Introduction
The pancreas is the tenth most common site of new
cancers, but pancreatic cancer is the fourth leading cause of
cancer deaths among men and women, with an overall 5-year survival
rate of 5% (1). One of the major
causes of death is peritoneal dissemination and liver metastasis
(2). The treatment of advanced
pancreatic cancer with gemcitabine has only modest activity with a
small survival benefit, and toxicity continues to be a major
obstacle (3). New therapeutic
strategies that notably lack cross resistance with established
treatment regimens are much needed in pancreatic cancer. Since
pancreatic carcinoma show strong tumor angiogenesis, overexpression
of VEGF, the inhibition of tumor angiogenesis has been one of the
promising strategies in the treatment of pancreatic carcinoma.
Luteolin, 3′,4′,5,7-tetrahydroxyflavone, is a common
flavonoid that exists in many types of plants including fruits,
vegetables and medicinal herbs. Plants rich in luteolin have been
used in Chinese traditional medicine for treating various diseases
such as hypertension, inflammatory disorders and cancer. The
luteolin anticancer property is associated with the induction of
apoptosis, and inhibition of cell proliferation, metastasis and
angiogenesis (4). However, whether
or not luteolin can inhibit proliferation of pancreatic carcinoma
cells was unclear. In this study, we assessed the antitumor and
anti-angiogenic activity of luteolin on pancreatic carcinoma cells,
and also investigated its effect on VEGF signal transduction.
Materials and methods
Materials
Luteolin was purchased from Nanjing TCM Institute of
Chinese Materia Medica, China. Luteolin was dissolved in dimethyl
sulfoxide (DMSO) and was used in all experiments. Trypsin, MTT
[3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide],
propidium iodide (PI) and DNase-free RNase were obtained from
Sigma, USA. Lysis buffer was purchased from Beyotime, China.
Antibodies (caspase-3, caspase-8, goat anti-mouse IgG-HRP and goat
anti-rabbit IgG-HRP) were obtained from Santa Cruz Biotechnology,
CA, USA. Mouse anti-Bax was obtained from BD Biosciences, Bedford,
MA, USA. Bcl-2, caspase-9, PARP (poly-ADP-ribose polymerase),
ERK1/2, p-ERK1/2, p-p38, p38, JNK and p-JNK Antibody were purchased
from Cell Signaling Technology, MA, USA. Monoclonal mouse
anti-glyceraldehyde-3-phosphate dehydrogease (GAPDH) was obtained
from KangChen, China. Endothelial cell growth supplement (ECGS) was
purchased from Millipore, MA, USA. Peripheral blood was purchased
from the Blood Center of Jiangsu Province, China. Ficoll-Hypaque
was obtained from Meijing, China.
Cell culture
Human pancreatic carcinoma cell lines PANC-1,
CoLo-357, BxPC-3 were purchased from CellBank of Shanghai Institute
of Biochemistry and Cell Biology. Cells were cultured in
DMEM-medium (for PANC-1, CoLo-357 cells) or RPMI-1640 (for BxPC-3
cells) supplemented with 10% fetal bovine serum (FBS), 100 U/ml
penicillin and 100 μg/ml streptomycin (all available from
Invitrogen, Grand Island, NY, USA). Human umbilical vein
endothelial cells (HUVEC) were isolated from human umbilical cord
veins by trypsin digestion method, improved from Jaffe et al
(5). Fresh human umbilical cord was
washed clear by phosphate-buffered saline (PBS), the vein was then
infused with 0.1% trypsin and digested for 15 min at 37°C. The
cells digested from the umbilical vein was maintained in M199
medium supplemented with 15% FBS, 0.1 mg/ml heparin, 0.03 mg/ml
endothelial cell growth supplement (ECGS). HUVEC were characterized
by immunofluorescence method with antibodies to Factor VIII related
antigen. Human peripheral blood mononuclear cells (PBMC) were
isolated from fresh human blood by Ficoll-Hypaque density gradient
centrifugation. The isolated cells were cultured in RPMI-1640
medium containing 10% FBS. All cultures were maintained in a
humidified environment with 5% CO2 at 37°C.
Endothelial cell proliferation assay
HUVEC were seeded in 96-well plates,
1×104 cells per well. The medium was replaced by
low-serum medium (1% fetal bovine serum in M199) containing
different concentrations of luteolin. DMSO (0.1%) was used as
control. After 24 h, the viability of HUVEC were analyzed by MTT
assay as described previously (6).
The percent inhibition of the treated cells was calculated by the
formula: % inhibition = 1 - (A570
nm–A630 nm) treated/(A570
nm–A630 nm) control × 100%. The
IC50 were further assayed.
Cytotoxicity assay
The cytotoxicity of luteolin, which had potent HUVEC
inhibition ability, was analyzed by MTT assay with PBMC and various
differentiated human pancreatic carcinoma cell lines, the poorly
differentiated cell line (PANC-1), the moderately differentiated
cell line (CoLo-357) and the well differentiated cell line
(BxPC-3).
Cell morphological assessment
Cells were cultured until mid-log phase. DMSO 0.1%
(control), 20, 40, 60 or 80 μmol/l luteolin was then added to the
culture medium. The morphology of cells was monitored under an
inverted microscope (Zeiss Axio Observer A1) at 6, 12, 18, 24 and
48 h.
Cell cycle analysis
Cells treated with 0.1% DMSO or increasing
concentrations of luteolin for different time periods were
trypsinized and washed twice with PBS, and fixed in 100% ethanol
overnight at 4°C. Fixed cells were washed with PBS before
incubation with 1 ml PBS containing 50 μg/ml PI and 1 mg/ml RNase
for 30 min at 37°C. DNA content and cell cycle were determined
using a FACScan laser flow cytometer (FACSCalibur,
Becton-Dickinson, NY, USA). The data were analyzed using the
software CellQuest.
Hoechst 33258 staining
Hoechst 33258 staining was used to visualize nuclear
change and apoptotic body formation. At the end of luteolin
treatment, attached cells were washed twice with PBS and fixed with
4% methanol at 4°C for 30 min. The fixing solution was removed and
cells were washed twice with PBS before staining with Hoechst 33258
(KeyGen, Nanjing, China). After staining for 10 min, cells were
washed again and observed under a fluorescence microscope (Zeiss
Axio Observer A1) at 340 nm.
Chicken chorioallantoic membrane (CAM)
assay
Fertilized chicken eggs were incubated at around 55%
relative humidity, 37.5°C incubator for 7 days. Eggs were set with
the blunt end up at 45°C to prevent microbial growth and turned
regularly (at least two times per day). At experiment day, eggs
were checked with an egg candler, only live, fertilized ones were
random divided, ten for each group. A 1.5×1.5 cm ‘window’ was
created on the blunt end of the egg, where the air sac was located.
The air sac membrane was punctured carefully using an injection
needle avoid breaking the blood vessel. About 50 μl sterilized
water was injected between the air sac membrane and chorioallantoic
membrane, and then the air sac membrane could be easily peeled off.
Sterilized filter paper (5×5 mm) saturated with 0.1% DMSO (control)
or luteolin (5 and 10 nmol/egg) was air dried and then placed on
the CAMs. The eggs were then covered with parafilm and put back to
the incubator. Three days later, 20% fat emulsion (Chia-tai
Tianqing Pharmaceutical Co., Jiangsu, China) was injected into the
chorioallantois and the blood vessels were photographed by Sony
α100 (Sony, Japan). Blood vessel density was calculated by
Image-pro plus 6.0 software.
Tube formation assay
Under sterile conditions, 24-well plates were coated
with 200 μl/well of growth factor-reduced Matrigel (BD Biosciences)
without introducing air bubbles. The plates were set at 37°C for 30
min to allow gelling of Matrigel. The PANC-1 cells and HUVEC were
cultured in serum-free medium for 12 h before the experiments. The
HUVEC were then cultured on Matrigel, whereas the PANC-1 cells were
pre-treated with 0.1% DMSO or 40 μmol/l luteolin for 30 min before
plating within the transwell inserts to prevent physical contact.
Both cells were cultured in serum-free medium. The cells were
co-cultured for 12 h, HUVEC differentiated and formed
capillary-like structures on Matrigel. The enclosed networks of
complete tubes from five randomly chosen fields were counted and
photographed under a microscope (x100).
VEGF detected by ELISA
The concentration of VEGF in the conditioned medium
from human pancreatic carcinoma cells was measured using a
commercially available ELISA kit (R&D Systems, Minneapolis, MN,
USA). The cells (3×105/well) were incubated overnight in
6-well dishes in medium containing 10% FBS. The media were then
replaced with serum-free media containing various doses of luteolin
for 24 h. VEGF was expressed as picogram of VEGF protein/ml medium
and per 105 cells.
RT-PCR analysis
The expression of VEGF from the cell samples was
studied by RT-PCR. Total cellular RNA was extracted from
luteolin-treated PANC-1 cells using TRIzol reagent (Invitrogen)
according to the manufacturer’s instructions. Complementary DNA
(cDNA) was generated with M-MLV reverse transcriptase (Takara, NY,
USA) according to the manufacturer’s instructions. The cDNA samples
were then subjected to PCR analysis using the following primers:
VEGF sense, 5′-ATGGCACCCATGGCAGAAG-3′; VEGF antisense,
5′-TCACCGCCTCGGCTTGTCAC-3′; GAPDH sense, 5′-ATGGGGAAGGTGAAGGTCG-3′;
GAPDH antisense, 5′-TTACTCCTTGGAGGCCATGTG-3′. PCR reaction
conditions were as follows: initial denaturation at 94°C for 3 min
and 30 cycles of amplification [94°C for 45 sec, 57°C for 45 sec
and 72°C for 30 sec (GAPDH for 60 sec)], followed by a final
extension step for 7 min at 72°C. The amplified PCR products were
separated by electrophoresis on a 2% agarose gel containing
ethidium bromide and quantitated by relative intensities of the
bands as compared to those of GAPDH using Gel Base/Gel Blot/Gel
Excel/Gel Sequence analysis software (UVP, UK). A value of 100% was
given to the relative intensity of untreated cells (control).
NF-κB activation detected by EMSA
To determine NF-κB activation by luteolin, we
examined the NF-κB-DNA binding by electrophoretic mobility shift
assay (EMSA). Briefly, nuclear extracts prepared from treated cells
with Nuclear and Cytoplasmic Extraction Reagents (Pierce, IL, USA)
were incubated with double-stranded NF-κB oligonucleotide
(5′-AGTTGAGGGGACTTTCCCAGG-3′) (Beyotime). EMSA followed the
instructions of LightShift chemiluminescent EMSA kit (Pierce).
Western blot analysis
Cells were cultured until mid-log phase and then
incubated with different luteolin for 24 h. Proteins were isolated
by lysis buffer (Beyotime) and measured using the Nanodrop 1000
Spectrophotometer (Thermo, IL, USA). Protein samples were separated
on 13% SDS-polyacrylamide gels (SDS-PAGE) and transferred onto the
PVDF membranes (Millipore). After blocked with 1% BSA in TBST
(Tris-buffered saline with Tween-20) for 2 h, membranes were
incubated with primary antibodies overnight at 4°C. Blots were
washed and incubated with secondary antibodies for 1 h at room
temperature. Membranes were again washed three times with TBST and
developed using enhanced chemiluminescence (Luminata Crescendo
Western HRP substrate, Millipore). Membranes were then exposed to
film.
Statistical analysis
Values were expressed as means ± SD from three
independent experiments. Statistical analysis was performed by
one-way analysis of variance. When significance was detected, the
t-test for multiple comparisons was performed on the data from
experimental groups. A probability value of P<0.01 was
considered statistically significant.
Results
Decreased cell viability and cell cycle
arrest in luteolin-treated pancreatic carcinoma cells
Exponentially growing pancreatic carcinoma cells
(PANC-1, CoLo-357, BxPC-3) were cultured continuously in the
absence or presence of different concentrations of luteolin. The
effects of luteolin on cell growth were assessed by the commonly
used MTT assay at different intervals (12, 24, 48 and 72 h) of
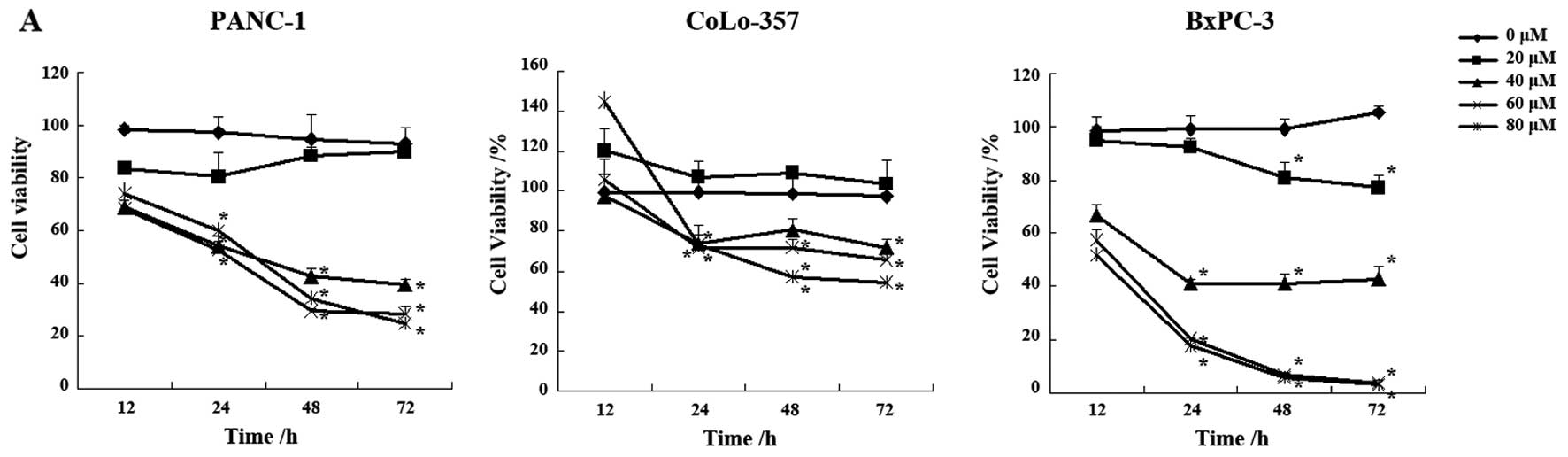
treatment. Luteolin treatment significantly inhibited the growth of
pancreatic carcinoma cells (Fig.
1A). The degree of growth inhibition depended on both the
concentration and the treatment time. At a given duration of
treatment, the number of viable cells decreased as the
concentration of luteolin increased. On the other hand, when
luteolin concentration was held constant, the number of viable
cells decreased regularly as the exposure time increased. The
effect of luteolin treatment was statistically significant when
compared with the control group (P<0.01).
To test whether luteolin could affect the cell cycle
of pancreatic carcinoma cells, cells treated with DMSO or different
concentration luteolin for 24 h were subjected to flow cytometric
analysis after DNA staining. As shown in Fig. 1B and C, exposure of PANC-1 and
CoLo-357 cells to growth suppressive concentrations of luteolin
resulted in a statistically significant increase in the G2 phase
that was accompanied by a decrease in the G1 phase. However,
luteolin had no effect on BxPC-3 cell cycle.
Luteolin-induced apoptosis in pancreatic
carcinoma cells through caspase pathway
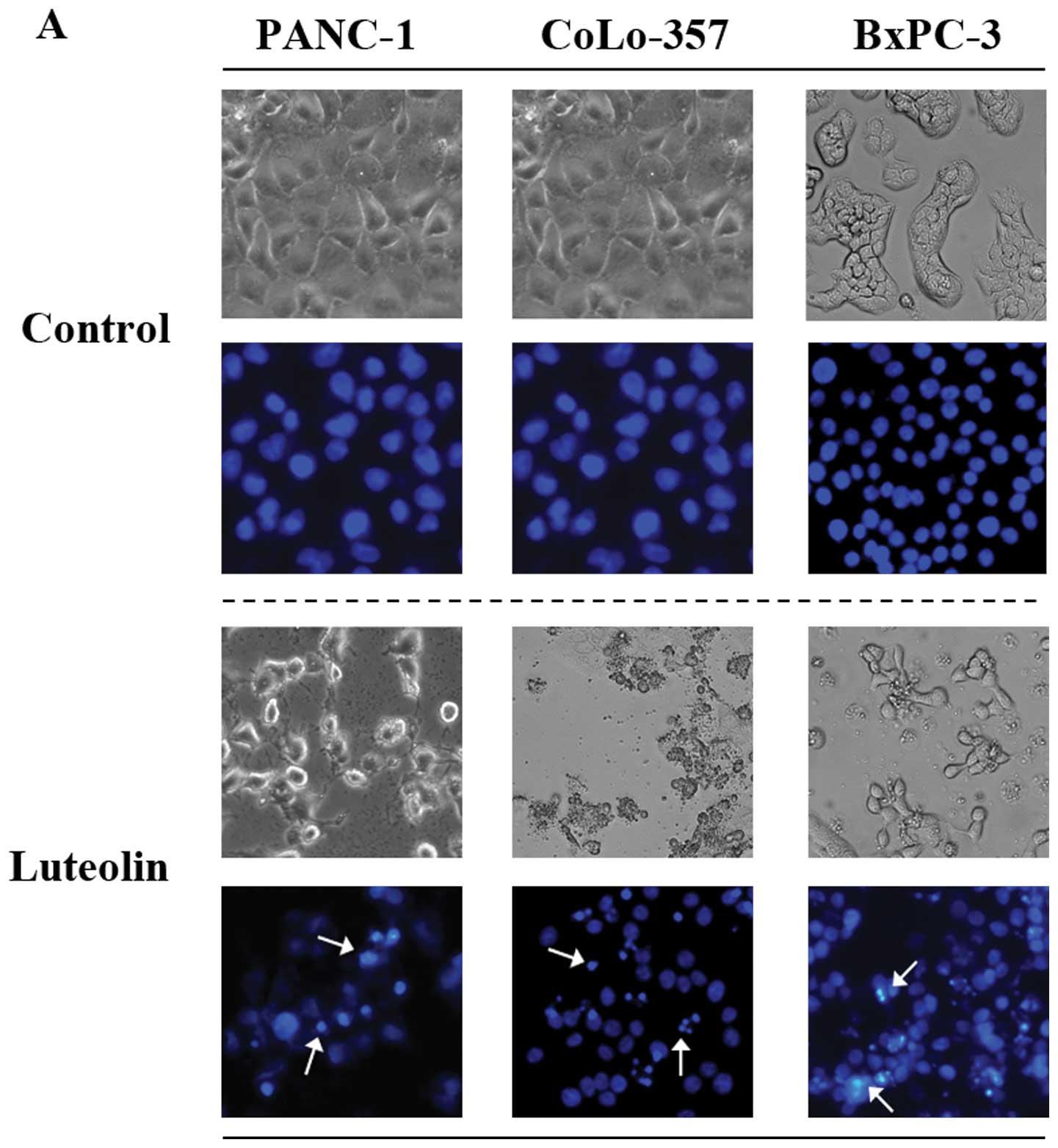
Differences in cell morphology were observed between
luteolin-treated and control cells by light microscopy. The most
conspicuous change observed in luteolin-treated cells included cell
shrinkage and extensive detachment of the cells from the cell
culture substratum (Fig. 2A). These
changes, which were characteristic of cell apoptotic death, became
visible after luteolin treatment, but were absent in control cells.
The morphological change became more remarkable with increased time
of drug treatment (data not shown). The occurrence of apoptosis was
further verified by Hoechst staining, which detects chromatin
condensation, one of the hallmarks of apoptotic cell death.
Differences were observed in the nuclei of luteolin-treated and
untreated pancreatic carcinoma cells after staining with Hoechst
33258 (Fig. 2A). The Hoechst 33258
dye stained morphologically normal nuclei dimly blue, whereas
luteolin-treated cells demonstrated smaller nuclei with brilliant
blue staining. The change in nuclear morphology was initially
observed after 24 h of luteolin treatment and increased thereafter
(data not shown). These results demonstrated that luteolin induces
morphological changes characteristic of cell apoptotic death.
The molecular mechanism for the potent pro-apoptotic
effect of luteolin on pancreatic carcinoma cells was further
studied. Western blot analysis was done as described in Materials
and methods. As shown in Fig. 2B,
luteolin increased the expression of pro-apoptotic protein (Bax)
and decreased anti-apoptotic protein (Bcl-2), with a concomitant
increase in the levels of caspase-3 and cleaved PARP in pancreatic
carcinoma cells after treatment for 24 h.
Differential growth inhibition of
luteolin on HUVEC and PBMC
In order to rule out the effects of physico-chemical
property of luteolin, such as pH, but not the pharmacologic actions
on the cells, human normal cells PMBC were used as control. The
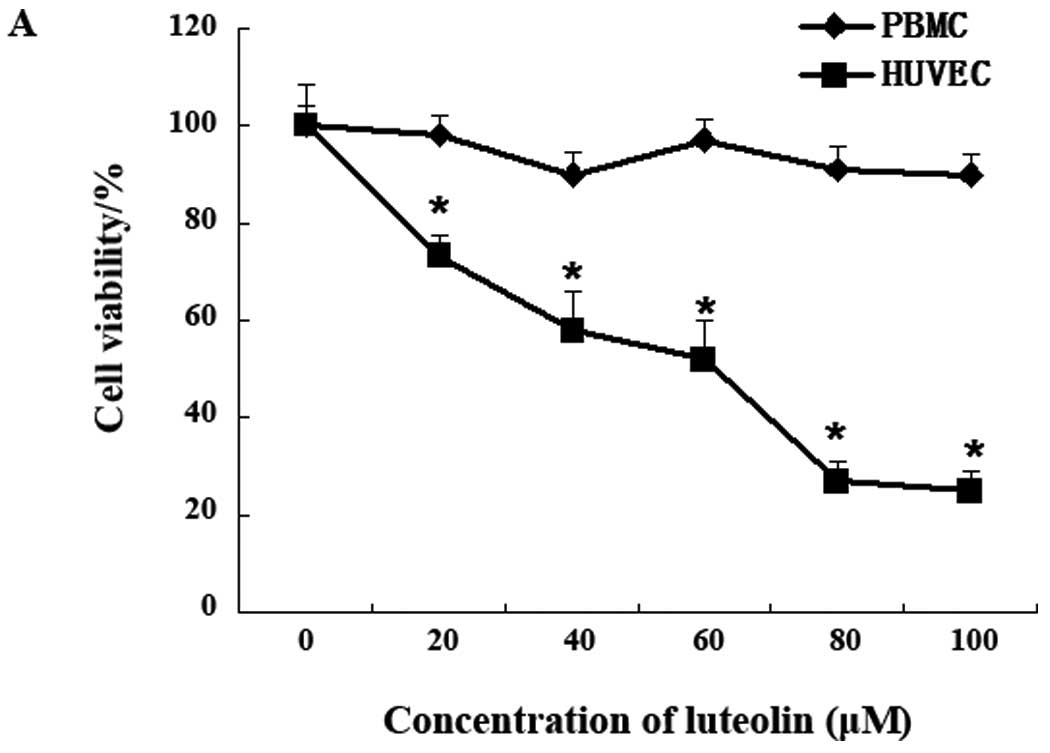
inhibition effect of luteolin on cell growth was assessed by the
commonly used MTT assay. Luteolin treatment significantly inhibited
the growth of HUVEC cells in concentration-dependent manner
(Fig. 3A) with IC50
value of 47.0 μM. However, no significant inhibitory effect was
observed on PBMC.
Luteolin inhibits angiogenesis in
vivo
Anti-angiogenic activity of luteolin on CAM was
assayed. The CAM of the chicken embryo provides a unique model for
investigating the process of new blood vessel formation and vessel
responses to anti-angiogenic agents. Using this model, we
additionally examined the potential in vivo anti-angiogenic
activity of luteolin. New blood vessels formed well on CAM in the
control group. Luteolin at 5 nmol/egg incubation for 72 h showed a
notable restraint. Up to 10 nmol/egg, the inhibition was getting
more prominent (Fig. 3B). These
results demonstrate that luteolin was able to suppress angiogenesis
in embryos.
Co-culture of HUVEC with PANC-1 activated
HUVEC tube formation, which were suppressed by luteolin
To investigate the effects of PANC-1 cells on HUVEC
angiogenesis, we used Transwell inserts with a pore size of 0.4 mm
and no collagen coating. HUVEC were cultured in the chamber coated
by Matrigel with PANC-1 cells in the inserts. When cultured alone
in the growth factor-reduced Matrigel, HUVEC barely formed
capillary structures 12 h after plating. However, HUVEC co-cultured
with PANC-1 cells displayed a dramatically increased network
formation and this effect disappeared when PANC-1 cells pre-treated
with 40 μmol/l luteolin for 30 min (Fig. 3C). These data suggested that
pancreatic carcinoma cells can induce endothelial cells to
differentiate into structures that resemble in vivo
neovascularization and this effect could be inhibited by
luteolin.
Luteolin suppresses VEGF secretion from
pancreatic carcinoma cells
It has been shown that VEGF, actively secreted from
hypoxic tumor cells, could potently trigger tumor angiogenesis.
Reduction of VEGF weakens its stimulation for tumor angiogenesis
(7). We additionally examined the
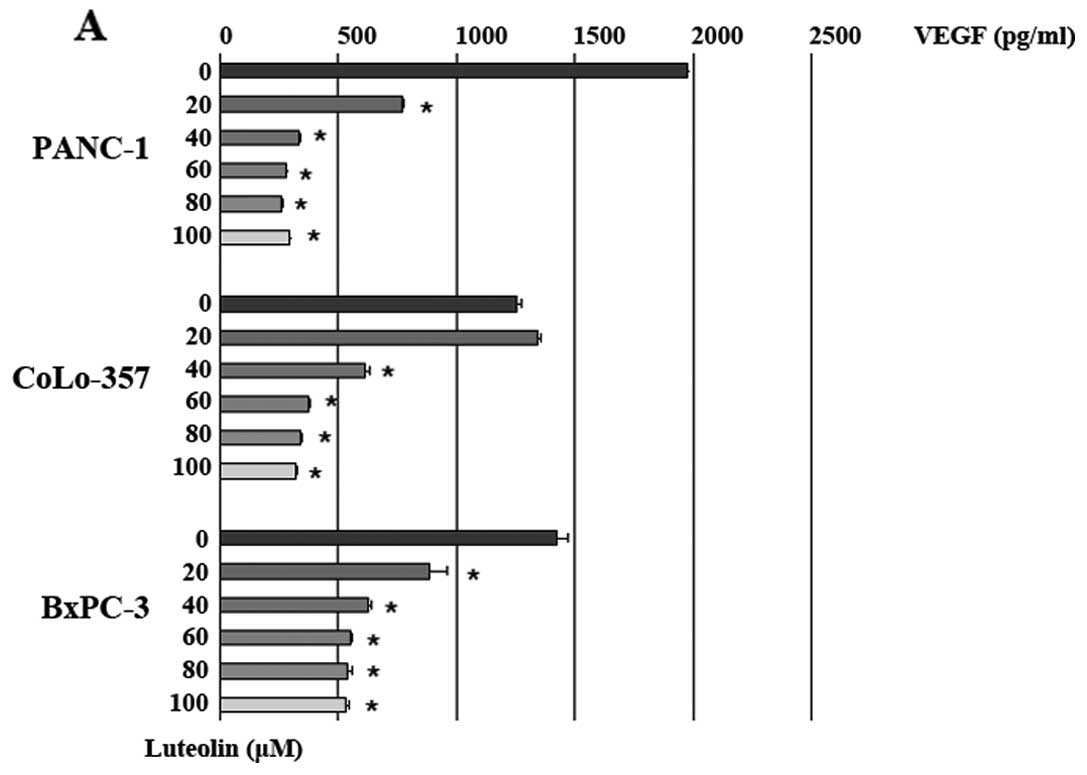
effect of luteolin on VEGF secretion from the pancreatic carcinoma
cells by ELISA analysis. The results showed that luteolin treatment
for 24 h decreased VEGF secretion compared to the vehicle control
group (Fig. 4A). Luteolin (40
μmol/l) decreased VEGF secretion by 83.1% (PANC-1 cells), 51.4%
(CoLo-357 cells) and 55.9% (BxPC-3 cells).
Luteolin suppresses production of VEGF
mRNA
The expression of intracellular VEGF protein was
detected by western blotting after treating with luteolin for 24 h.
As shown in Fig. 4B, VEGF protein
in untreated or treated-PANC-1 cells had no significant difference.
Further, we detected the VEGF mRNA level by RT-PCR. Gene expression
of these two growth factors were both decreased compared to the
vehicle control group (Fig. 4C).
These data suggested that the expression level of VEGF mRNA was
decreased after treatment with luteolin, but not the intracellular
VEGF protein level.
Luteolin suppresses VEGF secretion
related to JNK phosphorylation and NF-κB-DNA binding activity
Our results showed that luteolin suppressed VEGF
secretion from pancreatic carcinoma cells and antagonized
VEGF-indued angiogenesis in HUVEC. This prompted us to investigete
how luteolin regulate the secretion of VEGF. Since the secretion of
VEGF was regulated by MAP kinase pathways (8), we examined the expression and
phosphorylation of MAPK (ERK1/2, p38 and JNK). Western blotting
result showed that the expression and phosphorylation of ERK1/2 and
p38 had no significant difference compared with the control group
(Fig. 4D). However, phosphorylation
level of JNK was increased, which could induce apoptosis (6). These data suggested that the secretion
of VEGF may be regulate by JNK signaling pathway. The result from
RT-PCR revealed the VEGF mRNA level was decreased in a
concentration-dependent manner after luteolin treatment (Fig. 4C). Therefore, we hypothesized that
the abatement of VEGF secretion was due to inhibition of VEGF mRNA
expression. Our recent studies showed that luteolin inhibited TNFα
induced NF-κB translocation on A549 cells (6). VEGF mRNA expression was partly
regulated by nuclear transcription factor NF-κB. In view of the
above, EMSA assay was done to investigate the NF-κB-DNA binding
activity. The result showed that luteolin inhibited NF-κB
transcription activity in all three pancreatic carcinoma cells
(Fig. 4E).
Discussion
Angiogenesis is the physiological process involving
the growth of new blood vessels from pre-existing vessels (9). Angiogenesis is a necessary and
required step for transition from a small harmless cluster of cells
to a large tumor. Tumors cannot grow beyond a certain size,
generally 1–2 mm3 (10),
due to a lack of oxygen and other essential nutrients. Angiogenesis
is also required for the spread of a tumor or metastasis. Luteolin
was found to be a potent angiogenesis inhibitor (11). In a murine xenograft tumor model,
luteolin inhibited tumor growth and angiogenesis in xenografted
tumors (12). In this study, we
investigated the effect of luteolin on angiogenesis in vitro
with human umbilical vein endothelial cells. Luteolin showed
significant inhibitory to HUVEC, but had no effect on human normal
cell PBMC. In vivo experiment, luteolin at 5 nmol/egg
incubation for 72 h showed a notable inhibition of new blood vessel
formation in CAM. These result indicate that luteolin may be used
as an anti-angiogenesis agent with little side effect.
The significant role of vegetables and fruits in
prevention and treatment of cancer is due to their high polyphenol
content, particularly of flavonoids (13). Luteolin is a common dietary
flavonoid that can be found in a large quantity of plants and
foods. Luteolin was reported to inhibit the development of a series
of solid tumors (colonic HT-29, HCT116, hepatic HepG2, SK-Hep-1,
PLC/PRF/5, Hep3, cervical HeLa, oral SCC-4) (14–23),
but the mechanism of luteolin affecting pancreatic carcinoma cells
was not addressed. Therefore, we used three different human
pancreatic carcinoma cell lines, the poorly differentiated cell
line (PANC-1), the moderately differentiated cell line (CoLo-357)
and the well differentiated cell line (BxPC-3) as model to examine
the therapeutic effects of luteolin. In our studies, luteolin
inhibited the proliferation of those three pancreatic carcinoma
cell lines and induced apoptotic cell death through caspase
pathway. Luteolin induced G2 phase arrest of cell cycle progression
on PANC-1 and CoLo-357 cells. Since pancreatic carcinoma shows
strong tumor angiogenesis, overexpression of VEGF, we detected the
VEGF expression in pancreatic carcinoma cells with western blotting
and VEGF secreted into the medium with ELISA. The results showed
that intracellular VEGF protein expression level was constant, when
the VEGF secreted into the medium decreased significantly. This
prompted us to investigate whether the inhibition of VEGF secretion
on pancreatic carcinoma cells could be anti-angiogenic. Therefore,
we co-cultured the luteolin-treated PANC-1 cells with HUVEC. As
speculated, the capillary-like structures, consisting of HUVEC,
decreased compared with the luteolin-untreated group.
Since the secretion of VEGF was regulated by the MAP
kinase pathway, three key MAP kinases (ERK1/2, p38 and JNK) were
chosen for investigation. The phosphorylation of JNK was
upregulated by luteolin, when p-ERK1/2 and p-p38 did not change.
Substantial research on luteolin has revealed that luteolin could
either activate JNK (15,24) or downregulate JNK phosphorylation
(25–27), both leading to apoptosis. These data
suggested that the secretion of VEGF could be regulated by the JNK
signaling pathway. The result from RT-PCR revealed the VEGF mRNA
level was decreased in a concentration-dependent manner after
luteolin treatment. Therefore, we hypothesized that the abatement
of VEGF secretion was due to inhibition of VEGF mRNA expression.
VEGF mRNA expression was partly regulated by nuclear transcription
factor NF-κB. EMSA assay showed that luteolin inhibited NF-κB
transcription activity in all three pancreatic carcinoma cells. How
luteolin affects the NF-κB transcription activity is now under
study in our laboratory.
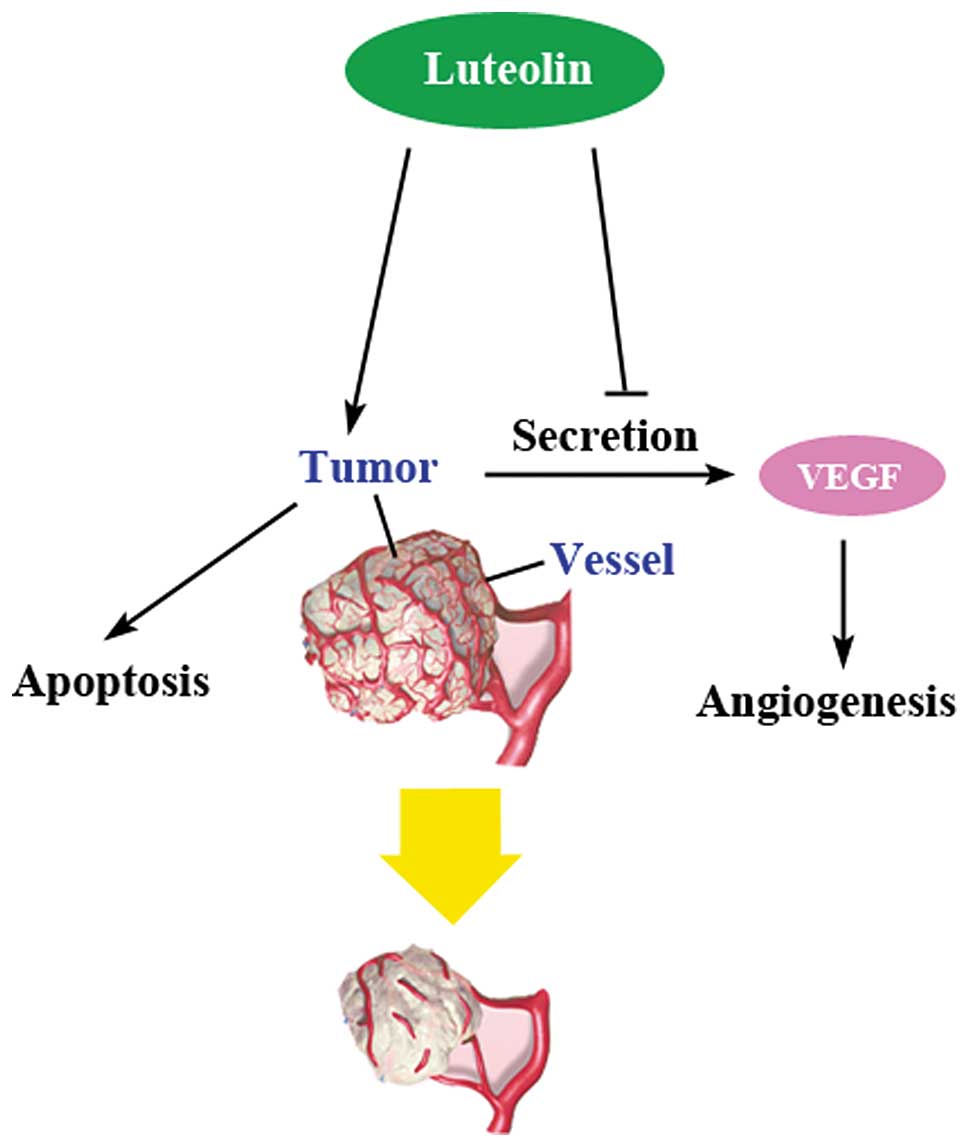
Taking these results together, we demonstrated that
luteolin possessed dual roles, the direct action on endothelial
cells and reduction of the VEGF secretion from tumor cells
(Fig. 5). Such dual activities of
luteolin, different from the relatively single effects of other
angiogenesis inhibitors, can be reasonably inferred to allow better
in vivo and even better clinical effects as a novel
antitumor agent.
Acknowledgements
This study was supported by the National Natural
Science Foundation of China (Nos. 30701098, 30873410 and 81073101),
Natural Science Foundation of Zhejiang Province (No. Y2090676) and
Jiangsu Province’s Outstanding Leader Program of Traditional
Chinese Medicine.
References
|
1
|
Neesse A, Gress TM and Michl P:
Therapeutic targeting of apoptotic pathways: novel aspects in
pancreatic cancer. Curr Pharm Biotechnol. May 24–2011.(Epub ahead
of print).
|
|
2
|
Kato H, Ishikura H, Kawarada Y, Furuya M,
Kondo S and Yoshiki T: Anti-angiogenic treatment for peritoneal
dissemination of pancreas adenocarcinoma: a study using TNP-470.
Jpn J Cancer Res. 92:67–73. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Saif MW: Anti-angiogenesis therapy in
pancreatic carcinoma. JOP. 7:163–173. 2006.PubMed/NCBI
|
|
4
|
Lin Y, Shi R, Wang X and Shen HM:
Luteolin, a flavonoid with potential for cancer prevention and
therapy. Curr Cancer Drug Targets. 8:634–646. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Jaffe EA, Nachman RL, Becker CG and Minick
CR: Culture of human endothelial cells derived from umbilical
veins. Identification by morphologic and immunologic criteria. J
Clin Invest. 52:2745–2756. 1973. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cai X, Ye T, Liu C, et al: Luteolin
induced G2 phase cell cycle arrest and apoptosis on non-small cell
lung cancer cells. Toxicol In Vitro. 25:1385–1391. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Minchenko A, Bauer T, Salceda S and Caro
J: Hypoxic stimulation of vascular endothelial growth factor
expression in vitro and in vivo. Lab Invest. 71:374–379.
1994.PubMed/NCBI
|
|
8
|
Nagineni CN, Samuel W, Nagineni S, et al:
Transforming growth factor-β induces expression of vascular
endothelial growth factor in human retinal pigment epithelial
cells: involvement of mitogen-activated protein kinases. J Cell
Physiol. 197:453–462. 2003.
|
|
9
|
Das A and McGuire PG: Retinal and
choroidal angiogenesis: pathophysiology and strategies for
inhibition. Prog Retin Eye Res. 22:721–748. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
McDougall SR, Anderson AR and Chaplain MA:
Mathematical modelling of dynamic adaptive tumour-induced
angiogenesis: clinical implications and therapeutic targeting
strategies. J Theor Biol. 241:564–589. 2006. View Article : Google Scholar
|
|
11
|
Joussen AM, Rohrschneider K, Reichling J,
Kirchhof B and Kruse FE: Treatment of corneal neovascularization
with dietary isoflavonoids and flavonoids. Exp Eye Res. 71:483–487.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bagli E, Stefaniotou M, Morbidelli L, et
al: Luteolin inhibits vascular endothelial growth factor-induced
angiogenesis; inhibition of endothelial cell survival and
proliferation by targeting phosphatidylinositol 3′-kinase activity.
Cancer Res. 64:7936–7946. 2004.PubMed/NCBI
|
|
13
|
Ramos S: Effects of dietary flavonoids on
apoptotic pathways related to cancer chemoprevention. J Nutr
Biochem. 18:427–442. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chang J, Hsu Y, Kuo P, Kuo Y, Chiang L and
Lin C: Increase of Bax/Bcl-XL ratio and arrest of cell cycle by
luteolin in immortalized human hepatoma cell line. Life Sci.
76:1883–1893. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lee HJ, Wang CJ, Kuo HC, Chou FP, Jean LF
and Tseng TH: Induction apoptosis of luteolin in human hepatoma
HepG2 cells involving mitochondria translocation of Bax/Bak and
activation of JNK. Toxicol Appl Pharmacol. 203:124–131. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lee WJ, Wu LF, Chen WK, Wang CJ and Tseng
TH: Inhibitory effect of luteolin on hepatocyte growth
factor/scatter factor-induced HepG2 cell invasion involving both
MAPK/ERKs and PI3K-Akt pathways. Chem Biol Interact. 160:123–133.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lim do Y, Jeong Y, Tyner AL and Park JH:
Induction of cell cycle arrest and apoptosis in HT-29 human colon
cancer cells by the dietary compound luteolin. Am J Physiol
Gastrointest Liver Physiol. 292:G66–G75. 2007.PubMed/NCBI
|
|
18
|
Plaumann B, Fritsche M, Rimpler H,
Brandner G and Hess RD: Flavonoids activate wild-type p53.
Oncogene. 13:1605–1614. 1996.PubMed/NCBI
|
|
19
|
Zhang Q, Zhao XH and Wang ZJ: Flavones and
flavonols exert cytotoxic effects on a human oesophageal
adenocarcinoma cell line (OE33) by causing G2/M arrest and inducing
apoptosis. Food Chem Toxicol. 46:2042–2053. 2008. View Article : Google Scholar
|
|
20
|
Schutte ME, Boersma MG, Verhallen DA,
Groten JP and Rietjens IM: Effects of flavonoid mixtures on the
transport of 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP)
through Caco-2 monolayers: an in vitro and kinetic modeling
approach to predict the combined effects on transporter inhibition.
Food Chem Toxicol. 46:557–566. 2008.PubMed/NCBI
|
|
21
|
Xavier CP, Lima CF, Preto A, Seruca R,
Fernandes-Ferreira M and Pereira-Wilson C: Luteolin, quercetin and
ursolic acid are potent inhibitors of proliferation and inducers of
apoptosis in both KRAS and BRAF mutated human colorectal cancer
cells. Cancer Lett. 281:162–170. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yang SF, Yang WE, Chang HR, Chu SC and
Hsieh YS: Luteolin induces apoptosis in oral squamous cancer cells.
J Dent Res. 87:401–406. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yee SB, Lee JH, Chung HY, et al:
Inhibitory effects of luteolin isolated from Ixeris sonchifolia
Hance on the proliferation of HepG2 human hepatocellular carcinoma
cells. Arch Pharm Res. 26:151–156. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Shi RX, Ong CN and Shen HM: Luteolin
sensitizes tumor necrosis factor-alpha-induced apoptosis in human
tumor cells. Oncogene. 23:7712–7721. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ando C, Takahashi N, Hirai S, et al:
Luteolin, a food-derived flavonoid, suppresses adipocyte-dependent
activation of macrophages by inhibiting JNK activation. FEBS Lett.
583:3649–3654. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Jang S, Kelley KW and Johnson RW: Luteolin
reduces IL-6 production in microglia by inhibiting JNK
phosphorylation and activation of AP-1. Proc Natl Acad Sci USA.
105:7534–7539. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kimata M, Shichijo M, Miura T, Serizawa I,
Inagaki N and Nagai H: Effects of luteolin, quercetin and baicalein
on immunoglobulin E-mediated mediator release from human cultured
mast cells. Clin Exp Allergy. 30:501–508. 2000. View Article : Google Scholar : PubMed/NCBI
|