Introduction
Hepatocellular carcinoma (HCC) is one of the most
common types of cancer of the digestive system. HCC is the third
most common cause of cancer-associated mortality worldwide
(1–4),
and the second in China (5). Surgery
is the preferred treatment for HCC, while chemotherapy is applied
in cases of unresectable HCC (6). A
poor prognosis and high mortality rate continue to be associated
with HCC. The tendency of intrahepatic metastasis in the early
stages means that HCC is frequently not identified while it remains
resectable (7). Damage to the immune
system following radiotherapy and chemotherapy further increases
the difficulty in the rehabilitation of patients (8).
Cancer is determined not only by the genetic
composition of the tumor cells, but also by the surrounding
environment, which supports the growth, proliferation and
metastasis of tumor cells (9). While
a significant body of research has focused on the characteristics
and mechanisms of HCC cells, the role of the tumor microenvironment
in HCC is largely unknown. Certain immune cells in the tumor
microenvironment directly promote the growth of tumor cells.
Macrophages can produce insulin-like growth factor-1 to activate
the extracellular signal-regulated kinase and protein kinase B
pathways in lung cancer, directly stimulating tumor proliferation
and growth (10). Moreover,
regulatory T cells (Tregs) can significantly increase the invasive
ability of cancer cells and inhibit the antitumor immune response
in ovarian cancer (11). The extent
of forkhead box P3-positive (FOXP3+) Treg infiltration in HCC
tissue is associated with high-grade, poorly differentiated tumors,
and may be indicative of a poor prognosis (12). Th17 is a type of helper T cell
produced by differentiation from Th0 cells upon stimulation with
interleukin (IL)-6 and IL-23. Th17 cells secrete proinflammatory
factors, including IL-17 and IL-22; and retinoic acid
receptor-related orphan receptor-γ (RORγ), especially the isoform
RORγ2 (also known as RORγt), is an important transcription factor
in this process (13). High
expression of IL-17 and IL-17 receptors is associated with a poor
prognosis in patients with HCC (14).
Cisplatin is one of the most commonly used and
effective anticancer chemotherapy drugs, but it is associated with
severe tissue damage and other adverse reactions (15). Previous studies have indicated that
cisplatin has higher efficacy and fewer side effects when used in
combination with other anticancer drugs (16). Moreover, herbs from traditional
Chinese medicines have been reported to improve the immune response
of patients with HCC (17).
Herbs, including safflower (Carthamus tinctorius
L.), have been demonstrated to promote blood circulation and remove
blockages to treat menstrual disorders, trauma, joint pain and
abdominal masses in traditional Chinese medicine (18,19). The
main active component of safflower is hydroxyl safflower yellow A
(HSYA). Recently, HSYA has been found to attenuate the apoptosis of
peripheral blood T lymphocytes positive for T-cell surface
glycoprotein CD4 (CD4) in a murine model of sepsis, to inhibit the
proliferation and metastasis of tumor cells and tumor angiogenesis,
and to induce tumor cell apoptosis (20–24).
Furthermore, HSYA treatment has been demonstrated to decrease the
expression of matrix metalloproteinase and hypoxia inducible factor
1α in mice with xenograft tumors (25,26).
In the present study, the potential anticancer
effect of HSYA on the tumor immune microenvironment in HCC was
investigated. Following the establishment of an HCC model in BALB/c
mice, the anticancer effect of HSYA at varying concentrations was
evaluated, the Treg ratio in the spleen was assessed and the
expression of several crucial molecules in the tumor
microenvironment was measured. We hypothesized that HSYA treatment
would have anticancer effects and low toxicity in mouse HCC,
highlighting the potential of this compound in cancer therapy.
Materials and methods
Reagents
HSYA (purity ≥95%) was purchased from China
Pharmaceutical and Biological Products (Beijing, China). Cisplatin
was purchased from Sigma-Aldrich; Merck KGaA (Darmstadt, Germany;
product number, P4394; concentration ≤100%). fetal bovine serum
(FBS) was obtained from Hyclone; GE Healthcare Life Sciences
(Logan, UT, USA), high glucose-DMEM from Gibco; Thermo Fisher
Scientific, Inc., (Waltham, MA, USA) and the Mouse Regulatory T
Cell Staining kit from eBioscience; Thermo Fisher Scientific, Inc.
The SV Total RNA Isolation system and SYBR Green mix were obtained
from Promega Corporation (Madison, WI, USA), the primers from
Sangon Biotech Co., Ltd. (Shanghai, China), the anti-FOXP3 (cat.
no. ab75763) and anti-RORγt (cat. no. ab207082) antibodies from
Abcam (Cambridge, MA, USA) and the SuperEnhanced chemiluminescence
detection kit from Applygen Technology, Inc. (Beijing, China).
Animals and cells
A total of 70 male BALB/c mice, aged 6–8 weeks,
weighing 19–21 g each, were purchased from Si Pei Fu Laboratory
Animal Technology Co. Ltd. (Beijing, China; certificate no: SCXK
Jing 2011–0004). The mice were raised in the animal room of Beijing
University of Chinese Medicine, at 25°C with 12 h of light and 12 h
of darkness, and fed with food and water. The Hepa1-6 murine HCC
cell line was purchased from the Institute of Basic Medical
Sciences (Chinese Academy of Medical Sciences, Beijing, China).
Cells were maintained in high-glucose DMEM supplemented with 10%
FBS at 37°C in a humidified atmosphere containing 5% CO2. Following
3 to 4 passages, cells in the logarithmic growth phase were used
for the subsequent experiments.
Model establishment
The experiment was approved by the Institutional
Animal Ethics Committee of Beijing University of Chinese Medicine
(Beijing, China). A PBS suspension was made of Hepa1-6 cells at
4×107 cells/ml. BALB/c mice were acclimated to their surroundings
for 7 days and fasted for 12 h. Anesthesia was then induced by
isoflurane inhalation (2–4%) and preoperative subcutaneous
application of buprenorphine (0.1 mg/kg) (27). Following anesthesia, the mice were
placed on a plate in the supine position and their fur was removed.
Subsequent to sterilization with iodine, a laparotomy was performed
along the linea alba, exposing the abdominal cavity. Light pressure
was applied to the thoracic cavity to expose the mouse liver. A
microsyringe was used to inject 1×106 Hepa1-6 cells into the
surface of the proximal body of the liver, while the mice in the
control group were injected with an equal volume of normal saline.
The syringe tip was inserted ~1 cm into the liver at a 15°-angle. A
total of 25 µl of the cell suspension was injected slowly. The
needle was slowly withdrawn, pressure was applied for a moment with
a sterile cotton swab and then the liver was reinserted into the
abdominal cavity. The abdomen was closed and the wound was
disinfected. All mice were sacrificed 11 days later by cervical
dislocation. Following laparotomy, a tumor could be observed in the
liver, indicating the success of establishing the model.
Animal grouping
In total, 70 mice were randomly divided into 7
groups (n=10). Subsequent to receiving an injection of Hepa1-6
cells (saline for the control group) into the liver on day 1, the
mice were treated as follows: The control group and model group
were injected intraperitoneally with 0.2 ml normal saline twice per
day. The high-, medium- and low-dose groups received
intraperitoneal injections of HSYA at 2.25, 1.13 and 0.57 mg/kg
twice per day, respectively. These doses were selected using
preliminary experimental results. The cisplatin group received an
intraperitoneal injection of 5 mg/kg cisplatin once every 2 days at
a fixed time in the morning, and the combination group received an
intraperitoneal injection of 0.57 mg/kg HSYA twice per day, plus 5
mg/kg cisplatin once every 2 days. After 11 days, all mice were
sacrificed.
Histological examination
Tumors were excised and fixed in 4% paraformaldehyde
for 48 h at room temperature, rinsed with water, dehydrated with
gradient ethanol and embedded in paraffin at 56°C. The slices were
cut into 4-µm layers and stained with hematoxylin for 5 min and
subsequently, with eosin for 1 min at room temperature.
Pathological changes to the tumor tissue was observed under an
optical microscope (Olympus Corporation, Tokyo, Japan).
Weight and index calculation
The body weights of the mice were recorded every 2
days. Subsequent to sacrifice, the spleen and thymus were
aseptically removed and weighed, and the spleen and thymus indexes
were calculated as follows: Spleen index=spleen weight (mg)/mouse
weight (g) × 10; and thymus index=thymus weight (mg)/mouse weight
(g) × 10. The spleen and thymus index reflect the weight of the
spleen and thymus, which are related to the number of immune cells
contained within. An increasing index indicates enhanced immunity
(28).
Flow cytometry
The Treg ratio in the spleen was assessed by flow
cytometry according to the protocol of the Mouse Regulatory T Cell
Staining kit. Briefly, a suspension of spleen cells (1×107
cells/ml) was prepared by grinding, filtering and washing the
spleen samples with PBS. A total of 100 µl cell suspension was
added to each flow tube. Anti-mouse CD4 (0.25 µl) and anti-mouse
IL-2 receptor subunit α CD25 (CD25) (0.3 µl) antibodies were added
and incubated for 60 min at 4°C. The cells were then washed and
lysed. Fc block (1 µl) was added and incubated for 15 min at room
temperature. Next, anti-mouse FOXP3 antibody (2.0 µl) was added.
Following incubation at room temperature for 60 min, the cells were
washed and analyzed using a flow cytometer (BD Biosciences, San
Jose, CA, USA). FlowJo software, version 10 (FlowJo LLC, Ashland,
OR, USA) was used to analyze the sample data.
Reverse transcription
quantitative-polymerase chain reaction analysis (RT-qPCR)
Total RNA was extracted from tumor samples using the
SV Total RNA Isolation system. The reverse transcription reaction
was performed as follows: Firstly, the reaction mixture (3 µg total
RNA, Oligo (dT), nuclease-free water) was incubated at 95°C for 5
min, and immediately placed on ice; secondly, the RT reaction was
performed at 42°C for 60 min, and at 75°C for 15 min in a 20 µl
volume with 3 µg total RNA, dNTPs, Oligo (dT), RNasin ribonuclease
inhibitor, Moloney murine leukemia virus (M-MLV) reverse
transcriptase, 5X reverse transcriptase buffer and nuclease-free
water. qPCR was performed with a CFX96 Real Time-PCR system
(Bio-Rad Laboratories, Inc., Hercules, CA, USA) in a 20 µl reaction
volume of 2 µl cDNA, 1 µl primer, 7 µl nuclease-free water and 10
µl SYBR Green mix. The sequences of the PCR primers were as
follows: Foxp3 forward, 5′-CTGCCTTGGTACATTCGTGAAC-3′ and reverse,
5′-ATGTTGTGGGTGAGTGCTTTG-3′; Rorγt forward,
5′-GCTCTGCCAGAATGACCAGA-3′ and reverse, 5′-CAGCTCCACACCACCGTATT-3′;
and β-actin forward, 5′-CCAGCCTTCCTTCTTGGGTATG-3′ and reverse,
5′-TGTTGGCATAGAGGTCTTTACGG-3′. PCR was performed as follows:
Denaturation at 95°C for 10 min, followed by 39 cycles at 95°C for
15 sec, 55°C for 30 sec and 72°C for 30 sec. Melting curve analysis
was performed to measure the specificity of the PCR products.
Expression was calculated using the 2−ΔΔCq method
(29), relative to β-actin.
Western blot analysis
Protein was isolated from tumor tissue by routine
methods with radioimmunoprecipitation assay buffer (Beijing
Solarbio Science & Technology Co., Ltd., Beijing, China) and
quantified using the BCA method. The protein was separated by 10%
SDS-PAGE (30 µg protein/lane), and transferred to polyvinylidene
fluoride membranes (EMD Millipore, Bedford, MA, USA). The membranes
were blocked with 5% skimmed milk [1 g skimmed milk powder in 20 ml
TBS with 1% Tween (TBST)] overnight. The membrane was then
incubated with unconjugated rabbit anti-mouse primary antibodies
against FOXP3+ and RORγt at 4°C overnight (anti-bodies were diluted
by TBST at 1:1,000 with 1% sodium azide), washed with TBST 3 times
(10 min each), incubated with a HRP-conjugated goat-anti-rabbit IgG
secondary antibody (cat. no. 7090; Abcam, Cambridge, MA, USA) for 1
h at room temperature and washed again. Finally, the bands were
visualized using the SuperEnhanced chemiluminescence detection
kit.
Statistical analysis
The experimental data are expressed as the mean ±
standard deviation. Multigroup comparisons were performed using
one-way analysis of variance and the least significant difference
post hoc test. All statistical analyses were performed with SPSS
20.0 statistical software (IBM Corporation, Armonk, NY, USA).
P<0.05 was considered to indicate a statistically significant
difference.
Results
Effect of HSYA on tumorigenesis and
pathological changes in HCC model mice
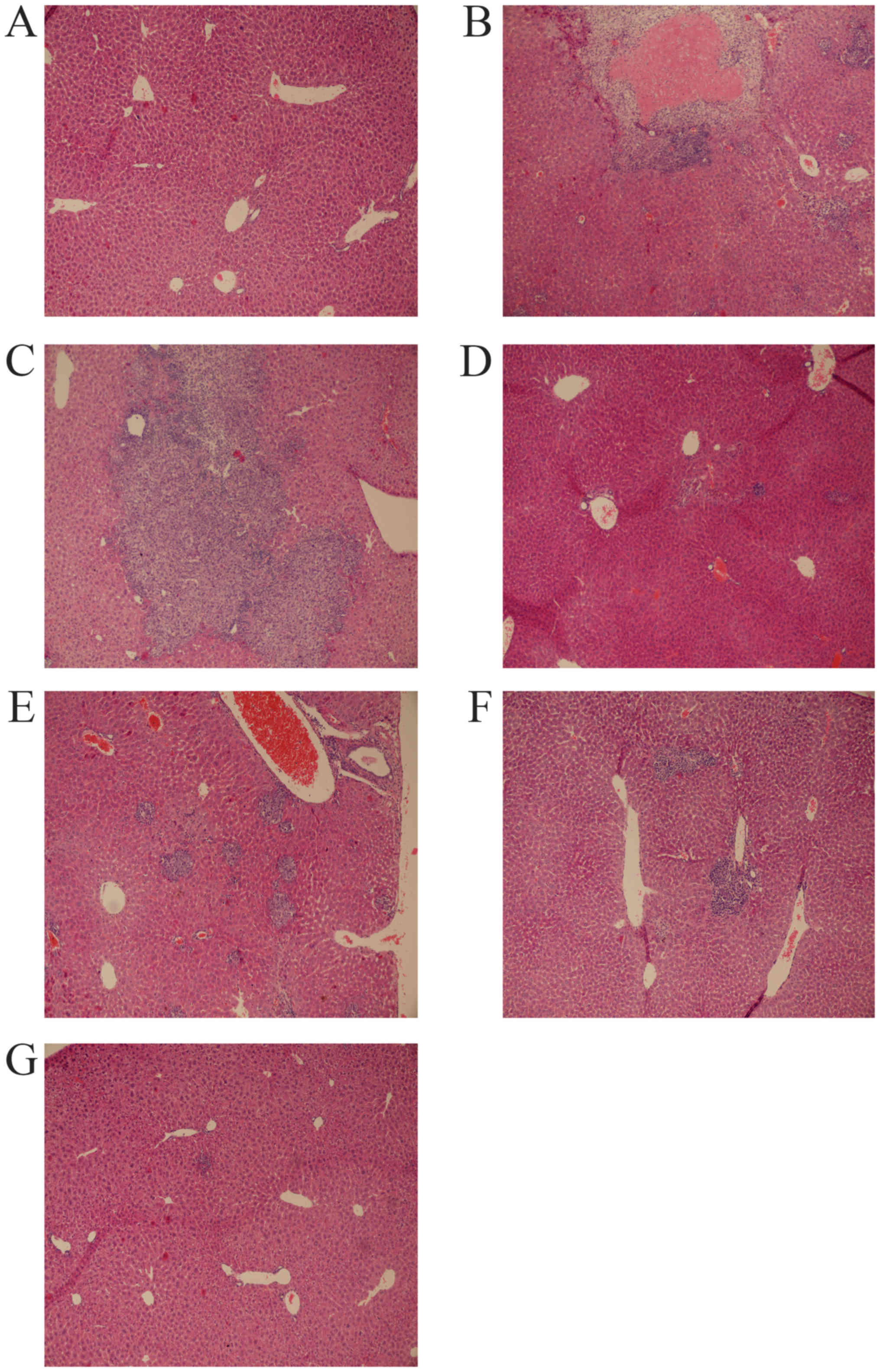
HE staining was performed to detect pathological
changes in the livers of the mice, particularly to observe any
alterations in tumor growth. Liver cells of the mice from the
control group displayed red-stained circular cytoplasm and
blue-stained nuclei, the liver lobular structural integrity was
highly ordered, and the largest vein of the liver was cord-like and
positioned centrally (Fig. 1A). A
large area of solid tumor tissue was found in the central
vasculature in the model group, with tumor cells occurring in
clumps (Fig. 1B). Tumor cells were
generally smaller and more deeply stained than the normal adjacent
cells. The HSYA high- (Fig. 1C),
middle- (Fig. 1D) and low- (Fig. 1E) concentration groups, and the
cisplatin group (Fig. 1F) all
exhibited scattered cancer cell clumps. The number of cancer cells
was markedly lower in these groups than in the model group, and
only precancerous pathological changes were observed. The liver
lobular structural integrity of the mice in the cisplatin group was
disordered. In the combination group (Fig. 1G), scattered liver cancer cells and
evidence of precancerous pathological alterations were observed,
but the normal structure of the liver cells was more intact.
Therefore, HSYA inhibited the proliferation of liver cancer cells
and reduced the extent of tissue damage induced by cisplatin.
Effect of HSYA on body weight of HCC model mice. In
order to examine whether HSYA induced adverse effects on mice or
reduced the side-effects caused by cisplatin, the body weight of
the mice was measured every 2 days. As shown in Table I, the mice treated with cisplatin or a
combination of cisplatin and HSYA exhibited a decreasing trend in
body weight, while mice treated with HSYA alone gained weight.
Compared with that of the control group, the body weight of the
model group increased, which may have been caused by tumor growth.
The body weight of the HSYA groups was not significantly different
to the body weight of the model group, while the weight of the mice
in the cisplatin group was significantly decreased from the 5th day
compared with that of the model group (P<0.01). The weight of
the mice in the combination group also decreased from the 7th day
(P<0.01). Compared with the cisplatin group, the body weight of
the mice in the combination group was significantly higher on the
5th day (P<0.05). These results indicate that HSYA treatment did
not lead to negative effects on mouse body weight and reduced the
extent of weight loss in the mice treated with cisplatin.
 | Table I.Effect of HSYA on body weight of
hepatocellular carcinoma model mice. |
Table I.
Effect of HSYA on body weight of
hepatocellular carcinoma model mice.
| Group | 1 day | 3 days | 5 days | 7 days | 9 days | 11 days |
|---|
| Control | 20.19±1.001 | 20.91±1.660 | 21.18±1.885 | 21.46±1.773 | 21.23±2.119 | 21.23±2.356 |
| Model | 20.47±0.472 | 21.11±0.522 | 21.68±0.611 | 21.70±0.668 | 21.81±0.869 | 21.12±1.319 |
| HSYA high dose | 20.90±0.572 | 21.17±0.539 | 22.09±0.610 | 21.88±0.601 | 22.35±0.721 | 22.31±0.574 |
| HSYA medium
dose | 20.83±0.699 | 21.26±0.704 | 21.89±0.948 | 21.68±0.836 | 22.02±0.885 | 21.84±0.773 |
| HSYA low dose | 20.74±0.654 | 20.59±0.749 | 21.17±0.815 | 20.78±0.722 | 20.75±0.428 | 21.18±0.464 |
| Cisplatin | 20.55±0.430 | 20.48±0.573 |
19.96±0.486b |
18.94±0.403b |
17.57±0.479b |
17.93±0.818b |
| Combination | 20.63±0.942 | 20.96±1.176 |
20.71±1.358a |
19.81±1.462b |
19.07±1.746b |
17.68±1.582b |
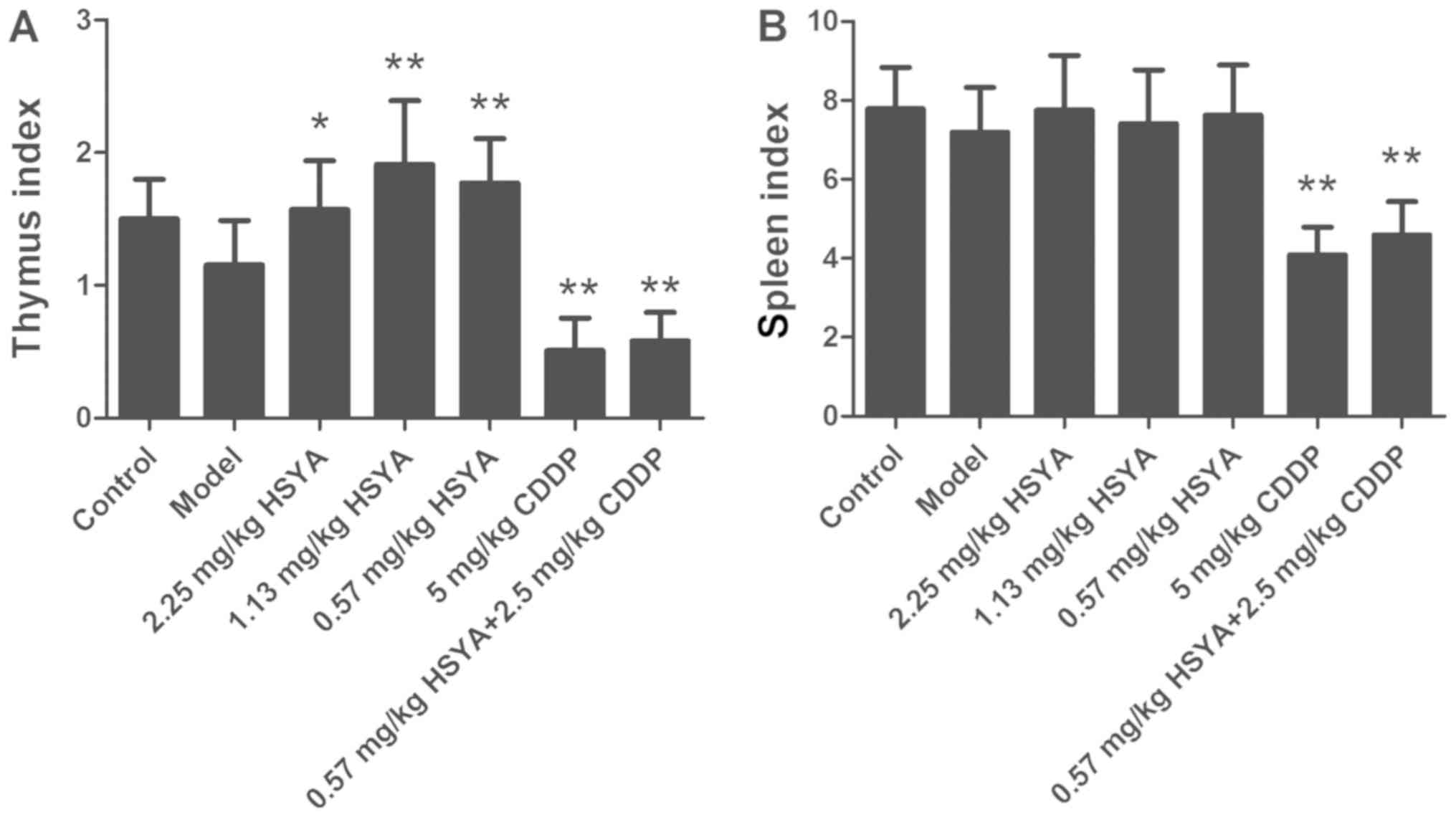
Effect of HSYA on the thymus and
spleen indexes in HCC model mice
To investigate whether HSYA protected the immune
system from damage by cisplatin treatment, the thymus and spleen
indexes were measured as indicators for immunity. The effect of
HSYA on the thymus and spleen indexes is displayed in Fig. 2. Compared with the control group, the
thymus and spleen indexes of the model group decreased (P>0.05),
whereas compared with the model group, the HSYA groups exhibited an
increased thymus index (P<0.05). The effects of middle (1.13
mg/kg) and low (0.57 mg/kg) concentrations of HSYA on the thymus
index were particularly significant (P<0.01). However, as for
the spleen index, compared with the control group, there was no
significant change in the model group and compared with the model
group, there was no significant change in the HSYA groups. In the
cisplatin and combination groups, the thymus and spleen indexes
were significantly decreased (P<0.01). Compared with the
cisplatin group, the combination group trended towards an increase
in the thymus and spleen indexes, but there was no significant
difference. These results indicated that HSYA treatment increased
the thymus index of HCC model mice, potentially reflecting an
improvement in immunity, whereas HSYA could not rescue the
decreased thymus index induced by cisplatin treatment.
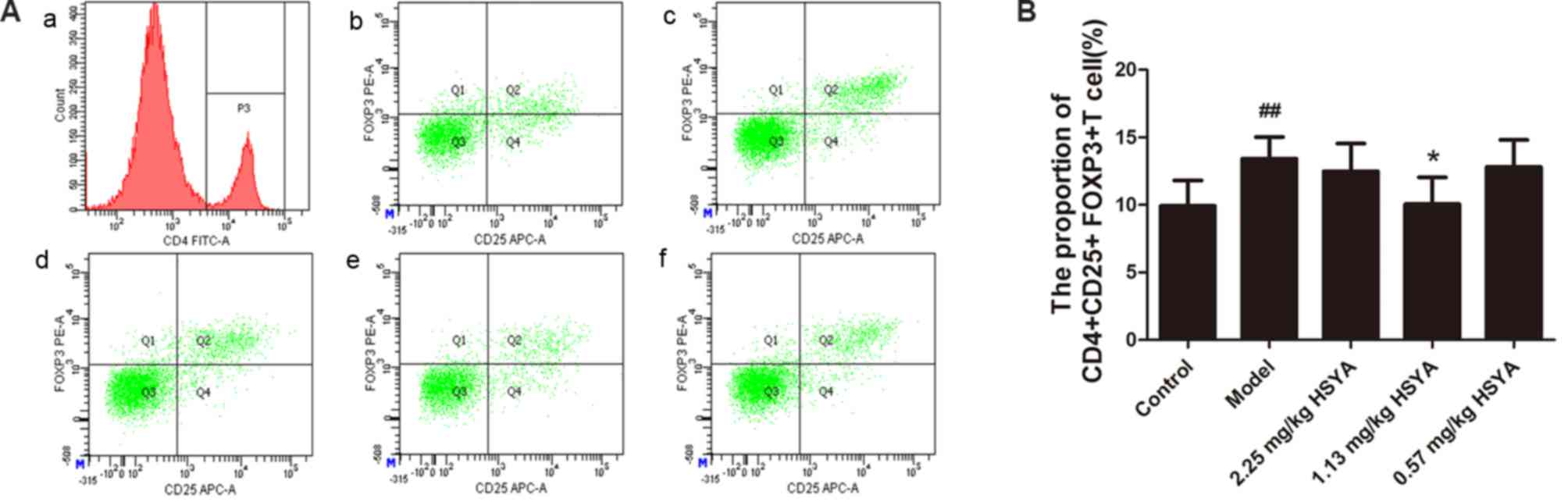
Effect of HSYA on the proportion of
CD4+CD25+FOXP3+ Tregs in the
spleen of HCC model mice
As the aforementioned data indicates, HSYA inhibited
tumor growth and improved the thymus index. The underlying
mechanisms were then considered. As illustrated in Fig. 3, the ratio of
CD4+CD25+FOXP3+ Tregs to all
CD4+ T lymphocytes
(CD4+CD25+FOXP3+
Tregs/CD4+ T lymphocytes) in the spleen of mice from the
model group was significantly higher than that of the control group
(P<0.01). The ratio of
CD4+CD25+FOXP3+ Tregs in the
spleen was significantly decreased in mice treated with 1.13 mg/kg
HSYA compared with the model group (P<0.05), whereas the ratio
change was not significant for the high (2.25 mg/kg) and low (0.57
mg/kg) HSYA groups. These results indicated that HSYA may decrease
the proportion of Tregs in the spleen of HCC model mice at specific
doses. This may be because Tregs only respond to HSYA at a certain
concentration range, but this conclusion requires further
investigation.
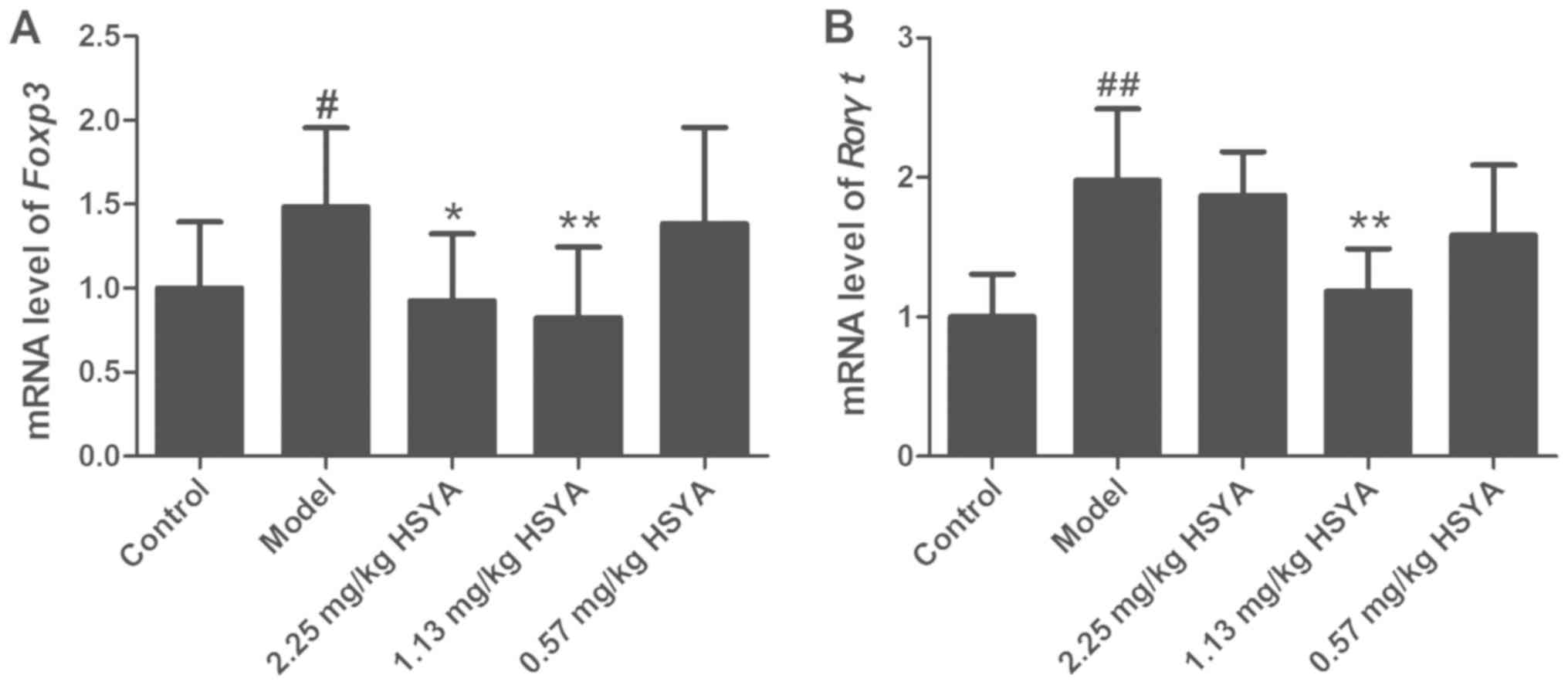
Effect of HSYA on the expression of
Foxp3 and Rorγt mRNA in tumor tissue from HCC model mice
The expression levels of Foxp3 and Rorγt mRNA in
tumor samples are displayed in Fig.
4. The expression of Foxp3 (P<0.05) and Rorγt (P<0.01)
was increased in the model group compared with that in the control
group. Compared with that in the model group, the expression of
Foxp3 was decreased in the high- and medium-dose HSYA groups,
particularly in the middle group (1.13 mg/kg) (P<0.01). Rorγt
expression was decreased in the medium-dose group only (P<0.01)
compared with that in the model group. These results indicated that
HSYA treatment reduced the expression of Foxp3 and Rorγt mRNA at
certain doses.
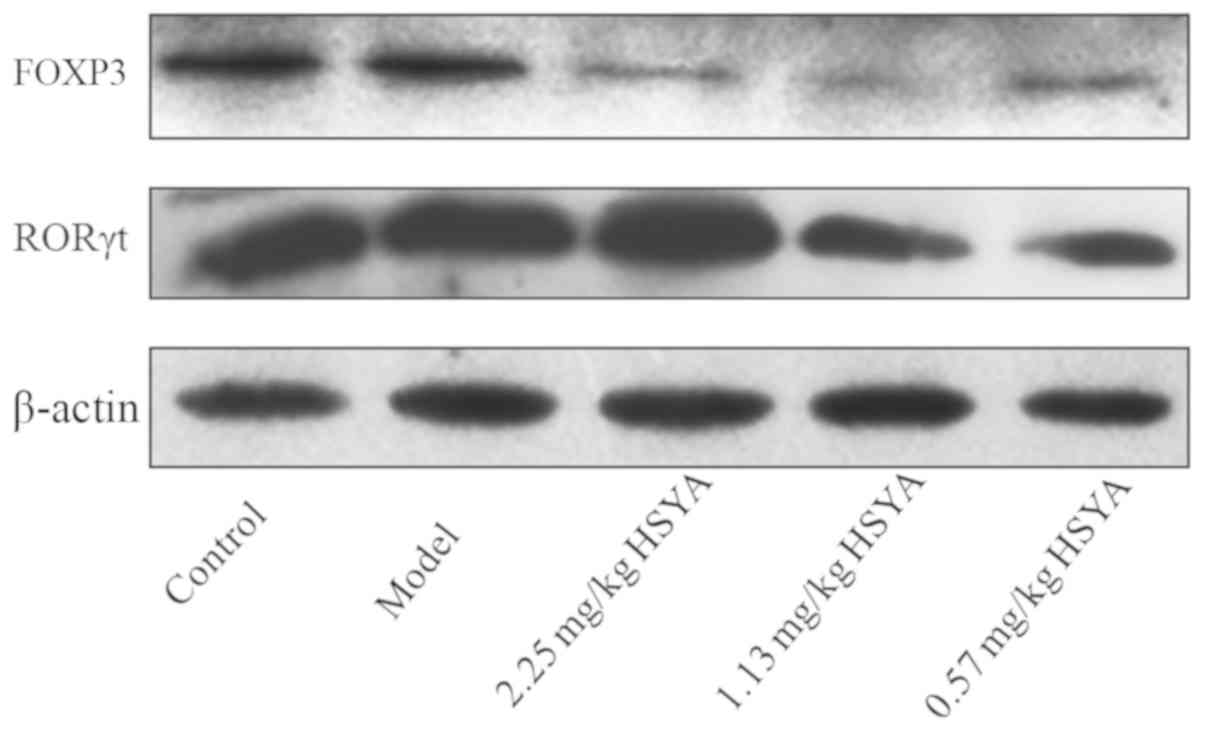
Effect of HSYA on FOXP3 and RORγt
protein expression in tumor tissue from HCC model mice
Compared with that in the model group, the
expression of FOXP3 protein in the HSYA groups was decreased, which
was most evident for the middle-concentration group (1.13 mg/kg)
(Fig. 5). The middle- and
low-concentration groups also induced a reduction in RORγt protein
expression. These results indicated that HSYA treatment decreases
the expression of FOXP3 and RORγt protein, two important modulators
of the tumor immune microenvironment. Therefore, HSYA treatment may
improve the tumor immune microenvironment of HCC model mice.
Discussion
HSYA has been demonstrated to protect
cardiovascular, cerebrovascular and nerve cells, and to prevent
inflammation, oxidation and tumors (30–33). As
aforementioned, cisplatin is one of the most commonly used cancer
chemotherapy drugs. Cisplatin inhibits DNA replication in cancer
cells and damages the structure of cancer cell membranes, but its
harmful side effects limit its use. Studies have revealed that
combining traditional Chinese medicine with cisplatin may enhance
its antitumor effect (34,35). Park et al (36) found that cisplatin combined with
HemoHIM, a preparation of three herbs, not only significantly
reduced tumor volumes, but also enhanced the activity of
interferon-γ and IL-2, and increased the ratio of cytotoxic T cells
and natural killer cells in the spleen of mice. The spleen and
thymus are important organs for immune surveillance. Therefore, the
spleen and thymus indexes are two important indicators for the
effect of HSYA on the immune system of mice (37). An increasing index indicates enhanced
immunity (28).
In the present study, the pathological results
demonstrated that the number of cancer cells was reduced following
treatment with HSYA, with the effect of 1.13 mg/kg being the most
apparent, indicating that HSYA inhibited tumor growth most
effectively at this concentration. In the cisplatin and HSYA
combined therapy group, the number of cancer cells was reduced more
than in the cisplatin alone group, and the extent of liver injury
was reduced, indicating that HSYA could enhance the anticancer
effect of cisplatin, and reduce liver tissue damage due to
cisplatin chemotherapy. The body weight and thymus index of mice
receiving HSYA tended to increase compared with the model group,
while mice receiving cisplatin exhibited a reduced weight, thymus
and spleen index. This indicated that cisplatin treatment has a
negative effect on the physical condition of mice and their immune
system, whereas HSYA did not affect the weight of HCC model mice.
Moreover, at a certain concentration, HSYA improved the immunity of
HCC model mice, as well as reducing cisplatin chemotherapy-induced
weight loss.
Tregs inhibit antitumor immune responses and FOXP3
is an inhibitory transcription factor within them (38). It was previously illustrated that
Tregs are enriched in HCC tissue (39). Another previous study demonstrated
that the proportion of FOXP3+ Tregs in tumor tissue and
the proportion of
CD4+CD25+Tregs/CD4+ T lymphocytes
in the spleen were negatively correlated with patient prognosis.
Therefore, there is clinical value in measuring FOXP3+
Tregs to predict HCC recurrence and survival (40). Reducing the proportion of Tregs may
inhibit tumor growth by regulating the immunosuppressive state of
the tumor microenvironment (41).
Th17 cells are a key component of the inflammatory
and immune responses that produce IL-17. In HCC patients, the
presence of Th17 cells is negatively associated with the overall
and recurrence-free survival rate (42). Huang et al (43) found that intrahepatic
IL-17+ Th17 cells and FOXP3+ Tregs exhibited
a synergistic effect, promoting the progress of HCC. RORγt is a
Th17 cell-specific transcription factor (13). A previous study found that the mRNA
levels of RORγt and FOXP3 were significantly increased in
peripheral blood mononuclear cells from patients with HCC,
indicating high Th17 and Treg numbers (44).
The results of the present study demonstrated that
1.13 mg/kg HSYA significantly reduced the proportion of Tregs in
the spleens of HCC model mice, and reduced the expression of Foxp3
and Rorγt in mouse cancer tissue, indicating that HSYA may suppress
tumor growth and immune escape by reducing the number of Tregs,
potentially reducing inflammation to improve the tumor immune
microenvironment and relieving the immunosuppressive state. The
specific mechanisms of this effect remain to be studied.
In summary, 1.13 mg/kg HSYA regulated the immune
microenvironment of HCC model mice by reducing the proportion of
Tregs in the spleen, and the expression of Foxp3 and Rorγt in tumor
tissue, enhancing the immunity of mice and reducing the side
effects of cisplatin when used at a particular concentration, thus
exerting an anticancer effect. However, the immune environment of
the body is complex, with a variety of immune factors, and the
present study selected a small portion of them to investigate. The
effect of HSYA on other important immune factors requires further
examination.
Acknowledgements
The authors would like to thank Dr Wenbin Ou
(Institution of Life Sciences, ZheJiang Sci-Tech University), who
assisted with the experimental design and data analysis.
Funding
The present study was supported by the National
Natural Science Foundation of China (grant no. 81473655, 31500640
and 31770849), the Beijing University of Traditional Chinese
Medicine Graduate Student Project (grant no. 2015-JYB-XS041), the
Natural Science Foundation of Zhejiang Province (grant no.
LY15C070002 and LY16C050001), the fund from the Science Technology
Department of Zhejiang Province (grant no. 2015C33131 and
2016F10005) and the fund from the Science and Technology Bureau of
Jiaxing (grant no. 2015AY23007).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
YM and CF analyzed the data and wrote the
manuscript. JW, ZC and PW performed the experiments. AF, XW, XY,
DG, HX and LL assisted in preparing the experiments. QZ and XL
designed the study. All authors have read and approved the final
manuscript.
Ethics approval and consent to
participate
The present study was approved by the Institutional
Animal Ethics Committee of Beijing University of Chinese
Medicine.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Yang JD, Nakamura I and Roberts LR: The
tumor microenvironment in hepatocellular carcinoma: Current status
and therapeutic targets. Semin Cancer Biol. 21:35–43. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Luo P, Yin P, Hua R, Tan Y, Li Z, Qiu G,
Yin Z, Xie X, Wang X, Chen W, et al: A Large-scale, multicenter
serum metabolite biomarker identification study for the early
detection of hepatocellular carcinoma. Hepatology.
28–Sep;2017.(Epub ahead of print).
|
|
3
|
Parkin DM, Bray F, Ferlay J and Pisani P:
Global cancer statistics, 2002. CA Cancer J Clin. 55:74–108. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ferlay J, Shin HR, Bray F, Forman D,
Mathers C and Parkin DM: Estimates of worldwide burden of cancer in
2008: GLOBOCAN 2008. Int J Cancer. 127:2893–2917. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chen JG: Trends in the incidence of liver
cancer and its primary prevention in China. J Clin Hepatol.
28:256–260. 2012.
|
|
6
|
Bruix J and Sherman M; American
Association for the Study of Liver Diseases, : Management of
hepatocellular carcinoma: An update. Hepatology. 53:1020–1022.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Karaman B, Battal B, Sari S and Verim S:
Hepatocellular carcinoma review: Current treatment, and
evidence-based medicine. World J Gastroenterol. 20:18059–18060.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Greten TF, Wang XW and Korangy F: Current
concepts of immune based treatments for patients with HCC: From
basic science to novel treatment approaches. Gut. 64:842–848. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Mbeunkui F and Johann DJ Jr: Cancer and
the tumor microenvironment: A review of an essential relationship.
Cancer Chemother Pharmacol. 63:571–582. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Fritz JM, Dwyer-Nield LD and Malkinson AM:
Stimulation of neoplastic mouse lung cell proliferation by alveolar
macrophage-derived, insulin-like growth factor-1 can be blocked by
inhibiting MEK and PI3K activation. Mol Cancer. 10:762011.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu J, Zhang H, Jia L and Sun H: Effects
of Treg cells and IDO on human epithelial ovarian cancer cells
under hypoxic conditions. Mol Med Rep. 11:1708–1714. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mathai AM, Kapadia MJ, Alexander J,
Kernochan LE, Swanson PE and Yeh MM: Role of Foxp3-positive
tumor-infiltrating lymphocytes in the histologic features and
clinical outcomes of hepatocellular carcinoma. Am J Surg Pathol.
36:980–986. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Castro G, Liu X, Ngo K, De Leon-Tabaldo A,
Zhao S, Luna-Roman R, Yu J, Cao T, Kuhn R, Wilkinson P, et al:
RORγt and RORα signature genes in human Th17 cells. PLoS One.
12:e01818682017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liao R, Sun J, Wu H, Yi Y, Wang JX, He HW,
Cai XY, Zhou J, Cheng YF, Fan J and Qiu SJ: High expression of
IL-17 and IL-17RE associate with poor prognosis of hepatocellular
carcinoma. J Exp Clin Cancer Res. 32:32013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhang Q, Zheng J, Wang L, et al:
Therapeutic effect of Ramosetron on preventing gastrointestinal
reaction induced by chemotherapeutic drugs including Cisplatin in
80 cases. Chin Pharm. 10:1024–1025. 2007.
|
|
16
|
Liu ZQ, Cheng JT, Zhu HX, et al:
Experimental study on increasing effect and decreasing its
side-effect of Cisplatin of LiuJunZi decoction. Lishizhen Medicine
and Materia Medical Research. 20:2492–2494. 2009.
|
|
17
|
Wu H, Lv SZ, Ying XZ, Ye YL and Tian ZF:
Effect of traditional Chinese medicine in immunity recovery of
radiotherapy in patients with liver cancer. Chin Archiv Trad Chin
Med. 8:1997–1999. 2015.(In Chinese).
|
|
18
|
Wu QL, Lu HD, Guo XL and Kong QZ:
Exploration of treatment of malignant tumors with Chinese herbs
that invigorate the blood and remove stasis. J Int Trad Western
Med. 5:530–531. 2010.
|
|
19
|
Zhou X, Tang L, Xu Y, Zhou G and Wang Z:
Towards a better understanding of medicinal uses of Carthamus
tinctorius L. in traditional Chinese medicine: A phytochemical and
pharmacological review. J Ethnopharmacol. 151:27–43. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wang J, Wang P, Gui S, Li Y, Chen R, Zeng
R, Zhao P, Wu H, Huang Z and Wu J: Hydroxysafflor yellow A
attenuates the apoptosis of peripheral blood CD4+ T
lymphocytes in a murine model of sepsis. Front Pharmacol.
8:6132017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Xie H, Zhang Q, Zhao YQ, Cui W and Niu X:
The experiment of safflor yellow effecting the cell growth cycle of
mouse H22 grafting tumor. China J Tradit Chin Med Pharm. 171–173.
2006.(In Chinese).
|
|
22
|
Ma L, Liu L, Ma Y, Xie H, Yu X, Wang X,
Fan A, Ge D, Xu Y, Zhang Q and Song C: The role of
E-cadherin/β-catenin in hydroxysafflor yellow A inhibiting
adhesion, invasion, migration and lung metastasis of hepatoma
cells. Biol Pharm Bull. 40:1706–1715. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Xi S, Zhang Q, Xie H, Liu L, Liu C, Gao X,
Zhang J, Wu L, Qian L and Deng X: Effects of hydroxy safflor yellow
A on blood vessel and mRNA expression with VEGF and bFGF of
transplantation tumor with gastric adenocarcinoma cell line BGC-823
in nude mice. Zhongguo Zhong Yao Za Zhi. 34:605–610. 2009.(In
Chinese). PubMed/NCBI
|
|
24
|
Wang J, Zhang Q, Gu LG, Cui W, Xie H and
Niu XY: Effect of hydroxy safflor yellow A on the cell cycle and
apoptosis of human umbilical vein endothelial cells with the
stimulus of tumor cell conditioned medium. J Beijing Univ Tradit
Chin Med. 31:741–744. 2008.(In Chinese).
|
|
25
|
Xi SY, Zhang Q, Liu CY, et al: Effects of
hydroxy safflower yellow A on the expression of bFGF protein and
MMP-9 in human gastric cancer xenografts. Chin J Chin Materia.
21:2877–2881. 2010.(In Chinese).
|
|
26
|
Xi SY, Zhang Q, Liu CY, Xie H, Yue LF, Li
WD, Zang BX and Gao XM: Effects of HSYA on protein and mRNA
expression of KDR, HIF-1α and protein expression of VEGF in nude
mice with BGC-823 transplantation tumor. Chin J Trad Chin Med
Pharm. 27:82–87. 2012.
|
|
27
|
Limani P, Borgeaud N, Linecker M, Tschuor
C, Kachaylo E, Schlegel A, Jang JH, Ungethüm U, Montani M, Graf R,
et al: Selective portal vein injection for the design of syngeneic
models of liver malignancy. Am J Physiol Gastrointest Liver
Physiol. 310:G682–G688. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lu YM and Zhang H: Effects of
electroacupuncture on T-lymphocytes, spleen index, thymus index and
lymphopoiesis levels in strenuous exercise-induced stress rat. Zhen
Ci Yan Jiu. 37:136–139. 2012.PubMed/NCBI
|
|
29
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Dong F, Xue C, Wang Y, Peng Y, Zhang Y,
Jin M and Zang B: Hydroxysafflor yellow A attenuates the expression
of inflammatory cytokines in acute soft tissue injury. Sci Rep.
7:405842017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Min J and Wei C: Hydroxysafflor yellow A
cardioprotection in ischemia-reperfusion (I/R) injury mainly via
Akt/hexokinase II independent of ERK/GSK-3β pathway. Biomed
Pharmacother. 87:419–426. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang T, Duan SJ, Wang SY, Lu Y, Zhu Q,
Wang LJ and Han B: Coadministration of hydroxysafflor yellow A with
levodopa attenuates the dyskinesia. Physiol Behav. 147:193–197.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Sun L, Yang L, Xu YW, Liang H, Han J, Zhao
RJ and Cheng Y: Neuroprotection of hydroxysafflor yellow A in the
transient focal ischemia: inhibition of protein
oxidation/nitration, 12/15-lipoxygenase and blood-brain barrier
disruption. Brain Res. 1473:227–235. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wu XL: Efficacy enhancing effect of
Shikonin on human liver cancer HepG2 cells treated by Cisplatin.
Chin Med J Res Prac. 31:35–38. 2017.(In Chinese).
|
|
35
|
Wu Y and Liu R: Study on the
drug-resistant reversal effects of ginsenoside Rh2 in human
hepatocellular carcinoma HepG2/ADM cells and its mechanism. J Med
Postgra. 30:476–480. 2017.
|
|
36
|
Park HR, Ju EJ, Jo SK, Jung U, Kim SH and
Yee ST: Enhanced antitumor efficancy of cisplatin in combination
with HemoHIM in tumor-bearing mice. BMC Cancer. 9:852009.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Liu Y, Jing Y, Guo LL, et al: Effects of
Guben Zhike Recipe on the spleen index, thymus index and Th1/Th2
unbalance in COPD mice. China J Tradit Chin Med Pharm. 2015.(In
Chinese).
|
|
38
|
Fontenot JD, Rasmussen JP, Williams LM,
Dooley JL, Farr AG and Rudensky AY: Regulatory T cell lineage
specification by the forkhead transcription factor foxp3. Immunity.
22:329–341. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Fu J, Xu D, Liu Z, Shi M, Zhao P, Fu B,
Zhang Z, Yang H, Zhang H, Zhou C, et al: Increased regulatory T
cells correlate with CD8 T-cell impairment and poor survival in
hepatocellular carcinoma patients. Gastroenterology. 132:2328–2339.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lin SZ, Chen KJ, Xu ZY, Chen H, Zhou L,
Xie HY and Zheng SS: Prediction of recurrence and survival in
hepatocellular carcinoma based on two Cox models mainly determined
by FoxP3+ regulatory T cells. Cancer Prev Res (Phila).
6:594–602. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Takeuchi Y and Nishikawa H: Roles of
regulatory T cells in cancer immunity. Int Immunol. 28:401–409.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zhang JP, Yan J, Xu J, Pang XH, Chen MS,
Li L, Wu C, Li SP and Zheng L: Increased intratumoral
IL17-producing cells correlate with poor survival in hepatocellular
carcinoma patients. J Hepatol. 50:980–989. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Huang Y, Wang F, Wang Y, Zhu Z, Gao Y, Ma
Z, Xu R and Du Z: Intrahepatic interleukin-17+ T cells
and FoxP3+ regulatory T cells cooperate to promote
development and affect the prognosis of hepatocellular carcinoma. J
Gastroenterol Hepatol. 29:851–859. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Lin ZW, Wu LX, Xie Y, Ou X, Tian PK, Liu
XP, Min J, Wang J, Chen RF, Chen YJ, et al: The expression levels
of transcription factors T-bet, GATA-3, RORγt and FOXP3 in
peripheral blood lymphocyte (PBL) of patients with liver cancer and
their significance. Int J Med Sci. 12:7–16. 2015. View Article : Google Scholar : PubMed/NCBI
|