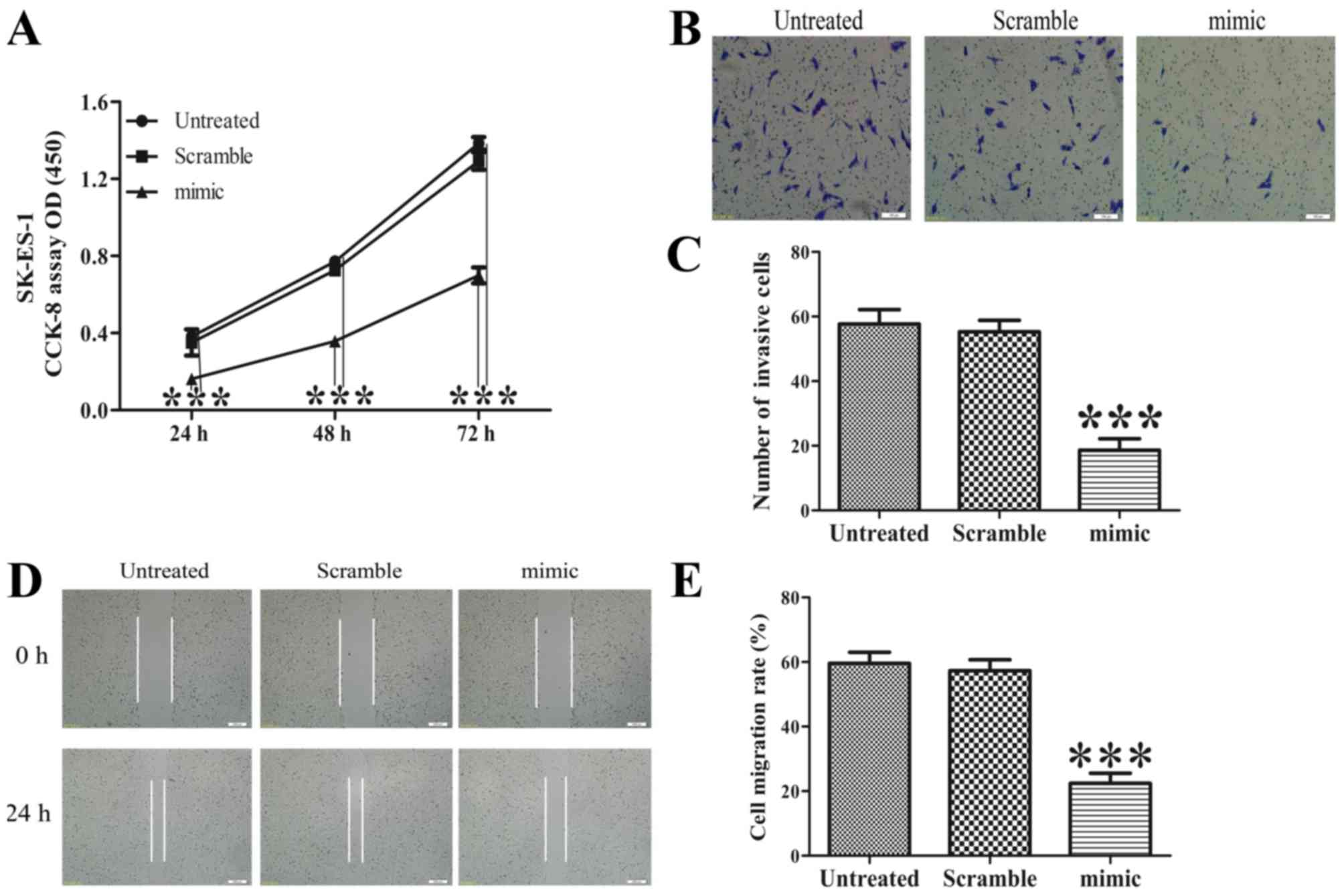
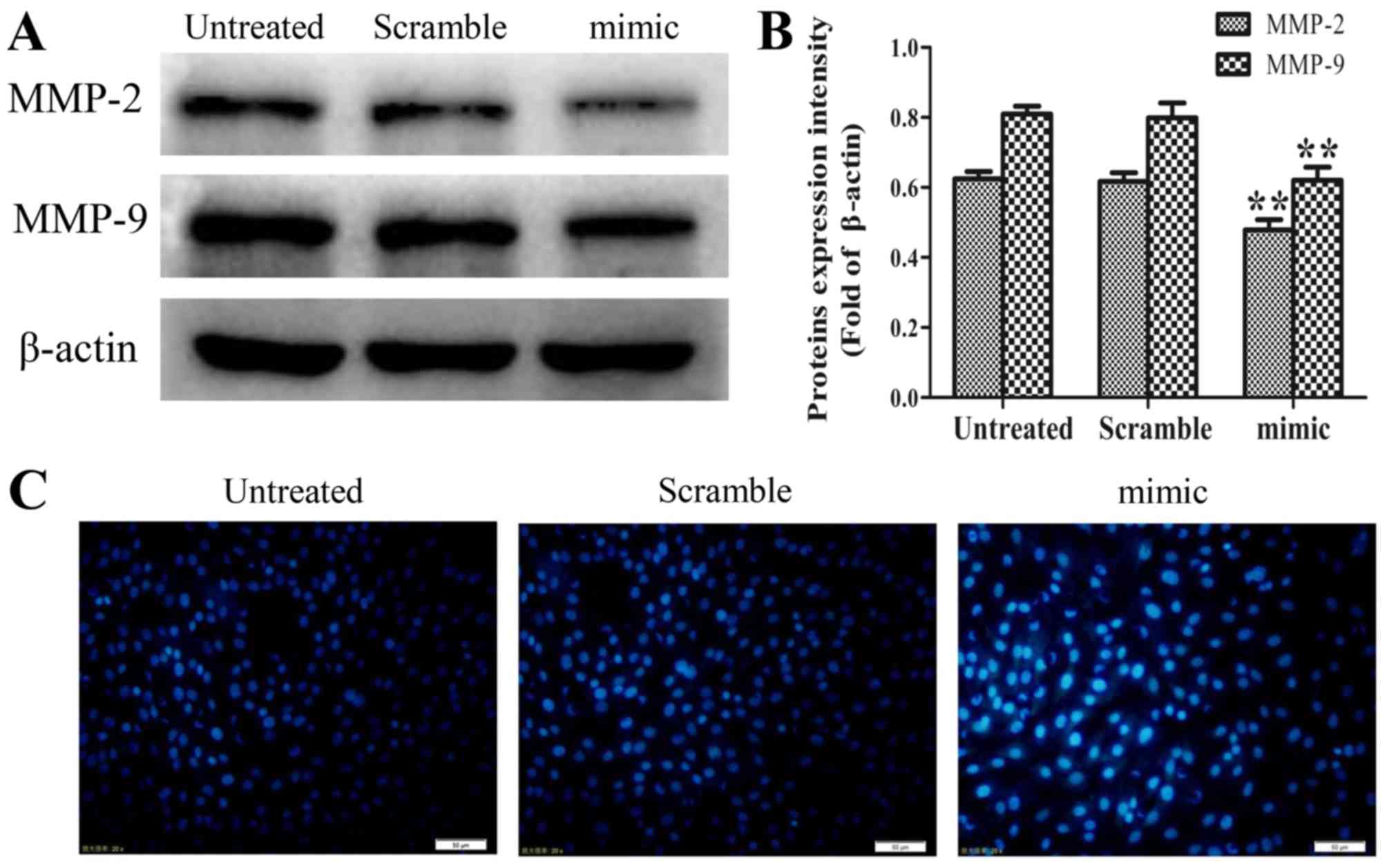
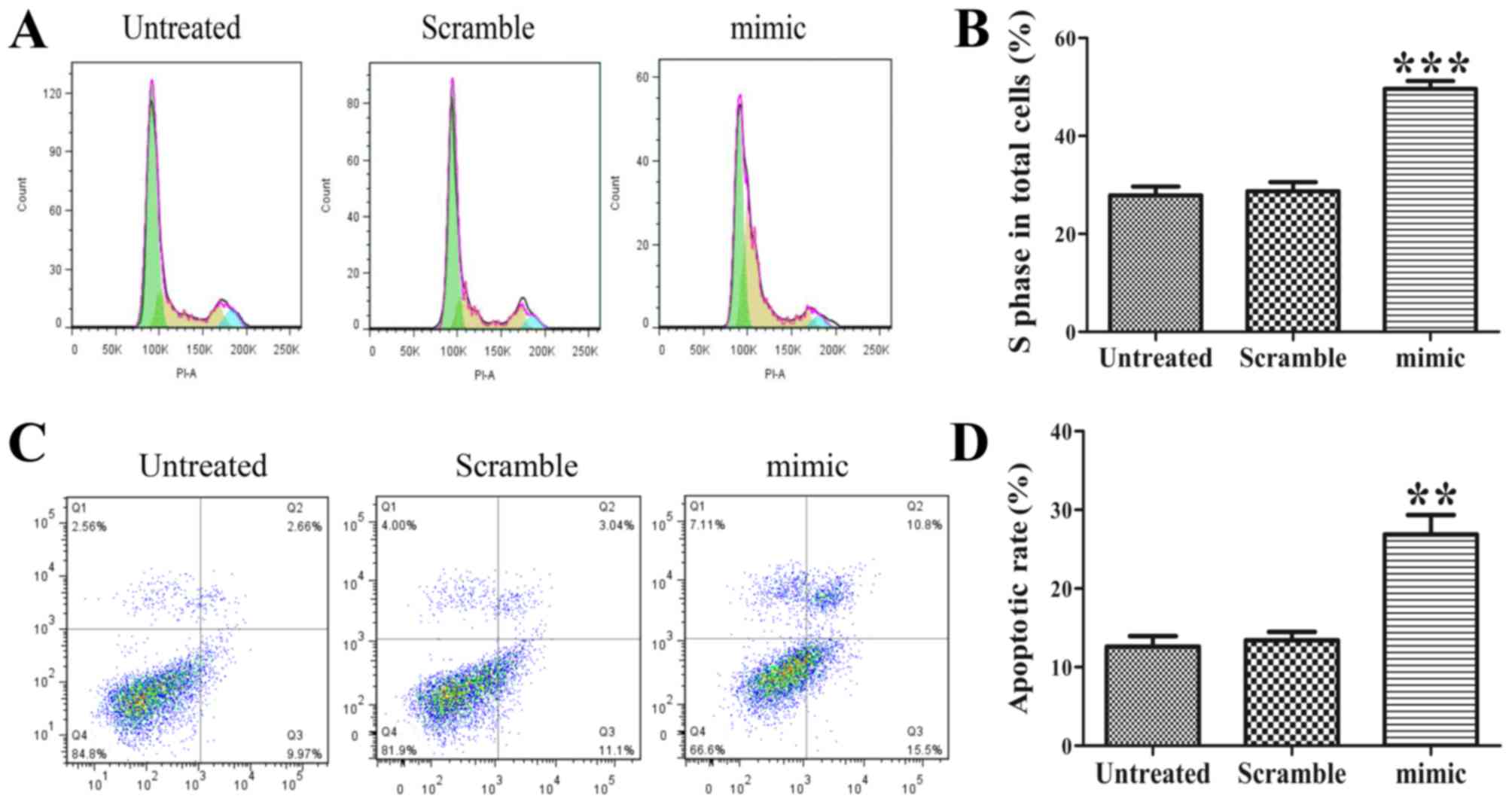
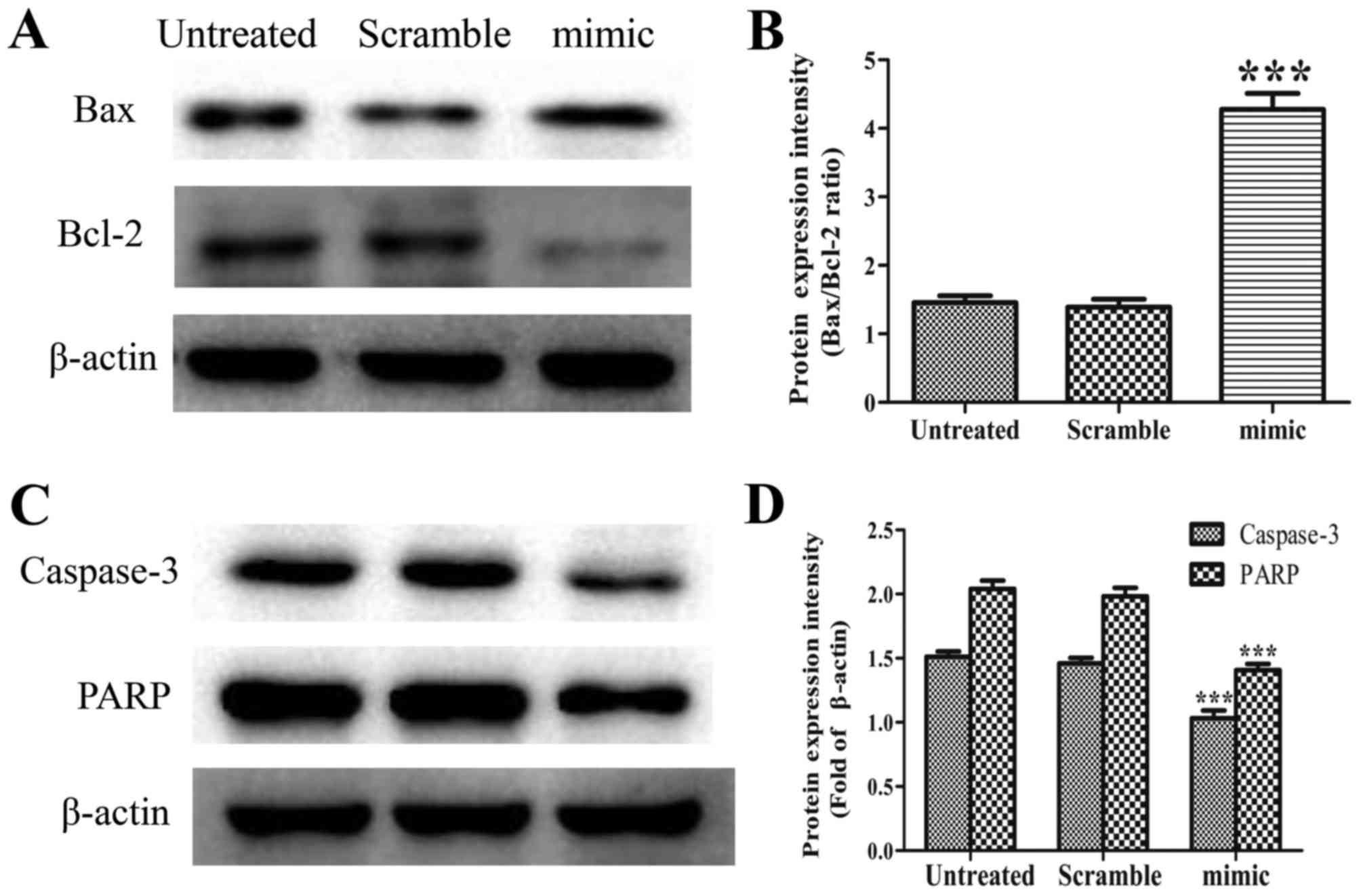
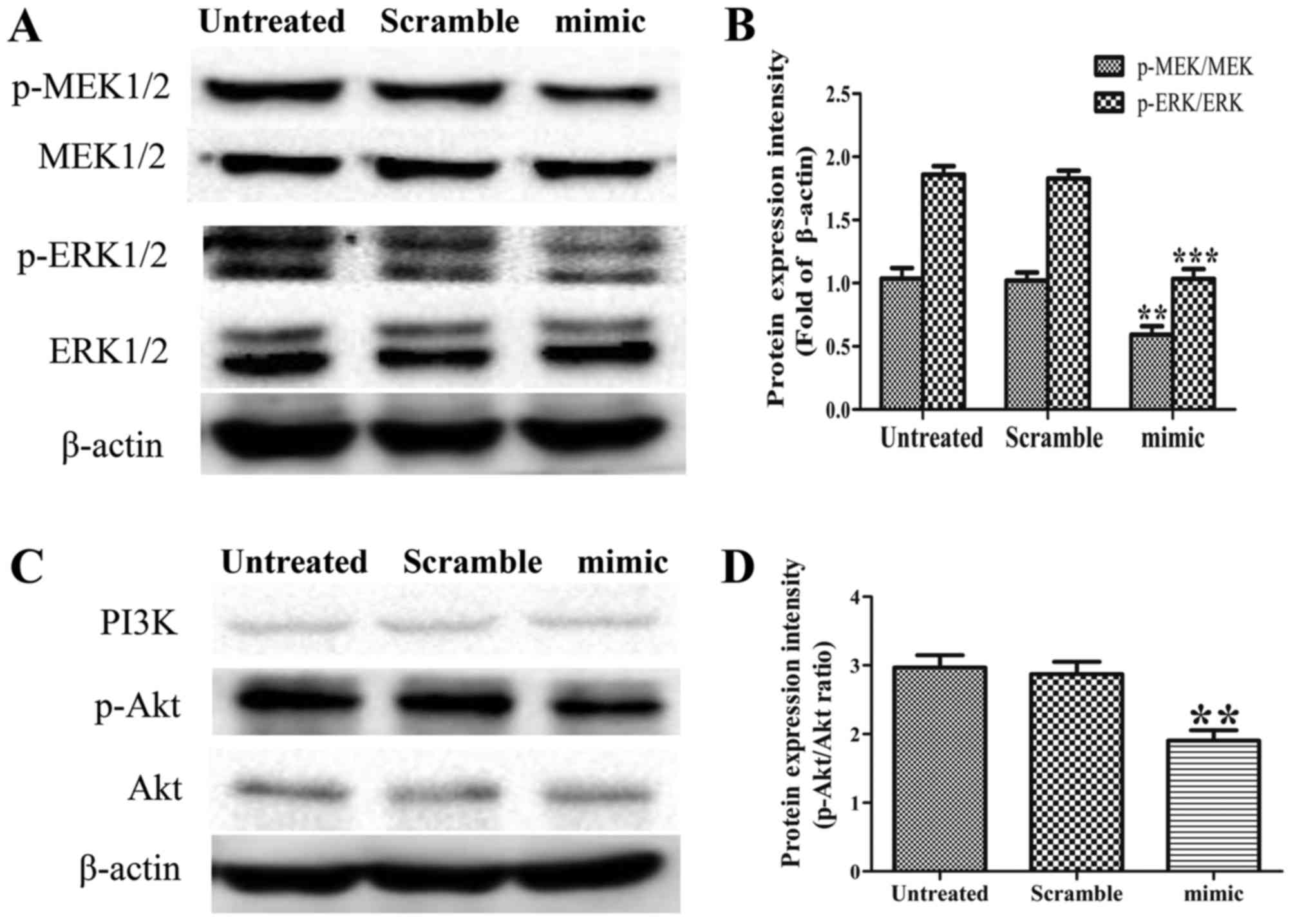
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Gaspar N, Hawkins DS, Dirksen U, Lewis IJ,
Ferrari S, Le Deley MC, Kovar H, Grimer R, Whelan J, Claude L, et
al: Ewing sarcoma: Current management and future approaches through
collaboration. J Clin Oncol. 33:3036–3046. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Medina-Villaamil V, Martínez-Breijo S,
Portela-Pereira P, Quindós-Varela M, Santamarina-Caínzos I,
Antón-Aparicio LM and Gómez-Veiga F: Circulating microRNAs in blood
of patients with prostate cancer. Actas Urol Esp. 38:633–639. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yang M, Liu R, Sheng J, Liao J, Wang Y,
Pan E, Guo W, Pu Y and Yin L: Differential expression profles of
microRNAs as potential biomarkers for the early diagnosis of
esophageal squamous cell carcinoma. Oncol Rep. 29:169–176. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Leite KR, Tomiyama A, Reis ST,
Sousa-Canavez JM, Sañudo A, Camara-Lopes LH and Srougi M: MicroRNA
expression profles in the progression of prostate cancer-from
high-grade prostate intraepithelial neoplasia to metastasis. Urol
Oncol. 31:796–801. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li Y, Shao G, Zhang M, Zhu F, Zhao B, He C
and Zhang Z: miR-124 represses the mesenchymal features and
suppresses metastasis in Ewing sarcoma. Oncotarget. 8:10274–10286.
2017.PubMed/NCBI
|
|
7
|
Schwentner R, Herrero-Martin D, Kauer MO,
Mutz CN, Katschnig AM, Sienski G, Alonso J, Aryee DN and Kovar H:
The role of miR-17-92 in the miRegulatory landscape of Ewing
sarcoma. Oncotarget. 8:10980–10993. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kawano M, Tanaka K, Itonaga I, Iwasaki T
and Tsumura H: MicroRNA-301a promotes cell proliferation via PTEN
targeting in Ewing's sarcoma cells. Int J Oncol. 48:1531–1540.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Esposito F, Tornincasa M, Pallante P,
Federico A, Borbone E, Pierantoni GM and Fusco A: Down-regulation
of the miR-25 and miR-30d contributes to the development of
anaplastic thyroid carcinoma targeting the polycomb protein EZH2. J
Clin Endocrinol Metab. 97:E710–E718. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yu H, Lin X, Wang F, Zhang B, Wang W, Shi
H, Zou B and Zhao J: Proliferation inhibition and the underlying
molecular mechanisms of microRNA-30d in renal carcinoma cells.
Oncol Lett. 7:799–804. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chen D, Guo W, Qiu Z, Wang Q, Li Y, Liang
L, Liu L, Huang S, Zhao Y and He X: MicroRNA-30d-5p inhibits tumour
cell proliferation and motility by directly targeting CCNE2 in
non-small cell lung cancer. Cancer Lett. 362:208–217. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kobayashi N, Uemura H, Nagahama K, Okudela
K, Furuya M, Ino Y, Ito Y, Hirano H, Inayama Y, Aoki I, et al:
Identification of miR-30d as a novel prognostic maker of prostate
cancer. Oncotarget. 3:1455–1471. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li H, Zhang K, Liu LH, Ouyang Y, Bu J, Guo
HB and Xiao T: A systematic review of matrix metalloproteinase 9 as
a biomarker of survival in patients with osteosarcoma. Tumour Biol.
35:5487–5491. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wang J, Shi Q, Yuan TX, Song QL, Zhang Y,
Wei Q, Zhou L, Luo J, Zuo G, Tang M, et al: Matrix
metalloproteinase 9 (MMP-9) in osteosarcoma: Review and
meta-analysis. Clin Chim Acta. 433:225–231. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shang HS, Chang JB, Lin JH, Lin JP, Hsu
SC, Liu CM, Liu JY, Wu PP, Lu HF, Au MK and Chung JG: Deguelin
inhibits the migration and invasion of U-2 OS human osteosarcoma
cells via the inhibition of matrix metalloproteinase-2/-9 in vitro.
Molecules. 19:16588–16608. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Vinatier D, Dufour P and Subtil D:
Caspases: Pharmacological manipulation of cell death. Eur J Obstet
Gynecol Reprod Biol. 67:85–102. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Norbury CJ and Zhivotovsky B: DNA
damage-induced apoptosis. Oncogene. 23:2797–2808. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Soldani C and Scovassi AI:
Poly(ADP-ribose) polymerase-1 cleavage during apoptosis: An update.
Apoptosis. 7:321–328. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Orrenius S: Mitochondrial regulation of
apoptotic cell death. Toxicol Lett. 149:19–23. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Scorrano L and Korsmeyer SJ: Mechanisms of
cytochrome c release by proapoptotic Bcl-2 family members. Biochem
Biophys Res Commun. 304:437–444. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Meeran SM and Katiyar SK: Grape seed
proanthocyanidins promote apoptosis in human epidermoid carcinoma
A431 cells through alterations in Cdki-Cdk-cyclin cascade, and
caspase-3 activation via loss of mitochondrial membrane potential.
Exp Dermatol. 16:405–415. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zenali MJ, Zhang PL, Bendel AE and Brown
RE: Morphoproteomic confrmation of constitutively activated mTOR,
ERK, and NF-kappaB pathways in Ewing family of tumors. Ann Clin Lab
Sci. 39:160–166. 2009.PubMed/NCBI
|
|
23
|
Roskoski R Jr: ERK1/2 MAP kinases:
Structure, function and regulation. Pharmacol Res. 66:105–143.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Datta SR, Brunet A and Greenberg ME:
Cellular survival: A play in three Akts. Genes Dev. 13:2905–2927.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Toretsky JA, Thakar M, Eskenazi AE and
Frantz CN: Phosphoinositide 3-hydroxide kinase blockade enhances
apoptosis in the Ewing's sarcoma family of tumors. Cancer Res.
59:5745–5750. 1999.PubMed/NCBI
|
|
26
|
Balamuth NJ and Womer RB: Ewing's sarcoma.
Lancet Oncol. 11:184–192. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Iwamoto Y: Diagnosis and treatment of
Ewing's sarcoma. Jpn J Clin Oncol. 37:79–89. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gorlick R, Janeway K, Lessnick S, Randall
RL and Marina N; COG bone tumor committee, : Children's oncology
group's 2013 blueprint for research: Bone tumors. Pediatr Blood
Cancer. 60:1009–1015. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lee RC, Feinbaum RL and Ambros V: The C.
elegans heterochronic gene lin-4 encodes small RNAs with antisense
complementarity to lin-14. Cell. 75:843–854. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Šulc M, Marín RM, Robins HS and Vaníček J:
PACCMIT/PACCMIT-CDS: Identifying microRNA targets in 3′ UTRs and
coding sequences. Nucleic Acids Res. 43:W474–W479. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Fang Z, Tang J, Bai Y, Lin H, You H, Jin
H, Lin L, You P, Li J, Dai Z, et al: Plasma levels of microRNA-24,
microRNA-320a, and microRNA-423-5p are potential biomarkers for
colorectal carcinoma. J Exp Clin Cancer Res. 34:862015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Nishikawa R, Goto Y, Kurozumi A,
Matsushita R, Enokida H, Kojima S, Naya Y, Nakagawa M, Ichikawa T
and Seki N: MicroRNA-205 inhibits cancer cell migration and
invasion via modulation of centromere protein F regulating pathways
in prostate cancer. Int J Urol. 22:867–877. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Volinia S, Calin GA, Liu CG, Ambs S,
Cimmino A, Petrocca F, Visone R, Iorio M, Roldo C, Ferracin M, et
al: A microRNA expression signature of human solid tumors defines
cancer gene targets. Proc Natl Acad Sci USA. 103:pp. 2257–2261.
2006; View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhang Y, Yang WQ, Zhu H, Qian YY, Zhou L,
Ren YJ, Ren XC, Zhang L, Liu XP, Liu CG, et al: Regulation of
autophagy by miR-30d impacts sensitivity of anaplastic thyroid
carcinoma to cisplatin. Biochem Pharmacol. 87:562–570. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Gaziel-Sovran A, Segura MF, Di Micco R,
Collins MK, Hanniford D, Vega-Saenz de Miera E, Rakus JF, Dankert
JF, Shang S, Kerbel RS, et al: miR-30b/30d regulation of GalNAc
transferases enhances invasion and immunosuppression during
metastasis. Cancer Cell. 20:104–118. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Yao J, Liang L, Huang S, Ding J, Tan N,
Zhao Y, Yan M, Ge C, Zhang Z, Chen T, et al: MicroRNA-30d promotes
tumor invasion and metastasis by targeting Galphai2 in
hepatocellular carcinoma. Hepatology. 51:846–856. 2010.PubMed/NCBI
|