Introduction
Breast cancer is currently the most common form of
cancer and the second-leading cause of death from cancer in women
(1). Comprehensive treatment
strategies using surgery in combination with chemotherapy,
radiotherapy, and endocrine therapy have achieved significant
progress in improving the outcome of breast cancer (2). This can be attributed not only to the
continuous improvement of surgical techniques and methods but also
to the improved efficacy of chemotherapy-based adjuvant therapy in
controlling the progression of primary lesions and tumor recurrence
and metastasis (3). However, clinical
challenges, such as cancer recurrence and metastasis, caused by
chemotherapy failure need to be addressed immediately (4).
Natural plants offer a treasure trove of resources
for antitumor drug development (5).
Currently, exploring natural plant-derived antitumor drugs with
definitive efficacy, high efficiency, and low toxicity has become
one of the most popular research topics in breast cancer treatment
(6). China has abundant sources of
medicinal species and a long history of using medicinal plants for
anticancer treatment. There are various successful examples of
medicinal monomer compounds, such as ginsenoside Rh2 (7) and tanshinone IIA (8) that have been developed and applied to
tumor treatment. In 1967, the US National Cancer Institute
discovered taxol, which was isolated from the bark of the Pacific
yew (9). Since then, more than 30
research groups worldwide have successfully semi-synthesized or
fully synthesized taxol or both, and Taxol quickly became the
first-line drug for adjuvant therapy of various tumors including
breast cancer (10).
Clematis belongs to the family Ranunculaceae
and has ~355 species worldwide. China is home to 155 species, of
which ~70 are widely used in traditional Chinese medicine (TCM)
(11). The TCM drug Radix Clematidis
recorded in the Chinese Pharmacopoeia is isolated from the dried
roots and rhizomes of Clematis chinensis Osbeck, Clematis
mandshurica Rupr, and Clematis hexapetala Pall. This
drug has analgesic, sedative, antibacterial, anti-inflammatory, and
diuretic effects (People's Republic of China Pharmacopoeia
Commission, 2005). Currently, studies on the active substances in
Clematis are scarce. We conducted a preliminary research
study on Clematis plants that are included in an antitumor
treatment formula by the Naxi ethnic group (Yunnan, China)
(11). We have successfully extracted
four monomer compounds from this species, and their inhibitory
effects on the growth of breast cancer cells were proven by using
bioactivity tests (11,12). Among them, Clematis hederagenin
saponin (hederagenin 3β-O-α-L-arabinopyranoside, CHS) belongs to
the class of triterpenoid saponins (13,14).
Triterpenoid saponins show bioactivities against various types of
malignancies including breast, colon, and lung cancers (15). Numerous noteworthy studies have
revealed that triterpenoid saponin compounds have relatively high
anti-breast cancer activity and could be potential drugs that would
contribute to chemoprevention and treatment of breast cancer
(16). Previous studies have found
that triterpenoid saponin compounds can exert an inhibitory effect
on numerous types of cancers by regulating different signaling
pathways, such as the epidermal growth factor receptor (EGFR),
estrogen receptor (ER) (16,17), and Fas/Fas ligand (FasL) pathways
(18). However, to the best of our
knowledge, studies on the triterpenoid saponins extracted from
Clematis ganpiniana are very scarce, and those on the
antitumor mechanism of Clematis species are even fewer.
This is an original research study on the
pro-apoptotic effect of saponins from the Clematis vine on
breast cancer cells and an exploration of the apoptotic pathways
involved. Cell apoptosis and proliferation are two basic
physiological processes, which are also basic measures that
maintain the dynamic equilibrium of the number of cells in the
body. There are two main apoptotic pathways: One involves
intracellular caspases activated by extracellular signals while the
other involves caspases activated by mitochondria-derived
activators of caspase. Activated caspases can degrade key cellular
proteins, thereby causing apoptosis (19,20).
Considering the lack of research on the effect and the underlying
mechanisms of saponins extracted from Clematis vine on
breast cancer cells, we investigated the role of this compound in
inducing apoptosis of breast cancer cells by using MCF-7 and
MDA-MB-231 breast cancer cell lines as experimental models.
Moreover, because of the pivotal role of the mitochondrial pathway
in apoptosis, the study was focused particularly on the effect of
this compound on the mitochondrial pathway to further clarify its
antitumor mechanisms.
Materials and methods
Compound preparation
The extraction and purification of CHS were
conducted using the method previously described in the literature
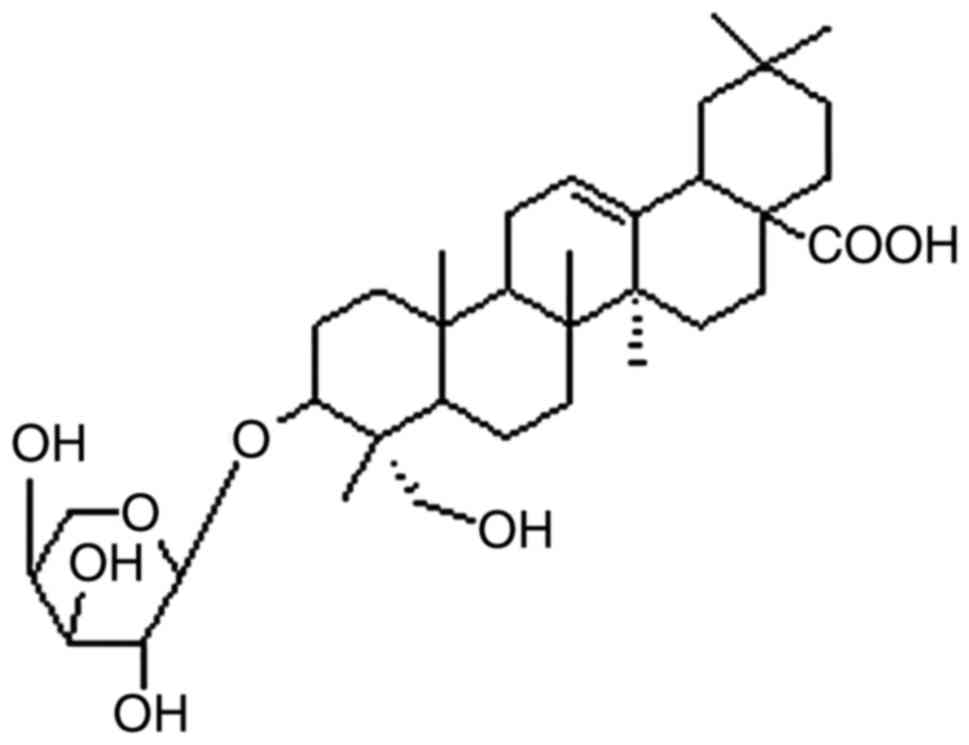
(11) and the chemical structure of
the saponin is shown in Fig. 1. The
extracted compound was dissolved in 100% dimethyl sulfoxide (DMSO)
and stored at −20°C. Before use, the drug was dissolved in culture
medium to final concentrations of 0.08, 0.4, 2 and 10 µg/ml. Cells
treated with only DMSO were used as the control. The possible
cytotoxic effects of DMSO were minimized by ensuring a final DMSO
concentration ≤0.1% (v/v).
Cell culture
MCF-7 and MDA-MB-231 breast cancer cell lines (ATCC,
Manassas, VA, USA) were cultured in Dulbecco's modified Eagle's
medium (DMEM) containing 10% fetal bovine serum (FBS), 100 U/ml
penicillin, and 100 g/ml streptomycin at 37°C in an atmosphere of
5% CO2.
For routine passages, cultures were split 1:3 when
they reached 80–90% confluence generally every 2–3 days. All
experiments were performed on exponentially growing cells. Two
breast cancer cell lines MCF-7 and MDA-MB-231 which represented
different phenotypes of this heterogeneous disease, were used to
evaluate the growth inhibition and to explore the underlying
molecular mechanisms of CHS.
Main reagents
The reagents and kits used in this study included
DMEM (Hyclone Laboratories, Inc., Logan, UT, USA), FBS (Wisent,
Nanjing, China), trypsin, DMSO,
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT)
(Beyotime Institute of Biotechnology, Nanjing, China),
mitochondrial protein extraction kit (KeyGen Biotech Co., Ltd.,
Nanjing, China), and Annexin V-FITC apoptosis detection kit (Bender
Medsystems, San Diego, CA, USA). Target antibodies, including
anti-Apaf-1 and anti-cytochrome c (Cyto C) primary antibodies, were
all purchased from Cell Signaling Technology, Inc. (Danvers, MA,
USA). The secondary antibody used was horseradish peroxidase
(HRP)-goat anti rabbit IgG (Cell Signaling Technology, Inc.).
β-actin (Beyotime Institute of Biotechnology) was used as an
internal standard. The enhanced chemiluminescence (ECL) kit and
polyvinylidene fluoride (PVDF) membranes were both purchased from
Pierce (Rockford, IL, USA) and EMD Millipore (Billerica, MA, USA),
respectively.
Cytotoxic effect assay (MTT
assay)
Cells were collected and diluted with culture medium
to a density of 105 cells/ml, and then seeded into
96-well plates at 100 µl/well. The cells were cultured overnight to
70% confluence, and treated with 0.08, 0.4, 2, or 10 µg/ml CHS.
Each concentration was tested in triplicate. Subsequently, the
cells were cultured in vitro for 12, 24, or 48 h. Breast
cancer cells cultured in medium with 0.05% DMSO as a negative
control for this experiment. After treatment, the culture medium in
each well was discarded and replaced with 180 µl of fresh culture
medium and 20 µl of MTT (5 mg/ml). The cells were incubated at 37°C
for 4 h, after which the culture medium was discarded and 150 µl of
DMSO was added to each well. The samples were placed on a shaker
for 15 min and and then read at 490 nm with a microplate reader
(5082; Tecan Austria GmbH, Grödig, Austria). Cell survival rate was
calculated as OD of the experiment samples/OD of the control ×
100.
Detection of the apoptosis rate using
flow cytometry
After exposure to 2 µg/ml CHS for 6, 12 and 24 h,
the breast cancer cells were collected and centrifuged.
Approximately 105 cells were analyzed for each
treatment. After the supernatant was discarded, the treated cells
were trypsinized and washed three times with PBS, followed by the
addition of Annexin V-FITC and propidium iodide (PI). The cells
were then kept in the dark at room temperature for 10 min prior to
evaluation using flow cytometry to determine the apoptosis rate.
Annexin V can bind to phosphatidylserineand PI can easily enter
dead or damaged cells. The combination of the two dyes can be used
to detect cellular apoptosis. FITC-positive cells are the cells in
the early stage of apoptosis, while PI-positive staining indicates
dead cells. The cells stained with both are in the late stage of
apoptosis, while cells that not stained by either are normal, live
cells.
Detection of caspase-3 and −9 activity
using flow cytometry
After treatment (24 h of exposure to 2 µg/ml CHS),
the breast cancer cells were collected and centrifuged. After the
supernatant was discarded, the treated cells were trypsinized and
resuspended in PBS. Next, 300 µl of each sample was added to a
centrifuge tube and mixed with 100 µM RED-DEVD-FMK (caspase-3) or
RED-LEHD-FMK (caspase-9), and incubated at 37°C for 30 min. After
centrifugation, the supernatant was discarded and the cell pellets
were washed twice with 500 µl of wash buffer. Subsequently, the
cells were resuspended and subjected to flow cytometry to detect
the number of positively stained cells. Z-DEVD-FMK is a caspase-3
inhibitor and Z-LEHD-FMK is a caspase-9 inhibitor, both of which
can inhibit apoptosis caused by caspase activation. To determine
the inhibitor-induced change in caspase activity, the cells in the
two treatment groups were pretreated with one of the two inhibitors
(100 µM) for 1 h prior to the CHS treatment (2.0 µg/ml). What's
more, after exposure to 10 µg/ml CHS for 24 h, breast cancer cells
(with or without pre-incubated with caspase inhibitors 1 h before
CHS treatment) were resuspended in stain containing Annexin
V-FITC/PI and analyzed with FACSAria flow cytometer using FACSDiva
software.
Western blotting (WB) assay
MCF-7 and MDA-MB-231 cells were seeded at
1×106 cells in 100 mm2 dishes. Cells were
treated in complete medium with CHS for 2, 6, 12 and 24 h. After
treatment, the cells treated with the compound (or control) were
collected, washed once with PBS, and centrifuged at 1,500 rpm for 5
min. The supernatant was discarded. A mitochondrial protein
extraction kit (Beyotime Institute of Biotechnology) was used to
extract mitochondrial proteins from cells in accordance with the
manufacturer's instructions. Briefly, after exposure, MCF-7 and
MDA-MB-231 cells were harvested and centrifuged at 800 × g at 4°C
for 10 min. The pellets were added with 20 mM
N-2-hydroxyethylpiperazine-N0-20-ethanesulfonic acid (HEPES) buffer
containing protease inhibitor cocktail and disrupted with a glass
tissue grinder. Homogenates were centrifuged at 800 × g at 4°C for
10 min, and the resulting supernatants were transferred to 0.5 ml
conical tubes, and further centrifuged at 10,000 × g at 4°C for 20
min. The final pellets, containing the mitochondrial fraction, were
analyzed for protein content using the Bradford method.
After protein quantification using the bicinchoninic
acid (BCA) method, the samples were added to 5X protein loading
buffer, boiled for 5 min to sufficiently denature proteins, and
then stored at −70°C. Forty micrograms of protein was loaded into
each lane for sodium dodecyl sulfate polyacrylamide gel
electrophoresis (SDS-PAGE). Next, the separated proteins were
blotted onto a PVDF membrane by wet transfer. After a 2 h block at
room temperature to eliminate non-specific signals, the membrane
was incubated with the primary anti-Apaf-1 or anti-Cyto C (1:1,000)
antibodies at 4°C overnight, followed by three 5 min washes.
Subsequently, the membrane was incubated with secondary antibodies
(1:1,000) at room temperature for 2 h, followed by three 10 min
washes. β-actin (1:1,000) was used as a standard to ensure equal
loading of protein into each lane of the gel. Finally, the protein
signal was visualized by using ECL reagent and an automated
chemiluminescence gel imaging system.
Statistical analysis
SPSS 20.0 statistical software (SPSS Inc., Chicago,
IL, USA) was used for statistical analysis. The results were
expressed as mean ± standard deviation. One-way analysis of
variance (ANOVA) was used for intergroup comparisons, followed by
pairwise comparisons between groups. The results were expressed as
mean ± standard deviation (mean± SD). P<0.05 was considered to
indicate a statistically significant difference.
Results
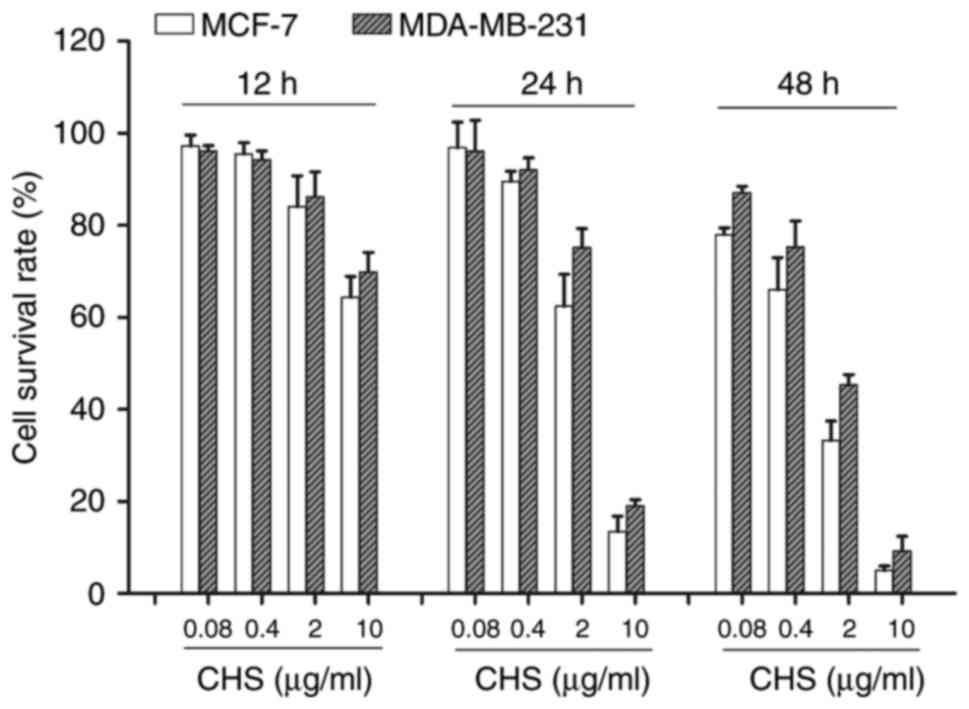
CHS showed cytotoxic effect on breast
cancer cells
In this study, MCF-7 and MDA-MB-231 breast cancer
cells were used to evaluate the anticancer effect of CHS. Compared
to the negative control group, CHS showed cytotoxic effect on both
types of breast cancer cells after 12, 24 and 48 h of treatment, in
a time- and dose-dependent manner (P<0.05) (Fig. 2).
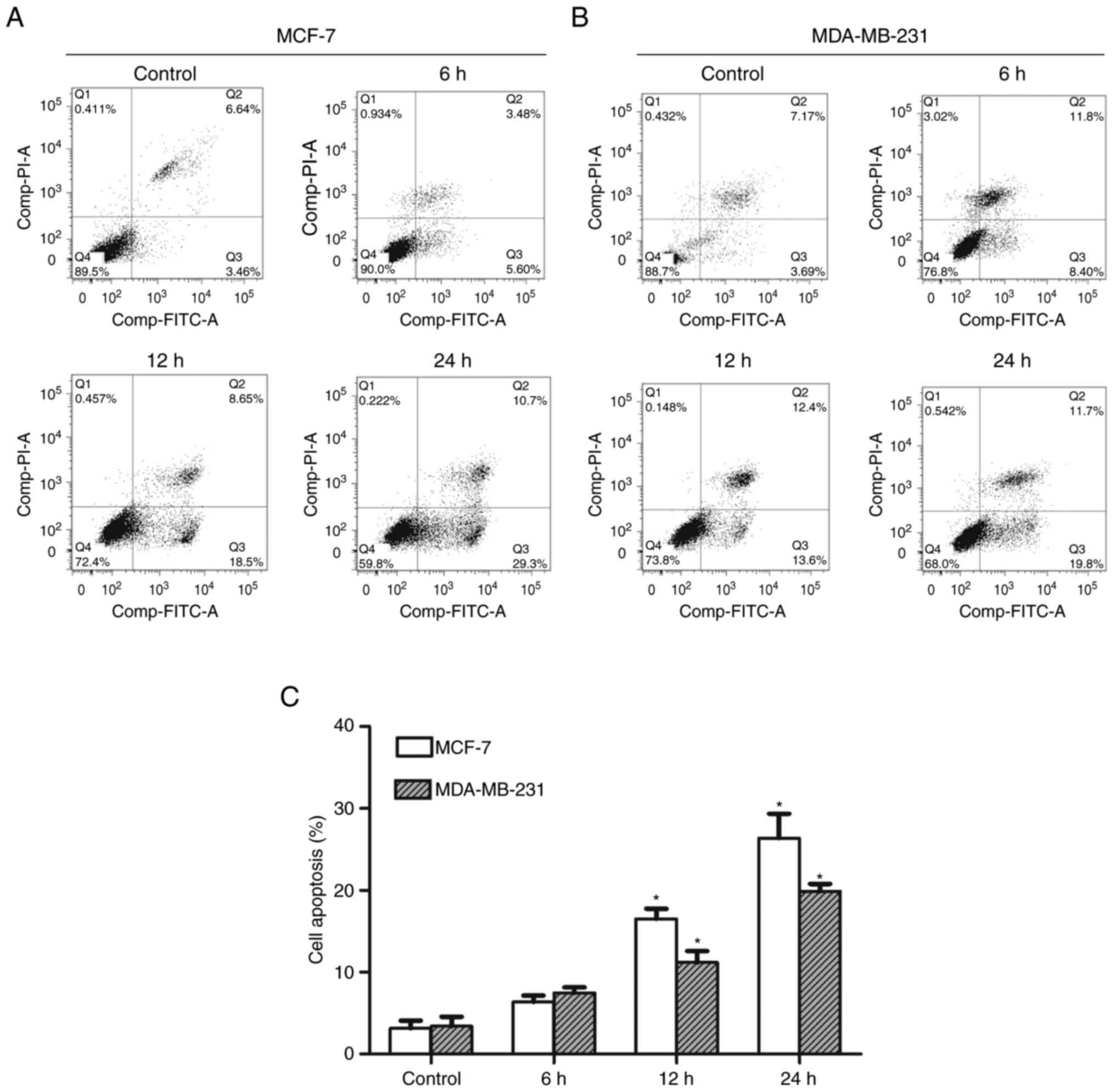
CHS induced apoptosis in breast cancer
cells
After 6, 12, and 24 h of treatment with 2.0 µg/ml
CHS, MCF-7 and MDA-MB-231 cells were evaluated using flow cytometry
to determine the apoptosis rate. The results showed that CHS
induced apoptosis in breast cancer cells and the apoptosis rate
increased over time. MCF-7 and MDA-MB-231 cells treated with 2.0
µg/ml CHS for 24 h showed an early apoptosis rate of 29.3 and
19.8%, respectively (Fig. 3A and B).
Early apoptosis rate of cells treated with CHS of three independent
experiments were shown in column statistics (Fig. 3C).
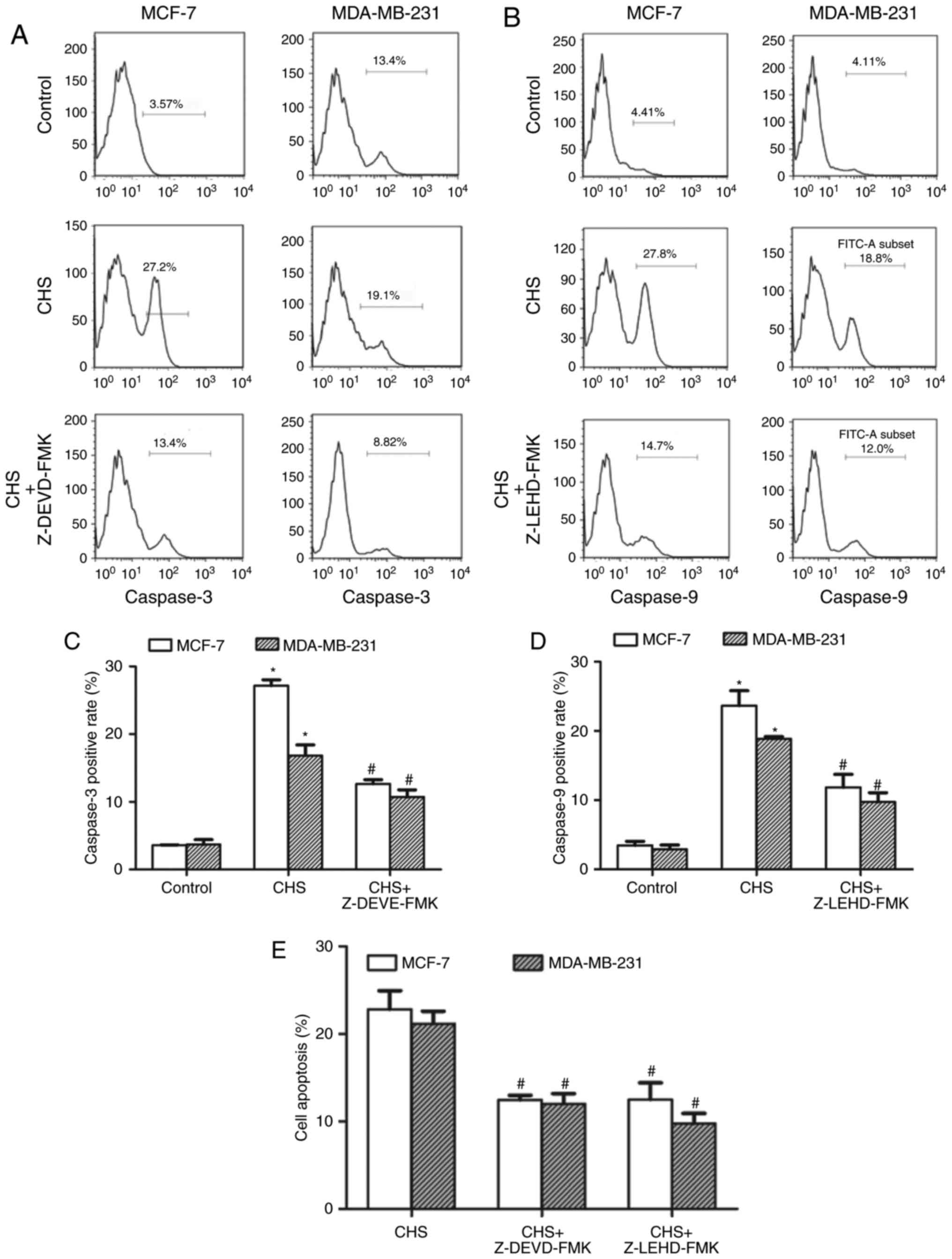
CHS upregulated the activity of
caspase-3 and caspase-9 in breast cancer cells
After treatment with CHS (2.0 µg/ml) for 24 h, the
breast cancer cells were subjected to RED-DEVD-FMK (caspase-3) or
RED-LEHD-FMK (caspase-9) staining and flow cytometry to determine
the changes in caspase activity. The results confirmed that CHS
increased both caspase-3 and −9 activity in breast cancer cells. In
the inhibitor-pretreatment groups, both caspase-3 and −9 inhibitors
showed an inhibitory effect on the CHS-induced increase of caspase
activity (Fig. 4A and B). Caspase-3,
−9 positive rate of cells treated with CHS with/without caspase
inhibitors of three independent experiments were shown in column
statistics (Fig. 4C and D). Moreover,
both caspase-3 and −9 inhibitors reduced CHS-induced apoptosis
(Fig. 4E), which demonstrated that
caspase-3 and −9 activation were involved in CHS-induced
apoptosis.
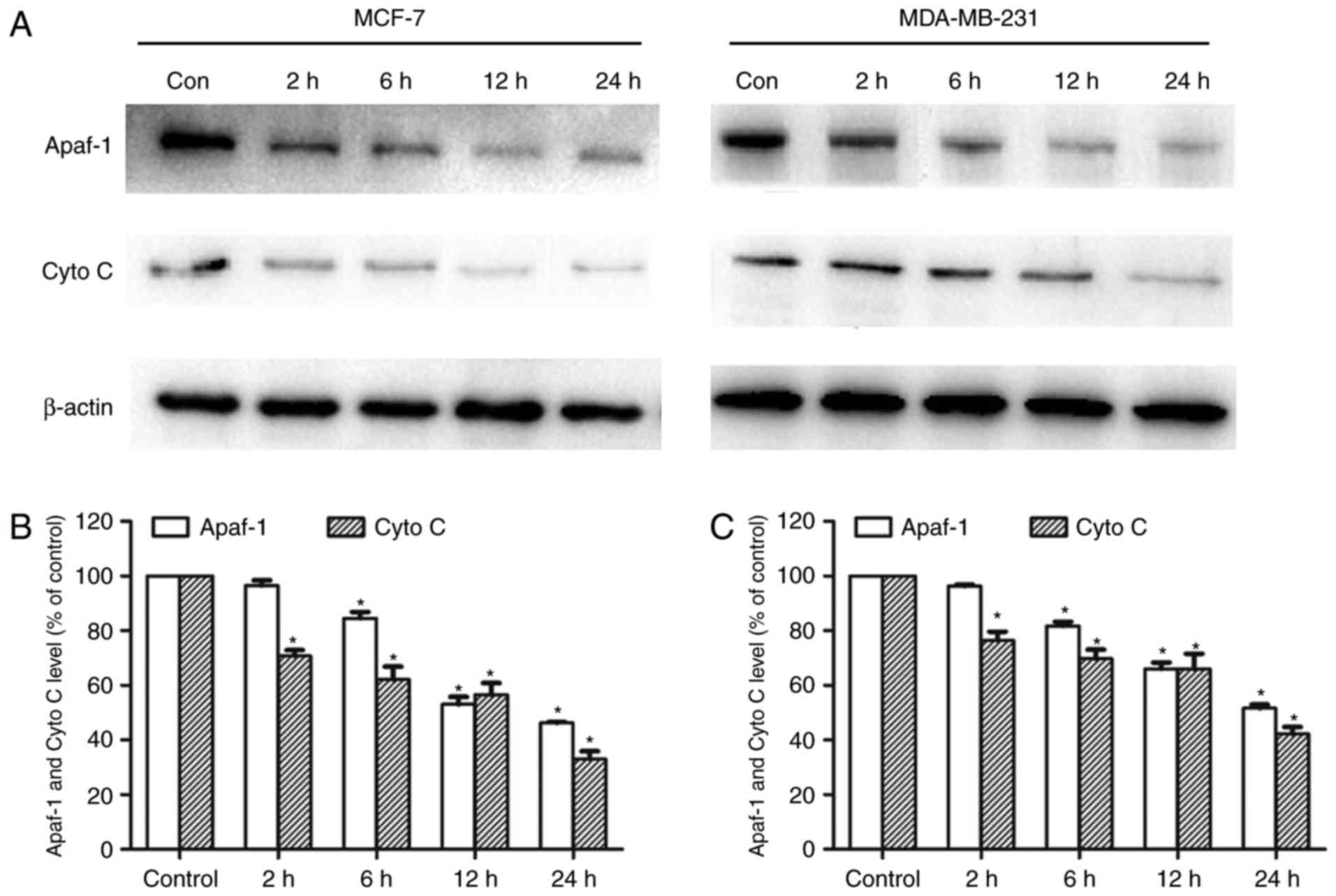
CHS regulated the mitochondrial Apaf-1
and Cyto c level
MCF-7 and MDA-MB-231 cells were treated for 2, 6, 12
or 24 h with CHS (2 µg/ml) and both mitochondrial Apaf-1 and Cyto C
level were detected by western blotting. CHS significantly reduced
mitochondrial Apaf-1 and Cyto C proteins in breast cancer cells,
indicating the enhanced release of Apaf-1 and Cyto C from
mitochondria in breast cancer cells (Fig.
5A). After the cells were treated with the compound for 2 to 24
h, there was gradual ruduction of mitochondria Apaf-1 and Cyto C
proteins. Expressions of mitochondrial Apaf-1 andCyto C of cells
treated with CHS of three independent experiments were shown in
column statistics (Fig. 5B and
C).
Discussion
In this study, we discovered that CHS, a new type of
triterpenoid saponin, showed strong cytotoxic effect on various
types of breast cancer cells. CHS can induce apoptosis in both
ER+ MCF-7 breast cancer cells and ER−
MDA-MB-231 breast cancer cells. Further, we found that the
mechanisms underlying the induction of apoptosis by CHS involved
the regulation of caspase-3 and −9 activity and the reduction of
mitochondrial Apaf-1 and Cyto C proteins in breast cancer
cells.
Recent studies have found that monomer components
extracted from Clematis ganpiniana can suppress cell
proliferation and promote cell apoptosis in malignant tumors. For
example, Clematis montana lectin can induce apoptosis in
MCF-7 breast cancer cells in a dose-dependent manner (21). Polygonatum cyrtonema lectin, a
mannose-binding lectin can induce apoptosis and autophagy in A375
melanoma cells through the mitochondria-mediated ROS-p38-p53
pathway (22). In this study, the
effects of a triterpenoid saponin extracted from Clematis on
the growth and apoptosis of human breast cancer cells and its
underlying mechanism were investigated.
CHS significantly reduced the mitochondrial Apaf-1
and Cyto C level. It also increased the activities of caspase-3 and
−9. Mitochondria play a core role in the progression of cellular
apoptosis. The cellular stress response or apoptotic signals can
cause mitochondria to release Cyto C to induce apoptosis (23). CHS may induce mitochondria to release
Cyto C, which acts as an apoptosis inducer to form apoptosomes with
Apaf-1, caspase-9, and ATP/dATP. As a result, caspase-3 is
recruited and activated, triggering a caspase cascade and
subsequent apoptosis (24,25).
Mitochondrial apoptotic pathway was reported widely
for the actions of triterpenoid saponins in other human cancers
including liver cancer (26–28), gastric cancer (29), esophageal cancer (30), and colorectal cancer (31). However, studies about mitochondrial
apoptotic activity in breast cancer of triterpenoid saponins are
scarce, most of which focus on ginsenosides. For example,
Ginsenoside Rh2 inhibited viability of both MCF-7 and MDA-MB-231
human breast cells by mitochondrial pathway (32). Ginsenoside F2 induced apoptosis in
breast cancer stem cells via mitochondria pathway (33). Moreover, α-hederin, a triterpenoid
saponin similar to CHS, induced apoptosis via mitochondrial
perturbations (34,35), substantiating our findings as a
possible common mechanistic pathway of triterpenoid
saponins-induced apoptosis.
However, to the best of our knowledge, there are no
studies on the effects of hederagenin saponin isolated from
Clematis on breast cancer cells. In this study, we found
that CHS had a strong cytotoxic effect on MDA-MB-231 and MCF-7
breast cancer cells, and can also cause apoptosis in breast cancer
cells. CHS significantly reduced the mitochondrial Apaf-1 and Cyto
C leveland also increased the activities of caspase-3 and −9. This
indicates that similar to numerous types of triterpenoid saponins,
CHS induces the apoptosis of tumor cells via the mitochondrial
pathway.
Natural plants are a treasure trove of resources for
antitumor drug development. Developing natural plant-derived
antitumor drugs with definitive efficacy, high efficacy, and
low-toxicity is a promising research direction for breast cancer
treatment. However, the use of Chinese medicinal herbs for tumor
treatment is currently undeveloped and unsystematic, with treatment
methods varying largely, mostly on a case-by-case basis. The
standardization of TCMs for clinical applications requires a large
number of rigorous in vitro and in vivo studies.
Domestic and international studies on the reactive monomer
substances from Clematis ganpiniana are relatively few, and
even fewer studies investigate the effect and the underlying
mechanisms of the antitumor activity of Clematis ganpiniana.
This study analyzed the antitumor function of CHS, a type of
triterpenoid saponin extracted from Clematis ganpiniana, and
the underlying mechanisms. The results suggest that CHS offers
great potential as a new anti-breast cancer drug.
Acknowledgements
This study was supported by Natural Science
Foundation of China (grant nos. 81502294, 81572595, 81272916 and
81602336) and Changzhou Sci and Tech Program (grant no.
CJ20159044).
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2016. CA Cancer J Clin. 66:7–30. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Song JL, Chen C, Yuan JP and Sun SR:
Progress in the clinical detection of heterogeneity in breast
cancer. Cancer Med. 5:3475–3488. 2016. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Liedtke C and Kolberg HC: Systemic therapy
of advanced/metastatic breast cancer-current evidence and future
concepts. Breast Care (Basel). 11:275–281. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Krol M, Pawłowski KM, Majchrzak K, Szyszko
K and Motyl T: Why chemotherapy can fail? Pol J Vet Sci.
13:399–406. 2010.PubMed/NCBI
|
|
5
|
Karikas GA: Anticancer and chemopreventing
natural products: Some biochemical and therapeutic aspects. J BUON.
15:627–638. 2010.PubMed/NCBI
|
|
6
|
Cragg GM and Newman DJ: Natural products:
A continuing source of novel drug leads. Biochim Biophys Acta.
1830:3670–3695. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yang J, Yuan D, Xing T, Su H, Zhang S, Wen
J, Bai Q and Dang D: Ginsenoside Rh2 inhibiting HCT116 colon cancer
cell proliferation through blocking PDZ-binding kinase/T-LAK
cell-originated protein kinase. J Ginseng Res. 40:400–408. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lin CY, Chang TW, Hsieh WH, Hung MC, Lin
IH, Lai SC and Tzeng YJ: Simultaneous induction of apoptosis and
necroptosis by tanshinone IIA in human hepatocellular carcinoma
HepG2 cells. Cell Death Discov. 2:160652016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Peltier S, Oger JM, Lagarce F, Couet W and
Benoît JP: Enhanced oral paclitaxel bioavailability after
administration of paclitaxel-loaded lipid nanocapsules. Pharm Res.
23:1243–1250. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang TH, Wang HS and Soong YK:
Paclitaxel-induced cell death: Where the cell cycle and apoptosis
come together. Cancer. 88:2619–2628. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ding Q, Yang LX, Yang HW, Jiang C, Wang YF
and Wang S: Cytotoxic and antibacterial triterpenoids derivatives
from Clematis ganpiniana. J Ethnopharmacol. 126:382–385. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cheng L, Xia TS, Wang YF, Zhou W, Liang
XQ, Xue JQ, Shi L, Wang Y and Ding Q: The apoptotic effect of D
rhamnose β-hederin, a novel oleanane-type triterpenoid saponin on
breast cancer cells. PLoS One. 9:e908482014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Podolak I, Galanty A and Sobolewska D:
Saponins as cytotoxic agents: A review. Phytochem Rev. 9:425–474.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Augustin JM, Kuzina V, Andersen SB and Bak
S: Molecular activities, biosynthesis and evolution of triterpenoid
saponins. Phytochemistry. 72:435–457. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ma G, Guo W, Zhao L, Zheng Q, Sun Z, Wei
J, Yang J and Xu X: Two new triterpenoid saponins from the root of
Platycodon grandiflorum. Chem Pharm Bull (Tokyo). 61:101–104. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bishayee A, Ahmed S, Brankov N and Perloff
M: Triterpenoids as potential agents for the chemoprevention and
therapy of breast cancer. Front Biosci (Landmark Ed). 16:980–996.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Rabi T, Huwiler A and Zangemeister-Wittke
U: AMR-Me inhibits PI3K/Akt signaling in hormone-dependent MCF-7
breast cancer cells and inactivates NF-κB in hormone-independent
MDA-MB-231 cells. Mol Carcinog. 53:578–588. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Xiong J, Cheng G, Tang H, Zhen HN and
Zhang X: Ardipusilloside I induces apoptosis in human glioblastoma
cells through a caspase-8-independent FasL/Fas-signaling pathway.
Environ Toxicol Pharmacol. 27:264–270. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Cohen GM: Caspases: The executioners of
apoptosis. Biochem J. 326:1–16. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kim R, Emi M and Tanabe K:
Caspase-dependent and -independent cell death pathways after DNA
damage (Review). Oncol Rep. 14:595–599. 2005.PubMed/NCBI
|
|
21
|
Peng H, Lv H, Wang Y, Liu YH, Li CY, Meng
L, Chen F and Bao JK: Clematis montana lectin, a novel
mannose-binding lectin from traditional Chinese medicine with
antiviral and apoptosis-inducing activities. Peptides.
30:1805–1815. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Liu B, Cheng Y, Zhang B, Bian HJ and Bao
JK: Polygonatum cyrtonema lectin induces apoptosis and autophagy in
human melanoma A375 cells through a mitochondria-mediated
ROS-p38-p53 pathway. Cancer Lett. 275:54–60. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Suen DF, Norris KL and Youle RJ:
Mitochondrial dynamics and apoptosis. Genes Dev. 22:1577–1590.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Merino D, Lok SW, Visvader JE and Lindeman
GJ: Targeting BCL-2 to enhance vulnerability to therapy in estrogen
receptor-positive breast cancer. Oncogene. 35:1877–1887. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zeng M, Zheng M, Lu D, Wang J, Jiang W and
Sha O: Anti-tumor activities and apoptotic mechanism of
ribosome-inactivating proteins. Chin J Cancer. 34:325–334. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wang QF, Chen JC, Hsieh SJ, Cheng CC and
Hsu SL: Regulation of Bcl-2 family molecules and activation of
caspase cascade involved in gypenosides-induced apoptosis in human
hepatoma cells. Cancer Lett. 183:169–178. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang J, Zhao XZ, Qi Q, Tao L, Zhao Q, Mu
R, Gu HY, Wang M, Feng X and Guo QL: Macranthoside B, a hederagenin
saponin extracted from Lonicera macranthoides and its anti-tumor
activities in vitro and in vivo. Food Chem Toxicol. 47:1716–1721.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Park HM, Kim SJ, Kim JS and Kang HS:
Reactive oxygen species mediated ginsenoside Rg3- and Rh2-induced
apoptosis in hepatoma cells through mitochondrial signaling
pathways. Food Chem Toxicol. 50:2736–2741. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Chun J, Ha IJ and Kim YS:
Antiproliferative and apoptotic activities of triterpenoid saponins
from the roots of Platycodon grandiflorum and their
structure-activity relationships. Planta Med. 79:639–645. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Mo S, Xiong H, Shu G, Yang X, Wang J,
Zheng C, Xiong W and Mei Z: Phaseoloideside E, a novel natural
triterpenoid saponin identified from entada phaseoloides, induces
apoptosis in Ec-109 esophageal cancer cells through reactive oxygen
species generation. J Pharmacol Sci. 122:163–175. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wang CZ, Li XL, Wang QF, Mehendale SR,
Fishbein AB, Han AH, Sun S and Yuan CS: The mitochondrial pathway
is involved in American ginseng-induced apoptosis of SW-480 colon
cancer cells. Oncol Rep. 21:577–584. 2009.PubMed/NCBI
|
|
32
|
Choi S, Oh JY and Kim SJ: Ginsenoside Rh2
induces Bcl-2 family proteins-mediated apoptosis in vitro and in
xenografts in vivo models. J Cell Biochem. 112:330–340. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Mai TT, Moon J, Song Y, Viet PQ, Phuc PV,
Lee JM, Yi TH, Cho M and Cho SK: Ginsenoside F2 induces apoptosis
accompanied by protective autophagy in breast cancer stem cells.
Cancer Lett. 321:144–153. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Swamy SM and Huat BT: Intracellular
glutathione depletion and reactive oxygen species generation are
important in alpha-hederin-induced apoptosis of P388 cells. Mol
Cell Biochem. 245:127–139. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Cheng L, Xia TS, Wang YF, Zhou W, Liang
XQ, Xue JQ, Shi L, Wang Y, Ding Q and Wang M: The anticancer effect
and mechanism of α-hederin on breast cancer cells. Int J Oncol.
45:757–763. 2014. View Article : Google Scholar : PubMed/NCBI
|