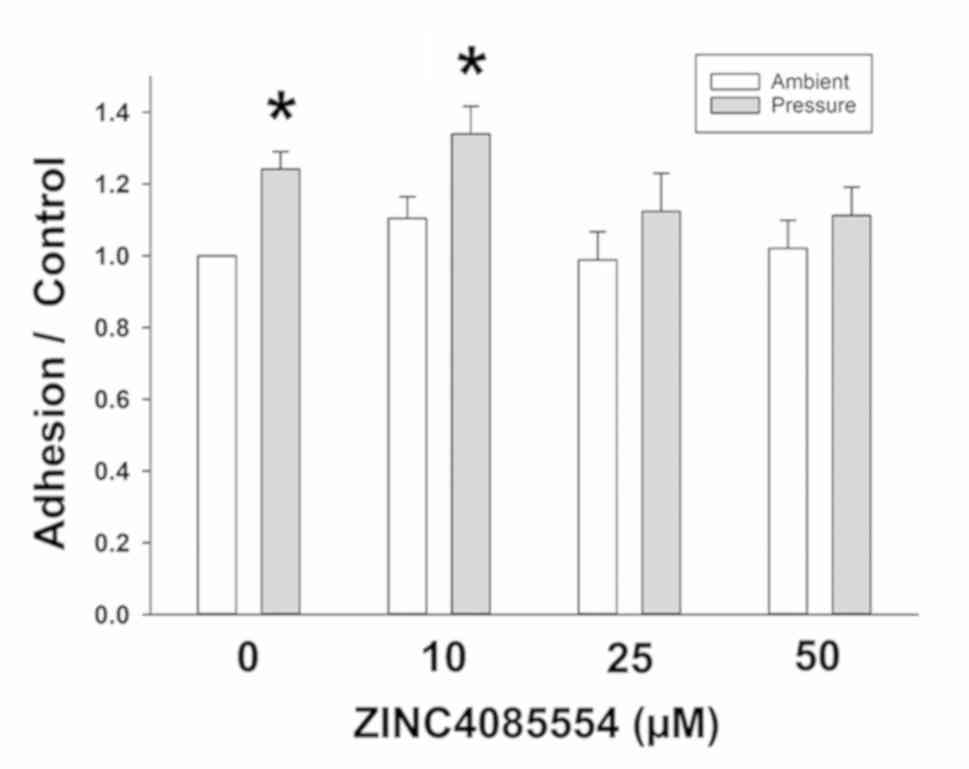
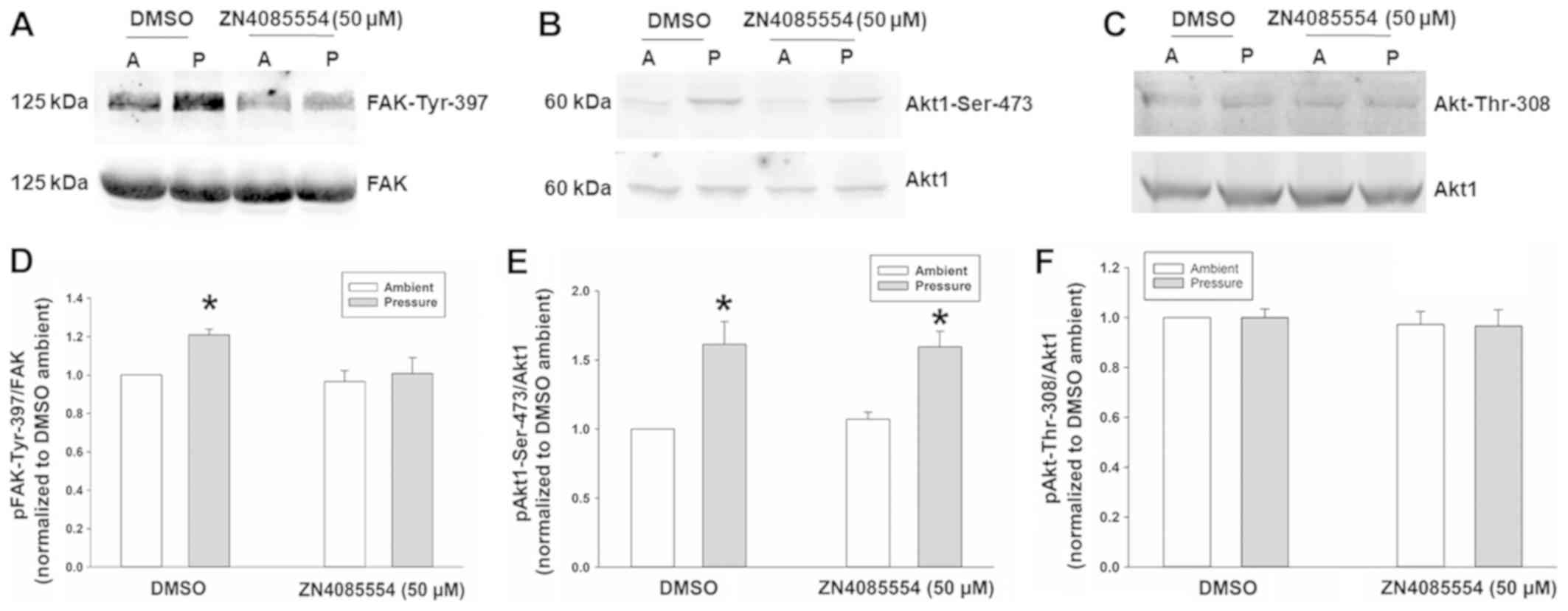
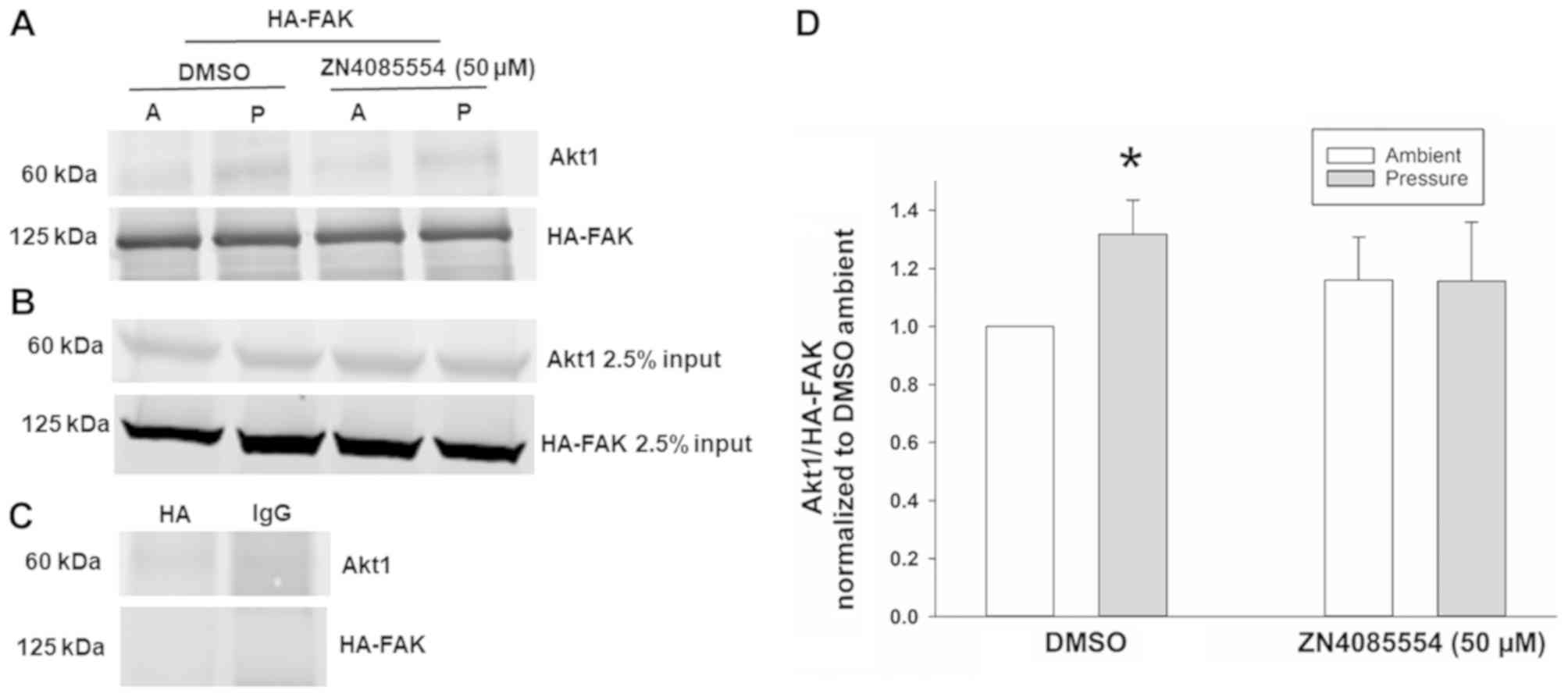
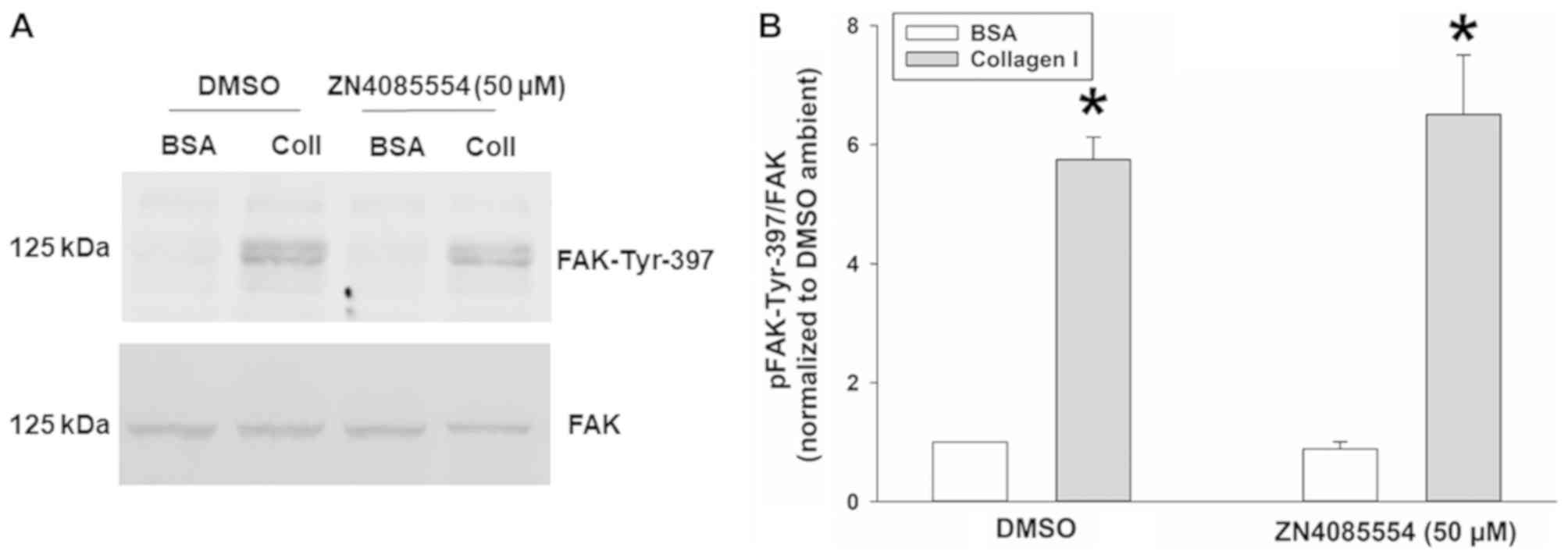
|
1
|
Lambert AW, Pattabiraman DR and Weinberg
RA: Emerging Biological Principles of Metastasis. Cell.
168:670–691. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Craig DH and Basson MD: Biological impact
of mechanical stimuli on tumor metastasis. Cell Cycle. 8:828–831.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Brooks SA, Lomax-Browne HJ, Carter TM,
Kinch CE and Hall DM: Molecular interactions in cancer cell
metastasis. Acta Histochem. 112:3–25. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Craig DH, Owen CR, Conway WC, Walsh MF,
Downey C and Basson MD: Colchicine inhibits pressure-induced tumor
cell implantation within surgical wounds and enhances tumor-free
survival in mice. J Clin Invest. 118:3170–3180. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lafrenie RM, Buchanan MR and Orr FW:
Adhesion molecules and their role in cancer metastasis. Cell
Biophys. 23:3–89. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kavic SM and Basson MD: Environmental
factors of temperature, humidity, serum accumulation, and cell
seeding increase colon cancer cell adhesion in vitro, with partial
characterization of the serum component responsible for
pressure-stimulated adhesion. J Surg Res. 98:89–96. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wirtz D, Konstantopoulos K and Searson PC:
The physics of cancer: The role of physical interactions and
mechanical forces in metastasis. Nat Rev Cancer. 11:512–522. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Basson MD: An intracellular signal pathway
that regulates cancer cell adhesion in response to extracellular
forces. Cancer Res. 68:2–4. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Downey C, Alwan K, Thamilselvan V, Zhang
L, Jiang Y, Rishi AK and Basson MD: Pressure stimulates breast
cancer cell adhesion independently of cell cycle and apoptosis
regulatory protein (CARP)-1 regulation of focal adhesion kinase. Am
J Surg. 192:631–635. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Conway WC, Van der Voort van Zyp J,
Thamilselvan V, Walsh MF, Crowe DL and Basson MD: Paxillin
modulates squamous cancer cell adhesion and is important in
pressure-augmented adhesion. J Cell Biochem. 98:1507–1516. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Perry BC, Wang S and Basson MD:
Extracellular pressure stimulates adhesion of sarcoma cells via
activation of focal adhesion kinase and Akt. Am J Surg.
200:610–614. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Basson MD, Yu CF, Herden-Kirchoff O,
Ellermeier M, Sanders MA, Merrell RC and Sumpio BE: Effects of
increased ambient pressure on colon cancer cell adhesion. J Cell
Biochem. 78:47–61. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Daskalakis M, Scheffel O and Weiner RA:
High flow insufflation for the maintenance of the pneumoperitoneum
during bariatric surgery. Obes Facts. 2 (Suppl 1):37–40. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Thamilselvan V and Basson MD: The role of
the cytoskeleton in differentially regulating pressure-mediated
effects on malignant colonocyte focal adhesion signaling and cell
adhesion. Carcinogenesis. 26:1687–1697. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Parsons JT: Focal adhesion kinase: The
first ten years. J Cell Sci. 116:1409–1416. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang S and Basson MD: Integrin-linked
kinase: Α multi-functional regulator modulating extracellular
pressure-stimulated cancer cell adhesion through focal adhesion
kinase and AKT. Cell Oncol. 31:273–289. 2009.PubMed/NCBI
|
|
17
|
Wang S and Basson MD: Akt directly
regulates focal adhesion kinase through association and serine
phosphorylation: Implication for pressure-induced colon cancer
metastasis. Am J Physiol Cell Physiol. 300:C657–C670. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Golubovskaya VM: Targeting FAK in human
cancer: From finding to first clinical trials. Front Biosci.
19:687–706. 2014. View
Article : Google Scholar
|
|
19
|
Lv PC, Jiang AQ, Zhang WM and Zhu HL: FAK
inhibitors in Cancer, a patent review. Expert Opin Ther Pat.
28:139–145. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Schultze A and Fiedler W: Therapeutic
potential and limitations of new FAK inhibitors in the treatment of
cancer. Expert Opin Investig Drugs. 19:777–788. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sulzmaier FJ, Jean C and Schlaepfer DD:
FAK in cancer: Mechanistic findings and clinical applications. Nat
Rev Cancer. 14:598–610. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Basson MD, Zeng B and Wang S: Akt1 binds
focal adhesion kinase via the Akt1 kinase domain independently of
the pleckstrin homology domain. J Physiol Pharmacol. 66:701–709.
2015.PubMed/NCBI
|
|
23
|
Basson MD, Zeng B and Wang S: The
C-terminal region of the focal adhesion kinase F1 domain binds Akt1
and inhibits pressure-induced cell adhesion. J Physiol Pharmacol.
68:375–383. 2017.PubMed/NCBI
|
|
24
|
Zeng B, Devadoss D, Wang S, Vomhof-DeKrey
EE, Kuhn LA and Basson MD: Inhibition of pressure-activated cancer
cell adhesion by FAK-derived peptides. Oncotarget. 8:98051–98067.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Marqus S, Pirogova E and Piva TJ:
Evaluation of the use of therapeutic peptides for cancer treatment.
J Biomed Sci. 24:212017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Otvos L Jr and Wade JD: Current challenges
in peptide-based drug discovery. Front Chem. 2:622014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Raschka S, More SK, Devadoss D, Zeng B,
Kuhn LA and Basson MD: Identification of potential small-molecule
protein-protein inhibitors of cancer metastasis by 3D epitope-based
computational screening. J Physiol Pharmacol. 69:doi:
10.26402/jpp.2018.2.11. PubMed/NCBI
|
|
28
|
Thamilselvan V, Craig DH and Basson MD:
FAK association with multiple signal proteins mediates
pressure-induced colon cancer cell adhesion via a Src-dependent
PI3K/Akt pathway. FASEB J. 21:1730–1741. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Allain F, Vanpouille C, Carpentier M,
Slomianny MC, Durieux S and Spik G: Interaction with
glycosaminoglycans is required for cyclophilin B to trigger
integrin-mediated adhesion of peripheral blood T lymphocytes to
extracellular matrix. Proc Natl Acad Sci USA. 99:2714–2719. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Canavan HE, Cheng X, Graham DJ, Ratner BD
and Castner DG: Cell sheet detachment affects the extracellular
matrix: A surface science study comparing thermal liftoff,
enzymatic, and mechanical methods. J Biomed Mater Res A. 75:1–13.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Craig DH, Downey C and Basson MD:
SiRNA-mediated reduction of alpha-actinin-1 inhibits
pressure-induced murine tumor cell wound implantation and enhances
tumor-free survival. Neoplasia. 10:217–222. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Claycombe KJ, Vomhof-DeKrey EE, Garcia R,
Johnson WT, Uthus E and Roemmich JN: Decreased beige adipocyte
number and mitochondrial respiration coincide with increased
histone methyl transferase (G9a) and reduced FGF21 gene expression
in Sprague-Dawley rats fed prenatal low protein and postnatal
high-fat diets. J Nutr Biochem. 31:113–121. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Vomhof-DeKrey E, Darland D, Ghribi O,
Bundy A, Roemmich J and Claycombe K: Maternal low protein diet
leads to placental angiogenic compensation via dysregulated M1/M2
macrophages and TNFα expression in Sprague-Dawley rats. J Reprod
Immunol. 118:9–17. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Velling T, Nilsson S, Stefansson A and
Johansson S: beta1-Integrins induce phosphorylation of Akt on
serine 473 independently of focal adhesion kinase and Src family
kinases. EMBO Rep. 5:901–905. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Huveneers S and Danen EH: Adhesion
signaling - crosstalk between integrins, Src and Rho. J Cell Sci.
122:1059–1069. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Hynes RO: Integrins: Bidirectional,
allosteric signaling machines. Cell. 110:673–687. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Turecková J, Vojtechová M, Krausová M,
Sloncová E and Korínek V: Focal adhesion kinase functions as an akt
downstream target in migration of colorectal cancer cells. Transl
Oncol. 2:281–290. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Xia H, Nho RS, Kahm J, Kleidon J and Henke
CA: Focal adhesion kinase is upstream of phosphatidylinositol
3-kinase/Akt in regulating fibroblast survival in response to
contraction of type I collagen matrices via a beta 1 integrin
viability signaling pathway. J Biol Chem. 279:33024–33034. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zhao J and Guan JL: Signal transduction by
focal adhesion kinase in cancer. Cancer Metastasis Rev. 28:35–49.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Gayer CP and Basson MD: The effects of
mechanical forces on intestinal physiology and pathology. Cell
Signal. 21:1237–1244. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Vleming LJ, Baelde JJ, Westendorp RG, Daha
MR, van Es LA and Bruijn JA: Progression of chronic renal disease
in humans is associated with the deposition of basement membrane
components and decorin in the interstitial extracellular matrix.
Clin Nephrol. 44:211–219. 1995.PubMed/NCBI
|
|
42
|
Tomar A and Schlaepfer DD: A PAK-activated
linker for EGFR and FAK. Dev Cell. 18:170–172. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Nagano M, Hoshino D, Koshikawa N, Akizawa
T and Seiki M: Turnover of focal adhesions and cancer cell
migration. Int J Cell Biol. 2012:3106162012. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Schlaepfer DD, Hauck CR and Sieg DJ:
Signaling through focal adhesion kinase. Prog Biophys Mol Biol.
71:435–478. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Higuchi M, Kihara R, Okazaki T, Aoki I,
Suetsugu S and Gotoh Y: Akt1 promotes focal adhesion disassembly
and cell motility through phosphorylation of FAK in growth
factor-stimulated cells. J Cell Sci. 126:745–755. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Zhao X and Guan JL: Focal adhesion kinase
and its signaling pathways in cell migration and angiogenesis. Adv
Drug Deliv Rev. 63:610–615. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Manning BD and Toker A: AKT/PKB Signaling:
Navigating the Network. Cell. 169:381–405. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
O'toole A, Moule SK, Lockyer PJ and
Halestrap AP: Tumour necrosis factor-alpha activation of protein
kinase B in WEHI-164 cells is accompanied by increased
phosphorylation of Ser473, but not Thr308. Biochem J. 359:119–127.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Kitagawa K, Takasawa K, Kuwabara K, Sasaki
T, Tanaka S, Mabuchi T, Sugiura S, Omura-Matsuoka E, Matsumoto M
and Hori M: Differential Akt phosphorylation at Ser473 and Thr308
in cultured neurons after exposure to glutamate in rats. Neurosci
Lett. 333:187–190. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Salazar EP, Hunger-Glaser I and Rozengurt
E: Dissociation of focal adhesion kinase and paxillin tyrosine
phosphorylation induced by bombesin and lysophosphatidic acid from
epidermal growth factor receptor transactivation in Swiss 3T3
cells. J Cell Physiol. 194:314–324. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Mitra SK, Hanson DA and Schlaepfer DD:
Focal adhesion kinase: In command and control of cell motility. Nat
Rev Mol Cell Biol. 6:56–68. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Mitra SK and Schlaepfer DD:
Integrin-regulated FAK-Src signaling in normal and cancer cells.
Curr Opin Cell Biol. 18:516–523. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Shiratsuchi H and Basson MD: Akt2, but not
Akt1 or Akt3 mediates pressure-stimulated serum-opsonized latex
bead phagocytosis through activating mTOR and p70 S6 kinase. J Cell
Biochem. 102:353–367. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Thamilselvan V and Basson MD: Pressure
activates colon cancer cell adhesion by inside-out focal adhesion
complex and actin cytoskeletal signaling. Gastroenterology.
126:8–18. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
van Zyp J, Conway WC, Craig DH, van Zyp N,
Thamilselvan V and Basson MD: Extracellular pressure stimulates
tumor cell adhesion in vitro by paxillin activation. Cancer Biol
Ther. 5:1169–1178. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Goreczny GJ, Ouderkirk-Pecone JL, Olson
EC, Krendel M and Turner CE: Hic-5 remodeling of the stromal matrix
promotes breast tumor progression. Oncogene. 36:2693–2703. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Lee BY, Hochgräfe F, Lin HM, Castillo L,
Wu J, Raftery MJ, Martin Shreeve S, Horvath LG and Daly RJ:
Phosphoproteomic profiling identifies focal adhesion kinase as a
mediator of docetaxel resistance in castrate-resistant prostate
cancer. Mol Cancer Ther. 13:190–201. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Srinivas V, Datta SA, Ramakrishna T and
Rao CM: Studies on the alpha-crystallin target protein binding
sites: Sequential binding with two target proteins. Mol Vis.
7:114–119. 2001.PubMed/NCBI
|
|
59
|
Wang H, Zhu Y, Zhao M, Wu C, Zhang P, Tang
L, Zhang H, Chen X, Yang Y and Liu G: miRNA-29c suppresses lung
cancer cell adhesion to extracellular matrix and metastasis by
targeting integrin β1 and matrix metalloproteinase2 (MMP2). PLoS
One. 8:e701922013. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Nelson H, Sargent DJ, Wieand HS, Fleshman
J, Anvari M, Stryker SJ, Beart RW Jr, Hellinger M, Flanagan R Jr,
Peters W, et al Clinical Outcomes of Surgical Therapy Study Group,
: A comparison of laparoscopically assisted and open colectomy for
colon cancer. N Engl J Med. 350:2050–2059. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Fujita S, Kudo N, Akasu T and Moriya Y:
Detection of cytokeratin 19 and 20 mRNA in peripheral and
mesenteric blood from colorectal cancer patients and their
prognosis. Int J Colorectal Dis. 16:141–146. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Guller U, Zajac P, Schnider A, Bösch B,
Vorburger S, Zuber M, Spagnoli GC, Oertli D, Maurer R, Metzger U,
et al: Disseminated single tumor cells as detected by real-time
quantitative polymerase chain reaction represent a prognostic
factor in patients undergoing surgery for colorectal cancer. Ann
Surg. 236:768–775; discussion 775–776. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Ishida H, Hashimoto D, Takeuchi I,
Yokoyama M, Okita T and Hoshino T: Liver metastases are less
established after gasless laparoscopy than after carbon dioxide
pneumoperitoneum and laparotomy in a mouse model. Surg Endosc.
16:193–196. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Lee SW, Whelan RL, Southall JC and Bessler
M: Abdominal wound tumor recurrence after open and
laparoscopic-assisted splenectomy in a murine model. Dis Colon
Rectum. 41:824–831. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Melamed A, Margul DJ, Chen L, Keating NL,
Del Carmen MG, Yang J, Seagle BL, Alexander A, Barber EL, Rice LW,
et al: Survival after Minimally Invasive Radical Hysterectomy for
Early-Stage Cervical Cancer. N Engl J Med. 379:1905–1914. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Ramirez PT, Frumovitz M, Pareja R, Lopez
A, Vieira M, Ribeiro R, Buda A, Yan X, Shuzhong Y, Chetty N, et al:
Minimally Invasive versus Abdominal Radical Hysterectomy for
Cervical Cancer. N Engl J Med. 379:1895–1904. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Umpleby HC, Fermor B, Symes MO and
Williamson RC: Viability of exfoliated colorectal carcinoma cells.
Br J Surg. 71:659–663. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Thamilselvan V, Patel A, van der Voort van
Zyp J and Basson MD: Colon cancer cell adhesion in response to Src
kinase activation and actin-cytoskeleton by non-laminar shear
stress. J Cell Biochem. 92:361–371. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Sugarbaker PH: Successful management of
microscopic residual disease in large bowel cancer. Cancer
Chemother Pharmacol. 43 (Suppl):S15–S25. 1999. View Article : Google Scholar : PubMed/NCBI
|