Introduction
Neuroendocrine tumors (NETs) represent rare tumors
that are derived from cells of the embryonic neural crest. They
account for ~0.5% of all malignancies and the incidence is
~2/100,000 (1). The ability to
secrete hormones and release them into the bloodstream is one
characteristic of NET cells. These tumors originate from endocrine
glands, whereby their origin and behavior can vary over a wide
range. MTCs are calcitonin producing tumors arising from the
parafollicular C-cells and occur in ~4–10% of all thyroid tumors
(2). MTCs (75%) mainly occur
sporadically, but there is also a familial form of MTCs without any
associated endocrinopathies (FMTC), in which germline mutations of
the ‘rearranged during transfection’ (RET) proto-oncogene have very
often been identified. The remaining 25% of MTC cases are autosomal
dominantly inherited and are combined with other endocrinopathies
summarized as multiple endocrine neoplasia (MEN) type 2A or 2B
(3,4). SI-NETs, formerly called carcinoids,
have their origin in enterochromaffin cells throughout the
intestinal tract. They have the ability to secrete and release
serotonin into the bloodstream, which can result in flushing,
diarrhea, bronchoconstriction and fibrosis, summarized as the
carcinoid syndrome (5).
SI-NETs can occur sporadically, but also in a
dominantly inherited form associated with multiple endocrine
neoplasia type 1 (MEN1), which occurs due to loss-of-function
germline mutations of the MEN1 gene. Since these NETs
usually have already metastasized when diagnosed, none of the
treatment options reported have been found to be effective and the
response rate has generally been low (6). As the only curative treatment for NETs
is early removal of all neoplastic tissue (7,8) new
strategies for the treatment remain crucial. Medicinal herbs are of
high interest in anticancer drug research (9–11).
Christia vespertilionis, a south-east Asian Fabaceae, is
known to exhibit antiplasmodial activity and to increase
cytotoxicity in HeLa cells compared to normal human lung cells
(12). Triterpenes, alkaloids,
fatty acids, phenols, alkanes and long chained alcohols have been
identified as main constituents of this plant.
Remarkably, isoquinoline alkaloids, usually typical
for Papaveraceae or closely related families, were found in
Christia plant extracts. In a previous study, pheophorbid-a,
a chlorophyll derivative in Christia vespertilionis plant
extracts showed high antiproliferative activity in MTC cells
(13). The aim of the present study
was to examine the in vitro effects of bioactive agents
extracted from Christia vespertilionis in chemo- and
radiation-resistant NET cells.
Materials and methods
Plant material
Specimen of Christia vespertilionis, a
south-east Asian Fabaceae, was deposited at the Institute of
Pharmacy at the University of Innsbruck. The plant was originally
bought at a market in Vietnam. Aerial parts of the plant were
successively extracted in dichloromethane in a Soxhlet apparatus
and four subfractions extracted by petroleum ether (CV-44), ethyl
acetate (CV-45), butanol (CV-46) and water (CV-47). Two pure
substances, palmitine (CV-48) and corynoxidine (CV-49), were
isolated from CV-46. The identity was confirmed by 1D and 2D NMR
spectroscopy and mass spectrometry. Dried extracts were redissolved
in dimethyl sulfoxide (DMSO, Sigma-Aldrich, Vienna, Austria) at a
concentration of 5 mg/ml and stored at −20°C.
Cell lines and cell culture
The human MTC cell line MTC-SK(14), the SI-NET cell line
KRJ-I(15) and the normal
human fibroblast cell line HF-SAR (Pfragner R, personal
communication) were established in our laboratory. MTC-SK
and KRJ-I cells were cultured in Ham’s F12:M199 medium (1:1)
(BioWhittaker; Lonza, Verviers, Belgium) with 10% fetal bovine
serum (FBS) (PAA Laboratories, Pasching, Austria) at an initial
cell number of 2×105 cells/ml. Human skin fibroblasts,
HF-SAR, isolated from a 2-year-old male served as control
and were cultured in DMEM (BioWhittaker; Lonza), supplemented with
L-glutamine and 10% FBS (PAA Laboratories, Vienna, Austria) at an
initial cell number of 1×105 cells/ml. All cells were
cultured at 37°C in a humidified atmosphere containing 5%
CO2. Cell lines were Mycoplasma-free, as tested
with MycoAlert Mycoplasma detection kit (Lonza, Rockland,
ME, USA). For all experiments, cells were cultured without
antibiotics.
Cell counting
Cells were counted with the CASY-1® Cell
Counter Analyser TTC (Schärfe system; Reutlingen, Germany).
MTC-SK as well as KRJ-I cells were seeded into
24-well plates at a density of 2×105 cells/ml and
incubated for 24, 48 and 72 h, respectively, with DMSO (control) or
supplemented with 10 μg/ml of CV extracts. Cell clusters were
pipetted into single cells and each sample was analyzed in
triplicate by cell counting.
Cell proliferation
WST-1 cell proliferation reagent (Roche Diagnostics,
Vienna, Austria) was used to quantify viability and proliferation
in MTC-SK, KRJ-I and HF-SAR after CV treatment.
Mitochondrial dehydrogenases are able to convert WST-1, a
tetrazolium salt, into formazan, which leads to a change of
absorbance. MTC-SK and KRJ-I cell suspensions were
seeded into 24-well plates and incubated with DMSO (control) or 10
μg/ml of plant extracts; then cell viability was measured. After
24, 48 and 72 h of treatment, cell aggregates were pipetted into
single cells. The measurement was carried out according to the
manufacturer’s protocol. Adherent HF-SAR cells were directly
seeded into 96-well plates and cell viability was measured as
above. Samples were tested in 6 replicates.
DAPI staining
DAPI (4′,6′-diamidino-2 phenylindole) is a
fluorochrome that forms fluorescence complexes with double-stranded
DNA. Treatment of cells with the fluorescent dye leads to a blue
coloring of the nuclei when irradiated at an excitation wavelength
of 350 nm. Apoptotic nuclei can be identified by the condensed
chromatin gathering at the periphery of the nuclear membrane or a
fragmented morphology of nuclear bodies, observed by fluorescence
microscopy. Cell pellets of MTC-SK as well as KRJ-I
cells were resuspended in DAPI (Sigma) solution pursuant to the
manufacturer’s instructions. The stained cells were pipetted onto
glass slides, covered with a cover slip and analyzed under a
fluorescence microscope (Leica DM 4000/EL6000; Leica, Wetzlar,
Germany). Nuclei that showed clearly condensed and fragmented
chromatin were scored as apoptotic.
Caspase-Glo 3/7 assay
To measure the activity of apoptotic key effector
caspases 3 and 7, the Caspase-Glo® 3/7 assay (Promega,
Mannheim, Germany), which is based on a luminescence reaction, was
used. The obtained luminescence signal is proportional to caspase
3/7 activity. In a time period of 5 or 8 h, respectively, CV
extract or DMSO was added to the cells. Samples were transferred
into white-walled 96-well plates (Nunc™, Thermo Fisher Scientific,
Vienna, Austria). Caspase-Glo 3/7 reagent was added according to
the manufacturer’s instructions and luminescence of each sample
measured with the GloMax®-Multi+ Microplate
Multimode Reader with Instinct™ (Promega). Medium supplemented with
10% FBS was used for blank reactions. DMSO treated cells were used
as control. Each treatment was measured in triplicate; arithmetic
mean and SD were calculated automatically by the GloMax
software.
Cell morphology
Effects of CV-45 on cell morphology of treated
MTC-SK and KRJ-I cells were observed with the Nikon
inverted microscope (Eclipse TE 300; Nikon, Tokyo, Japan).
MTC-SK and KRJ-I cells (2×105 cells/ml)
were treated with either 10 μg/ml of CV-45 or DMSO (control) and
incubated for 24, 48 and 72 h in 24-well plates. After each
incubation day, cell morphology was observed and images were taken
(Nikon 12-bit CCD camera, Nikon).
RNA extraction and reverse
transcription
MTC-SK and KRJ-I cells
(2×105 cells/ml) were transferred into cell culture
flasks (75 cm2) and incubated for 2 h with 10 μg/ml of
CV-45, or DMSO (control). After incubation, cells were transferred
into 50 ml tubes (Sarstedt), and centrifuged for 10 min at 300 × g;
the cell pellets were used for RNA isolation with the TRI
Reagent® (Molecular Research Centre, Cincinnati, OH,
USA). The concentration of isolated RNA was determined using the
Nanodrop (NanoDrop® Spectrophotometer ND-1000, Peqlab
Biotechnology GmbH, Erlangen, Germany). Each preparation (1 μg) was
reverse-transcribed using the High Capacity RNA to cDNA kit
(Applied Biosystems, Carlsbad, CA, USA) following the
manufacturer’s instructions.
Quantitative real-time polymerase chain
reaction (q RT-PCR)
The relative gene expression of MTDH
(Metadherin, also known as lyric), PDCD5
(programmed cell death 5) and TNFRSF10b (tumor necrosis
factor receptor superfamily, member 10b) in CV treated cells in
comparison to untreated cells (control) was analyzed. Primers are
summarized in Table I. SYBR Green I
fluorescence dye (Bio-Rad) was used for detection. IQ Sybergreen
Supermix,12.5 μl, with 5.5 μl RNAse free water and 0.5 μl of each
forward and reversed primer (10 μM) were mixed thoroughly and 6 μl
cDNA (5 ng/μl) of CV-45 treated and untreated MTC-SK and
KRJ-I cells added to the Master mix in PCR tubes. For the
calibration curve of each gene of interest, cDNA of pooled RNA
(treated + untreated) was added and combined with Master mixes for
each gene. The following PCR conditions were used: 95°C for 3 min,
44 cycles at 95°C/10 sec, 60°C/45 sec, 72°C/40 sec, followed by
55°C for 10 sec and the condition for the melting curve (55°C–95°C
for 5 sec). Data were normalized using the expression of the
housekeeping gene GAPDH. Relative gene expression was
assessed using the ΔΔCq-method (16).
 | Table IPrimer sequences for quantitative RT
PCR. |
Table I
Primer sequences for quantitative RT
PCR.
| GenBank Acc.
no. | Gene ID | Gene name | Forward primer | Reverse primer | Product size
(bp) |
|---|
| NM_178812 | MTDH | Metadherin |
CAGATCAGTGCAGGTGAGGA |
GGAATTGGTTCCTGGGACTT | 374 |
| NM_004708 | PDCD5 | Programmed cell
death 5 |
AAAGCACAGGGAAGCAGAAA |
GGGCACATAGTCATCCCACT | 275 |
| NM_003842 | TNFRSF10B | Tumor necrosis
factor receptor superfamily, member 10b |
CACCAGGTGTGATTCAGGTG |
CCCCACTGTGCTTTGTACCT | 221 |
| NM_001256799 | GAPDH | Glyceraldehyde-
3-phosphate dehydrogenase |
AATGCCTTGTCGAAGTCCAG |
TCTGTGTCTGCCTTTGTTGG | 236 |
Statistical analysis
Medians and SD were calculated using Microsoft Excel
software (Microsoft Office, version 2011). A statistical analysis
of differences between treated and untreated tumor cells was
carried out with a two-tailed unpaired Student’s t-test. A P-value
<0.05 was considered to indicate significance; P-values <0.01
indicated ‘very significant’. Experimental results are shown in the
graphs as mean value ± standard error of the mean.
Results
Cell proliferation
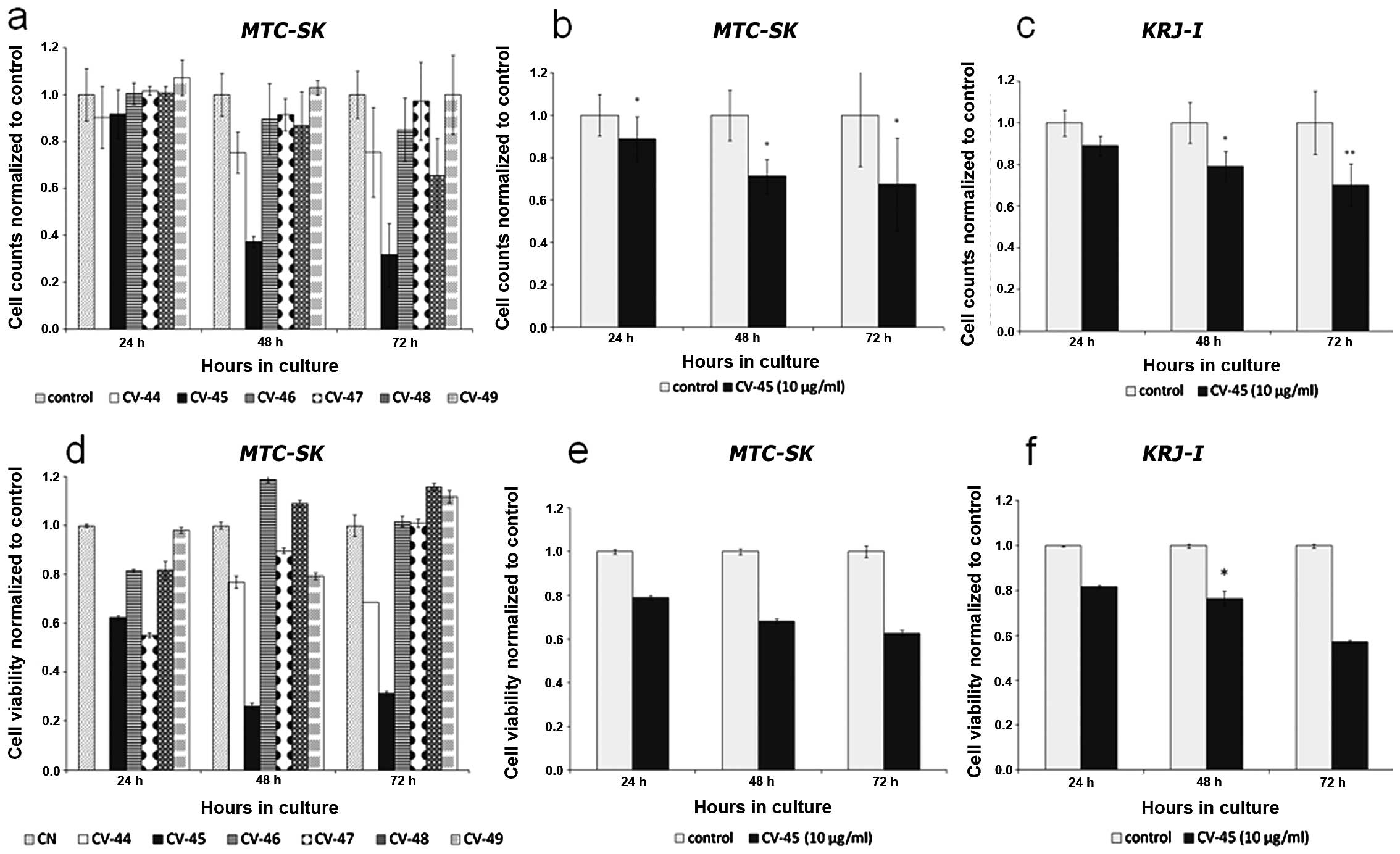
The effect of the petroleum ether (CV-44),
ethylacetate (CV-45), butanol (CV-46) and water (CV-47) fractions
and the two isolated pure substances (CV-48 and CV-49) of
Christia vespertilionis on cell-proliferation and
cell-viability was tested by cell counting in MTC-SK cells.
At a concentration of 10 μg/ml all subfractions showed a reduction
of cell viability within 72 h (Fig.
1a). After 72 h treatment with CV-45, numbers of total viable
MTC-SK cells were significantly lower than those treated
with DMSO or other fractions. Isolated pure Christia
vespertilionis showed very weak inhibition of cell viability;
only after 72 h did CV-48 show decreased number of viable cells as
compared to DMSO treated control cells. We obtained the highest
inhibition of proliferation with the ethylacetate fraction CV-45
compared to other tested fractions and pure substances. Treatment
of MTC-SK as well as KRJ-I cells with 10 μg/ml of
CV-45 showed significant inhibition of cell growth. As shown in
Fig. 1b, the cell number obtained
by cell counting of MTC-SK cells after CV-45 treatment
compared to control cells (DMSO) was 11.3% less after 24 h, 28.9%
less after 48 h and 32.5% less after 72 h (t-test p<0.05). The
cell number obtained by counting KRJ-I cells (Fig. 1c) after CV-45 treatment was also
significantly decreased as compared to control cells. Following
CV-45 treatment, total viable cell counts decreased by 11.1% after
24 h, by 20.8% after 48 h and by 29.9% after 72 h (t-test
p<0.01).
Cell viability
The potency of CV plant extracts to reduce cell
viability of MTC-SK cells is shown in Fig. 1d. After 48 h of exposure, we
measured a decrease in cell viability with CV-44, CV-45 and CV-47,
but not with CV-46 and only very weakly with the isolated pure
substances CV-48 and CV-49; after 72 h of treatment CV-46 and CV-47
as well as CV-48 and CV-49 we did not observe antiproliferative
effects; on the contrary, CV-44 and CV-45 showed the highest
antiproliferative effects after 48 and 72 h of exposure (77% cell
viability after 48 h and 69% cell viability after 72 h with CV-44
compared to control cells and 26% cell viability after 48 h and 31%
cell viability after 72 h with CV-45 compared to control cells).
The highest antiproliferative effect was measured for CV-45. As
shown in Fig. 1e, cell viability of
MTC-SK decreased 21.1% after 24 h, 31.8% after 48 h and
37.4% after 72 h exposure to 10 μg/ml of CV-45 when compared to
control cells (DMSO). Similarly to MTC-SK cells,
KRJ-I cells were also suppressed by CV-45 fraction (Fig. 1f). After 24 h of CV-45 treatment (10
μg/ml) cell viability decreased by ~18.3%, after 48 h,
significantly by 23.5% (t-test p<0.05) and after 72 h by 42.8%
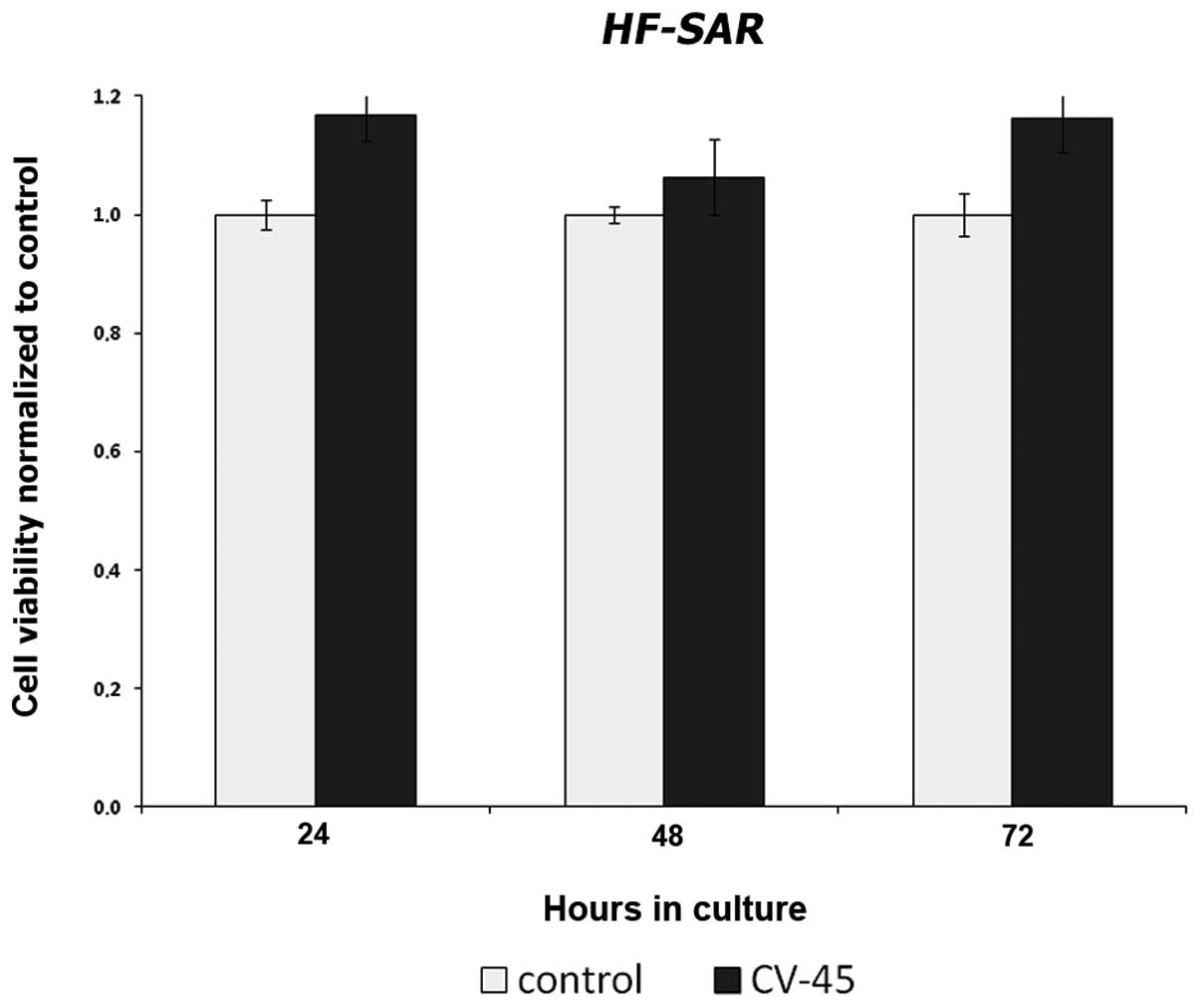
compared to DMSO treated cells (control). In human skin fibroblast
HF-SAR serving as control cells, the same concentration of
CV-45 did not inhibit cell proliferation (Fig. 2).
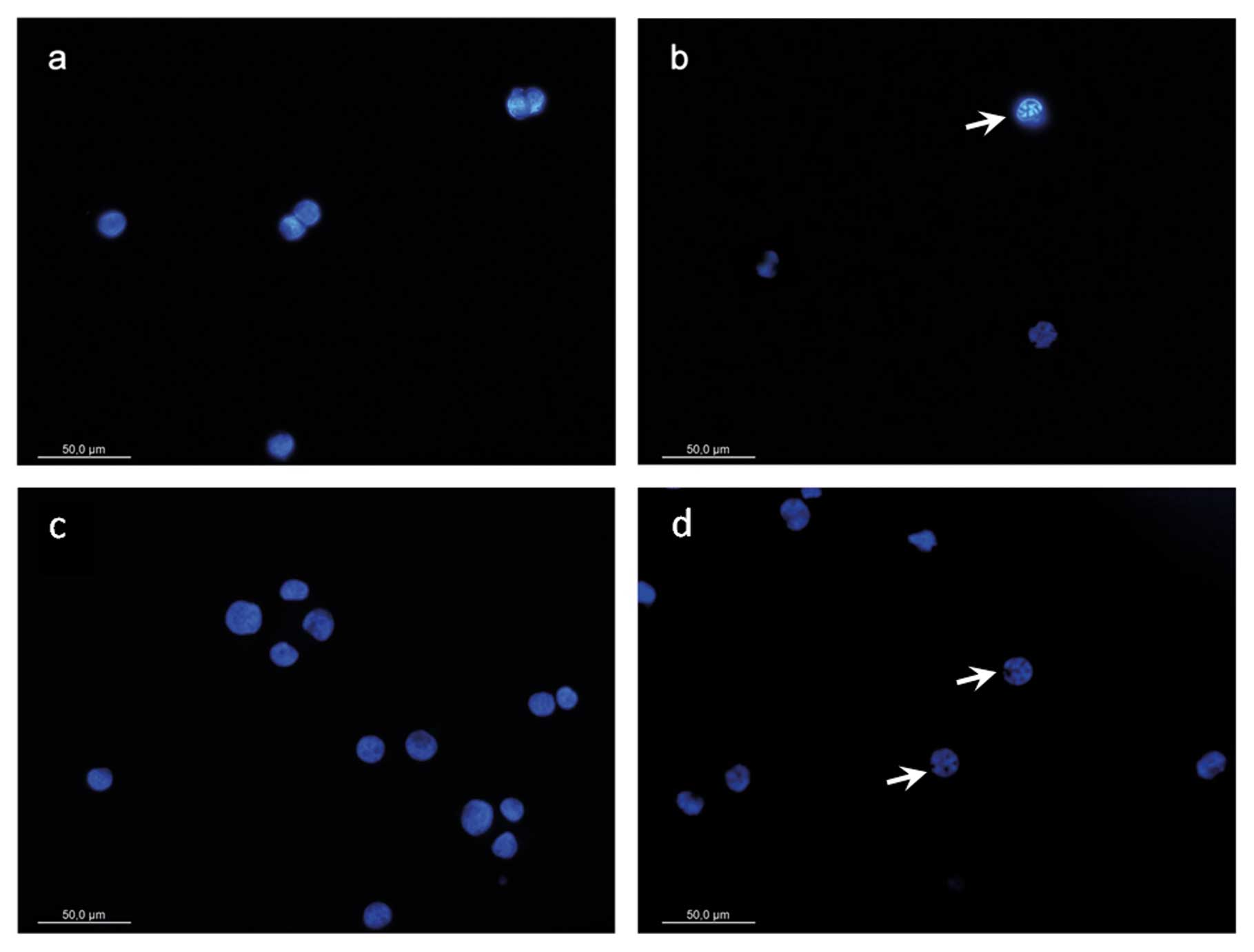
DAPI-staining
We found morphological changes in MTC-SK as
well as in KRJ-I cells including chromatin condensation,
cell shrinking and apoptotic bodies when cells were treated for 48
and 72 h with CV-45 (Fig. 3). No
significant changes were observed in MTC-SK and KRJ-I
control cells (DMSO).
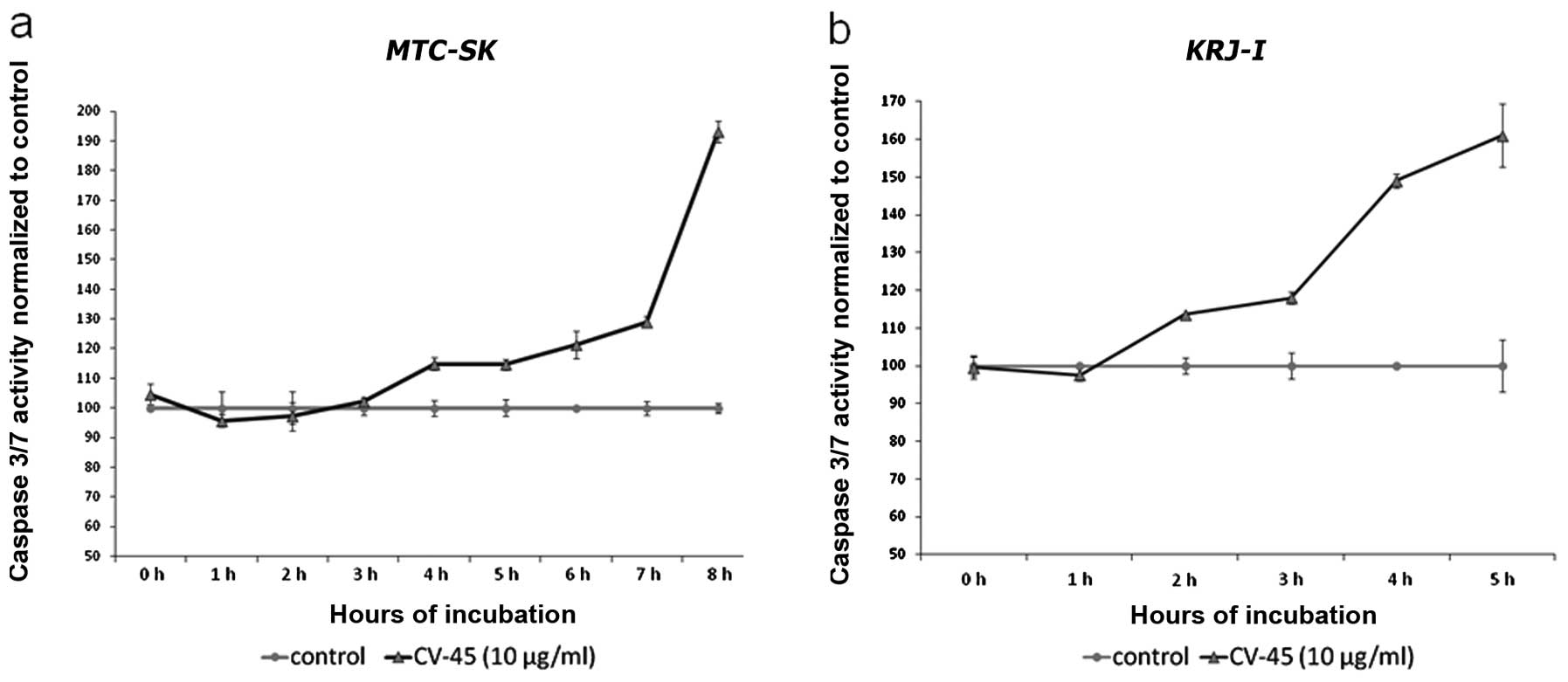
Induction of apoptotic pathway
CV-45 treatment increased caspase 3/7 activity in
MTC-SK and KRJ-I cells within 24 h, which correlated
with a decrease in viable cells. An increase of caspase 3/7
activity was no longer evident after 48 and 72 h of CV-45
treatment, although the amount of viable cells after CV-45
treatment still decreased after 48 and 72 h (data not shown). After
6 h of exposure at a concentration of 10 μg/ml, the caspase 3/7
activity of CV-45 treated MTC-SK cells was ~21.4% higher and
after 8 h caspase 3/7 activity nearly doubled (93.1%) compared to
control cells (Fig. 4a). In
KRJ-I cells, caspase 3/7 activity had increased by 13.5%
after 2 h of CV-45 treatment, 17.9% after 3 h, 49.1% after 4 h and
61% after 5 h CV-45 compared to control cells (Fig. 4b).
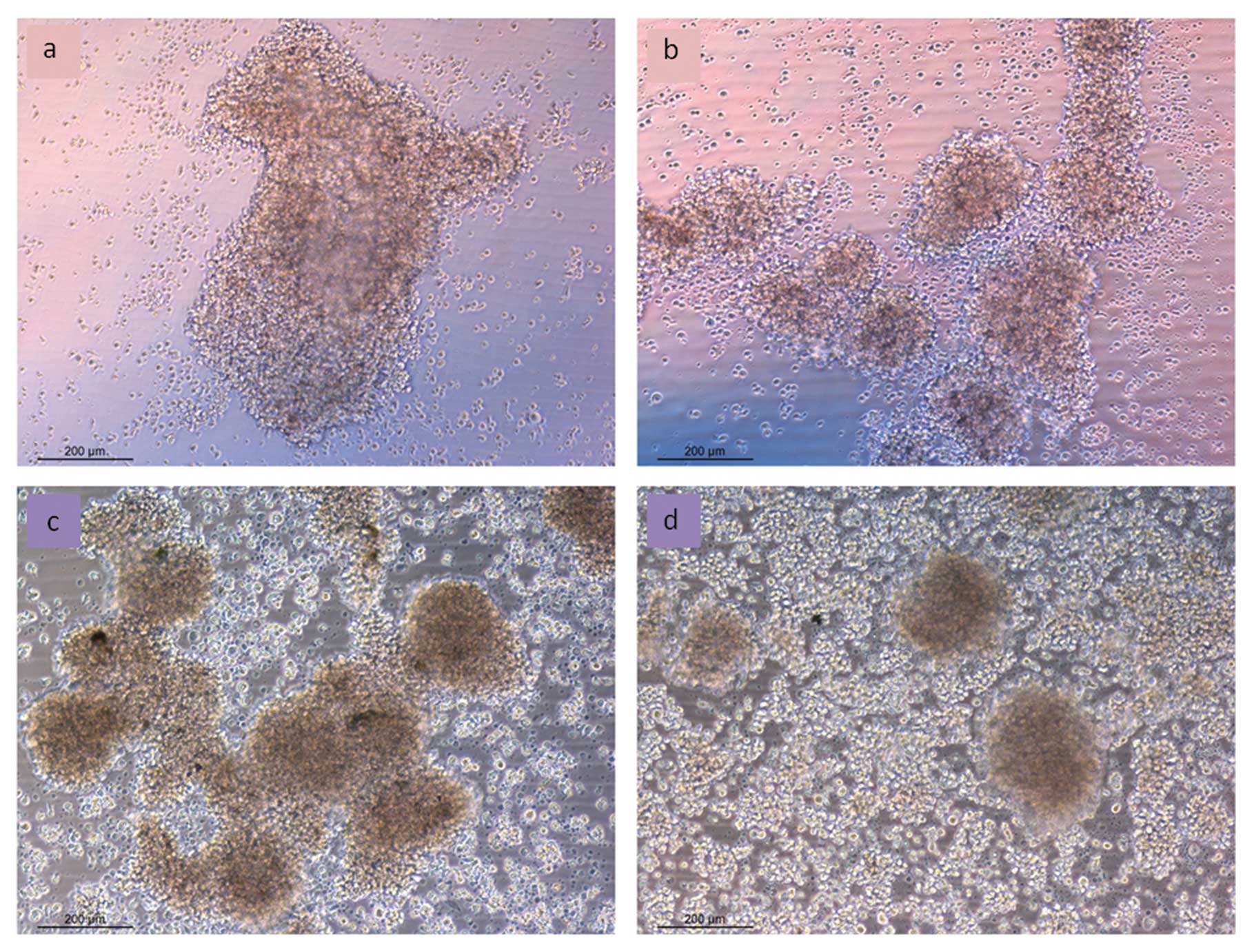
Cell morphology in vitro
MTC-SK as well as KRJ-I cells grow in
multicellular spheroids. Fig. 6
shows MTC-SK and KRJ-I cells after treatment with 10
μg/ml of CV-45 compared to control. Forty-eight hours after adding
CV-45 to MTC-SK cells, treated cells showed multicellular
aggregates of ~300 μm diameter and more single cells were visible,
compared to the much larger aggregates (800–900 μm) formed by
control cells (Fig. 5a and b). In
KRJ-I cells, CV-45 treatment also led to reduction of
aggregate formation. After 48 h of CV-45 treatment, multicellular
clusters of KRJ-I were mostly dissociated into single cells.
Aggregates of control cells were twice as large as those of treated
cells (Fig. 5c and d).
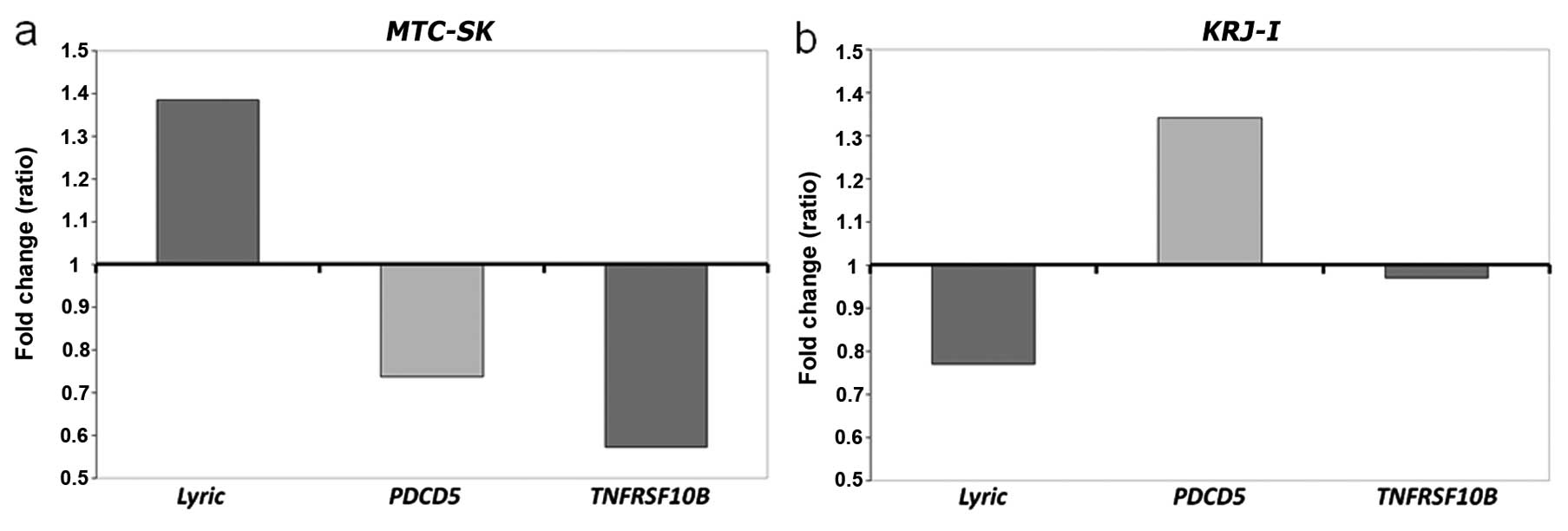
Gene expression of PDCD5, MTDH and
TNFRSF10b
The data on cycles of quantity (Cq-value)
of the reference gene GAPDH showed stable results in
MTC-SK, indicating that expression of GAPDH was not
influenced by CV-45 treatment (17.96 in CV-45 treated versus 18.01
in control, data not shown). Quantitative RT-PCR showed increased
expression of MTDH and downregulation of PDCD5 and
TNFRSF10b in CV-45 treated MTC-SK cells (Fig. 6a). Although GAPDH gene
expression was slightly influenced by CV-45 treatment in
KRJ-I cells, the gene expression analysis showed that CV-45
treatment lead to downregulation of MTDH and upregulation of
PDCD5 gene expression in KRJ-I cells. The expression
of TNFRSF10b was not significantly altered in KRJ-I
cells when treated for 3 h with CV-45 (Fig. 6b).
Discussion
The establishment of new treatment options for
chemo- and radiation-resistant NETs is essential because of the
inefficacy of conventional chemotherapy. Medicinal herbs have come
increasingly into the spotlight as complementary medicines. In the
present study, we provide a first report of the antitumor activity
of plant extracts from Christia vespertilionis, in which the
ethylacetate fraction CV-45 showed significant antiproliferative
and pro-apoptotic effects in MTC-SK as well as in
KRJ-I cells.
It is known that many chemotherapeutic agents are
able to induce apoptosis in cancer cells, as with sorafenib or
5-fluorouracil for human hepatoma cells (18,19).
One goal in the establishment of new therapies against NETs is to
define substances that have the ability to trigger anticancer
effects and to induce apoptosis specifically in tumor cells, but
not in normal cells.
In the human fibroblasts (HF-SAR) tested, the
same concentration of CV-45 (10 μg/ml) as used for tumor cells did
not inhibit proliferation, suggesting that the composition of this
fraction does affect tumor cells specifically. Moreover, bioactive
ingredients of Christia vespertilionis extracts, only some
of which have been identified to date, look like good candidates
for further evaluation. The CV plant extracts were resolved in
DMSO, which is often discussed to be cell toxic. However, Da
Violante et al showed, DMSO can be used as solvent at even
higher concentrations, up to 10%, without significant cell damage
(17).
The tendency of MTC as well as SI-NETs to grow in
multicellular aggregates when cultured in vitro may be
evidence for the resistance of NETs to conventional chemo- and
radiation therapies (20,21). As reported by Pfragner et
al(15), both MTC-SK as
well as KRJ-I cells grow in multicellular spheroids. To
optimize treatments for MTCs and carcinoid cells, these cell
clusters must be dissociated into single cells to make them more
accessible to chemotherapeutics. Treatment of MTC-SK and
KRJ-I cells with CV-45 showed only weak effects on
disruption of cellular aggregates. This may be due to the low
concentration of the plant extract in contrast to the pure
substance. It should be noted that we do not yet know whether this
spheroid destroying effect could also be achieved by other
components.
Many chemotherapeutic drugs degrade tumor cells by
activating cascades of reactions that generally result in
apoptosis. The induction of apoptosis follows a very complex
pathway involving many cell proteins. Caspases 3 and 7 are known to
be proteases inducing and activating proteins responsible for cell
fragmentation and cell shrinking. Further, an evaluation of the
activity of caspase 3/7 is a meaningful method to identify
induction of apoptosis. The natural compounds of CV-45 showed
strong evidence of triggering apoptosis in NET cells. Within 24 h
of CV-45 treatment, caspase 3/7 activity was increased in
MTC-SK as well as in KRJ-I cells, but no longer after
48 and 72 h. When compared to cell viability, these results
correlate within 24 h of CV-45 treatment, with a decrease in cell
counts compared to an increase of caspase 3/7 activity, but no
longer correlate after 48 and 72 h. We know accordingly that
caspase 3 activation is an early-stage event in apoptosis; thus,
this could be the reason why a significant decrease in cell
viability and proliferation occurred later than caspase 3/7
activation. The apoptotic potential of CV-45 was also evident in
the formation of apoptotic bodies that were observed with
fluorescence microscopy after DAPI staining. Nucleic aberrations,
manifested in chromatin condensation and nucleic shrinking, were
determined in MTC-SK as well as in KRJ-I cells,
confirming the apoptosis inducing effect of Christia
vespertilionis plant extracts.
The object of the present study was also to examine
the effects of CV-45 treatment on the relative gene expression of
MTDH, PDCD5 and TNFRSF10b in MTC-SK and
KRJ-I cells. We found these genes to be expressed in our
cell lines and their expression was altered after treatment.
Proteins encoded by the genes PDCD5, MTDH and
TNFRSF10b are known to be involved in programmed cell death.
PDCD5 protein is believed to participate in regulation of
apoptosis. Its expression was downregulated in gastric tumor tissue
as compared to normal gastric tissue. However, when treated with
diallyl trisulfide, which induced apoptosis in gastric cancer,
PDCD5 expression was upregulated (22). In KRJ-I cells, PDCD5
gene expression was also upregulated with 3 h of treatment with
CV-45, in agreement with literature data. Unlike gene expression in
KRJ-I, PDCD5 gene expression was downregulated in
MTC-SK with 2 h stimulation with apoptosis inducing CV-45.
This result could possibly be related to insufficient incubation
time with CV-45 in these cells. Altered expression of PDCD5
in MTC-SK occurred also in MTC-SK treated for a
longer time with CV-45. MTDH is known to have a multifaceted
role in cancer progression, taking part in the complex network of
oncogenic signaling pathways. It is involved in the processes of
proliferation, metastasis, survival, chemoresistance and invasion
of tumor cells. Furthermore, it interacts with NF-κB by binding
directly to the p65 subunit of the necrosis factor (23).
The upregulation of MTDH in CV-45 treated
MTC-SK cells, might lead to the apoptotic effect by the
activation of NF-κB. NF-κB on its part is known for its
anti-apoptotic activity, acting as transcription factor for cell
survival genes. In contrast MTDH was downregulated in
KRJ-I. A possible explanation could be that downregulation
of MTDH leads to less NF-κB activation and further to an
inhibition of the expression of cell survival genes involved in
cell death. In the present study, the apoptotic potential of CV-45
was demonstrated via caspase 3/7 activation and fluorescence
staining in both MTC-SK and KRJ-I cell lines. Besides
inhibition of MTDH, we suppose the presence of another
pathway for CV-45 to activate apoptosis in MTC-SK. Hu et
al(23) also described a
connection between MTDH and tumor necrosis factor-related
apoptosis-inducing ligand (TRAIL), which is known to
activate TRAIL induced apoptosis via the following pathway: TRAIL
binds to TNFRSF10b and activates further proteins, known as FADD
and initiator caspase 8, which thereupon activate effector caspases
3 and 7 and finally apoptosis.
Hu et al explained that overexpressed
MTDH leads to downregulation of TRAIL. Thus,
decreased TRAIL could possibly mean less TRAIL-induced apoptosis,
suggesting that this probably does not occur in CV-45 treated
MTC-SK cells. However, knowing that many proteins, also ones
yet unknown, are involved in the complex pathway of apoptosis,
explanations can only be vague and speculative. The protein encoded
by TNFRSF10b gene is a receptor that is also known as death
receptor 5 (DR 5), containing a death domain (DD). Some parts of
this receptor interact with TRAIL (24), whereas FADD binds with its DD at the
DD of TNFRSF10b resulting in formation of the death-inducing
signaling complex (DISC). Further, it recruits caspase 8 to DISC,
engaging the caspase cascade to induce apoptosis (25). It seems that TNFRSF10b is
selectively expressed in cancer cells and may induce TRAIL-induced
apoptosis in cancer cells (26).
Overexpression of TNFRSF10b led to
significant activation of NF-κB in human embryonic kidney cells
(HEK 293). Thus, TNFRSF10b was involved in both induction of
apoptosis and activation of NF-κB (27). The downregulation of
TNFRSF10b in CV-45 treated MTC-SK cells remains to be
clarified. In KRJ-I cells, TNFRSF10b gene expression
was not significantly altered when compared to control cells.
Chaudhary et al showed that overexpression of
TNFRSF10b induced apoptosis in mammalian cells via a
caspase-dependent mechanism (27).
Although caspases 3 and 7 were activated after CV-45 treatment in
MTC-SK and KRJ-I cells, there was no significant
upregulation of TNFRSF10b, but quite the opposite. These
results lead to the conclusion that apoptosis might not be induced
by the TRAIL/TNFRSF10b mechanism.
The present in vitro study demonstrates the
antitumor effects of novel plant derived agents in human
neuroendocrine tumor cells. The ethyl acetate fraction of
Christia vespertilionis (CV-45) had an antiproliferative and
pro-apoptotic effect in MTC cells as well as in SI-NET cells.
Fibroblasts were not impaired, indicating a lack of side effects.
The in vitro effects of the selected plant-derived compounds
suggest potential clinical effects in patients with neuroendocrine
tumors.
Acknowledgements
The authors thank Eugenia Lamont for editing the
manuscript. This work was supported by the Jubilee Fund of the
Austrian National Bank (Project no. 14394), The Franz Lanyar
Foundation (Project no. 357), Stadt Graz Wissenschaft, and Land
Steiermark Wissenschaft and Forschung.
References
|
1
|
Taal BG and Visser O: Epidemiology of
neuroendocrine tumors. Neuroendocrinology. 80(Suppl 1): 3–7. 2004.
View Article : Google Scholar
|
|
2
|
Saad MF, Ordonez NG, Rashid RK, Guido JJ,
Hill CS Jr, Hickey RC and Samaan NA: Medullary carcinoma of the
thyroid. A study of the clinical features and prognostic factors in
161 patients. Medicine. 63:319–342. 1984.PubMed/NCBI
|
|
3
|
Vitale G, Caraglia M, Ciccarelli A and
Lupoli G, Abbruzzese A, Tagliaferri P and Lupoli G: Current
approaches and perspectives in the therapy of medullary thyroid
carcinoma. Cancer. 91:1797–1808. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Eng C and Mulligan LM: Mutations of the
RET proto-oncogene in the multiple endocrine neoplasia type 2
syndromes, related sporadic tumors, and Hirschsprung disease. Hum
Mutat. 9:97–109. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Modlin IM, Kidd M, Latich I, Zikusoka MN
and Shapiro MD: Current status of gastrointestinal carcinoids.
Gastroenterology. 128:1717–1751. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Scheuba C, Kaserer K, Weinhausl A, Pandev
R, Kaider A, Passler C, Prager G, Vierhapper H, Haas OA and
Niederle B: Is medullary thyroid cancer predictable? A prospective
study of 86 patients with abnormal pentagastrin tests. Surgery.
126:1089–1095. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Orlandi F, Caraci P, Mussa A, Saggiorata
E, Pancani G and Angeli A: Treatment of medullary thyroid
carcinoma: an update. Endocr Relat Cancer. 8:135–147. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Modlin IM, Latich I, Kidd M, Zikusoka M
and Eick G: Therapeutic options for gastrointestinal carcinoids.
Clin Gastroenterol Hepatol. 4:526–547. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li ZX, Sturm S, Stuppner H, Schraml E,
Aguiriano Moser V, Siegl V and Pfragner R: The dichloromethane
fraction of Stemona tuberosae Lour inhibits tumor cell
growth and induces apoptosis of human medullary thyroid carcinoma
cells. Biologics. 1:455–463. 2007.
|
|
10
|
Li ZX, Sturm S, Svejda B, Höger H, Schraml
E, Ingolic E, Siegl V, Stuppner H and Pfragner R: Anticancer
activity of novel extracts from Cautleya gracilis (Smith)
Dandy: Apoptosis in human medullary thyroid carcinoma cells.
Anticancer Res. 28:2705–2714. 2008.PubMed/NCBI
|
|
11
|
Rinner B, Li ZX, Haas H, Siegl V, Sturm S,
Stuppner H and Pfragner R: Antiproliferative effects of Uncaria
tomentosa in human medullary thyroid carcinoma cell. Anticancer
Res. 29:4519–4528. 2009.PubMed/NCBI
|
|
12
|
Nguyen-Pouplin J, Tran Ho, Tran Hu, Phan
TA, Dolecek C, Farrar J, Tran TH, Caron P, Bodo B and Grellier P:
Antimalarial and cytotoxic activities of ethnopharmacologically
selected medicinal plants from South Vietnam. J Ethnopharmacol.
109:417–427. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Strasser M: Phytochemische Untersuchung
von Christia vespertilionis. Diploma thesis. University of
Innsbruck; 2010
|
|
14
|
Pfragner R, Höfler H, Behmel A, Ingolic E
and Walser V: Establishment and characterization of continuous cell
line MTC-SK derived from a human medullary thyroid carcinoma.
Cancer Res. 50:4160–4166. 1990.PubMed/NCBI
|
|
15
|
Pfragner R, Wirnsberger G, Niederle B,
Behmel A, Rinner I, Mandl A, Wawrina F, Luo J-S, Adamiker D, Höger
H, Ingolic E and Schauenstein K: Establishment of a continuous cell
line from a human carcinoid of the small intestine (KRJ-I).
Int J Oncol. 8:513–520. 1996.PubMed/NCBI
|
|
16
|
Pfaffl MW: A new mathematical model for
relative quantification in real-time RT-PCR. Nucleic Acids Res.
29:2002–2007. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Da Violante G, Zerrouk N, Richard I,
Provot G, Chaumeil JC and Arnaud P: Evaluation of the cytotoxicity
of dimethyl sulfoxide (DMSO) on Caco2/TC7 colon tumor cell
cultures. Biol Pharm Bull. 25:1600–1603. 2002.PubMed/NCBI
|
|
18
|
Li ZD, Liu Y, Liao Y and Zuo GQ: Sorafenib
and octreotide combination therapy can inhibit proliferation of and
induce apoptosis in human hepatoma cells. Zhonghua Gan Zang Bing Za
Zhi. 20:126–130. 2012.(in Chinese).
|
|
19
|
Cao Z, Liao L, Chen X, Lan L, Hu H, Liu Z,
Chen L, Huang S and Du J: Enhancement of antitumor activity of
low-dose 5-fluorouracil by combination with fuzheng-yiliu granules
in hepatoma 22 tumor-bearing mice. Integr Cancer Ther. Jul
12–2012.(Epub ahead of print).
|
|
20
|
Yang KP, Liang YF and Samaan NA: Intrinsic
drug resistance in a human medullary thyroid carcinoma cell line:
association with overexpression of mdr1 gene and low proliferation
fraction. Anticancer Res. 11:1065–1068. 1991.PubMed/NCBI
|
|
21
|
Carlomagno F, Salvatore D, Santoro M, de
Franciscis V, Quadro L, Panariello L, Colantuoni V and Fusco A:
Point mutation of the RET protooncogene in the TT human
medullary thyroid carcinoma cell line. Biochem Biophys Res Commun.
207:1022–1028. 1995.PubMed/NCBI
|
|
22
|
Yang YH, Zhao M, Li WM, Chen YY, Kang B
and Lu YY: Expression of programmed cell death 5 gene involves in
regulation of apoptosis in gastric tumor cells. Apoptosis.
11:993–1001. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Hu G, Wei Y and Kang Y: The multifaceted
role of MTDH/AEG-1 in cancer progression. Clin Cancer Res.
15:5615–5620. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhuang L, Lee CS, Scolyer RA, McCarthy SW,
Zhang XD, Thompson JF, Screaton G and Hersey P: Progression in
melanoma is associated with decreased expression of death receptors
for tumor necrosis factor-related apoptosis-inducing ligand. Hum
Pathol. 37:1286–1294. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Thomas LR, Bender LM, Morgan MJ and
Thorburn A: Extensive regions of the FADD death domain are required
for binding to the TRAIL receptor DR5. Cell Death Differ.
13:160–162. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sheikh MS, Burns TF, Huang Y, Wu GS,
Amundson S, Brooks KS, Fornace AJ Jr and El-Deiry WS: p53-dependent
and -independent regulation of the death receptor KILLER/DR5 gene
expression in response to genotoxic stress and tumor necrosis
factor alpha. Cancer Res. 58:1593–1598. 1998.PubMed/NCBI
|
|
27
|
Chaudhary PM, Eby M, Jasmin A, Bookwalter
A, Murray J and Hood L: Death receptor 5, a new member of the TNFR
family, and DR4 induce FADD-dependent apoptosis and activate the
NFκB pathway. Immunity. 7:821–830. 1997.PubMed/NCBI
|