Introduction
Combined hepatocellular cholangiocarcinoma
(cHCC-CC), a rare and unique form of primary liver malignancy, was
first described in 1949 by Allen and Lisa (1), and accounts for 0.4–14.2% of primary
liver malignancies (2,3). The World Health Organization
classification defines cHCC-CC as a tumor containing unequivocal
HCC and CC components; the condition is distinguished from separate
HCC and intrahepatic CC (ICC) arising in the same liver (4).
The prognosis of patients with cHCC-CC undergoing
liver resection has been reported to be poor. The 5-year overall
survival (OS) rate was reported to be 37.2%, and the 5-year disease
free survival rate was 10.7% (5);
therefore, it is necessary to identify the prognostic factors of
patients diagnosed with cHCC-CC who underwent surgical
resection.
Due to its low prevalence, the prognostic factors of
cHCC-CC remain unclear. The well-known Tumor-Node-Metastasis (TNM)
staging system for cHCC-CC remains controversial and poorly
understood (6). Whether common
prognostic factors (particularly lymph nodal metastasis, which has
been most frequently revealed to be associated with the prognosis
of various solid tumors, including HCC and ICC) (5,7) leads to a
poorer prognosis in patients with HCC-CC is also controversial
(8). Certain previous studies
demonstrated that lymph node metastasis was a significant
prognostic factor in patients with cHCC-CC (5), while in other reports (6), lymphatic metastasis failed to represent
a significant prognostic factor. Thus, novel prognostic markers are
required to predict the prognosis of patients with cHCC-CC.
Inflammation serves an important role in the
development and progression of numerous malignancies by
participating in the neoplastic process, proliferation and
migration (9). Systemic inflammation
is a complex progress, the response to which can be assessed using
the neutrophil-to-lymphocyte ratio (NLR) (10). Elevated pre-treatment NLR has been
confirmed to be associated with poor outcomes in various types of
cancer, including non-small cell lung cancer (11), gastric cancer (12), ovarian cancer (13), advanced pancreatic cancer (14), hepatocellular carcinoma (15–19) and
cholangiocarcinoma (20). However,
the utility of the NLR has not been validated in cHCC-CC.
The present study hypothesized that the NLR may be a
practical predictor of the inflammatory process, and investigated
the association between inflammation and the prognosis of patients
with cHCC-CC. Therefore, the present retrospective study evaluated
the association between the NLR and prognosis in patients with
cHCC-CC who underwent surgical resection.
Patients and methods
Patient cohort
A total of 59 patients who underwent surgical
resection and were histologically diagnosed with cHCC-CC between
January 2000 and October 2014 at the Department of Hepatobiliary
and Pancreatic Surgery (Sun Yat-sen University Cancer Center,
Guangzhou, China) were retrospectively recruited. Exclusion
criteria were as follows: i) other treatments, including
transarterial chemoembolization, radiofrequency treatment and liver
transplantation for patients with HCC before surgical resection;
ii) inadequate renal function (serum creatinine level and blood
urea nitrogen level higher than the upper limits of normal); iii)
severe coagulopathy (prothrombin activity <40% or platelet count
<40,000/mm3); iv) Child-Pugh C liver function or
evidence of hepatocellular decompensation, including refractory
ascites, esophageal or gastric variceal bleeding, and hepatic
encephalopathy; v) obstructive jaundice; vi) other concurrent
primary tumors; vii) pathological confirmed subtype of cHCC-CC with
stem cell features or viii) follow-up period of <3 months or
lost to follow up.
Clinical data collection
All clinicopathological data were retrieved from
medical records at the Department of Hepatobiliary and Pancreatic
Surgery of the Sun Yat-sen University Cancer Center.
Clinicopathological parameters included histologically confirmed
cHCC-CC, age, gender, leukocyte cell count, neutrophil cell count
and lymphocyte cell count, levels of hemoglobin, platelets,
α-fetoprotein (AFP), carbohydrate antigen 19-9 (CA19-9), γ-glutamyl
transpeptidase (GGT) and hepatitis B surface antigen (HbsAg), tumor
size, tumor number, lymph node metastasis, major thrombi,
microvascular thrombi, and conventional TNM stage for HCC and ICC,
as established by the Union for International Cancer Control and
the American Joint Committee on Cancer (AJCC) (21) (Table I).
The laboratory data were obtained prior to surgical resection. The
present study was approved by the Institutional Review Board of the
Sun Yat-sen University Cancer Center. Written informed consent was
obtained from all patients prior to enrollment in the present
study.
 | Table I.Clinicopathological factors in
NLR≤2.75 (n=41) and NLR>2.75 (n=11) groups at diagnosis. |
Table I.
Clinicopathological factors in
NLR≤2.75 (n=41) and NLR>2.75 (n=11) groups at diagnosis.
| Variables | NLR≤2.75, n | NLR≤2.75, n | P-value |
|---|
| Age, years |
|
|
|
| ≤60 | 35 | 9 |
|
|
>60 | 6 | 2 | 0.856 |
| Gender |
|
|
|
|
Female | 11 | 4 |
|
| Male | 30 | 7 | 0.709 |
| AFP, ng/ml |
|
|
|
| ≤25 | 16 | 4 |
|
|
>25 | 25 | 7 | 0.851 |
| CA19-9, U/ml |
|
|
|
| ≤35 | 27 | 9 |
|
|
>35 | 14 | 2 | 0.468 |
| GGT, U/l |
|
|
|
| ≤50 | 21 | 2 |
|
|
>50 | 20 | 9 | 0.086 |
| HbsAg |
|
|
|
|
Negative | 5 | 1 |
|
|
Positive | 36 | 10 | 0.806 |
| Tumor size, cm |
|
|
|
|
<5 | 24 | 2 |
|
| ≥5 | 17 | 9 | 0.017 |
| Tumor number |
|
|
|
|
Solitary | 11 | 3 |
|
|
Multiple | 30 | 8 | 0.724 |
| Lymph node
metastasis |
|
|
|
|
Negative | 37 | 8 |
|
|
Positive | 4 | 3 | 0.154 |
| Major thrombi |
|
|
|
|
Negative | 37 | 11 |
|
|
Positive | 4 | 0 | 0.567 |
| Microvascular
thrombi |
|
|
|
|
Negative | 35 | 9 |
|
|
Positive | 6 | 2 | 0.856 |
| HCC stage |
|
|
|
| Early
stagea | 30 | 5 |
|
|
Advanced stageb | 11 | 6 | 0.145 |
| ICC stage |
|
|
|
| Early
stage | 32 | 7 |
|
|
Advanced stage | 9 | 4 | 0.435 |
Follow-up
Patients were followed up at least every 2 months
during the first year and every 3 months thereafter. Tumor markers,
including AFP, CEA and CA19-9 tests, liver ultrasonography,
computed tomography and magnetic resonance imaging, were selected
as required. OS was defined as the duration (in months) from the
date of surgery until cancer-specific mortality or last follow-up.
The final follow-up date was August 1, 2015.
Statistical analysis
The optimal cut-off values for NLR were determined
using time-dependent receiver operating curve (ROC) analysis.
Time-dependent ROC analysis was performed using R software version
3.2.2 (The R Foundation for Statistical Computing, Vienna, Austria;
http://www.r-project.org) and the ‘survival ROC’
package (22). The NLR was evaluated
by dividing the neutrophil cell counts by the lymphocyte cell
counts. The NLR value was categorized into two groups: NLR≤2.75 and
NLR>2.75.
SPSS version 22 (IBM Corp., Armonk, NY, USA) was
used to analyze the data. Continuous variables were expressed as
the mean ± standard deviation and the range, and were compared
between the NLR≤2.75 and NLR>2.75 groups using Student's t-test.
The χ2 test and Fisher's exact test were used to compare
categorical variables, which were presented as the number and
percentage of patients.
Survival curves for OS were analyzed using the
Kaplan-Meier method. Significant differences between groups were
identified using the log-rank test. A univariate analysis was
performed to assess significant differences in clinicopathological
characteristics. A multivariate analysis was performed via Cox
regression for variables significant in a univariate analysis, and
the associated 95% confidence interval (CI) was determined.
P<0.05 was considered to indicate a statistically significant
difference.
Results
Patient characteristics
A total of 59 patients were diagnosed with cHCC-CC
between January 2000 and October 2014, and consecutively enrolled
in the present retrospective study. The median patient age was 49
years (range, 25–75 years). Among these patients, 43 (72.9%) were
male and 16 (27.1%) were female. There were 40 (67.8%) patients in
the HCC early-stage group and 19 (32.2%) patients in the HCC
advanced-stage group, according to the TNM staging system.
Furthermore, according to the ICC TNM staging system, 45 (76.3%)
patients were in the early-stage group and 14 (23.7%) patients were
in the advanced-stage group at diagnosis.
Among the patients diagnosed with cHCC-CC, 52 were
included in the present study for prognostic analysis; the
remaining 7 were excluded, as their survival time was <3 months.
The study cohort for the prognostic analysis consisted of 37
(71.2%) males and 15 (28.8%) females, and was prospectively
recruited and retrospectively analyzed. The median age of the
patients in the prognostic analysis was 50 years (range 27–75
years).
The clinicopathological characteristics of the
investigated patients in the prognostic analysis are presented in
Table I. A total of 8 patients
(15.4%) were older than 60 years, and the majority of the patients
(71.2%) were male. Depending on the selected NLR value, patients
were divided into two groups: NLR≤2.75 and NLR>2.75. A total of
41 patients (78.8%) were in the NLR≤2.75 group, whereas 11 patients
(21.2%) were in the NLR>2.75 group. There were no significant
differences regarding age, gender, AFP, CA19-9, GGT, HbsAg, tumor
number, lymph node metastasis, major thrombi, microvascular
thrombi, HCC stage or ICC stage between the two groups; however,
tumor size was significantly larger in the NLR>2.75 group
compared with that in the NLR≤2.75 group (log-rank test,
P=0.017).
Association between NLR and tumor
stage
The association between NLR and tumor stage, which
was the primary determinant of patient prognosis, was evaluated. As
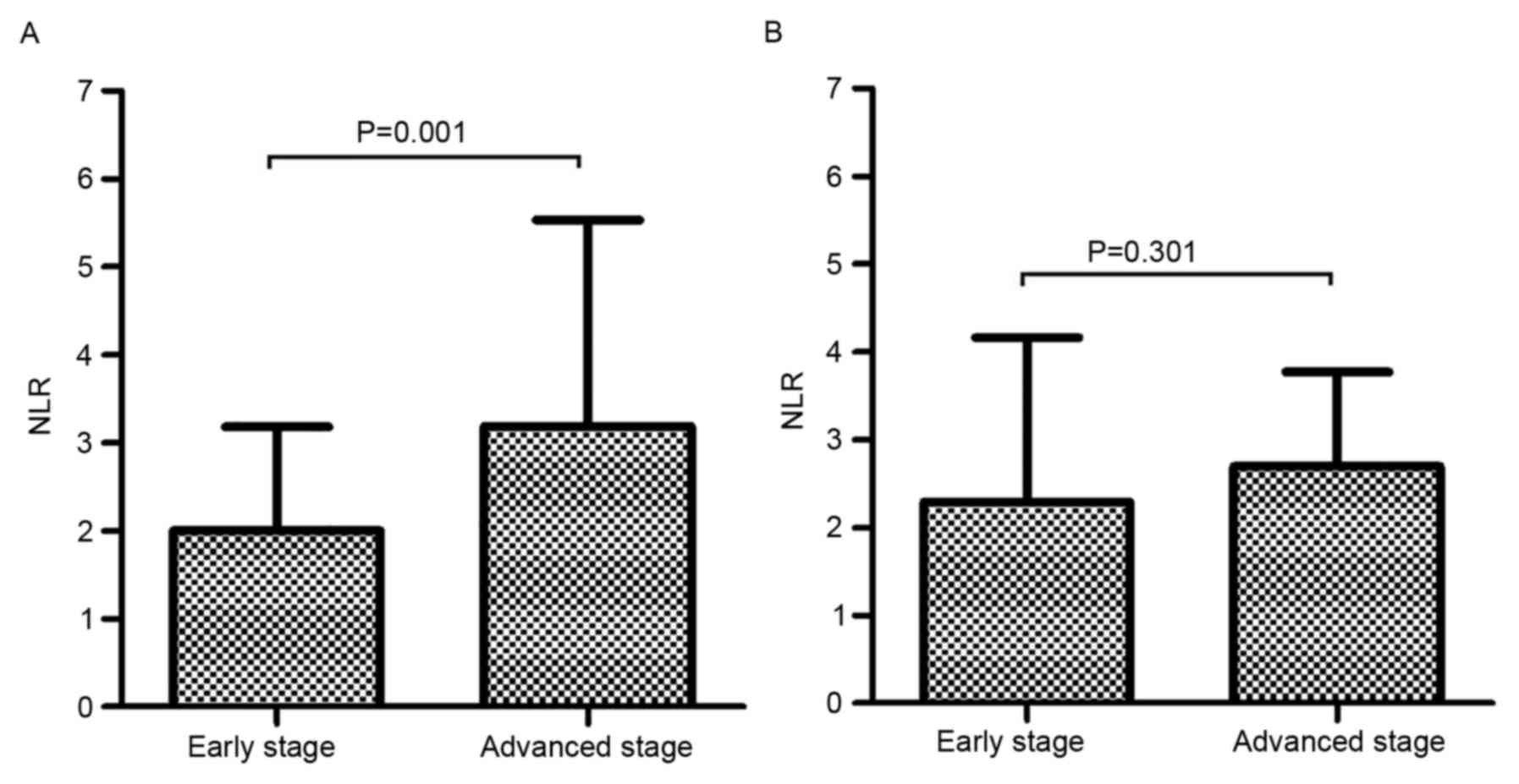
presented in Fig. 1A, the NLR value
was compared between the early-stage group (HCC stage I and II) and
the advanced-stage group (HCC stage III and IV), according to the
TNM staging system. The NLR value was significantly higher in the
advanced-stage group compared with that in the early-stage group
(3.19±2.34 vs. 2.00±1.17; P=0.001). However, there was no
difference in NLR value between the ICC early-stage group and the
ICC advanced-stage group according to TNM staging system (2.29±1.87
vs. 1.70±1.08; P=0.301; Fig. 1B).
Overall survival according to NLR
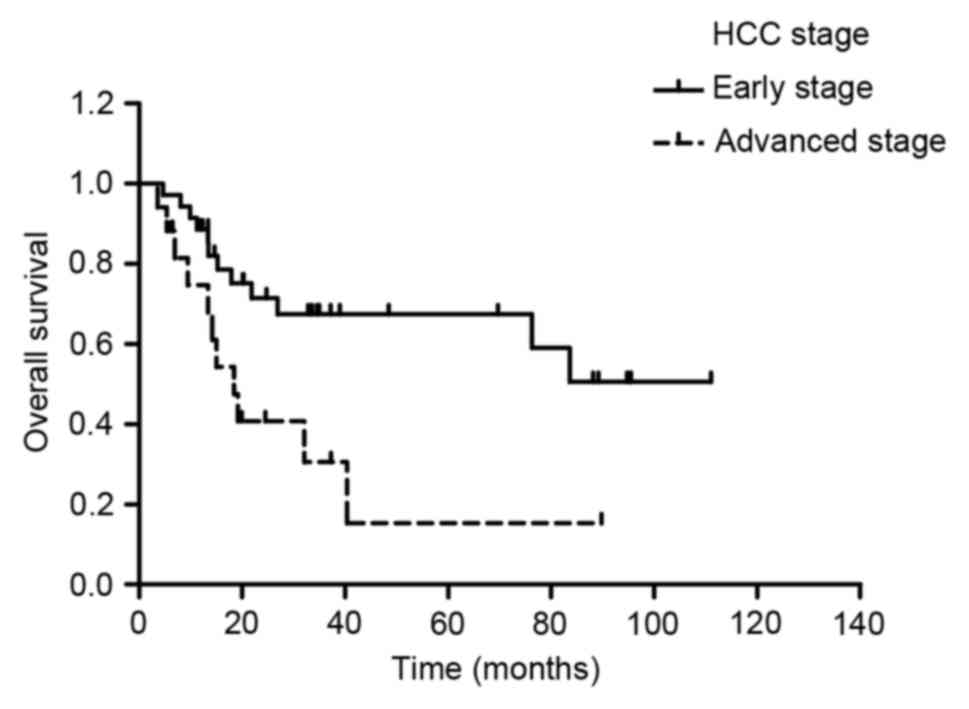
The OS rates of the patients in the advanced-stage
group were significantly poorer compared with those in the
early-stage group, according to the HCC staging system (1-year OS:
74.7 vs. 88.6%; 2-year OS: 40.7 vs. 71.4%; P=0.009; Fig. 2). However, the difference in OS rates
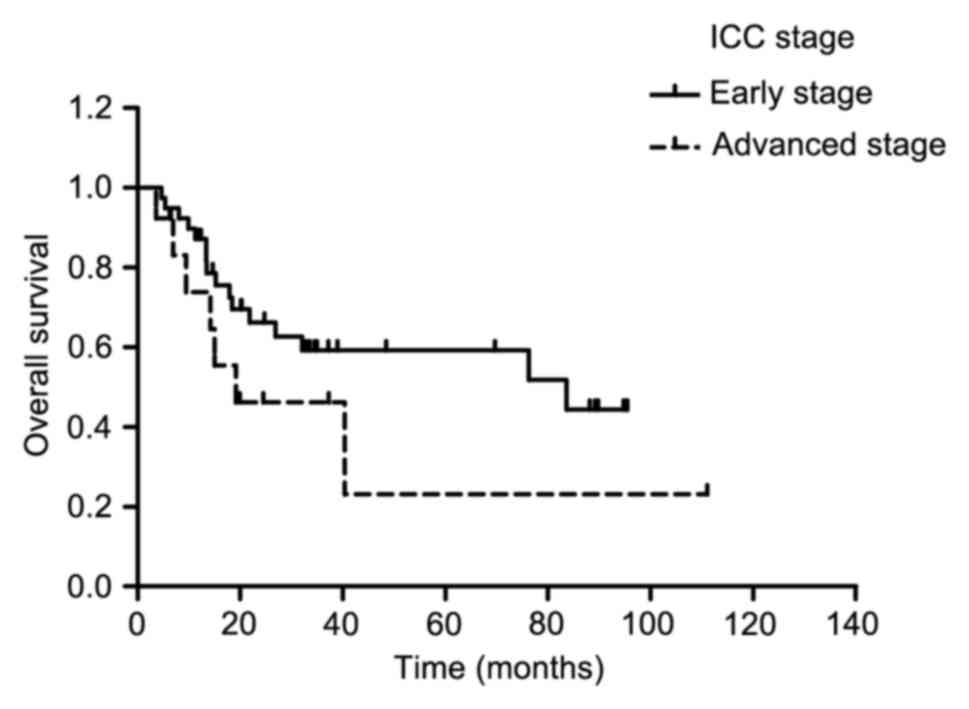
between the early-stage and advanced-stage patients in the ICC
stage system were not significant (1-year OS: 87.7 vs. 66.2%;
2-year OS: 73.8 vs. 46.2%; P=0.169; Fig.
3).
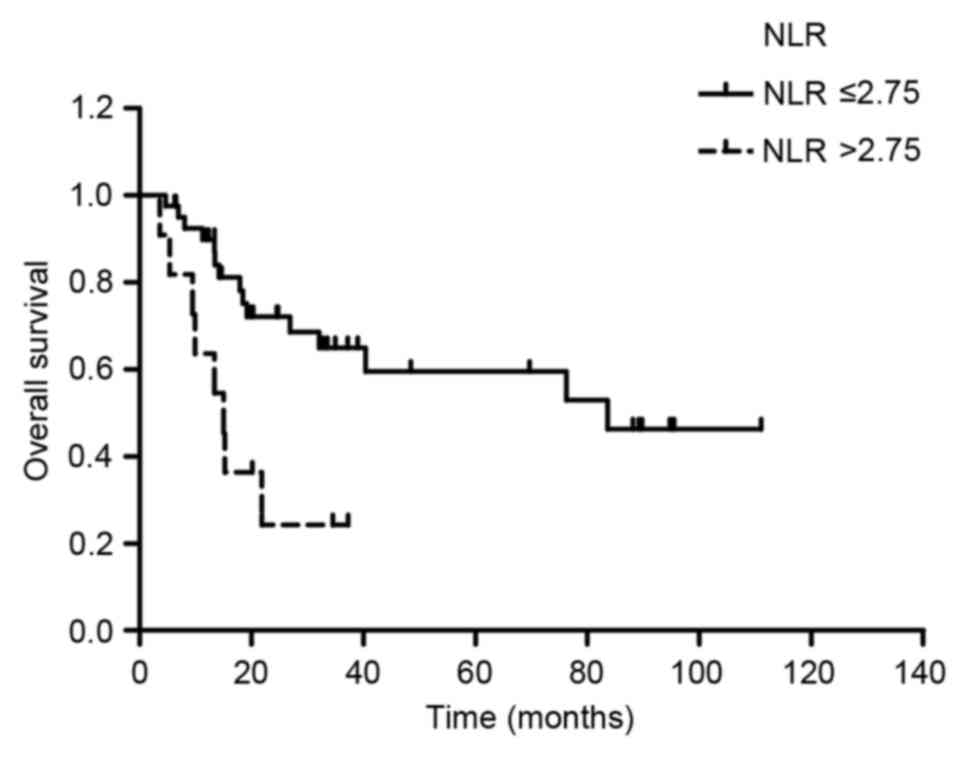
The OS rates of the patients with NLR≥2.75 were
significantly lower compared with those of the patients with
NLR≤2.75 (1-year OS: 63.6 vs. 89.9%; 2-year OS: 24.2 vs. 72.1%;
P=0.004). The median survival time was longer in the NLR≤2.75 group
compared with that in the NLR>2.75 group (83.6 vs. 15 months;
P=0.004) (Fig. 4).
Prognostic significance of NLR in
patients with cHCC-CC
The clinicopathological parameters were included in
the univariate and multivariate analyses to further investigate the
prognostic factors of patients with cHCC-CC. As presented in
Table II, tumor size, HCC TNM stage
and NLR were significant factors associated with OS in the
univariate analysis. The significant predictors were then utilized
for multivariate proportional hazard regression analysis. The
multiple analysis results revealed that advanced stage in the HCC
TNM staging system [hazard ratio (HR), 2.527; 95% CI, 1.088–5.872;
P=0.031] and NLR>2.75 (HR, 2.990; 95% CI, 1.198–7.462; P=0.019)
were independent prognostic factors of poor OS in cHCC-CC (Table II).
 | Table II.Univariate and multivariate analyses
of factors associated with overall survival in 52 patients with
combined hepatocellular-cholangiocarcinoma. |
Table II.
Univariate and multivariate analyses
of factors associated with overall survival in 52 patients with
combined hepatocellular-cholangiocarcinoma.
|
|
|
| Univariate | Multivariate |
|---|
|
|
|
|
|
|
|---|
|
Characteristics | n | % | HR (95% CI) | P-value | HR (95% CI) | P-value |
|---|
| Age, years |
|
|
|
|
|
|
|
≤60 | 44 | 84.6 | Reference
value |
|
|
|
|
>60 | 8 | 15.4 | 0.716
(0.212–2.417) | 0.590 |
|
|
| Gender |
|
|
|
|
|
|
|
Female | 15 | 28.8 | Reference
value |
|
|
|
|
Male | 37 | 71.2 | 1.487
(0.551–4.015) | 0.434 |
|
|
| AFP, ng/ml |
|
|
|
|
|
|
|
≤25 | 20 | 38.5 | Reference
value |
|
|
|
|
>25 | 32 | 61.5 | 1.296
(0.546–3.077) | 0.556 |
|
|
| CA19-9, U/ml |
|
|
|
|
|
|
|
≤35 | 36 | 69.2 | Reference
value |
|
|
|
|
>35 | 16 | 30.8 | 1.127
(0.477–2.664) | 0.785 |
|
|
| GGT, U/l |
|
|
|
|
|
|
|
≤50 | 23 | 44.2 | Reference
value |
|
|
|
|
>50 | 29 | 55.8 | 1.788
(0.756–4.229) | 0.186 |
|
|
| HbsAg |
|
|
|
|
|
|
|
Negative | 6 | 11.5 | Reference
value |
|
|
|
|
Positive | 46 | 88.5 | 3.510
(0.470–26.239) | 0.221 |
|
|
| Tumor size, cm |
|
|
|
|
|
|
|
<5 | 26 | 50.0 | Reference
value |
|
|
|
| ≥5 | 26 | 50.0 | 3.475
(1.411–8.562) | 0.007 | 1.113
(0.959–1.291) | 0.159 |
| Tumor number |
|
|
|
|
|
|
|
Solitary | 14 | 26.9 | Reference
value |
|
|
|
|
Multiple | 38 | 73.1 | 0.955
(0.375–2.429) | 0.923 |
|
|
| Lymph node
metastasis |
|
|
|
|
|
|
|
Negative | 45 | 26.5 | Reference
value |
|
|
|
|
Positive | 7 | 13.5 | 3.036
(0.992–9.292) | 0.052 |
|
|
| Major thrombus |
|
|
|
|
|
|
|
Negative | 48 | 92.3 | Reference
value |
|
|
|
|
Positive | 4 | 7.7 | 3.121
(0.912–10.684) | 0.070 |
|
|
| Microvascular
thrombus |
|
|
|
|
|
|
|
Negative | 44 | 84.6 | Reference
value |
|
|
|
|
Positive | 8 | 15.4 | 1.676
(0.556–5.054) | 0.305 |
|
|
| HCC stage |
|
|
|
|
|
|
| Early
stagea | 35 | 67.3 | Reference
value |
|
|
|
|
Advanced stageb | 17 | 32.7 | 2.882
(1.258–6.607) | 0.012 | 2.527
(1.088–5.872) | 0.031 |
| ICC stage |
|
|
|
|
|
|
| Early
stage | 39 | 75.0 | Reference
value |
|
|
|
|
Advanced stage | 13 | 25.0 | 1.853
(0.758–4.528) | 0.176 |
|
|
| NLR |
|
|
|
|
|
|
|
≤2.75 | 41 | 78.8 | Reference
value |
|
|
|
|
>2.75 | 11 | 21.2 | 3.474
(1.409–8.563) | 0.007 | 2.990
(1.198–7.462) | 0.019 |
Discussion
cHCC-CC is a mixed carcinoma that is composed of two
distinct tumor elements in which HCC and CC intimately coexist
(4). The present study confirmed the
prognostic value of NLR in a cohort of patients with cHCC-CC.
Inflammatory processes have been identified to serve
a role in tumor progression (9).
Growth and survival factors released from inflammatory cells can
stimulate tumor formation, progression, angiogenesis, invasion and
metastasis (23–25). The paradoxical roles of adaptive
(lymphocyte immune cells) and innate leukocytes (circulating
neutrophils) in inflammatory processes act as crucial opposing
regulators in cancer occurrence (26). Immune cell-like neutrophils have been
associated with increased angiogenesis and/or a poor prognosis,
which is in part explained by the upregulation of cyclooxygenase-2
or the suppression of an antitumor adaptive immune response
(27–29). However, lymphocytes have been
essential components in tumor defense via killing tumor cells and
inhibiting cell proliferation or migration (9,30).
Additionally, certain previous studies suggested that adaptive
immune cells, including B-lymphocytes, cluster of differentiation
(CD)8+ cytotoxic T-lymphocytes and CD4+
helper T-lymphocytes, served important roles in the modulation of
cancer development via the lysis of tumor cells (26,31).
Inflammation is a complex process that may be
reflected by NLR, a practical biomarker. The present study
demonstrated a substantial difference in NLR between early- and
advanced-stage HCC (P=0.001); however, there were no significant
differences in NLR values between early and advanced ICC stage
according to the TNM staging system (P=0.301). cHCC-CC has been
included in the ICC section of the TNM staging system in accordance
with the AJCC manual (21); however,
a study with a larger cohort suggested that the HCC TNM staging
system provided an better prognostic stratification for patients
with cHCC-CC (6). Similarly, in the
present study, the OS rates of patients with advanced HCC stage
were significantly lower compared with those of patients with early
stage disease, according to the TNM staging system. However, there
were no differences in OS between the early and advanced ICC
stages, according to the TNM staging system (P=0.301). Furthermore,
a recent study revealed that the NLR value exhibited a linear
association with cancer progression staging (17). This may partly explain the difference
in NLR value between early- and advanced-stage HCC present in the
TNM staging system but not the ICC TNM staging system in the
present study.
Increasing evidence has suggested that NLR is a
prognostic factor for OS in various types of cancer. Various
cut-off values have been used to describe the association between
NLR and survival in these cancer types (32,33). In
the present study, a time-dependent ROC was performed to determine
the optimal cut-off points for NLR. According to this, the present
study demonstrated that patients could be divided into two groups
(NLR>2.75 and NLR≤2.75). Upon univariate and multivariate
analyses, the present study confirmed that NLR was an independent
prognostic factor. In contrast with the present study, a previous
study revealed that an NLR value of >2 did not independently
predict poorer overall survival in patients with cHCC-CC (34). This discrepancy may be due to
differences in the assays using NLR. The previous study selected
the median value as a cut-off point, whereas the present study used
a time-dependent ROC curve to select a high-sensitivity value
cut-off point for NLR.
There are certain limitations to the present study.
Firstly, the total number of recruited patients was relatively
small. Secondly, it was a retrospective study and was thus
susceptible to bias in data selection and analysis. Other
inflammatory markers, including C-reactive protein or
procalcitonin, which have been demonstrated to be independent
prognostic factors in patients with HCC (17) and ICC (35), are not routinely evaluated at Sun
Yat-sen University. Finally, the present study did not evaluate NLR
following surgery, and further investigation is required to
determine whether this was associated with OS.
Despite these limitations, the present study
suggested that pre-treatment NLR may be associated with OS in
patients diagnosed with cHCC-CC and that analysis of the NLR should
be introduced in clinical practice. Further prospective studies are
required to evaluate cut-off values and confirm the results of the
present study.
Acknowledgements
The present study was supported by National Natural
Science Foundation of China (grant no. 81171890) and the Major
National Scientific Research Projects of China (grant no.
2013CB910304).
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Allen RA and Lisa JR: Combined liver cell
and bile duct carcinoma. Am J Pathol. 25:647–655. 1949.PubMed/NCBI
|
|
2
|
Jarnagin WR, Weber S, Tickoo SK, Koea JB,
Obiekwe S, Fong Y, DeMatteo RP, Blumgart LH and Klimstra D:
Combined hepatocellular and cholangiocarcinoma: Demographic,
clinical, and prognostic factors. Cancer. 94:2040–2046. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Panjala C, Senecal DL, Bridges MD, Kim GP,
Nakhleh RE, Nguyen JH and Harnois DM: The diagnostic conundrum and
liver transplantation outcome for combined
hepatocellular-cholangiocarcinoma. Am J Transplant. 10:1263–1267.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bosman FT, Carneiro F, Hruban RH and
Theise ND: WHO classification of tumours of the digestive system.
4th. International Agency for Research on Cancer Publisher, Lyon.
World Health Organization; 2010
|
|
5
|
Kim SH, Park YN, Lim JH, Choi GH, Choi JS
and Kim KS: Characteristics of combined
hepatocelluar-cholangiocarcinoma and comparison with intrahepatic
cholangiocarcinoma. Eur J Surg Oncol. 40:976–981. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chu KJ, Lu CD, Dong H, Fu XH, Zhang HW and
Yao XP: Hepatitis B virus-related combined
hepatocellular-cholangiocarcinoma: Clinicopathological and
prognostic analysis of 390 cases. Eur J Gastroenterol Hepatol.
26:192–199. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zheng W, Yao M, Sai W, Qian Q, Pan L, Qiu
L, Huang J, Wu W and Yao D: Diagnostic and prognostic significance
of secretory clusterin expression in patients with hepatocellular
carcinoma. Tumour Biol. 37:999–1008. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kassahun WT and Hauss J: Management of
combined hepatocellular and cholangiocarcinoma. Int J Clin Pract.
62:1271–1278. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Coussens LM and Werb Z: Inflammation and
cancer. Nature. 420:860–867. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zahorec R: Ratio of neutrophil to
lymphocyte counts-rapid and simple parameter of systemic
inflammation and stress in critically ill. Bratisl Lek Listy.
102:5–14. 2001.(In English, Slovak). PubMed/NCBI
|
|
11
|
Sarraf KM, Belcher E, Raevsky E, Nicholson
AG, Goldstraw P and Lim E: Neutrophil/lymphocyte ratio and its
association with survival after complete resection in non-small
cell lung cancer. J Thorac Cardiovasc Surg. 137:425–428. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yamanaka T, Matsumoto S, Teramukai S,
Ishiwata R, Nagai Y and Fukushima M: The baseline ratio of
neutrophils to lymphocytes is associated with patient prognosis in
advanced gastric cancer. Oncology. 73:215–220. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Cho H, Hur HW, Kim SW, Kim SH, Kim JH, Kim
YT and Lee K: Pre-treatment neutrophil to lymphocyte ratio is
elevated in epithelial ovarian cancer and predicts survival after
treatment. Cancer Immunol Immunother. 58:15–23. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
An X, Ding PR, Li YH, Wang FH, Shi YX,
Wang ZQ, He YJ, Xu RH and Jiang WQ: Elevated neutrophil to
lymphocyte ratio predicts survival in advanced pancreatic cancer.
Biomarkers. 15:516–522. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gomez D, Farid S, Malik HZ, Young AL,
Toogood GJ, Lodge JP and Prasad KR: Preoperative
neutrophil-to-lymphocyte ratio as a prognostic predictor after
curative resection for hepatocellular carcinoma. World J Surg.
32:1757–1762. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kayadibi H, Sertoglu E, Uyanik M and Tapan
S: Neutrophil-lymphocyte ratio is useful for the prognosis of
patients with hepatocellular carcinoma. World J Gastroenterol.
20:9631–9632. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Oh BS, Jang JW, Kwon JH, You CR, Chung KW,
Kay CS, Jung HS and Lee S: Prognostic value of C-reactive protein
and neutrophil-to-lymphocyte ratio in patients with hepatocellular
carcinoma. BMC Cancer. 13:782013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Xiao WK, Chen D, Li SQ, Fu SJ, Peng BG and
Liang LJ: Prognostic significance of neutrophil-lymphocyte ratio in
hepatocellular carcinoma: A meta-analysis. BMC Cancer. 14:1172014.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Shen SL, Fu SJ, Huang XQ, Chen B, Kuang M,
Li SQ, Hua YP, Liang LJ and Peng BG: Elevated preoperative
peripheral blood monocyte count predicts poor prognosis for
hepatocellular carcinoma after curative resection. BMC Cancer.
14:7442014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chen Q, Yang LX, Li XD, Yin D, Shi SM,
Chen EB, Yu L, Zhou ZJ, Zhou SL, Shi YH, et al: The elevated
preoperative neutrophil-to-lymphocyte ratio predicts poor prognosis
in intrahepatic cholangiocarcinoma patients undergoing hepatectomy.
Tumour Biol. 36:5283–5289. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Edge S, Byrd D, Compton C, Fritz A, Greene
F and Trotti A: AJCC cancer staging manual. 7. American Joint
Committee on Cancer; 2010
|
|
22
|
Heagerty PJ, Lumley T and Pepe MS:
Time-dependent ROC curves for censored survival data and a
diagnostic marker. Biometrics. 56:337–344. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Jaiswal M, LaRusso NF, Burgart LJ and
Gores GJ: Inflammatory cytokines induce DNA damage and inhibit DNA
repair in cholangiocarcinoma cells by a nitric oxide-dependent
mechanism. Cancer Res. 60:184–190. 2000.PubMed/NCBI
|
|
24
|
de Visser KE, Eichten A and Coussens LM:
Paradoxical roles of the immune system during cancer development.
Nat Rev Cancer. 6:24–37. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jackson JR, Seed MP, Kircher CH,
Willoughby DA and Winkler JD: The codependence of angiogenesis and
chronic inflammation. FASEB J. 11:457–465. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ishigami S, Natsugoe S, Tokuda K, Nakajo
A, Che X, Iwashige H, Aridome K, Hokita S and Aikou T: Prognostic
value of intratumoral natural killer cells in gastric carcinoma.
Cancer. 88:577–583. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Liu CH, Chang SH, Narko K, Trifan OC, Wu
MT, Smith E, Haudenschild C, Lane TF and Hla T: Overexpression of
cyclooxygenase-2 is sufficient to induce tumorigenesis in
transgenic mice. J Biol Chem. 276:18563–18569. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Schoppmann SF, Birner P, Stöckl J, Kalt R,
Ullrich R, Caucig C, Kriehuber E, Nagy K, Alitalo K and Kerjaschki
D: Tumor-associated macrophages express lymphatic endothelial
growth factors and are related to peritumoral lymphangiogenesis. Am
J Pathol. 161:947–956. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Dannenberg AJ and Subbaramaiah K:
Targeting cyclooxygenase-2 in human neoplasia: Rationale and
promise. Cancer Cell. 4:431–436. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Mantovani A, Allavena P, Sica A and
Balkwill F: Cancer-related inflammation. Nature. 454:436–444. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zou W: Immunosuppressive networks in the
tumour environment and their therapeutic relevance. Nat Rev Cancer.
5:263–274. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
32
|
Perez DR, Baser RE, Cavnar MJ,
Balachandran VP, Antonescu CR, Tap WD, Strong VE, Brennan MF, Coit
DG, Singer S and Dematteo RP: Blood neutrophil-to-lymphocyte ratio
is prognostic in gastrointestinal stromal tumor. Ann Surg Oncol.
20:593–599. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Williams KA, Labidi-Galy SI, Terry KL,
Vitonis AF, Welch WR, Goodman A and Cramer DW: Prognostic
significance and predictors of the neutrophil-to-lymphocyte ratio
in ovarian cancer. Gynecol Oncol. 132:542–550. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Cai X, Zhai J, Kaplan DE, Zhang Y, Zhou L,
Chen X, Qian G, Zhao Q, Li Y, Gao L, et al: Background progenitor
activation is associated with recurrence after hepatectomy of
combined hepatocellular-cholangiocarcinoma. Hepatology.
56:1804–1816. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zeng X and Tao H: Diagnostic and
prognostic serum marker of cholangiocarcinoma (Review). Oncol Lett.
9:3–8. 2015. View Article : Google Scholar : PubMed/NCBI
|