Introduction
Prostate cancer is the most common male
genitourinary system malignancy, and causes major public health
problems worldwide (1,2). The incidence of prostate cancer in
older men is rapidly increasing every year (3). Cancer often compromises adjacent
tissues and organs during the development of the disease and can
result in a wide range of bone metastases, seriously affecting the
survival and quality of life of patients. Thus far, the underlying
molecular mechanism of prostate cancer remains unclear. To this
end, it is necessary to investigate the biological mechanism
driving prostate cancer to provide novel insight into the
prevention and therapeutic management of this disease.
Forkhead box Q1 (FOXQ1) is a transcription factor
that exerts its biological effects by controlling gene
transcription activity (4–6). FOXQ1 encodes a forkhead box (FOX)
family related protein (6–8). The FOX family is involved in the
regulation of various biological activities, such as development
(7,9), metabolism (10), cancer (11), and aging (12). In recent years, the role of FOXQ1 as
a proto-oncogene has gradually been recognized, and it is found in
colon cancer (7), ovarian cancer
(13), breast cancer (7,14),
nasopharyngeal carcinoma (15),
glioma (16), non-small cell lung
cancer (NSCLC) (17) and other
tumors. Moreover, FOXQ1 plays a role in proliferation (18), drug resistance (19), epithelial-mesenchymal transition
(EMT) (19), angiogenesis (17) and metastasis (20) in cancer. It has been reported that
FOXQ1 is upregulated in colorectal cancer, inhibiting apoptosis and
accelerating tumor growth (21).
Moreover, FOXQ1 has a significant function in tumor growth in
bladder cancer (22). FOXQ1
promotes the invasive ability of laryngeal carcinoma (23) and enhances the metastasis of
hepatocellular carcinoma and non-small cell lung cancer by
regulating EMT (4,17). However, the role of FOXQ1 in
prostate cancer remains poorly understood.
BCL (B cell lymphoma/leukemia) 11A is a member of
the BCL family, and is a transcription factor that negatively
regulates fetal hemoglobin generation (24). BCL11A is expressed in the bone
marrow, spleen, B and T cells, monocytes and megakaryocytes
(25). Additionally, BCL11A is
related to the development of B cell malignancies, in which it is
expressed at high levels (26).
BCL11A is upregulated by FOXQ1 overexpression in colorectal cancer
and acute myeloid leukemia (21,27).
Studies have indicated that BCL11A regulates the
expression of murine double minute 2 (MDM2) (25,28).
MDM2 encodes for a protein that acts as a major regulator of the
tumor-suppressor gene p53, and is involved in the regulation of
cell growth, apoptosis and the cell cycle (29,30).
MDM2 is overexpressed in human gastric cancer, bladder cancer and
sarcoma, and expressed at lower levels in the corresponding normal
tissues, suggesting that MDM2 plays a role in the course of cancer
development (31–33). It has been indicated that MDM2
inhibition induces apoptosis in hepatoma and constrains the
proliferation and invasion of these cells (34). Moreover, studies have indicated that
MDM2 is a potential target for prostate cancer therapy, as
downregulation of MDM2 suppresses prostate cancer cell
proliferation and metastasis and promotes apoptosis (35).
In this study, we aimed to investigate the role of
FOXQ1 in regulating prostate cancer cell proliferation and
apoptosis and its potential relationship with BCL11A and MDM2. We
found that FOXQ1 is overexpressed in prostate cancer tissues and
cell lines and the inhibition of FOXQ1 induces cells apoptosis and
suppresses the proliferation and invasion of prostate cancer cells.
Moreover, the data demonstrate that FOXQ1 may play a role in
regulating MDM2 by controlling BCL11A in prostate cancer. Our study
suggests that FOXQ1 plays an important role in prostate cancer and
provided a novel insight into preventing and treating prostate
cancer through targeting DOXQ1.
Materials and methods
Cell lines
Human prostate cancer cell lines (PC-3, DU-145 and
LNCaP) were purchased from the Cell Bank of the Chinese Academy of
Sciences (Shanghai, China). These cell lines were cultured in
RPMI-1640 with 10% fetal bovine serum (FBS) (both from Hyclone,
Salt Lake City, UT, USA) and 1% penicillin/streptomycin (Gibco,
Rockville, MD, USA). Additionally, the human non-neoplastic
prostatic epithelial RWPE-1 obtained from the American Type Culture
Collection (ATCC; Rockwell, MD, USA) were maintained in
keratinocyte serum-free medium (Gibco) containing 50 μg/ml
of bovine pituitary extract, 0.5% penicillin-streptomycin mix, and
5 ng/ml epidermal growth factor. The cell lines were cultured in a
humidified atmosphere containing 5% CO2 at 37°C.
Tissue specimens
Tumor tissues and matched adjacent normal tissues
were obtained from Fourth Military Medical University and tissues
were cryopreserved immediately following laparoscopic radical
prostatectomy. The set of prostate tissues included 10 tumor
tissues and 10 matched adjacent normal tissues. The study was
approved by the hospital ethics board.
Real-time quantitative polymerase chain
reaction (RT-qPCR) assay
Total RNA was extracted from cell lines using TRIzol
(Thermo Fisher Scientific, Waltham, MA, USA). The method of total
RNA extraction from tissues was performed according to the
literature (36). Subsequently, the
total RNA was synthesized into cDNA using the RevertAid First
Strand cDNA Synthesis kit (Thermo Fisher Scientific) as per the
protocol. All of the RT-qPCR reactions used 20 μl, with 10
μl of SYBR Green PCR Master Mix (Applied Biosystems,
Carlsbad, CA, USA) and primers as follows: FOXQ1 forward,
5′-ATTTCTTGCTATTGACCGATGC-3′ and reverse,
5′-CCCAAGGAGACCACAGTTAGAG-3′; GAPDH forward,
5′-GGAAGATGGTGATGGGATT-3′ and reverse, 5′-GGATTTGGTCGTATTGGG-3′.
E-cadherin forward, 5′-GGCTGCGGCTGCAGAGACTGG-3′ and reverse,
5′-TACACTGCCCAGGAGCCAGA-3′; and vimentin forward,
5′-TGAGTACCGGAGACAGGTGCAG-3′ and reverse,
5′-GGAGCCACTGCCTTCATAGTCAA-3′. The relative levels of gene
expression were estimated by the 2−ΔΔCt method.
Western blot analysis
Total proteins were extracted from the cell lines
using lysis buffer (Beyotime, Nantong, China) and
phenylmethanesulfonyl fluoride (100:1). The concentrations of
proteins were measured using a BCA kit (Beyotime), and the
equivalent volume containing 25 μg of protein was separated
by 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis
(SDS-PAGE). The proteins were then transferred to a nitrocellulose
membrane using a semi-dry blotting apparatus (both from Bio-Rad,
Hercules, CA, USA). The membrane was blocked in 5% skim milk
diluted in Tris-buffered saline at room temperature for 2 h and
then incubated with primary antibodies against anti-FOXQ1 (1:500),
anti-BCL11A (1:800), anti-MDM2 (1:500), anti-E-cadherin (1:500),
anti-vimentin (1:500) and anti-GAPDH (1:1,000) purchased from Santa
Cruz Biotechnology Inc. (Santa Cruz, CA, USA) overnight at 4°C. The
membrane was blotted with a horseradish peroxidase-conjugated
secondary antibody (Cell Signaling Technology, Danvers, MA, USA;
1:1,000) for 2 h followed by washing with Tris-buffered saline with
Tween (TBST). Finally, the target proteins were analyzed using a
Bio-Rad ChemiDoc apparatus, and the signal intensity was measured
by Image-Pro Plus 6.0 software.
Caspase-3 activity assay
Caspase-3 activity assay in the cells was assessed
on the basis of the standard instructions of a Caspase-3 activity
assay kit (Beyotime).
Annexin-V fluorescein isothiocyanate
conjugate and propidium iodide Annexin V (Annexin V-FITC/PI)
Apoptosis was assessed according to the
manufacturer's instructions in the Annexin V-FITC Apoptosis
detection kit (Bender MedSystems, Vienna, Austria). The cells
(5×105 cells/ml) were resuspended in the binding buffer
diluted in distilled deionized water (1:4) and mixed with 5
μl of Annexin V-FITC in 195 μl of the cell
suspension. The mixture was then incubated at room temperature for
10 min, then washed in phosphate-buffered saline. Thereafter, the
cells were suspended in 190 μl of diluted binding buffer and
10 μl of PI was added. Apoptotic cells were counted using a
FACS analyzer (Beckman Coulter, Kraemer Boulevard Brea, CA,
USA).
Measurement of cell growth and
viability
Cell growth and viability were tested using the
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT)
assay. The cells were plated in 96-well plates and cultured in an
incubator with 5% CO2 at 37°C. A total of 25 μl
MTT (5 g/l) was added after removing the cell culture medium and
allowed to react for 5 h at 37°C. Afterwards, the crystals were
dissolved in 150 μl of dimethyl sulfoxide. The results were
measured using an ELISA reader (Titertek Plus MS 212; ICN,
Eschwege, Germany) at a wavelength of 490 nm. All experiments were
performed in triplicate.
Bromodeoxyuridine (BrdU) assay
Cell proliferation was assessed by a BrdU cell
proliferation assay kit (Millipore, Billerica, MA, USA). The
protocols were performed as per the instructions. Cells were plated
in 96-well plates and 10 μl of BrdU solution was added per
well and allowed to react for 1 h. The culture medium was replaced
by denaturing solution (100 μl/well) and incubated for 20
min. Thereafter, the detection antibody solution was added and
incubated for 1.5 h at room temperature. The secondary antibody
solution (100 μl/well) was added following three washes with
wash buffer. Next, 100 μl tetramethylbenzidine (TMB)
substrate was added and allowed to incubate for 25 min. The DNA
incorporation of BrdU was detected at 450 nm on a SpectroFluor Plus
Multiwell plate reader (Tecan, Research Triangle Park, NC, USA).
All experiments were performed in triplicate.
Cell invasion assay
Prostate cancer cell invasion ability was detected
using Bio-Coat cell migration chambers (BD Biosciences, San Jose,
CA, USA). Filters (Corning Incorporated, Toledo, NY, USA) were
coated with Matrigel (Becton Dickinson, Bedford, MA, USA). The
cells were resuspended in 300 μl of serum-free medium
(2×105 cells) and added to the insert (top chamber). A
total of 500 μl of culture medium with 10% FBS was then
added to the lower chamber and cultured in an incubator with 5%
CO2 at 37°C for 48 h. Non-invading cells were gently
removed by a cotton swab, and invasive cells were fixed, stained
and observed using a microscope. The number of invaded cells was
counted in six random fields per membrane and averaged. Each assay
was repeated three times.
Construction of recombinant plasmids
Full-length human BCL11A (NM_022893.3) was amplified
using the reverse transcription polymerase chain reaction and then
subcloned into the plasmid pcDNA.3.1/myc-His(−)Avector (Invitrogen,
Carlsbad, CA, USA). The restriction sites of BCL11A cDNA and
pcDNA.3.1/myc-His(−)Avector were EcoRI and BamHI.
Next, the transformation of recombined plasmids into E. coli
DH5α (Tiangen, Beijing, China) was conducted, and the recombined
plasmids were amplified overnight at 37°C. Finally, the amplified
plasmids were extracted and sequenced, and designated as
pcDNA.3.1/-BCL11A.
Cell transfection
The PC-3 (3×104 cells/well) and DU-145
(3×104 cells/well) cells were plated in 24-well plates,
separately. Transfection of FOXQ1 small-interfering RNA (siRNA,
5′-CGCGGACTTTGCACTTTGA-3′), non-specific siRNA
(5′-TTCTCCGAACGTGTCACGT-3′) or co-transfection of pcDNA.3.1/-BCL11A
and FOXQ1 siRNA was performed using Turbofect (Thermo Fisher
Scientific) in accordance with the manufacturer's instructions. The
transfected cells were maintained in an incubator (Thermo Fisher
Scientific) with 5% CO2 at 37°C for 48 h, and the
transfection efficiency was measured by RT-qPCR and western blot
analyses.
Statistical analysis
The statistical analyses were performed to test for
statistically significant differences between two groups using
Student's t-test and between multiple groups using one-way ANOVA.
Data are expressed as the mean ± standard deviation (SD). A p-value
of <0.05 was considered statistically significant.
Results
High expression of FOXQ1 in prostate
cancer tissues and cell lines
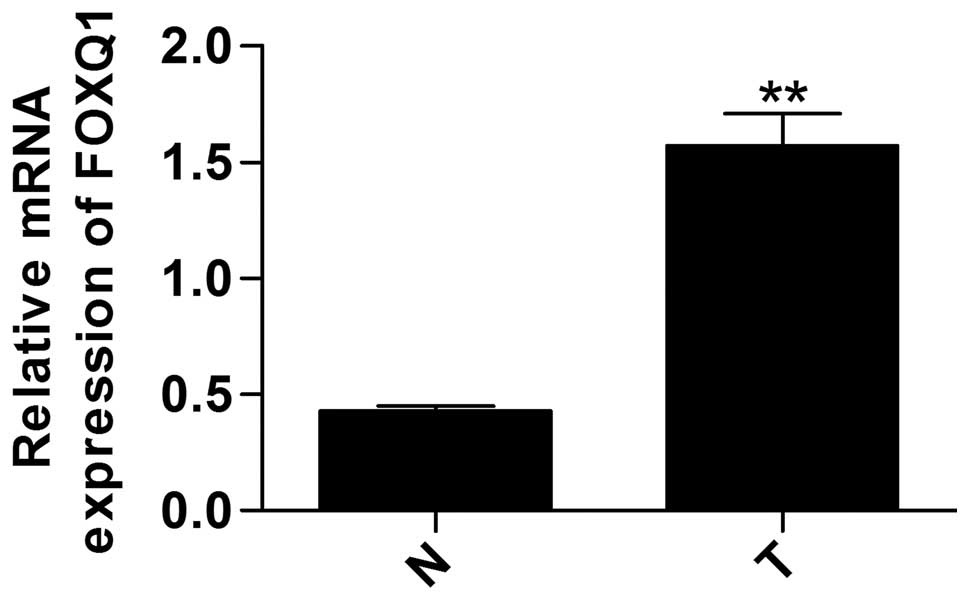
To investigate the expression of FOXQ1 in prostate
cancer, we tested the relative mRNA expression using RT-qPCR in
tumor tissues and matched adjacent normal tissues. The data show
that the mRNA expression of FOXQ1 was significantly higher in tumor
tissue than in the matched adjacent normal tissues (Fig. 1). Furthermore, FOXQ1 mRNA and
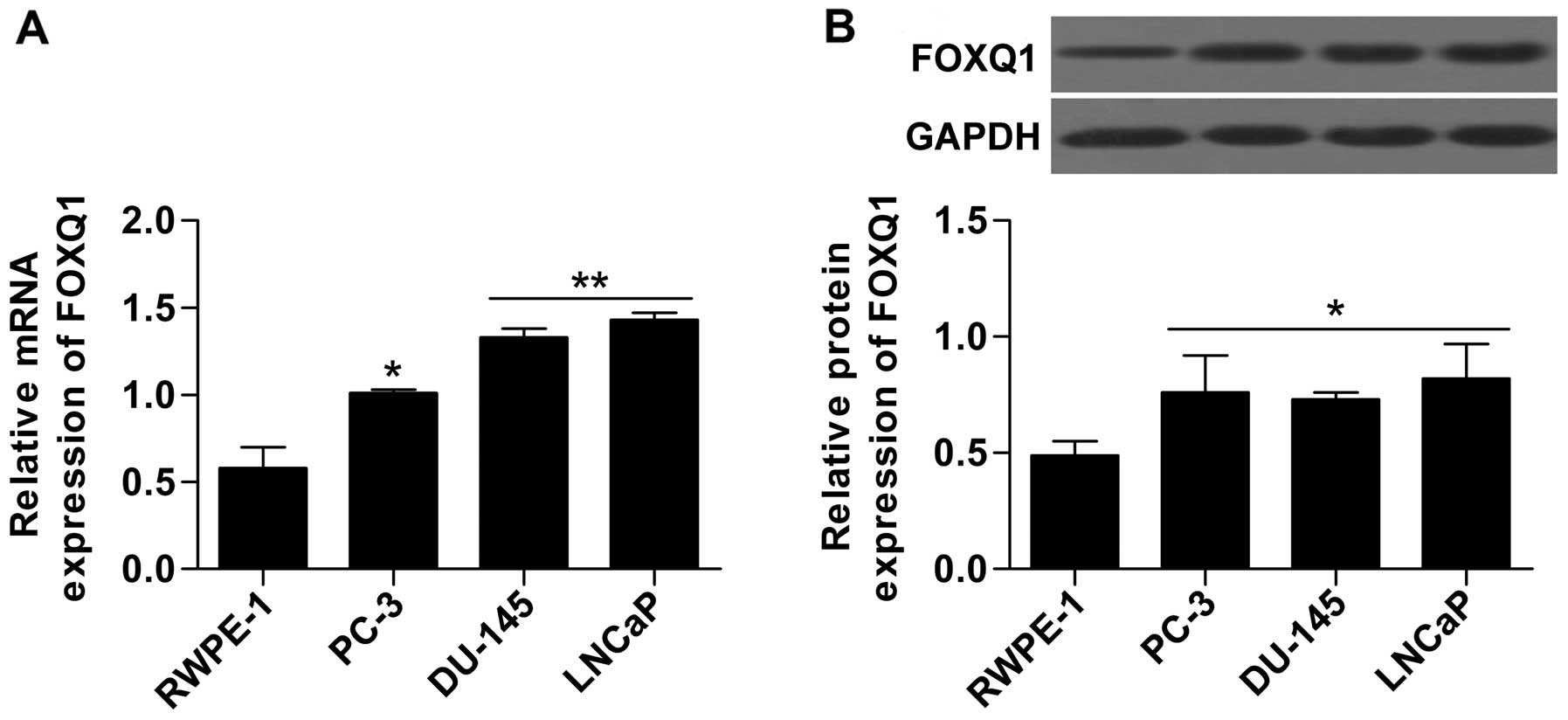
protein expression in prostate cancer cell lines, including PC-3,
DU-145 and LNCaP, were measured by RT-qPCR and western blot
analysis, respectively. The results show that the mRNA (Fig. 2A) and protein (Fig. 2B) expression of FOXQ1 in prostate
cancer lines (PC-3, DU-145 and LNCaP) was obviously upregulated
compared with the non-neoplastic cell line RWPE-1. These results
indicate that FOXQ1 may play an important role in prostate
cancer.
FOXQ1 inhibition constrains the
expression of BCL11A and MDM2
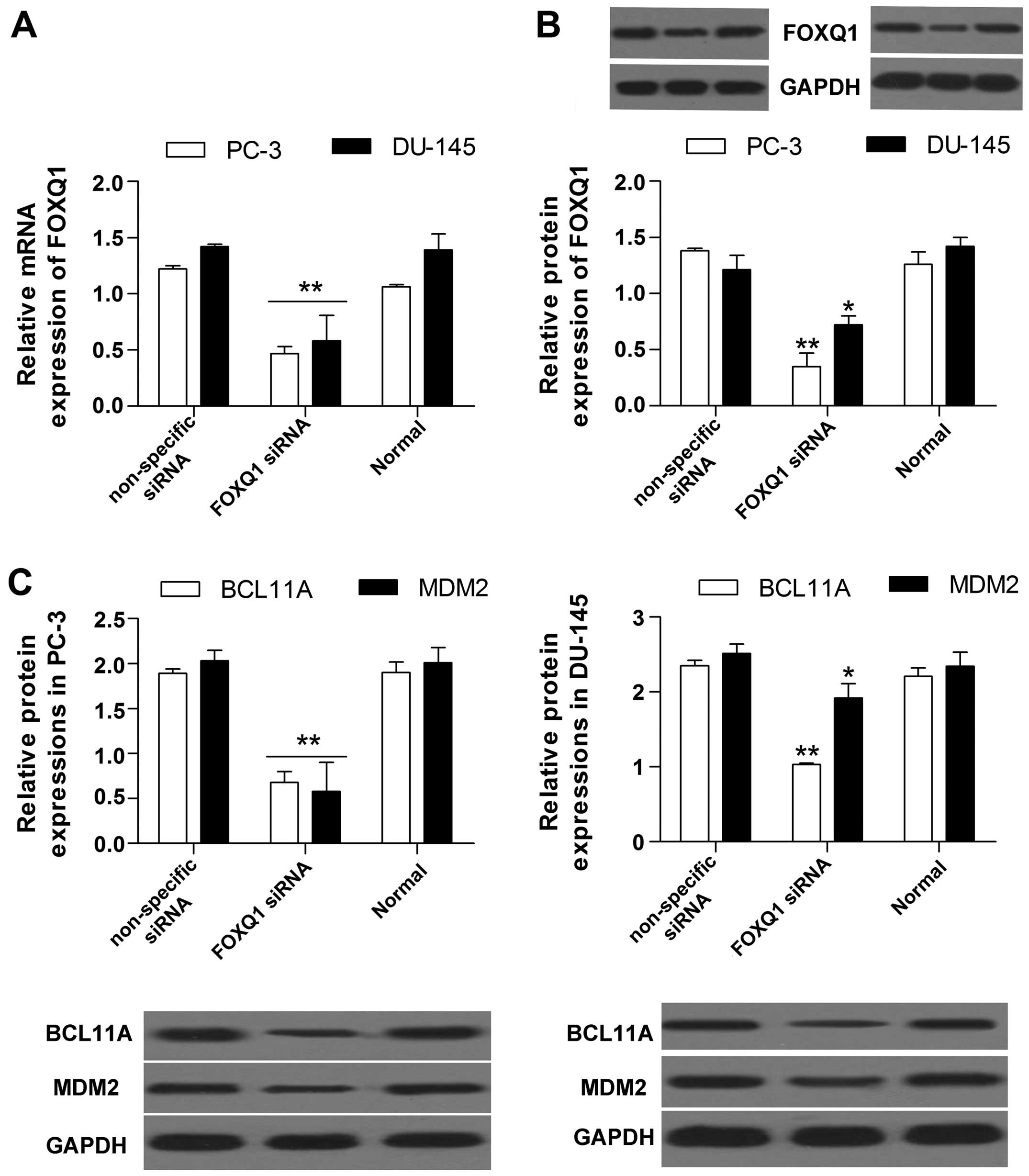
To explore the influence of FOXQ1 on BCL11A and
MDM2, we performed a FOXQ1 loss-of-function experiment in PC-3 and
DU-145 cells by transfection with siRNA targeted to FOXQ1. The
results show that the mRNA (Fig.
3A) and protein (Fig. 3B)
expression of FOXQ1 were both markedly decreased by FOXQ1 siRNA, as
determined by RT-qPCR and western blot analysis, indicating that
FOXQ1 inhibition was successful. Moreover, we found that FOXQ1
inhibition significantly decreased the protein expression of BCL11A
and MDM2 (Fig. 3C) in prostate
cancer cells.
FOXQ1 inhibition induces prostate cancer
cell apoptosis
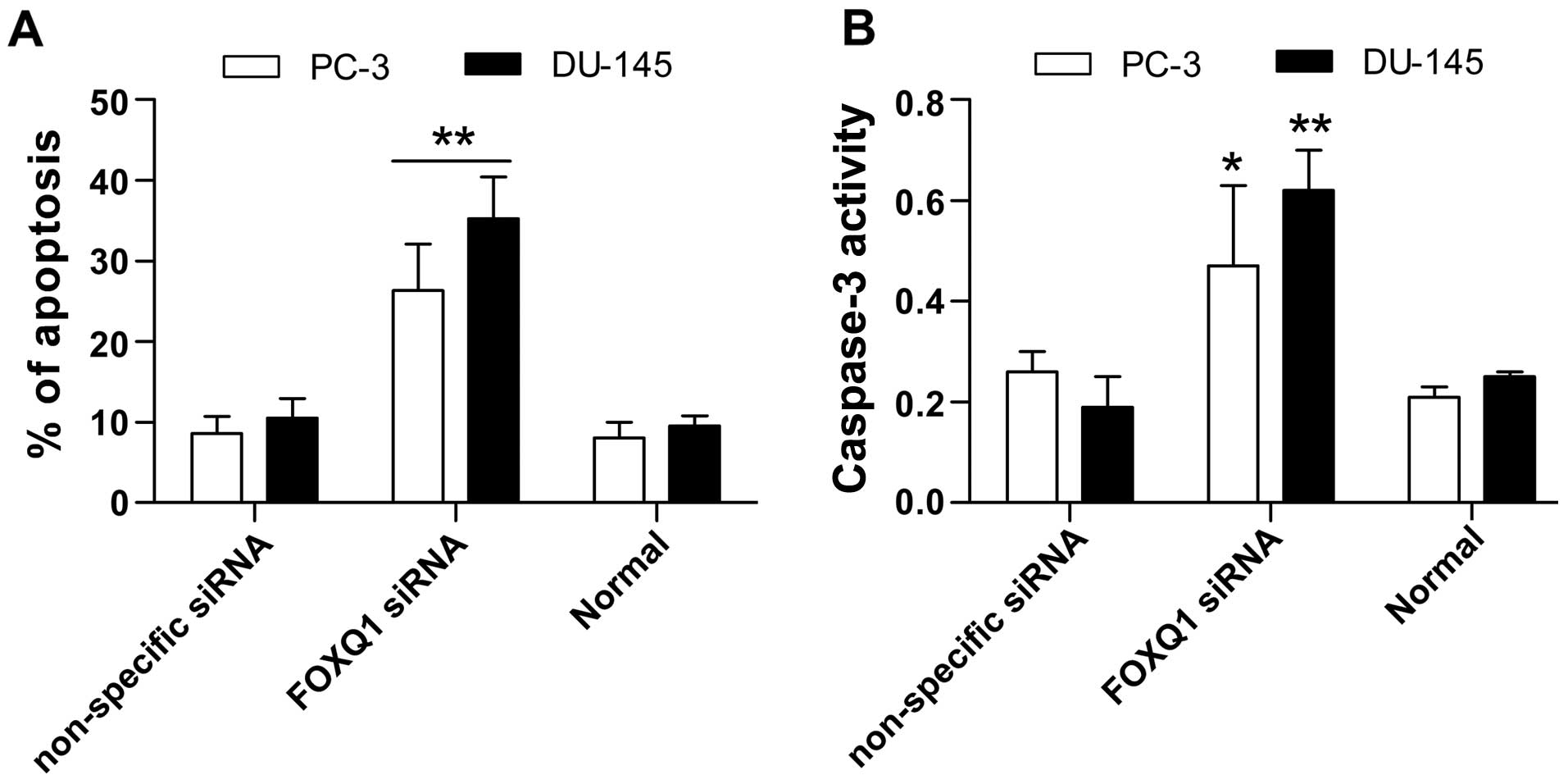
Next, we tested the effect of FOXQ1 inhibition on
apoptosis in PC-3 and DU-145 cells using the Annexin V-FITC/PI
assay and by assessing caspase-3 activity. In the Annexin V-FITC/PI
assay, apoptosis was markedly increased by FOXQ1 siRNA (Fig. 4A). Furthermore, the results show
that caspase-3 activity was greatly increased by FOXQ1 inhibition
(Fig. 4B). These results
demonstrate that suppression of FOXQ1 promotes apoptosis in
prostate cancer cells.
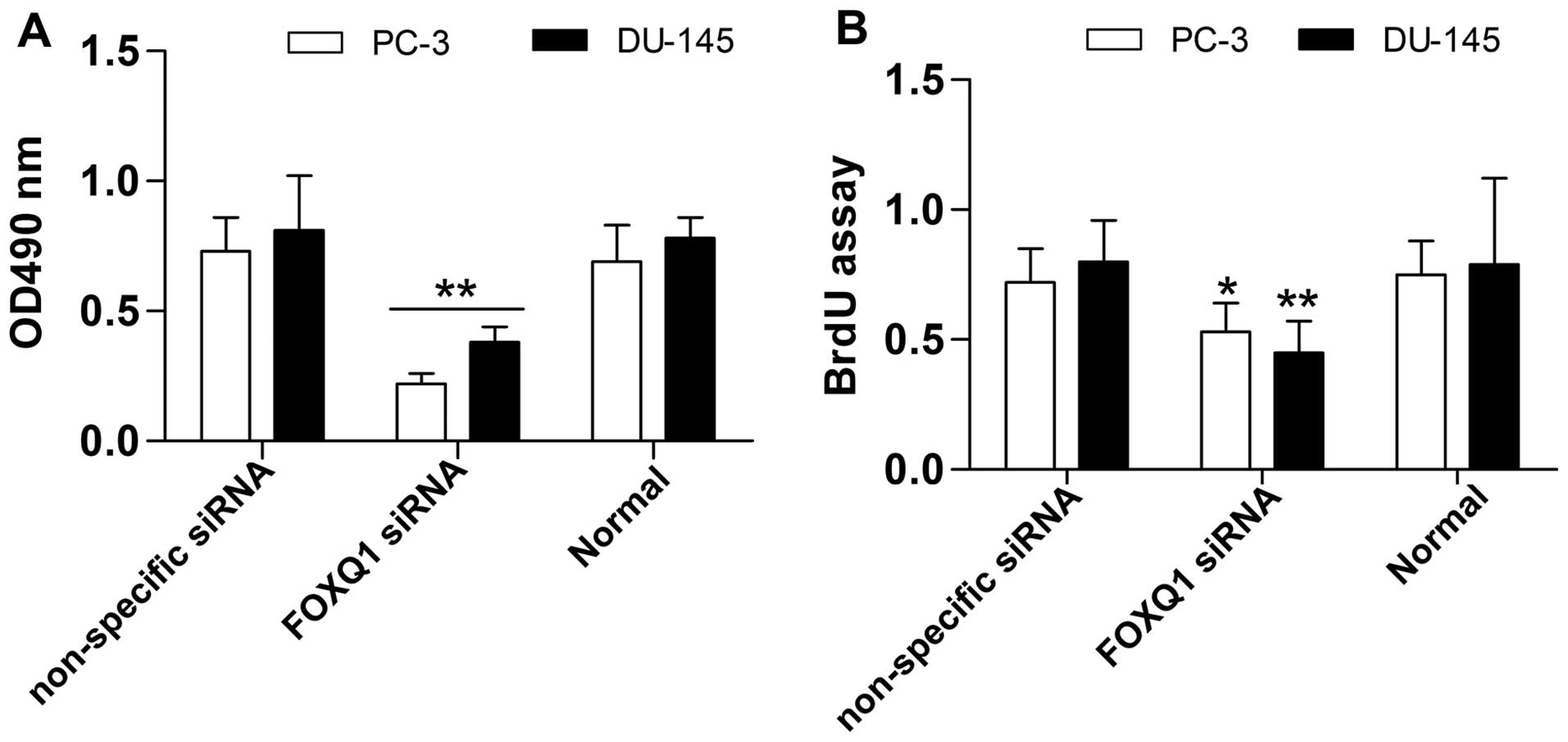
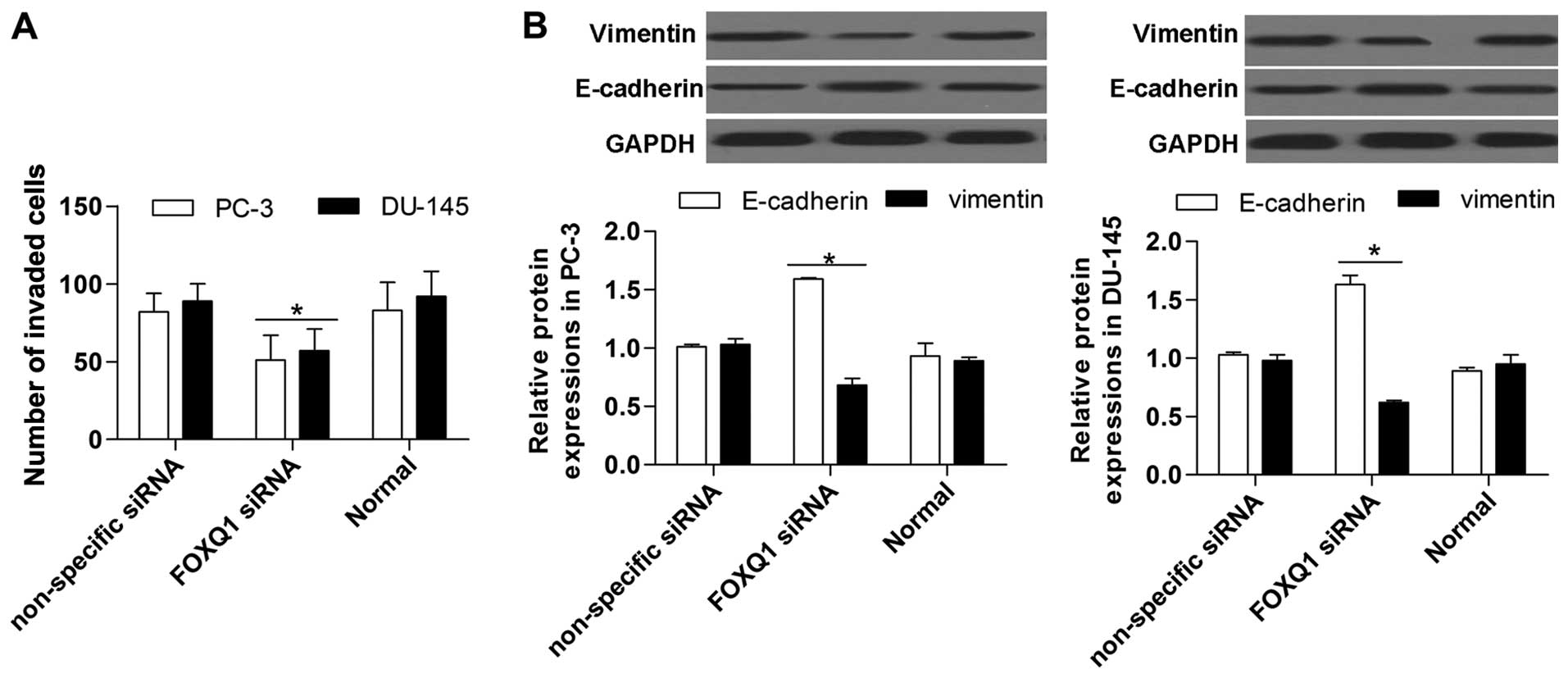
Suppression of FOXQ1 inhibits prostate
cancer cell proliferation and invasion
To further investigate the biological effect of
FOXQ1 inhibition in prostate cancer cells, we measured its effect
on prostate cancer cell proliferation and invasion. The results
show that prostate cancer cell proliferation was significantly
restrained by FOXQ1 inhibition, as detected by the MTT (Fig. 5A) and BrdU (Fig. 5B) assays. Moreover, the Transwell
invasion assay showed that FOXQ1 suppression significantly
restrained the invasion ability of prostate cancer cells (Fig. 6A). Western blot analysis indicated
that FOXQ1 suppression markedly increased E-cadherin expression and
inhibited vimentin expression in prostate cancer cells (Fig. 6B). Above all, these results suggest
that FOXQ1 inhibition suppresses the proliferation and invasion of
prostate cancer cells.
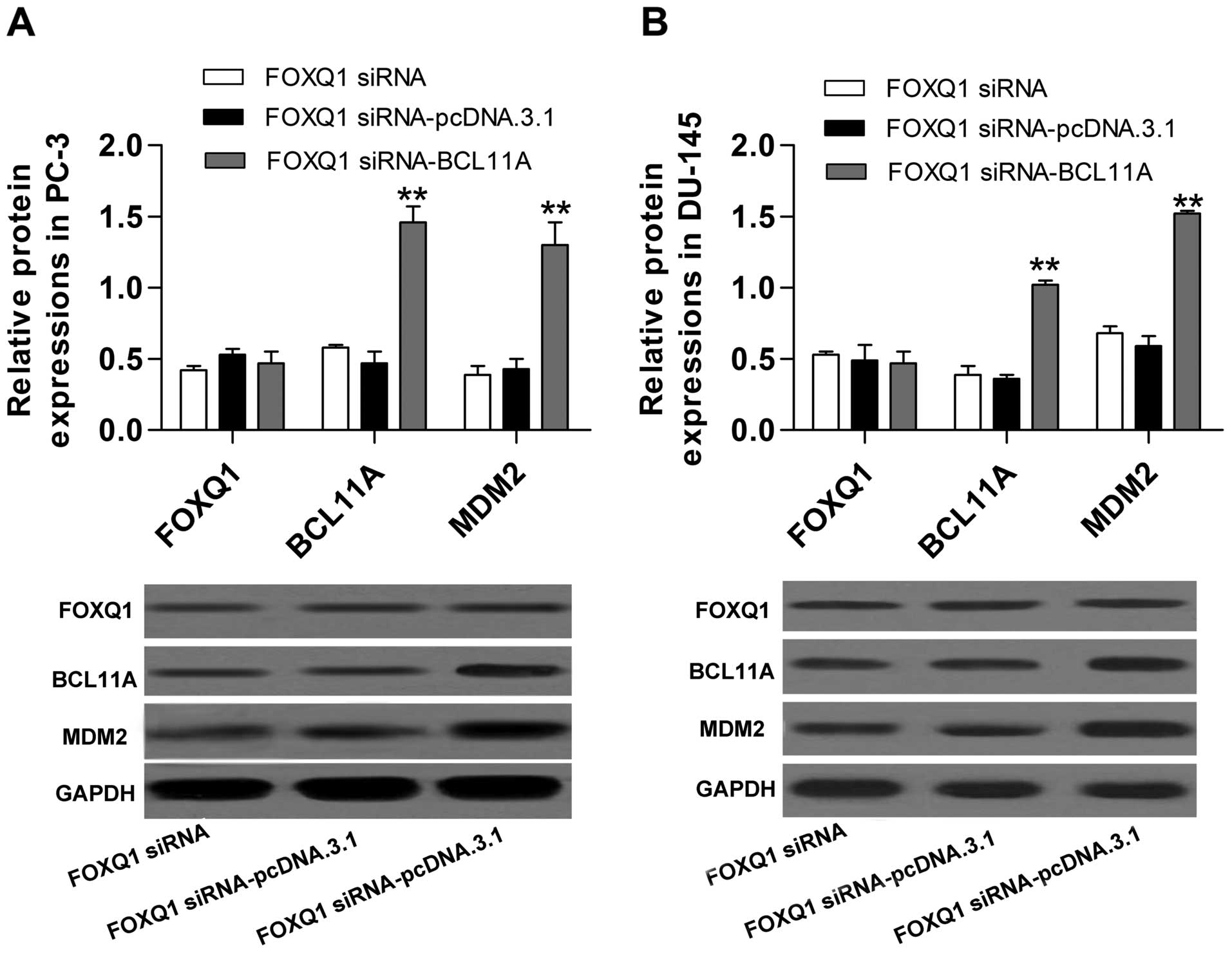
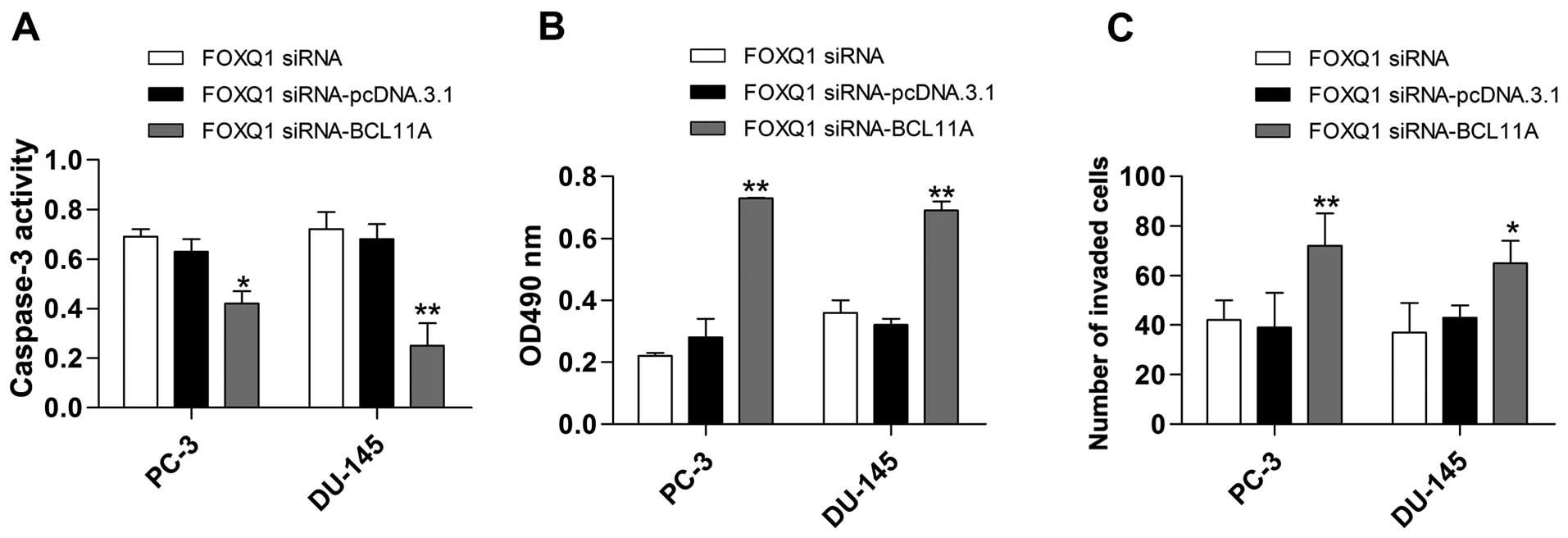
Inhibition of FOXQ1 constrains MDM2 by
controlling BCL11A expression
To investigate the potential molecular mechanism of
FOXQ1 in prostate cancer, we performed co-transfection of FOXQ1
siRNA with the recombinant plasmid pcDNA.3.1/-BCL11A. The western
blot analysis showed that upregulation of BCL11A significantly
reversed the inhibitory effect of FOXQ1 siRNA on BCL11A expression
(Fig. 7A and B). Furthermore, the
decreased expression of MDM2 induced by FOXQ1 siRNA was also
significantly reversed by BCL11A overexpression (Fig. 7A and B). Additionally, the induction
of apoptosis (Fig. 8A) and
inhibition of proliferation (Fig.
8B) and invasion (Fig. 8C)
caused by FOXQ1 suppression were all obviously reversed by BCL11A
overexpression.
Discussion
As a malignant tumor, the morbidity of prostate
cancer is increasing and the therapy for this disease is still a
challenge (37). In recent years,
the FOXQ1 oncogene has attracted increasing attention in the cancer
field. Studies have shown that FOXQ1 is a transcription factor that
plays a role in cancer metastasis and development (38). It has been reported that high
expression of FOXQ1 is found in cancers such as NSCLC, colorectal
cancer, breast cancer, glioma, and hepatocellular carcinoma
(39). FOXQ1 overexpression
enhances invasion and metastasis by promoting EMT activity in
bladder cancer and breast cancer, and FOXQ1 inhibition has an
inhibitory effect on tumor growth and metastasis in bladder cancer
and nasopharyngeal carcinoma (22,40,41).
It has also been demonstrated that FOXQ1 overexpression indicates a
poor prognosis in NSCLC (4).
However, whether FOXQ1 plays an important role in prostate cancer
remained unclear. In this study, we found that FOXQ1 is upregulated
in tumor tissues and cell lines, and suppression of it can, in
addition to constraining proliferation, invasion and EMT, induce
apoptosis in prostate cancer cells. The foundation in our study is
in line with previous studies in other cancers, further confirming
its oncogenic function and providing a novel target in the
prevention and therapy for this disease.
BCL11A is a transcription factor with a Krüppel
zinc-finger motif, and the expression of BCL11A is required for not
only the development pre-B-cells as well as thymocyte maturity, but
also immunoglobulin switching (42–44).
Moreover, BCL11A has been related to cancer. It is predicated that
BCL11A has a positive role in cancers such as NSCLC, acute
lymphoblastic leukemia, triple-negative breast cancer, squamous
cell carcinoma as well as large cell carcinoma (45,46).
Additionally, studies have shown that FOXQ1 has an effect on the
expression of BCL11A in colorectal cancer (21). Thus, we speculated that FOXQ1
facilitates the expression of BCL11A in prostate cancer. As
expected, inhibition of FOXQ1 had a significant repressive
influence on BCL11A expression in our research. Furthermore, a
recent study showed that BCL11A deficient B cells exhibit reduced
expression of MDM2 (25).
MDM2 exerts oncogenic functions in many cancers
mainly via the inhibition of the tumor suppressor p53 (47). MDM2 gene mutation or abnormal
expression leads to the destruction of the physiological balance,
and is associated with cell transformation and tumorigenesis
(48). Targeting the p53-MDM2
interaction has been considered as a method of treating cancer,
includes the inhibition of MDM2 expression by antisense
oligonucleotides (49). Studies
have reported that prostate cancer is characterized by the
overexpression of MDM2, and suppression of MDM2 has been reported
as an experimental therapy for prostate cancer (50). Additionally, it has been found that
MDM2 promotes EMT activity, whereas MDM2 inhibition constrains EMT
and reduces the migratory potential of breast cancer cells
(51). In our study, we found that
silencing FOXQ1 exerted an inhibitory function on MDM2, while
overexpression of BCL11A reversed the reduction in MDM2 induced by
FOXQ1 downregulation in prostate cancer cells. Further data
demonstrated that an increase in MDM2 enhanced EMT activity and
cell invasion and suppressed apoptosis in prostate cancer cells,
similar to the findings of Zhang et al (50). These results confirm that MDM2 is
regulated by FOXQ1 via BCL11A and further has an influence on
proliferation and apoptosis in prostate cancer cells.
Taken together, these data indicate that FOXQ1 acts
as an oncogene in prostate cancer and is expressed at a high level
in clinical prostate tissue and cell lines. Moreover, FOXQ1
inhibition constrains MDM2 by controlling BCL11A, resulting in the
suppression of proliferation and invasion, and the induction of
apoptosis in prostate cancer cells. Thus, our study provides novel
insight into the molecular mechanism underling prostate cancer and
provides a novel theoretical basis for the therapy of prostate
cancer.
Abbreviations:
|
FOXQ1
|
forkhead box Q1
|
|
BCL
|
B cell lymphoma/leukemia
|
|
MDM2
|
murine double minute 2
|
|
FBS
|
fetal bovine serum
|
|
SDS-PAGE
|
sodium dodecyl sulfate-polyacrylamide
gel electrophoresis
|
|
Annexin V-FITC/PI
|
Annexin V-fluorescein
isothiocyanate/propidium iodide
|
|
MTT
|
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
|
|
BrdU
|
bromodeoxyuridine
|
References
|
1
|
Carver BS, Chapinski C, Wongvipat J,
Hieronymus H, Chen Y, Chandarlapaty S, Arora VK, Le C, Koutcher J,
Scher H, et al: Reciprocal feedback regulation of PI3K and androgen
receptor signaling in PTEN-deficient prostate cancer. Cancer Cell.
19:575–586. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rastinehad AR, Baccala AA Jr, Chung PH,
Proano JM, Kruecker J, Xu S, Locklin JK, Turkbey B, Shih J,
Bratslavsky G, et al: D'Amico risk stratification correlates with
degree of suspicion of prostate cancer on multiparametric magnetic
resonance imaging. J Urol. 185:815–820. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Brawley OW: Prostate cancer epidemiology
in the United States. World J Urol. 30:195–200. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Feng J, Zhang X, Zhu H, Wang X, Ni S and
Huang J: FoxQ1 overexpression influences poor prognosis in
non-small cell lung cancer, associates with the phenomenon of EMT.
PLoS One. 7:e399372012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gao M, Shih IeM and Wang TL: The role of
forkhead box Q1 transcription factor in ovarian epithelial
carcinomas. Int J Mol Sci. 13:13881–13893. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Qin J, Xu Y, Li X, Wu Y, Zhou J, Wang G
and Chen L: Effects of lentiviral-mediated Foxp1 and Foxq1 RNAi on
the hepatocarcinoma cell. Exp Mol Pathol. 96:1–8. 2014. View Article : Google Scholar
|
|
7
|
Zhang H, Meng F, Liu G, Zhang B, Zhu J, Wu
F, Ethier SP, Miller F and Wu G: Forkhead transcription factor
foxq1 promotes epithelial-mesenchymal transition and breast cancer
metastasis. Cancer Res. 71:1292–1301. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Feuerborn A, Srivastava PK, Küffer S,
Grandy WA, Sijmonsma TP, Gretz N, Brors B and Gröne HJ: The
Forkhead factor FoxQ1 influences epithelial differentiation. J Cell
Physiol. 226:710–719. 2011. View Article : Google Scholar
|
|
9
|
Candelario J, Chen LY, Marjoram P, Reddy S
and Comai L: A filtering strategy identifies FOXQ1 as a potential
effector of lamin A dysfunction. Aging (Albany, NY). 4:567–577.
2012. View Article : Google Scholar
|
|
10
|
Liang SH, Yan XZ, Wang BL, Jin HF, Yao LP,
Li YN, Chen M, Nie YZ, Wang X, Guo XG, et al: Increased expression
of FOXQ1 is a prognostic marker for patients with gastric cancer.
Tumour Biol. 34:2605–2609. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Christensen J, Bentz S, Sengstag T,
Shastri VP and Anderle P: FOXQ1, a novel target of the Wnt pathway
and a new marker for activation of Wnt signaling in solid tumors.
PLoS One. 8:e600512013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Xiang XJ, Deng J, Liu YW, Wan LY, Feng M,
Chen J and Xiong JP: miR-1271 inhibits cell proliferation, invasion
and EMT in gastric cancer by targeting FOXQ1. Cell Physiol Biochem.
36:1382–1394. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yuan H, Kajiyama H, Ito S, Yoshikawa N,
Hyodo T, Asano E, Hasegawa H, Maeda M, Shibata K, Hamaguchi M, et
al: ALX1 induces snail expression to promote
epithelial-to-mesenchymal transition and invasion of ovarian cancer
cells. Cancer Res. 73:1581–1590. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Drasin DJ, Robin TP and Ford HL: Breast
cancer epithelial-to-mesenchymal transition: Examining the
functional consequences of plasticity. Breast Cancer Res.
13:2262011. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
Qi X, Li J, Zhou C, Lv C and Tian M:
miR-142-3p suppresses SOCS6 expression and promotes cell
proliferation in nasopharyngeal carcinoma. Cell Physiol Biochem.
36:1743–1752. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sun HT, Cheng SX, Tu Y, Li XH and Zhang S:
FoxQ1 promotes glioma cells proliferation and migration by
regulating NRXN3 expression. PLoS One. 8:e556932013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang W, He S, Ji J, Huang J, Zhang S and
Zhang Y: The prognostic significance of FOXQ1 oncogene
overexpression in human hepatocellular carcinoma. Pathol Res Pract.
209:353–358. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Pei Y, Wang P, Liu H, He F and Ming L:
FOXQ1 promotes esophageal cancer proliferation and metastasis by
negatively modulating CDH1. Biomed Pharmacother. 74:89–94. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Feng J, Xu L, Ni S, Gu J, Zhu H, Wang H,
Zhang S, Zhang W and Huang J: Involvement of FoxQ1 in NSCLC through
regulating EMT and increasing chemosensitivity. Oncotarget.
5:9689–9702. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Su L, Liu X, Chai N, Lv L, Wang R, Li X,
Nie Y, Shi Y and Fan D: The transcription factor FOXO4 is
down-regulated and inhibits tumor proliferation and metastasis in
gastric cancer. BMC Cancer. 14:3782014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kaneda H, Arao T, Tanaka K, Tamura D,
Aomatsu K, Kudo K, Sakai K, De Velasco MA, Matsumoto K, Fujita Y,
et al: FOXQ1 is overexpressed in colorectal cancer and enhances
tumorigenicity and tumor growth. Cancer Res. 70:2053–2063. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhu Z, Zhu Z, Pang Z, Xing Y, Wan F, Lan D
and Wang H: Short hairpin RNA targeting FOXQ1 inhibits invasion and
metastasis via the reversal of epithelial-mesenchymal transition in
bladder cancer. Int J Oncol. 42:1271–1278. 2013.PubMed/NCBI
|
|
23
|
Zhang J, Li W, Dai S, Tai X, Jia J and Guo
X: FOXQ1 is overexpressed in laryngeal carcinoma and affects cell
growth, cell cycle progression and cell invasion. Oncol Lett.
10:2499–2504. 2015.PubMed/NCBI
|
|
24
|
Kutlar A, Ataga K, Reid M, Vichinsky EP,
Neumayr L, Blair-Britt L, Labotka R, Glass J, Keefer JR, Wargin WA,
et al: A phase 1/2 trial of HQK-1001, an oral fetal globin inducer,
in sickle cell disease. Am J Hematol. 87:1017–1021. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yu Y, Wang J, Khaled W, Burke S, Li P,
Chen X, Yang W, Jenkins NA, Copeland NG, Zhang S, et al: Bcl11a is
essential for lymphoid development and negatively regulates p53. J
Exp Med. 209:2467–2483. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Staszewski O, Baker RE, Ucher AJ, Martier
R, Stavnezer J and Guikema JE: Activation-induced cytidine
deaminase induces reproducible DNA breaks at many non-Ig loci in
activated B cells. Mol Cell. 41:232–242. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tao H, Ma X, Su G, Yin J, Xie X, Hu C,
Chen Z, Tan D, Xu Z, Zheng Y, et al: BCL11A expression in acute
myeloid leukemia. Leuk Res. 41:71–75. 2016. View Article : Google Scholar
|
|
28
|
Jiang BY, Zhang XC, Su J, Meng W, Yang XN,
Yang JJ, Zhou Q, Chen ZY, Chen ZH, Xie Z, et al: BCL11A
overexpression predicts survival and relapse in non-small cell lung
cancer and is modulated by microRNA-30a and gene amplification. Mol
Cancer. 12:612013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wade M, Li YC and Wahl GM: MDM2, MDMX and
p53 in oncogenesis and cancer therapy. Nat Rev Cancer. 13:83–96.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li T, Kon N, Jiang L, Tan M, Ludwig T,
Zhao Y, Baer R and Gu W: Tumor suppression in the absence of
p53-mediated cell-cycle arrest, apoptosis, and senescence. Cell.
149:1269–1283. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wan Y, Wu W, Yin Z, Guan P and Zhou B:
MDM2 SNP309, gene-gene interaction, and tumor susceptibility: An
updated meta-analysis. BMC Cancer. 11:2082011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhang J, Sun Q, Zhang Z, Ge S, Han ZG and
Chen WT: Loss of microRNA-143/145 disturbs cellular growth and
apoptosis of human epithelial cancers by impairing the MDM2-p53
feedback loop. Oncogene. 32:61–69. 2013. View Article : Google Scholar
|
|
33
|
Li Q and Lozano G: Molecular pathways:
Targeting Mdm2 and Mdm4 in cancer therapy. Clin Cancer Res.
19:34–41. 2013. View Article : Google Scholar :
|
|
34
|
Zhang YY and Zhou LM: Sirt3 inhibits
hepatocellular carcinoma cell growth through reducing Mdm2-mediated
p53 degradation. Biochem Biophys Res Commun. 423:26–31. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Tovar C, Higgins B, Kolinsky K, Xia M,
Packman K, Heimbrook DC and Vassilev LT: MDM2 antagonists boost
antitumor effect of androgen withdrawal: Implications for therapy
of prostate cancer. Mol Cancer. 10:492011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Welsh JB, Sapinoso LM, Su AI, Kern SG,
Wang-Rodriguez J, Moskaluk CA, Frierson HF Jr and Hampton GM:
Analysis of gene expression identifies candidate markers and
pharmacological targets in prostate cancer. Cancer Res.
61:5974–5978. 2001.PubMed/NCBI
|
|
37
|
Shiozawa Y, Pedersen EA, Havens AM, Jung
Y, Mishra A, Joseph J, Kim JK, Patel LR, Ying C, Ziegler AM, et al:
Human prostate cancer metastases target the hematopoietic stem cell
niche to establish footholds in mouse bone marrow. J Clin Invest.
121:1298–1312. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Xia L, Huang W, Tian D, Zhang L, Qi X,
Chen Z, Shang X, Nie Y and Wu K: Forkhead box Q1 promotes
hepatocellular carcinoma metastasis by transactivating ZEB2 and
VersicanV1 expression. Hepatology. 59:958–973. 2014. View Article : Google Scholar
|
|
39
|
Bao B, Azmi AS, Aboukameel A, Ahmad A,
Bolling-Fischer A, Sethi S, Ali S, Li Y, Kong D, Banerjee S, et al:
Pancreatic cancer stem-like cells display aggressive behavior
mediated via activation of FoxQ1. J Biol Chem. 289:14520–14533.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Qiao Y, Jiang X, Lee ST, Karuturi RK, Hooi
SC and Yu Q: FOXQ1 regulates epithelial-mesenchymal transition in
human cancers. Cancer Res. 71:3076–3086. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Peng XH, Huang HR, Lu J, Liu X, Zhao FP,
Zhang B, Lin SX, Wang L, Chen HH, Xu X, et al: miR-124 suppresses
tumor growth and metastasis by targeting Foxq1 in nasopharyngeal
carcinoma. Mol Cancer. 13:1862014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Costa FC, Fedosyuk H, Neades R, de Los
Rios JB, Barbas CF III and Peterson KR: Induction of fetal
hemoglobin in vivo mediated by a synthetic γ-globin zinc finger
activator. Anemia. 2012:5078942012. View Article : Google Scholar
|
|
43
|
Esteghamat F, Gillemans N, Bilic I, van
den Akker E, Cantù I, van Gent T, Klingmüller U, van Lom K, von
Lindern M, Grosveld F, et al: Erythropoiesis and globin switching
in compound Klf1:Bcl11a mutant mice. Blood. 121:2553–2562. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Liu P, Keller JR, Ortiz M, Tessarollo L,
Rachel RA, Nakamura T, Jenkins NA and Copeland NG: Bcl11a is
essential for normal lymphoid development. Nat Immunol. 4:525–532.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Kadoch C, Hargreaves DC, Hodges C, Elias
L, Ho L, Ranish J and Crabtree GR: Proteomic and bioinformatic
analysis of mammalian SWI/SNF complexes identifies extensive roles
in human malignancy. Nat Genet. 45:592–601. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Khaled WT, Choon Lee S, Stingl J, Chen X,
Raza Ali H, Rueda OM, Hadi F, Wang J, Yu Y, Chin SF, et al: BCL11A
is a triple-negative breast cancer gene with critical functions in
stem and progenitor cells. Nat Commun. 6:59872015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Bond GL, Hu W, Bond EE, Robins H, Lutzker
SG, Arva NC, Bargonetti J, Bartel F, Taubert H, Wuerl P, et al: A
single nucleotide polymorphism in the MDM2 promoter attenuates the
p53 tumor suppressor pathway and accelerates tumor formation in
humans. Cell. 119:591–602. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Jones SN, Hancock AR, Vogel H, Donehower
LA and Bradley A: Overexpression of Mdm2 in mice reveals a
p53-independent role for Mdm2 in tumorigenesis. Proc Natl Acad Sci
USA. 95:15608–15612. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Klein C and Vassilev LT: Targeting the
p53-MDM2 interaction to treat cancer. Br J Cancer. 91:1415–1419.
2004.PubMed/NCBI
|
|
50
|
Zhang Z, Li M, Wang H, Agrawal S and Zhang
R: Antisense therapy targeting MDM2 oncogene in prostate cancer:
Effects on proliferation, apoptosis, multiple gene expression, and
chemotherapy. Proc Natl Acad Sci USA. 100:11636–11641. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Wang W, Zhang X, Qin JJ, Voruganti S, Nag
SA, Wang MH, Wang H and Zhang R: Natural product ginsenoside
25-OCH3-PPD inhibits breast cancer growth and metastasis through
down-regulating MDM2. PLoS One. 7:e415862012. View Article : Google Scholar : PubMed/NCBI
|