T, Litzow M, Rytting M, Borthakur G
and Bleyer A: Prevention and management of
asparaginase/pegasparaginase-associated toxicities in adults and
older adolescents: Recommendations of an expert panel. Leuk
Lymphoma. 52:2237–2253. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kopeček J: Polymer-drug conjugates:
Origins, progress to date and future directions. Adv Drug Deliver
Rev. 65:49–59. 2013. View Article : Google Scholar
|
|
13
|
Pasut G, Sergi M and Veronese FM:
Anti-cancer PEG-enzymes: 30 years old, but still a current
approach. Adv Drug Deliv Rev. 60:69–78. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Dinndorf PA, Gootenberg J, Cohen MH,
Keegan P and Pazdur R: FDA drug approval summary: Pegaspargase
(oncaspar) for the first-line treatment of children with acute
lymphoblastic leukemia (ALL). Oncologist. 12:991–998. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kurre HA, Ettinger AG, Veenstra DL, Gaynon
PS, Franklin J, Sencer SF, Reaman GH, Lange BJ and Holcenberg JS: A
pharmacoeconomic analysis of pegaspargase versus native Escherichia
coli L-asparaginase for the treatment of children with
standard-risk, acute lymphoblastic leukemia: The Children's Cancer
Group study (CCG-1962). J Pediatr Hematol Oncol. 24:175–181. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
NCCN Clinical Practice Guidelines in
Oncology (NCCN Guidelines)[S/OL]. Acute Lymphoblastic Leukemia.
Version 1. 2015, http://www.nccn.org/professionals/physician_gls/f_guidelines.asp
|
|
17
|
National Cancer Institute [S/OL], . Common
Terminology Criteria for Adverse Events (CTCAE) Mar 4. 2010,
http://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03_2010-06-14_QuickReference_5×7.pdf
|
|
18
|
Vrooman LM, Stevenson KE, Supko JG,
O'Brien J, Dahlberg SE, Asselin BL, Athale UH, Clavell LA, Kelly
KM, Kutok JL, et al: Postinduction dexamethasone and individualized
dosing of Escherichia coli L-asparaginase each improve outcome of
children and adolescents with newly diagnosed acute lymphoblastic
leukemia: Results from a randomized study-Dana-Farber Cancer
Institute ALL Consortium Protocol 00–01. J Clin Oncol.
31:1202–1210. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Panosyan EH, Seibel NL, Martin-Aragon S,
Gaynon PS, Avramis IA, Sather H, Franklin J, Nachman J, Ettinger
LJ, La M, et al: Asparaginase antibody and asparaginase activity in
children with higher-risk acute lymphoblastic leukemia: Children's
Cancer Group Study CCG-1961. J Pediatr Hematol Oncol. 26:217–226.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Rytting M: Peg-asparaginase for acute
lymphoblastic leukemia. Expert Opin Biol Ther. 10:833–839. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Thomas X, Cannas G, Chelghoum Y and
Gougounon A: Therapeutic alternatives to native L-asparaginase in
the treatment of adult acute lymphoblastic leukemia. Bull Cancer.
97:1105–1117. 2010.(In French). PubMed/NCBI
|
|
22
|
Graham ML: Pegaspargase: A review of
clinical studies. Adv Drug Deliv Rev. 55:1293–1302. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Avramis VI, Sencer S, Periclou AP, Sather
H, Bostrom BC, Cohen LJ, Ettinger AG, Ettinger LJ, Franklin J,
Gaynon PS, et al: A randomized comparison of native Escherichia
coli asparaginase and polyethylene glycol conjugated asparaginase
for treatment of children with newly diagnosed standard-risk acute
lymphoblastic leukemia: A Children's Cancer Group study. Blood.
99:1986–1994. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Panetta JC, Gajjar A, Hijiya N, Hak LJ,
Cheng C, Liu W, Pui CH and Relling MV: Comparison of native E. coli
and PEG asparaginase pharmacokinetics and pharmacodynamics in
pediatric acute lymphoblastic leukemia. Clin Pharmacol Ther.
86:651–658. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sirvent N, Suciu S, Rialland X, Millot F,
Benoit Y, Plantaz D, Ferster A, Robert A, Lutz P, Nelken B, et al:
Prognostic significance of the initial cerebro-spinal fluid (CSF)
involvement of children with acute lymphoblastic leukaemia (ALL)
treated without cranial irradiation: Results of European
Organization for Research and Treatment of Cancer (EORTC) Children
Leukemia Group study 58881. Eur J Cancer. 47:239–247. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Pieters R, Hunger SP, Boos J, Rizzari C,
Silverman L, Baruchel A, Goekbuget N, Schrappe M and Pui CH:
L-asparaginase treatment in acute lymphoblastic leukemia: A focus
on Erwinia asparaginase. Cancer. 117:238–249. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Henriksen LT, Nersting J, Raja RA,
Frandsen TL, Rosthøj S, Schrøder H and Albertsen BK; Nordic Society
of Paediatric Haematology and Oncology (NOPHO) group, :
Cerebrospinal fluid asparagine depletion during pegylated
asparaginase therapy in children with acute lymphoblastic
leukaemia. Br J Haematol. 166:213–220. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kawedia JD, Liu C, Pei D, Cheng C,
Fernandez CA, Howard SC, Campana D, Panetta JC, Bowman WP, Evans
WE, et al: Dexamethasone exposure and asparaginase antibodies
affect relapse risk in acute lymphoblastic leukemia. Blood.
119:1658–1664. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Merryman R, Stevenson KE, Gostic WN,
Neuberg D II, O'Brien J, Sallan SE and Silverman LB:
Asparaginase-associated myelosuppression and effects on dosing of
other chemotherapeutic agents in childhood acute lymphoblastic
leukemia. Pediatr Blood Cancer. 59:925–927. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Avramis VI and Tiwari PN: Asparaginase
(native ASNase or pegylated ASNase) in the treatment of acute
lymphoblastic leukemia. Int J Nanomedicine. 1:241–254.
2006.PubMed/NCBI
|
|
31
|
Tong WH, Pieters R, Kaspers GJ, Te LD,
Bierings MB, van den Bos C, Kollen WJ, Hop WC, Lanvers-Kaminsky C,
Relling MV, et al: A prospective study on drug monitoring of
PEGasparaginase and Erwinia asparaginase and asparaginase
antibodies in pediatric acute lymphoblastic leukemia. Blood.
123:2026–2033. 2014. View Article : Google Scholar : PubMed/NCBI
|