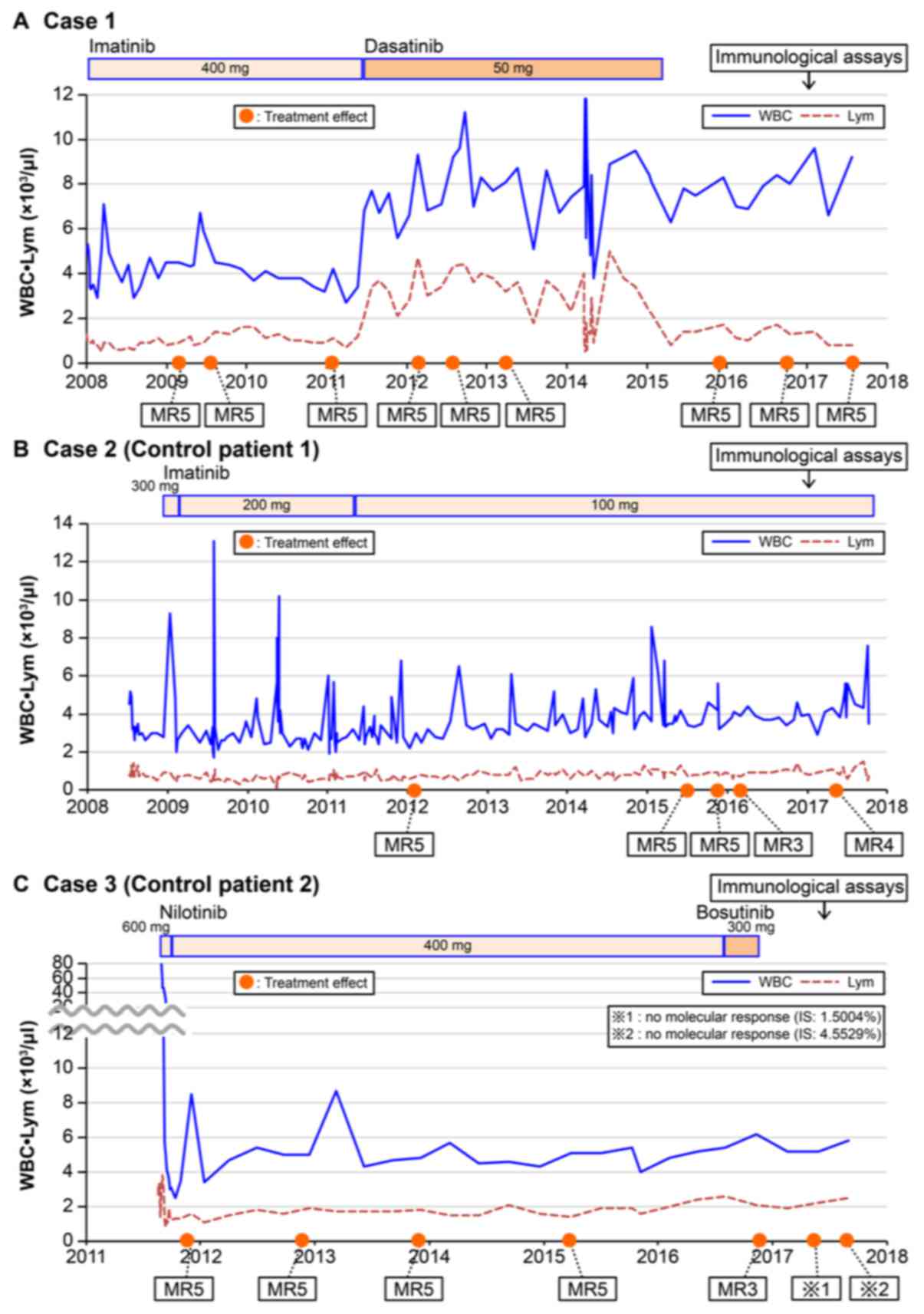
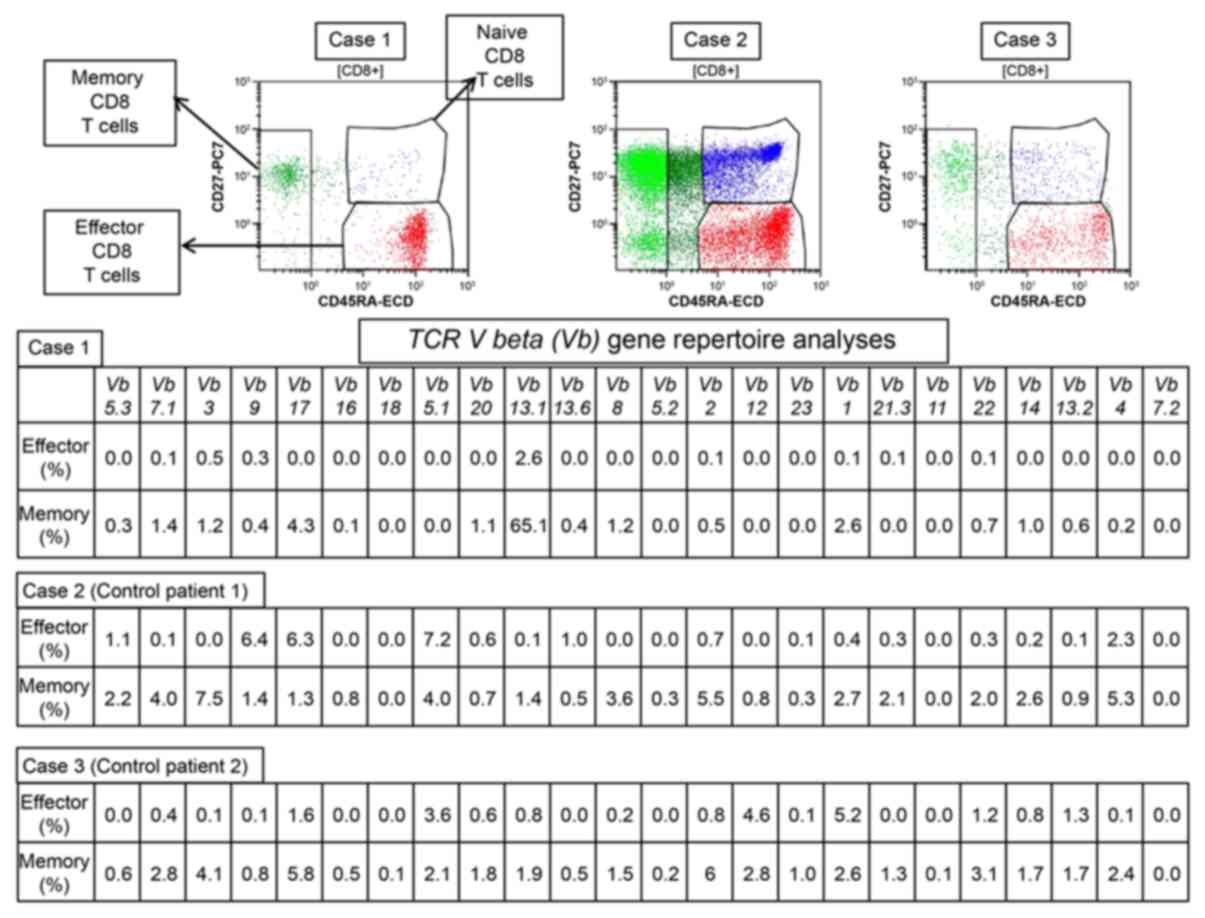
|
1
|
O'Brien SG, Guilhot F, Larson RA, Gathmann
I, Baccarani M, Cervantes F, Cornelissen JJ, Fischer T, Hochhaus A,
Hughes T, et al: Imatinib compared with interferon and low-dose
cytarabine for newly diagnosed chronic-phase chronic myeloid
leukemia. N Engl J Med. 348:994–1004. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kantarjian H, Shah NP, Hochhaus A, Cortes
J, Shah S, Ayala M, Moiraghi B, Shen Z, Mayer J, Pasquini R, et al:
Dasatinib versus imatinib in newly diagnosed chronic-phase chronic
myeloid leukemia. N Engl J Med. 362:2260–2270. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Saglio G, Kim DW, Issaragrisil S, le
Coutre P, Etienne G, Lobo C, Pasquini R, Clark RE, Hochhaus A,
Hughes TP, et al: Nilotinib versus imatinib for newly diagnosed
chronic myeloid leukemia. N Engl J Med. 362:2251–2259. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hantschel O, Rix U and Superti-Furga G:
Target spectrum of the BCR-ABL inhibitors imatinib, nilotinib and
dasatinib. Leuk Lymphoma. 49:615–619. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mahon FX, Réa D, Guilhot J, Guilhot F,
Huguet F, Nicolini F, Legros L, Charbonnier A, Guerci A, Varet B,
et al: Discontinuation of imatinib in patients with chronic myeloid
leukaemia who have maintained complete molecular remission for at
least 2 years: The prospective, multicentre Stop Imatinib (STIM)
trial. Lancet Oncol. 11:1029–1035. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ross DM, Branford S, Seymour JF, Schwarer
AP, Arthur C, Yeung DT, Dang P, Goyne JM, Slader C, Filshie RJ, et
al: Safety and efficacy of imatinib cessation for CML patients with
stable undetectable minimal residual disease: Results from the
TWISTER study. Blood. 122:515–522. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Imagawa J, Tanaka H, Okada M, Nakamae H,
Hino M, Murai K, Ishida Y, Kumagai T, Sato S, Ohashi K, et al:
Discontinuation of dasatinib in patients with chronic myeloid
leukaemia who have maintained deep molecular response for longer
than 1 year (DADI trial): A multicentre phase 2 trial. Lancet
Haematol. 2:e528–e535. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kolb HJ, Schattenberg A, Goldman JM,
Hertenstein B, Jacobsen N, Arcese W, Ljungman P, Ferrant A,
Verdonck L, Niederwieser D, et al: Graft-versus-leukemia effect of
donor lymphocyte transfusions in marrow grafted patients. Blood.
86:2041–2050. 1995.PubMed/NCBI
|
|
9
|
Mustjoki S, Ekblom M, Arstila TP, Dybedal
I, Epling-Burnette PK, Guilhot F, Hjorth-Hansen H, Höglund M,
Kovanen P, Laurinolli T, et al: Clonal expansion of T/NK-cells
during tyrosine kinase inhibitor dasatinib therapy. Leukemia.
23:1398–1405. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kim DH, Kamel-Reid S, Chang H, Sutherland
R, Jung CW, Kim HJ, Lee JJ and Lipton JH: Natural killer or natural
killer/T cell lineage large granular lymphocytosis associated with
dasatinib therapy for Philadelphia chromosome positive leukemia.
Haematologica. 94:135–139. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
White HE, Matejtschuk P, Rigsby P, Gabert
J, Lin F, Lynn Wang Y, Branford S, Müller MC, Beaufils N, Beillard
E, et al: Establishment of the first World Health Organization
International Genetic Reference Panel for quantitation of BCR-ABL
mRNA. Blood. 116:e111–e117. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sasaki K, Strom SS, O'Brien S, Jabbour E,
Ravandi F, Konopleva M, Borthakur G, Pemmaraju N, Daver N, Jain P,
et al: Relative survival in patients with chronic-phase chronic
myeloid leukaemia in the tyrosine-kinase inhibitor era: Analysis of
patient data from six prospective clinical trials. Lancet Haematol.
2:e186–e193. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Rohon P, Porkka K and Mustjoki S:
Immunoprofiling of patients with chronic myeloid leukemia at
diagnosis and during tyrosine kinase inhibitor therapy. Eur J
Haematol. 85:387–398. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kreutzman A, Rohon P, Faber E, Indrak K,
Juvonen V, Kairisto V, Voglová J, Sinisalo M, Flochová E, Vakkila
J, et al: Chronic myeloid leukemia patients in prolonged remission
following interferon-α monotherapy have distinct cytokine and
oligoclonal lymphocyte profile. PLoS One. 6:e230222011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
de Castro FA, Palma PV, Morais FR, Simões
BP, Carvalho PV, Ismael SJ, Lima CP and Voltarelli JC:
Immunological effects of interferon-alpha on chronic myelogenous
leukemia. Leuk Lymphoma. 44:2061–2067. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Schiffer CA, Cortes JE, Hochhaus A, Saglio
G, le Coutre P, Porkka K, Mustjoki S, Mohamed H and Shah NP:
Lymphocytosis after treatment with dasatinib in chronic myeloid
leukemia: Effects on response and toxicity. Cancer. 122:1398–1407.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Powers JJ, Dubovsky JA, Epling-Burnette
PK, Moscinski L, Zhang L, Mustjoki S, Sotomayor EM and
Pinilla-Ibarz JA: A molecular and functional analysis of large
granular lymphocyte expansions in patients with chronic myelogenous
leukemia treated with tyrosine kinase inhibitors. Leuk Lymphoma.
52:668–679. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kreutzman A, Juvonen V, Kairisto V, Ekblom
M, Stenke L, Seggewiss R, Porkka K and Mustjoki S: Mono/oligoclonal
T and NK cells are common in chronic myeloid leukemia patients at
diagnosis and expand during dasatinib therapy. Blood. 116:772–782.
2010. View Article : Google Scholar : PubMed/NCBI
|