Introduction
Most patients with advanced non-small cell lung
cancer (NSCLC) are candidates for systemic therapy. Platinum-based
doublet chemotherapy has been the cornerstone of first line
treatment regimens with response rates of ~30% (1). Immunotherapy with antibodies that target
the programmed cell death protein 1 pathway (PD-1/PD-L1) are now
approved in both the first- and second-line settings (2–4). Response
rates of ~20% have been observed; many of the responses have been
durable. Cytotoxic chemotherapy can enhance immune responses
through several mechanisms and have been effectively combined with
immunotherapeutics. Several platinum-based combination chemotherapy
regimens have been tested with the anti-PD-1 antibody, nivolumab,
in patients with NSCLC (5). The
combination of another anti-PD-1 antibody, pembrolizumab, with
carboplatin and pemetrexed has recently been approved for the
first-line treatment of metastatic non-squamous NSCLC. The approval
was based on data from the KEYNOTE-021 trial in which the response
rate with the combination was 55% compared to 29% in patients
treated with chemotherapy alone (6).
One of the strongest predictors of response and
survival in patients with advanced NSCLC is performance status (PS)
(7). Although platinum-based
combination chemotherapy has been shown to be beneficial for
patients with a good PS, 0 or 1, there is still debate regarding
its efficacy for patients with a poor PS, 2 or worse (8). PS 2 patients account for up to 30% of
patients with advanced NSCLC. In addition to poor responses and
survival outcomes, many of the treatment options can cause
substantial toxicity in this subset of patients which is why no
regimen is accepted as the standard-of-care (9,10). Accrual
to most NSCLC clinical trials, including the
PD-1-inhibitor-chemotherapy trials, has been limited to patients
with PS 0 or 1. The frequent administration of low doses of
chemotherapy, referred to as metronomic chemotherapy, has been
tested in patients with NSCLC with poor PS (11–16).
Although these approaches have been shown to be active and well
tolerated, patients with advanced NSCLC and poor PS represent a
large unmet need for new treatment options.
Cancer patients with poor PS have been characterized
by decreases in T cell function and increases in myeloid suppressor
cells (MDSC) and T regulatory (Treg) cells, cells that suppress
antitumor immunity (17). Platinum
and taxane compounds have been shown to stimulate cytolytic T
lymphocytes and to inhibit MDSC and Treg cells (18,19). There
is evidence that weekly low-dose carboplatin and paclitaxel
produces superior antitumor immune responses, including T cell
activation and MDSC and Treg cell inhibition, when compared with
maximally tolerated dosing (20). We
report the interim analysis of an ongoing study which examines the
feasibility of using weekly low-dose carboplatin and paclitaxel
with pembrolizumab in patients with advanced NSCLC and poor PS.
Patients and methods
Patients
Patients were enrolled to this clinical trial, which
was reviewed and approved by the Wake Forest Health Sciences
Institutional Review Board (Winston-Salem, NC, USA). All patients
provided written informed consent before undergoing any
study-related procedures or testing. Eligibility criteria included
histologically confirmed NSCLC that was recurrent (progression
after surgery or radiation or chemo-radiation treatment for
loco-regional disease) or metastatic; ECOG PS of 2 or 3; age
>18; and at least one measurable lesion as defined by Response
Evaluation Criteria in Solid Tumors (RECIST) version 1.1 (21). Key exclusion criteria included more
than two previous lines of systemic treatment; active, untreated
central nervous system metastases requiring steroids; previous
exposure to PD-1/PD-L1 inhibitors; history of pneumonitis;
infection with HIV or hepatitis B/C; pregnancy; a requirement for
immunosuppression or chronic steroids; and active infection on
antibiotics, antifungals, or antivirals.
Study design and treatment
Patients were randomized to pembrolizumab alone or
pembrolizumab combined with weekly carboplatin and paclitaxel. The
dosing for pembrolizumab was the same in both arms, 200 mg
intravenously over 30 min every 3 weeks. Carboplatin and paclitaxel
were administered every 7 days at an AUC 1 (Calvert formula)
intravenously over 1 h and 25 mg/m2 intravenously over 1
h, respectively. Standard-of-care dosing adjustments were used.
Treatment continued until unacceptable toxicity or disease
progression. Three weeks of treatment were considered to be one
cycle. This study is registered with NCI (NCT02581943).
Study assessments
Baseline assessments included a complete medical
history, physical exam, electrocardiogram, complete blood count
(CBC), comprehensive metabolic panel (CMP), and a CT scan of the
chest and abdomen. A physical exam was performed every three weeks
in all patients. Weekly CBC and CMP were obtained prior to each
chemotherapy infusion for patients receiving the combination of
low-dose chemotherapy and pembrolizumab and every three weeks for
patients receiving pembrolizumab alone. Thyroid function studies
were obtained every six weeks. Adverse events were characterized
and graded using Common Terminology Criteria for Adverse Events
version 4.0. The causal relationship (related or not related)
between treatment and adverse events was determined by the
investigator. Tumor response was assessed by CT scan at 7 weeks of
initiation of therapy and every 12 weeks thereafter. In case of
clinical suspicion of tumor progression, CT scans were obtained at
a shorter interval. Complete response (CR) partial response (PR)
and stable disease (SD) and progressive disease (PD) were evaluated
and assigned according to RECIST version 1.1. Patients who
manifested a CR, PR, or SD were considered to have achieved disease
control. All imaging assessments were reviewed by at least one
investigator and one radiologist. Blood, for immune laboratory
correlates was collected into heparin from each patient before the
first dosing and at weeks 4 and 7, after the completion of cycles 1
and 2, respectively.
Flow cytometry
An aliquot of whole peripheral blood was evaluated
by multicolor flow cytometry using a FACSCalibur flow cytometer (BD
Biosciences, Mountain View, CA, USA). Immune cell populations were
identified using phycoerythrin-labeled FoxP3 and HLA-DR,
fluorescein-isothiocynate-labeled cluster of differentation (CD)4,
CD14, and inducible costimulator (ICOS); and
allophycocyanin-labeled CD8. All labeled antibodies were purchased
from BD Biosciences with the exception of FoxP3, which was
purchased from eBiosciences (San Diego, CA, USA). The percentage of
populations of interest was determined using gate statistics. The
absolute number of MDSCs was calculated as follows: (total white
blood cell count (cells/µl) × percent MDSCs). Treg cell frequencies
were calculated by normalization to total CD4 T cell numbers.
miRs
Total RNA was isolated from plasma using the
miRNeasy Mini kit (Qiagen Inc., Valencia, CA, USA) according to the
manufacturer's instructions. Reverse transcription reactions were
performed using a TaqMan MicroRNA Reverse Transcription kit
(Applied Biosystems; Thermo Fisher Scientific, Inc., Waltham, MA,
USA) according to the manufacturer's instructions. Reverse
transcription-quantitative polymerase chain reaction (RT-qPCR) was
performed using the reverse transcription reaction product, TaqMan
MicroRNA Assay kit, and TaqMan Universal PCR Master Mix (Applied
Biosystems; Thermo Fisher Scientific, Inc.) according to the
manufacturer's instructions. TaqMan MicroRNA Assay kits for human
miRs were used. Reactions were loaded onto a 96-well plate and run
in duplicate on an ABI 7500 Fast Real-Time PCR System (Applied
Biosystems; Thermo Fisher Scientific, Inc.). The reactions were
incubated at 50°C for 20 sec and 95°C for 10 min, followed by 40
cycles of denaturation at 95°C for 15 sec, then 1 min of
annealing/extension at 60°C. The ΔΔCq method was used to determine
relative number of copies (RQ) of miR (22). Data were normalized to a C.
elegans synthetic miR sequence, cel-miR-39 (Qiagen, Inc.),
which was spiked in as a control during RNA isolation.
Soluble PD-L1
Plasma levels of soluble PD-L1 (sPD-L1) were
determined by an ELISA kit (MyBioSource, San Diego, CA, USA) as
recommended by the manufacturer.
Statistical analysis
Data were summarized as percentages, means and
standard deviation, or medians and ranges. Repeated-measures ANOVA
(using PROC MIXED in SAS), considering treatment group (single vs.
combination) and time period (pre-treatment, cycle 1, and cycle 2)
as categorical (class) variables, as well as the treatment*time
period interaction, were used to model levels of immune cells and
factors and circulating miRs. We used post-hoc contrast statements
on estimated least-squares means to compare the two treatment
groups at specific time points, and to compare first and last time
point measures within treatment group. A significant treatment*time
interaction in a model means that the null hypothesis that the two
treatment groups have similar biomarker patterns over the 3 time
points can be rejected. For each outcome variable we also conducted
a planned contrast comparing differences in estimated least squares
means at cycle 2 compared to pretreatment levels within each
treatment group separately (i.e., 2 contrasts per outcome
variable). The null hypothesis being tested in these contrasts is
that the difference between pretreatment and cycle 2 levels within
a treatment group is zero. We applied a Bonferroni correction
within each model to the two P-values obtained from the contrast
statements. A two-sided alpha of 0.05 was used to evaluate main and
interactive model effects, and a two-sided alpha of 0.025 was used
for the contrast statements, per the Bonferroni method of adjusting
for the two extra contrasts within each model. All statistical
analyses were performed using SAS version 9.4 (SAS Institute, Inc.,
Cary, NC, USA) and StatXact version 10 (Cytel Inc., Cambridge, MA,
USA). All tests were two-sided, and P<0.05 was considered to
indicate a statistically significant difference.
Results
Clinical efficacy
All patients enrolled had a PS of 2. Ten were
randomized to the pembrolizumab alone, the single-agent arm, and 10
to pembrolizumab, carboplatin, and paclitaxel, the combination arm.
Patient characteristics are outlined in Table I. The mean number of 3-week cycles per
patient was 7 (range, 2–14) in the single-agent arm and 9 (range,
4–16) in the combination arm. Adverse events are displayed in
Table II. One patient in the
single-agent arm discontinued treatment due to A-V block that was
not considered related to treatment and that was successfully
managed with pacemaker insertion. Two patients in the single-agent
arm required replacement therapy due to treatment-induced
hypothyroidism. One patient in the combination arm discontinued
therapy due to Grade 3 fatigue. Four patients in the combination
arm discontinued carboplatin due to allergic reactions at 3, 7, 9,
and 10 months of treatment. The patient who had a reaction during
the third month of treatment had received carboplatin prior. These
patients were able to continue treatment with pembolizumab and
weekly paclitaxel. One patient in the combination arm required
replacement therapy due to treatment-induced hypothyroidism.
Paclitaxel infusion reactions were not observed nor were
pneumonitis or renal failure. Grade 4 and 5 adverse events were
also not observed. Tumor response and disease control rates are
summarized in Table III.
 | Table I.Patient characteristics. |
Table I.
Patient characteristics.
| Treatment arm | Pembrolizumab +
low-dosechemotherapy (n=10) | Pembrolizumab
(n=10) |
|---|
| Sex (M/F) | 5/5 | 7/3 |
| Mean age, years
(range) | 67.9 (56–85) | 68.6 (54–83) |
| Histology, n
(%) |
|
|
|
Non-squamous | 8 (80) | 6 (60) |
|
Squamous | 0 (0) | 4 (40) |
| Not
otherwise specified | 2 (20) | 0 (0) |
| Prior treatment, n
(%) |
|
|
|
None | 7 (70) | 6 (60) |
|
Chemotherapy | 3 (30) | 4 (40) |
 | Table II.Adverse events. |
Table II.
Adverse events.
|
| Pembrolizumab +
low-dose chemotherapy (n=10) | Pembrolizumab
(n=10) |
|---|
|
|
|
|
|---|
| Event type | Grade 1–2 | Grade 3 | Grade 1–2 | Grade 3 |
|---|
| Any event | 28 | 0 | 16 | 1 |
| Alopecia | 0 | 0 | 0 | 0 |
| Anemia | 1 | 0 | 0 | 0 |
| Anorexia | 2 | 0 | 1 | 0 |
| Arthralgia | 1 | 0 | 1 | 0 |
| Constipation | 0 | 0 | 0 | 0 |
| Cough | 0 | 0 | 2 | 0 |
| Diarrhea | 1 | 0 | 2 | 0 |
| Dizziness | 0 | 0 | 0 | 0 |
| Dry skin | 1 | 0 | 1 | 0 |
| Dysgeusia | 0 | 0 | 0 | 0 |
| Epistaxis | 0 | 0 | 0 | 0 |
| Fatigue | 2 | 0 | 1 | 1 |
| Headache | 1 | 0 | 1 | 0 |
|
Hypersensitivity | 3 | 0 | 0 | 0 |
| Hypoesthesia | 0 | 0 | 0 | 0 |
| Hypomagnesemia | 0 | 0 | 0 | 0 |
| Hypothyroidism | 1 | 0 | 2 | 0 |
| Influenza-like
illness | 0 | 0 | 0 | 0 |
| Infusion
reactions | 0 | 0 | 0 | 0 |
| Leukopenia | 2 | 0 | 0 | 0 |
| Lymphopenia | 2 | 0 | 0 | 0 |
| Mucositis | 0 | 0 | 0 | 0 |
| Musculoskeletal
pain | 2 | 0 | 0 | 0 |
| Nausea | 2 | 0 | 0 | 0 |
| Neutropenia | 2 | 0 | 0 | 0 |
| Peripheral
neuropathy | 1 | 0 | 0 | 0 |
| Pneumonitis | 1 | 0 | 2 | 0 |
| Pruritus | 1 | 0 | 2 | 0 |
| Pyrexia | 0 | 0 | 0 | 0 |
| Rash | 1 | 0 | 1 | 0 |
| Renal failure
(acute) | 0 | 0 | 0 | 0 |
|
Thrombocytopenia | 1 | 0 | 0 | 0 |
| Tinnitus | 0 | 0 | 0 | 0 |
| Transaminase
increase | 0 | 0 | 0 | 0 |
| Vomiting | 0 | 0 | 0 | 0 |
 | Table III.Tumor response. |
Table III.
Tumor response.
| Treatment | Pembrolizumab +
low-dose chemotherapy (n=10) | Pembrolizumab
(n=10) |
|---|
| Complete response,
n (%) | 0 (0) | 0 (0) |
| Partial response, n
(%) | 7 (70) | 2 (20) |
| Stable disease, n
(%) | 2 (20) | 4 (40) |
| Disease control, n
(%) | 9 (90) | 6 (60) |
| Progressive
disease, n (%) | 1 (10) | 4 (40) |
Immunologic effects
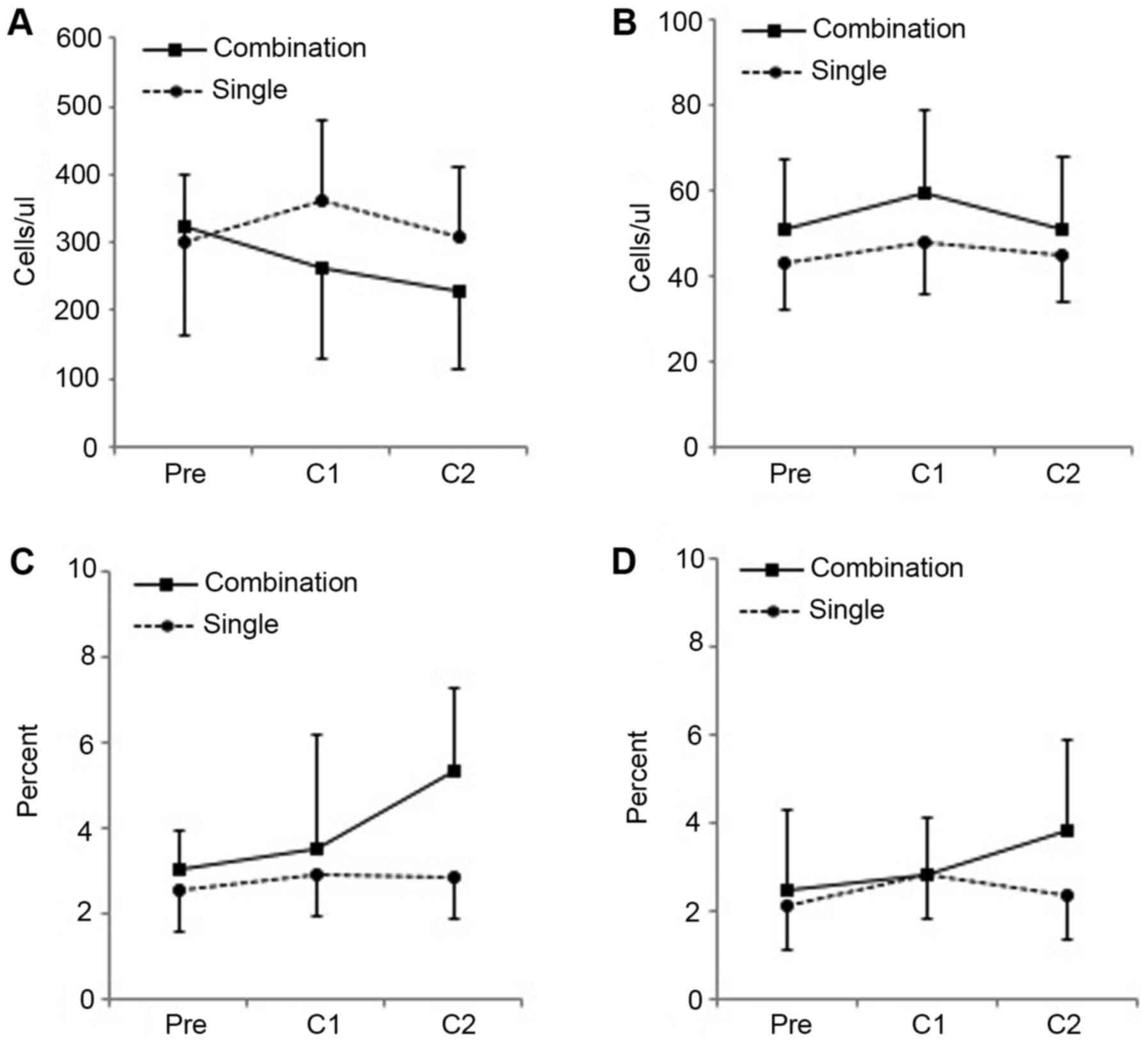
Circulating immune cells and soluble factors
previously reported to be modulated by platinum and taxane
chemotherapeutics and/or by PD-1 blockade were examined.
Circulating CD14+HLA-DR−/low MDSC, which have
been associated with therapeutic resistance in patients with NSCLC,
were examined (23) Decreases in
absolute numbers of this MDSC subpopulation were observed in the
combination arm; significant changes were not observed in the
single-agent arm (Fig. 1A).
Significant changes in circulating CD4+FOXP3+
Treg cells were not observed in either arm (Fig. 1B). Expression of ICOS, a T cell
costimulatory receptor related to CD28 expressed on activated T
cells that has been associated clinical response in patients with
NSCLC treated with PD-1 pathway blockade, was also examined
(24). Increases in
ICOS+CD8+ cell were observed in the
combination arm but not in the single-agent arm (Fig. 1C). Significant changes in
ICOS+CD4+ cells were not observed.
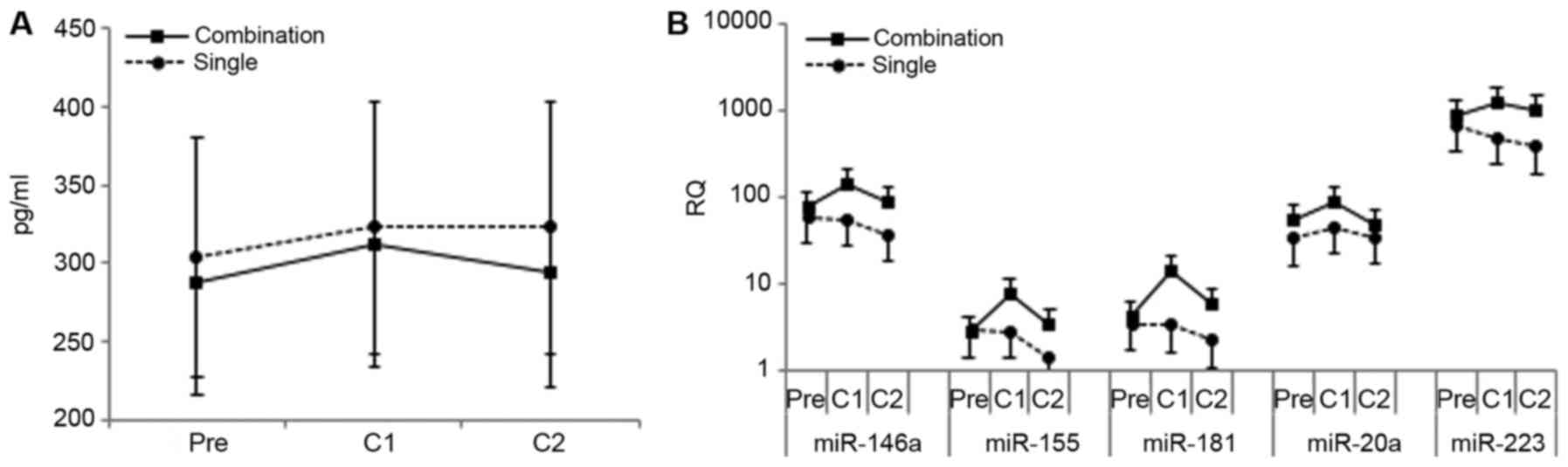
Significant changes in circulating sPD-L1, which has been
associated with immune suppression and clinical outcome, were also
not observed (Fig. 2A) (25–28).
Pre-treatment sPD-L1 levels tended to be higher in patients that
manifested PD (346±85 pg/ml, n=5), compared to patients that
manifested PR (272±70 pg/ml, n=8), but this difference was not
statistically significant.
Circulating levels of specific miRs implicated in
regulating antitumor immune responses were also examined (29). Prominent among these are miRs-146a,
155, 181a, 20a and 223, plasma levels of which have been associated
with changes in immune effector and regulatory cells in cancer
patients (30,31). Enhanced immunity effected by taxanes
has been associated with increases in miR-146a and 155 (32,33).
Increases in plasma levels of all the immune-regulatory miRs tested
were observed with treatment in the combination arm; increases in
miR-155, 181, and 223 were statistically significant (Fig. 2B).
Discussion
Patients who are able to care for themselves but are
unable to perform work-related activities are considered to have a
PS of 2. These patients constitute a significant proportion of
patients with NSCLC. Poor PS patients with NSCLC, irrespective of
age, have poorer outcomes as well as an increased incidence of
adverse events with therapy. The feasibility of applying weekly
low-dose carboplatin and paclitaxel with pembrolizumab in this
patient population was examined by randomizing patients to the
combination or to pembrolizumab alone. Weekly carboplatin, at AUC 3
or 2, and paclitaxel, at 75 or 100 mg/m2, respectively,
has resulted in acceptable toxicity and response rates in patients
with advanced NSCLC, including those with poor PS (34,35).
Immune activation was demonstrated in patients with ovarian cancer
with weekly carboplatin at AUC 2 and paclitaxel at 60 to 80
mg/m2 (20). Because of
potentially superior immune effects, we examined lower dosing,
carboplatin at AUC 1 and paclitaxel at 25 mg/m2. We
found that administering this chemotherapy-immunotherapy regimen to
patients with advanced NSCLC and PS 2 was well tolerated and
immunologically and clinically active.
Treatments in both the combination and single-agent
arms were well tolerated. The incidences of Grade 3 adverse events
were low. Carboplatin hypersensitivity was observed in 4 of the 10
patients treated with the combination. Repeated exposure is the
most important predictive factor for platinum hypersensitivity, and
the patients who did manifest hypersensitivity did receive more
than 6 months of repeated exposures (36). How PD-1 blockade may influence
platinum hypersensitivity, which is mediated primarily by IgE
and/or mast cells/basophils, is not known. The toxicity observed
compares favorably with other chemotherapy-PD-1 inhibitor
combinations tested in advanced NSCLC patients with good PS. In a
study of two doses of nivolumab combined with standard-dose,
platinum-based doublet chemotherapy, Grade 3 or 4 treatment-related
adverse events led to discontinuation in eight (14%) of 56
patients, most commonly pneumonitis and acute renal failure
(5). These included four of 14
patients in the 5 mg/kg nivolumab plus carboplatin-paclitaxel arm
and two of 15 patients in the 10 mg/kg nivolumab plus
carboplatin-paclitaxel arm. Hypersensitivity reactions occurred in
14 (25%) of patients, all of whom received 10 mg/kg nivolumab,
including six of 15 administered carboplatin-paclitaxel, seven of
15 administered cisplatin-pemetrexed, and one of 12 administered
cisplatin-gemcitabine. In KEYNOTE-21, which involved 123 patients,
the incidence of Grade 3 or worse adverse events was 39% patients
in the pembrolizumab, carboplatin, and pemetrexed group and 26% in
the chemotherapy alone group. Infusion reactions were observed in
only one (2%) of 59 in the pembrolizumab plus chemotherapy group
vs. none in the chemotherapy alone group (6).
Circulating cell regulators of the immune effects of
chemotherapy and PD-1 inhibitors were examined. Decreases in MDSC
were observed in the combination arm; changes in Treg cells were
not. That T cells were activated in the combination arm was
supported by the increase in ICOS expression on CD4+
cells. Although these findings are statistically non-significant
after applying the Bonferroni correction, given the small sample
size, we consider the results to be mainly hypothesis generating
and warrant further study in a larger group of patients. Increased
expression was not observed on CD8+ cells and not in the
single-agent arm. ICOS expression was reported to increase on
CD8+ cells of patients with NSCLC responding to
single-agent PD-1 or PD-L1 blocking antibodies (22). Increases in CD4+ ICOS have
been associated with response to the anti-CTLA-4 antibody,
ipilimumab, in patients with melanoma (37). Significant changes in circulating
immune suppressor and effector cells were not observed in the
single-agent arm. It has been noted that whereas changes in MDSC
have been evident intratumorally in patients treated with PD-1
inhibitors, changes in peripheral blood MDSC have not (38). In patients with melanoma treated with
nivolumab combined with a vaccine, a decline in circulating Treg
cells during treatment was associated with clinical benefit
(39).
Circulating soluble immune regulators were also
examined. Levels of sPD-L1 have had predictive value in patients
with melanoma treated with CTLA-4 or PD-1 blockade (24). High plasma levels of sPD-L1 have been
associated with reduced survival in lung cancer (25,26).
Although there is evidence that high baseline sPD-L1 is associated
with disease progression, it remains unclear whether sPD-L1 levels
are associated with clinical response to PD-1 blockade in patients
with NSCLC (27). Patients in our
study who manifested PD tended to have higher levels of sPD-L1 than
patients who manifested PR, but differences were not statistically
significant. Plasma miRs are emerging as a new class of biomarkers.
Circulating levels of many miRs with immune-regulatory effects have
demonstrated diagnostic or prognostic potential in NCSLC, including
the miRs tested, miR-146a, 155, 181a, 20a, and 223 (40–45). As
noted, enhanced immunity effected by taxanes in mice has been
associated with increases in miR-146a and miR-155 (31,32).
Cisplatin has been shown to increase miR-146a and to decrease
miR-181a (46–49). Increases in plasma levels of miR-155,
181a and 223 were associated with disease control the combination
arm. miR-155 and miR-223 are involved in the development of MDSC as
are miR-146a and miR-20a (50–53).
miR-155 and 181a are involved in T cell regulation (54). miRs with immune regulatory activities
also regulate a variety of other cellular processes. Of note,
miR-181a and miR-20a has been implicated in regulating the
sensitivity of tumor cells to platinums and taxanes (55–58).
A variety of factors are involved in the poor
systemic therapy response rates observed in patients with NSCLC
with poor PS (9,10). Although this clinical trial was not
designed to establish or compare response rates, 7 of 10 patients
(70%) in the combination arm manifested a PR compared to 2 of 10
patients (20%) in the single-agent arm. Weekly low-dose carboplatin
at AUC 1 and paclitaxel at 25 mg/m2 can be safely
administered with pembrolizumab at standard dosing to patients with
advanced NSCLC and poor PS. This regimen is immunologically active.
Reductions in MDSC, increases in activated CD4+ T cells,
and changes in immune-regulatory miRs are observed. The combination
of this low-dose chemotherapy regimen and pembrolizumab is also
clinically active and merits further study in this patient
population.
Acknowledgements
Not applicable.
Funding
The present study was funded by Merck & Co. as a
part of an investigator initiated clinical trial (Merck
Investigator Studies Program grant no. 52346).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
MB, TA and PLT made substantial contributions to the
conception and design of the study, interpreted the data, were
involved in the drafting and revising of the manuscript critically
for important intellectual content and gave final approval of the
version to be published. BJL and WJP interpreted the data,
assisting the drafting and revising of the manuscript critically
for important intellectual content, and gave final approval of the
version to be published. SA, MK, SG, JR and DP interpreted the data
and critically revised the manuscript for important intellectual
content.
Ethics approval and consent to
participate
The present study was approved by Wake Forest Health
Sciences Institutional Review Board (Clinical Trial Identifier,
NCT02581943; Date of registration, 5/1/16). All patients provided
written informed consent before undergoing any study-related
procedures or testing.
Patient consent for publication
Not applicable.
Competing interests
The authors declare they have no competing
interests.
References
|
1
|
Azzoli CG, Temin S, Aliff T, Baker S Jr,
Brahmer J, Johnson DH, Laskin JL, Masters G, Milton D, Nordquist L,
et al: 2011 focused update of 2009 American society of clinical
oncology clinical practice guideline update on chemotherapy for
stage IV non-small-cell lung cancer. J Clin Oncol. 29:3825–3831.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Reck M, Rodríguez-Abreu D, Robinson AG,
Hui R, Csőszi T, Fülöp A, Gottfried M, Peled N, Tafreshi A, Cuffe
S, et al: Pembrolizumab versus Chemotherapy for PD-L1-positive
non-small-cell lung cancer. N Engl J Med. 375:1823–1833. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Brahmer J, Reckamp KL, Baas P, Crinò L,
Eberhardt WE, Poddubskaya E, Antonia S, Pluzanski A, Vokes EE,
Holgado E, et al: Nivolumab versus docetaxel in advanced
squamous-cell non-small-cell lung cancer. N Engl J Med.
373:123–135. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rittmeyer A, Barlesi F, Waterkamp D, Park
K, Ciardiello F, von Pawel J, Gadgeel SM, Hida T, Kowalski DM, Dols
MC, et al: Atezolizumab versus docetaxel in patients with
previously treated non-small-cell lung cancer (OAK): A phase 3,
open-label, multicentre randomised controlled trial. Lancet.
389:255–265. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rizvi NA, Hellmann MD, Brahmer JR,
Juergens RA, Borghaei H, Gettinger S, Chow LQ, Gerber DE, Laurie
SA, Goldman JW, et al: Nivolumab in combination with platinum-based
doublet chemotherapy for first-line treatment of advanced
non-small-cell lung cancer. J Clin Oncol. 34:2969–2979. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Langer CJ, Gadgeel SM, Borghaei H,
Papadimitrakopoulou VA, Patnaik A, Powell SF, Gentzler RD, Martins
RG, Stevenson JP, Jalal SI, et al: Carboplatin and pemetrexed with
or without pembrolizumab for advanced, non-squamous non-small-cell
lung cancer: A randomised, phase 2 cohort of the open-label
KEYNOTE-021 study. Lancet Oncol. 17:1497–1508. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sculier JP, Chansky K, Crowley JJ, Van
Meerbeeck J and Goldstraw P: International Staging Committee and
Participating Institutions: The impact of additional prognostic
factors on survival and their relationship with the anatomical
extent of disease expressed by the 6th edition of the TNM
classification of malignant tumors and the proposals for the 7th
edition. J Thorac Oncol. 3:457–466. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bunn PA Jr: Chemotherapy for advanced
non-small-cell lung cancer: Who, what, when, why? J Clin Oncol. 20
18 Suppl:23S–33S. 2002.PubMed/NCBI
|
|
9
|
Tartarone A and Aieta M: Treatment of
performance status 2 patients with advanced non-small-cell lung
cancer: What we know and what we don't know. Future Oncol.
5:837–841. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lilenbaum RC, Cashy J, Hensing TA, Young S
and Cella D: Prevalence of poor performance status in lung cancer
patients: Implications for research. J Thorac Oncol. 3:125–129.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Correale P, Cerretani D, Remondo C,
Martellucci I, Marsili S, La Placa M, Sciandivasci A, Paolelli L,
Pascucci A, Rossi M, et al: A novel metronomic chemotherapy regimen
of weekly platinum and daily oral etoposide in high-risk non-small
cell lung cancer patients. Oncol Rep. 16:133–140. 2006.PubMed/NCBI
|
|
12
|
Bilir C, Durak S, Kızılkaya B,
Hacıbekiroglu I, Nayır E and Engin H: Efficacy of metronomic
vinorelbine in elderly patients with advanced non-small-cell lung
cancer and poor performance status. Curr Oncol. 24:e199–e204. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zwitter M, Kovac V, Rajer M, Vrankar M and
Smrdel U: Two schedules of chemotherapy for patients with non-small
cell lung cancer in poor performance status: A phase II randomized
trial. Anticancer Drugs. 21:662–668. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lee KW, Lim JH, Kim JH, Lee CT and Lee JS:
Weekly low-dose docetaxel for salvage chemotherapy in pretreated
elderly or poor performance status patients with non-small cell
lung cancer. J Korean Med Sci. 23:992–998. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Juan O, Albert A, Ordoño F, Casany R,
Carañana V, Campos JM and Alberola V: Low-dose weekly paclitaxel as
second-line treatment for advanced non-small cell lung cancer: A
phase II study. Jpn J Clin Oncol. 32:449–454. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hainsworth JD, Burris HA III and Greco FA:
Weekly docetaxel as a single agent and in combination with
gemcitabine in elderly and poor performance status patients with
advanced non-small cell lung cancer. Semin Oncol. 28 3 Suppl
9:S21–S25. 2001. View Article : Google Scholar
|
|
17
|
Santegoets SJ, Welters MJ and van der Burg
SH: Monitoring of the immune dysfunction in cancer patients.
Vaccines (Basel). 4:pii: E29. 2016.PubMed/NCBI
|
|
18
|
de Biasi AR, Villena-Vargas J and
Adusumilli PS: Cisplatin-induced antitumor immunomodulation: A
review of preclinical and clinical evidence. Clin Cancer Res.
20:5384–5391. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Javeed A, Ashraf M, Riaz A, Ghafoor A,
Afzal S and Mukhtar MM: Paclitaxel and immune system. Eur J Pharm
Sci. 38:283–290. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chang CL, Hsu YT, Wu CC, Lai YZ, Wang C,
Yang YC, Wu TC and Hung CF: Dose-dense chemotherapy improves
mechanisms of antitumor immune response. Cancer Res. 73:119–127.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–47.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Huang A, Zhang B, Wang B, ZAhang F, Fan KX
and Guo YJ: Increased CD14(+)HLA-DR (−/low) myeloid-derived
suppressor cells correlate with extrathoracic metastasis and poor
response to chemotherapy in non-small cell lung cancer patients.
Cancer Immunol Immunother. 62:1439–1451. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kamphorst AO, Pillai RN, Yang S, Nasti TH,
Akondy RS, Wieland A, Sica GL, Yu K, Koenig L, Patel NT, et al:
Proliferation of PD-1+ CD8 T cells in peripheral blood after
PD-1-targeted therapy in lung cancer patients. Proc Natl Acad Sci
USA. 114:4993–4998. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhou J, Mahoney KM, Giobbie-Hurder A, Zhao
F, Lee S, Liao X, Rodig S, Li J, Wu X, Butterfield LH, et al:
Soluble PD-L1 as a biomarker in malignant melanoma treated with
checkpoint blockade. Cancer Immunol Res. 5:480–492. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Okuma Y, Hosomi Y, Nakahara Y, Watanabe K,
Sagawa Y and Homma S: High plasma levels of soluble programmed cell
death ligand 1 are prognostic for reduced survival in advanced lung
cancer. Lung Cancer. 104:1–6. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhang J, Gao J, Li Y, Nie J, Dai L, Hu W,
Chen X, Han J, Ma X, Tian G, et al: Circulating PD-L1 in NSCLC
patients and the correlation between the level of PD-L1 expression
and the clinical characteristics. Thorac Cancer. 6:534–538. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Dronca RS, Mansfield AS, Liu X, Harrington
S, Enninga EA, Kottschade LA, Koo CW, McWilliams RR, Block MS,
Nevala WK, et al: Bim and soluble PD-L1 (sPD-L1) as predictive
biomarkers of response to anti-PD-1 therapy in patients with
melanoma and lung carcinoma. J Clin Oncol. 35 15 Suppl:S115342017.
View Article : Google Scholar
|
|
29
|
Paladini L, Fabris L, Bottai G, Raschioni
C, Calin GA and Santarpia L: Targeting microRNAs as key modulators
of tumor immune response. J Exp Clin Cancer Res. 35:1032016.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Triozzi PL, Achberger S, Aldrich W, Elson
P, Garcia J and Dreicer R: Differential immunologic and microRNA
effects of 2 dosing regimens of recombinant human
granulocyte/macrophage colony stimulating factor. J Immunother.
35:587–594. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Achberger S, Aldrich W, Tubbs R, Crabb JW,
Singh AD and Triozzi PL: Circulating immune cell and microRNA in
patients with uveal melanoma developing metastatic disease. Mol
Immunol. 58:182–186. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chen J, Yuan L, Fan Q, Su F, Chen Y and Hu
S: Adjuvant effect of docetaxel on the immune responses to
influenza A H1N1 vaccine in mice. BMC Immunol. 13:362012.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Yuan L, Wu L, Chen J, Wu Q and Hu S:
Paclitaxel acts as an adjuvant to promote both Th1 and Th2 immune
responses induced by ovalbumin in mice. Vaccine. 28:4402–4410.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Volk V, Cathomas R, Mark M, von Moos R,
Klingbiel D, Brossart P and Mey U: Weekly carboplatin in
combination with weekly paclitaxel in the treatment of metastatic
non-small cell lung cancer: A single center 10-year experience.
Support Care Cancer. 24:2119–2128. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Schuette W, Blankenburg T, Guschall W,
Dittrich I, Schroeder M, Schweisfurth H, Chemaissani A, Schumann C,
Dickgreber N, Appel T and Ukena D: Multicenter randomized trial for
stage IIIB/IV non-small-cell lung cancer using every-3-week versus
weekly paclitaxel/carboplatin. Clin Lung Cancer. 7:338–343. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Caiado J and Castells M: Presentation and
diagnosis of hypersensitivity to platinum drugs. Curr Allergy
Asthma Rep. 15:152015. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Carthon BC, Wolchok JD, Yuan J, Kamat A,
Tang Ng DS, Sun J, Ku G, Troncoso P, Logothetis CJ, Allison JP and
Sharma P: Preoperative CTLA-4 blockade: Tolerability and immune
monitoring in the setting of a presurgical clinical trial. Clin
Cancer Res. 16:2861–2871. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ribas A, Shin DS, Zaretsky J, Frederiksen
J, Cornish A, Avramis E, Seja E, Kivork C, Siebert J, Kaplan-Lefko
P, et al: PD-1 blockade expands intratumoral memory T cells. Cancer
Immunol Res. 4:194–203. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Weber JS, Kudchadkar RR, Yu B, Gallenstein
D, Horak CE, Inzunza HD, Zhao X, Martinez AJ, Wang W, Gibney G, et
al: Safety, efficacy, and biomarkers of nivolumab with vaccine in
ipilimumab-refractory or -naive melanoma. J Clin Oncol.
31:4311–4318. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Heegaard NH, Schetter AJ, Welsh JA, Yoneda
M, Bowman ED and Harris CC: Circulating micro-RNA expression
profiles in early stage nonsmall cell lung cancer. Int J Cancer.
130:1378–2886. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Shen J, Todd NW, Zhang H, Yu L, Lingxiao
X, Mei Y, Guarnera M, Liao J, Chou A, Lu CL, et al: Plasma
microRNAs as potential biomarkers for non-small-cell lung cancer.
Lab Invest. 91:579–587. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zheng D, Haddadin S, Wang Y, Gu LQ, Perry
MC, Freter CE and Wang MX: Plasma microRNAs as novel biomarkers for
early detection of lung cancer. Int J Clin Exp Pathol. 4:575–586.
2011.PubMed/NCBI
|
|
43
|
Zhu W, He J, Chen D, Zhang B, Xu L, Ma H,
Liu X, Zhang Y and Le H: Expression of miR-29c, miR-93, and miR-429
as potential biomarkers for detection of early stage non-small lung
cancer. PLoS One. 9:e877802014. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Zhang H, Mao F, Shen T, Luo Q, Ding Z,
Qian L and Huang J: Plasma miR-145, miR-20a, miR-21 and miR-223 as
novel biomarkers for screening early-stage non-small cell lung
cancer. Oncol Lett. 13:669–676. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Wu C, Cao Y, He Z, He J, Hu C, Duan H and
Jiang J: Serum levels of miR-19b and miR-146a as prognostic
biomarkers for non-small cell lung cancer. Tohoku J Exp Med.
232:85–95. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Zhou M, Liu Z, Zhao Y, Ding Y, Liu H, Xi
Y, Xiong W, Li G, Lu J, Fodstad O, Riker AI and Tan M:
MicroRNA-125b confers the resistance of breast cancer cells to
paclitaxel through suppression of pro-apoptotic Bcl-2 antagonist
killer 1 (Bak1) expression. J Biol Chem. 285:21496–21507. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Kong F, Sun C, Wang Z, Han L, Weng D, Lu Y
and Chen G: miR-125b confers resistance of ovarian cancer cells to
cisplatin by targeting pro-apoptotic Bcl-2 antagonist killer 1. J
Huazhong Univ Sci Technolog Med Sci. 31:5432011. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Liu M, Wang J, Huang H, Hou J, Zhang B and
Wang A: miR-181a-Twist1 pathway in the chemoresistance of tongue
squamous cell carcinoma. Biochem Biophys Res Commun. 441:364–370.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Pogribny IP, Filkowski JN, Tryndyak VP,
Golubov A, Shpyleva SI and Kovalchuk O: Alterations of microRNAs
and their targets are associated with acquired resistance of MCF-7
breast cancer cells to cisplatin. Int J Cancer. 127:1785–1794.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Boldin MP, Taganov KD, Rao DS, Yang L,
Zhao JL, Kalwani M, Garcia-Flores Y, Luong M, Devrekanli A, Xu J,
et al: miR-146a is a significant brake on autoimmunity,
myeloproliferation, and cancer in mice. J Exp Med. 208:1189–1201.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Li L, Zhang J, Diao W, Wang D, Wei Y,
Zhang CY and Zen K: MicroRNA-155 and MicroRNA-21 promote the
expansion of functional myeloid-derived suppressor cells. J
Immunol. 192:1034–1043. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Zhang M, Liu Q, Mi S, Liang X, Zhang Z, Su
X, Liu J, Chen Y, Wang M, Zhang Y, et al: Both miR-17-5p and
miR-20a alleviate suppressive potential of myeloid-derived
suppressor cellsby modulating STAT3 expression. J Immunol.
186:4716–4724. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Liu Q, Zhang M, Jiang X, Zhang Z, Dai L,
Min S, Wu X, He Q, Liu J, Zhang Y, et al: miR-223 suppresses
differentiation of tumor-induced CD11b+ Gr1+
myeloid-derived suppressor cells from bone marrow cells. Int J
Cancer. 129:2662–2673. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Dooley J, Linterman MA and Liston A:
MicroRNA regulation of T-cell development. Immunol Rev. 253:53–64.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Galluzzi L, Morselli E, Vitale I, Kepp O,
Senovilla L, Criollo A, Servant N, Paccard C, Hupé P, Robert T, et
al: miR-181a and miR-630 regulate cisplatin-induced cancer cell
death. Cancer Res. 70:1793–1803. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Li H, Zhang P, Sun X, Sun Y, Shi C, Liu H
and Liu X: MicroRNA-181a regulates epithelial-mesenchymal
transition by targeting PTEN in drug-resistant lung adenocarcinoma
cells. Int J Oncol. 47:1379–1392. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Zhu M, Zhou X, Du Y, Huang Z, Zhu J, Xu J,
Cheng G, Shu Y, Liu P, Zhu W and Wang T: miR-20a induces cisplatin
resistance of a human gastric cancer cell line via targeting CYLD.
Mol Med Rep. 14:1742–1750. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Lin HM, Castillo L, Mahon KL, Chiam K, Lee
BY, Nguyen Q, Boyer MJ, Stockler MR, Pavlakis N, Marx G, et al:
Circulating microRNAs are associated with docetaxel chemotherapy
outcome in castration-resistant prostate cancer. Br J Cancer.
110:2462–2471. 2014. View Article : Google Scholar : PubMed/NCBI
|