Introduction
Gastric cancer is a disease with one of the poorest
prognoses, and is the second leading cause of tumor-related
mortality worldwide. The 5-year overall survival is 25% or less,
particularly in the USA, Europe and China (1,2). Every
year, 1 million new cases of gastric cancer are diagnosed and
700,000 individuals succumb to this disease worldwide (3). Most patients with gastric cancer are
diagnosed with advanced gastric cancer, and the overall survival
rate remains poor. There is a need for new prognostic tumor markers
that provide more effective therapeutic targets for gastric
cancer.
Resistin-like molecule (RELM)-α belongs to the RELM
family which are potent innate immune-modulating molecules which
are implicated in Th2-associated mucosal immune responses (4). RELM-α was originally identified in
inflammatory zones in an experimental allergic airway disease model
and was therefore also termed found in inflammatory zone 1 (FIZZ1)
(5,6). Moreover, related studies found that
RELM-α is a hallmark signature gene of activated macrophages.
RELM-α expression is tightly regulated by IL-13, IL-4 and signal
transducer and activator of transcription protein signaling pathway
(7). RELM-α also has a key
functions in fibrosis in the setting of experimental asthma
(8). Notably, RELM-α was identified
in the gastrointestinal tract and is strongly linked with the
induction of Th2 immune responses and mucosal immunity, involving
inflammatory bowel disease (9).
Accumulating the evidence also indicates that RELM-α is involved in
the angiogenesis of endothelial cells and induces vascular
remodeling (10). Yet, RELM-α
expression in gastric cancer and its correlation with gastric
cancer clinicopathological characteristics remain unclear.
In the present study, we first examined RELM-α
expression in 92 paired cases of gastric cancer and adjacent
non-cancerous mucosa tissues and in SGC7901 and MKN45 gastric
cancer cell lines to investigate the relevance of RELM-α expression
in gastric cancer and its functional mechanism. In addition, an
in vitro study was performed to observe the silencing effect
of RELM-α on gastric cancer cells, to investigated its effective
mechanism in order to determine its use as a potential therapeutic
target.
Materials and methods
Patients and specimens
Ninety-two cases of gastric cancer and adjacent
non-cancerous tissues were collected at Ruijin Hospital and Ruijin
Hospital North, Shanghai Jiaotong University School of Medicine
from April 2007 to February 2013. Tissue samples for diagnostic
purposes were obtained with the consent of each patient. All tumor
specimens and corresponding adjacent non-cancerous tissues were
fixed in 10% buffered formalin, embedded in paraffin and then made
into continuous 4-μm tissue sections for examination. The
study group consisted of 52 males and 40 females, aged 18–92 years,
with an average age of 61±14 years. Each specimen was analyzed by
routine pathological analysis and was classified according to the
pathological criteria published by the World Health organization
(4th edition) and the tumor-node-metastasis (TNM) staging system of
the American Joint Committee on Cancer Staging Manual (7th edition)
and the Japanese Gastric Cancer Association Guidelines (3rd
edition). None of the gastric cancer patients had synchronous
cancers or previous gastrointestinal diseases, nor had undergone
abdominal surgery, chemotherapy or radiotherapy prior to specimen
collection. The present study was performed with pre-approval from
the ethics committees of the participating hospitals.
Immunohistochemistry
Gastric cancer tissue paraffin sections were placed
in citrate buffer (pH 6.0) for antigen retrieval. The negative
control antibody was replaced by phosphate-buffered saline (PBS).
The procedure was in accordance with the SP detection instructions
(Maixin Co., Fujian, China). Paraffin sections (4-μm) were
used for histological staining. The diluted density of rabbit
anti-human RELM-α, vascular endothelial growth factor (VEGF), CD34
antibody (Santa Cruz Biotechnology, Santa Cruz, CA, USA) as the
primary antibodies was 1:50. Thereafter, each section was incubated
with the horseradish peroxidase (HRP)-labeled anti-rabbit IgG
antibody (Maixin Co.). The immunostained specimens were assessed by
two independent observers without prior knowledge of the
clinicopathological characteristics.
The RELM-α or VEGF protein positive expression was
mainly localized in the cytoplasm of gastric cancer tissue. The
staining intensity score was 0 (negative), 1 (weak), 2 (medium) and
3 (strong). The integral of the rate of positive cells was 0 (0%),
1 (1-25%), 2 (26-50%), 3 (51-75%) and 4 (76-100%). The proportional
score and the intensity score were then added to obtain a total
score. A score ≥3 was considered to indicate positive
expression.
Microvessel density (MVD) was assessed according to
the criteria proposed by Weidner et al (11); the brownish cytoplasmic staining of
vascular endothelial cells with CD34 antibody was considered as
positive. The microvessels were counted according to the number of
single endothelial cells or endothelial cell clusters showing
brownish yellow granules in the cytoplasm. The sections were
observed first under low-power magnification (×40), and then the
most dense area of microvessel sections was selected under
high-power magnification (×400; the surface area of every vision
field being 0.785 mm2). The number of microvessels in
three vision fields was counted and averaged as MVD of the given
specimen.
In vitro
Cell culture
Two gastric cancer cell lines (MKN45 and SGC7901)
and the 293 cell line were obtained from Ruijin Hospital, Shanghai
Jiaotong University School of Medicine. Cells were cultured in
RPMI-1640 medium supplemented with 10% fetal bovine serum (FBS)
(Gibco-BRL, Gaithersburg, MD, USA) and incubated in 5%
CO2 at 37°C in 95% humidity.
Construction of RELM-α-siRNA
The specific silencing of the human RELM-α gene
expression was achieved by the siRNA technique.
The study used replication defective adenovirus
serotype 5/F35 (Ad5/F35) as the vector. Ad5/F35-RELM-α-siRNA was
constructed by Hongming Bio. (Shanghai, China) using a previously
described method (12).
RELM-α-siRNA was chemically synthesized, and subcloned into the
PDC316-MCMV-EGFP transfer plasmid (Mierobix Biosystems Co.,
Mississauga, ON, Canada). This plasmid was cotransfected into 293
cells, along with a fragment of the plasmid containing the Ad5/F35
adenoviral vector (Bengyuanzhengyang Bio., Beijing, China).
Additionally, an Ad5/F35 containing an empty expression cassette
was constructed for use as a control (Ad5/F35-vector). All of the
viral constructs were similar with the exception of the trans-gene,
and the production and purification procedures were identical. The
SGC7901 and MKN45 cells at 50-60% confluency in a total volume of
500 ml/well growth medium, were randomly allocated into 3 groups:
cells treated with PBS served as control cells; cells treated with
Ad5/F35-RELM-α-siRNA at a multiplicity of infection (MOI) of 10;
and cells treated with the Ad5/F35-vector (MOI of 5) for 24 h for
further examination.
RELM-α expression in cells
Total RNA was isolated from the cells using TRIzol
reagent kits (Gibco-BRL, Rockville, MD, USA). The cDNA obtained
from this reaction was mixed with PCR buffer, MgCl2,
dNTPs, Taq DNA polymerase and human RELM-α gene-specific
primers [the primer sequences of RELM-α: forward primer, 5′-GCT GGG
ATG ACT GCT ACT GG-3′ and reverse primer, 5′-AAG CGG GGT TAA TGG
GCA AT-3′ (synthesis by Sangon Biotech Co., Shanghai, China)] and
amplified in an automated thermal cycler (Bio-Rad MJ Mini; Bio-Rad
Laboratories, Inc., Hercules, CA, USA). The conditions of RT-PCR
were as follows: 1 cycle for 5 min at 95°C, 35 cycles for 45 sec at
94°C, for 45 sec at 55°C, for 1 min at 72°C, and 1 cycle for 10 min
at 72°C. The PCR products were separated by electrophoresis on 1.2%
agarose gels and stained with ethidium bromide. The densities of
the cDNA bands were analyzed by scanning densitometry using GelDoc
2000 software (Bio-Rad Laboratories, Inc.). The band densities were
normalized to GAPDH (the primer sequence of GAPDH: forward primer,
5′-GG C TGAGAA CGG GAA GCT TGT C-3′ and reverse primer, 5′-CAGCCT
TCT CCA TGG TGG TGA AGA-3′; synthesis by Sangon Biotech Co.) band
densities, and the results are expressed as ratios.
Cell viability assay
The effect of RELM-α silencing on gastric cancer
cell viability was monitored using the
3-(4,5-dimethyl-thiazolyl-2)-2,5-diphenyltetrazolium bromide (MTT)
(Sigma, St. Louis, MO, USA) colorimetric assay. In brief, a total
of 2×103 cells were seeded in 96-well plates. After 24
h, the cells were treated with Ad5/F35-RELM-α-siRNA (MOI of 10) or
transfected with the Ad5/F35 vector (MOI of 5). After 24 h, 20
μl MTT (5 mg/ml) was added to each well. Four hours later,
100 μl of dimethyl sulfoxide was added to each well after
the medium was removed. Finally, the absorbance (A) was detected
with an enzyme calibrator at 570 nm, The cell viability = (A of
study group/A of control group) × 100%.
Scratch wound-healing assay
To measure cell motility, 4×105 cells
were seeded into 6-well plates. A central linear wound was created
by scraping the cell monolayer with a 200-μl sterile pipette
tip. The media were carefully changed to remove any floating cells
and cultured in 5% CO2 at 37°C. The migration of cells
into the denuded areas in the scraped region was calculated at 48
h, respectively. The wound at 0 h was considered 100% of the
average gap.
Cell invasion assays
Invasion assays were performed using Transwell
chambers (Corning Inc., Corning, NY, USA). Briefly,
2×106 cells/dish were seeded in a 10-cm dish overnight
in RPMI-1640 supplemented with 10% FBS. Then, the medium was
replaced with serum-free RPMI-1640 for 12 h, and the cells were
treated with or without Ad5/F35-RELM-α-siRNA for 2 h in 10% FBS
medium. The cells were collected and centrifuged at 500 × g for 3
min. The treated cells were seeded at a density of 1×105
cells/well in 10% FBS medium in the upper chamber. The lower
chamber was filled with complete medium. The cells were allowed to
migrate for 24 h at 37°C. After the incubation, the cells that did
not migrate, which remained on the top surface of the membrane,
were removed with a cotton swab. The cell that migrated to the
bottom side of the membrane were fixed in cold 75% methanol for 15
min and washed three times with PBS. Next, the cells were stained
with Giemsa staining solution (Sigma) and then destained with PBS.
The images were captured using an optical microscope (Nikon Co.,
Tokyo, Japan; magnification, ×200), and invading cells were
quantified by manual counting.
Western blot analysis
The levels of nuclear factor (NF)-κB p65, vascular
endothelial growth factor (VEGF) and matrix metallopeptidase
(MMP)-9 proteins were investigated in each group using western
blotting. The cells were washed twice with PBS and then homogenized
in RIPA buffer (Biyuntian Co., Shanghai, China). Following
centrifugation at 12,000 × g at 4°C for 10 min, the supernatant was
collected and stored at 80°C. Protein concentration of each sample
was determined by the BCA protein assay (Biyuntian Co.). Each
sample was adjusted to a desired protein content of 40 μg,
then denatured in loading buffer and separated by electrophoresis
on 9% SDS polyacrylamide gel at 100 V for 120 min. The separated
proteins were transferred to polyvinylidene difluoride membranes
using transfer buffer at 200 mA for 90 min. The membranes were
blocked with 5% non-fat dry milk in Tris-buffered saline (TBS)-0.1%
Tween for 1 h at room temperature, washed three times for 10 min
each in TBS-0.1% Tween, and incubated with a primary antibody
including NF-κB p65, VEGF and MMP-9 (Santa Cruz Biotechnology) at a
1:1,000 dilution in TBS-0.1% Tween overnight at 4°C, respectively.
After three 10-min washings in TBS-0.1% Tween, the membranes were
incubated with a secondary antibody, HRP-conjugated rat anti-rabbit
immunoglobulin G (Kangcheng Inc., Shanghai, China) for 1 h at room
temperature. After washing, the membranes were detected using
enhanced chemiluminescence methods (Amersham Biosciences,
Piscataway, NJ, USA), and then were scanned for densitometry using
Bio-Image Analysis System (Bio-Rad Laboratories, Inc.). For
quantification, GAPDH was determined in a similar manner with the
anti-GAPDH antibody (diluted to 1:1,000; Santa Cruz Biotechnology)
as an endogenous control for other proteins.
Electrophoretic and mobility shift
assay (EMSA)
The activity of NF-κB of cells was examined by EMSA
reagent kits (Pierce Chemical Co., Rockford, IL, USA). Nuclear
extracts of cells were prepared from gastric cancer cells at 24 h
after stimulation. The biotinylated double-stranded DNA probe
sequence of NF-κB was: sense, 5′-AGT TGA GGG GAC TTT CCC AGG C-3′
and antisense, 3′-TCA ACT CCC CTG AAA GGG TCC G-5′.
A total of 5 μg of nuclear extract was
incubated with 5 ng of biotinylated double-stranded
oligonucleotides in 20 μl of reaction mixture containing
binding buffer, 2.5% glycerol, 0.05% NP-40 and 50 μg/ml
poly(dI:dC) (Biyuntian Co.). After 20 min at room temperature, the
reaction was terminated by addition of 2 μl of 10X loading
buffer. The mixture was run on 6% polyacrylamide gel. DNA was
transferred to a membrane, followed by detection using streptavidin
HRP-conjugated and chemiluminescence substrate from Pierce Chemical
Co.
Statistical analysis
All statistical analyses were carried out using the
SPSS software statistical package (version 13.0; SPSS, Inc.,
Chicago, IL, USA). The relationships between the differential
expression of RELM-α and clinicopathological characteristics were
evaluated by the χ2 test. The one-way analysis of
variance (ANOVA) with Dunnett’s multiple comparison tests was used
for comparisons. A P-value <0.05 was considered to indicate a
statistically significant result.
Results
RELM-α expression in the gastric cancer
cases
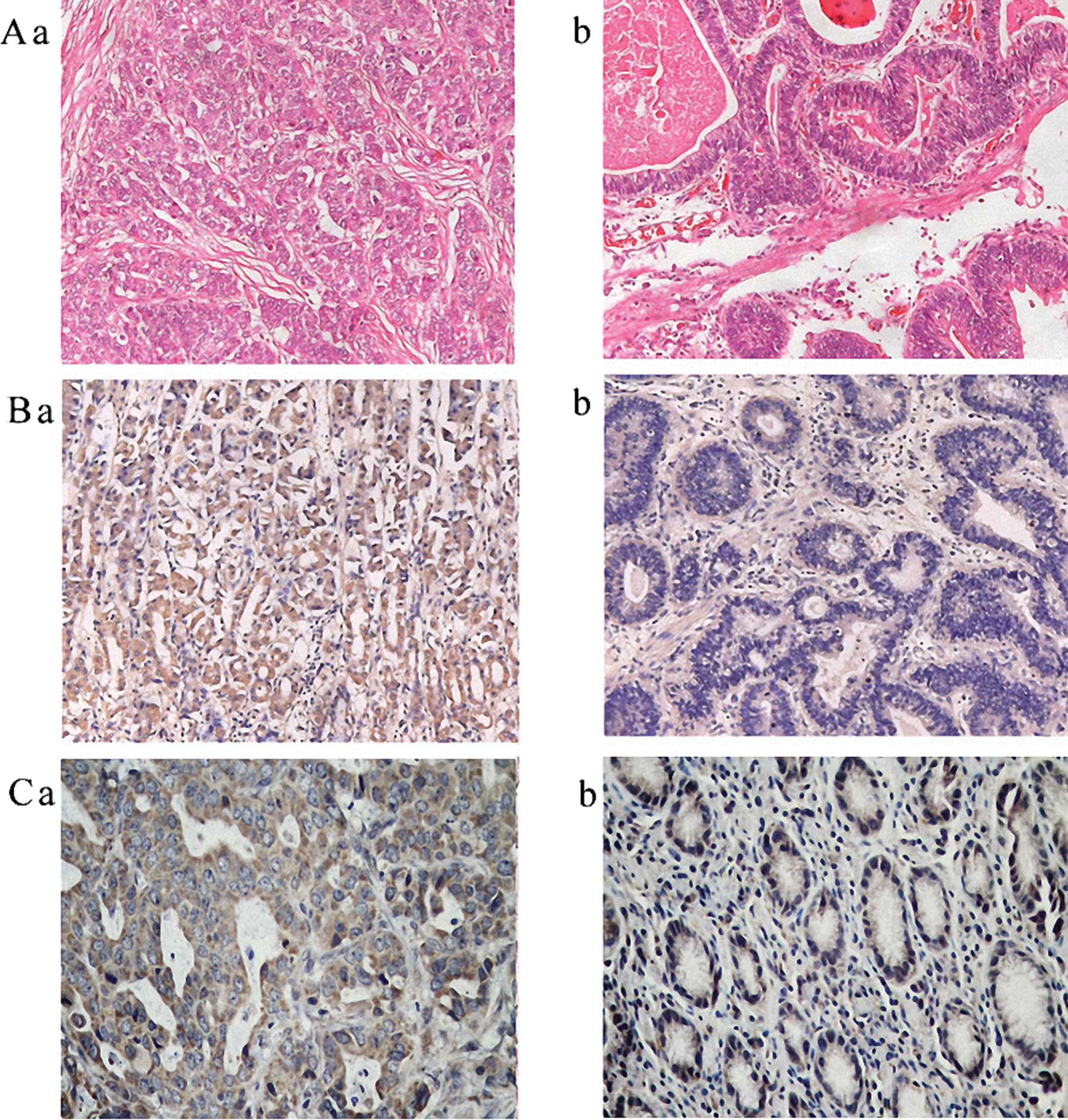
Expression of RELM-α and VEGF showed a predominant
localization to the cytoplasm or on the cell membrane in the tissue
specimens. Specimens with positive expression are presented in
Fig. 1. The results showed that
positive expression of RELM-α was found in 64.1% (59/92) of the
gastric cancer tissues compared with 16.3% (15/92) of the adjacent
normal tissues.
Among the 92 cases of gastric cancer, 59 (64.1%)
cases had positive expression and 33 (35.8%) cases had negative
expression of RELM-α; which showed a statistically significant
difference (P<0.05). Moroever, among the gastric cancer cases,
77 (83.6%) cases had positive expression and 15 (16.4%) cases had
negative expression of VEGF; which achieved a statistically
significant difference (P<0.05).
Furthermore, the clinical relevance was confirmed by
the observation that RELM-α expression was correlated with the
prognosis in gastric cancer (Table
I). Tumor stage and size were significantly associated with
high expression of RELM-α protein (P<0.05). No correlation was
observed between RELM-α protein expression and age, gender, degree
of differentiation, and gross classification of tumors (P>0.05).
In addition, the gastric cancer cases with positive expression of
RELM-α and VEGF (48/92, 52.1%) were compared with those having
negative expression of RELM-α and VEGF (4/92, 4.3%), which showed a
difference and thus may be correlated with each other (Table II).
 | Table IRelationship of RELM-α and VEGF
expression with clinicopathological parameters in the patients with
gastric cancer. |
Table I
Relationship of RELM-α and VEGF
expression with clinicopathological parameters in the patients with
gastric cancer.
| Total | Positive expression
of RELM-α (%, cases) | P-value | Positive expression
of VEGF (%, cases) | P-value |
|---|
| Total cases | 92 | | | | |
| Age (years) |
| ≥55 | 58 | 68.9 (40/58) | 0.207 | 81.0 (47/58) | 0.367 |
| <55 | 34 | 55.8 (19/34) | | 88.2 (30/34) | |
| Gender |
| Male | 52 | 55.7 (29/52) | 0.057 | 76.9 (40/52) | 0.607 |
| Female | 40 | 75.0 (30/40) | | 92.5 (37/40) | |
| Stage |
| T2 | 47 | 48.9 (23/47) | 0.020 | 80.8 (38/47) | 0.450 |
| T3, T4 | 45 | 86.8 (36/45) | | 86.6 (39/45) | |
| Size (cm) |
| ≥5 | 61 | 85.2 (52/61) | 0.000 | 90.1 (55/61) | 0.018 |
| <5 | 31 | 22.5 (7/31) | | 70.9 (22/31) | |
|
Differentiation |
| Undifferentiated,
poorly differentiated | 57 | 64.9 (37/57) | 0.842 | 87.7 (50/57) | 0.182 |
| Moderately, well
differentiated | 35 | 62.8 (22/35) | | 77.1 (27/35) | |
| Gross
classification |
| Early gastric
cancer | 5 | 60.0 (3/5) | 0.250 | 80.0 (4/5) | 0.670 |
| Borrmann I | 27 | 51.8 (14/27) | | 88.8 (24/27) | |
| Borrmann II,
III | 49 | 73.4 (36/49) | | 83.6 (41/49) | |
| Borrmann IV | 11 | 54.5 (6/11) | | 72.2 (8/11) | |
 | Table IIRelationship between expression of
RELM-α and VEGF with tumor stage, size and MVD in patients with
gastric cancer. |
Table II
Relationship between expression of
RELM-α and VEGF with tumor stage, size and MVD in patients with
gastric cancer.
| RELM-α | VEGF | Cases (n=92) | MVD | Stage (T3, T4) | Size (≥5 cm) |
|---|
| + | + | 48 | 23.77±6.33 | 30 | 40 |
| + | − | 11 | 14.15±5.24a | 2a | 5a |
| − | + | 29 | 8.39±4.18a | 10a | 15a |
| − | − | 4 | 7.80±2.28a | 3a | 1a |
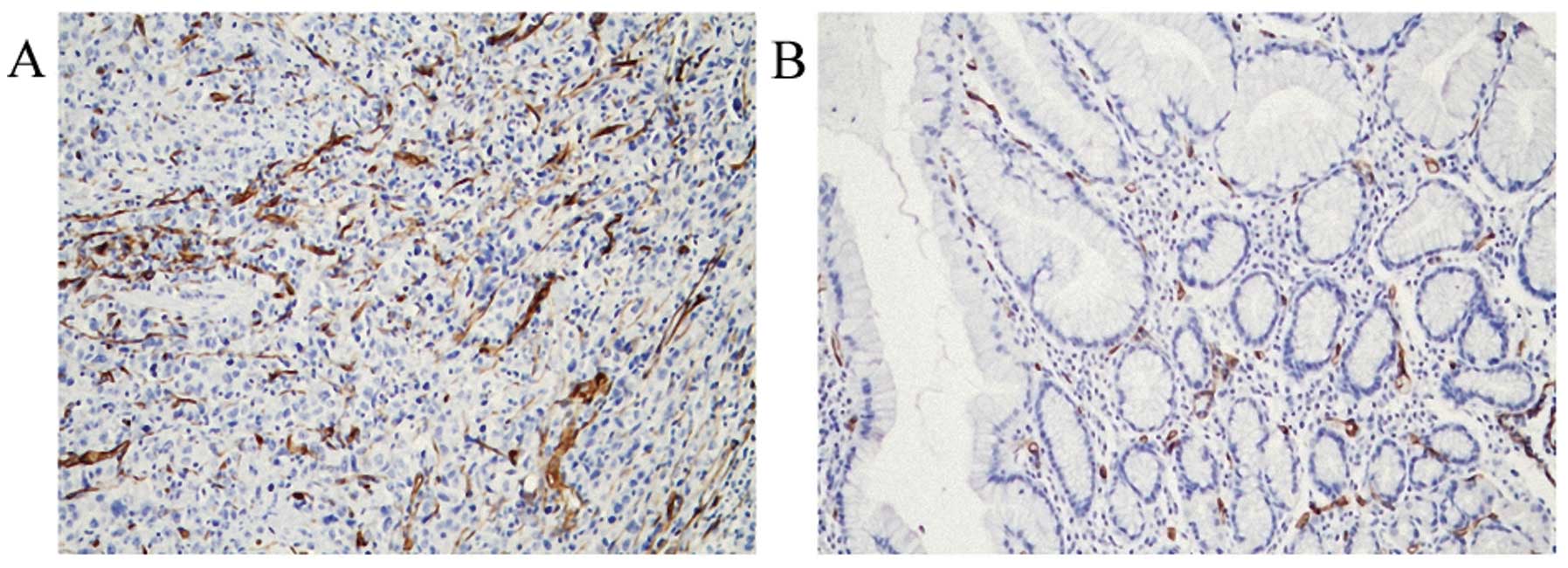
As shown in Fig. 2,
the MVD of tumors was determined by CD34 staining. The MVD value of
the 92 gastric cancer specimens was 17.23±8.94, while the MVD value
in the 92 adjacent normal tissues was 6.58±2.32, showing a
statistically significant difference between the gastric cancer and
adjacent normal tissues (P<0.05).
In the 59 gastric cancer cases with positive RELM-α
expression and the 33 cases with negative of RELM-α expression, the
MVD was 22.23±6.79 and 8.30±3.93, respectively, which showed a
statistically significant difference (P<0.05). Meanwhile, in the
77 gastric cancer cases with positive VEGF expression and the 15
cases with negative VEGF expression, the MVD was 19.12±8.49 and
7.53±2.74, respectively, which showed a statistically significant
difference (P<0.05).
The tumor size, tumor stage and MVD value in the
RELM-α-positive/VEGF-positive gastric cancer patients were found to
be higher when compared with these parameters in the
RELM-α-negative/VEGF-negative gastric cancer patients (P<0.01)
(Table II).
In vitro
Effect of the silencing of RELM-α
expression on gastric cancer cells
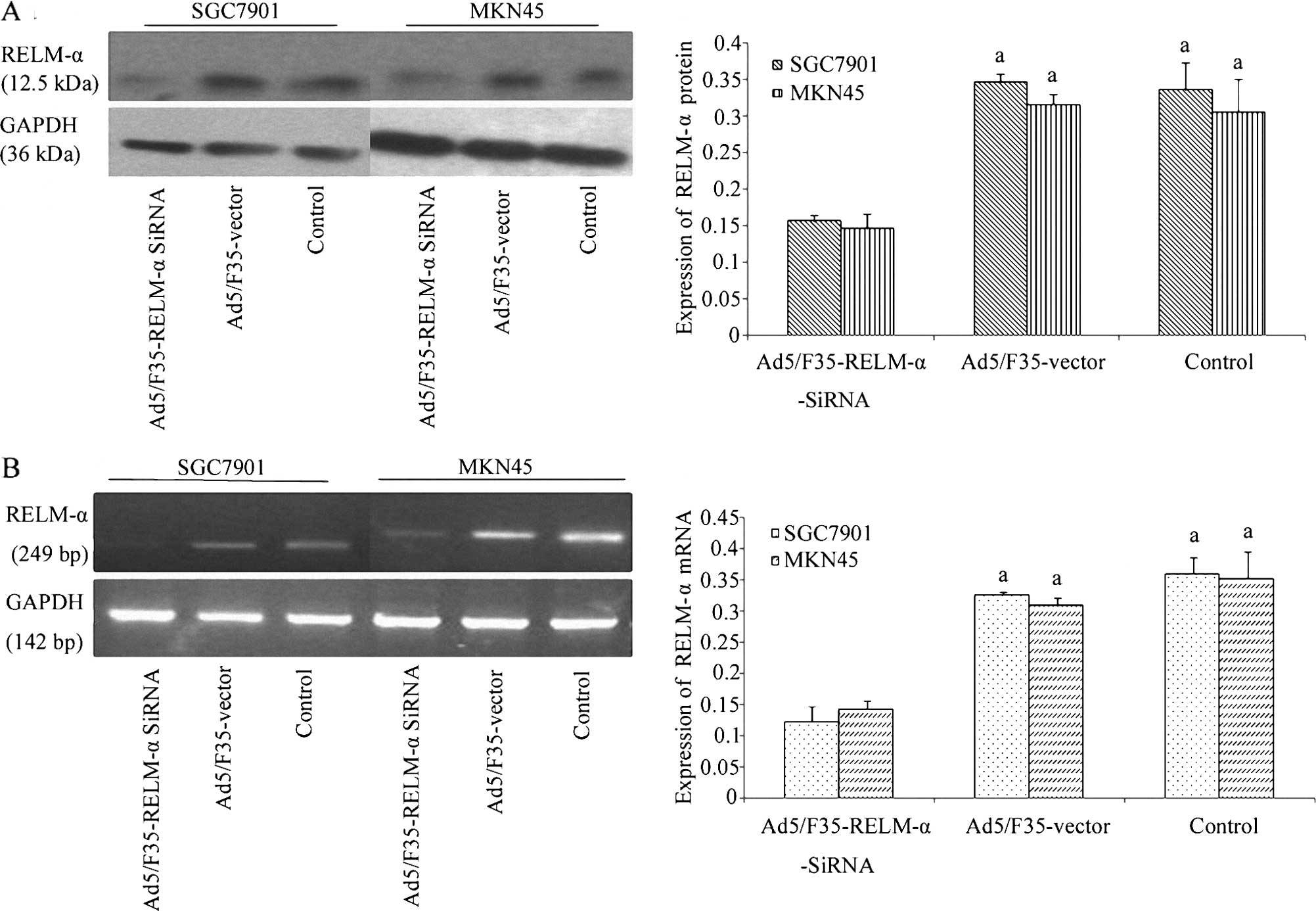
The levels of RELM-α expression in the SGC7901 and
MKN45 cells were assessed by western blotting and RT-PCR assays
methods (Fig. 3). The data showed
that at 24 h following Ad5/F35-RELM-α-siRNA treatment, RELM-α
protein levels were decreased in the SGC7901 and MKN45 cells, yet
not in the cells treated with the Ad5/F35-vector and the control
cells (P<0.01) (Fig. 3A).
Similar to the western blotting data, the SGC7901 and MKN45 cells
with Ad5/F35-RELM-α-siRNA treatment expressed a lower level of
RELM-α mRNA compared with the cells treated with the Ad5/F35 vector
and the control cells (P<0.01) (Fig.
3B).
Effects of RELM-α silencing on cell
proliferation
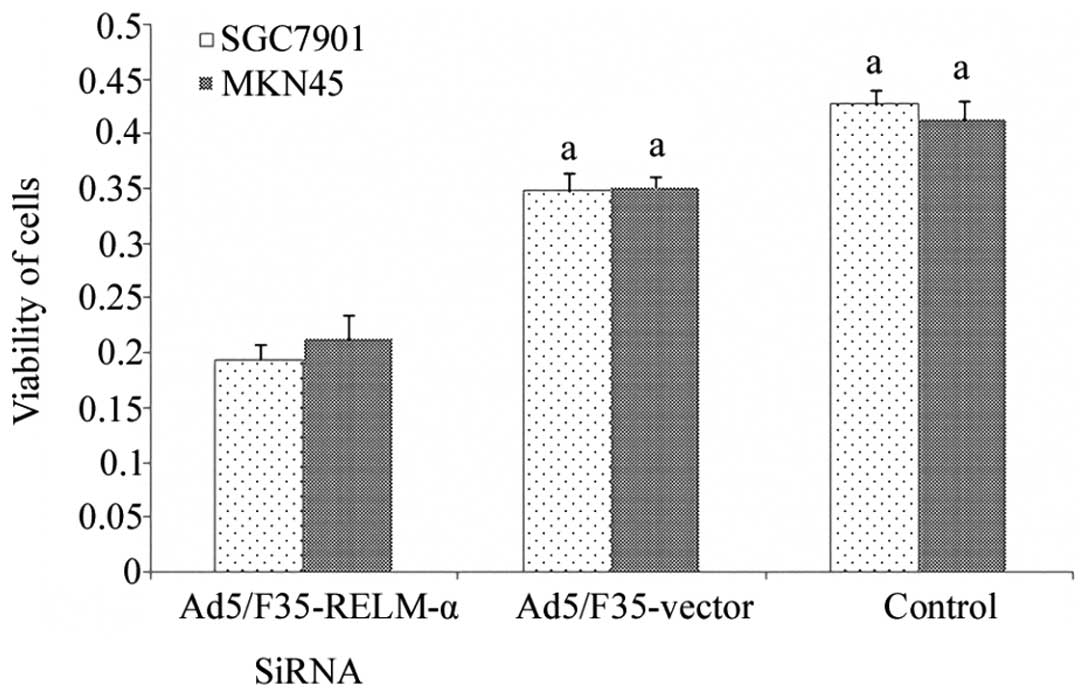
We aimed to assess whether RELM-α silencing
suppressed the growth of gastric cancer cells (Fig. 4). The results showed that the
percentage of viable cells following Ad5/F35-RELM-α-siRNA treatment
for 24 h was reduced in the two gastric cancer cell lines when
compared with the control and Ad5/F35 vector-treated cells as
determined by MTT assay (P<0.01).
Furthermore, to examine the effect of RELM-α
silencing on cell motility, an in vitro scratch
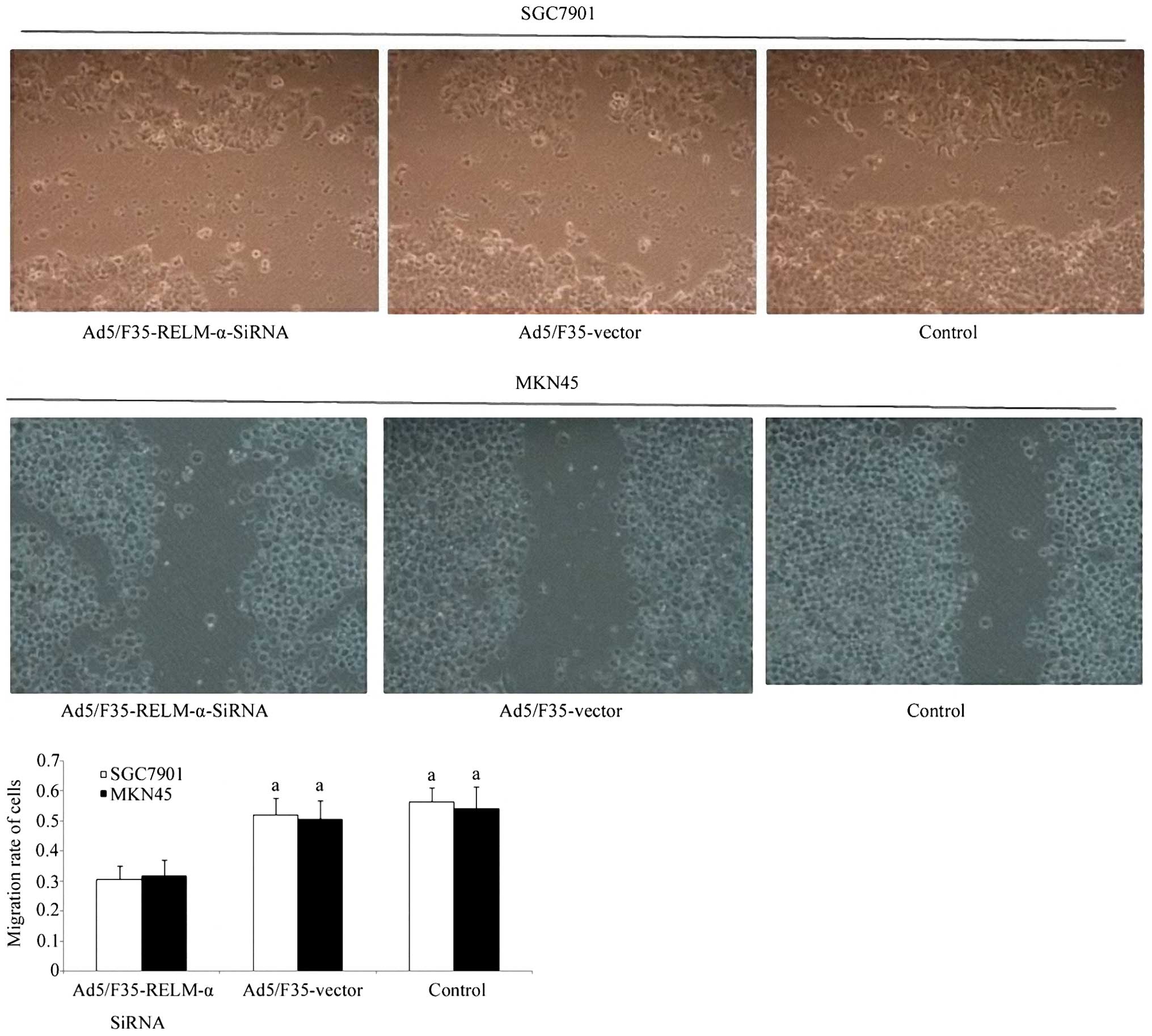
wound-healing assay was performed. The results indicated that the
Ad5/F35-RELM-α-siRNA-treated cells significantly (P<0.01)
exhibited slower repair of the scratched wound when compared with
the control and the RELM-α vector-treated cells (Fig. 5). This result suggests that the
silencing of RELM-α expression inhibited cell migration.
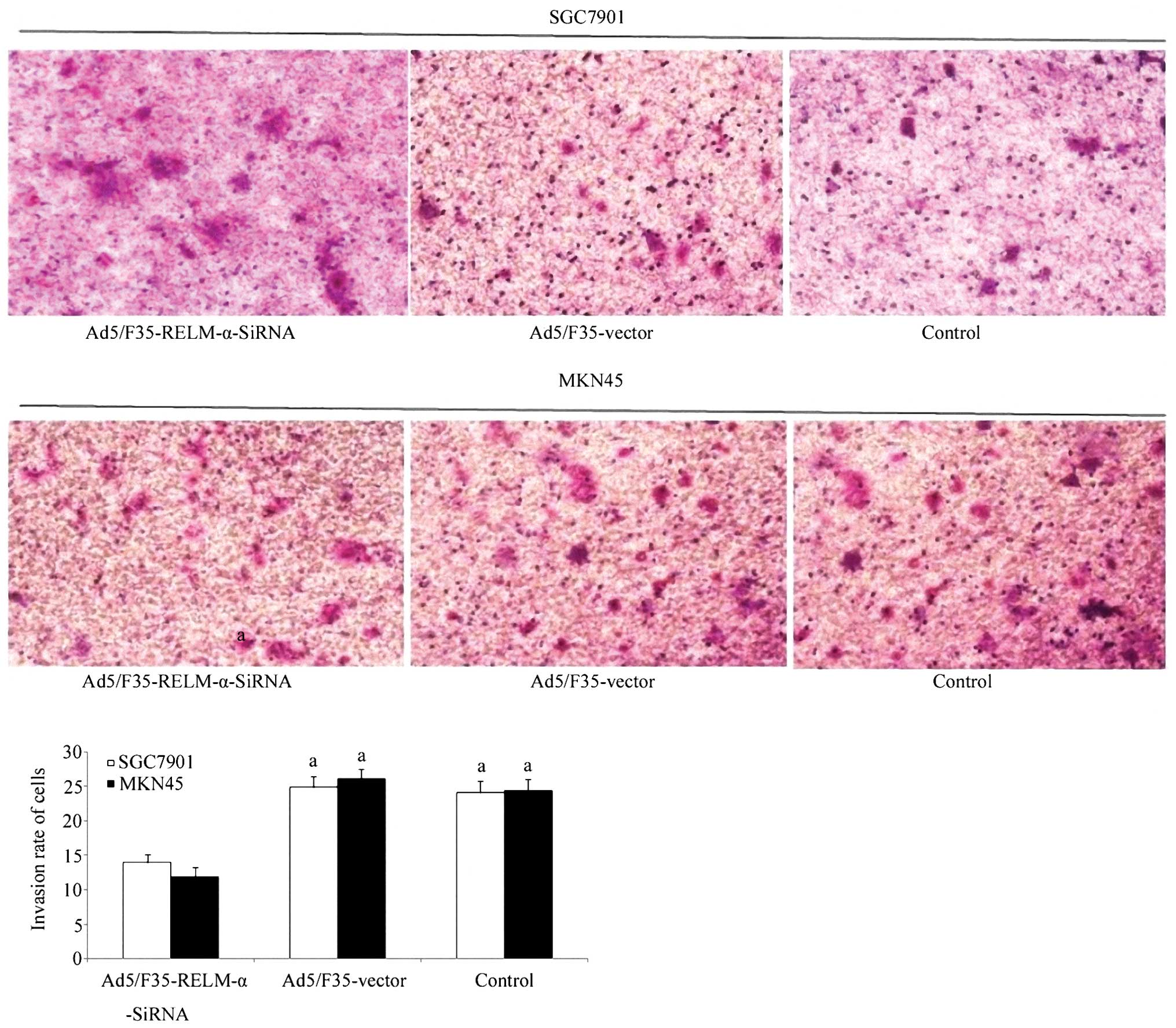
To further investigate the effect of RELM-α
silencing on cell invasion, the invasive ability of the cells was
examined using a Transwell chamber assay (Fig. 6). After incubation for 24 h, the
number of control and Ad5/F35 vector-treated cells which had
invaded the polycarbonate membrane of the Matrigel chamber was
~2-fold higher than that of the Ad5/F35-RELM-α-siRNA-treated group,
respectively, which showed a statistically significant difference
(P<0.01). These results provide evidences that RELM-α plays a
role in enhancing the metastasis of gastric cancer cells.
The mechanism involved in RELM-α
silencing in gastric cancer cells
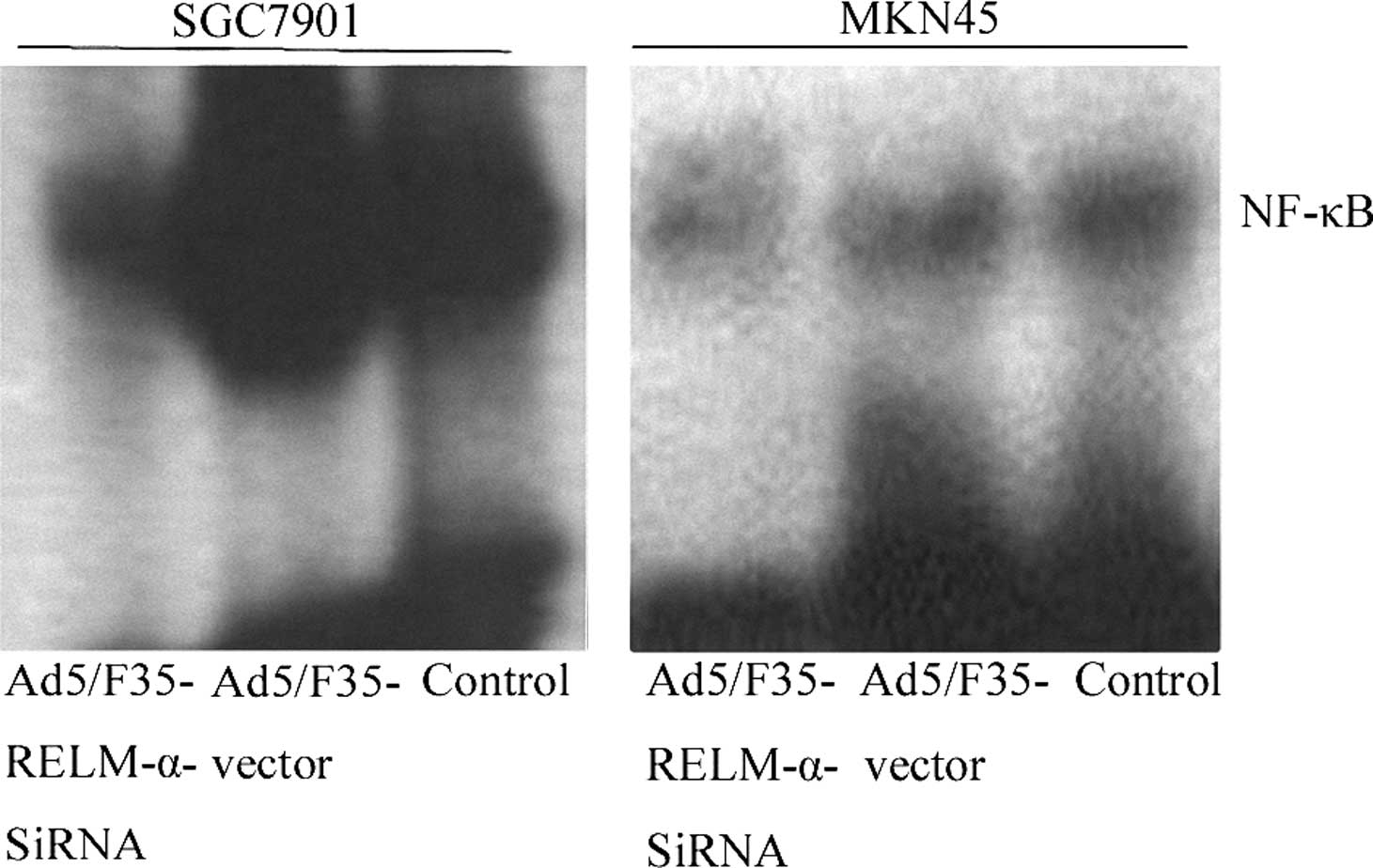
To determine the effect of RELM-α on NF-κB
activation, NF-κB-DNA binding activity was determined in the
different treated and control gastric cancer cells by EMSA. In
Fig. 7, after treatment with
Ad5/F35-RELM-α-siRNA for 24 h, the NF-κB-DNA binding activity was
decreased when compared with the Ad5/F35 vector-treated and control
cells.
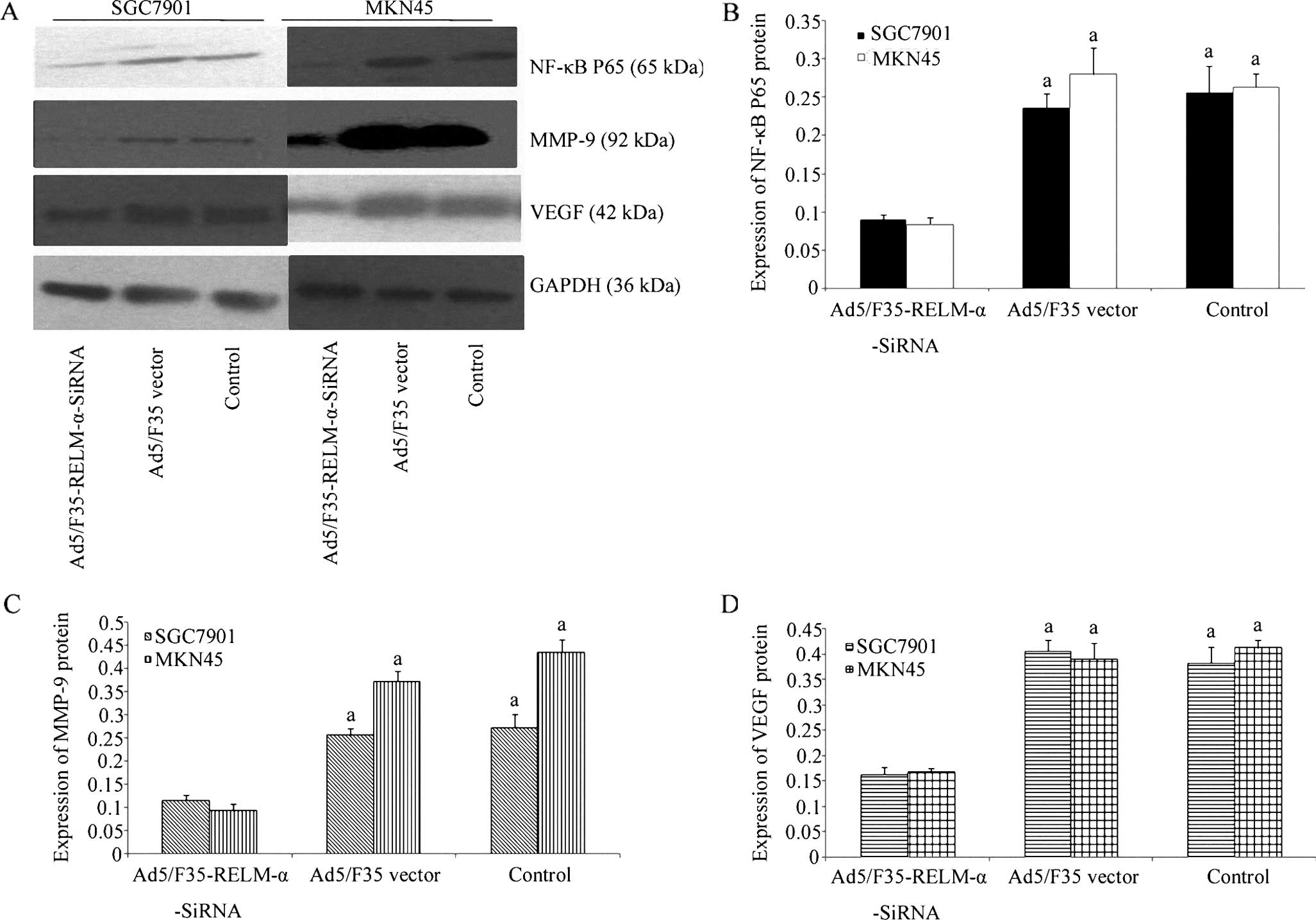
The expression level of p65, as the active subunit
of NF-κB, was significantly decreased in the
Ad5/F35-RELM-α-siRNA-treated cells, compared with the level in the
Ad5/F35-vector and control cells (P<0.01). Additionally, the
downstream proteins regulated by NF-κB, MMP-9 and VEGF, were also
suppressed in the Ad5/F35-RELM-α-siRNA-treated cells when compared
with the Ad5/F35 vector-treated and the control cells, respectively
(P<0.01) (Fig. 8).
Discussion
RELM-α belongs to the RELM family of cysteine-rich
secretory proteins that share homology with resistin. To date, four
members of this family have been identified: RELM-α, RELM-β, FIZZ-3
and RELM-γ (13). Some studies in
animals suggest that the members of the RELM family have important
pro-inflammatory and remodeling roles. For example, RELM-α was
first discovered in the ‘inflammatory zone’ of mice with allergic
pulmonary inflammation and has also been described as a
hypoxia-inducible mitogenic factor implicated in hypoxia-associated
vascular remodeling. RELM-α was found to be upregulated in several
infectious and inflammatory settings, including infection, allergic
airway inflammation and colitis (14,15).
Related research demonstrated that RELM-α promotes intestinal APC
activation, Th17 cell responses and intestinal inflammation
(16). A previous study
demonstrated that RELM-α was consistently detectable in the serum,
and the expression levels in colonic inflammation (17). In addition, substantial evidence
exists that RELM-α regulates pro-inflammatory cytokines (IL-6 and
TNF-α) and activates intracellular pro-inflammatory NF-κB signaling
pathway (18,19).
Angiogenesis is a process of neovascular formation
from pre-existing blood vessels, which consists of sequential steps
for vascular destabilization, lumen formation and vascular
stabilization. Furthermore, angiogenesis is a critical process in
the invasion, growth and metastasis of most solid tumors, and
induction of angiogenesis represents one of the major hallmarks of
cancer (20). Angiogenesis is
complex and involves a large number of molecules including VEGF and
MMP-9 (21). VEGF is a key mediator
in the angiogenesis of cancers through signaling pathways including
phosphoinositide 3 kinase (PI3K)/Akt and NF-κB, which stimulate
endothelial cells of microvessels to proliferate, migrate and alter
their pattern of gene expression (22). The high levels of VEGF in tumors are
predictive of high metastatic risk and poor prognosis (23). In the present study, VEGF expression
was highly correlated with angiogenesis, malignancy and metastasis
of gastric cancer. MVD has been used to evaluate the angiogenic
activity of tumors (24). In the
present study, the MVD value was significantly increased in the
gastric cancer tissues when compared with that in the adjacent
normal tissues. This finding suggests that angiogenesis plays an
important role in the development of gastric cancer. Furthermore,
upregulation of RELM-α was observed in the gastric cancer but not
in the normal gastric tissues. In addition, expression of RELM-α
was correlation with the expression of VEGF and the MVD in the
tissues. In conclusion, our results suggest that RELM-α is a novel
independent prognostic marker with functional relevance in gastric
cancer, yet its related mechanisms remain unclear.
In the present study, the results demonstrated for
the first time that RELM-α was silenced in gastric cancer cell
lines by siRNA treatment. We then explored the molecular mechanisms
of RELM-α, to identify key regulators of the RELM-α mediated effect
on gastric cancer.
The activation of NF-κB and its signaling pathways
are central coordinators of innate and adaptive immune responses.
More recently, it has become clear that NF-κB also has a critical
role in cancer development and progression. Inactivation of NF-κB
decreases tumor multiplicity or size in cancer by downregulating
anti-apoptotic gene expression and dampened production of
growth-stimulating cytokines (25).
Moreover, NF-κB also regulates tumor angiogenesis and invasiveness
(26). NF-κB p65 is sequestered in
the cytoplasm by its inhibitor proteins in cells, and in response
to a variety of stimuli, its inhibitor proteins can be
phosphorylated resulting in the translocation of cytoplasmic NF-κB
p65 into the nucleus, thereby activating the transcription of NF-κB
target genes (27).
In keeping with this, in the present study, the
activation of NF-κB was increased in gastric cancer, yet RELM-α
silencing decreased its activation. Furthermore, the expression of
NF-κB p65 was decreased in gastric cancer cells by
Ad5/F35-RELM-α-siRNA treatment.
Previous studies have described RELM-α expression in
vascular smooth muscle and endothelial cells of the remodeling
vasculature in animals (28). The
present study also demonstrated that RELM-α expression was
positively correlated with angiogenesis of gastric cancers. Based
on this result, the study further explored the relationship between
RELM-α and VEGF expression.
A recent study suggested that RELM-α upregulated
VEGF expression in mouse epithelial cells via an NF-κB-dependent
pathway (29) which is in
accordance with our present study that found that the VEGF
production in cells was significantly attenuated by NF-κB signaling
pathway inhibitor that was involved in RELM-α silencing in gastric
cancer. On the other hand, MMP-9 is a downstream target gene of
NF-κB, and has also been found to be upregulated in gastric cancer
cells. Furthermore, it has been reported that the expression of
MMP-9 is closely correlates with tumor angiogenesis (30). MMP-9 has also been shown to trigger
an angiogenic switch during tumor progression by releasing VEGF
(31). We found that cells with
silenced expression of RELM-α exhibited decreased expression of
MMP-9 and VEGF protein when compared to the control cells. Thus,
RELM-α is involved in the development of gastric cancer by the
NF-κB-MMP-9/VEGF pathway.
In summary, the positive expression of RELM-α in the
gastric cancer tissues was related to tumor size, clinical stage
and promoted the progression of gastric cancer by angiogenesis. The
anti-angiogenesis following RELM-α silencing may be involved in the
regulation of the production of angiogenesis factor VEGF and NF-κB
signaling pathway activation. Inhibition of RELM-α expression was
associated with the inhibition of tumor progression and
invasiveness in gastric cancer. Finally, RELM-α targeted gene and
protein therapy can be used as an effective molecular targeted
therapy for gastric cancer.
References
|
1
|
Zhang D and Fan D: New insights into the
mechanisms of gastric cancer multidrug resistance and future
perspectives. Future Oncol. 6:527–537. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hartgrink HH, Jansen EP, van Grieken NC
and van de Velde CJ: Gastric cancer. Lancet. 374:477–490. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Santoro R, Carboni F, Lepiane P, Ettorre
GM and Santoro E: Clinicopathological features and prognosis of
gastric cancer in young European adults. Br J Surg. 94:737–742.
2007. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Liu T, Dhanasekaran SM, Jin H, Hu B,
Tomlins SA, Chinnaiyan AM and Phan SH: FIZZ1 stimulation of
myofibroblast differentiation. Am J Pathol. 164:1315–1326. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Holcomb IN, Kabakoff RC, Chan B, Baker TW,
Gurney A, Henzel W, Nelson C, Lowman HB, Wright BD, Skelton NJ, et
al: FIZZ1, a novel cysteine-rich secreted protein associated with
pulmonary inflammation, defines a new gene family. EMBO J.
19:4046–4055. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Raes G, De Baetselier P, Noël W, Beschin
A, Brombacher F and Hassanzadeh Gh G: Differential expression of
FIZZ1 and ym1 in alternatively versus classically activated
macrophages. J Leukoc Biol. 71:597–602. 2002.PubMed/NCBI
|
|
7
|
Stütz AM, Pickart LA, Trifilieff A,
Baumruker T, Prieschl-Strassmayr E and Woisetschläger M: The Th2
cell cytokines IL-4 and IL-13 regulate found in inflammatory zone
1/resistin-like molecule alpha gene expression by a STAT6 and
CCAAT/enhancer-binding protein-dependent mechanism. J Immunol.
170:1789–1796. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Dong L, Wang SJ, Camoretti-Mercado B, Li
HJ, Chen M and Bi WX: FIZZ1 plays a crucial role in early stage
airway remodeling of OVA-induced asthma. J Asthma. 45:648–653.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Filbey KJ, Grainger JR, Smith KA, Boon L,
van Rooijen N, Harcus y, Jenkins S, Hewitson JP and Maizels RM:
Innate and adaptive type 2 immune cell responses in genetically
controlled resistance to intestinal helminth infection. Immunol
Cell Biol. 92:436–448. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Angelini DJ, Su Q, Kolosova IA, Fan C,
Skinner JT, Yamaji-Kegan K, Collector M, Sharkis SJ and Johns RA:
Hypoxia-induced mitogenic factor (HIMF/FIZZ1/RELM alpha) recruits
bone marrow-derived cells to the murine pulmonary vasculature. PLoS
One. 5:e112512010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Weidner N, Folkman J, Pozza F, Bevilacqua
P, Allred EN, Moore DH, Meli S and Gasparini G: Tumor angiogenesis:
A new significant and independent prognostic indicator in
early-stage breast carcinoma. J Natl Cancer Inst. 84:1875–1887.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Huang L, Ma J, Tang Y, Chen P, Zhang S,
Zhang Y and Yuan YZ: siRNA-based targeting of fractalkine
overexpression suppresses inflammation development in a severe
acute pancreatitis rat model. Int J Mol Med. 30:514–520.
2012.PubMed/NCBI
|
|
13
|
Steppan CM and Lazar MA: The current
biology of resistin. J Intern Med. 255:439–447. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sun Y, Wang J, Li H and Han X: Found in
inflammatory zone 1 induces angiogenesis in murine models of
asthma. Lung. 186:375–380. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Li X, Yang Y, Fang J and Zhang H: FIZZ1
could enhance the angiogenic ability of rat aortic endothelial
cells. Int J Clin Exp Pathol. 6:1847–1853. 2013.PubMed/NCBI
|
|
16
|
Osborne LC, Joyce KL, Alenghat T,
Sonnenberg GF, Giacomin PR, Du Y, Bergstrom KS, Vallance BA and
Nair MG: Resistin-like molecule α promotes pathogenic Th17 cell
responses and bacterial-induced intestinal inflammation. J Immunol.
190:2292–2300. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yamaji-Kegan K, Su Q, Angelini DJ,
Champion HC and Johns RA: Hypoxia-induced mitogenic factor has
proangiogenic and proinflammatory effects in the lung via VEGF and
VEGF receptor-2. Am J Physiol Lung Cell Mol Physiol.
291:L1159–L1168. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Munitz A, Waddell A, Seidu L, Cole ET,
Ahrens R, Hogan SP and Rothenberg ME: Resistin-like molecule alpha
enhances myeloid cell activation and promotes colitis. J Allergy
Clin Immunol. 122:1200.e1–1207.e1. 2008. View Article : Google Scholar
|
|
19
|
Munitz A, Seidu L, Cole ET, Ahrens R,
Hogan SP and Rothenberg ME: Resistin-like molecule alpha decreases
glucose tolerance during intestinal inflammation. J Immunol.
182:2357–2363. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Suzuki S, Dobashi Y, Hatakeyama Y, Tajiri
R, Fujimura T, Heldin CH and Ooi A: Clinicopathological
significance of platelet-derived growth factor (PDGF)-B and
vascular endothelial growth factor-A expression, PDGF receptor-β
phosphorylation, and microvessel density in gastric cancer. BMC
Cancer. 10:6592010. View Article : Google Scholar
|
|
21
|
Sia D, Alsinet C, Newell P and Villanueva
A: VEGF signaling in cancer treatment. Curr Pharm Des.
20:2834–2842. 2014. View Article : Google Scholar
|
|
22
|
Abid MR, Schoots IG, Spokes KC, Wu SQ,
Mawhinney C and Aird WC: Vascular endothelial growth
factor-mediated induction of manganese superoxide dismutase occurs
through redox-dependent regulation of forkhead and
IkappaB/NF-kappaB. J Biol Chem. 279:44030–44038. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yu YF, Zhang Y, Shen N, Zhang RY and Lu
XQ: Effect of VEGF, P53 and telomerase on angiogenesis of gastric
carcinoma tissue. Asian Pac J Trop Med. 7:293–296. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Vermeulen PB, Gasparini G, Fox SB, Toi M,
Martin L, McCulloch P, Pezzella F, Viale G, Weidner N, Harris AL,
et al: Quantification of angiogenesis in solid human tumours: An
international consensus on the methodology and criteria of
evaluation. Eur J Cancer. 32A:2474–2484. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Pikarsky E, Porat RM, Stein I, Abramovitch
R, Amit S, Kasem S, Gutkovich-Pyest E, Urieli-Shoval S, Galun E and
Ben-Neriah Y: NF-kappaB functions as a tumour promoter in
inflammation-associated cancer. Nature. 431:461–466. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Karin M and Greten FR: NF-kappaB: Linking
inflammation and immunity to cancer development and progression.
Nat Rev Immunol. 5:749–759. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yin Y, Si X, Gao Y, Gao L and Wang J: The
nuclear factor-κB correlates with increased expression of
interleukin-6 and promotes progression of gastric carcinoma. Oncol
Rep. 29:34–38. 2013.
|
|
28
|
Teng X, Li D, Champion HC and Johns RA:
FIZZ1/RELMalpha, a novel hypoxia-induced mitogenic factor in lung
with vasoconstrictive and angiogenic properties. Circ Res.
92:1065–1067. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tong Q, Zheng L, Lin L, Li B, Wang D,
Huang C and Li D: VEGF is upregulated by hypoxia-induced mitogenic
factor via the PI-3K/Akt-NF-kappaB signaling pathway. Respir Res.
7:372006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yan C, Wang H, Aggarwal B and Boyd DD: A
novel homologous recombination system to study 92 kDa type IV
collagenase transcription demonstrates that the NF-kappaB motif
drives the transition from a repressed to an activated state of
gene expression. FASEB J. 18:540–541. 2004.PubMed/NCBI
|
|
31
|
Bergers G, Brekken R, McMahon G, Vu TH,
Itoh T, Tamaki K, Tanzawa K, Thorpe P, Itohara S, Werb Z, et al:
Matrix metallo-proteinase-9 triggers the angiogenic switch during
carcinogenesis. Nat Cell Biol. 2:737–744. 2000. View Article : Google Scholar : PubMed/NCBI
|