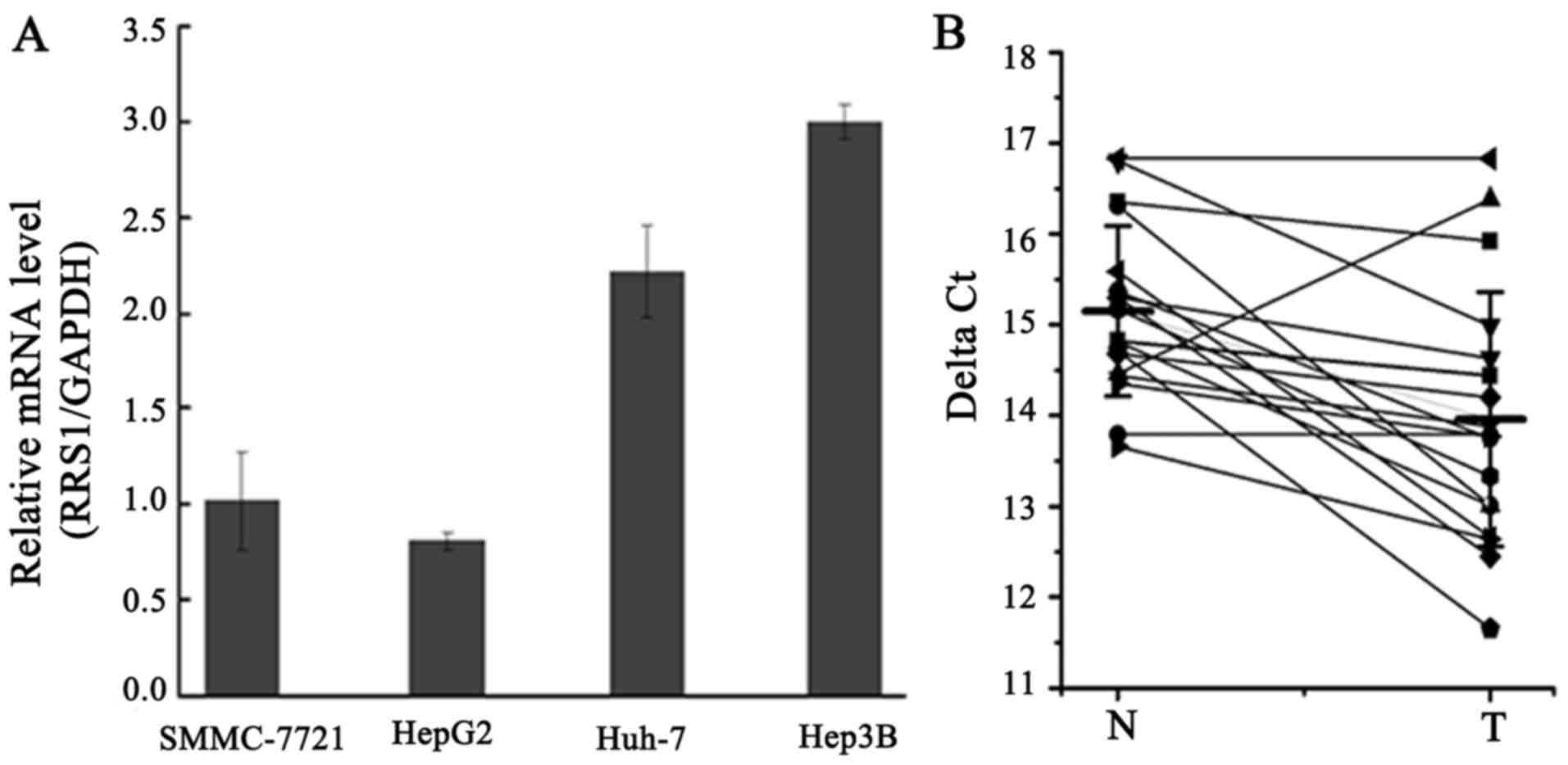
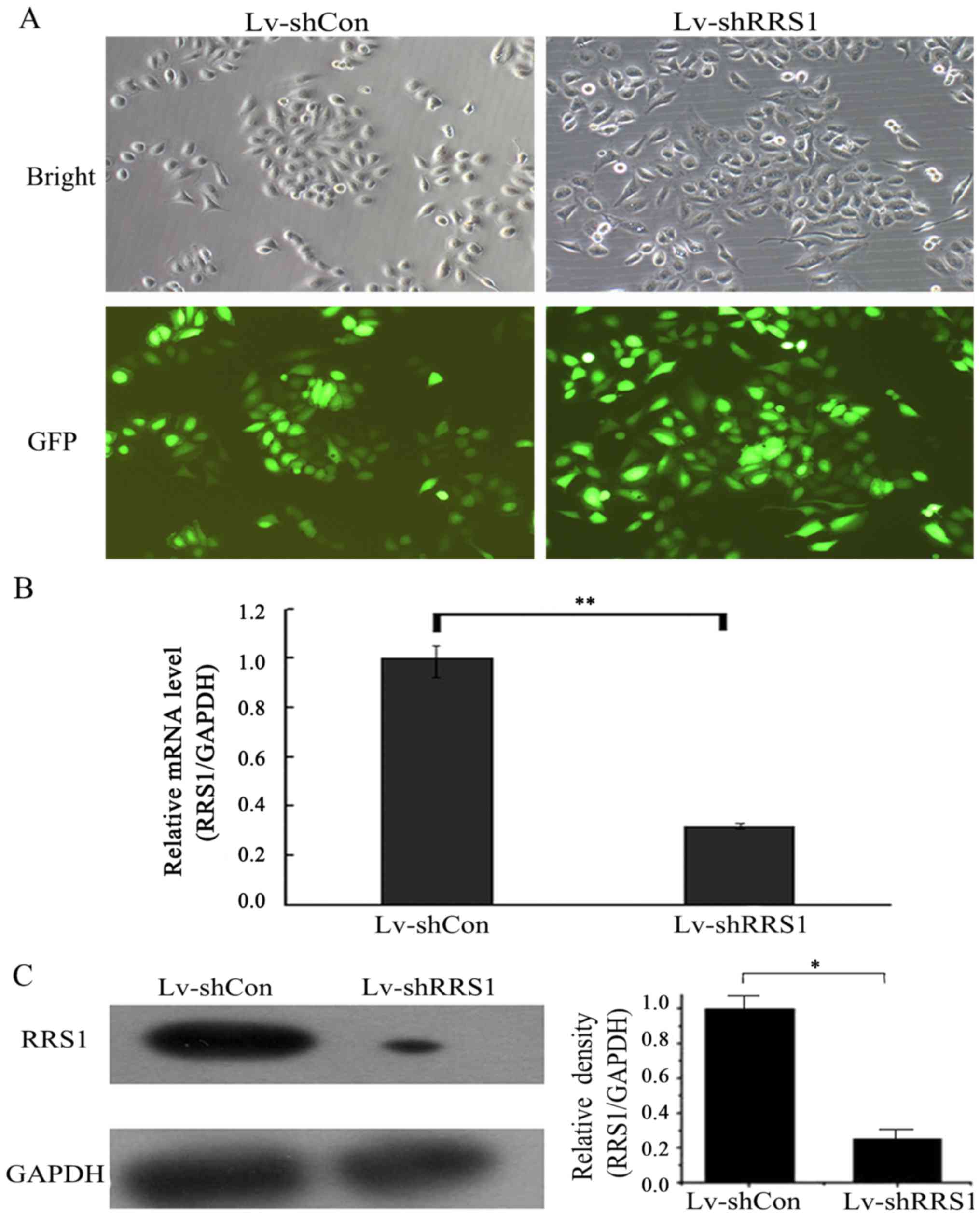
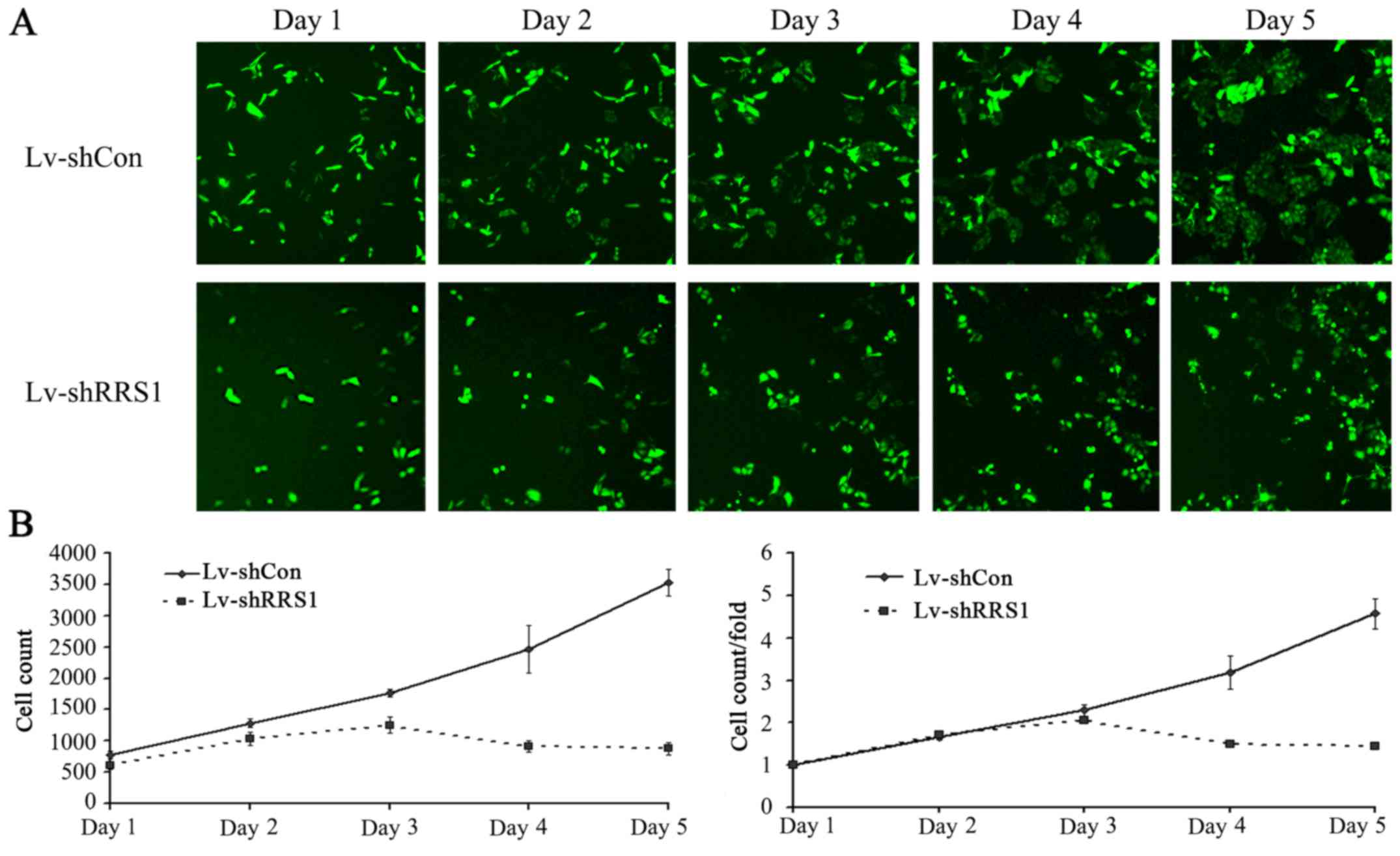
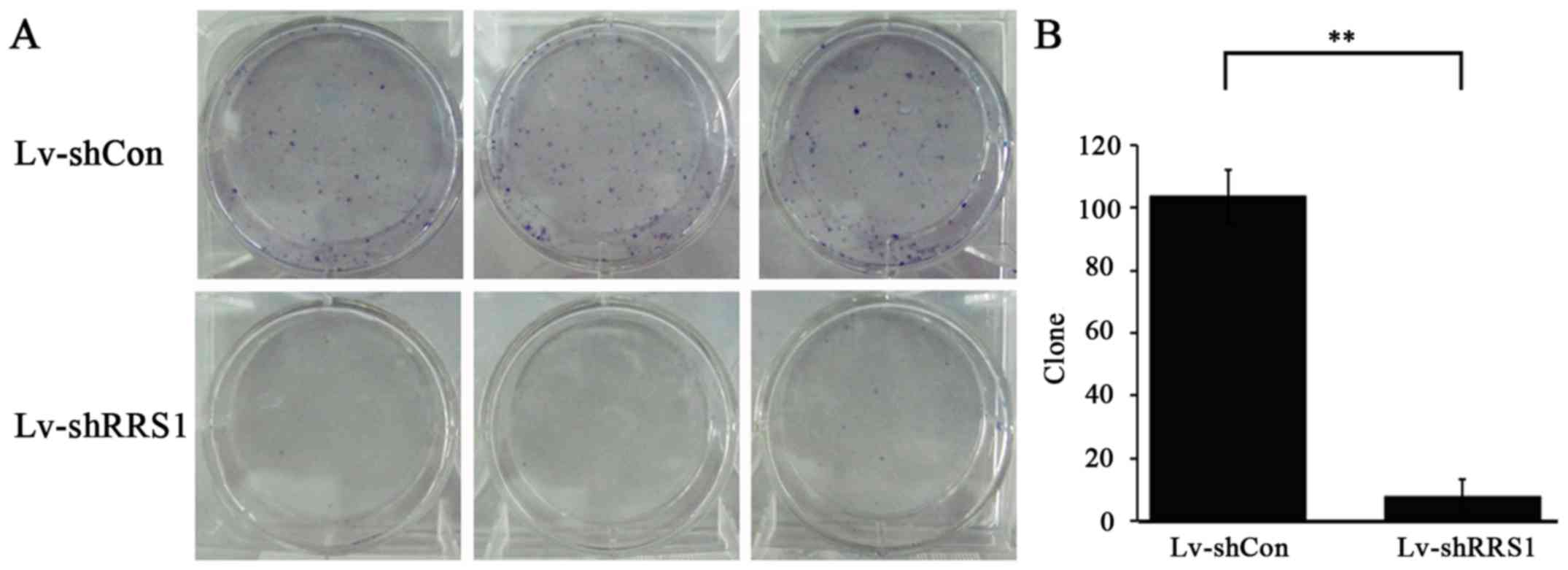
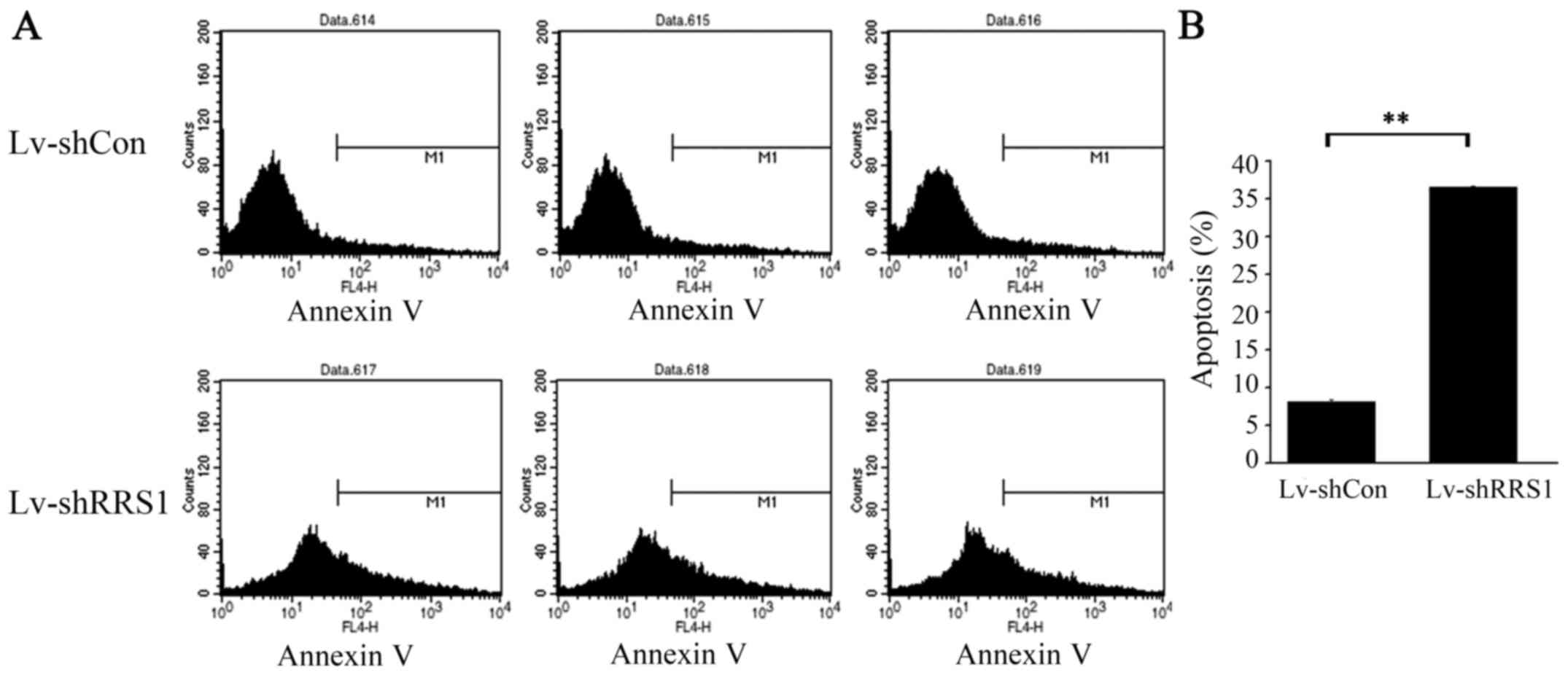
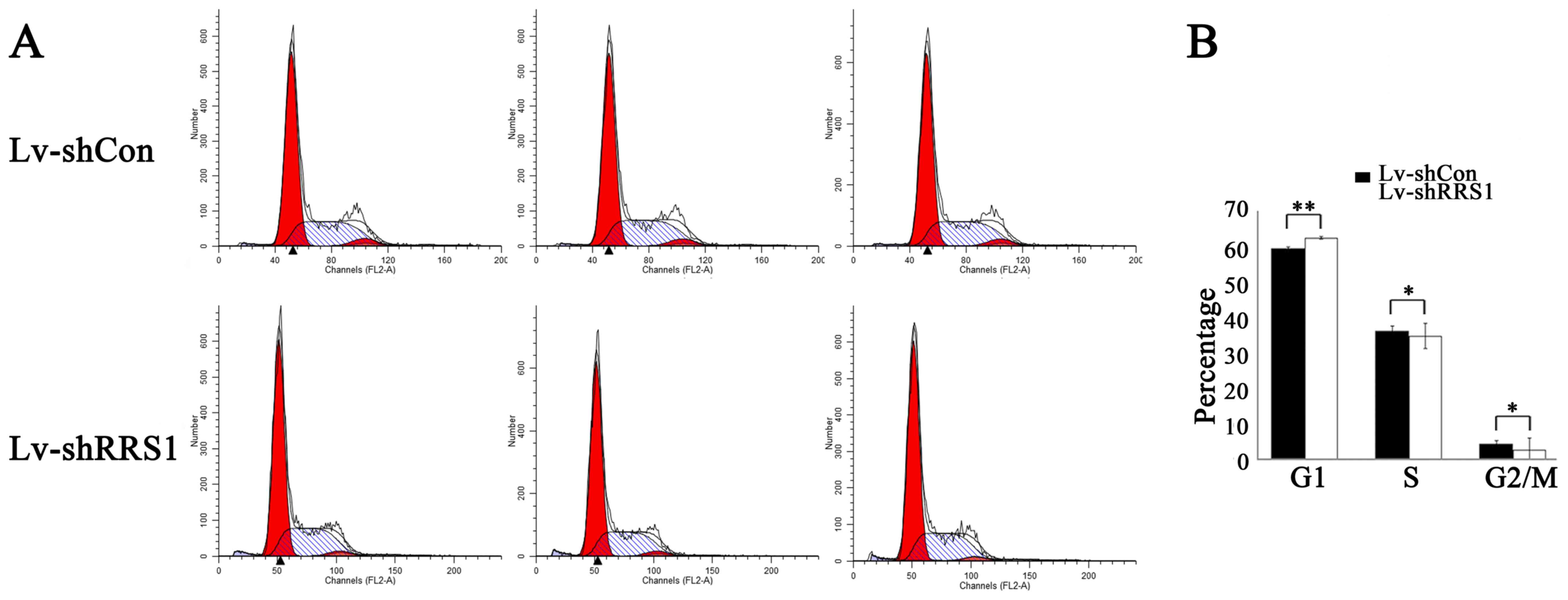
|
1
|
World Health Organization, . World Cancer
Report 2014Stewart BW and Wild CP: World Health Organization;
International Agency for Research on Cancer. WHO Press, Geneva:
2015, View Article : Google Scholar
|
|
2
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zuo TT, Zheng RS, Zhang SW, Zeng HM and
Chen WQ: Incidence and mortality of liver cancer in China in 2011.
Chin J Cancer. 34:508–513. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wei KR, Yu X, Zheng RS, Peng XB, Zhang SW,
Ji MF, Liang ZH, Ou ZX and Chen WQ: Incidence and mortality of
liver cancer in China, 2010. Chin J Cancer. 33:388–394.
2014.PubMed/NCBI
|
|
5
|
Petrick JL, Braunlin M, Laversanne M,
Valery PC, Bray F and McGlynn KA: International trends in liver
cancer incidence, overall and by histologic subtype, 1978–2007. Int
J Cancer. 139:1534–1545. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Fitzmorris P, Shoreibah M, Anand BS and
Singal AK: Management of hepatocellular carcinoma. J Cancer Res
Clin Oncol. 141:861–876. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nguyen MH and Keeffe EB: Screening for
hepatocellular carcinoma. J Clin Gastroenterol. 35 Suppl 2:S86–S91.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Shimizu D, Inokawa Y, Sonohara F, Inaoka K
and Nomoto S: Search for useful biomarkers in hepatocellular
carcinoma, tumor factors and background liver factors (Review).
Oncol Rep. 37:2527–2542. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pecot CV, Calin GA, Coleman RL,
Lopez-Berestein G and Sood AK: RNA interference in the clinic:
challenges and future directions. Nat Rev Cancer. 11:59–67. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sioud M: RNA interference: Mechanisms,
technical challenges, and therapeutic opportunities. Methods Mol
Biol. 1218:1–15. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Holoch D and Moazed D: RNA-mediated
epigenetic regulation of gene expression. Nat Rev Genet. 16:71–84.
2015. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Liu H, Liang S, Yang X, Ji Z, Zhao W, Ye X
and Rui J: RNAi-mediated RPL34 knockdown suppresses the growth of
human gastric cancer cells. Oncol Rep. 34:2267–2272. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu K, Li X, Cao Y, Ge Y, Wang J and Shi
B: MiR-132 inhibits cell proliferation, invasion and migration of
hepatocellular carcinoma by targeting PIK3R3. Int J Oncol.
47:1585–1593. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang Q, Hu H, Shi X and Tang W: Knockdown
of S100P by lentiviral-mediated RNAi promotes apoptosis and
suppresses the colony-formation ability of gastric cancer cells.
Oncol Rep. 31:2344–2350. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Du P, Ye L, Yang Y and Jiang WG: Candidate
of metastasis 1 regulates in vitro growth and invasion of
bladder cancer cells. Int J Oncol. 42:1249–1256. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Roop RP and Ma CX: Endocrine resistance in
breast cancer: Molecular pathways and rational development of
targeted therapies. Future Oncol. 8:273–292. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Li J and Huang L: Targeted delivery of
RNAi therapeutics for cancer therapy. Nanomedicine (Lond).
5:1483–1486. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Montanaro L, Treré D and Derenzini M:
Changes in ribosome biogenesis may induce cancer by down-regulating
the cell tumor suppressor potential. Biochim Biophys Acta.
1825:101–110. 2012.PubMed/NCBI
|
|
19
|
Donati G, Montanaro L and Derenzini M:
Ribosome biogenesis and control of cell proliferation: p53 is not
alone. Cancer Res. 72:1602–1607. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Montanaro L, Treré D and Derenzini M:
Nucleolus, ribosomes, and cancer. Am J Pathol. 173:301–310. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Tsuno A, Miyoshi K, Tsujii R, Miyakawa T
and Mizuta K: RRS1, a conserved essential gene, encodes a novel
regulatory protein required for ribosome biogenesis in
Saccharomyces cerevisiae. Mol Cell Biol. 20:2066–2074. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhang J, Harnpicharnchai P, Jakovljevic J,
Tang L, Guo Y, Oeffinger M, Rout MP, Hiley SL, Hughes T and
Woolford JL Jr: Assembly factors Rpf2 and Rrs1 recruit 5S rRNA and
ribosomal proteins rpL5 and rpL11 into nascent ribosomes. Genes
Dev. 21:2580–2592. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Fossale E, Wheeler VC, Vrbanac V, Lebel
LA, Teed A, Mysore JS, Gusella JF, MacDonald ME and Persichetti F:
Identification of a presymptomatic molecular phenotype in Hdh CAG
knock-in mice. Hum Mol Genet. 11:2233–2241. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gambe AE, Matsunaga S, Takata H,
Ono-Maniwa R, Baba A, Uchiyama S and Fukui K: A nucleolar protein
RRS1 contributes to chromosome congression. FEBS Lett.
583:1951–1956. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wan K, Kawara H, Yamamoto T, Kume K,
Yabuki Y, Goshima T, Kitamura K, Ueno M, Kanai M, Hirata D, et al:
The essential function of Rrs1 in ribosome biogenesis is conserved
in budding and fission yeasts. Yeast. 32:607–614. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Carnemolla A, Fossale E, Agostoni E,
Michelazzi S, Calligaris R, De Maso L, Del Sal G, MacDonald ME and
Persichetti F: Rrs1 is involved in endoplasmic reticulum stress
response in Huntington disease. J Biol Chem. 284:18167–18173. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Miyoshi K, Tsujii R, Yoshida H, Maki Y,
Wada A, Matsui Y, Toh-E A and Mizuta K: Normal assembly of 60 S
ribosomal subunits is required for the signaling in response to a
secretory defect in Saccharomyces cerevisiae. J Biol Chem.
277:18334–18339. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Morita D, Miyoshi K, Matsui Y, Toh-E A,
Shinkawa H, Miyakawa T and Mizuta K: Rpf2p, an evolutionarily
conserved protein, interacts with ribosomal protein L11 and is
essential for the processing of 27 SB Pre-rRNA to 25 S rRNA and the
60 S ribosomal subunit assembly in Saccharomyces cerevisiae.
J Biol Chem. 277:28780–28786. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Nariai M, Tanaka T, Okada T, Shirai C,
Horigome C and Mizuta K: Synergistic defect in 60S ribosomal
subunit assembly caused by a mutation of Rrs1p, a ribosomal protein
L11-binding protein, and 3′-extension of 5S rRNA in
Saccharomyces cerevisiae. Nucleic Acids Res. 33:4553–4562.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Miyoshi K, Shirai C, Horigome C, Takenami
K, Kawasaki J and Mizuta K: Rrs1p, a ribosomal protein L11-binding
protein, is required for nuclear export of the 60S pre-ribosomal
subunit in Saccharomyces cerevisiae. FEBS Lett. 565:106–110.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lois C, Hong EJ, Pease S, Brown EJ and
Baltimore D: Germline transmission and tissue-specific expression
of transgenes delivered by lentiviral vectors. Science.
295:868–872. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Laemmli UK: Cleavage of structural
proteins during the assembly of the head of bacteriophage T4.
Nature. 227:680–685. 1970. View
Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhang C, Liu K, Li T, Fang J, Ding Y, Sun
L, Tu T, Jiang X, Du S, Hu J, et al: miR-21: A gene of dual
regulation in breast cancer. Int J Oncol. 48:161–172. 2016.
View Article : Google Scholar : PubMed/NCBI
|