Introduction
Bladder cancer, second to prostate cancer, is a
commonly diagnosed cancer of the genito-urinary system. According
to 2014 cancer statistics of the American Cancer Society (ACS),
141,610 new cases of bladder cancer and 30,350 cancer-related
deaths were estimated in the USA in the genito-urinary system
(1). The most common type of
bladder cancer tissue is transitional cell carcinoma, which
accounts for more than 90% of all bladder carcinoma cases (2). Additionally, ~70% transitional cell
carcinomas are superficial tumors. Transurethral resection (TUR) is
the primary regimen for patients with superficial bladder tumors
(stages Ta, T1 or Tis) (3). However, the recurrence rate within 5
years after TUR is as high as 50–70% (4), and 10–20% of them progressed to
muscle-invasive disease (5). The
anthracycline epirubicin (EPI) is the first line treatment
routinely used in patients with bladder cancer after TUR (6). Although prophylactic EPI can
effectively reduce the recurrence rate of bladder cancer after TUR,
the acquired drug resistance still remains a serious issue in
bladder cancer patients.
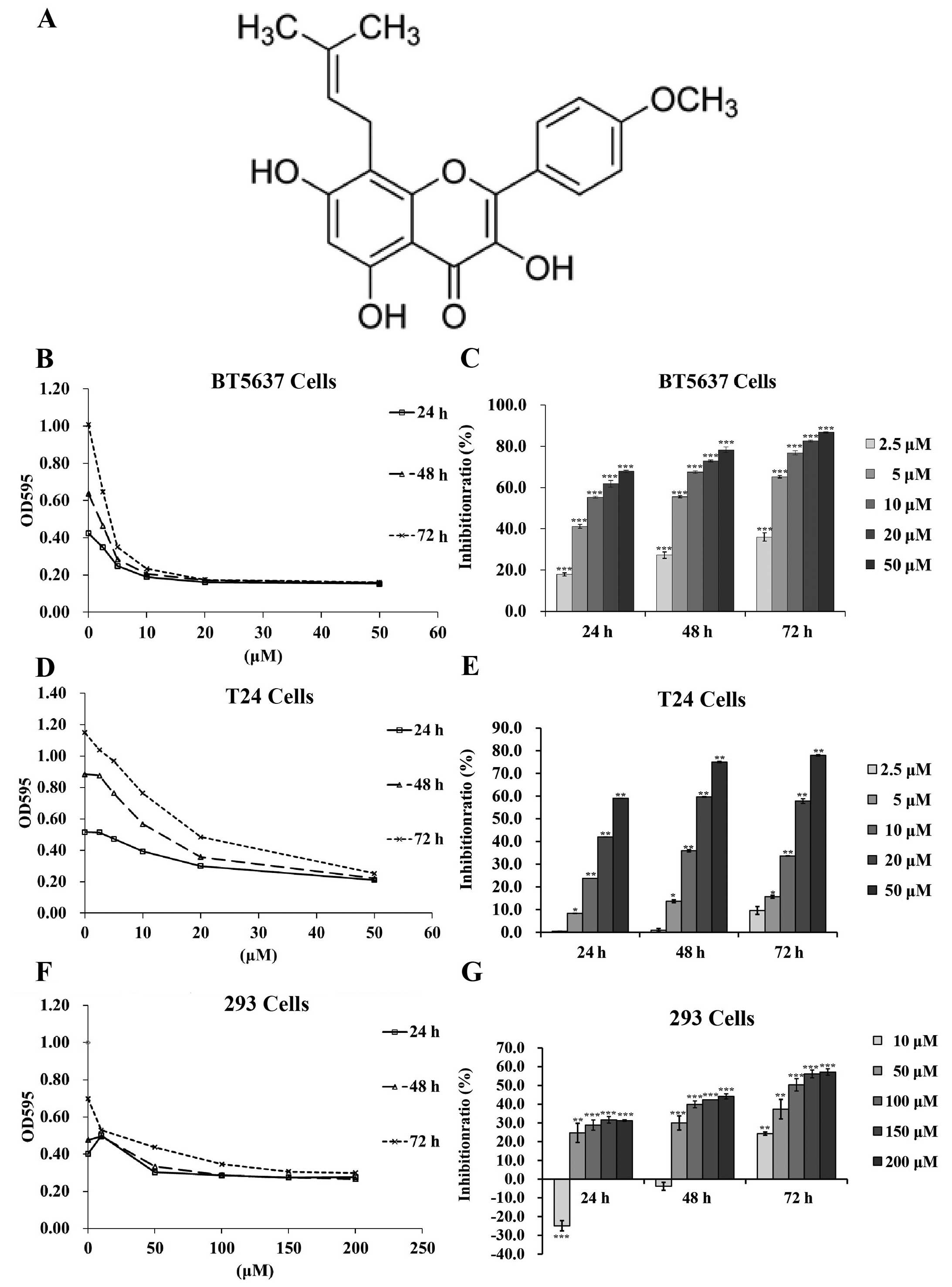
Icaritin (Fig. 1A)
is a hydrolytic form of icariin which is one of the traditional
Chinese herbals (7). Icaritin has
been shown to have a wide range of pharmacological and biological
activities, including stimulating neuronal differentiation via
estrogen-dependent pathway (8,9),
promoting differentiation of embryonic stem cells into
cardiomyocytes (10,11), inhibiting osteoclast differentiation
(12) and promoting apoptosis and
suppressing proliferation of prostatic smooth muscle cells
(13). In addition, numerous
studies have demonstrated that icaritin effectively promoted
apoptosis, inhibited cell proliferation and induced cell cycle
arrest in breast, endometrial and prostate cancer, and renal cancer
cells (14–18). Icaritin is expected to work as an
effective antitumor agent to regulate cancer cell growth via
various pathways. However, the function of icaritin against bladder
cancer has not been studied yet.
Autophagy has dual roles in cancer, acting as a
tumor suppressor and as a mechanism of cell survival that can
promote the growth of established tumors (19). In normal cells, a well regulated
autophagy may prevent the accumulation of damaged proteins and
organelles to protect cells from apoptosis or tumorigenesis.
However, in tumor cells, autophagy will convey a pro-survival
signal to adapt tumor cells to various adverse conditions, such as
starvation and chemotoxicity. Autophagy is upregulated upon
interferon-α (IFN-α) treatment to induce IFN-α resistance in
bladder cancer cells (20). In
addition, autophagy protects breast cancer cells from EPI-induced
apoptosis and facilitates epirubicin-resistance development
(21). The p53-induced glycolysis
and apoptosis regulator (TIGAR) inhibits autophagy, resulting in
reduced sensitivity of cancer cells to epirubicin treatment
(22). Several studies have
reported that apoptosis of cancer cells is markedly enhanced by
pharmacological or genetic mediated inhibition of autophagy
(23–25). Furthermore, autophagy inhibitors
such as 3-methyladenine or inhibition of autophagy regulatory
pathways have been proved to sensitize cancer cells to
chemotherapeutic agents (20–22).
The primary objective of the present investigation
was to explore the antitumor molecular mechanism of icaritin in the
treatment of human bladder cancer cells. To better understand the
function of icaritin in treating bladder cancer, we studied the
effects of icaritin on proliferation, apoptosis and autophagy of
human bladder cancer BT5637 and T24 cells, and explored the
sensitivity of cancer cells to EPI treatment.
Materials and methods
Reagents and antibodies
Icaritin (purity 98%; Shanghai Yuanye Biotechnology
Co., Ltd., China) was dissolved in dimethylsulfoxide (DMSO)
solution to reach a concentration of 50 mM, and then stored at
−20°C before use. The stock solution was diluted to the final
concentration using correspondent cell culture medium, and the
final DMSO concentration in experimental culture medium was
<0.1%. Therefore, 0.1% DMSO was used as control vehicle
throughout the present study. Epirubicin was purchased from AdooQ
BioScience (10 mg). The antibodies of ATG3 (#3415), ATG5 (#8540),
ATG7 (#2631) and LC3B (#3868) were purchased from Cell Signaling
Technology. GAPDH was purchased from ProteinTech Group
(10494-1-AP).
Cell culture
The bladder cancer cell line BT5637 and T24 and the
normal renal cell line HEK293 were purchased from the Cell Bank of
Chinese Academy of Science. BT5637 cells were cultured in RPMI-1640
culture medium (Hyclone) with 10% fetal bovine serum (FBS), 1%
non-essential amino acid (100X) and 2 g/l glucose. T24 cells were
cultured in McCoys' 5A culture medium (Sigma) with 10% FBS. HEK293
cells were cultured in DMEM culture medium (HyClone) with 10% FBS
and 2 mmol/l glutamine. All these cells were cultured in a
humidified atmosphere of 95% air and 5% CO2 at 37°C.
Cell proliferation assay
The
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT)
assay was carried out to evaluate the viability and proliferation
of cells. A total of 6×103 cells/well for BT5637 and
HEK293 cells and 4×103 cells/well for T24 cells were,
respectively, seeded into a 96-well plate, and incubated in
respective culture medium for 24 h at 37°C. Then culture medium was
replaced with fresh culture medium containing varying
concentrations of icaritin, and these cells were incubated for 24,
48 and 72 h. At each time point, cells of each well were added with
20 µl of 5 mg/ml MTT solution (Sigma). After 6-h incubation
at 37°C, the formazan crystals were dissolved by adding 100
µl DMSO into each well. The absorbance rate was measured at
570 nm using a spectrophotometer (Epoch Synergy 2; BioTek, USA).
Each experiment was carried out in triplicates.
Colony formation assay
BT5637 and T24 cells were plated into 6-well plates
(6×102 cells/well) in triplicate and were incubated for
24 h at 37°C. The culture medium was then removed and fresh culture
medium containing varying concentrations of icaritin was added into
each well. After 48-h incubation at 37°C, the special culture
medium was changed to culture medium without icaritin. Cell culture
was terminated when most colonies contained >50 cells.
Subsequently, cells were washed twice with phosphate-buffered
saline (PBS), fixed in 1 ml paraformaldehyde for 30 min, stained
with 500 µl Giemsa dye for 20 min and washed again with
ddH2O. Individual colonies were photographed using a
microscope, the plates were photographed using a digital camera,
and the colonies with >50 cells were counted. Each experiment
was repeated three times.
Measurement of mitochondrial membrane
potential (MMP)
JC-1 (Beyotime) staining assay as previously
described (26) was carried out to
measure MMP. A total of 2×104 cells/well BT5637 or T24
cells were seeded into a 6 cm plate in triplicate and cultured at
37°C. After 24-h incubation, the culture medium was changed to
fresh culture medium containing varying concentrations of icaritin
and then cells were incubated for 72 h at 37°C. At the designated
time point, cells were trypsinized, resuspended, incubated with
special culture medium containing JC-1 staining solution for 30 min
at 37°C, stained, washed twice using staining buffer and
resuspended with 500 µl staining buffer. Healthy cells with
high-MMP in the form of JC-1 aggregates showed red fluorescence,
and apoptotic cells with low-MMP in the form of JC-1 monomer
emitted green fluorescence only. Finally, the stained cells were
analyzed using flow cytometry (BD Biosciences).
Western blotting
The T24 and BT5637 cells were previously treated
with five concentrations of icaritin for 24, 48 and 72 h,
respectively. Proteins were extracted in lysis buffer. Following
quantification by ultraviolet spectrophotometry, ~30–60 µg
total proteins in each lane were separated by SDS-PAGE (10 and 12%
gels), and electrophoretically transferred onto nitrocellulose
membranes. The membranes were blocked for 1 h in 5% non-fat milk in
TBS/T and then incubated with various autophagy antibodies in 3%
non-fat milk at 4°C overnight (1:500; Cell Signaling Technology).
The next day, the membranes were washed three times for 10 min,
incubated in secondary antibody of anti-rabbit HRP (1:10,000),
washed again three times for 10 min, and finally exposed to
enhanced chemiluminescence (ECL) detection reagents (Amersham
Pharmacia Biotech), and photographed.
Synergistic effect
The synergistic effect of drug combinations was
evaluated by Chou-Talalay combination index (CI) equation as
follows: CI = Da/(Dx)a +
Db/(Dx)b (27), where Da,
(Dx)a, Db and
(Dx)b indicate the IC50 value of
drug A and B combination, drug A alone, drug B and A combination
and drug B alone, respectively. CI value of <1, 1, or >1
indicate a synergic, additive, or antagonistic effect of two drugs,
respectively.
Statistical analysis
The SPSS 19.0 (SPSS, Inc.) was used to carry out
statistical analyses. The data are shown as mean ± SD.
Statistically significant differences between groups were analyzed
using Student's t-test and P<0.05 was considered to indicate a
statistical significant result.
Results
Icaritin time- and dose-dependently
inhibits proliferation of bladder cancer cells
To explore the effect of icaritin in bladder cancer
cells, a dose-time-effect study was performed in BT5637 and T24
cells treated with concentration gradient of icaritin (2.5, 5, 10,
20 and 50 µM) for 24, 48 and 72 h. As shown in Fig. 1B–F, icaritin inhibited the
proliferation of both BT5637 and T24 cells dose- and
time-dependently, as compared with DMSO vehicle control.
To verify the specificity of icaritin on bladder
cancer cells rather than on normal human cells, we explored the
IC50 of icaritin on normal human HEK293 cells. As shown
in Fig. 1F and G, cell
proliferation was minimally suppressed in 293 cells after icaritin
treatment for 24 and 48 h. The IC50 of icaritin on 293
cells at 72 h was 126.58 µM. Whereas, the IC50 of
icaritin was 11.6, 7.21 and 4.46 µM on BT5637 cells; 36.8,
21.7 and 20.3 µM on T24 cells at 24, 48 and 72 h,
respectively, indicating that cancer cells have a higher
sensitivity towards icaritin than normal human cells, and that
icaritin functioned as a promising anticancer agent with low
cytotoxicity on normal cells.
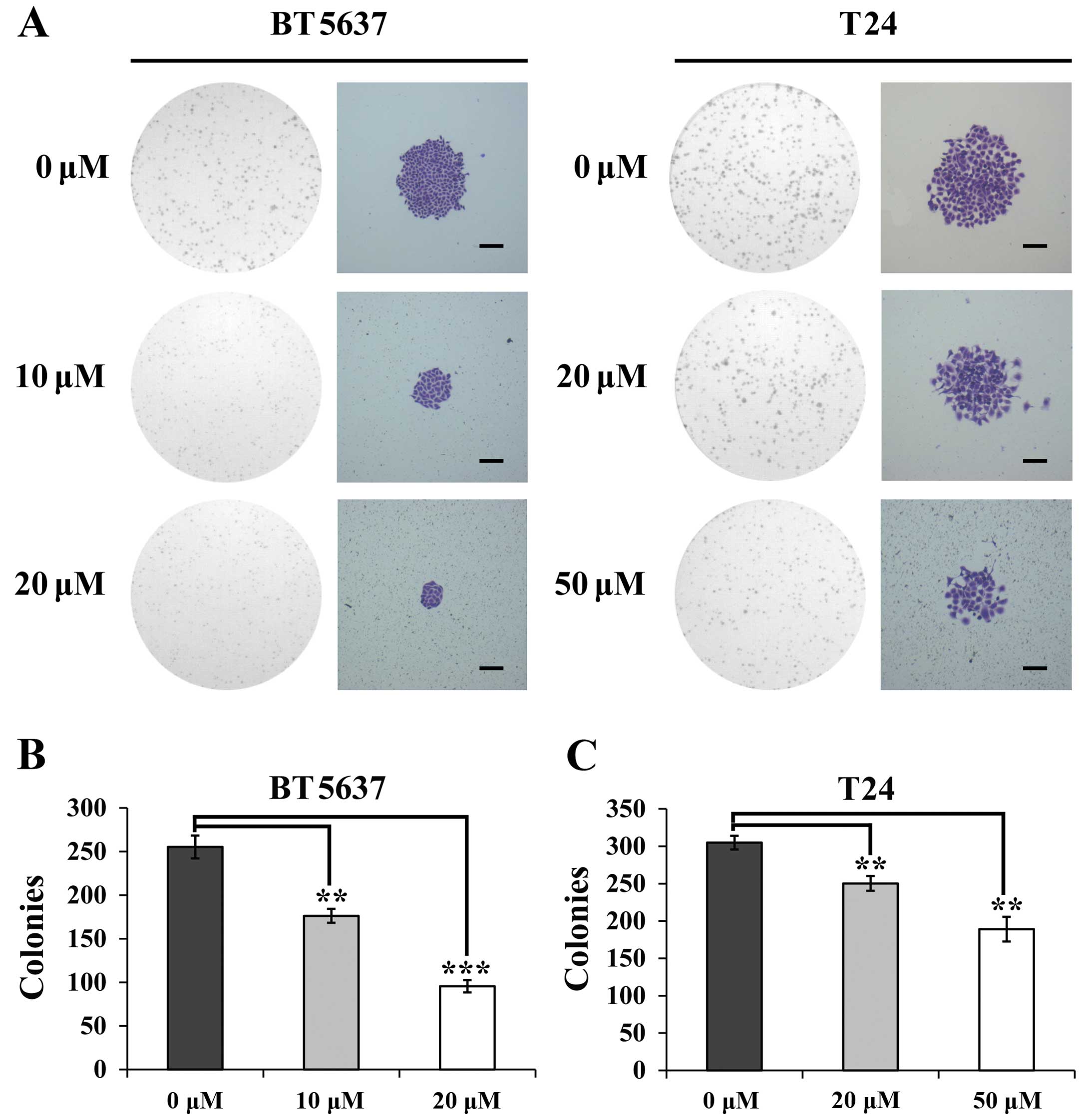
Icaritin reduces the capacity of colony
formation of bladder cancer cells
To confirm the effect of icaritin on colony-forming
ability of bladder cancer cells, cell colony formation assay was
performed on BT5637 and T24 cells treated with various
concentrations of icaritin for 48 h. As shown in Fig. 2A, the sizes of colony and the
numbers of cells/colony in icaritin groups were smaller as compared
with the DMSO vehicle control, particularly for BT5637 cells.
Moreover, the result showed that the higher concentration of
icaritin, the stronger the inhibitory effect. In addition, the
number of colonies of BT5637 cells treated with icaritin at the
concentrations of 10 and 20 µM was 176.3±8.0 and 95.7±7.1,
respectively, and that in T24 cells treated with icaritin at the
concentrations of 20 and 50 µM was 250.3±10 and 189.0±16.5,
respectively, which was significantly decreased comparing to the
DMSO vehicle groups in BT5637 and T24 cells (255.3±13.0 and
305±9.2, respectively) (Fig. 2B).
These results suggest that icartin suppressed the capacity of
colony formation of bladder cancer cells.
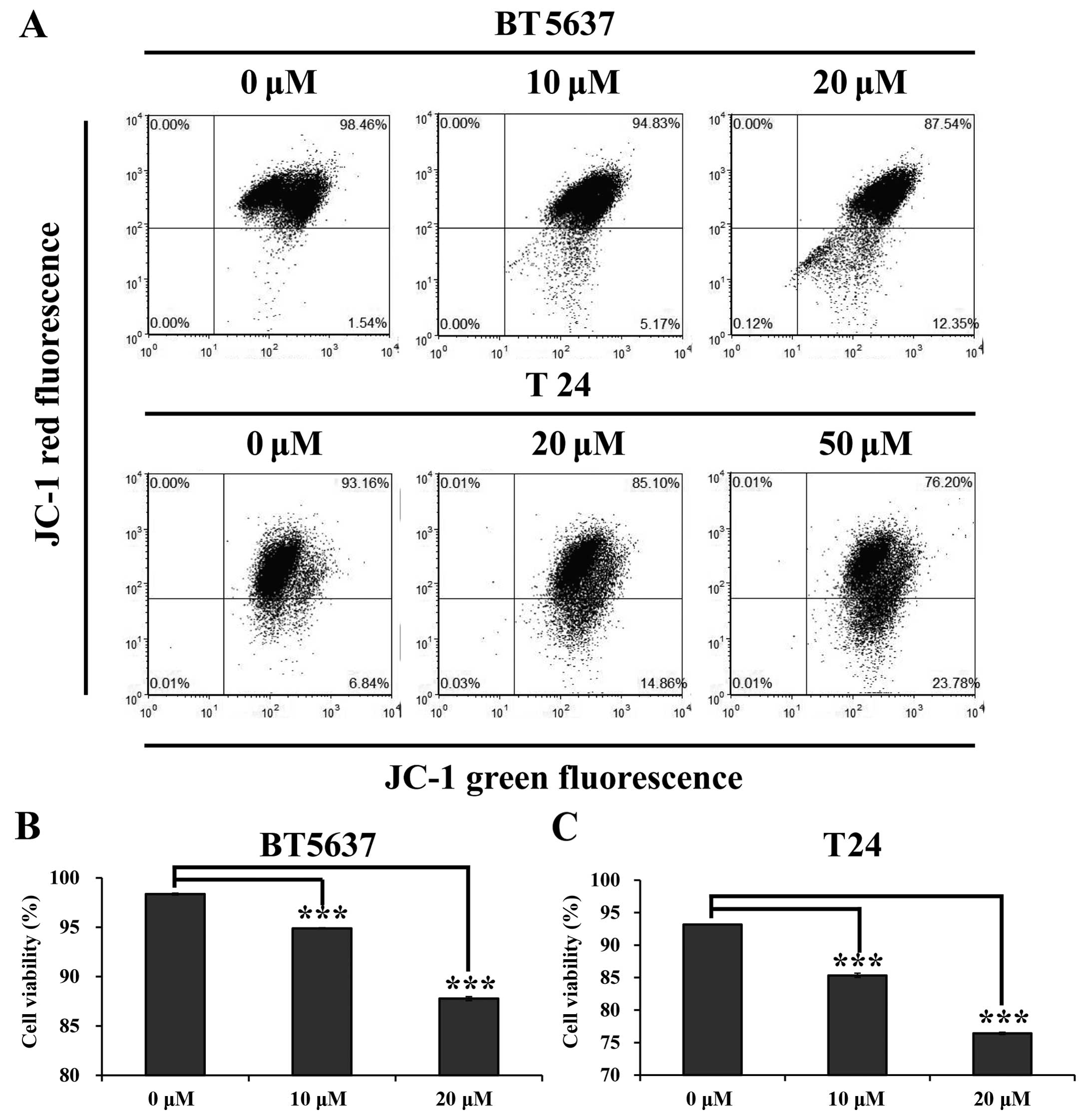
Icaritin reduces mitochondrial membrane
potential (MMP) in bladder cancer cells
Mitochondria, known as 'the power house', are
important cellular structures that generate adenosine triphosphate
(ATP) to supply most cellular functions. They are closely
associated with cell cycle progression, cell growth and cell death
(28). Therefore, we carried out
JC-1 staining assay to evaluate whether icaritin could affect
mitochondrial function in BT5637 and T24 cells. The result showed
that MMP was reduced in BT5637 and T24 cells treated with icaritin
(Fig. 3A). As shown in Fig. 3B and C, the cell viability in BT5637
cells were significantly decreased in 10 and 20 µM icaritin
groups as compared with the DMSO vehicle control (94.88±0.06 and
87.77±0.20 vs. 98.35±0.10, P<0.001), and a similar result was
seen in T24 cells treated with 20 and 50 µM icaritin
(85.34±0.32 and 76.4±10.18 vs. 93.15±0.02, P<0.001), indicating
that the cell viability of bladder cancer cells was significantly
inhibited after icaritin treatment in a dose-dependent manner.
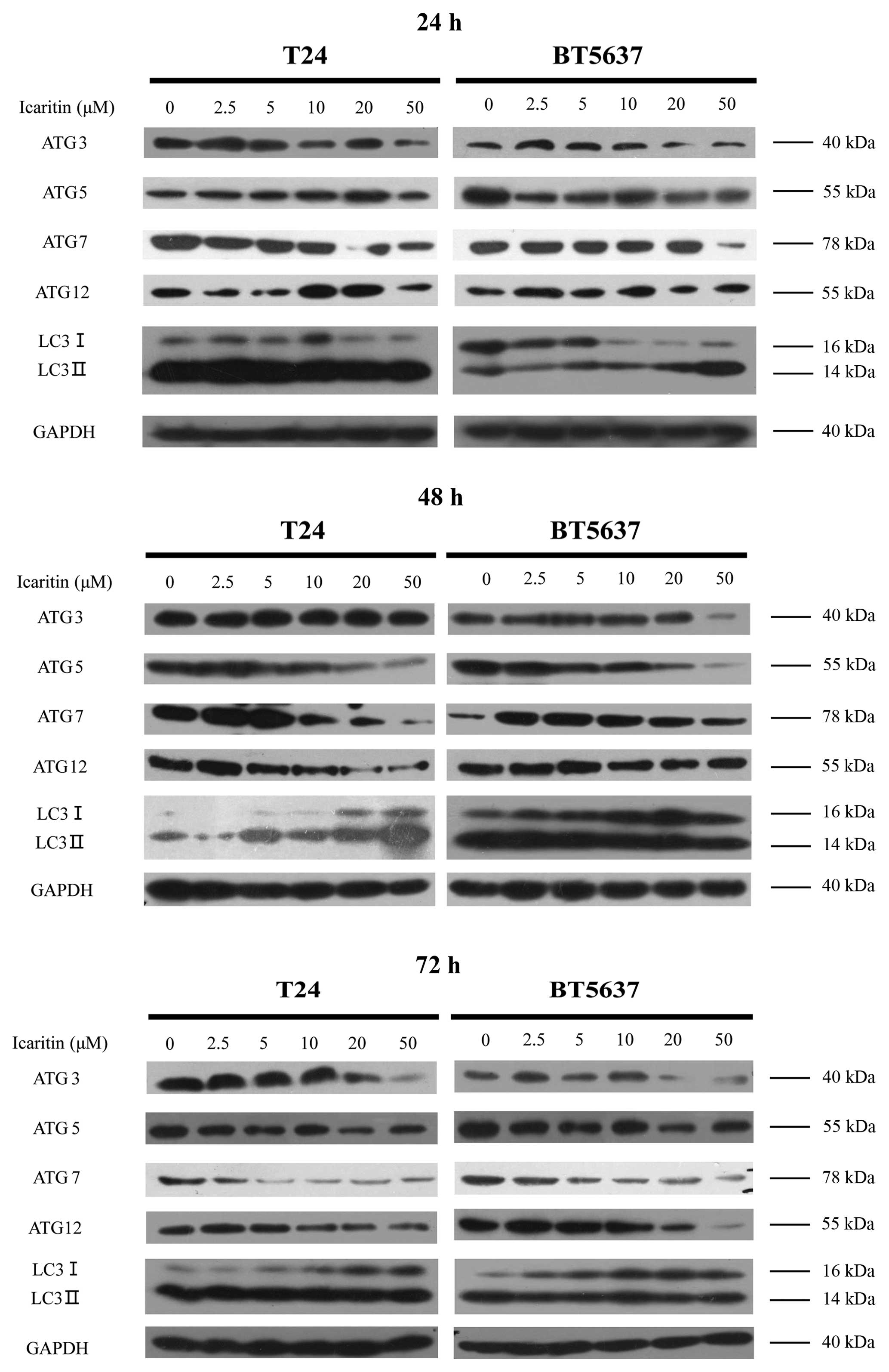
Icaritin inhibits autophagy of bladder
cancer cells
There was no significant change in cell cycle arrest
and apoptosis of bladder cancer cells treated with icaritin (data
not shown). To verify other potential mechanism of icaritin-induced
cell inhibition, we examined the autophagy pathways and postulated
that autophagy, which plays a vital role in cleaning injured
mitochondria, may act as a major factor in regulating the growth of
bladder cancer cells treated with icaritin. Using western blotting,
major autophagy proteins including ATG3, ATG5, ATG7, ATG12 and LC3
were investigated to evaluate the autophagy levels of BT5637 and
T24 treated with icaritin at 24, 48 and 72 h. Given the lack of the
duration of drug action (icaritin treatment for 24 h), both bladder
cancer T24 and BT5637 cells showed a slight difference in autophagy
or even LC3 was initially upregulated in BT5637 (Fig. 4A). After 48-h treatment, the
conversion of LC3-I to LC3-II was reduced (Fig. 4B), indicating that inhibition of
autophagy in BT5637 and T24 cells treated with icaritin was
initiated. Comparing with the DMSO vehicle control, autophagy of
BT5637 and T24 cells significantly decreased in a dose-dependent
manner, particularly at 48 and 72 h after 72-h icaritin treatment.
The above results suggest that icaritin inhibited the activation of
constitutive autophagy.
Incaritin sensitizes bladder cancer cells
to EPI by inhibiting EPI-induced autophagy
Knowing that inhibition of autophagy could sensitize
cancer cells to chemotherapeutic agents (21,22),
we aimed to clarify whether combination of incaritin and EPI for
treatment of BT5637 and T24 cells had a synergistic effect on
cancer cells. To clarify this, we treated BT5637 and T24 bladder
cancer cells with a combination of icaritin and EPI for 24 h and
then detected the proliferation of these cells by MTT assay. The
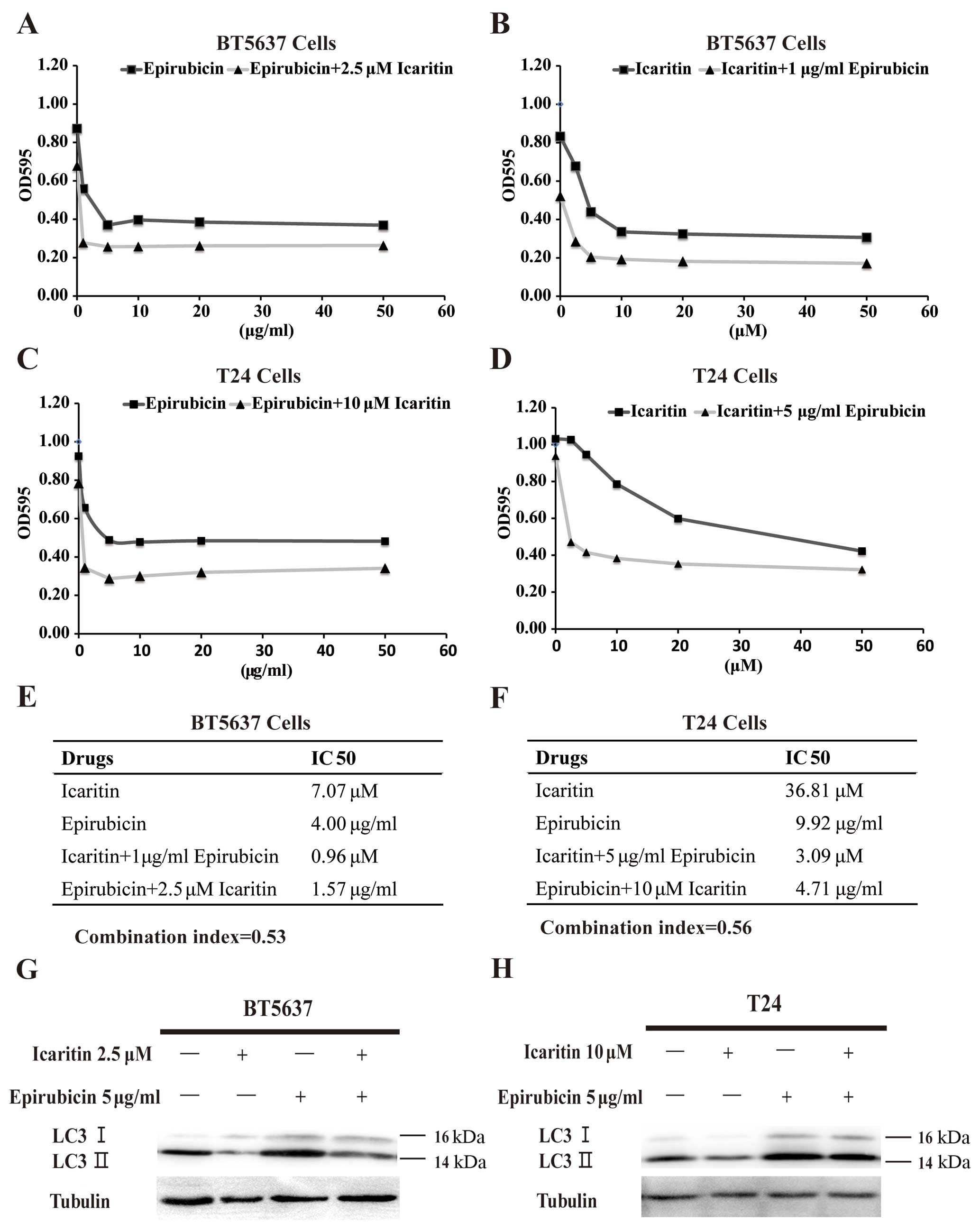
results in Fig. 5A and B showed
that the IC50 value of icaritin alone (7.07 µM)
and EPI alone (4.00 µg/ml) in inhibiting BT5637 cells was
significantly higher than that of icaritin in combination with 1.00
µg/ml EPI (0.96 µM) and EPI in combination with 2.5
µM icaritin (1.57 µg/ml). The same interesting
results were also observed in T24 cells (Fig. 5C and D). To confirm whether
combination of incaritin and EPI had a synergistic effect, we
adopted the Chou-Talalay combination index (CI) equation. Based on
the calculations, the CI value of icaritin and EPI combination in
treating BT5637 and T24 cells was 0.53 and 0.56, respectively
(Fig. 5E and F), indicating that
icaritin and EPI acted synergistically to suppress the
proliferation of bladder cancer cells. To verify our result
concerning the synergistic antitumor effect, western blotting was
carried out to confirm whether incaritin inhibited EPI-induced
autophagy and enhanced the susceptibility of bladder cancer cells
to EPI. The result showed that icaritin not only suppressed the
production of LC3-I as an autophagic marker in cytoplasm but
inhibited the conversion of LC3-I to LC3-II (Fig. 5E and F), suggesting that icaritin
could sensitize bladder cancer cells to epirubicin treatment via
downregulating EPI-induced autophagy.
Discussion
Bladder cancer is notorious for its high recurrence
rate. Although a routine intravesical perfusion with EPI after
surgery may reduce the recurrence rate, the acquired resistance to
EPI is common in cancer patients (29). Therefore, increased attention has
been paid to uncover the mechanism of drug resistance, and seeking
novel agents among natural herbal ingredients (29).
In the present study, in an effort to find effective
compounds to suppress the malignant proliferation of bladder cancer
cells, we screen a library of small compound extracted from herbal
medicine. We identified that icaritin may inhibit the proliferation
of T24 and BT5637 cells. Furthermore, as it has been reported that
icariin, an analogue of icaritin, may induce apoptosis in human
hepatoma SMMC-7721 cells via a ROS/JNK-dependent mitochondrial
pathway (30), we also investigated
the effect of icariin on bladder cancer cells. However, differently
to that previously reported in hepatoma cells, icariin showed a
very low cytotoxicity on bladder cancer cells compared to icaritin.
Therefore, icaritin was chosen to further explore the antitumor
molecular mechanism in bladder cancer cells. Icaritin significantly
inhibited the proliferation and colony formation of bladder cancer
cells in a time- and dose-dependent manner. BT5637 cells
(IC50 at 24, 48 and 72 h, 11.6, 7.21 and 4.46 µM)
showed higher sensitivity to icaritin than T24 cells
(IC50 at 24, 48 and 72 h, 36.8, 21.7 and 20.3
µM).
In order to explore the molecular mechanism how
icaritin inhibited the proliferation of BT5637 and T24 cells, we
analyzed the cell cycle distribution and apoptosis of both cells
using PI/Annexin V and flow cytometric analysis. However, the
results showed that icaritin hardly affected cell cycle
distribution or apoptosis in the cells (data not shown). However,
we discovered that mitochondrial membrane potential (MMP) of BT5637
and T24 cells was obviously impaired by icaritin treatment.
Mitochondria are known to play a vital role in cell growth as they
provide large amounts of ATP for various cellular activities
(27). Since tumor cells show
extremely active growth, large amounts of ATP are required for
tumor cells to support malignant cell proliferation (31). Therefore, decrease of mitochondrial
membrane potential may lead to a reduced ATP production and
suppressed cell proliferation rate. In the present study, growth
inhibitory effect of icaritin in bladder cancer cells may be
mediated through MMP suppression.
For eukaryotic cells, autophagy is a crucial pathway
to recycle organelles and proteins, especially injured
mitochondria. An elevated autophagic activity is utilized by tumor
cells to acquire malignant proliferation. The accumulation of
injured mitochondria in icaritin-treated bladder cancer cells
prompted us to analyze autophagy in these cells. During autophagy,
microtubule-associated protein 1A/1B-light chain 3 (LC3) is
conjugated to phosphatidylethanolamine to form
LC3-phosphatidylethanolamine (LC3-II)-conjugated by two consecutive
ubiquitylation-like reactions catalyzed by the E1-like enzyme Atg7
and the E2-like enzyme Atg3 (32,33).
LC3-II is recruited to autophagosomal membranes and degraded in
autolysosomes after being fused to lysosomes (32,33).
Therefore, the protein levels of ATG3, ATG7, LC3 and LC3-II have
been demonstrated to be reliable markers to monitor autophagy
(32,33). Moreover, ATG12, as the first
ubiquitin-like ATG protein to be found, could bind to and was
activated by ATG7, an E1-like enzyme (34). The ATG12 is sent to ATG10, an E2
enzyme, and finally forms a conjugation with ATG5, and then the
ATG12-ATG5 conjugation futher coacts with ATG16, a small
coiled-coil protein, and forms the ATG12-ATG5-ATG6 multimeric
complex (34). We found that
icaritin downregulated the expressions of ATG3, ATG7, ATG5, ATG12
and suppressed the transformation of LC3 to LC3-II, indicating that
icaritin inhibited autophagy of bladder cancer cells.
Previous studies reported that ERK and AKT signaling
are the dominant pathway for icaritin to inhibit cancer cells
growth in chronic myeloid leukemia, endometrial cancer renal cell
carcinoma and breast cancer cells (15,17,18,35).
Moreover, Li et al revealed that the apoptosis of human
hepatoma was also induced via icariin treatment with
ROS/JNK-dependent mitochondrial pathway (30). The above signaling pathways of
icaritin were explored in the present study, however, the
inhibition of bladder cancer cells undergoing icaritin treatment
had no correlation with the signaling (data was not shown). We
revealed that icaritin inhibited the autophagy pathway to effective
inhibition of bladder cancer cells.
Autophagy is activated in tumor cells compared to
normal cells, in response to cellular stress and/or increased
metabolic demands, leading to rapid cell proliferation and acquired
resistance to chemotherapies (19).
Therefore, autophagy which is differentially regulated in cancer
and normal cells may serve as a promising target for anticancer
therapy. We found that icaritin significantly inhibited the
proliferation of bladder cancer cells via downregulated autophagy.
However, the IC50 value of icaritin for normal human
HEK293 cells at 72 h was 28.4- and 6.2-fold higher than that for
BT5637 and T24 cells at 72 h, respectively, suggesting that human
bladder cancer BT5637 and T24 cells which have active autophagic
activity were more sensitive to icaritin compared to normal human
HEK293 cells, and that icaritin may serve as an anticancer agent
with low adverse effects.
In clinical practice, intravesical perfusion with
epirubicin is routinely used after transurethral resection to
prevent bladder cancer recurrence. Nevertheless, there exists an
even more serious problem regarding EPI-resistance which limits its
successful anticancer effect. In addition, adverse events
accompanying high-dose intravesical usage of epirubicin, including
chemical cystitis, urinary tract infection, hematuria and cardiac
adverse events, have also been shown as a problem for bladder
cancer patients (6). Therefore, a
combined usage of an agent that can improve the sensitivity of EPI
may reduce the side-effects due to high-dose EPI treatment. In the
present study, we found that when combined with icaritin, EPI
reached the same inhibitory effect on bladder cancer cells at a
much lower dose compared to EPI treatment alone, suggesting that
icaritin may serve as an adjuvant agent to boost EPI therapy.
Recently, Chittaranjan et al found a similar result that
EPI-resistant breast cancers cells treated with combination of EPI
and autophagy inhibitor (hydroxychloroquine) markedly enhanced the
inhibition of tumor growth comparing to those treated with EPI
alone (36).
Autophagy is a major way that tumor cells utilize to
develop chemoresistances to EPI. It is reported that EPI induces
autophagy in human breast cancer MCF-7 cells, and EPI-induced
autophagy protects MCF-7 cells from EPI-induced apoptosis (20). Also, EPI induces autophagy and
serves as a pro-survival mechanism in triple negative breast
cancers (TNBCs) (36). However, it
has not been reported whether EPI may cause autophagy in bladder
cancer. In the present study, we found that EPI treatment promoted
the conversion of LC3I to LC3II, a marker of autophagosome
(Fig. 5G and H), implying that EPI
may induce autophagy in bladder cancer cells. As identified in
Fig. 4 that icaritin acted as an
autophagy inhibitor, we tested whether icaritin can suppress
autophagy to increase EPI sensitivity in bladder cancer cells. The
results showed that EPI-inducing autophagy was suppressed by
icaritin treatment. Therefore, we found a novel compound icaritin
functioning as an autophagy inhibitor to restore the sensitivity of
bladder cancer to EPI.
In conclusion, the present study proposes that
icaritin inhibited the malignant proliferation of bladder cancer
cells through inhibition of autophagy. Moreover, as an autophagy
inhibitor, icaritin functioned synergistically with EPI to treat
bladder cancer cells by depressing EPI-induced autophagy. These
results provide reasonable evidence that the natural product
icaritin may prove to be a novel potent anticancer agent against
bladder cancer.
Acknowledgments
The present study was supported by grants from the
National Natural Science Foundation of China for Youths (no.
81202020), the National Natural Science Foundation of China (no.
81170637), the Shanghai Committee of Science and Technology General
Program for Medicine (no. 11JC1402302), the Military Fund for
Health Care (13BJZ29), and the Key Project of Science and
Innovation Foundation of Shanghai Ministry of Education
(14zz084).
Abbreviations:
|
DMSO
|
dimethylsulfoxide
|
|
EPI
|
anthracycline epirubicin
|
|
MMP
|
mitochondrial membrane potential
|
|
TUR
|
transurethral resection
|
References
|
1
|
Siegel R, Ma J, Zou Z and Jemal A: Cancer
statistics, 2014. CA Cancer J Clin. 64:9–29. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kaufman DS, Shipley WU and Feldman AS:
Bladder cancer. Lancet. 374:239–249. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Parekh DJ, Bochner BH and Dalbagni G:
Superficial and muscle-invasive bladder cancer: Principles of
management for outcomes assessments. J Clin Oncol. 24:5519–5527.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Herr HW: Natural history of superficial
bladder tumors: 10- to 20-year follow-up of treated patients. World
J Urol. 15:84–88. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rübben H, Lutzeyer W, Fischer N, Deutz F,
Lagrange W and Giani G: Natural history and treatment of low and
high risk superficial bladder tumors. J Urol. 139:283–285.
1988.PubMed/NCBI
|
|
6
|
Onrust SV, Wiseman LR and Goa KL:
Epirubicin: A review of its intravesical use in superficial bladder
cancer. Drugs Aging. 15:307–333. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wu H, Lien EJ and Lien LL: Chemical and
pharmacological investigations of Epimedium species: A survey. Prog
Drug Res. 60:1–57. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang Z, Zhang X, Wang H, Qi L and Lou Y:
Neuroprotective effects of icaritin against beta amyloid-induced
neurotoxicity in primary cultured rat neuronal cells via
estrogen-dependent pathway. Neuroscience. 145:911–922. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang Z, Wang H, Wu J, Zhu D, Zhang X, Ou
L, Yu Y and Lou Y: Enhanced co-expression of beta-tubulin III and
choline acetyltransferase in neurons from mouse embryonic stem
cells promoted by icaritin in an estrogen receptor-independent
manner. Chem Biol Interact. 179:375–385. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wo YB, Zhu DY, Hu Y, Wang ZQ, Liu J and
Lou YJ: Reactive oxygen species involved in prenylflavonoids,
icariin and icaritin, initiating cardiac differentiation of mouse
embryonic stem cells. J Cell Biochem. 103:1536–1550. 2008.
View Article : Google Scholar
|
|
11
|
Zhu DY and Lou YJ: Inducible effects of
icariin, icaritin, and desmethylicaritin on directional
differentiation of embryonic stem cells into cardiomyocytes in
vitro. Acta Pharmacol Sin. 26:477–485. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Huang J, Yuan L, Wang X, Zhang TL and Wang
K: Icaritin and its glycosides enhance osteoblastic, but suppress
osteoclastic, differentiation and activity in vitro. Life Sci.
81:832–840. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chen MF, Qi L, Li Y, Zu XB, Dai YQ and
Zhang P: Icaritin induces growth inhibition and apoptosis of human
prostatic smooth muscle cells in an estrogen receptor-independent
manner. Amino Acids. 38:1505–1513. 2010. View Article : Google Scholar
|
|
14
|
Hong J, Zhang Z, Lv W, Zhang M, Chen C,
Yang S, Li S, Zhang L, Han D and Zhang W: Icaritin synergistically
enhances the radio-sensitivity of 4T1 breast cancer cells. PLoS
One. 8:e713472013. View Article : Google Scholar
|
|
15
|
Tong JS, Zhang QH, Huang X, Fu XQ, Qi ST,
Wang YP, Hou Y, Sheng J and Sun QY: Icaritin causes sustained
ERK1/2 activation and induces apoptosis in human endometrial cancer
cells. PLoS One. 6:e167812011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Huang X, Zhu D and Lou Y: A novel
anticancer agent, icaritin, induced cell growth inhibition, G1
arrest and mitochondrial transmembrane potential drop in human
prostate carcinoma PC-3 cells. Eur J Pharmacol. 564:26–36. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Guo Y, Zhang X, Meng J and Wang ZY: An
anticancer agent icaritin induces sustained activation of the
extracellular signal-regulated kinase (ERK) pathway and inhibits
growth of breast cancer cells. Eur J Pharmacol. 658:114–122. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Li S, Priceman SJ, Xin H, Zhang W, Deng J,
Liu Y, Huang J, Zhu W, Chen M, Hu W, et al: Icaritin inhibits
JAK/STAT3 signaling and growth of renal cell carcinoma. PLoS One.
8:e816572013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yang ZJ, Chee CE, Huang S and Sinicrope
FA: The role of autophagy in cancer: Therapeutic implications. Mol
Cancer Ther. 10:1533–1541. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sun WL, Chen J, Wang YP and Zheng H:
Autophagy protects breast cancer cells from epirubicin-induced
apoptosis and facilitates epirubicin-resistance development.
Autophagy. 7:1035–1044. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Xie JM, Li B, Yu HP, Gao QG, Li W, Wu HR
and Qin ZH: TIGAR has a dual role in cancer cell survival through
regulating apoptosis and autophagy. Cancer Res. 74:5127–5138. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chen WC, Hsu KY, Hung CM, Lin YC, Yang NS,
Ho CT, Kuo SC and Way TD: The anti-tumor efficiency of
pterostilbene is promoted with a combined treatment of Fas
signaling or autophagy inhibitors in triple negative breast cancer
cells. Food Funct. 5:1856–1865. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tang Q, Li G, Wei X, Zhang J, Chiu JF,
Hasenmayer D, Zhang D and Zhang H: Resveratrol-induced apoptosis is
enhanced by inhibition of autophagy in esophageal squamous cell
carcinoma. Cancer Lett. 336:325–337. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Cui Q, Tashiro S, Onodera S, Minami M and
Ikejima T: Autophagy preceded apoptosis in oridonin-treated human
breast cancer MCF-7 cells. Biol Pharm Bull. 30:859–864. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhang XQ, Dunner K Jr and Benedict WF:
Autophagy is induced by adenoviral-mediated interferon alpha
treatment in interferon resistant bladder cancer and normal
urothelial cells as a cell death protective mechanism but not by
the bystander factors produced. Cancer Gene Ther. 17:579–584. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sang H, Zhang L and Li J:
Anti-benzopyrene-7,8-diol-9,10-epoxide induces apoptosis via
mitochondrial pathway in human bronchiolar epithelium cells
independent of the mitochondria permeability transition pore. Food
Chem Toxicol. 50:2417–2423. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chou TC, Motzer RJ, Tong Y and Bosl GJ:
Computerized quantitation of synergism and antagonism of taxol,
topotecan, and cisplatin against human teratocarcinoma cell growth:
A rational approach to clinical protocol design. J Natl Cancer
Inst. 86:1517–1524. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
McBride HM, Neuspiel M and Wasiak S:
Mitochondria: More than just a powerhouse. Curr Biol. 16:R551–R560.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yang CC, Chen GW, Lu HF, Wang DY, Chen YS
and Chung JG: Paclitaxel (taxol) inhibits the arylamine
N-acetyltransferase activity and gene expression (mRNA NAT1) and
2-aminofluo-rene-DNA adduct formation in human bladder carcinoma
cells (T24 and TSGH 8301). Pharmacol Toxicol. 92:287–294. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li S, Dong P, Wang J, Zhang J, Gu J, Wu X,
Wu W, Fei X, Zhang Z, Wang Y, et al: Icariin, a natural flavonol
glycoside, induces apoptosis in human hepatoma SMMC-7721 cells via
a ROS/JNK-dependent mitochondrial pathway. Cancer Lett.
298:222–230. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Weinberg F and Chandel NS: Mitochondrial
metabolism and cancer. Ann NY Acad Sci. 1177:66–73. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mizushima N and Yoshimori T: How to
interpret LC3 immunoblotting. Autophagy. 3:542–545. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tanida I, Ueno T and Kominami E: LC3 and
Autophagy. Methods Mol Biol. 445:77–88. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yamaguchi M, Noda NN, Yamamoto H, Shima T,
Kumeta H, Kobashigawa Y, Akada R, Ohsumi Y and Inagaki F:
Structural insights into Atg10-mediated formation of the
autophagy-essential Atg12-Atg5 conjugate. Structure. 20:1244–1254.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhu J, Li Z, Zhang G, Meng K, Kuang W, Li
J, Zhou X, Li R, Peng H, Dai C, et al: Icaritin shows potent
anti-leukemia activity on chronic myeloid leukemia in vitro and in
vivo by regulating MAPK/ERK/JNK and JAK2/STAT3/AKT signalings. PLoS
One. 6:e237202011. View Article : Google Scholar
|
|
36
|
Chittaranjan S, Bortnik S, Dragowska WH,
Xu J, Abeysundara N, Leung A, Go NE, DeVorkin L, Weppler SA, Gelmon
K, et al: Autophagy inhibition augments the anticancer effects of
epirubicin treatment in anthracycline-sensitive and -resistant
triple-negative breast cancer. Clin Cancer Res. 20:3159–3173. 2014.
View Article : Google Scholar : PubMed/NCBI
|