Introduction
In 1977, a group of scientists from Japan identified
adult T-cell lymphoma (ATL) as a helper T-cell malignancy (1). A year later, HuT-102 cells were
developed from a patient with cutaneous T-cell Lymphoma (2). HuT-102 cells were identified as a subset
of helper T-cells with high expression of IL-2 receptor and
constitutive IL-2 production, aiding in an autonomous proliferation
(2). In 1980, the human
T-lymphotropic virus 1 (HTLV-1) was identified in HuT-102 cells,
followed by its discovery in cells from ATL patient. Analysis of
serum samples from around the world provided information of endemic
areas for HTLV-1, and its role as an etiologic agent for ATL
(1).
Similar to freshly obtained cells from ATL patients,
HuT-102 cells is a helper T-cell cell that express Tax protein,
which promote these cells particularly as an excellent model for
studying ATL (3). Our previous
findings have identified the interferon regulatory factor (IRF)
family members as efficient mediators that modulate cytokines
profile in those cells, hence providing therapeutic solutions
(3). Although the microarray data
upon which we based our previous studies were used to elaborately
cover the roles of IRFs, particularly IRF3 and IRF4, on
Interferon-inducible genes, it was performed by manual selection of
related genes mentioned in the literature (3,4).
Therefore, we try in the current study to reanalyze the previous
data through more efficient computational tools. The aim of
browsing our previous data for further analysis is to find
important genes that we might have passed away, in order to
discover promising opportunities against ATL.
Materials and methods
Gene expression was analyzed using a GeneChip system
with Human Genome Array U133 Plus2.0 (Affymetrix; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) as described previously
(3,4).
The arrays were performed independently in two studies as follow, a
total of four arrays for the first study: Two for HuT102-shLuc
cells and two for HuT102-shTAK1 cells (deposited in the GEO
database under accession no. GSE16219). In the second study, six
arrays were used: Two for HuT102-siLuc cells, two for HuT102-siIRF4
cells, and two for HuT102-siIRF3 cells (deposited in the GEO
Database under accession no. 22036). The findings of both studies
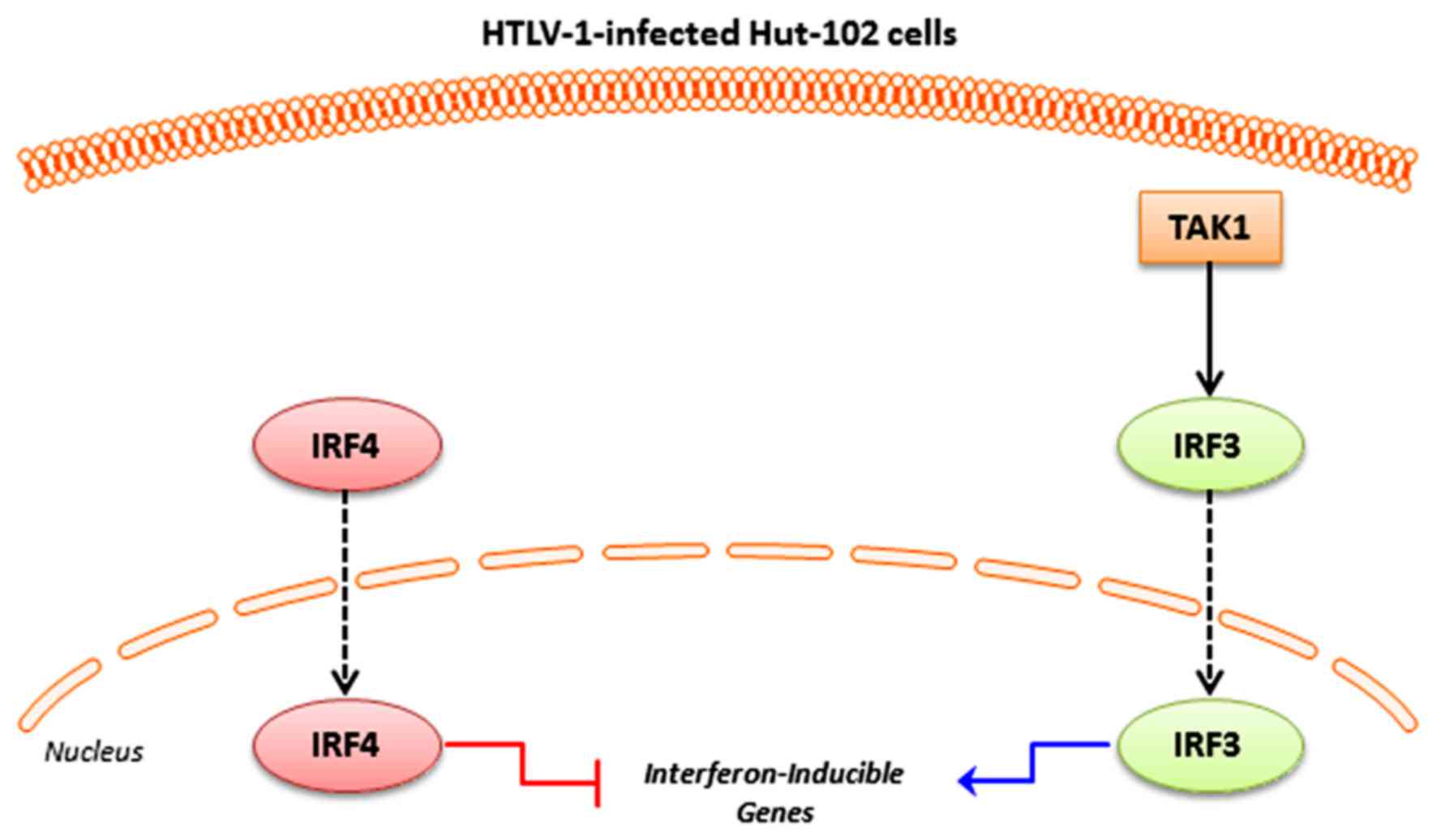
are summarized in Fig. 1. In order to
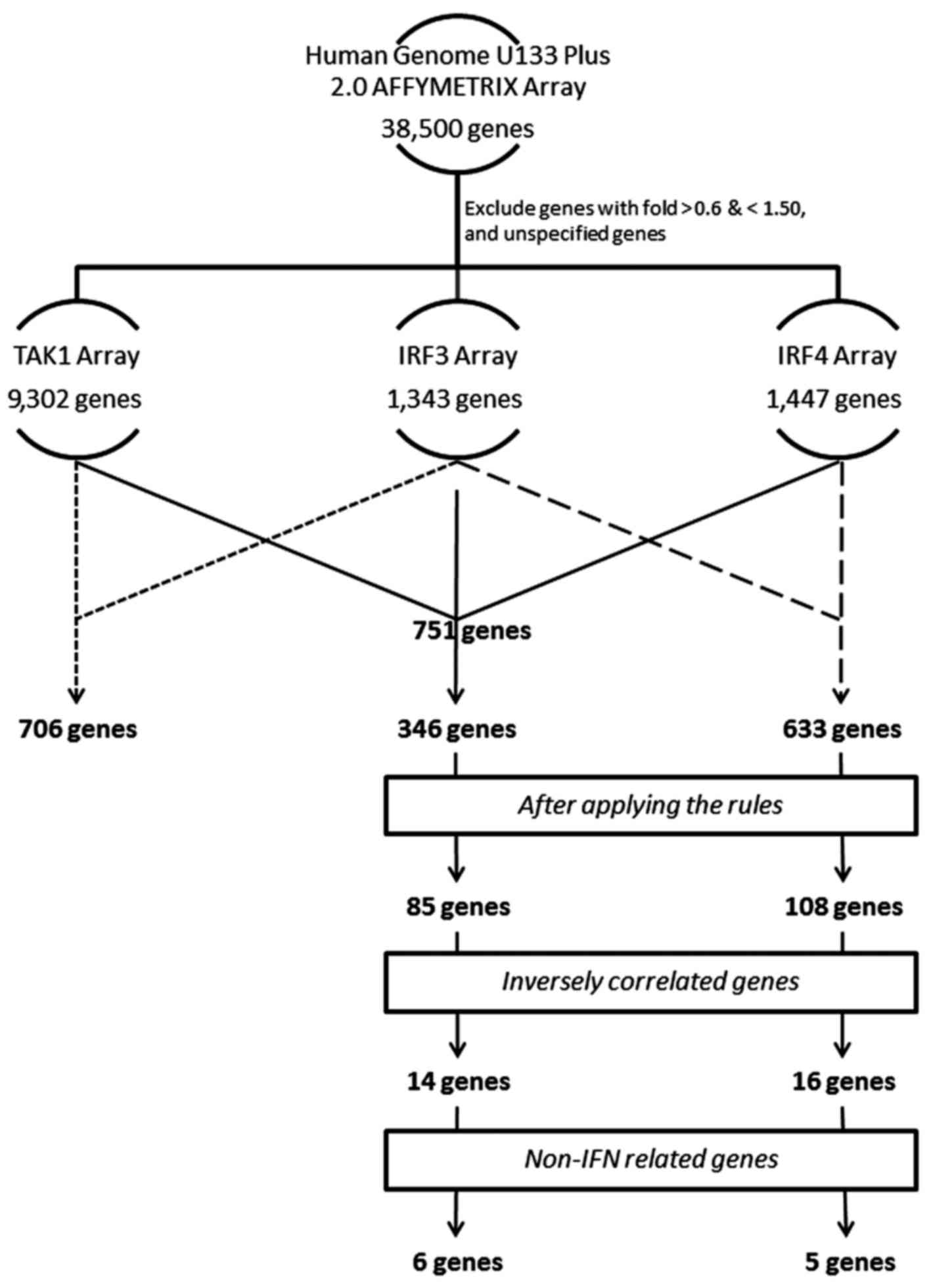
better analyze our study, we restricted the number of the analyzed
genes. Our criteria was to focus on the genes which have markedly
changed. We primarily excluded genes with values between 0.60 and
1.5 fold changes. Genes equal to or less than 0.60 are considered
downregulated, and genes equal to or more than 1.50 are considered
upregulated. The number of genes remaining after this step was
9,302, 1,343, and 1,447 genes for TAK1, IRF3, and IRF4 arrays,
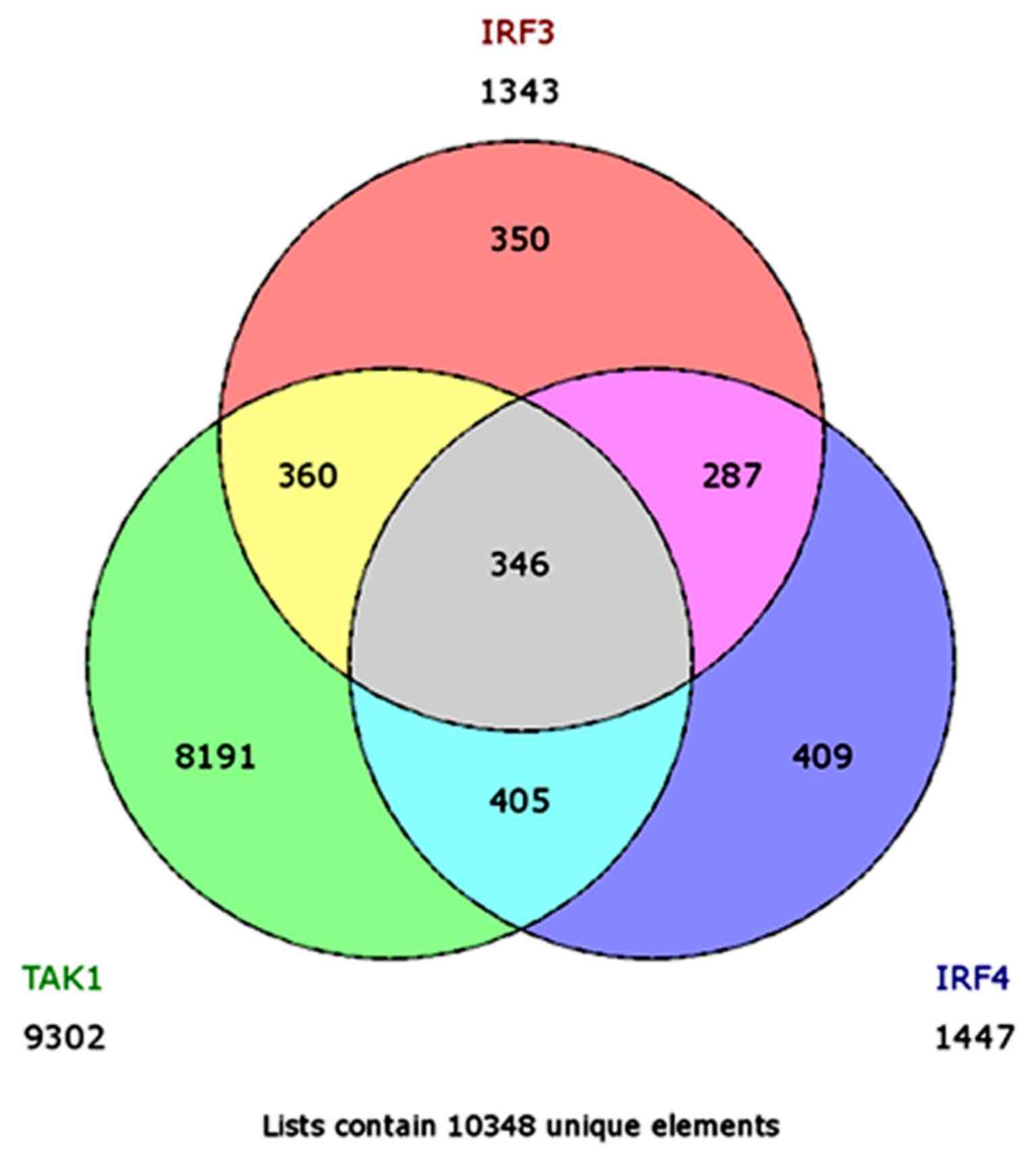
respectively. Next, we used GeneVenn (genevenn.sourceforge.net) to generate a Venn chart
demonstrating the set of common genes between the different sets of
arrays. The Venn chart indicated a list of 706 common genes between
TAK1 and IRF3, 751 common genes between TAK1 and IRF4, 633 common
genes between IRF3 and IRF4, and finally 346 common genes between
the three datasets. Finally, R software for statistical computing
of data sets was used, as explained below, to restrict our
selection to the most promising genes, as indicated.
Results
Rules applied on the identified
datasets
In this study, we focused on IRF4 compared to both
TAK1 and IRF3, or IRF3 only. Therefore, we continued our analysis
using either the 346 common genes between the three datasets (Set
A), or the 633 common genes between IRF3 and IRF4 (Set B) (Fig. 2). To filter down the list of genes
generated from the Venn chart, we applied simple rules for the
inclusion of genes. We firstly denoted the basic level of a given
gene as the average of luciferase transfected sample in the given
arrays. We then identified the basic level with different class
categories, and applied rules of inclusion for genes according to
Tables I and II. Through the applied criteria, we
obtained 85 genes out of Set A, and 108 genes out of Set B
(Figs. 3 and 4). The next stage was to identify genes
inversely correlated between IRF4 against both TAK1 and IRF3, and
IRF4 against IRF3 (Fig. 4). From this
step, we obtained 14 genes from Set A, and 16 genes from Set B. As
our goal from this analysis is to identify new genes inversely
controlled by the IRF4 axis vs. the TAK1-IRF3 axis, we excluded
unspecified genes, and genes related to interferon for being
already covered in previous studies (3,4) (Tables III and IV, and Fig.
4).
 | Table I.Rules implemented for set A
(IRF4#IRF3/TAK1) and the perspective number of genes. |
Table I.
Rules implemented for set A
(IRF4#IRF3/TAK1) and the perspective number of genes.
| Basic level | Class | No. of genes | Rule | No. of genes after
applying the rule |
|---|
| >100 | 5 | 13 | Include all | 13 |
| 50–100 | 4 | 18 | Include if 2 arrays
are below 0.5 or more than 2 | 15 |
| 25–50 | 3 | 94 | Include if 2 arrays
are below 0.4 or more than 4 | 32 |
| 10–25 | 2 | 132 | Include if 2 arrays
are below 0.3 or more than 6 | 21 |
| 0–10 | 1 | 89 | Include if 2 arrays
are below 0.2 or more than 8 | 4 |
| Total |
| 346 |
| 85 |
 | Table II.Rules implemented for set B
(IRF4#IRF3) and the perspective number of genes. |
Table II.
Rules implemented for set B
(IRF4#IRF3) and the perspective number of genes.
| Basic level | Class | No. of genes | Rule | No. of genes after
applying the rule |
|---|
| >100 | 5 | 13 | Include all | 13 |
| 50–100 | 4 | 72 | Include if 2 arrays
are below 0.5 or more than 2 | 49 |
| 25–50 | 3 | 186 | Include if 2 arrays
are below 0.4 or more than 4 | 38 |
| 10–25 | 2 | 201 | Include if 2 arrays
are below 0.3 or more than 6 |
8 |
| 0–10 | 1 | 161 | Include if 2 arrays
are below 0.2 or more than 8 |
0 |
| Total |
| 633 |
| 108 |
 | Table III.Final list of inversely correlated
genes created from set A (IRF4#IRF3/TAK1). |
Table III.
Final list of inversely correlated
genes created from set A (IRF4#IRF3/TAK1).
| Gene symbol | Gene name | Array 1
(TAK1/luc) | Array 2
(IRF3/luc) | Array 3
(IRF4/luc) | Class |
|---|
| ATM | Ataxia telangiectasia
mutated | 0.52 | 0.26 | 12.87 | 2 |
| CFTR | Cystic fibrosis
transmembrane conductance regulator | 4.31 | 2.11 | 0.34 | 3 |
| CXCL10 | Chemokine (C-X-C
Motif) ligand 10 | 0.35 | 0.19 | 6.55 | 5 |
| CXCL11 | Chemokine (C-X-C
Motif) ligand 11 | 0.21 | 0.19 | 8.98 | 5 |
| FLJ10213 | Hypothetical
protein LOC55096 | 0.58 | 0.16 | 4.23 | 3 |
| GBP4 | Guanylate binding
protein 4 | 0.51 | 0.45 | 7.51 | 4 |
| IFIT1 | Interferon-induced
protein with tetratricopeptide repeats 1 | 0.36 | 0.33 | 5.27 | 5 |
| IFIT2 | Interferon-induced
protein with tetratricopeptide repeats 2 | 0.5 | 0.29 | 2.49 | 5 |
| IFIT3 | Interferon-induced
protein with tetratricopeptide repeats 3 | 0.48 | 0.43 | 2.23 | 5 |
| IL18RAP | Interleukin 18
receptor accessory protein | 0.22 | 0.49 | 2.21 | 5 |
| MUC4 | Mucin 4, cell
surface associated | 1.66 | 3.29 | 0.31 | 3 |
| PARP14 | Poly (ADP-ribose)
polymerase family, member 14 | 0.1 | 0.58 | 2.94 | 5 |
| QKI | QKI, KH domain
containing, RNA binding | 4.38 | 5.4 | 0.59 | 2 |
| UBR2 | Ubiquitin protein
ligase E3 component N-recognin 2 | 0.29 | 0.23 | 1.94 | 2 |
 | Table IV.Final list of inversely correlated
genes created from set B (IRF4#IRF3). |
Table IV.
Final list of inversely correlated
genes created from set B (IRF4#IRF3).
| Gene symbol | Gene name | Fold 1
(IRF3/luc) | Fold 2
(IRF4/luc) | Class |
|---|
| ATM | Ataxia
telangiectasia mutated | 0.26 | 12.87 | 3 |
| CLEC7A | C-type lectin
domain family 7, member A | 0.14 | 4.62 | 2 |
| CXCL10 | Chemokine (C-X-C
Motif) ligand 10 | 0.19 | 6.55 | 5 |
| CXCL11 | Chemokine (C-X-C
Motif) ligand 11 | 0.19 | 8.98 | 5 |
| FLJ10213 | (unidentified) | 0.16 | 4.23 | 3 |
| GBP4 | Guanylate binding
protein 4 | 0.45 | 7.51 | 4 |
| IFIT1 | Interferon-induced
protein with tetratricopeptide repeats 1 | 0.33 | 5.27 | 5 |
| IFIT2 | Interferon-induced
protein with tetratricopeptide repeats 2 | 0.29 | 2.49 | 5 |
| IFIT3 | Interferon-induced
protein with tetratricopeptide repeats 3 | 0.43 | 2.23 | 5 |
| IL18RAP | Interleukin 18
receptor accessory protein | 0.49 | 2.21 | 5 |
| ISG20 | Interferon
stimulated exonuclease gene 20 kDa | 0.45 | 2.24 | 5 |
| L3MBTL | Lethal(3)malignant
brain tumor-like gene | 0.13 | 4.84 | 3 |
| LOC729397 | Hypothetical
LOC729397 | 0.31 | 2.23 | 4 |
| PML | Promyelocytic
leukemia | 0.53 | 2.06 | 5 |
| SEC24D | SEC24 family member
D | 0.31 | 3.24 | 3 |
| TMEM140 | Transmembrane
protein 140 | 0.35 | 2.61 | 4 |
List of genes identified as possible
future therapeutic targets
The final list we obtained consists of 10 genes that
we highly recommend as potential candidate for therapies targeting
the HTLV-1 infected cancer cells, and might be potential in other
cancers as well.
i) Ataxia telangiectasia mutated (ATM) is a
Serine/threonine protein kinase with a distinct role in
double-strand breaks. Its activation causes the phosphorylation and
the activation of several downstream DNA damage and cell cycle
arrest checkpoints including the histone variant H2ax, the effector
protein kinases, and the tumor suppressor p53 (5–7). Germ-line
mutations of ATM lead to ataxia-telangiectasia and shows a high
risk of breast cancer (8).
ii) Cystic Fibrosis Transmembrane conductance
Regulator (CFTR) is Chloride channel membrane protein (9). It is a member of the ABC transporter
superfamily and is activated by phosphorylation by PKA. CFTR
conducts anions to flow down their electrochemical gradient and
facilitate the passive movement of the positively charged ions. The
CFTR gene has been reported to be mutated in patients with
cystic fibrosis (CF) and has been a potential therapeutic target.
In 2012, the FDA approved Ivacaftor as the first targeted therapy
for patient with CF (10). Recently
CFTR was identified as s a tumor suppressor including intestinal,
prostate and ovarian cancer (11–13).
Moreover, CFTR was shown as a potent suppressor of
epithelial-to-mesenchymal transition (EMT) breast cancer cells and
was associated with poor prognosis in patients (14).
iii) Mucin 4 (MUC4) is a high molecular weight
glycosylated protein, which plays various roles in promoting cancer
progression (15). Overexpression of
mucins has been shown to reduce of cell adhesion and promote cancer
cell migration and metastasis. MUC4 also phosphorylate the ErbB2
leading to increasing the tumor cell proliferation independent on
the activation of neither the MAPK nor AKT pathways. Importantly,
MUC4 has been shown as a promising biomarker for diagnoses of
pancreatic cancer with undetectable levels in normal pancreas
(16).
iv) Poly (ADP-ribose) polymerase 14 (PARP14) is a
member of the PARP family of proteins, which involves in DNA repair
and programmed cell death. PARP14 was shown to regulate the
STAT6-dependent transcription (17).
Moreover, Iansante et al has reported the vital role of
PARP14 to mechanistically link apoptosis to metabolism. Through
inhibiting the JNK1/PKMa2 regulatory axis, PARP14 potentially
promoted Warburg effect in hepatocellular carcinoma (18).
v) Quaking homolog, KH domain RNA binding (QK1) is a
RNA-binding protein. It is a member of the Signal Transduction and
Activation of RNA (STAR) proteins family. QK1 was reported to play
a distinct role in schizophrenia via regulating the
myelin-related genes activity (19).
QKI has been recently shown to be downregulated in lung cancer
resulted in poorer prognosis (20).
Additionally, it suppresses glioblastoma by stabilizing
microRNA-20a leading to regulating TGFβ pathway (21).
vi) UBR2 is an E3 ubiquitin-protein ligase, which
acts by recognizing and binding to the N-terminal residues-carrying
proteins leading to their ubiquitination and degradation. Notably,
UBR2 together with UBR1 were shown as potential negative regulators
of mammalian target of rapamycin (mTOR) pathway via
degrading the Leucine protein, and therefore its expression can be
associated with cancer elimination (22).
vii) C-type lectin domain family 7 member A (CLEC7A,
also known as Dectin-1) is a glycoprotein with a distinct role in
regulating innate immunity. It is predominantly expressed in
macrophages, neutrophils and dendritic cells and binds both CD4+
and CD8+ T cells (23). Dectin-1 also
recognizes several fungal species, which triggers the induction of
numerous cytokines and chemokines including TNF-α, IL-2, IL-6, and
IL-23 (24). Mechanistically,
Dectin-1 ligand has been shown to act via recruiting and activating
the NFkB inflammatory pathway (25).
Importantly, activation of dectin-1 on macrophages induces an
adaptive immune suppression and promotes pancreatic cancer
progression (26).
viii) Lethal (3)
malignant brain tumor-like protein (L3MBTL) is a polycomb group
protein (PcG) that recognizes and binds methyllysine residues on
the target proteins leading to post-translational modifications.
L3MBTL was shown to be associated with myeloid malignancies
(27).
ix) SEC24D is a member of the SEC23/SEC24 family,
which is involved in vesicle trafficking at the endoplasmic
reticulum (28).
x) TMEM140 is the transmembrane protein 140, shown
as a promising prognostic marker for patients with glioma where its
overexpression strongly correlates with tumor size and overall
patients' survival rates. Importantly, silencing TMEM140 suppressed
the viability, migration, and invasion of glioma cells suggesting
its importance as an attractive therapeutic target (29).
Discussion
In this study, we have identified a set of 10 genes
relevant to interferon signaling. To date, interferon based therapy
is widely used to treat ATL. Some of the identified genes might
have wider scope of functions irrespective to the
interferon-related signaling, per se. With the exception of
SEC24D, which has very limited data, the remaining genes in the
list has several roles in cancer progression through their
overexpression or mutation. As a matter of interest, PARP-14 is of
an exceptionally rising importance in cancer research and
metabolism. PARP-14 and other molecular targets identified in this
study can act as potential therapeutic targets for Cancer (18,30).
References
|
1
|
Coffin JM: The discovery of HTLV-1, the
first pathogenic human retrovirus. Proc Natl Acad Sci USA.
112:15525–15529. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Poiesz BJ, Ruscetti FW, Gazdar AF, Bunn
PA, Minna JD and Gallo RC: Detection and isolation of type C
retrovirus particles from fresh and cultured lymphocytes of a
patient with cutaneous T-cell lymphoma. Proc Natl Acad Sci USA.
77:7415–7419. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Refaat A, Zhou Y, Suzuki S, Takasaki I,
Koizumi K, Yamaoka S, Tabuchi Y, Saiki I and Sakurai H: Distinct
roles of transforming growth factor-beta-activated kinase 1
(TAK1)-c-Rel and interferon regulatory factor 4 (IRF4) pathways in
human T cell lymphotropic virus 1-transformed T helper 17 cells
producing interleukin-9. J Biol Chem. 286:21092–21099. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Suzuki S, Zhou Y, Refaat A, Takasaki I,
Koizumi K, Yamaoka S, Tabuchi Y, Saiki I and Sakurai H: Human T
cell lymphotropic virus 1 manipulates interferon regulatory signals
by controlling the TAK1-IRF3 and IRF4 pathways. J Biol Chem.
285:4441–4446. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lee JH and Paull TT: Activation and
regulation of ATM kinase activity in response to DNA double-strand
breaks. Oncogene. 26:7741–7748. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Huang X, Halicka HD and Darzynkiewicz Z:
Detection of histone H2AX phosphorylation on Ser-139 as an
indicator of DNA damage (DNA double-strand breaks). Curr Protoc
Cytom Chapter. 7:Unit 7.27. 2004. View Article : Google Scholar
|
|
7
|
Canman CE, Lim DS, Cimprich KA, Taya Y,
Tamai K, Sakaguchi K, Appella E, Kastan MB and Siliciano JD:
Activation of the ATM kinase by ionizing radiation and
phosphorylation of p53. Science. 281:1677–1679. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ahmed M and Rahman N: ATM and breast
cancer susceptibility. Oncogene. 25:5906–5911. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gadsby DC, Vergani P and Csanády L: The
ABC protein turned chloride channel whose failure causes cystic
fibrosis. Nature. 440:477–483. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Jones AM and Helm JM: Emerging treatments
in cystic fibrosis. Drugs. 69:1903–1910. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Than BLN, Linnekamp JF, Starr TK,
Largaespada DA, Rod A, Zhang Y, Bruner V, Abrahante J, Schumann A,
Luczak T, et al: CFTR is a tumor suppressor gene in murine and
human intestinal cancer. Oncogene. 36:35042017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Xie C, Jiang XH, Zhang JT, Sun TT, Dong
JD, Sanders AJ, Diao RY, Wang Y, Fok KL, Tsang LL, et al: CFTR
suppresses tumor progression through miR-193b targeting urokinase
plasminogen activator (uPA) in prostate cancer. Oncogene.
32:2282–2291, 2291.e1-7. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Xu J, Yong M, Li J, Dong X, Yu T, Fu X and
Hu L: High level of CFTR expression is associated with tumor
aggression and knockdown of CFTR suppresses proliferation of
ovarian cancer in vitro and in vivo. Oncol Rep. 33:2227–2234. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang JT, Jiang XH, Xie C, Cheng H, Da
Dong J, Wang Y, Fok KL, Zhang XH, Sun TT, Tsang LL, et al:
Downregulation of CFTR promotes epithelial-to-mesenchymal
transition and is associated with poor prognosis of breast cancer.
Biochim Biophys Acta. 1833:2961–2969. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Jhala N, Jhala D, Vickers SM, Eltoum I,
Batra SK, Manne U, Eloubeidi M, Jones JJ and Grizzle WE: Biomarkers
in Diagnosis of pancreatic carcinoma in fine-needle aspirates. Am J
Clin Pathol. 126:572–579. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Srivastava SK, Bhardwaj A, Singh S, Arora
S, Wang B, Grizzle WE and Singh AP: MicroRNA-150 directly targets
MUC4 and suppresses growth and malignant behavior of pancreatic
cancer cells. Carcinogenesis. 32:1832–1839. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mehrotra P, Riley JP, Patel R, Li F, Voss
L and Goenka S: PARP-14 functions as a transcriptional switch for
Stat6-dependent gene activation. J Biol Chem. 286:1767–1776. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Iansante V, Choy PM, Fung SW, Liu Y, Chai
JG, Dyson J, Del Rio A, D'Santos C, Williams R, Chokshi S, et al:
PARP14 promotes the Warburg effect in hepatocellular carcinoma by
inhibiting JNK1-dependent PKM2 phosphorylation and activation. Nat
Commun. 6:78822015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Aberg K, Saetre P, Jareborg N and Jazin E:
Human QKI, a potential regulator of mRNA expression of human
oligodendrocyte-related genes involved in schizophrenia. Proc Natl
Acad Sci USA. 103:7482–7487. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zong FY, Fu X, Wei WJ, Luo YG, Heiner M,
Cao LJ, Fang Z, Fang R, Lu D, Ji H and Hui J: The RNA-binding
protein QKI suppresses cancer-associated aberrant splicing. PLoS
Genet. 10:e10042892014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen AJ, Paik JH, Zhang H, Shukla SA,
Mortensen R, Hu J, Ying H, Hu B, Hurt J, Farny N, et al: STAR
RNA-binding protein Quaking suppresses cancer via stabilization of
specific miRNA. Genes Dev. 26:1459–1472. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kume K, Iizumi Y, Shimada M, Ito Y, Kishi
T, Yamaguchi Y and Handa H: Role of N-end rule ubiquitin ligases
UBR1 and UBR2 in regulating the leucine-mTOR signaling pathway.
Genes Cells. 15:339–349. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Taylor PR, Brown GD, Reid DM, Willment JA,
Martinez-Pomares L, Gordon S and Wong SY: The beta-glucan receptor,
dectin-1, is predominantly expressed on the surface of cells of the
monocyte/macrophage and neutrophil lineages. J Immunol.
169:3876–3882. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Huysamen C and Brown GD: The fungal
pattern recognition receptor, Dectin-1, and the associated cluster
of C-type lectin-like receptors. FEMS Microbiol Lett. 290:121–128.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Saijo S and Iwakura Y: Dectin-1 and
Dectin-2 in innate immunity against fungi. Int Immunol. 23:467–472.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Activation of Dectin-1 on Macrophages
Promotes Pancreatic Cancer. Cancer Discov. 7:5492017.
|
|
27
|
Li J, Bench AJ, Vassiliou GS, Fourouclas
N, Ferguson-Smith AC and Green AR: Imprinting of the human L3MBTL
gene, a polycomb family member located in a region of chromosome 20
deleted in human myeloid malignancies. Proc Natl Acad Sci USA.
101:7341–7346. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Pagano A, Letourneur F, Garcia-Estefania
D, Carpentier JL, Orci L and Paccaud JP: Sec24 proteins and sorting
at the endoplasmic reticulum. J Biol Chem. 274:7833–7840. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Li B, Huang MZ, Wang XQ, Tao BB, Zhong J,
Wang XH, Zhang WC and Li ST: TMEM140 is associated with the
prognosis of glioma by promoting cell viability and invasion. J
Hematol Oncol. 8:892015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Camicia R, Winkler HC and Hassa PO: Novel
drug targets for personalized precision medicine in
relapsed/refractory diffuse large B-cell lymphoma: A comprehensive
review. Mol Cancer. 14:2072015. View Article : Google Scholar : PubMed/NCBI
|