Introduction
Aggressive cancer therapy including chemo- and
chemoradiotherapy can help patients with various malignancies to
achieve greater improvement in survival and prognosis. However,
these therapies frequently cause some serious adverse events. Of
these, oral mucositis is one of the most frequent and clinically
significant complications induced by chemotherapy, with or without
radiotherapy (1). Serious oral
mucositis, which involves both erythema and painful mucosal
ulceration, can cause difficulty in swallowing, dehydration,
malnutrition and increased risk for infection (2,3). These
clinical sequelae can limit the tolerated dose of chemotherapy,
leading to poor prognosis, and severely impair the health-related
quality of life (3,4). It has been reported that the incidence
of oral mucositis induced by conventional chemotherapy for
esophageal cancer is relatively high (35–60%) and that the DCF
regimen (docetaxel, cisplatin and 5-fluorouracil), which is
occasionally used for patients with esophageal cancer in Japan, is
one of the high-risk regimens for oral mucositis (3,5–7).
Unfortunately, there are few agents that have been confirmed to
reveal consistent protective action against chemo (chemoradio)
therapy-induced oral mucositis to date.
Involuntary weight loss is considered a hallmark of
advanced cancer, and esophageal cancer has been reported to have
the highest median percentage weight loss among the primary tumor
sites evaluated (8). Cancer
cachexia leads to body weight loss by reducing both lean and fat
mass, and muscle loss has recently emerged as a more serious
concern. Sarcopenia, which was initially defined as the loss of
skeletal muscle mass that occurs with aging, has also been
recognized as a clinically important phenomenon that is associated
with reduced quality of life and poor clinical outcome in malignant
conditions (9,10). There is mounting evidence that
sarcopenia or sarcopenic obese patients are independently
associated with a poor response to cancer therapy in various
malignancies, such as melanoma (11), renal cell (12), hepatic carcinoma (13), colorectal (14), pancreatic (15), breast (16) and lung cancer (17). However, there is limited knowledge
on the impact of sarcopenia on the prognostic value and response to
chemo (chemoradio) therapy for esophageal cancer. A recent study
demonstrated that body composition was markedly changed following
chemotherapy for esophageal cancer, and a significant reduction of
fat free mass (skeletal muscle) was observed during chemotherapy
and the incidence of sarcopenia increased from 57% before
chemotherapy to 78.7% after completion of chemotherapy (18). Counteraction of sarcopenia
progression during chemotherapy in some way (e.g. nutritional
supplement) is considered to be important, since it may attenuate
the toxicity of anticancer agents and improve the therapeutic
efficacy of chemo (chemoradio) therapy. Although supplementation
has been suggested as means of attenuating the reduction of fat
free mass during chemotherapy (19,20),
robust data supporting that supplemental intervention in cancer
patients improves sarcopenia progression during chemo (chemoradio)
therapy are currently lacking.
Elental® is one of the widely used
nutritional supplements and its special formula contains a variety
of amino acids, together with easily digestible nutritions,
minerals, vitamins and a major energy source, dextrin (Table I). Fat is present in a very small
amount, whereas L-glutamine is present at an especially high dose
(1.932 mg/package). Several reports have demonstrated that
glutamine supplementation decreases the incidence and severity of
chemo (chemoradio) therapy-induced mucositis (21–23).
Moreover, it has been reported that amino acids, such as leucine,
can stimulate muscle protein synthesis (24) and that supplementation of amino
acids improves or prevents sarcopenia in several diseases,
including cancer (25). These
observations led us to examine the preventive effects of Elental on
oral mucositis and sarcopenia progression during chemo (chemoradio)
therapy for esophageal cancer.
 | Table IComposition of Elental (one package =
80 g). |
Table I
Composition of Elental (one package =
80 g).
| Composition | Amount |
|---|
| Energy (kcal) | 300 |
| Carbohydrate
(g) |
| Dextrin | 63.41 |
| Fat (g) |
| Soy bean oil | 0.51 |
| Amino acid (g) | 14.1 |
| Amino acid
(mg) |
| L-Isoleucine | 642 |
| L-Leucine | 899 |
| Lysine
hydrochloride | 888 |
| L-Methionine | 648 |
|
L-Phenylalanine | 871 |
| L-Threonine | 523 |
| L-Tryptophan | 151 |
| L-Valine | 701 |
| L-Histidine
hydrochloride monohydrate | 501 |
| L-Arginine
hydrochloride | 1,125 |
| L-Alanine | 899 |
| L-Aspartic acid
magnesium potassium | 1,036 |
| L-Aspartic acid
sodium monohydrate | 867 |
| L-Glutamine | 1,932 |
| Aminoacetic
acid | 505 |
| L-Proline | 630 |
| L-Serine | 1,159 |
| L-Tyrosine | 110 |
| BCAA (mg) | 2,242 |
| BCAA
branched-chain amino acids | |
The primary endpoint of this trial was to explore
the potential preventive effect of the amino acid-rich elemental
diet Elental on chemo (chemoradio) therapy-induced oral mucositis
in patients with esophageal cancer. The impact of the anticatabolic
properties of Elental was also studied as a secondary endpoint.
Herein, we report that Elental nutrition could counteract
sarcopenia progression during chemo (chemoradio) therapy for
esophageal cancer, but we did not demonstrate efficacy against
chemo (chemoradio) therapy-induced oral mucositis.
Materials and methods
Patients
Participants with pathologically diagnosed primary
squamous cell carcinoma of the esophagus who were scheduled to
undergo chemotherapy or chemoradiotherapy were recruited. They
fulfilled the following eligibility criteria: age between 20 to 80
years; an Eastern Cooperative Oncology Group (ECOG) performance
status of 2 or less; at least six months since the last
chemotherapy or radiation therapy; and adequate hematologic,
hepatic, renal and cardiac function. The exclusion criteria were:
presence of uncontrolled infection; presence of severe
gastrointestinal stenosis or bleeding; requirement for total
parental nutrition; the use of other nutritional supplements; poor
control of diabetes or the use of insulin treatment.
Study design
This was a randomized, open label, phase 2 clinical
trial with patients randomized in a 1:1 allocation to one of two
arms: Arm 1 (control arm), azulene (Azunol® Gargle
liquid 4%; Nippon Shinyaku Co., Kyoto, Japan) oral rinse; Arm 2,
the elemental diet Elental®. Azulene was used as a
control agent since this compound is approved for oral mucositis
therapy in Japan. Randomization was stratified by treatment
(chemotherapy/chemoradiotherapy) and stage using a stratified
permuted block-randomization scheme. Azulene oral rinse was
prepared by pouring 5 drops of 4% liquid solution into 100 ml of
water. At the start of chemo (chemoradio) therapy, patients
underwent oral rinsing with azulene solution 3 times a day until
the end of the treatment course. Elental was administered at 80 g
(one package)/day to patients in Arm 2 from the start of the
treatment until the end of the treatment course. The composition of
Elental is shown in the Table
I.
The primary objective of the present study was to
estimate the incidence and severity of oral mucositis during one
cycle of treatment (4 weeks). Changes of nutrition indicators (e.g.
serum total protein, albumin, hemoglobin and body composition)
during chemo (chemoradio) therapy were also evaluated as secondary
objectives. The present study was approved by the ethics committee
of Kyoto Prefectural University of Medicine. The trial was designed
and conducted in accordance with the Helsinki Declaration and the
Ethical Guidelines for Clinical Research (the Ministry of Health,
Labor and Welfare, Japan). All participants provided written
informed consent. This trial was registered as the University
Hospital Medical Information Network (UMIN) Clinical Trial Registry
as ID: UMIN 000007960.
Assessment
Evaluation of oral mucositis and other
adverse events
The incidence of oral mucositis was evaluated and
the severity of all side-effects was graded by one researcher
according to the Common Terminology Criteria for Adverse Events
v.4.0 (CTCAE). Oral pain was assessed using a numerical rating
scale (NRS). The side-effects were checked every week and the worst
score obtained during the observation period (4 weeks after
commencing the treatment) was included in the data for
comparison.
Nutritional status assessment
Body composition measurement
Body composition was assessed at the initiation of
the chemo (chemoradio) therapy and ~4 weeks after commencing the
treatment, using multifrequency bioelectrical impedance with eight
tactile electrodes (InBody 720; Biospace Co., Ltd., Seoul, Korea).
With this method, body weight, body mass index (BMI), body fat mass
and lean body mass (LBM) were measured automatically and
simultaneously. The InBody has been reported to be an accurate
substitute for dual-energy X-ray absorptiometry (DXA) for the
measurement of body composition (26). The skeletal muscle index (SMI)
(kg/m2) is calculated as the appendicular skeletal
muscle mass divided by the square of the height. In the present
study, cut-off values for sarcopenia were based on the consensus
report from the Asian Working Group for Sarcopenia (AWGS) (27); (i.e., SMI: <7.0 kg/m2
for men and 5.7 kg/m2 for women using bioimpedance
analysis).
Blood test
Blood tests, such as serum total protein, albumin,
blood hemoglobin, C-reactive protein (CRP), erythrocyte, white
blood cell, neutrophil and lymphocyte counts were determined
routinely. Outcomes were assessed at the initiation of the chemo
(chemoradio) therapy and 4 weeks after commencing the
treatment.
Statistical analysis
Patients who received at least one dose of azulene
or Elental were included in the analyses. Based on the results of
previous studies (28–30), the difference between the incidences
of oral mucositis for two arms was assumed to be 35% (the incidence
for the azulene arm and Elental Arm being 50 and 15%,
respectively). With one-sided type I error=0.1 and type II
error=0.2, it was necessary to include 16 patients in each arm.
Assuming 10% drop-out, we calculated that a total of 18 patients
were required in both treatment arms. The baseline characteristics
of the patients were compared between the treatment arms using
Chi-squared test (applying the Yates correction when necessary) for
categorical variables and using Student's t-test for continuous
variables. Differences between paired observations were determined
using the Student's paired t-test. P-values <0.05 were
considered significant. All statistical analyses were performed
with GraphPad Prism 5 for Windows (Graphpad Software, Inc., San
Diego, CA, USA).
Results
Patient characteristics
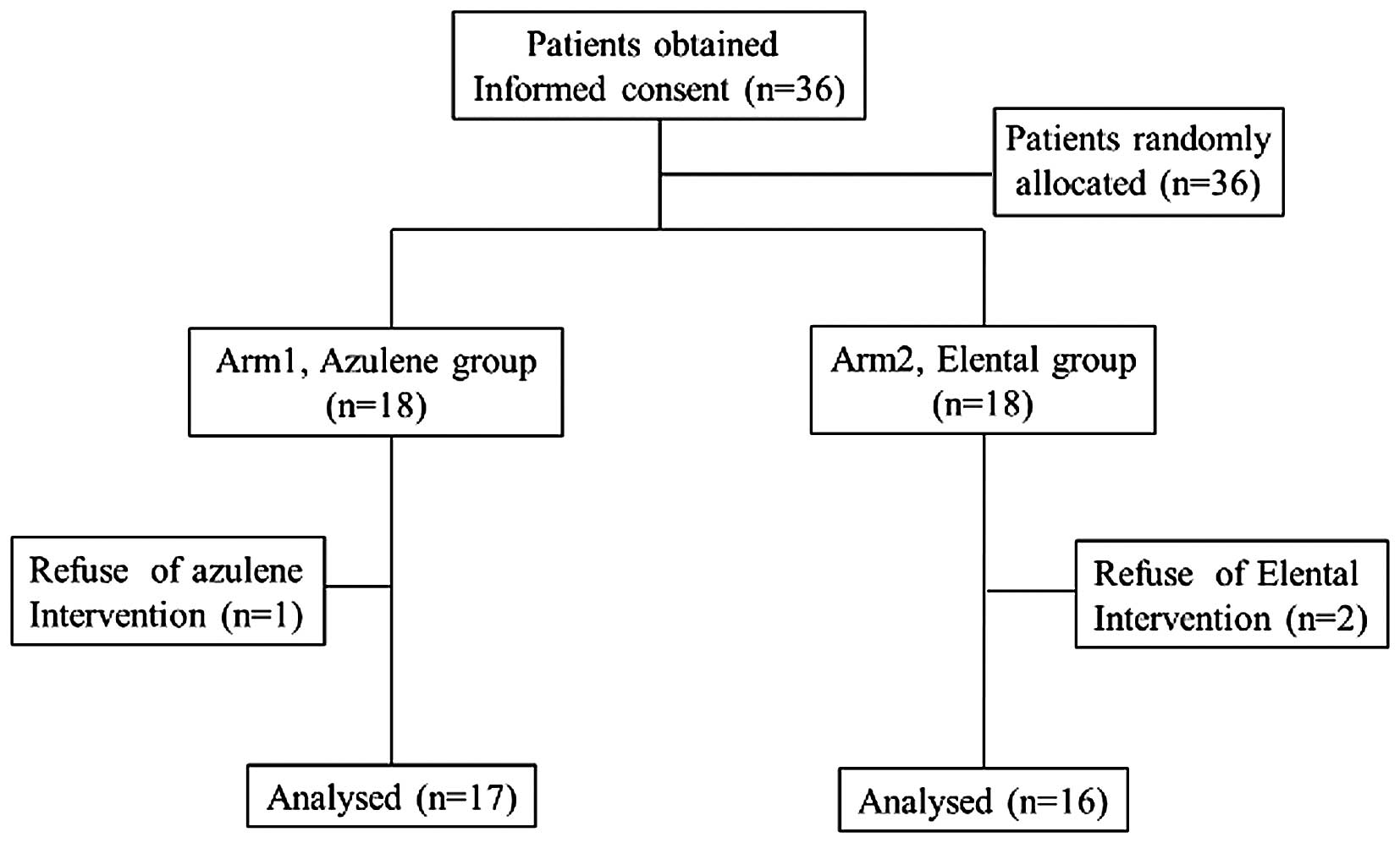
Thirty-six patients were enrolled in the study
between February 2012 and April 2015, after signing the written
informed consent, and they were randomly assigned to receive oral
rinse with azulene (Arm 1, n=18) or to undergo administration of
Elental® (Arm 2, n=18). Of them, three patients were
excluded from analysis (Arm 1, 1; Arm 2, 2), who did not receive
either treatment (i.e., azulene or elental). Consequently, 33
patients were included in the complete analysis (Fig. 1). The average BMI and SMI for the 33
patients was 20.42 and 6.59 kg/m2, respectively.
Although sarcopenia was observed in 20 patients (60.6%) prior to
treatment, there was no sarcopenic obesity, i.e., sarcopenia
combined with BMI ≥25 kg/m2. The baseline patient
characteristics were similar between the groups (Table II). As randomization was stratified
by treatment (chemotherapy/chemoradiotherapy) and stage, these were
well balanced between the treatment groups. However, with respect
to the chemotherapy regimen, triplet chemotherapy regimen, which is
a more toxic regimen with an increased incidence of stomatitis
(31), was more frequently used for
patients in Arm 2 (Arm 1=22.2%; Arm 2=50.0%). Although CRP
(P=0.24), white blood cell count (P=0.12) and total neutrophil
count (P=0.14) tended to be higher in patients in Arm 2, the
differences were not statistically significant. BMI and body
composition (i.e., body fat mass and LBM) were comparable between
the groups.
 | Table IIBaseline patient characteristics. |
Table II
Baseline patient characteristics.
|
Characteristics | Arm 1 (Azulene,
n=17) | Arm 2 (Elental,
n=16) | P-value |
|---|
| Gender
(Male/female) | 13/4 | 14/2 | 1 |
| Age, median
(range) | 66 (44–79) | 68 (50–76) | 0.46 |
| ECOG performance
status |
| 0 | 8 | 6 | 0.51 |
| 1 | 9 | 10 | |
| Stage |
| I | 3 | 4 | 0.95 |
| II | 1 | 1 | |
| III | 9 | 7 | |
| IV | 4 | 4 | |
| Treatment |
| Chemotherapy
(doublet/triplet) | 9 (7/2) | 8 (4/4) | 1 |
|
Chemo-radiotherapy | 8 | 8 | |
| BMI
(kg/m2) | 20.39±2.84 | 20.46±1.76 | 0.93 |
| Body fat weight
(kg) | 12.08±5.65 | 12.39±3.20 | 0.85 |
| LBM (kg) | 23.78±0.94 | 24.35±1.06 | 0.69 |
| SMI
(kg/m2) | 6.57±0.85 | 6.60±0.74 | 0.92 |
| Albumin (g/dl) | 3.78±0.38 | 3.78±0.37 | 0.99 |
| Total cholesterol
(mg/dl) | 187.1±41.8 | 193.6±28.6 | 0.63 |
| CRP (mg/dl) | 0.16
(0.02–5.32) | 0.43
(0.02–8.66) | 0.24 |
| White blood cell
count (/mm3) | 5706±1756 | 7169±3293 | 0.12 |
| Total neutrophil
count (/mm3) | 3408±1695 | 4694±3035 | 0.14 |
| Total lymphocyte
count (/mm3) | 1719±478 | 1658±574 | 0.74 |
| Hemoglobin
(g/dl) | 12.9±1.14 | 13.0±1.75 | 0.84 |
Incidence and severity of oral
mucositis
During the treatment cycle, oral mucositis CTCAE
grade ≥2 developed in 6 patients (18.2%). The incidence of oral
mucositis grade ≥2 tended to be lower for patients treated with
Elental (Arm 1=23.5%; Arm 2=12.5%), but this was not statistically
significant (Table III). Analysis
of the severity of oral pain is shown in Table IV. Most of the patients (72.7%) did
not complain of oral pain and oral pain grade ≥2 was observed in
only 5 patients (15.2%). There was no significant difference in the
maximum severity of oral pain between Arm 1 (azulene) and Arm 2
(Elental).
 | Table IIIMaximum severity of oral mucositis.
Table IV. Maximum severity of oral
pain. |
Table III
Maximum severity of oral mucositis.
Table IV. Maximum severity of oral
pain.
| Maximumgrade | Patients, n (%)
| P-value |
|---|
| Arm 1 (Azulene,
n=17) | Arm 2 (Elental,
n=16) |
|---|
| 1 | 13 (76.5) | 14 (87.5) | |
| 2 | 4 (23.5) | 1 (6.25) | 0.246 |
| 3 | 0 (0) | 1 (6.25) | |
 | Table IVMaximum severity of oral pain. |
Table IV
Maximum severity of oral pain.
| Maximumseverity
grade of oral pain | Patients, n (%)
| P-value |
|---|
| Arm 1 (Azulene,
n=17) | Arm 2 (Elental,
n=16) |
|---|
| 0 | 12 (70.6) | 12 (75) | 0.728 |
| 1 | 3 (17.6) | 1 (6.25) | |
| 2 | 1 (5.9) | 2 (12.5) | |
| ≥3 | 1 (5.9) | 1 (6.25) | |
Changes of nutrition indicators from
baseline
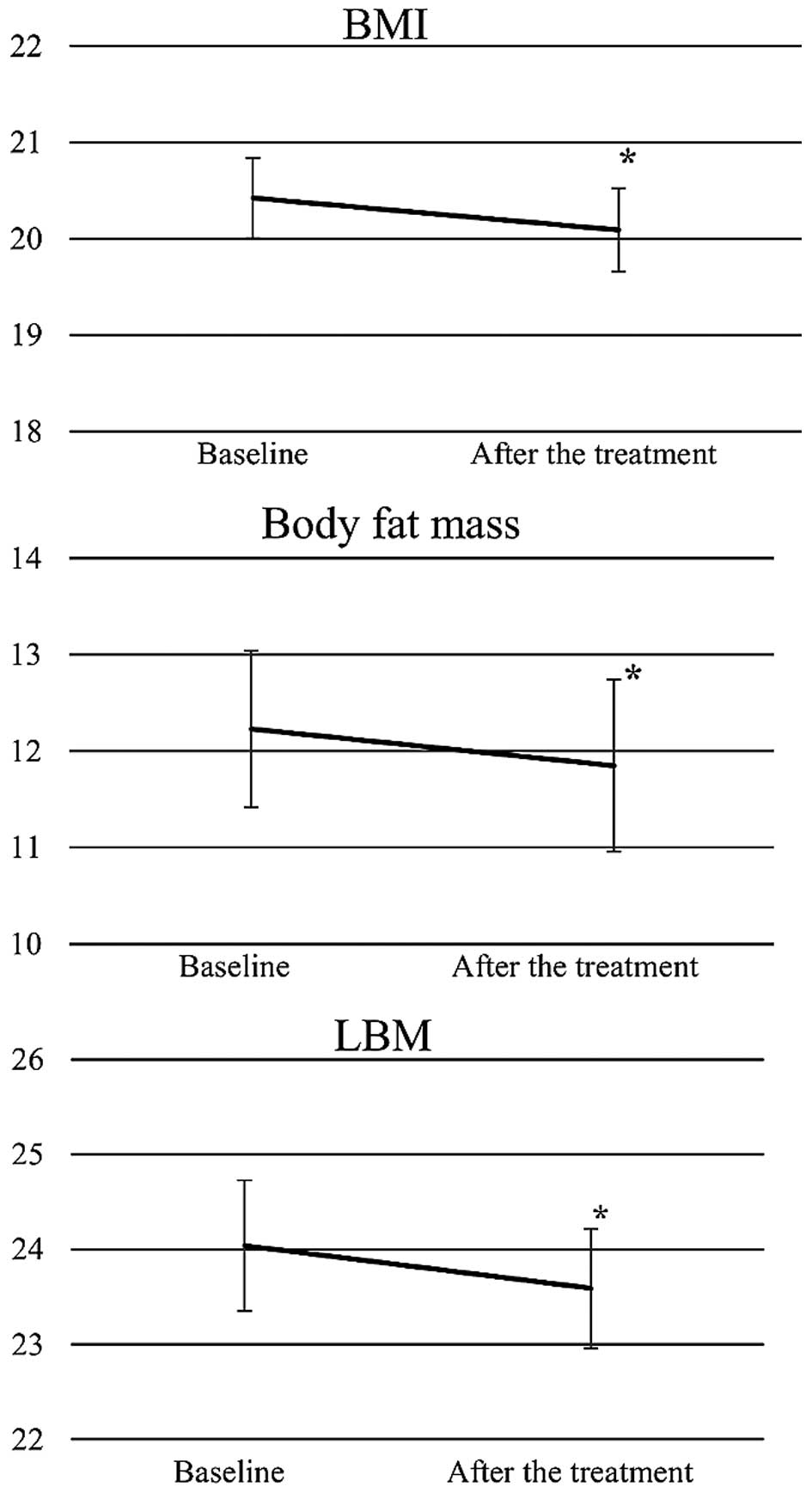
The average BMI, body fat mass and LBM in the 33
patients decreased significantly after chemo (chemoradio) therapy
(Fig. 2). Relative changes of
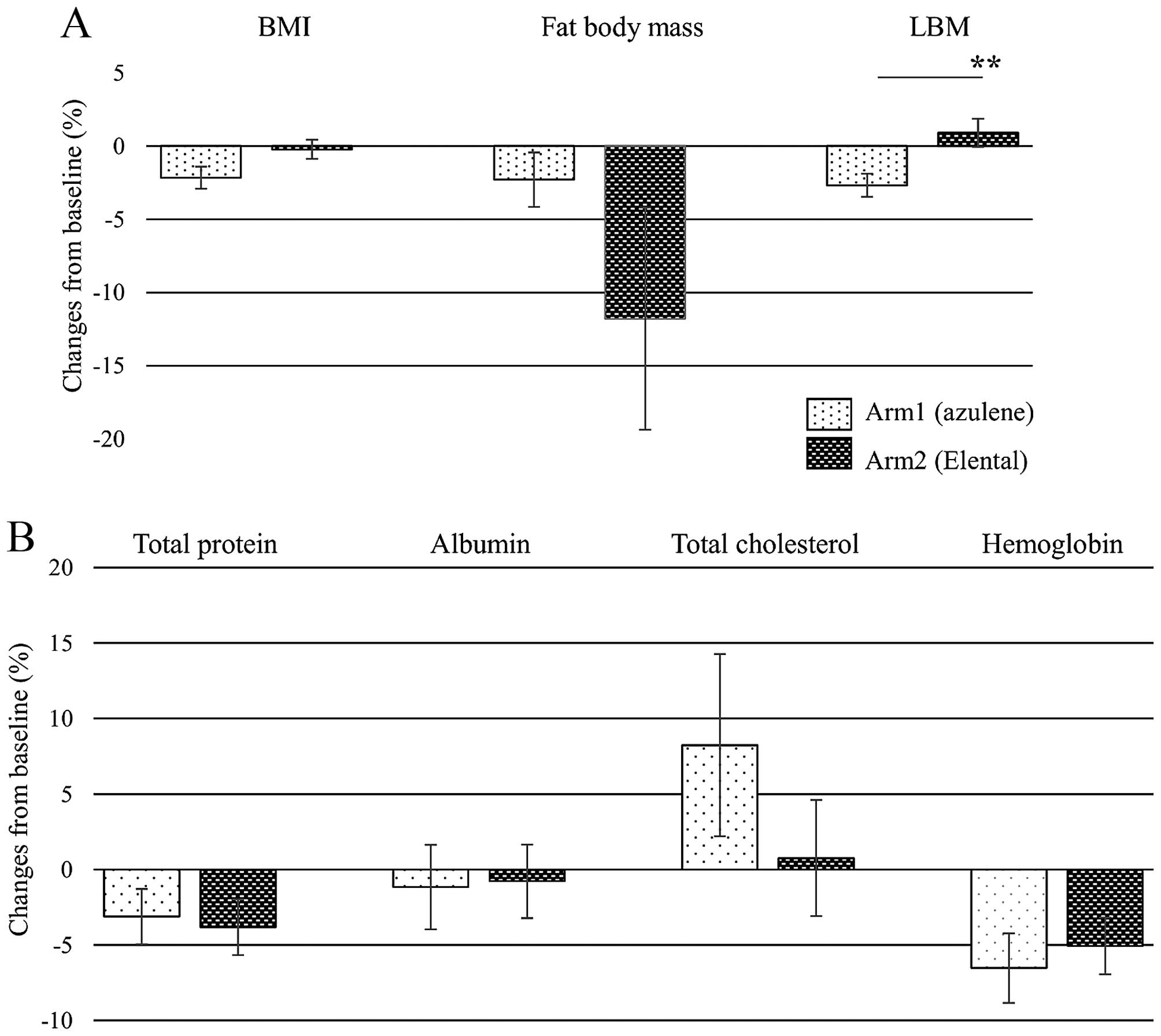
nutrition indicators after the treatment from baseline were
calculated using the following formula: 100 x (value after-value
before)/value before. When the relative changes of nutrition
indicators were compared between Arm 1 (azulene) and Arm 2
(Elental) groups (Fig. 3), a
different pattern appeared when BMI, body fat mass and LBM were
expressed as the percent change from baseline for each group. BMI
and body fat mass were reduced after the treatment relative to
baseline in both groups. By contrast, LBM was lost in Arm 1, but
increased in Arm 2 after the treatment compared with the baseline
values. There was a significant difference between the change of
LBM in Arm 1 and in Arm 2 (P=0.007). The laboratory values for
total protein, albumin and hemoglobin decreased but total
cholesterol increased in both groups after treatment. The relative
change of total cholesterol increased more in Arm 1 than in Arm 2,
but there was no statistically significant difference between the
two groups (P=0.33).
Adverse events of chemotherapy
With respect to adverse events other than oral
mucositis during the treatment cycle, common grade 3/4 toxicities
included leukopenia, neutropenia and lymphocytopenia (Table V). Although the grade 3/4
neutropenia occurred more often in Arm 1 compared with Arm 2 (41.2
and 25.0%, respectively), this was not statistically significant
(P=0.46). The incidence of diarrhea was greater in Arm 2 than in
Arm 1 (31.3 and 11.8%, respectively), and was grade 2 or less.
There were no statistically significant differences in the
incidence or severity of hematological and non-hematological
adverse effects between the two arms.
 | Table VAdverse events according to treatment
group. |
Table V
Adverse events according to treatment
group.
| Toxicity | Arm 1 (Azulene,
n=17)
| Arm 2 (Elental,
n=16)
|
|---|
| Grade1 | Grade 2 | Grade 3 | Grade 4 | Grade1 | Grade 2 | Grade 3 | Grade 4 |
|---|
| Hematological |
| Leukopenia | 0 | 5 | 4 | 2 | 3 | 3 | 3 | 1 |
| Neutropenia | 0 | 2 | 3 | 4 | 0 | 1 | 2 | 2 |
|
Lymphocytopenia | 2 | 5 | 6 | 0 | 3 | 4 | 7 | 0 |
| Anemia | 11 | 2 | 0 | 0 | 9 | 4 | 0 | 0 |
|
Thrombocytopenia | 6 | 3 | 0 | 1 | 6 | 1 | 0 | 0 |
| Elevated AST | 3 | 0 | 0 | 0 | 4 | 0 | 0 | 0 |
| Elevated ALT | 6 | 1 | 0 | 0 | 8 | 0 | 0 | 0 |
|
Non-hematological |
| Fatigue | 8 | 5 | 0 | 0 | 6 | 1 | 0 | 0 |
| Anorexia | 3 | 12 | 0 | 0 | 2 | 8 | 1 | 0 |
| Nausea | 4 | 8 | 0 | 0 | 5 | 1 | 0 | 0 |
| Diarrhea | 2 | 0 | 0 | 0 | 4 | 1 | 0 | 0 |
| Constipation | 13 | 0 | 0 | 0 | 10 | 0 | 0 | 0 |
Discussion
Profound weight loss and malnutrition subsequent to
severe dysphagia and cancer cachexia are cardinal symptoms in
esophageal cancer. Recently, it has been reported that ~25–55% of
patients with esophageal cancer undergoing neo-adjuvant therapy
were sarcopenic at the time of diagnosis (18,32–35).
Moreover, two recent studies in esophageal cancer patients have
shown that muscle wasting can worsen with cancer chemo- or
chemoradiotherapy (18,34). Although the primary endpoint of a
preventive effect against oral mucositis was not met, the present
study demonstrated several noteworthy findings. First, the overall
prevalence of sarcopenia among Japanese esophageal cancer patients
was 60.6% in the present study, which is higher than the
prevalences described in previous reports for esophageal cancer
patients mainly from Europe (18,32–35).
On the other hand, sarcopenic obesity was not observed in this
study, however, Anandavadivelan et al (32) reported sarcopenic obesity in 14% of
esophageal cancer patients in Sweden. Second, in most patients,
both body fat mass and LBM were reduced during the treatment cycle,
and average values among all participants were decreased
significantly after chemo (chemoradio) therapy. Third, the most
important finding in this study is that enteral nutrition using
Elental could maintain or even increase LBM post chemo (chemoradio)
therapy.
Sarcopenia was recently identified as a poor
prognostic factor in patients with various malignancies, such as
melanoma, hepatic carcinoma, pancreatic and lung cancer, and liver
metastasis from colorectal cancer (11,13,15,17,36).
Moreover, sarcopenia and sarcopenic obesity are associated
independently with a poor response to cancer chemotherapy in
melanoma (11), renal-cell
(12), hepatic carcinoma (13), colorectal (14), pancreatic (15), breast (16) and lung cancer (17). More recently, sarcopenic obesity has
been reported to be a probable risk factor for dose limiting
toxicity during chemotherapy for esophageal cancer (32). Thus, it has become apparent that
sarcopenia has a clinically significant impact on treatment
response and survival in various malignancies, including esophageal
cancer.
The rate of malnutrition in patients with esophageal
cancer has been estimated to be as high as 78.9% (37) and marked reductions in both body fat
mass and skeletal muscle mass occur during chemotherapy for
esophageal cancer (18).
Nutritional support is important to decrease major postoperative
complications in esophageal cancer patients (38,39).
However, the significance of nutritional intervention in patients
undergoing chemo- or chemoradiotherapy for esophageal cancer is not
clear. In the present study, LBM decreased during treatment in the
azulene (control) group, but increased in the Elental®
group relative to the baseline and there was a significant
difference between the two groups. To the best of our knowledge,
this is the first report demonstrating that nutritional
intervention can counteract skeletal muscle wasting during chemo-
or chemoradiotherapy for esophageal cancer. The treatment response
to chemo (chemoradio) therapy did not differ between the azulene
and Elental groups in the present study (average change in tumor
size after 1 cycle treatment; −18.4 vs. −18.1%, respectively; data
not shown), however, this may be due to the short observation
period. We believe that nutritional intervention during cancer
treatment is important, and can contribute to the improvement of
both malnutrition as well as the clinical outcome in cancer
patients. Further studies over a longer follow-up period of time
are needed to confirm these observations.
It has been reported that amino acid supplementation
can stimulate muscle protein synthesis (24,40,41),
and among the amino acids, leucine has been recognized to play a
significant role in muscle protein synthesis (42). Elental, which does not contain a
great amount of leucine (6.84%) (Table
I), does contain a large amount of amino acids (12.66 g/one
pack). In considering the possible explanations for the results of
this study, it appears likely that muscle protein synthesis was
enhanced by the high amount of amino acids contained in Elental.
Another possible explanation is that Elental could help to maintain
mucosal integrity in the gastrointestinal tract, thereby resulting
in maintained nutrient absorption. Anticancer drugs, such as
5-fluorouracil, can induce small intestinal mucosal injury and
plasma diamine oxidase (DAO) has been reported to be a useful
indicator of mucosal injury of the gastrointestinal tract (43). Tanaka et al (7) recently reported that plasma DAO
activity was decreased after chemotherapy in patients with
esophageal cancer, and patients treated with the Elental diet
exhibited a significant increase in DAO activity after
chemotherapy. While we did not measure plasma DAO activity during
chemo (chemoradio) therapy, amino acids, such as glutamine, which
is abundant in Elental, could prevent anticancer drug-induced
intestinal mucosal injury and maintain nutrient absorption,
including the amino acids, thus contributing to stimulate muscle
protein synthesis. However, plasma amino acid concentrations were
not measured during this study, and doing this in future
experiments could help to validate the results presented here. We
also believe further experimental studies are needed to confirm the
underlying mechanism.
The present study did not provide evidence of the
effectiveness of Elental in protecting against the development of
oral mucositis during chemo (chemoradio) therapy. The incidence of
oral mucositis (≥ grade 2) in this study was 18.2% among all
enrolled patients, which was much lower than previously assumed
(the incidence for the azulene arm being 50%), which makes it
difficult to achieve statistical significance. Despite lacking
statistical significance, the incidence of oral mucositis in the
Elental arm was lower than that in the azulene arm (12.5 vs. 23.5%,
respectively). It is possible that evidence supporting the efficacy
for Elental against oral mucositis might be achieved, if the study
were to focus on a high-risk population susceptible to oral
mucositis, such as patients who have experienced it prior to
undergoing chemo (chemoradio) therapy.
In conclusion, this study revealed that Elental
could counteract the development of sarcopenia during the treatment
cycle, though we could not demonstrate the efficacy of Elental
against chemo (chemoradio) therapy-induced oral mucositis in
esophageal cancer patients. Further clinical studies with a larger
sample size should be conducted to confirm these results and to
elucidate whether the preventive effect of Elental against
sarcopenia leads to an improvement of the clinical outcome of
esophageal cancer patients undergoing treatment with chemo
(chemoradio) therapy.
Acknowledgments
We are grateful to Saeko Tsuchiya for her help and
support in the present study.
Abbreviations:
|
BMI
|
body mass index
|
|
LBM
|
lean body mass
|
|
ECOG
|
Eastern Cooperative Oncology Group
|
|
CTCAE
|
the Common Terminology Criteria for
Adverse Events
|
|
SMI
|
skeletal muscle index
|
|
DAO
|
diamine oxidase
|
|
BCAA
|
branched-chain amino acids
|
References
|
1
|
Jones JA, Avritscher EB, Cooksley CD,
Michelet M, Bekele BN and Elting LS: Epidemiology of
treatment-associated mucosal injury after treatment with newer
regimens for lymphoma, breast, lung, or colorectal cancer. Support
Care Cancer. 14:505–515. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Elting LS, Cooksley C, Chambers M, Cantor
SB, Manzullo E and Rubenstein EB: The burdens of cancer therapy.
Clinical and economic outcomes of chemotherapy-induced mucositis.
Cancer. 98:1531–1539. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sonis ST, Elting LS, Keefe D, Peterson DE,
Schubert M, Hauer-Jensen M, Bekele BN, Raber-Durlacher J, Donnelly
JP and Rubenstein EB; Mucositis Study Section of the Multinational
Association for Supportive Care in Cancer; International Society
for Oral Oncology: Perspectives on cancer therapy-induced mucosal
injury: Pathogenesis, measurement, epidemiology, and consequences
for patients. Cancer. 100(Suppl 9): 1995–2025. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rubenstein EB, Peterson DE, Schubert M,
Keefe D, McGuire D, Epstein J, Elting LS, Fox PC, Cooksley C and
Sonis ST; Mucositis Study Section of the Multinational Association
for Supportive Care in Cancer; International Society for Oral
Oncology: Clinical practice guidelines for the prevention and
treatment of cancer therapy-induced oral and gastrointestinal
mucositis. Cancer. 100(Suppl 9): 2026–2046. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Nishimura N, Nakano K, Ueda K, Kodaira M,
Yamada S, Mishima Y, Yokoyama M, Terui Y, Takahashi S and Hatake K:
Prospective evaluation of incidence and severity of oral mucositis
induced by conventional chemotherapy in solid tumors and malignant
lymphomas. Support Care Cancer. 20:2053–2059. 2012. View Article : Google Scholar
|
|
6
|
Hayashi K, Ando N, Watanabe H, Ide H,
Nagai K, Aoyama N, Takiyama W, Ishida K, Isono K, Makuuchi H, et
al: Phase II evaluation of protracted infusion of cisplatin and
5-fluorouracil in advanced squamous cell carcinoma of the
esophagus: A Japan Esophageal Oncology Group (JEOG) Trial
(JCOG9407). Jpn J Clin Oncol. 31:419–423. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tanaka Y, Takahashi T, Yamaguchi K, Osada
S, Shimokawa T and Yoshida K: Elemental diet plus glutamine for the
prevention of mucositis in esophageal cancer patients receiving
chemotherapy: a feasibility study. Support Care Cancer. 24:933–941.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bozzetti F and Group SW: Screening the
nutritional status in oncology: a preliminary report on 1,000
outpatients. Support Care Cancer. 17:279–284. 2009. View Article : Google Scholar
|
|
9
|
Norman K, Stobäus N, Zocher D,
Bosy-Westphal A, Szramek A, Scheufele R, Smoliner C and Pirlich M:
Cutoff percentiles of bioelectrical phase angle predict
functionality, quality of life, and mortality in patients with
cancer. Am J Clin Nutr. 92:612–619. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Martin L, Birdsell L, Macdonald N, Reiman
T, Clandinin MT, McCargar LJ, Murphy R, Ghosh S, Sawyer MB and
Baracos VE: Cancer cachexia in the age of obesity: Skeletal muscle
depletion is a powerful prognostic factor, independent of body mass
index. J Clin Oncol. 31:1539–1547. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sabel MS, Lee J, Cai S, Englesbe MJ,
Holcombe S and Wang S: Sarcopenia as a prognostic factor among
patients with stage III melanoma. Ann Surg Oncol. 18:3579–3585.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Antoun S, Lanoy E, Iacovelli R,
Albiges-Sauvin L, Loriot Y, Merad-Taoufik M, Fizazi K, di Palma M,
Baracos VE and Escudier B: Skeletal muscle density predicts
prognosis in patients with metastatic renal cell carcinoma treated
with targeted therapies. Cancer. 119:3377–3384. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Iritani S, Imai K, Takai K, Hanai T, Ideta
T, Miyazaki T, Suetsugu A, Shiraki M, Shimizu M and Moriwaki H:
Skeletal muscle depletion is an independent prognostic factor for
hepatocellular carcinoma. J Gastroenterol. 50:323–332. 2015.
View Article : Google Scholar
|
|
14
|
Barret M, Antoun S, Dalban C, Malka D,
Mansourbakht T, Zaanan A, Latko E and Taieb J: Sarcopenia is linked
to treatment toxicity in patients with metastatic colorectal
cancer. Nutr Cancer. 66:583–589. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tan BH, Birdsell LA, Martin L, Baracos VE
and Fearon KC: Sarcopenia in an overweight or obese patient is an
adverse prognostic factor in pancreatic cancer. Clin Cancer Res.
15:6973–6979. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Prado CM, Baracos VE, McCargar LJ, Reiman
T, Mourtzakis M, Tonkin K, Mackey JR, Koski S, Pituskin E and
Sawyer MB: Sarcopenia as a determinant of chemotherapy toxicity and
time to tumor progression in metastatic breast cancer patients
receiving capecitabine treatment. Clin Cancer Res. 15:2920–2926.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Prado CM, Lieffers JR, McCargar LJ, Reiman
T, Sawyer MB, Martin L and Baracos VE: Prevalence and clinical
implications of sarcopenic obesity in patients with solid tumours
of the respiratory and gastrointestinal tracts: A population-based
study. Lancet Oncol. 9:629–635. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Awad S, Tan BH, Cui H, Bhalla A, Fearon
KC, Parsons SL, Catton JA and Lobo DN: Marked changes in body
composition following neoadjuvant chemotherapy for oesophagogastric
cancer. Clin Nutr. 31:74–77. 2012. View Article : Google Scholar
|
|
19
|
Baracos VE, Reiman T, Mourtzakis M,
Gioulbasanis I and Antoun S: Body composition in patients with
non-small cell lung cancer: A contemporary view of cancer cachexia
with the use of computed tomography image analysis. Am J Clin Nutr.
91:1133S–1137S. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bower MR and Martin RC II: Nutritional
management during neoadjuvant therapy for esophageal cancer. J Surg
Oncol. 100:82–87. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Peterson DE, Jones JB and Petit RG II:
Randomized, placebo-controlled trial of Saforis for prevention and
treatment of oral mucositis in breast cancer patients receiving
anthracycline-based chemotherapy. Cancer. 109:322–331. 2007.
View Article : Google Scholar
|
|
22
|
Choi K, Lee SS, Oh SJ, Lim SY, Lim SY,
Jeon WK, Oh TY and Kim JW: The effect of oral glutamine on
5-fluorouracil/leucovorin-induced mucositis/stomatitis assessed by
intestinal permeability test. Clin Nutr. 26:57–62. 2007. View Article : Google Scholar
|
|
23
|
Tsujimoto T, Yamamoto Y, Wasa M, Takenaka
Y, Nakahara S, Takagi T, Tsugane M, Hayashi N, Maeda K, Inohara H,
et al: L-glutamine decreases the severity of mucositis induced by
chemoradiotherapy in patients with locally advanced head and neck
cancer: A double-blind, randomized, placebo-controlled trial. Oncol
Rep. 33:33–39. 2015.
|
|
24
|
Wolfe RR: Regulation of muscle protein by
amino acids. J Nutr. 132:3219S–3224S. 2002.PubMed/NCBI
|
|
25
|
de Campos-Ferraz PL, Andrade I, das Neves
W, Hangai I, Alves CR and Lancha AH Jr: An overview of amines as
nutritional supplements to counteract cancer cachexia. J Cachexia
Sarcopenia Muscle. 5:105–110. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Malavolti M, Mussi C, Poli M, Fantuzzi AL,
Salvioli G, Battistini N and Bedogni G: Cross-calibration of
eight-polar bioelectrical impedance analysis versus dual-energy
X-ray absorptiometry for the assessment of total and appendicular
body composition in healthy subjects aged 21–82 years. Ann Hum
Biol. 30:380–391. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chen LK, Liu LK, Woo J, Assantachai P,
Auyeung TW, Bahyah KS, Chou MY, Chen LY, Hsu PS, Krairit O, et al:
Sarcopenia in Asia: Consensus report of the Asian Working Group for
Sarcopenia. J Am Med Dir Assoc. 15:95–101. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Fukui T, Itoh Y, Orihara M, Yoshizawa K,
Takeda H, Kawada S and Yoshioka T: Elental prevented and reduced
oral mucositis during chemotherapy in patients esophageal cancer.
Gan To Kagaku Ryoho. 38:2597–2601. 2011.In Japanese. PubMed/NCBI
|
|
29
|
Ogata Y, Takeuchi M, Ishibashi N, Kibe S,
Takahashi K, Uchida S, Murakami N, Yahara T and Shirouzu K:
Efficacy of Elental on prevention for chemotherapy-induced oral
mucositis in colorectal cancer patients. Gan To Kagaku Ryoho.
39:583–587. 2012.In Japanese. PubMed/NCBI
|
|
30
|
Ogata Y, Ishibashi N, Yamaguchi K, Uchida
S, Kamei H, Nakayama G, Hirakawa H, Tanigawa M and Akagi Y:
Preventive effects of amino-acid-rich elemental diet Elental on
chemotherapy-induced oral mucositis in patients with colorectal
cancer: a prospective pilot study. Support Care Cancer. 24:783–789.
2015. View Article : Google Scholar
|
|
31
|
Tebbutt NC, Cummins MM, Sourjina T,
Strickland A, Van Hazel G, Ganju V, Gibbs D, Stockler M, Gebski V
and Zalcberg J; Australasian Gastro-Intestinal Trials Group:
Randomised, non-comparative phase II study of weekly docetaxel with
cisplatin and 5-fluorouracil or with capecitabine in
oesophagogastric cancer: The AGITG ATTAX trial. Br J Cancer.
102:475–481. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Anandavadivelan P, Brismar TB, Nilsson M,
Johar AM and Martin L: Sarcopenic obesity: A probable risk factor
for dose limiting toxicity during neo-adjuvant chemotherapy in
oesophageal cancer patients. Clin Nutr. 35:724–730. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tan BH, Brammer K, Randhawa N, Welch NT,
Parsons SL, James EJ and Catton JA: Sarcopenia is associated with
toxicity in patients undergoing neo-adjuvant chemotherapy for
oesophagogastric cancer. Eur J Surg Oncol. 41:333–338. 2015.
View Article : Google Scholar
|
|
34
|
Yip C, Goh V, Davies A, Gossage J,
Mitchell-Hay R, Hynes O, Maisey N, Ross P, Gaya A, Landau DB, et
al: Assessment of sarcopenia and changes in body composition after
neoadjuvant chemotherapy and associations with clinical outcomes in
oesophageal cancer. Eur Radiol. 24:998–1005. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Harada K, Ida S, Baba Y, Ishimoto T,
Kosumi K, Tokunaga R, Izumi D, Ohuchi M, Nakamura K, Kiyozumi Y, et
al: Prognostic and clinical impact of sarcopenia in esophageal
squamous cell carcinoma. Dis Esophagus. Jun 30–2015.Epub ahead of
print. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
van Vledder MG, Levolger S, Ayez N,
Verhoef C, Tran TC and Ijzermans JN: Body composition and outcome
in patients undergoing resection of colorectal liver metastases. Br
J Surg. 99:550–557. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Larrea J, Vega S, Martínez T, Torrent JM,
Vega V and Núñez V: The nutritional status and immunological
situation of cancer patients. Nutr Hosp. 7:178–184. 1992.In
Spanish. PubMed/NCBI
|
|
38
|
Daly JM, Massar E, Giacco G, Frazier OH,
Mountain CF, Dudrick SJ and Copeland EM III: Parenteral nutrition
in esophageal cancer patients. Ann Surg. 196:203–208. 1982.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Takesue T, Takeuchi H, Ogura M, Fukuda K,
Nakamura R, Takahashi T, Wada N, Kawakubo H and Kitagawa Y: A
prospective randomized trial of enteral nutrition after
thoracoscopic esophagectomy for esophageal cancer. Ann Surg Oncol.
22(Suppl 3): S802–S809. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Paddon-Jones D, Sheffield-Moore M,
Katsanos CS, Zhang XJ and Wolfe RR: Differential stimulation of
muscle protein synthesis in elderly humans following isocaloric
ingestion of amino acids or whey protein. Exp Gerontol. 41:215–219.
2006. View Article : Google Scholar
|
|
41
|
Katsanos CS, Kobayashi H, Sheffield-Moore
M, Aarsland A and Wolfe RR: A high proportion of leucine is
required for optimal stimulation of the rate of muscle protein
synthesis by essential amino acids in the elderly. Am J Physiol
Endocrinol Metab. 291:E381–E387. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Garlick PJ: The role of leucine in the
regulation of protein metabolism. J Nutr. 135(Suppl): 1553S–1556S.
2005.PubMed/NCBI
|
|
43
|
Miyoshi J, Miyamoto H, Goji T, Taniguchi
T, Tomonari T, Sogabe M, Kimura T, Kitamura S, Okamoto K, Fujino Y,
et al: Serum diamine oxidase activity as a predictor of
gastrointestinal toxicity and malnutrition due to anticancer drugs.
J Gastroenterol Hepatol. 30:1582–1590. 2015. View Article : Google Scholar : PubMed/NCBI
|