Introduction
Epidemiological studies have demonstrated that diets
rich in fruits and vegetables have a chemopreventive effect on the
development of cancer. It has been reported that phenolic
compounds, a large family of natural compounds, abundant in fruits
and vegetables, have been associated with a possible reduced risk
of prostate cancer (1), breast
cancer (2), liver cancer (3) and cervical cancer (4). The potential application of phenolic
compounds in the development of therapeutic agents for cancer
treatment and for their use as valuable additive or nutritional
supplements to prevent cancer risk has gained increasing
importance, and research in this field is currently expanding
(5–7).
Ovarian cancer is one of the leading causes of
cancer-related mortality in women in developed areas (8). In 2015, an estimated 21,290 new cases
and 14,180 deaths due to ovarian cancer occurred in the USA
(9). Unfortunately, the overall
survival rate at 5 years is only 50%, which has not significantly
improved in the past 30 years (10). Even though the majority of women
have successful initial therapy, the low rate of survival is due to
eventual recurrence and succumbing to the disease. The treatment
for ovarian cancer is still unsatisfactory, and new treatments for
patients with recurrent ovarian cancer who are incurable with
current management are needed. Another challenge is that more than
70% of women present with advanced stage disease (11). Symptoms of the disease generally do
not present themselves until after the cancer has metastasized from
the ovaries to the surfaces of the peritoneal cavity. Once
metastasis has occurred, surgical removal of all lesions is no
longer possible, making ovarian cancer extremely difficult to
combat. The remaining first-line treatment option is
chemotherapy.
Cytoreductive surgery followed by platinum and
taxane-based combination chemotherapy is currently the standard
treatment for ovarian cancer (12).
Ovarian cancer is one of the most sensitive solid tumors, with
objective responses ranging from 60 to 80% to chemotherapy even in
patients with advanced stage disease. However, most patients
ultimately recur and develop resistance to chemotherapy. Resistance
to chemotherapy presents a major obstacle to improving the
prognosis of patients with ovarian cancer. The response rate to
current second-line or third-line (after interim non-platinum
therapy) chemotherapy is <33% due to the rise in resistance to
these drugs (13,14). Therefore, selecting new chemicals as
cancer therapeutics from natural compounds is still crucial for
ovarian cancer research. Finding new natural compounds that match
or exceed the effects of common chemical drugs, or are able to be
given in combination with cisplatin to overcome resistance is of
great significance (15). Our
previous studies have shown that phenolic compounds are effective
inhibitors of cell growth and vascular endothelial growth factor
(VEGF) expression in ovarian cancer cells (16–18).
Gallic acid (3,4,5-trihydroxybenzoic acid, GA), a
predominant polyphenol, is an endogenous plant polyphenol
abundantly found in tea, grapes, berries and other fruits as well
as in wine. GA has been shown to exhibit a variety of
pharmacological and biological properties, including antibacterial,
antiviral, and antitumor activities in many human cancer cell
lines. Its anticancer activity has been reported in oral (19), lung (20), pancreatic (21), glioma (22) and cervical cancer cells (23).
In our previous experiments, we found that GA has
the greatest inhibitory activity on human ovarian cancer cells
among eight natural phenolic compounds from traditional Chinese
medicine (24). However, the
molecular mechanisms underlying the anti-angiogenic effects of GA
on ovarian cancer have not been discussed in detail to date. In the
present study, the inhibitory effect on cell proliferation, in
vitro angiogenesis and VEGF expression by GA on human ovarian
cancer cell lines was examined. In particular, the mechanism of
GA-induced anti-angiogenesis in OVCAR-3 cells was further
studied.
Materials and methods
Cell culture and treatment
Ovarian cancer cell line OVCAR-3 was provided by Dr
BingHua Jiang of West Virginia University. IOSE-364, normal ovarian
surface epithelial cells from healthy women, but immortalized with
SV40 T/t, were courtesy of Dr Auersperg of the University of
British Columbia, Canada. All cells were maintained in RPMI-1640
medium (Sigma) supplemented with 10% US-qualified fetal bovine
serum (Invitrogen, Grand Island, NY, USA) in a cell culture
incubator with 5% CO2 at 37°C. A stock solution of GA
(Sigma) was prepared in dimethyl sulfoxide (DMSO) at 100 mM and
stored at −20°C. Different concentrations of GA were prepared in
RPMI-1640 medium for cell treatments, and DMSO was included in the
preparations to ensure equal concentrations of DMSO in each
treatment.
Cell viability assay
To measure cell viability, cells were seeded in
96-well plates at a density of 1×104/well and allowed to
recover, attach to the substrate, and grown to log phase overnight.
After incubation 37°C, the culture medium was removed and incubated
with different concentrations of GA for 24 h. Each treatment was
performed in triplicate. After treatment, the cells were washed
twice with phosphate-buffered saline (PBS), introduced to 100
μl freshly prepared AQueous One Solution (MTS tetrazolium
compound) (Promega, Madison, WI, USA) in medium, and incubated for
1 h at 37°C. The OD values, at 490 nm, were measured by an
enzyme-linked immunosorbent assay (ELISA) plate reader (Bio-Tek
Instruments, Winooski, VT, USA). Cell viability was expressed as a
percentage of the control.
VEGF protein quantification
The effect of GA on VEGF protein secretion was
analyzed by ELISA with a Quantikine Human VEGF Immunoassay kit
(R&D Systems, Minneapolis, MN, USA), targeting VEGF165 in the
cell culture supernatant. Cells (6×105) were seeded in
60-mm cell culture dishes and allowed to attach to the substrate
and grow for 16 h at 37°C before treatment with various
concentrations of GA for 24 h. Culture supernatants were collected
for the VEGF assay. The inhibition of VEGF protein secretion was
expressed as a percentage of the control. Cell lysates were also
assayed for total protein levels using the BCA protein assay kit
(Pierce, Rockford, IL, USA) to adjust VEGF levels.
In vitro angiogenesis assay
OVCAR-3 cancer cells were seeded in 60-mm cell
culture dishes at 6×105 cells/dish and incubated
overnight. Cells were then treated with 4 ml serum-free medium
containing 0, 5, 10, or 20 μM GA for 24 h and collected for
the in vitro angiogenesis assay. Growth factor-reduced
Matrigel (BD Biosciences, San Jose, CA, USA) was transferred into
96-well plates at 50 μl/well and incubated at 37°C for 1 h
to gel. Human umbilical vein endothelial cells (HUVECs) were
harvested in PBS, counted, and seeded onto the Matrigel beds at
2×104 cells/90 μl PBS. Thereafter, volumes of 10
μl of the collected conditioned media were added to each
well. The system was incubated at 37°C for 8 h, and tube formation
was visualized under a microscope with images captured for
comparison.
Western blot analysis
OVCAR-3 cancer cells were seeded and incubated
overnight before treatment with GA. After a double wash with cold
PBS, the cells were harvested with M-PER Mammalian Protein
Extraction reagent supplemented with Halt protease and phosphatase
inhibitor (both from Pierce), and total protein levels were assayed
with the BCA protein assay kit. Cell lysates (50 μg total
protein) were separated by SDS-PAGE and blotted onto a
nitrocellulose membrane with a Mini-Protean 3 system (Bio-Rad,
Hercules, CA, USA). For immunodetection, antibodies against HIF-1α,
HIF-1β, AKT, p-AKT, PTEN and GAPDH (Santa Cruz Biotechnology, Santa
Cruz, CA, USA) were applied, and signals were visualized with
phycoerythrin-conjugated anti-mouse IgG secondary antibodies,
SuperSignal West Pico Substrate, and X-ray film (Pierce). Protein
bands were quantitated with NIH ImageJ software and normalized by
GAPDH bands for analysis.
Transient transfection and luciferase
assay
OVCAR-3 cancer cells (1×104 cells/well)
were seeded onto 96-well plates and incubated overnight. For
transfection with hypoxia-inducible factor 1α (HIF-1α) plasmids
(Addgene, Cambridge, MA, USA), cells were transfected with VEGF
luciferase reporter (0.05 μg) and HIF-1α plasmids (0,
0.0625, 0.125 and 0.25 μg) or SR-a (as vehicle) plasmids
using 0.6 μl of jetPRIME reagent (VWR, West Chester, PA,
USA) for 4 h. For transfection with mAkt plasmids (Addgene), the
cells were transfected with HIF-1α or VEGF luciferase reporter
(0.05 μg) and mAkt (0, 0.0625, and 0.125 μg) or the
SR-a plasmids using 0.6 μl of jetPRIME reagent for 4 h.
After transfection, all the cells were treated with or without GA
(5 μM) for 16 h. The cells were harvested and analyzed for
luciferase activity and total protein levels using a BCA protein
assay kit, and the activities of VEGF reporter or the HIF-1α
reporter were normalized to corresponding total protein levels for
statistical analysis. The experiments were conducted 3–8 times.
Statistical analysis
One-way ANOVA followed by Dunnett's test or by the
Student's t-test was applied to compare the effects between
chemical treatments. A p-value of <0.05 was considered as
indicative of a statistically significant result.
Results and Discussion
GA inhibits cell proliferation and VEGF
secretion dose-dependently in an ovarian cancer cell line
Phenolic compounds, a large family of natural
compounds with phenolic hydroxyls found in plants, fruits,
vegetables and teas, have long been regarded as significant
secondary metabolites for their chemopreventive and
chemotherapeutic effects in cancer (25,26).
In our previous experiments, eight natural phenolic compounds from
traditional Chinese medicine were compared for their
anti-proliferative effects on an ovarian cancer cell line at
concentrations of 20 and 40 μM using a cell viability assay.
We found that GA had the greatest inhibitory activity among these
phenolic compounds (24).
Therefore, GA was selected for subsequent experiments. Yet, we
needed to clarify the appropriate dosage of GA needed to exert
significant effects and to determine the relative cytotoxic effects
on ovarian cancer cell lines compared to normal ovarian cells. In
the present study, we further examined the suppression of cell
viability after treatment with varying concentrations of GA (0, 5,
10, 20 and 40 μM) in ovarian cancer cell lines (OVCAR-3,
A2780/CP70) and in a normal ovarian cell line (ISOE-364).
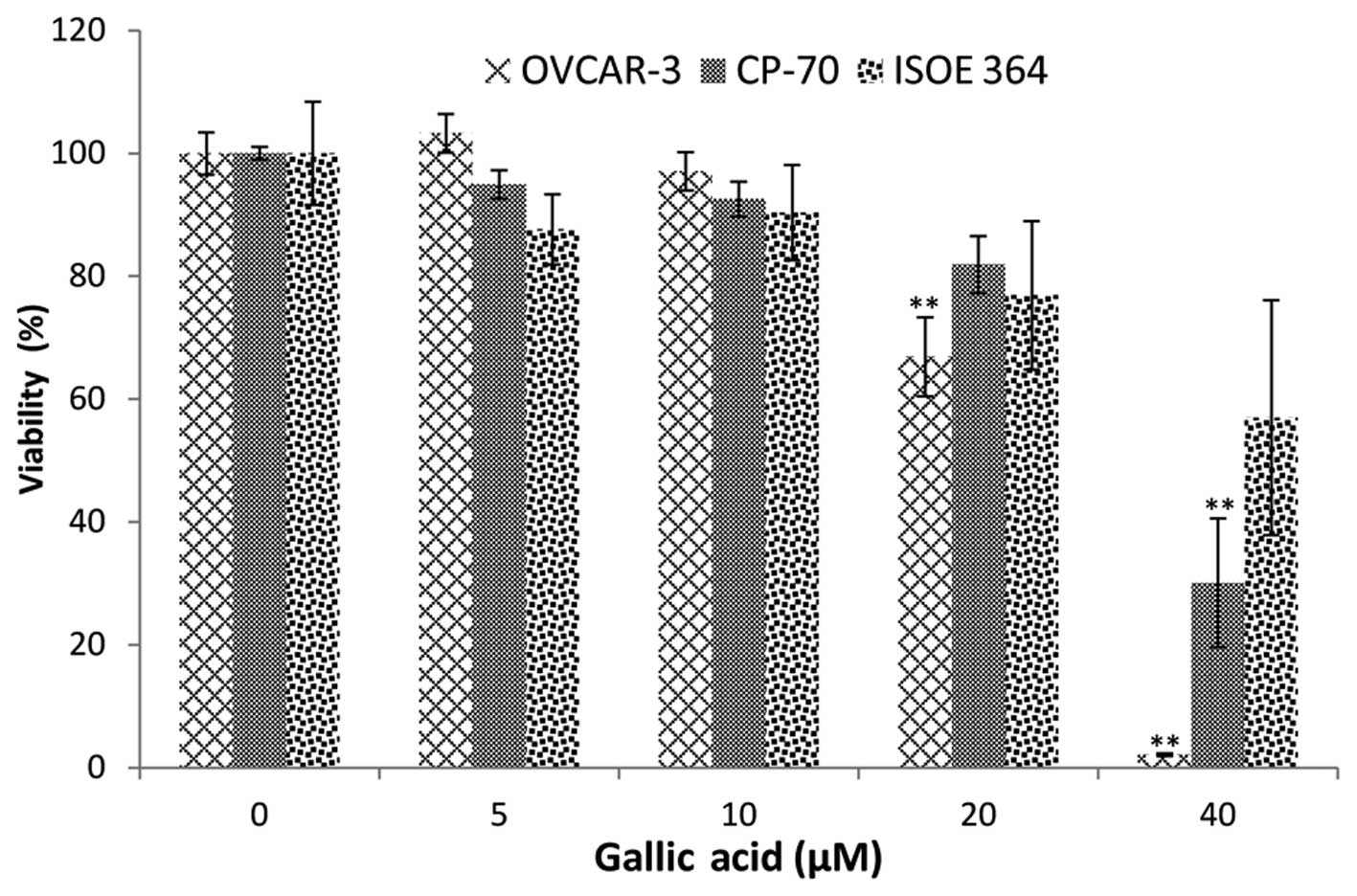
As shown in Fig. 1,
an overall inhibitory effect of GA on ovarian cancer cell lines and
the normal ovarian cell line was tested. A2780/CP70 cell viability
was significantly inhibited to 30.10% at 40 μM GA treatment.
The inhibitory effect on OVCAR-3 cells was much higher than that on
the A2780/CP70 cells. OVCAR-3 cell viability was inhibited to 66.86
and 2.11% by 20 and 40 μM GA treatment, respectively.
Compared to the inhibitory effect of cisplatin at 20 μM
(16), the lower cell viabilities
demonstrate that GA has high potential in the prevention and
therapy of ovarian cancer.
Notably, GA did not have a significant inhibitory
effect on the normal cell line (IOSE-364) at 40 μM. This
result indicates that the inhibitory effects of GA on normal
ovarian cells were less than that on the ovarian cancer cells.
Therefore, GA selectively inhibits cancer cells while having
significantly weaker inhibitory effects on normal ovarian
cells.
Many polyphenols have poor bioavailability, with
about a 100–1,000 nM concentration in human serum. However,
analyses of the bioavailability of polyphenols in humans reveal
that GA performs better than other polyphenols (27). A concentration of ~8–10 μM of
GA was detected in the serum of healthy volunteers, after oral
intake of a combination of a dietary herbal supplement and 800 mg
GA (28). The GA concentration
found in this study is very close to therapeutic concentrations. A
slight decrease in cell viability at 10 μM GA treatment was
also observed, although this decrease did not reach statistical
significance.
VEGF is the best studied and the most potent
pro-angiogenic factor known (29,30).
For the purpose of angio-prevention, levels of VEGF were examined.
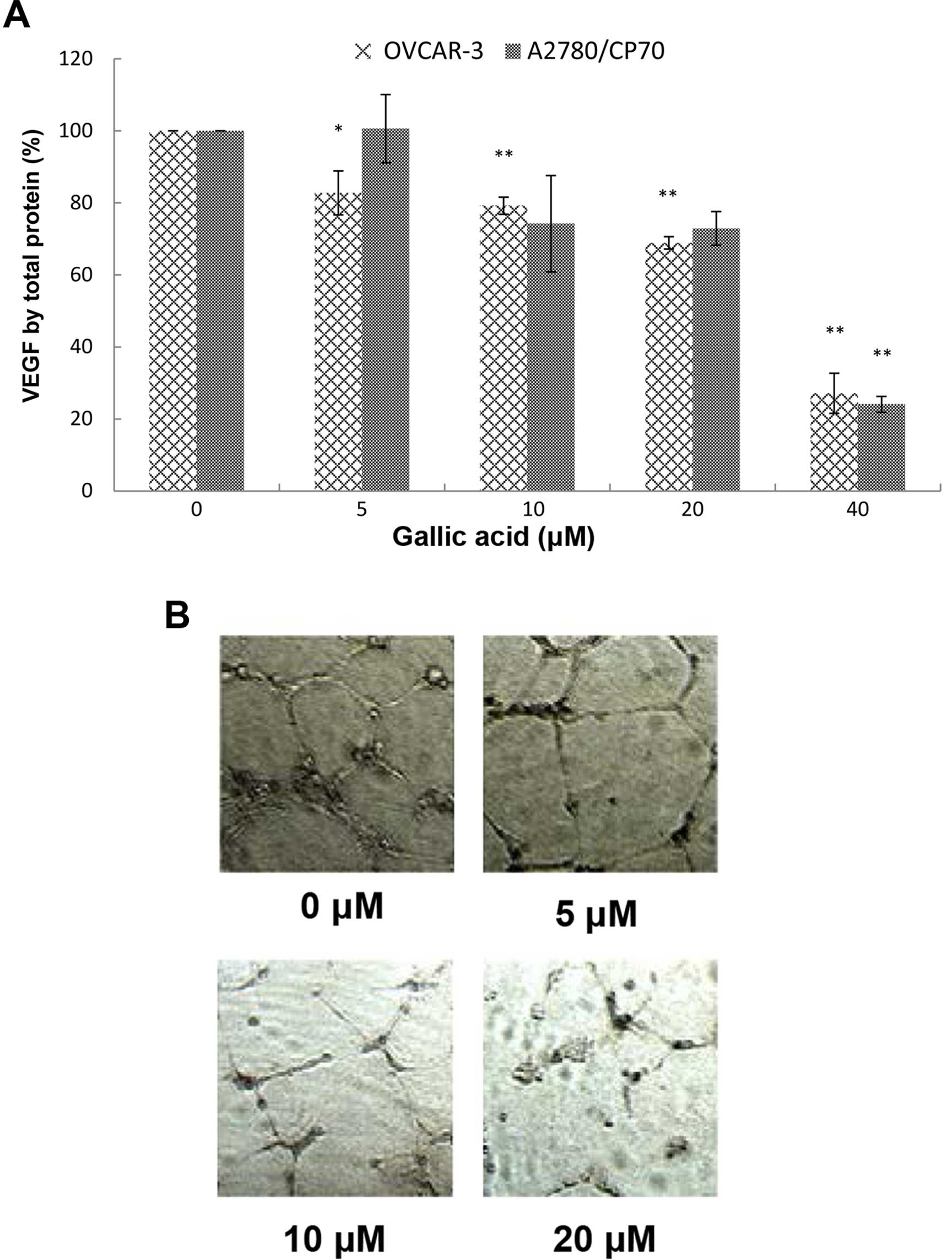
As shown in Fig. 2A, VEGF
expression in the A2780/CP70 cells and OVCAR-3 cells with 40
μM GA treatment was significantly inhibited to 24.05 and
27.12%, respectively. The inhibitory effect on OVCAR-3 cells
increased significantly when the concentrations were >5
μM while for A2780/CP70 cells the concentrations were >40
μM. Levels of VEGF excreted by OVCAR-3 cancer cells were
inhibited to 82.75, 79.22, 68.88, and 27.12% by 5, 10, 20, and 40
μM GA treatments, respectively.
The inhibitory effect of GA on the OVCAR-3 cells was
more pronounced than that on the A2780/CP70 cells. As shown in
Fig. 2A, the inhibitory effects on
A2780/CP70 cells treated with 10 and 20 μM GA were different
from the control but they were not significant. This may be due to
the lack of functional DNA mismatch repair of A2780/CP70 cells,
which hampers the inhibitory effect of GA (31).
Since 8 to 10 μM of GA in the blood is
attainable in humans following a plant-rich diet, our research
suggests that GA can directly inhibit VEGF secretion of ovarian
cancer cells at physiologically relevant concentrations. This is an
exciting result which indicates that GA may have legitimate
potential in treating cancer patients in the clinic.
GA inhibits in vitro angiogenesis induced
by OVCAR-3 in ovarian cancer cells
Angiogenesis, the formation of new blood vessels, is
essential for tumor development and subsequent growth, invasion and
metastasis. Suppressed expression of VEGF by kaempferol and
chaetoglobosin K has been shown to inhibit in vitro
angiogenesis in our previous research (17,32).
GA may have a similar effect of inhibiting tube formation by HUVECs
induced by VEGF. Therefore, we investigated the effect of GA on
in vitro tube formation by HUVECs induced by the culture
medium of ovarian cancer cells treated with different
concentrations of GA. We found that the culture media conditioned
by OVCAR-3 ovarian cancer cells promoted in vitro
angiogenesis and presented a well-established network of HUVECs
(Fig. 2B). Following treatment with
increasing concentrations of GA treatment, the network was
scattered down and HUVECs gradually lost their ability to form tube
structures. A 20-μM GA treatment completely destroyed the
network structure, and HUVECs were presented as mostly individual
clumps. Similar to other compounds in our earlier research, GA
inhibits in vitro angiogenesis through suppression of VEGF
expression.
GA inhibits VEGF production through
downregulation of HIF-1α expression
It has been well established that hypoxia stimulates
transcriptional activation of VEGF through induction of HIF-1α, and
HIF-1α is considered a primary regulator of VEGF production. Thus,
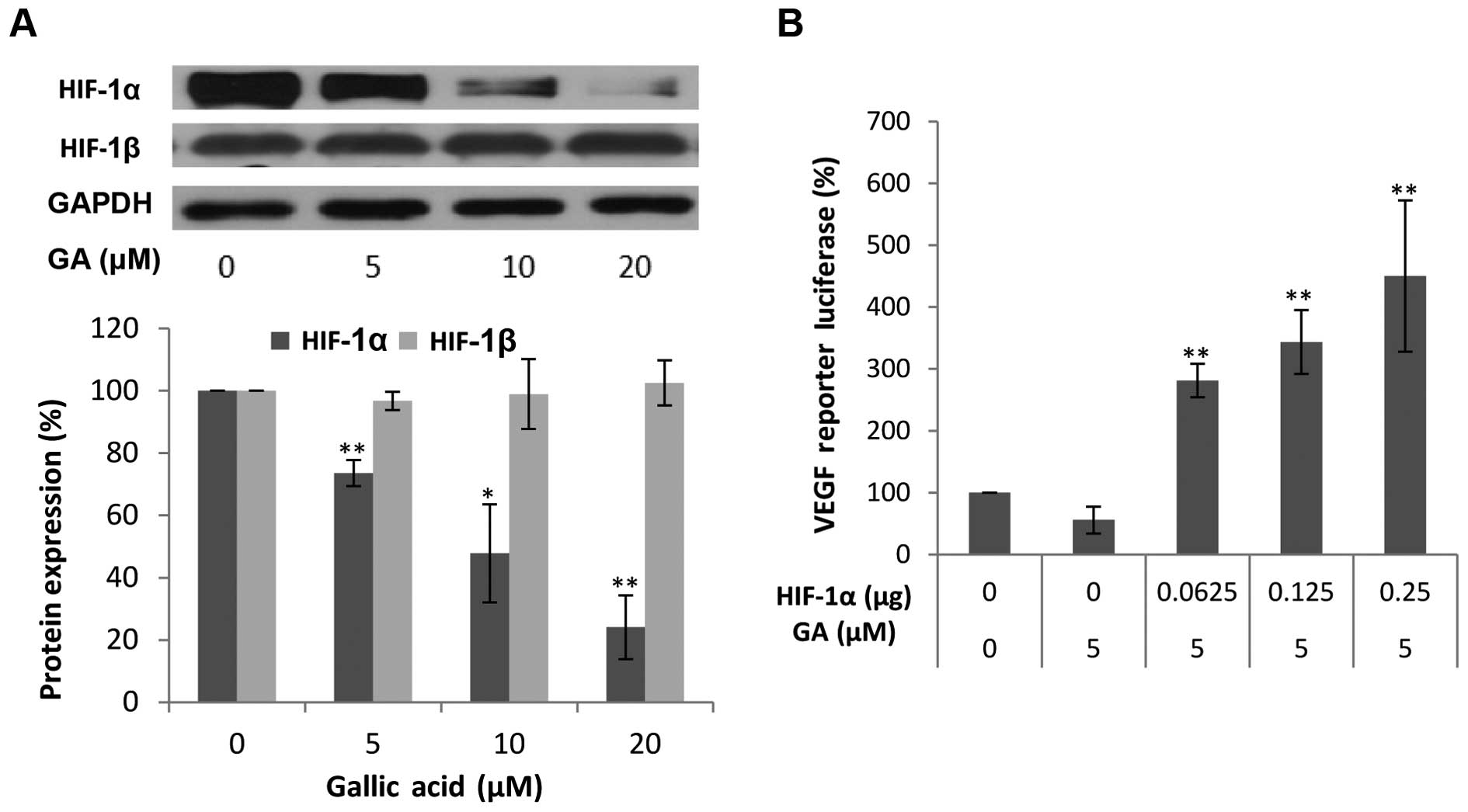
we first examined the influence of GA treatment on HIF-1α
expression. It is generally thought that HIF-1α is regulated mainly
by low oxygen or hypoxia as its name indicates. However, in
concordance with our previous findings, HIF-1α is highly expressed
in normoxic conditions in ovarian cancer cells (32). As expected, the protein levels of
HIF-1α in the OVCAR-3 cancer cell line were markedly decreased with
higher concentrations of GA. At 20 μM, the protein levels of
HIF-1α were 23.48% of the levels of HIF-1β. This means that GA
effectively decreased the protein expression of HIF-1α. These
results suggest that HIF-1α may be involved in the inhibitory
mechanism of VEGF production by GA treatment. To confirm that
HIF-1α expression is not only regulated by GA treatment but also
plays a role in the inhibition of VEGF secretion by GA, OVCAR-3
cells were transfected with the VEGF-promoter reporter, together
with HIF-1α plasmids. As shown in Fig.
3B, this inhibition of VEGF was significantly reversed by
forced expression of the HIF-1α protein. The higher the expression
of HIF-1α, the higher the increase in the expression of the VEGF
reporter. These results demonstrated that GA inhibits VEGF
production through a HIF-1α-dependent pathway.
The transcription factor HIF-1 consists of two
subunits, HIF-1α and HIF-1β. Research has shown that HIF-1β may be
regulated through an HIF-1α-dependent pathway (33). Thus, the protein levels of HIF-1β
were also investigated. As shown in Fig. 3A, GA did not have a significant
effect on the expression of HIF-1β. As generally regarded, HIF-1β
is constitutively expressed in ovarian cancer cells. GA inhibits
VEGF production through transcription factor, HIF-1, by reducing
the expression of HIF-1α.
GA inhibits HIF-1α expression through
blocking the phosphorylation of AKT
It is commonly recognized that the PI3K pathway
plays an important role in the regulation of cell growth, motility,
survival and metabolism, as well as angiogenesis. AKT, identified
as an oncogene in the 1980's, serves as a central sensor in the
PI3K pathway and represents an attractive therapeutic target since
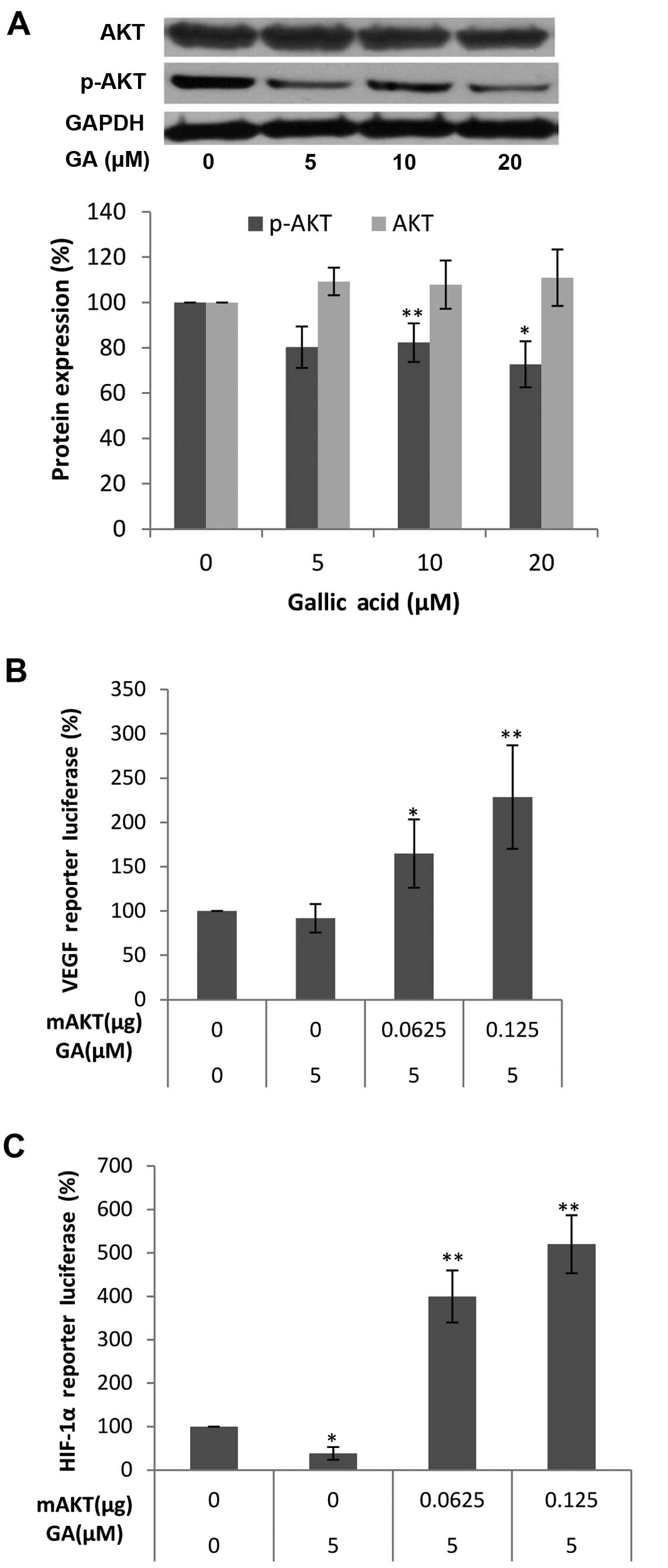
multiple signaling components converge in AKT (34). To check whether the inhibition of
the expression of HIF-1α and VEGF protein by GA is mediated by the
AKT pathway, we first examined the levels of AKT and p-AKT protein
after GA (0, 5, 10 and 20 μM) treatment. As shown in
Fig. 4A, the protein levels of
p-AKT in the OVCAR-3 cancer cell line were decreased with higher
concentrations of GA while no significant change in the protein
levels of AKT was found. These results demonstrate that GA inhibits
the phosphorylation of AKT. To show that the activation of AKT
plays a role in the inhibition of HIF-1α and VEGF secretion by GA,
OVCAR-3 cells were transfected with the HIF-1α promoter reporter,
together with the mAKT plasmids. As shown in Fig. 4B and C, the inhibition of HIF-1α and
VEGF was significantly reversed by forced expression of the mAKT
protein, and the higher the expression of mAKT, the higher the
expression of the HIF-1α reporter and VEGF reporter. These results
demonstrate that GA inhibits HIF-1α and VEGF expression through
blocking the phosphorylation of AKT. The regulating effect of AKT
on HIF-1α or VEGF expression in ovarian cancer cells was also
reported (35,36). Research into how the PI3K/AKT/mTOR
pathway affects the progression and tumorigenesis of ovarian cancer
may lead to new therapies that will increase the survival of these
patients (37). The inhibitory
effect of GA on the PI3K/Akt pathway demonstrates great therapeutic
potential for ovarian cancer.
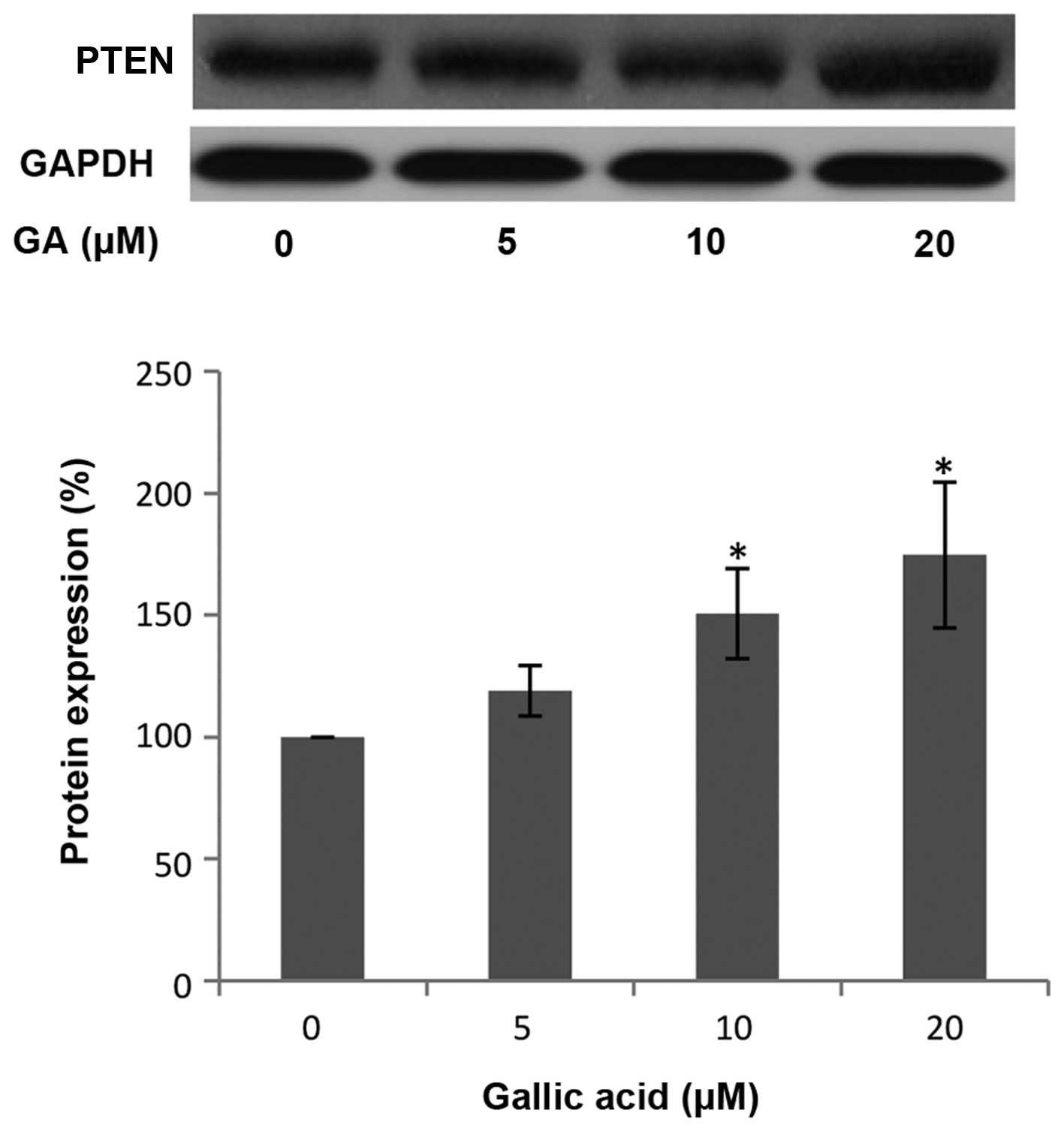
GA upregulates PTEN expression in OVCAR-3
ovarian cancer cells
Phosphatase and tensin homolog deleted on chromosome
10 (PTEN) is a tumor suppressor commonly mutated in many human
cancers. PTEN hydrolyzes the 3-phosphate on PIP3 to generate PIP2
and negatively regulates PIP3-mediated signaling pathways. AKT is
one of the most important downstream targets of PI3K. Therefore,
PTEN can control HIF-1α and VEGF expression through the PI3K/AKT
pathway (38). To examine whether
PTEN is involved in the inhibition of HIF-1α and VEGF expression by
GA, the level of PTEN protein was investigated by western blot
analysis. As shown in Fig. 5, the
protein levels of PTEN in the OVCAR-3 cancer cell line were
increased following treatment with a high concentration of GA. This
increase in PTEN protein expression indicates that GA may inhibit
HIF-1α and VEGF expression through upregulating PTEN protein.
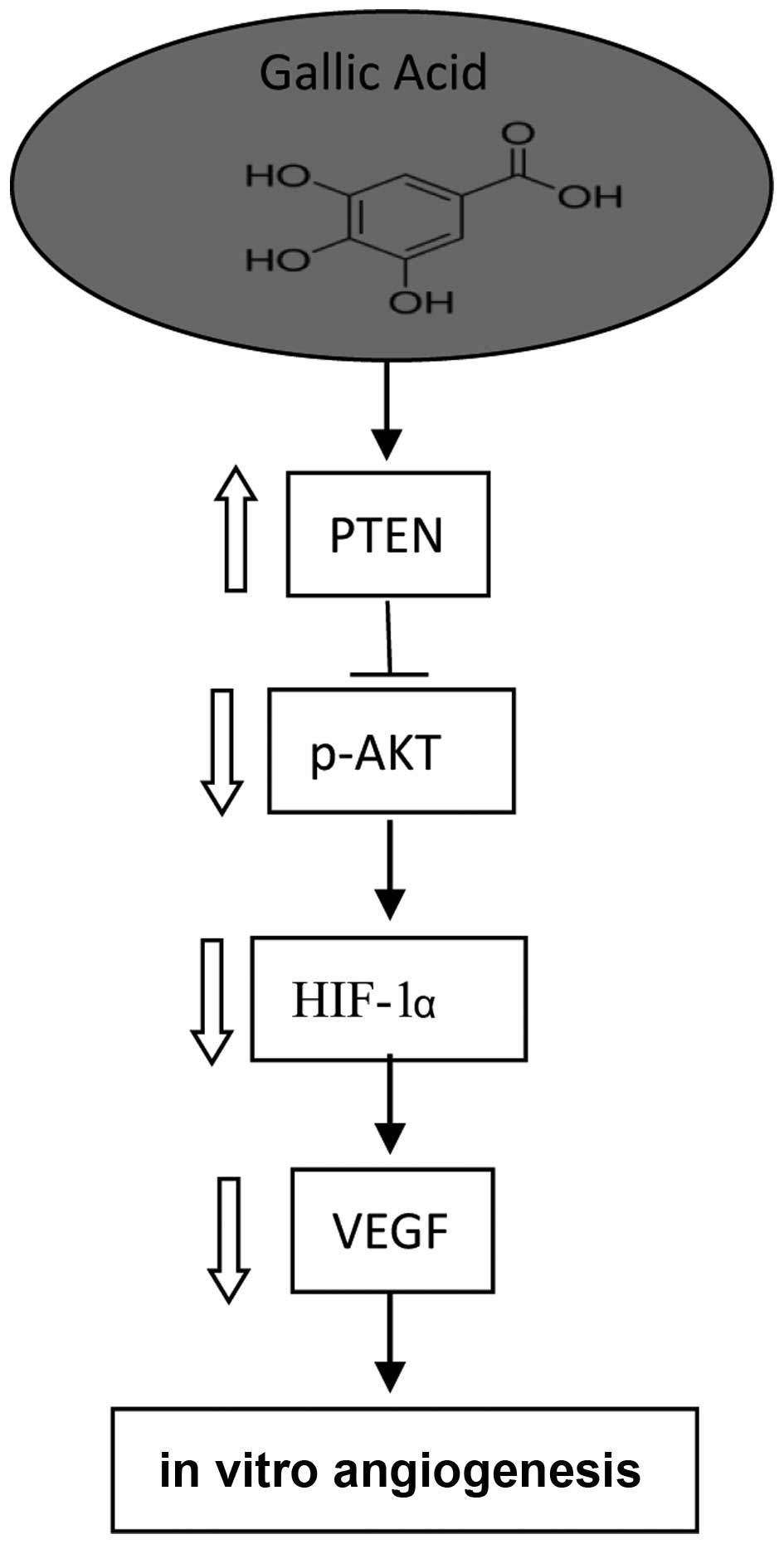
In conclusion, we investigated the molecular
mechanism of GA concerning its inhibition of VEGF expression and
in vitro angiogenesis activity. In the present study, GA was
found to directly inhibit VEGF secretion of ovarian cancer cells at
physiologically relevant concentrations (5 μM). These
experiments demonstrate that GA has great potential in the
prevention and therapy of ovarian cancer. To our knowledge, this is
the first study that addresses the anti-angiogenesis effect of GA
on ovarian cancer.
HIF-1, a transcription factor, can directly activate
VEGF expression. Our research shows that GA inhibits VEGF
production through the transcription factor, HIF-1 by markedly
reducing the expression of HIF-1α. GA can inhibit the expression of
VEGF at low concentrations possibly due to the strong inhibition of
the expression of HIF-1α. Furthermore, the inhibitory effect of GA
may be involved in the suppression of AKT phosphorylation and
upregulation of PTEN expression. A mechanism/pathway for the
inhibition of VEGF expression by GA in ovarian cancer cells is
proposed (Fig. 6). These findings
enhance our understanding of its mechanism of action and provide
strong support for using this compound in future animal and
clinical studies.
AKT is an important serine-threonine protein kinase
and carries out various cellular functions such as angiogenesis,
cell metabolism, cell survival/inhibition of apoptosis, and the
cell cycle. The inhibition of AKT phosphorylation by GA may imply
that GA induces various other cellular functions that require
further study.
Acknowledgments
This research was supported by an NIH grant
(5P20RR016477 and 8P20GM104434) from the National Center for
Research Resources awarded to the West Virginia IDeA Network of
Biomedical Research Excellence and the National Science Foundation
(1003907), the West Virginia Higher Education Policy
Commission/Division of Science Research. The authors also thank the
financial support from Natural Science Foundation of Zhejiang
Province (LY13C200013) and the Research and Development Fund of
Zhejiang A & F University (2012FR024).
References
|
1
|
Sánchez-González C, Ciudad CJ, Noé V and
Izquierdo-Pulido M: Walnut polyphenol metabolites, urolithins A and
B, inhibit the expression of the prostate-specific antigen and the
androgen receptor in prostate cancer cells. Food Funct.
5:2922–2930. 2014.
|
|
2
|
Lewandowska U, Owczarek K, Szewczyk K,
Podsędek A, Koziołkiewicz M and Hrabec E: Influence of polyphenol
extract from evening primrose (Oenothera paradoxa) seeds on
human prostate and breast cancer cell lines. Postepy Hig Med Dosw
(Online). 68:110–118. 2014.
|
|
3
|
Stagos D, Amoutzias GD, Matakos A, Spyrou
A, Tsatsakis AM and Kouretas D: Chemoprevention of liver cancer by
plant polyphenols. Food Chem Toxicol. 50:2155–2170. 2012.
|
|
4
|
Di Domenico F, Foppoli C, Coccia R and
Perluigi M: Antioxidants in cervical cancer: Chemopreventive and
chemotherapeutic effects of polyphenols. Biochim Biophys Acta.
1822:737–747. 2012.
|
|
5
|
Abbas A, Patterson W III and Georgel PT:
The epigenetic potentials of dietary polyphenols in prostate cancer
management. Biochem Cell Biol. 91:361–368. 2013.
|
|
6
|
Haraguchi T, Kayashima T, Okazaki Y, Inoue
J, Mineo S, Matsubara K, Sakaguchi E, Yanaka N and Kato N: Cecal
succinate elevated by some dietary polyphenols may inhibit colon
cancer cell proliferation and angiogenesis. J Agric Food Chem.
62:5589–5594. 2014.
|
|
7
|
Lall RK, Syed DN, Adhami VM, Khan MI and
Mukhtar H: Dietary polyphenols in prevention and treatment of
prostate cancer. Int J Mol Sci. 16:3350–3376. 2015.
|
|
8
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011.
|
|
9
|
Siegel R, Miller KD and Jemal A: Cancer
statistics, 2015. CA: A Cancer Journal for Clinicians. 65:5–29.
2015.
|
|
10
|
Vargas-Hernández VM, Moreno-Eutimio MA,
Acosta-Altamirano G and Vargas-Aguilar VM: Management of recurrent
epithelial ovarian cancer. Gland Surg. 3:198–202. 2014.
|
|
11
|
Cohen JG, White M, Cruz A and
Farias-Eisner R: In 2014, can we do better than CA125 in the early
detection of ovarian cancer? World J Biol Chem. 5:286–300.
2014.
|
|
12
|
Kigawa J: New strategy for overcoming
resistance to chemotherapy of ovarian cancer. Yonago Acta Med.
56:43–50. 2013.
|
|
13
|
Ott I and Gust R: Non platinum metal
complexes as anti-cancer drugs. Arch Pharm (Weinheim). 340:117–126.
2007.
|
|
14
|
Ayyagari VN and Brard L: Bithionol
inhibits ovarian cancer cell growth in vitro - studies on
mechanism(s) of action. BMC Cancer. 14:612014.
|
|
15
|
Agarwal R and Kaye SB: Ovarian cancer:
Strategies for overcoming resistance to chemotherapy. Nat Rev
Cancer. 3:502–516. 2003.
|
|
16
|
Luo H, Jiang BH, King SM and Chen YC:
Inhibition of cell growth and VEGF expression in ovarian cancer
cells by flavonoids. Nutr Cancer. 60:800–809. 2008.
|
|
17
|
Luo H, Rankin GO, Juliano N, Jiang BH and
Chen YC: Kaempferol inhibits VEGF expression and in vitro
angiogenesis through a novel ERK-NFκB-cMyc-p21 pathway. Food Chem.
130:321–328. 2012.
|
|
18
|
Chen AY and Chen YC: A review of the
dietary flavonoid, kaempferol on human health and cancer
chemoprevention. Food Chem. 138:2099–2107. 2013.
|
|
19
|
Kuo CL, Lai KC, Ma YS, Weng SW, Lin JP and
Chung JG: Gallic acid inhibits migration and invasion of SCC-4
human oral cancer cells through actions of NF-κB, Ras and matrix
metal-loproteinase-2 and -9. Oncol Rep. 32:355–361. 2014.
|
|
20
|
You BR, Kim SZ, Kim SH and Park WH: Gallic
acid-induced lung cancer cell death is accompanied by ROS increase
and glutathione depletion. Mol Cell Biochem. 357:295–303. 2011.
|
|
21
|
Cedó L, Castell-Auví A, Pallarès V, Macià
A, Blay M, Ardévol A, Motilva MJ and Pinent M: Gallic acid is an
active component for the anticarcinogenic action of grape seed
procyanidins in pancreatic cancer cells. Nutr Cancer. 66:88–96.
2014.
|
|
22
|
Lu Y, Jiang F, Jiang H, Wu K, Zheng X, Cai
Y, Katakowski M, Chopp M and To SS: Gallic acid suppresses cell
viability, proliferation, invasion and angiogenesis in human glioma
cells. Eur J Pharmacol. 641:102–107. 2010.
|
|
23
|
Zhao B and Hu M: Gallic acid reduces cell
viability, proliferation, invasion and angiogenesis in human
cervical cancer cells. Oncol Lett. 6:1749–1755. 2013.
|
|
24
|
He Z, Li B, Rankin GO, Rojanasakul Y and
Chen YC: Selecting bioactive phenolic compounds as potential agents
to inhibit proliferation and VEGF expression in human ovarian
cancer cells. Oncol Lett. 9:1444–1450. 2015.
|
|
25
|
Jafari S, Saeidnia S and Abdollahi M: Role
of natural phenolic compounds in cancer chemoprevention via
regulation of the cell cycle. Curr Pharm Biotechnol. 15:409–421.
2014.
|
|
26
|
Carocho M and Ferreira IC: The role of
phenolic compounds in the fight against cancer - a review.
Anticancer Agents Med Chem. 13:1236–1258. 2013.
|
|
27
|
Manach C, Williamson G, Morand C, Scalbert
A and Rémésy C: Bioavailability and bioefficacy of polyphenols in
humans. I. Review of 97 bioavailability studies. Am J Clin Nutr.
81(Suppl 1): 230S–242S. 2005.
|
|
28
|
Roberts AT, Martin CK, Liu Z, Amen RJ,
Woltering EA, Rood JC, Caruso MK, Yu Y, Xie H and Greenway FL: The
safety and efficacy of a dietary herbal supplement and gallic acid
for weight loss. J Med Food. 10:184–188. 2007.
|
|
29
|
Yamamizu K, Furuta S, Hamada Y, Yamashita
A, Kuzumaki N and Narita M: Dio K, Katayama S, Nagase H, Yamashita
JK, et al κ opioids inhibit tumor angiogenesis by suppressing VEGF
signaling. Sci Rep. 3:32132013.
|
|
30
|
Huang KF, Yang HY, Xing YM, Lin JS and
Diao Y: Recombinant human kallistatin inhibits angiogenesis by
blocking VEGF signaling pathway. J Cell Biochem. 115:575–584.
2014.
|
|
31
|
Strathdee G, MacKean MJ, Illand M and
Brown R: A role for methylation of the hMLH1 promoter in loss of
hMLH1 expression and drug resistance in ovarian cancer. Oncogene.
18:2335–2341. 1999.
|
|
32
|
Luo H, Li B, Li Z, Cutler SJ, Rankin GO
and Chen YC: Chaetoglobosin K inhibits tumor angiogenesis through
downregulation of vascular epithelial growth factor-binding
hypoxia-inducible factor 1α. Anticancer Drugs. 24:715–724.
2013.
|
|
33
|
Mandl M, Kapeller B, Lieber R and Macfelda
K: Hypoxia-inducible factor-1β (HIF-1β) is upregulated in a
HIF-1α-dependent manner in 518A2 human melanoma cells under hypoxic
conditions. Biochem Biophys Res Commun. 434:166–172. 2013.
|
|
34
|
Li H, Zeng J and Shen K: PI3K/AKT/mTOR
signaling pathway as a therapeutic target for ovarian cancer. Arch
Gynecol Obstet. 290:1067–1078. 2014.
|
|
35
|
Park ST, Kim BR, Park SH, Lee JH, Lee EJ,
Lee SH and Rho SB: Suppression of VEGF expression through
interruption of the HIF 1α and Akt signaling cascade modulates the
anti angiogenic activity of DAPK in ovarian carcinoma cells. Oncol
Rep. 31:1021–1029. 2014.
|
|
36
|
Mao Y, Xu J, Song G, Zhang N and Yin H:
Twist2 promotes ovarian cancer cell survival through activation of
Akt. Oncol Lett. 6:169–174. 2013.
|
|
37
|
Dobbin ZC and Landen CN: The importance of
the PI3K/AKT/mTOR pathway in the progression of ovarian cancer. Int
J Mol Sci. 14:8213–8227. 2013.
|
|
38
|
Jiang BH and Liu LZ: PI3K/PTEN signaling
in angiogenesis and tumorigenesis. Adv Cancer Res. 102:19–65.
2009.
|