1. Introduction
Infertility and fecundity problems concern 10-18% of
partners in their reproductive years comprising around one million
females and males in Poland (1).
According to the World Health Organization definition, infertility
is a disease of the reproductive system defined by the failure to
achieve a clinical pregnancy after 12 months or more of regular
unprotected sexual intercourse (2).
There are multiple factors affecting fertility in both sexes.
Female causes include defects in ovulation, as well as
transplantation and implantation of the ovum. Male causes involve
abnormalities in spermatogenesis and delivery of the sperm
(3). Moreover, epidemiological
factors and change in the general pattern of living have a
significant impact on fecundity problems. One of them is postponed
decision about pregnancy.
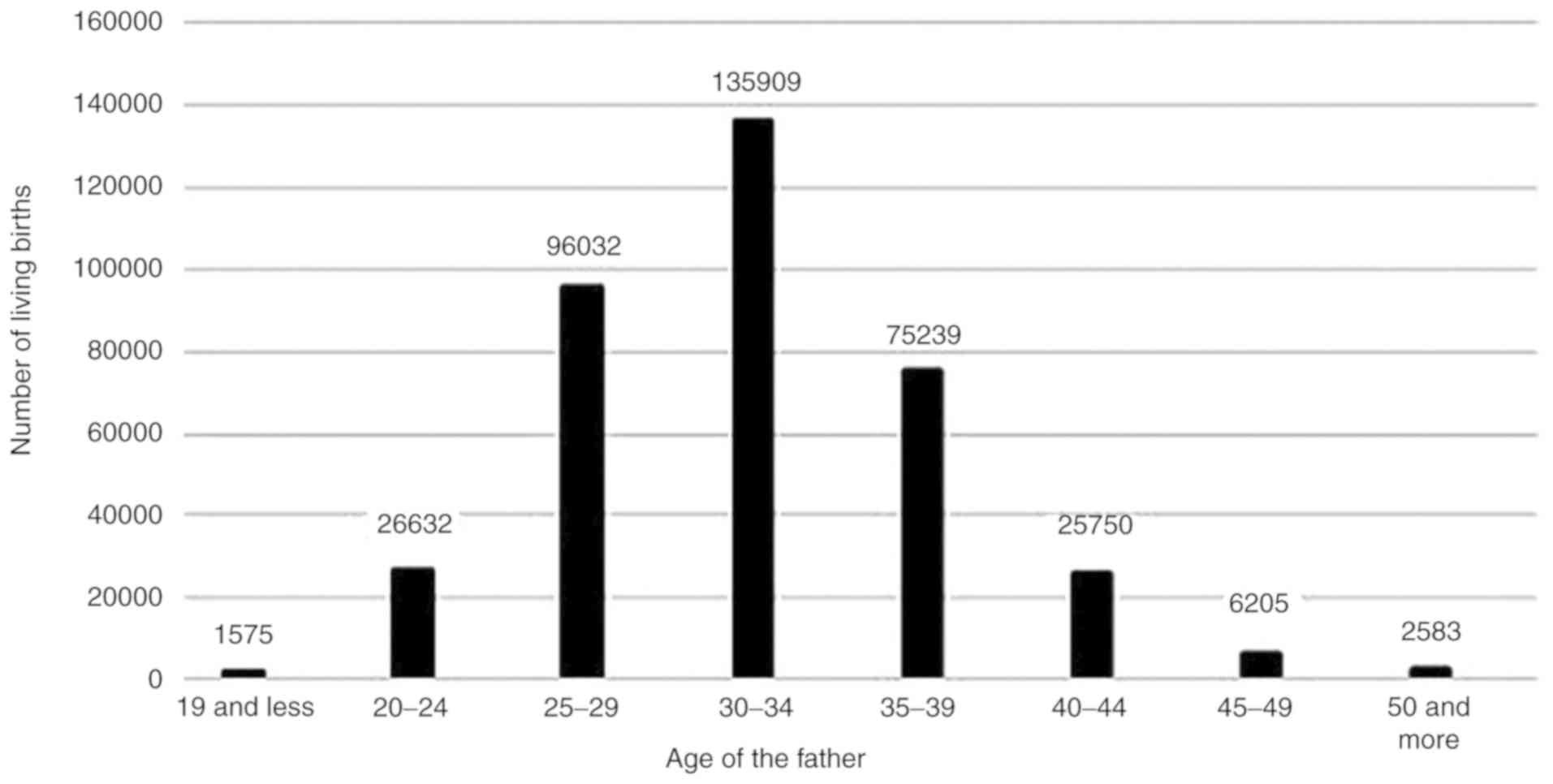
As reported by The Demographic Yearbook of Poland
2017, in 2016, Poland recorded 382,257 live births (4). The Polish population was divided into
groups according to age and sex in order to indicate
differentiation in the fertility model. The highest rate of living
births among women was observed within the group aged 25-29 years
(126,651), whereas in the case of men this value applies to the
group aged 30-34 years (135,909) (Fig.
1). Within the period 1980-2000, the statistics were different.
The highest number of living births was observed in a group of
women aged 20-24 years: 293,944 births in 1980; 199,575 in 1990 and
127,319 in 2000. In 2010, the pattern changed and the highest
number of living births was evident in females aged 25-29 years
(152,711). Data regarding fathers were not recorded in the
statistics. It underlines the fact that the impact of paternal age
regarding the offspring was neglected. While taking into account
the fertility rate, it also revealed a decreasing tendency from
17.0 per 1,000 women (2000) to 12.3 per 1,000 (2015). Only female
fertility was assessed (4).
The problem of infertility is shared equally (50%)
by both sexes (5). Research and
analysis of factors that affect male fertility are limited,
especially, regarding the age of the father (6). Data show that, with advanced age the
quality of semen decreases resulting in 3-22% decrease in semen
volume, 3-37% in motility and 4-18% in normal morphology (7). These modifications follow anatomical and
functional changes of the aging organism. Reduced volume of semen
is a consequence of insufficiency of seminal vesicles and decreased
motility follows smooth muscular atrophy in prostate (7). Moskovtsev et al (8) indicated the genetic implications
followed by advanced parental age. That study presented decreased
DNA integrity in a group of men above 45 years of age. In addition,
males with higher DNA fragmentations have lowered chances in both
in vitro and in vivo fertilization and further
development of the embryo (8).
Additionally, parental age has a significant impact on gene
mutation. One example is single gene mutation of fibroblast growth
factor receptor 3 (FGFR3), the cause of achondroplasia,
commonly known as dwarfism. Similarly, advanced age of the father
may increase the risk of FGFR2 mutations resulting in
Crouzon, Apert, and Pfeiffer syndromes. Genetic mutations linked
with advanced paternal age are associated with the stimulation of
carcinogenesis especially in terms of breast cancer, childhood
leukemia and childhood central nervous system tumors (9). Despite the fact that Down syndrome is
commonly known as related to advanced maternal age, previous
findings indicate that increased paternal age influences the chance
of mutational development as well (9).
Apart from enhanced occurrence of somatic disorders,
the effect of father's age on mental health and neurodevelopmental
disorders in offspring is continuously being examined. A
significant aspect is the correlation of autism and age of the
parent. Previous findings suggest that the development of this
disorder may result from epigenetic differences in paternal sperm.
These changes may directly affect the cerebellum of the offspring
(10). Moreover, advanced paternal
age is an important factor with psychiatric conditions such as
schizophrenia, bipolar disorder and even intelligence of the child
(11). It also has an impact on
future learning potential.
Currently, researchers have focused on determining a
threshold level of the age of the father that is followed by
negative implications on the developing fetus. Literature shows
differentiated statistics ranging from 30 to 50 years of age,
indicating the age of 40 as the threshold of advanced paternal age
most frequently (9). Nevertheless,
findings indicate the importance of the young age of the father in
order that possible somatic and mental complications of the future
offspring may be prevented.
2. Genetic syndromes
Paternal age has a greater impact on cases of
sporadic autosomal dominant congenital diseases than maternal age,
such as Apert, Crouzon, Pfeiffer, Noonan and Costello syndromes,
multiple endocrine neoplasia (types 2A and 2B) and achondroplasia
(12) (Table I). The main cause is the difference in
gametogenesis between men and women. Female oocytes do not go
through DNA replication in the mature age in contrast to male
spermatogenesis. Ageing leads to more DNA replications during
spermatogenesis in testicles. It increases the risk of copy error
mutations such as small deletions and insertions (13).
 | Table IGenetic disorders connected with
advanced paternal age. |
Table I
Genetic disorders connected with
advanced paternal age.
| Name of the
disorder | Mutated gene | Phenotype |
|---|
| Apert syndrome | FGFR2 | Craniosynostosis,
syndactyly, high-arched palate, mandibular prognathism, crowded
teeth |
| Crouzon
syndrome | FGFR2 | Craniosynostosis,
exophthalmos, hypertelorism, hypoplastic maxilla, hypodontia |
| Pfeiffer
syndrome | FGFR2 | Craniosynostosis,
turribrachycephaly, proptosis, hypertelorism, maxillary hypoplasia,
syndactyly |
| Noonan
syndrome | PTPN11 | Short height,
congenital heart disease, hypertelorism, skeletal malformations,
bleeding problems |
| Costello
syndrome | HRAS | Intellectual
disability, cardiac abnormalities, loose folds of skin on hands and
legs, too loose joints |
| Achondroplasia | FGFR3 | Shortened arms and
legs, prominent forehead, large head, trident hands |
It is estimated that spermatic chromosomes are
replicated 35 times by age 15, 150 times by age 20, 380 times by
age 30, 610 times by age 40, and 840 times by age 50 (14,15). In
opposition to men, female gametogenesis can lead to aneuploidy, and
it is also related to ageing (16).
Moreover, men over the age of 45 are more likely to have a child
with trisomy 21, versus men less than 30 years of age (17). The examples of autosomal dominant
diseases are Apert syndrome and achondroplasia, caused by mutations
in fibroblast growth factor receptor 2 and 3 (FGFR2, FGFR3).
Multiple endocrine neoplasia syndrome, is caused by mutations in
RET gene, and it presents with tumors in two or more glands
of the endocrine system (9).
3. Perinatal complications
There are only a few reports taking into
consideration advanced paternal age as a reason for pre-mature
birth, low Apgar score or admission to a neonatal intensive care
department. This topic requires further investigation. There is no
doubt that, older fathers are more likely to have infant with birth
defects, stillbirth and low birthweight (18-22).
An Italian study, conducted during the period 1990-1998 revealed
that men aged 45 to 49 years had a higher risk of having a pre-term
birth, than men aged 25 to 29 years (23). In the United States, a
population-based cohort study between 2007 and 2016 reported that
children born to fathers over the age of 45 had 14% higher risk of
preterm birth (21). Additionally,
for fathers aged over 35 years there was a higher risk of having a
baby with low birthweight. Mothers with older partners had a 34%
higher risk of gestational diabetes. Furthermore, fathers over the
age of 55, had a higher risk of having an infant with low (below 8)
Apgar score (21).
4. Congenital defects
The most common congenital defects are heart
malformations, which affect 5-10/1000 live births (24-26).
Nevertheless there are divergences between studies, some of which
confirm the connection of advanced paternal age with heart defects
in offspring (27-30),
while others are contradictory and show that older fathers have
fewer heart defects in their children (31-33).
One study reported that men over the age of 40 years were more
likely to have a child with ventricular septal defect (VSD) but not
atrial septal defect (ASD) (34).
Moreover, surveys prove the increased risk of having patent ductus
arteriosus (PDA) in the offspring of fathers older than 45 years
(28,35).
Oral cleft occurs in approximately 1-2/1000 live
births. The probability of this defect increases with both paternal
and maternal advanced age. In addition, the risk of an infant
having a cleft lip decreases the younger both of the parents are.
On the contrary, the risk of having a baby with a cleft palate is
only connected with paternal advanced age (36,37). The
probability of having a child with cataract, esophageal atresia,
diaphragmatic hernia, encephalocele or coarctation of the aorta
increases with advanced paternal age. Babies born with omphalocele,
spina bifida, orafacial clefts and septal heart defects were more
common among older men with younger partners (38).
5. Mental disorders and children's
neurocognitive abilities
A significant amount of attention has been devoted
to the influence of the father's age on the child's mental
development, the occurrence of mental disorders, cognitive and
developmental abilities, intelligence, and even learning outcomes.
Many studies linked advanced age of the father with behavioral and
psychiatric conditions, whereas only a few studies are associated
with developmental psychology (11,39). The
connection is particularly strong in autistic diseases and
schizophrenia, while in the other cases a correlation is not well
established.
Autism belongs to neurodevelopmental disorders.
Social deficiencies, language difficulties, stereotypical behavior
and early beginning of symptoms are the typical signs of the
disease. Advancing paternal age is a significant risk factor for
autism in offspring, which has been shown in many studies in recent
years. Reichenberg and co-investigators found that in a
cohort of nearly 400,000 individuals of paternal age above 40 years
significantly increases the risk of having a child with autism
(39). Additionally, the father's age
can also affect subsequent generations. The levels of risk were
similar for maternal and paternal grandfathers. The grand-paternal
age risk estimates were lower than the risk estimates for paternal
age, which is true to the hypothesis that the strength of a
genetically mediated effect should reflect the genetic relatedness.
The results of one of the most recent meta-analysis report a 2-fold
higher probability of autism in the offspring of a father aged 50
than a father at the age of 30(40).
Moreover, the risk of autism increased with advancing
grand-paternal age, which may be independent of the age of the
mother and father. It is also observed that men above 50 years who
have daughters are more likely to have grandchildren with autism
(41). Associations between advanced
paternal age and other mental disorders (schizophrenia, bipolar
disorder, and general neurocognitive development) have also been
observed (42-45).
Furthermore, the relationship between a higher incidence of the
disease and the childbirth sequence was also demonstrated. The risk
of developing autism was the most significant for firstborn
children (46). Paternal age effect
was also connected with a higher risk of autism spectrum disorder
with intellectual disability (47).
The connection between paternal age and occurrence
of autism has been well established by many studies. However, the
exact mechanism has yet to be identified. Sebat et al
(48) reported a severe connection of
de novo copy number variations (CNVs) with autism in cases
of sporadic autism. CNVs are the changes in the number of
particular places of DNA that allows cells to contain deletions or
duplications of given regions. Authors of that study showed that
de novo mutations potentially result in both gain or loss of
DNA region copy number, which can partially explain autism spectrum
disorders (48,49).
Different mutations associated with advanced age and
epigenetic alterations are also considered to be the main reason
for this phenomenon; however, the specific mechanisms remain to be
determined. The basis for these changes may be the de novo
mutations arising in male germ cells (50). The quality of semen decreases with age
and the number of changes in the genetic materials increases. It is
observed that levels of repair enzymes are lower and DNA
fragmentations increase (51,52). De novo mutations in autism have
mainly paternal descent, as indicated in recent studies (53-56).
The advanced age of the father also contributes to
schizophrenia and the spectrum of schizophrenia disorders.
Researchers from the United States conducted a study involving
12,000 patients in which they showed an increased risk of having a
child with autism with every 10-year increase in the father's age
(57). Research carried out on a
group of 750,000 individuals in Sweden has published similar
results (44). Moreover, the
connection between paternal age and schizophrenia was evident only
in cases with no family history, which may suggest that collection
of de novo mutations in the sperm may increase the risk
(58).
The influence of paternal age on schizophrenia and
autism has been investigated for decades. It was debated whether it
is also important in the development of psychotic-like symptoms in
adult offspring that appear more frequently in the society. Data
collected by researchers show the connection between advanced
paternal age and the development of psychotic-like symptoms.
Children with fathers above 35 years of age had 2.1-fold higher
odds of psychotic-like symptoms. Interestingly, maternal age had no
impact (45).
Another disease related to the father's age is
bipolar disorder. It is a disease characterized by alternating
periods of depression and manic mood. Recent findings have shown
that, children of men above 55 years of age are almost 1.5-fold
more often diagnosed with bipolar disorder than offspring of
fathers less than 25 years of age. Interestingly, the mother's age
had no impact on the development of this disease (59).
Paternal age effect on other mental disorders was
also studied. Interestingly, in the case of ADHD (attention-deficit
hyperactivity disorder) an inverse relationship was observed. As
the age of father decreases, the risk of having a child with ADHD
increases. This connection was observed in a comprehensive,
population-based record linkage study; however, scientists did not
manage to identify a specific reason (60).
A few studies also examined the influence of the
father's age on behavior and cognitive ability. Using
neurocognitive measures such as the Wechsler Intelligence Scale for
Children researchers have shown that children of older fathers had
lower results (61). Another study
showed the association of an advanced father's age with the
occurrence of negative behaviors in children such as aggression,
vandalism, and disruptive behavior. It was elucidated that for
every 5-year increase in the father's age, the likelihood of higher
defective demeanor increased by 12% (62,63). It
was also indicated that offspring of fathers aged 45 years and
older gained lower grades in science (64).
Association between the father's age and
intelligence of the descendants was also studied. Due to many
possible confounders such as socio-economic status, the level of
parents' education, and parents' intelligence indicators, it may be
complicated to analyze the link between paternal age effect and
intelligence of offspring. However, there are many premises that
should not be omitted. Investigators revealed a non-linear
relationship with male children's verbal intelligence quotient (IQ)
scores, despite considering possible disruptive factors (65). Moreover, it has been determined that
children of older fathers have increased risk of obtaining a lower
level of education and worse scores (66). Notably, research on a group of
adolescents born to fathers younger than 20 years and older than 50
years of age, revealed that these groups achieved the lowest
results in measures of non-verbal IQ (66).
There are also some signs that paternal age at birth
appears to have an impact on psychosocial development in offspring.
Children of younger and older men seem to have lower social skills
and an increased risk of developing major depressive disorder
(65,66).
6. Conclusions
Paternal age appears to have a significant impact on
a child's health and development. The quality of semen decreases
with aging, resulting in DNA copy mutations. Similarly, a decrease
in the volume of semen, can lead to problems with conception. It
can therefore be said that the male biological clock is ticking,
too. Available studies clearly link advanced paternal age at the
age of conception with increased frequency of mental health
disorders, such as bipolar disease, schizophrenia, autism or even a
lower IQ level of the offspring. Advanced paternal age also has
serious implications on genetic disorders, perinatal complications,
congenital defects, or even carcinogenesis. Currently, much more
data are available on the effects of maternal age on fetus. To
summarize, more research is needed regarding the impact of paternal
age on the developing fetus, health of mothers during pregnancy,
growing child and complications in their adult age. However, at
this point, we can certainly conclude, that paternal age is as
significant as maternal age, when it comes to reproductive
matters.
Acknowledgements
The authors would like to thank Barbara Zielinski
Roman and Stacie Rhoades (Department of Epidemiology, Nationwide
Children's Hospital, Columbus, OH, USA), and Filip Kacki MD, for
their support with linguistic correctness.
Funding
This study was supported by a grant from the Medical
University of Lublin (Lublin, Poland) to AS (Dz. St. 326/19).
Availability of data and materials
All the data and materials used and analyzed in this
review are available from the corresponding Author on reasonable
request.
Authors' contributions
DJ conceived and designed the review. DJ, AO, MH, BK
found literature and analyzed it. DJ, AO, MH, BK drafted the
manuscript. AS critically revised and corrected the manuscript. All
authors have read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare they have no competing
interests.
References
|
1
|
Łepecka-Klusek C, Pilewska-Kozak AB and
Jakiel G: Niepłodność w świetle definicji choroby podanej przez
WHO. Med Og Nauk o Zdro. 18:163–166. 2012.(In Polish).
|
|
2
|
World Health Organization: Sexual and
reproductive health. https://www.who.int/reproductivehealth/topics/infertility/definitions/en/.
Accessed June 4, 2019.
|
|
3
|
Anwar S and Anwar A: Infertility: A review
on causes, treatment and management. Womens Health Gynecol. 2:1–5.
2016.
|
|
4
|
Demographic Yearbook of Poland 2017.
Central Statistical Office, pp266-272, 2017. https://stat.gov.pl/en/topics/statistical-yearbooks/statistical-yearbooks/demographic-yearbook-of-poland-2017,3,11.html.
Accessed October 13, 2017.
|
|
5
|
Szkodziak P, Wozniak P, Czuczwar P,
Woźniakowska E, Milart P, Mroczkowski A and Paszkowski T:
Infertility in the light of new scientific reports-focus on male
factor. Ann Agric Environ Med. 23:227–230. 2016.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Agarwal A, Mulgund A, Hamada A and Chyatte
MR: A unique view on male infertility around the globe. Reprod Biol
Endocrinol. 13(37)2015.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Rogel R, Luján S, Broseta E, Age P and
Concerns F: Paternal age and fertility concerns. Encyclopedia Repr.
4:301–306. 2018. View Article : Google Scholar
|
|
8
|
Moskovtsev SI, Willis J and Mullen JB:
Age-related decline in sperm deoxyribonucleic acid integrity in
patients evaluated for male infertility. Fertil Steril. 85:496–499.
2006.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Ramasamy R, Chiba K, Butler P and Lamb DJ:
Male biological clock: A critical analysis of advanced paternal
age. Fertil Steril. 103:1402–1406. 2015.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Feinberg JI, Bakulski KM, Jaffe AE,
Tryggvadottir R, Brown SC, Goldman LR, Croen LA, Hertz-Picciotto I,
Newschaffer CJ, Fallin MD and Feinberg AP: Paternal sperm DNA
methylation associated with early signs of autism risk in an
autism-enriched cohort. Int J Epidemiol. 44:1199–1210.
2015.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Malaspina D, Gilman C and Kranz TM:
Paternal age and mental health of offspring. Fertil Steril.
103:1392–1396. 2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Goriely A and Wilkie AO: Paternal age
effect mutations and selfish spermatogonial selection: Causes and
consequences for human disease. Am J Hum Genet. 10:175–200.
2012.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Conti SL and Eisenberg ML: Paternal aging
and increased risk of congenital disease, psychiatric disorders,
and cancer. Asian J Androl. 18:420–424. 2016.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Speicher M, Antonarakis SE and Motulsky AG
(eds): Vogel and Motulsky's Human Genetics: Problems and
Approaches. 4th edition. Springer, 2010.
|
|
15
|
Kong A, Frigge ML, Masson G, Besenbacher
S, Sulem P, Magnusson G, Gudjonsson SA, Sigurdsson A, Jonasdottir
A, Jonasdottir A, et al: Rate of de novo mutations, father's age,
and disease risk. Nature. 488:471–475. 2012.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Kurahashi H, Tsutsumi M, Nishiyama S, Kogo
H, Inagaki H and Ohye T: Molecular basis of maternal age-related
increase in oocyte aneuploidy. Congenit Anom (Kyoto). 52:8–15.
2012.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Zhu JL, Madsen KM, Vestergaard M, Olesen
AV, Basso O and Olsen J: Paternal age and congenital malformations.
Hum Reprod. 20:3173–3177. 2005.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Meng Y and Groth SW: Fathers count: The
impact of paternal risk factors on birth outcomes. Matern Child
Health J. 22:401–408. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Nybo Andersen AM, Hansen KD, Andersen PK
and Davey Smith G: Advanced paternal age and risk of fetal death: A
cohort study. Am J Epidemiol. 160:1214–1222. 2004.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Urhoj SK, Andersen PK, Mortensen LH, Davey
Smith G and Nybo Andersen AM: Advanced paternal age and stillbirth
rate: A nationwide register-based cohort study of 944,031
pregnancies in Denmark. Eur J Epidemiol. 32:227–234.
2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Alio AP, Salihu HM, McIntosh C, August EM,
Weldeselasse H, Sanchez E and Mbah AK: The effect of paternal age
on fetal birth outcomes. Am J Mens Health. 6:427–435.
2012.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Khandwala YS, Baker VL, Shaw GM, Stevenson
DK, Lu Y and Eisenberg ML: Association of paternal age with
perinatal outcomes between 2007 and 2016 in the United States:
Population based cohort study. BMJ. 363(k4372)2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Yang Q, Wen SW, Leader A, Chen XK, Lipson
J and Walker M: Paternal age and birth defects: How strong is the
association? Hum Reprod. 22:696–701. 2007.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Astolfi P, De Pasquale A and Zonta LA:
Paternal age and preterm birth in Italy, 1990 to 1998.
Epidemiology. 17:218–221. 2006.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Bjornard K, Riehle-Colarusso T, Gilboa SM
and Correa A: Patterns in the prevalence of congenital heart
defects, metropolitan Atlanta, 1978 to 2005. Birth Defects Res A
Clin Mol Teratol. 97:87–94. 2013.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Oyen N, Poulsen G, Boyd HA, Wohlfahrt J,
Jensen PK and Melbye M: National time trends in congenital heart
defects, Denmark, 1977-2005. Am Heart J. 157:467.e1–473.e1.
2009.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Reller MD, Strickland MJ, Riehle-Colarusso
T, Mahle WT and Correa A: Prevalence of congenital heart defects in
metropolitan Atlanta, 1998-2005. J Pediatr. 153:807–813.
2008.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Materna-Kiryluk A, Wiśniewska K,
Badura-Stronka M, Mejnartowicz J, Wieckowska B, Balcar-Boroń A,
Czerwionka-Szaflarska M, Gajewska E, Godula-Stuglik U, Krawczyński
M, et al: Parental age as a risk factor for isolated congenital
malformations in a Polish population. Paediatr Perinat Epidemiol.
23:29–40. 2009.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Olshan AF, Schnitzer PG and Baird PA:
Paternal age and the risk of congenital heart defects. Teratology.
50:80–84. 1994.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Baltaxe E and Zarante I: Prevalence of
congenital heart disease in 44,985 newborns in Colombia. Arch
Cardiol Mex. 76:263–268. 2006.(In Spanish). PubMed/NCBI
|
|
31
|
Cedergren MI, Selbing AJ and Källén BA:
Risk factors for cardiovascular malformation-a study based on
prospectively collected data. Scand J Work Environ Health.
28:12–17. 2002.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Zhan SY, Lian ZH, Zheng DZ and Gao L:
Effect of fathers' age and birth order on occurrence of congenital
heart disease. J Epidemiol Community Health. 45:299–301.
1991.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Ewing CK, Loffredo CA and Beaty TH:
Paternal risk factors for isolated membranous ventricular septal
defects. Am J Med Genet. 71:42–46. 1997.PubMed/NCBI
|
|
34
|
Bassili A, Mokhtar SA, Dabous NI, Zaher
SR, Mokhtar MM and Zaki A: Risk factors for congenital heart
diseases in Alexandria, Egypt. Eur J Epidemiol. 16:805–814.
2000.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Su XJ, Yuan W, Huang GY, Olsen J and Li J:
Paternal age and offspring congenital heart defects: A national
cohort study. PLoS One. 10(e0121030)2015.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Bille C, Skytthe A, Vach W, Knudsen LB,
Andersen AM, Murray JC and Christensen K: Parent's age and the risk
of oral clefts. Epidemiology. 16:311–316. 2005.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Berg E, Lie RT, Sivertsen Å and Haaland
ØA: Parental age and the risk of isolated cleft lip: A
registry-based study. Ann Epidemiol. 25:942.e1–947.e1.
2015.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Green RF, Devine O, Crider KS, Olney RS,
Archer N, Olshan AF and Shapira SK: National Birth Defects
Prevention Study: Association of paternal age and risk for major
congenital anomalies from the National Birth Defects Prevention
Study, 1997 to 2004. Ann Epidemiol. 20:241–249. 2010.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Reichenberg A, Gross R, Weiser M,
Bresnahan M, Silverman J, Harlap S, Rabinowitz J, Shulman C,
Malaspina D, Lubin G, et al: Advancing paternal age and autism.
Arch Gen Psychiatry. 63:1026–1032. 2006.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Hultman CM, Sandin S, Levine SZ,
Lichtenstein P and Reichenberg A: Advancing paternal age and risk
of autism: New evidence from a population-based study and a
meta-analysis of epidemiological studies. Mol Psychiatry.
16:1203–1212. 2011.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Frans EM, Sandin S, Reichenberg A,
Långström N, Lichtenstein P, McGrath JJ and Hultman CM: Risk across
generations: A population-based study of advancing grandpaternal
and paternal age. JAMA Psychiatry. 70:516–521. 2013.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Byrne M, Agerbo E, Ewald H, Eaton WW and
Mortensen PB: Parental age and risk of schizophrenia: A
case-control study. Arch Gen Psychiatry. 60:673–678.
2003.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Sipos A, Rasmussen F, Harrison G, Tynelius
P, Lewis G, Leon DA and Gunnell D: Paternal age and schizophrenia:
A population based cohort study. BMJ. 329(1070)2004.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Frans EM, Sandin S, Reichenberg A,
Lichtenstein P, Långström N and Hultman CM: Advancing paternal age
and bipolar disorder. Arch Gen Psychiatry. 65:1034–1040.
2008.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Glasson EJ, Bower C, Petterson B, de Klerk
N, Chaney G and Hallmayer JF: Perinatal factors and the development
of autism: A population study. Arch Gen Psychiatry. 61:618–627.
2004.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Idring S, Magnusson C, Lundberg M, Ek M,
Rai D, Svensson AC, Dalman C, Karlsson H and Lee BK: Parental age
and the risk of autism spectrum disorders: Findings from a Swedish
population-based cohort. Int J Epidemiol. 43:107–115.
2014.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Puleo CM, Schmeidler J, Reichenberg A,
Kolevzon A, Soorya LV, Buxbaum JD and Silverman JM: Advancing
paternal age and simplex autism. Autism. 16:367–380.
2012.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Sebat J, Lakshmi B, Malhotra D, Troge J,
Lese-Martin C, Walsh T, Yamrom B, Yoon S, Krasnitz A, Kendall J, et
al: Strong association of de novo copy number mutations with
autism. Science. 316:445–449. 2007.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Crow JF: The origins, patterns and
implications of human spontaneous mutation. Nat Rev Genet. 1:40–47.
2000.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Malaspina D: Paternal factors and
schizophrenia risk: De novo mutations and imprinting. Schizophr
Bull. 27:379–393. 2001.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Tarín JJ, Brines J and Cano A: Long-term
effects of delayed parenthood. Hum Reprod. 13:2371–2376.
1998.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Iossifov I, Ronemus M, Levy D, Wang Z,
Hakker I, Rosenbaum J, Yamrom B, Lee YH, Narzisi G, Leotta A, et
al: De novo gene disruptions in children on the autistic spectrum.
Neuron. 74:285–299. 2012.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Neale BM, Kou Y, Liu L, Ma'ayan A, Samocha
KE, Sabo A, Lin CF, Stevens C, Wang LS, Makarov V, et al: Patterns
and rates of exonic de novo mutations in autism spectrum disorders.
Nature. 485:242–245. 2012.PubMed/NCBI View Article : Google Scholar
|
|
54
|
O'Roak BJ, Vives L, Girirajan S, Karakoc
E, Krumm N, Coe BP, Levy R, Ko A, Lee C, Smith JD, et al: Sporadic
autism exomes reveal a highly interconnected protein network of de
novo mutations. Nature. 485:246–250. 2012.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Sanders SJ, Murtha MT, Gupta AR, Murdoch
JD, Raubeson MJ, Willsey AJ, Ercan-Sencicek AG, DiLullo NM,
Parikshak NN, Stein JL, et al: De novo mutations revealed by
whole-exome sequencing are strongly associated with autism. Nature.
485:237–241. 2012.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Brown AS, Schaefer CA, Wyatt RJ, Begg MD,
Goetz R, Bresnahan MA, Harkavy-Friedman J, Gorman JM, Malaspina D
and Susser ES: Paternal age and risk of schizophrenia in adult
offspring. Am J Psychiatry. 159:1528–1533. 2002.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Malaspina D, Harlap S, Fennig S, Heiman D,
Nahon D, Feldman D and Susser ES: Advancing paternal age and the
risk of schizophrenia. Arch Gen Psychiatry. 58:361–367.
2001.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Foutz J and Mezuk B: Advanced paternal age
and risk of psychotic-like symptoms in adult offspring. Schizophr
Res. 165:123–127. 2015.PubMed/NCBI View Article : Google Scholar
|
|
59
|
McGrath JJ, Petersen L, Agerbo E, Mors O,
Mortensen PB and Pedersen CB: A comprehensive assessment of
parental age and psychiatric disorders. JAMA Psychiatry.
71:301–309. 2014.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Saha S, Barnett AG, Foldi C, Burne TH,
Eyles DW, Buka SL and McGrath JJ: Advanced paternal age is
associated with impaired neurocognitive outcomes during infancy and
childhood. PLoS Med. 6(e40)2009.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Wechsler D: Manual for the Wechsler
Intelligence Scale for Children. New York: The Psychological
Corporation, 1949.
|
|
62
|
Saha S, Barnett AG, Buka SL and McGrath
JJ: Maternal age and paternal age are associated with distinct
childhood behavioural outcomes in a general population birth
cohort. Schizophr Res. 115:130–135. 2009.PubMed/NCBI View Article : Google Scholar
|
|
63
|
D'Onofrio BM, Rickert ME, Frans E,
Kuja-Halkola R, Almqvist C, Sjölander A, Larsson H and Lichtenstein
P: Paternal age at childbearing and offspring psychiatric and
academic morbidity. JAMA Psychiatry. 71:432–438. 2014.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Janecka M, Haworth CMA, Ronald A, Krapohl
E, Happé F, Mill J, Schalkwyk LC, Fernandes C, Reichenberg A and
Rijsdijk F: Paternal age alters social development in offspring. J
Am Acad Child Adolesc Psychiatry. 56:383–390. 2017.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Malaspina D, Reichenberg A, Weiser M,
Fennig S, Davidson M, Harlap S, Wolitzky R, Rabinowitz J, Susser E
and Knobler HY: Paternal age and intelligence: Implications for
age-related genomic changes in male germ cells. Psychiatr Genet.
15:117–125. 2005.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Buizer-Voskamp JE, Laan W, Staal WG,
Hennekam EA, Aukes MF, Termorshuizen F, Kahn RS, Boks MP and Ophoff
RA: Paternal age and psychiatric disorders: Findings from a Dutch
population registry. Schizophr Res. 129:128–132. 2011.PubMed/NCBI View Article : Google Scholar
|